1. Introduction
Chronic kidney disease (CKD) is a significant global health challenge, contributing substantially to morbidity and mortality in both developed and developing countries, particularly when it progresses to end-stage renal disease (ESRD) [
1,
2]. The global prevalence of CKD is estimated at approximately 13.4%, and 4.9 to 7.1 million individuals with ESRD require renal replacement therapy [
3,
4]. Thyroid hormones play a crucial role in the physical and cognitive development of children and adults [
5]. These hormones are vital for supporting the optimal functioning of all major organs, including the kidneys, thereby helping to preserve overall homeostasis and essential bodily processes; consequently, any complications arising from thyroid hormone imbalances can significantly disrupt normal physiological functions [
6,
7].
Thyroid hormones such as triiodothyronine (T3) and thyroxine (T4) are produced by the thyroid gland [
8]. The thyroid gland primarily produces T4, which accounts for approximately 80% of thyroid hormone production, and T3, which accounts for about 20%. While all T4 in the bloodstream originates in the thyroid gland, only 20% of circulating T3 is directly produced by the thyroid; the remaining 80% is converted from T4 outside the thyroid (extrathyroidally) [
9,
10]. This process underscores the intricate dynamics of thyroid hormone synthesis and conversion, as discussed in studies on thyroid function assessment and thyroid disorders in chronic kidney disease [
11]. Thyroid-stimulating hormone (TSH), secreted by the pituitary gland, is crucial for normal thyroid function [
12]. TSH production is regulated by a hypothalamic hormone, thyrotropin-releasing hormone (TRH), which is influenced by environmental, developmental, and daily rhythms [
13,
14]. The regulation of thyroid hormone secretion is achieved through feedback inhibition by the hypothalamic-pituitary-thyroid axis (HPT). Initially, the hypothalamus secretes TRH, which then travels to the anterior pituitary gland and stimulates the synthesis of TSH [
15].
T3 and T4 play crucial roles in regulating metabolism, energy production, and overall growth and development [
16,
17]. Elevated TSH levels often signal conditions such as hypothyroidism and Hashimoto’s thyroiditis [
12]. The kidneys play a crucial role in thyroid hormone metabolism, breakdown, and elimination; consequently, renal impairment significantly alters thyroid physiology [
18]. Nevertheless, thyroid hormones are essential for regulating the glomerular filtration rate (GFR) by controlling renal blood flow [
6].
Previous studies indicate that thyroid comorbidities are more prevalent in individuals with CKD compared to the general population [
16,
19]. Endocrine disorders were found to be highly prevalent among patients with CKD, especially those undergoing dialysis. CKD affects multiple endocrine and metabolic processes [
3,
20]. Conversely, a previous study reported that hemodialysis induced a significant rise in thyroid hormone levels among ESRD patients [
21].
Therefore, the objective of this cohort study was to investigate the association and compare the levels of T3, T4, and TSH in patients undergoing renal replacement therapy (hemodialysis), patients not receiving dialysis, and patients suspected of having thyroid complications only, with no other confirmed kidney or other complications.
2. Materials and Methods
2.1. Ethical Approval, Study Settings, and Participants
The study was conducted from December 2024 to August 2025 in the hospital settings of Gonoshasthaya Nagar Hospital, Dhaka, Bangladesh. Ethical approval was obtained from the Center for Multidisciplinary Research, Gono Bishwabidyalay, Dhaka, Bangladesh (Registration no: CMR/ER/026).
2.2. Inclusion and Exclusion Criteria
Patients suspected of having thyroid complications and tested for T3, T4, and TSH were included in this study. The patients in this study cohort were categorized into the following three groups-
Normal patients (NP): individuals suspected of having thyroid complications and having no kidney complications or other confirmed diagnosed diseases.
Non-dialysis kidney patients (NDKP): individuals with kidney or renal complications confirmed by a creatinine level > the reference range (0.6-1.3 mg/dL) who are suspected of having thyroid complications.
Dialysis patients (DP): ESRD patients receiving dialysis for ≥1 year were suspected of having thyroid complications.
The patients who did not have or were not suspected of having thyroid complications and were not tested for all three hormones (i.e., T3, T4, and TSH) were excluded from this study.
2.3. Sample Collection
Blood samples from DP patients were collected before each session through the fistula. For NP and NDKP, blood was drawn by venipuncture. The serum was separated from the hematocrit by centrifugation. If the test was not performed immediately after separation, the serum was stored at -20 °C.
2.4. Measurement of T3, T4 and TSH Levels
All hormones, including triiodothyronine (T3), thyroxine (T4), and thyrotropin (TSH), were quantified using a colorimetric enzyme-linked immunosorbent assay (ELISA). Total triiodothyronine (T3) levels were measured with the Biorex Diagnostics TT3 kit (BXE0701A, United Kingdom), with a reference range of 0.8 to 2.0 ng/mL. Total thyroxine (T4) concentrations were assessed with the Biorex Diagnostics TT4 kit (BXE0711A, United Kingdom), with reference values ranging from 4.5 to 12.0 µg/dL. Thyrotropin (TSH) levels in human serum were quantified with a colorimetric ELISA using the Biorex Diagnostics TSH kit (BXE0681A, United Kingdom), with a reference range of 0.4 to 4.0 µIU/mL. The optical density (OD) of all analyzed hormones (T3, T4, and TSH) was measured at 450 nm, a wavelength commonly used in ELISA procedures to accurately quantify thyroid hormone concentrations in biological samples. Measurements were conducted with a Multiskan FC microplate photometer (Multiskan FC, USA).
2.5. Statistical Analysis
Statistical analyses were conducted using SPSS (version 16.0), and data assembly, general calculations, percentage determinations, and graphical presentations were performed using Microsoft Excel (2010). Mean, standard deviation, Pearson correlation, and p-value were assessed for the statistical analysis.
3. Results
3.1. Participant Demographics and Hormonal Level Determination
The study included 161 patients, divided into three groups: 106 NP, 32 NDKP, and 23 DP. The mean age of the participants was recorded as 42.49 ± 17.89 years. Detailed demographic information is provided in
Table 1.
The distribution of patients exhibiting normal (within reference values), elevated (> reference values), and diminished (< reference values) hormone levels is presented in
Table 2.
3.2. Comparison of Thyroid Hormone Levels
A comparative analysis of TSH, T3, and T4 across the three groups indicated that the DP group exhibited the highest levels of all three hormones. Notably, TSH levels in the NP group were significantly elevated compared to those in the NDKP group. Conversely, T3 levels were slightly higher in the NDKP group than in the NP group. Analogous trends were observed in T4 levels, with NP and NDKP showing results similar to those of T3 (
Figure 1).
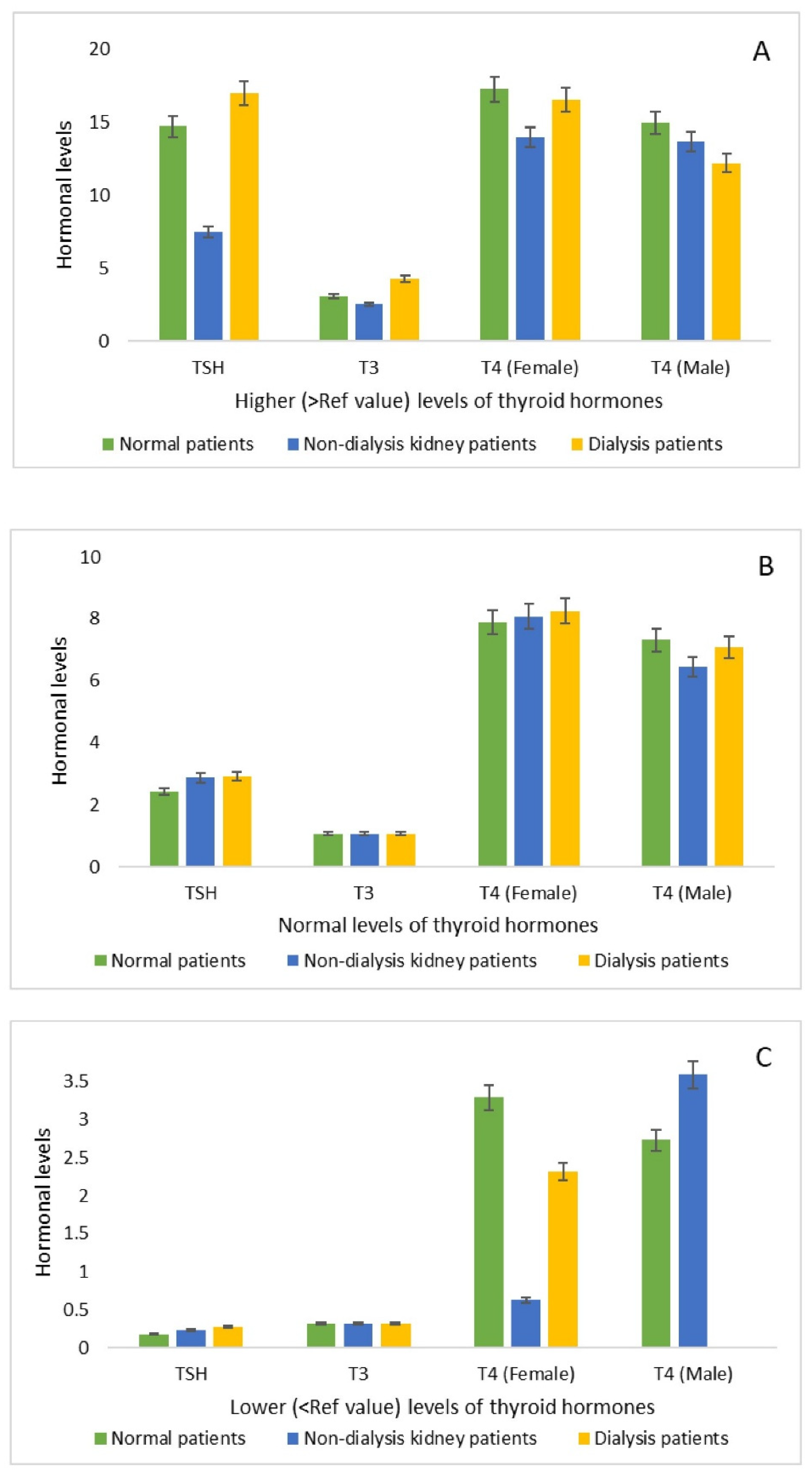
Further examination of hormonal levels across lower, normal, and higher categories revealed that, for elevated hormonal levels, TSH, T3, and T4 were substantially higher in the DP group than in NDKP, except for T4 levels among male patients. Similar patterns were observed for normal hormone levels in TSH and T4, although T3 levels remained consistent across all groups. When focusing solely on lower hormone levels, T3 levels were equivalent between DP and NDKP, whereas TSH and T4 (in females) were elevated in DP compared to NDKP. Moreover, no instances of low T4 levels were identified among males (
Figure 2).
3.3. Comparison of Hormone Levels Based on Sex and Age
Hormonal levels were analyzed with respect to sex and age. In male participants, both TSH and T4 levels were highest in the DP group compared with NP and NDKP. Among female participants, all three hormones showed elevated levels in the DP group compared with the other two groups (
Table 3).
In terms of age stratification, TSH and T3 levels were higher in the DP group than in the other groups. Conversely, T4 levels in the NDKP group exceed those in both the NP and DP groups among patients aged <45 years. In the cohort of patients over 45, T4 levels were generally higher in the NP group than in the other two groups, except in the DP group.
3.4. Association Between Hormone Levels
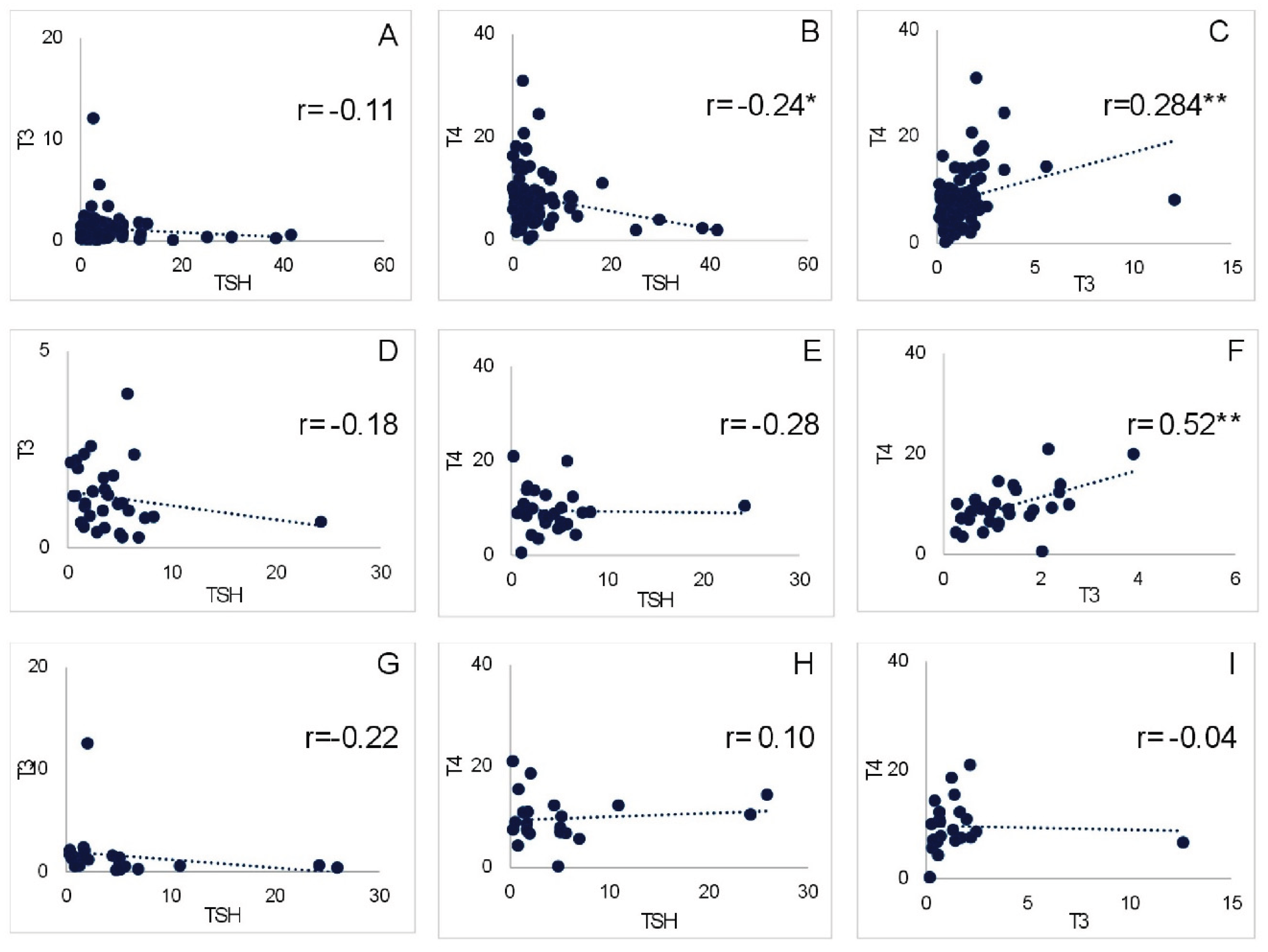
The assessment of correlations between pairs of hormones (TSH, T3, and T4) across all three groups revealed a negative correlation between TSH and T3. A positive correlation between TSH and T4 was observed in the DP group, whereas a negative correlation was observed in the NP and NDKP groups. Furthermore, negative correlations between T3 and T4 were observed exclusively in the DP group, whereas positive correlations were significantly established in the other two groups. Notably, the negative correlation between TSH and T4 was statistically significant in the NP group (
Figure 3).
4. Discussion
Thyroid autoimmunity and subclinical primary hypothyroidism are prevalent in CKD patients who do not require long-term dialysis [
22]. In a study by Lo et al. (2005), 23.1% of CKD patients with a GFR below 30 mL/min/1.73 m
2 were found to have hypothyroidism. Furthermore, it has been reported that 18-20% of CKD patients not needing renal replacement therapy experience either subclinical or clinically apparent hypothyroidism [
23].
The kidney plays a crucial role in iodine clearance, predominantly via glomerular filtration [
24,
25]. In patients with CKD, reduced GFR decreases iodine clearance, leading to higher plasma iodide levels, which eventually result in increased iodide uptake by thyroid tissue [
26,
27]. Excess iodide may inhibit thyroid hormone production by disrupting the pituitary-thyroid axis, a phenomenon known as the Wolff-Chaikoff effect. These physiological alterations provide a plausible explanation for the higher incidence of hypothyroidism observed in CKD patients [
28]. Both subclinical and overt hypothyroidism are common in CKD patients, indicating a prevalence of diminished thyroid function, whether symptomatic or asymptomatic [
29]. Our present study indicates that patients with kidney disease (NDKP) had lower TSH levels than patients without kidney disease (NP). Interestingly, patients receiving regular hemodialysis (DP) had higher hormonal levels for all three evaluated thyroid hormones (i.e., TSH, T3, and T4) (
Figure 1). While analyzing the higher, normal, and lower hormonal levels other than normal T4 (males), the DP group was found to have higher hormonal levels as compared to NDKP, and no cases were found in the T4 (males) within the category of lower hormonal level (<reference value) (
Figure 2). Previously, a study by Sanai et al. reported rising serum free T3 and T4 levels within the control or normal reference range after hemodialysis, especially among ESRD patients with chronic glomerulonephritis [
30]. Findings from our study also align with those of a study by Shamsadini et al., which reported that hemodialysis restored serum levels of thyroid hormone to normal ranges among patients with chronic renal failure [
31] . During participant selection, it was found that the majority of the patients were female, which is supported by a study reporting that thyroid dysfunction occurs predominantly in females (
Table 1) [
32]. Therefore, the effect of dialysis was more pronounced in female patients than in male patients, with mean hormone levels (TSH, T3, and T4) higher in the DP group than in the other two groups (
Table 3). Again, most patients in the cohort are close to 40 years of age, and in the NDKP and DP groups, the mean age was >40 years, which aligns with previous studies reporting that hyperthyroidism is quite common in older people as compared to the younger [
33,
34]. This phenomenon was observed by comparing the levels between age groups, as we observed that in the age group <45 years, dialysis did have an impact on elevating the hormone levels in most of the cases, whereas in the age group >45 years, dialysis did not have such an effect on the hormonal levels (
Table 3).
In the association analysis, we observed a similar pattern between the NP and NDKP groups: both showed negative correlations between T3 and TSH and between T4 and TSH, whereas T3 and T4 showed a significant positive association. However, in the case of DP, we observed that T4 vs. TSH showed a slight positive correlation, and T3 vs. T4 showed an inverse correlation, both of which were not significant (
Figure 3). A moderate inverse relationship between TSH and T4 was determined in a previous study among patients with primary thyroid failure [
35,
36]. Besides, T4 primarily serves as a prohormone reservoir that is converted to the more biologically active T3 in tissues, making T3 pivotal in mediating metabolic processes, while T4 ensures a steady supply necessary for physiological homeostasis, which indicates that, due to regular dialysis, T4 levels and activity were slightly higher in the DP group than in the T3 group [
37,
38]. Overall, our findings suggest that regular dialysis moderately improves thyroid hormone levels in kidney patients, especially compared with those not on dialysis (
Table 4). Further research is required to elucidate the underlying molecular mechanisms governing these observed changes.
5. Limitations of This Study
Including additional participants in this study, particularly in the NDKP and DP groups, was not feasible. Furthermore, the absence of a longitudinal analysis involving a larger cohort represents a notable limitation of the research. Additionally, the study was conducted at a single center, which may constrain the generalizability of the findings; this limitation is duly acknowledged.
6. Future Perspectives and Recommendations
• Research Gap: Limited research exists on the interplay among thyroid function, hormone levels, and dialysis outcomes in CKD patients, especially in the Bangladeshi context.
• Future Research Direction: Multifactorial longitudinal studies with multicenter designs, larger and more diverse cohorts are needed to robustly evaluate the interplay among thyroid function, hormone levels, and dialysis outcomes in CKD patients.
• Study Design Justification: Such studies are crucial to enhance generalizability, mitigate selection bias, and enable comprehensive statistical analysis.
7. Conclusions
Dialysis has been shown to moderately enhance thyroid hormone levels and activity in patients with renal complications compared to those with kidney disease who are not undergoing dialysis. These findings indicate that routine dialysis may confer potential benefits for individuals with chronic kidney disease (CKD) or end-stage renal disease (ESRD). Further investigation involving larger cohorts of dialysis and non-dialysis patients, stratified by hypo- and hyperthyroid conditions, is warranted to elucidate these observations more comprehensively.
Author Contributions
Conceptualization, S.S.K.; methodology, S.S.K. and H.I.; software, S.S.K., H.I; validation, X.X., Y.Y. and Z.Z.; formal analysis, S.S.K., H.I., and E.S.; investigation, S.S.K. and H.I.; resources, A.K., S.S.K; data curation, A.K., and H.I.; writing—original draft preparation, H.I., E.S., A.H.P, D.Z.I, R.B; writing—review and editing, S.S.K, R.B, M.N.U, M.A.H, M.S.A. and A.N.M.M-O-R; visualization, S.S.K, H.I. and E.S.; supervision, S.S.K, M.A.H and A.N.M.M-O-R; project administration, S.S.K and A.N.M.M-O-R. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the Center for Multidisciplinary Research of Gono Bishwabidyalay (CMR/EC/024) on 1 April 2025.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. The authors reviewed and approved the final version and agreed to be accountable for all aspects of the work.
Data Availability Statement
All data underlying the findings in our study are freely available in the manuscript. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Neuen, Brendon L; Bello, Aminu K; Levin, Adeera; Lunney, Meaghan; Osman, Mohamed A; Ye, Feng; Ashuntantang, Gloria E; Bellorin-Font, Ezequiel; Benghanem Gharbi, Mohammed; Davison, Sara. National Health Policies and Strategies for Addressing Chronic Kidney Disease: Data from the International Society of Nephrology Global Kidney Health Atlas. PLOS Global Public Health 2023, 3(no. 2), e0001467. [Google Scholar] [CrossRef]
- Khandker, Shahad Saif; Jahan, Safrin; Khan, Adiba Ayesha; Abrittee, Begum Faijunnesa; Nanjiba, Rifah; Islam, Dewan Zubaer; Suez, Ehsan; Rahman, Md Habibur; Farahim, Farha; Ali, Talat. Global Epidemiology of Hiv among Dialysis Patients: A Systematic Review and Meta-Analysis. International Urology and Nephrology 2025, 1–18. [Google Scholar]
- Cotoi, Laura; Borcan, Florin; Sporea, Ioan; Amzar, Daniela; Schiller, Oana; Schiller, Adalbert; Dehelean, Cristina A; Nicusor Pop, Gheorghe; Borlea, Andreea; Stoian, Dana. Thyroid Pathology in End-Stage Renal Disease Patients on Hemodialysis. Diagnostics 2020, 10(no. 4), 245. [Google Scholar] [CrossRef] [PubMed]
- Khandker, Shahad Saif; Jannat, Nurani; Sarkar, Deepannita; Pranto, Alif Hasan; Hoque, Ismoth Ara; Zaman, Jemema; Uddin, Md Nizam; Suez, Ehsan. Association between Glomerular Filtration Rate and Β-Thalassemia Major: A Systematic Review and Meta-Analysis. Thalassemia Reports 2023, 13(no. 3), 195–205. [Google Scholar] [CrossRef]
- Zoccali, Carmine; Kramer, Anneke; Jager, Kitty J. Chronic Kidney Disease and End-Stage Renal Disease—a Review Produced to Contribute to the Report ‘the Status of Health in the European Union: Towards a Healthier Europe’. NDT plus 2010, 3(no. 3), 213–24. [Google Scholar] [CrossRef]
- Fan, Jingxian; Yan, Peng; Wang, Yingdeng; Shen, Bo; Ding, Feng; Liu, Yingli. Prevalence and Clinical Significance of Low T3 Syndrome in Non-Dialysis Patients with Chronic Kidney Disease. Medical science monitor: international medical journal of experimental and clinical research 2016, 22, 1171. [Google Scholar] [PubMed]
- Shahid, Muhammad A; Ashraf, Muhammad A; Sharma, Sandeep. Physiology, Thyroid Hormone; 2018. [Google Scholar]
- Schroeder, Amy C; Privalsky, Martin L. Thyroid Hormones, T3 and T4, in the Brain. Frontiers in endocrinology 2014, 5, 40. [Google Scholar]
- Gomes-Lima, Cristiane; Wartofsky, Leonard; Burman, Kenneth. Can Reverse T3 Assay Be Employed to Guide T4 Vs. T4/T3 Therapy in Hypothyroidism? Frontiers in endocrinology 2019, 10, 856. [Google Scholar] [CrossRef]
- Wiersinga, WM. L-T4 and L-T3 Combined Treatment Vs L-T4 Alone. Paper presented at the Annales d’endocrinologie, 2007. [Google Scholar]
- Sapin, R; Schlienger, JL. Thyroxine (T4) and Tri-Iodothyronine (T3) Determinations: Techniques and Value in the Assessment of Thyroid Function. Paper presented at the Annales de biologie clinique, 2003. [Google Scholar]
- Biondi, Bernadette; Cappola, Anne R; Cooper, David S. Subclinical Hypothyroidism: A Review. Jama 2019, 322(no. 2), 153–60. [Google Scholar]
- Nillni, Eduardo A. Regulation of the Hypothalamic Thyrotropin Releasing Hormone (Trh) Neuron by Neuronal and Peripheral Inputs. Frontiers in neuroendocrinology 2010, 31(no. 2), 134–56. [Google Scholar]
- Duntas, LH. New Insights into the Hypothalamic-Pituitary-Thyroid Axis. Acta Endocrinologica (Bucharest) 2016, 12(no. 2), 125. [Google Scholar] [CrossRef]
- Salih, Sama S; Yenzeel, Jabbar H. Evaluation of Thyroid Hormones and Some Biochemical Variables in Patients with Chronic Kidney Disease. Iraqi Journal of Science 2020, 985–92. [Google Scholar] [CrossRef]
- Mohamedali, Mohamed; Reddy Maddika, Srikanth; Vyas, Anix; Iyer, Viswanathan; Cheriyath, Pramil. Thyroid Disorders and Chronic Kidney Disease. International journal of nephrology 2014, no. 1, 520281. [Google Scholar]
- Rhee, Connie M; You, Amy S; Nguyen, Danh V; Brunelli, Steven M; Budoff, Matthew J; Streja, Elani; Nakata, Tracy; Kovesdy, Csaba P; Brent, Gregory A; Kalantar-Zadeh, Kamyar. Thyroid Status and Mortality in a Prospective Hemodialysis Cohort. The Journal of Clinical Endocrinology & Metabolism 2017, 102(no. 5), 1568–77. [Google Scholar] [CrossRef]
- Agahi, Sadaf; Amouzegar, Atieh; Honarvar, Mohammadjavad; Azizi, Fereidoun; Mehran, Ladan. Interrelationship between Thyroid Hormones and Reduced Renal Function, a Review Article. Thyroid Research 2024, 17(no. 1), 14. [Google Scholar] [CrossRef]
- Raj, Rishav; Kumar, Vijay; Bhushan, Divendu; Biswas, Ratnadeep; Ojha, Vishnu S; KUMAR, VIJAY. The Prevalence of Thyroid Abnormalities in Patients with Chronic Kidney Disease: A Cross-Sectional Study at a Tertiary Care Hospital. Cureus 2023, 15(no. 8). [Google Scholar] [CrossRef]
- Kaka, Nirja; Sethi, Yashendra; Patel, Neil; Kaiwan, Okashah; Al-Inaya, Yana; Manchanda, Kshitij; Uniyal, Nidhi. Endocrine Manifestations of Chronic Kidney Disease and Their Evolving Management: A Systematic Review. Disease-a-Month 2022, 68(no. 12), 101466. [Google Scholar] [CrossRef]
- Horacek, Jiri; Dusilova Sulkova, Sylvie; Malirova, Eva; Dlabalova, Blanka; Safranek, Roman; Kubisova, Michaela; Kalousova, Marta; Maly, Jaroslav; Zak, Pavel. Haemodialysis-Induced Changes in Thyroid Hormones and Thyrotropin. Paper presented at the Endocrine Abstracts, 2013. [Google Scholar]
- Weaver, Virginia M; Fadrowski, Jeffrey J; Jaar, Bernard G. Global Dimensions of Chronic Kidney Disease of Unknown Etiology (Ckdu): A Modern Era Environmental and/or Occupational Nephropathy? BMC nephrology 2015, 16(no. 1), 145. [Google Scholar]
- Lo, Joan C; Chertow, Glenn M; Go, Alan S; Hsu, Chi-Yuan. Increased Prevalence of Subclinical and Clinical Hypothyroidism in Persons with Chronic Kidney Disease. Kidney international 2005, 67(no. 3), 1047–52. [Google Scholar] [CrossRef] [PubMed]
- Delanaye, Pierre; Ebert, Natalie; Melsom, Toralf; Gaspari, Flavio; Mariat, Christophe; Cavalier, Etienne; Björk, Jonas; Christensson, Anders; Nyman, Ulf; Porrini, Esteban. Iohexol Plasma Clearance for Measuring Glomerular Filtration Rate in Clinical Practice and Research: A Review. Part 1: How to Measure Glomerular Filtration Rate with Iohexol? Clinical kidney journal 2016, 9(no. 5), 682–99. [Google Scholar] [CrossRef]
- Zahra, Sabahat; Saleem, Muhammad Khurram; Ejaz, Khawaja Faizan; Akbar, Amna; Khan Jadoon, Sarosh; Hussain, Shoukat; Ali, Amir Iqbal; Ifty, Mehzabeen; Jannati, Sadia Zafur; Armin, Fariza. Prevalence of Nephropathy among Diabetic Patients in North American Region: A Systematic Review and Meta-Analysis. Medicine 2024, 103(no. 38), e39759. [Google Scholar] [CrossRef]
- Kashif, Md; Hussain, Md S; Anis, Mudassir; Shah, Papu K; Siddiqui, Mudassir A, Sr. Thyroid Dysfunction and Chronic Kidney Disease: A Study among the Northeastern Population of India. Cureus 2023, 15(no. 5). [Google Scholar] [CrossRef]
- Chehade, Joe M; Belal, Heiba F. Cross-Section of Thyroidology and Nephrology: Literature Review and Key Points for Clinicians. Journal of clinical & translational endocrinology 2024, 37, 100359. [Google Scholar]
- Rhee, Connie M; Brent, Gregory A; Kovesdy, Csaba P; Soldin, Offie P; Nguyen, Danh; Budoff, Matthew J; Brunelli, Steven M; Kalantar-Zadeh, Kamyar. Thyroid Functional Disease: An under-Recognized Cardiovascular Risk Factor in Kidney Disease Patients. Nephrology Dialysis Transplantation 2015, 30(no. 5), 724–37. [Google Scholar] [CrossRef]
- Reque Santivañez, Javier; Garcia Peris, Beatriz; Panizo Gonzalez, Nayara; Perez Alba, Alejandro; D’Marco, Luis; Collado Boira, Eládio. Subclinical Hypothyroidism in Advanced Chronic Kidney Disease Patients: Prevalence and Associated Factors. Journal of Thyroid Research 2022, no. 1, 1077553. [Google Scholar] [CrossRef] [PubMed]
- Sanai, Toru; Okamura, Ken; Onoue, Tomoaki; Ono, Takashi; Motomura, Kenichi; Miyazono, Motoaki; Shimamatsu, Kazumasa. Hemodilution Impacts Assessment of Thyroid Status before and after Hemodialysis in Patients with End-Stage Renal Disease. American Journal of Nephrology 2021, 51(no. 12), 988–94. [Google Scholar] [CrossRef]
- Shamsadini, S; Darvish-Moghaddam, S; Abdollahi, H; Fekri, AR; Ebrahimi, HA. Creatinine, Blood Urea Nitrogen and Thyroid Hormone Levels before and after Haemodialysis. Eastern Mediterranean Health Journal 2006, 12(no. 1/2), 231. [Google Scholar]
- Bauer, Michael; Glenn, Tasha; Pilhatsch, Maximilian; Pfennig, Andrea; Whybrow, Peter C. Gender Differences in Thyroid System Function: Relevance to Bipolar Disorder and Its Treatment. Bipolar disorders 2014, 16(no. 1), 58–71. [Google Scholar] [CrossRef] [PubMed]
- Mirahmad, Maryam; Mansour, Asieh; Moodi, Mitra; Safkhani, Elaheh; Haghpanah, Vahid; Asili, Pooria; Fakhrzadeh, Hossein; Payab, Moloud; Ebrahimpur, Mahbube; Khorashadi, Masoumeh. Prevalence of Thyroid Dysfunction among Iranian Older Adults: A Cross-Sectional Study. Scientific Reports 2023, 13(no. 1), 21651. [Google Scholar] [CrossRef] [PubMed]
- Samuels, Mary H. Hyperthyroidism in Aging. In Endotext [Internet]; 2021. [Google Scholar]
- Kumar, Manjula S; Safa, Ali M; Deodhar, Sharad D; Schumacher, O Peter. The Relationship of Thyroid-Stimulating Hormone (Tsh), Thyroxine (T4), and Triiodothyronine (T3) in Primary Thyroid Failure. American Journal of Clinical Pathology 1977, 68(no. 6), 747–51. [Google Scholar] [CrossRef]
- Fitzgerald, Stephen Paul; Bean, Nigel Geoffrey. The Relationship between Population T4/Tsh Set Point Data and T4/Tsh Physiology. Journal of Thyroid Research 2016, no. 1, 6351473. [Google Scholar] [CrossRef] [PubMed]
- Wiersinga, Wilmar M. T4+ T3 Combination Therapy: An Unsolved Problem of Increasing Magnitude and Complexity. Endocrinology and Metabolism 2021, 36(no. 5), 938–51. [Google Scholar] [CrossRef] [PubMed]
- Li, Hong; Yuan, Xiaolan; Liu, Lu; Zhou, Jiaojiao; Li, Chunyan; Yang, Peng; Bu, Le; Zhang, Manna; Qu, Shen. Clinical Evaluation of Various Thyroid Hormones on Thyroid Function. International journal of endocrinology 2014, no. 1, 618572. [Google Scholar] [CrossRef] [PubMed]
Figure 1.
Comparison of mean hormonal levels of thyroid hormones (i.e., TSH, T3, and T4) among normal (NP), non-dialysis kidney (NDKP), and dialysis patients (DP).
Figure 1.
Comparison of mean hormonal levels of thyroid hormones (i.e., TSH, T3, and T4) among normal (NP), non-dialysis kidney (NDKP), and dialysis patients (DP).
Figure 2.
Comparisons among the thyroid hormones (i.e., TSH, T3, and T4) based on (A) higher levels (>Ref value), (B) normal levels (within the Ref value), and (C) lower levels (<Ref value) among normal, non-dialysis kidney and dialysis patients. The TSH, T3, and T4 units were µIU/mL, ng/mL, and (µg/dL), respectively.
Figure 2.
Comparisons among the thyroid hormones (i.e., TSH, T3, and T4) based on (A) higher levels (>Ref value), (B) normal levels (within the Ref value), and (C) lower levels (<Ref value) among normal, non-dialysis kidney and dialysis patients. The TSH, T3, and T4 units were µIU/mL, ng/mL, and (µg/dL), respectively.
Figure 3.
Association of thyroid hormones (i.e., TSH, T3, and T4) among NP, NDKP, and DP. The plot shows the association between the levels of (A) TSH and T3, (B) TSH and T4, (C) T3 and T4 in NP; (D) TSH and T3, (E) TSH and T4, (F) T3 and T4 in NDKP; and (G) TSH and T3, (H) TSH and T4, (I) T3 and T4 in DP. * denotes a significant correlation at the 0.05 level, and ** denotes a significant correlation at the 0.01 level. The TSH, T3, and T4 units were µIU/mL, ng/mL, and µg/dL, respectively.
Figure 3.
Association of thyroid hormones (i.e., TSH, T3, and T4) among NP, NDKP, and DP. The plot shows the association between the levels of (A) TSH and T3, (B) TSH and T4, (C) T3 and T4 in NP; (D) TSH and T3, (E) TSH and T4, (F) T3 and T4 in NDKP; and (G) TSH and T3, (H) TSH and T4, (I) T3 and T4 in DP. * denotes a significant correlation at the 0.05 level, and ** denotes a significant correlation at the 0.01 level. The TSH, T3, and T4 units were µIU/mL, ng/mL, and µg/dL, respectively.
Table 1.
Participant demographics.
Table 1.
Participant demographics.
| |
NP |
NDKP |
DP |
| Overall, patients, n |
106 |
32 |
23 |
| Age in years (mean ± SD) |
38.64 ± 17.05 |
43.75 ± 17.54 |
43.00 ± 17.51 |
| Male, n |
32 |
10 |
8 |
| Age in years (mean ± SD) |
37.55 ± 17.01 |
45.7 ± 18.22 |
45 ± 15.90 |
| Female, n |
74 |
22 |
15 |
| Age in years (mean ± SD) |
39.12 ± 17.10 |
42.86 ± 17.53 |
41.93 ± 17.51 |
Table 2.
Participants (number and percentage) identified as having normal, high, and low levels of TSH, T3, and T4.
Table 2.
Participants (number and percentage) identified as having normal, high, and low levels of TSH, T3, and T4.
| Patients of different groups, n (%) |
Normal Level, n (%) |
Higher level, n (%) |
Lower level, n (%) |
| NP, n= 106 (100) |
| TSH |
80 (75.47) |
19 (17.92) |
7 (6.60) |
| T3 |
67 (63.20) |
17 (16.03) |
22 (20.75) |
| T4 (female) |
43 (40.56) |
11 (10.37) |
18 (16.98) |
| T4 (male) |
19 (17.92) |
9 (8.49) |
6 (5.66) |
| T4 overall |
62 (58.49) |
20 (18.86) |
24 (22.64) |
| NDKP, n= 32 (100) |
| TSH |
26 (81.25) |
5 (15.62) |
1 (3.12) |
| T3 |
20 (62.50) |
7 (21.32) |
5 (15.62) |
| T4 (female) |
17 (53.12) |
5 (15.62) |
1 (3.12) |
| T4 (male) |
4 (12.5) |
4 (12.5) |
1 (3.12) |
| T4 overall |
21 (65.62) |
9 (28.12) |
2 (6.25) |
| DP, n= 23 (100) |
| TSH |
17 (73.91) |
4 (17.39) |
2 (8.69) |
| T3 |
13 (56.52) |
5 (21.73) |
5 (21.73) |
| T4 (female) |
9 (39.13) |
4 (17.39) |
2 (8.69) |
| T4 (male) |
4 (17.39) |
4 (17.39) |
0 (0.0) |
| T4 overall |
13 (56.52) |
8 (34.78) |
2 (8.69) |
Table 3.
The levels of thyroid hormones (i.e., TSH, T3, and T4) among normal, non-dialysis kidney, and dialysis patients, stratified by sex and age category.
Table 3.
The levels of thyroid hormones (i.e., TSH, T3, and T4) among normal, non-dialysis kidney, and dialysis patients, stratified by sex and age category.
| Category |
Thyroid hormones |
Hormonal levels (mean) |
| NP |
NDKP |
DP |
| Male |
TSH |
3.97 |
4.10 |
4.26 |
| T3 |
1.54 |
1.39 |
1.13 |
| T4 |
8.2 |
9.33 |
9.65 |
| Female |
TSH |
4.71 |
3.96 |
5.60 |
| T3 |
1.10 |
1.24 |
1.86 |
| T4 |
8.36 |
9.55 |
9.67 |
| Age<45 y |
TSH |
3.94 |
4.64 |
5.54 |
| T3 |
1.24 |
1.40 |
2.32 |
| T4 |
8.59 |
11.07 |
8.74 |
| Age>45 y |
TSH |
5.89 |
3.25 |
4.64 |
| T3 |
1.24 |
1.23 |
1.12 |
| T4 |
8.11 |
7.95 |
10.77 |
Table 4.
Summary of Implications of Thyroid Hormone Levels in Kidney Disease Patients.
Table 4.
Summary of Implications of Thyroid Hormone Levels in Kidney Disease Patients.
| Patient Group |
Thyroid Hormone Levels/Activity |
Implications |
| Dialysis patients with renal complications |
Moderately enhanced thyroid hormone levels and activity |
Suggests potential benefits of routine dialysis for CKD/ESRD patients |
| Non-dialysis patients with kidney disease |
Not significantly enhanced thyroid hormone levels and activity |
Supports the need for further investigation with larger cohorts |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).