Introduction
The clinical and molecular delineation of Ehlers–Danlos syndrome (EDS) has been hampered by a continued focus on extreme findings [
1,
2] and one gene-one type correlations [
3]. Skeletal and skin [
1,
2,
3,
4] but not integral neuro-autonomic findings [
5] dominate diagnostic criteria [
6] while mutations in genes like COL5A1/A2 are exclusively associated with EDS types--classical EDS 1 and 2 [
7] in this case [130000/130010, 6 digit numbers referencing entries in the
www.omim.org database, 8]--without acknowledging that collagen fibers are formed from multiple collagen types with type V being a catalyst for fibril formation [
9]. Lacking in these approaches are a) the systematic documentation of findings that can differentiate the multisystemic patterns of EDS types, b) recognition that mutations of different type or position cause different disorders (seven diseases besides Marfan syndrome (154700] are associated with FBN1 gene mutations, 134797), c) assumptions, despite contrary evidence [
9], that the striking trait of double-jointedness as manifest in EDS hypermobile type [
4] was associated with no gene changes, and d) appreciation that one-copy (heterozygous) variants in a gene may contribute to different EDS phenotypes than two-copy (homozygous, compound heterozygous) variants. A cogent example is the Ulrich muscular dystrophy (616470) associated with biallelic collagen type XII alpha-1 chain
COL12A1 (120320) variants versus the Bethlem myopathy/myopathic EDS overlap disorder (616471) associated with monoallelic variants [
10,
11].
Systematic documention of 120 history-physical findings in 1261 EDS patients coupled with whole exome sequencing in 906 of them [
12,
13] agreed with prior work [
14,
15] that showed type-independent, multigenic, and novel heterozygous variation in common forms of EDS [
4,
7]. Among the latter EDS patients were six (four in one family) with variations in the zinc finger 469 (
ZNF469) gene (612078) that, when present in both gene copies, were previously associated with brittle cornea syndrome (229200). Brittle cornea syndrome shares an asthenic (Marfanoid) habitus, joint laxity, scoliosis, unusual scarring, and mitral valve prolapse with EDS but includes severe corneal fragility that leads to keratoconus and blindness [
16,
17]. Hypothesis that one-copy
ZNF469 variants contribute to a milder connective tissue dysplasia best classified as hypermobile EDS is here addressed by describing 8 EDS patients plus 14 from the literature [
18,
19,
20,
21] with heterozygous
ZNF469 gene variants.
Key for this study is holistic delineation [
12] of craniofacial (myopia, corneal issues, high palate), skeletal (asthenic habitus, joint flexibility-instability), skin (elasticity, unusual scarring), cardiac (mitral valve prolapse, aneurysms), neural (migraines, neuropathy, poor balance), and dysautonomia findings that occur in EDS. The latter include those of postural orthostatic tachycardia syndrome or POTS [chronic fatigue, syncope, tachycardia, etc., 22], of mast cell activation syndrome or MACS [reactive skin, asthma, etc., 23], and of irritable bowel syndrome or IBS [irregularity, bloating-nausea, swallowing difficulties, etc., 24].
3. Results
Clinical Presentations
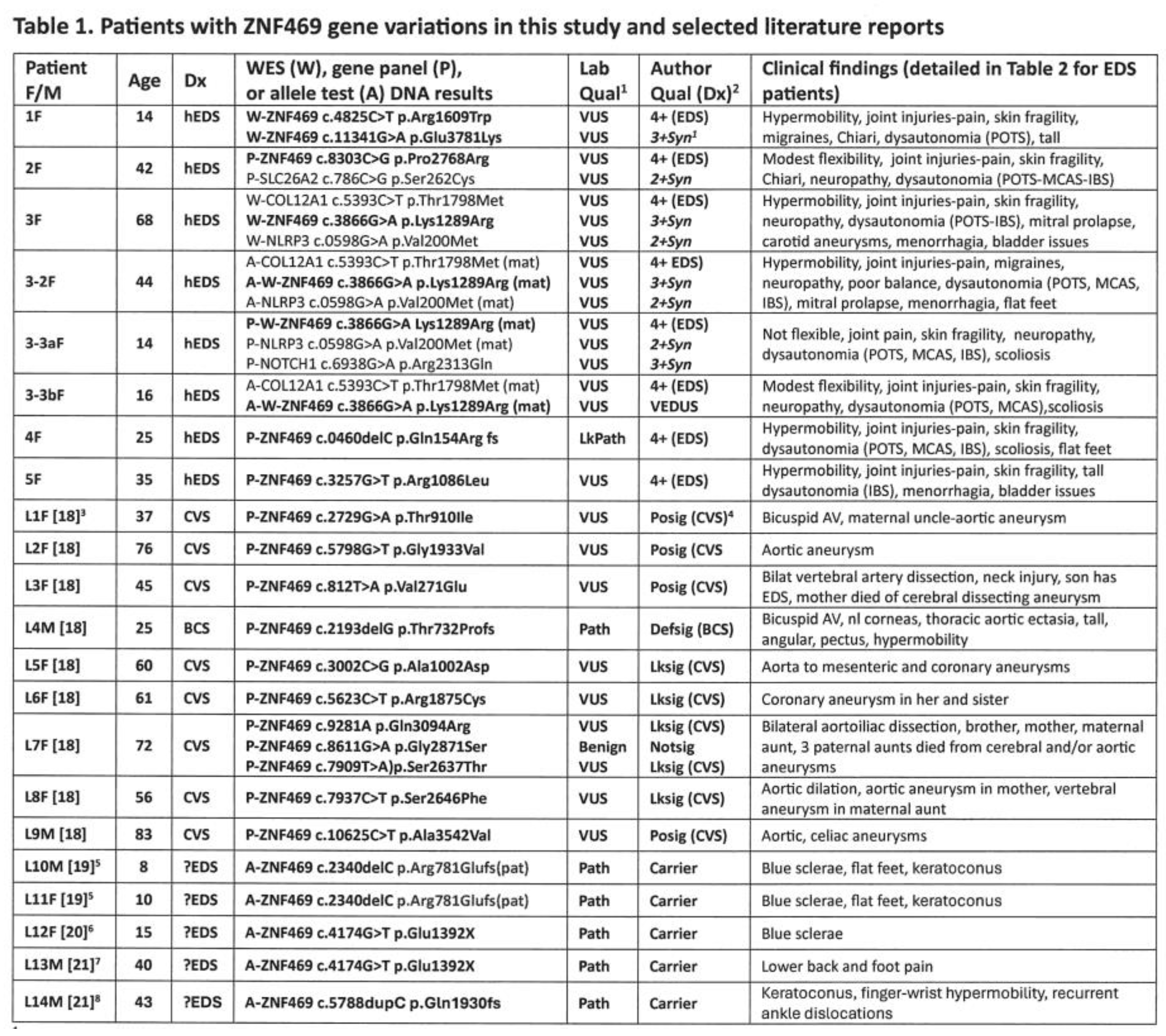
Three patients of ages 13, 42, and 68 years (numbers 1-3 in Table 1) are presented in detail to show the variable impact of one-copy ZNF469 gene changes. It should be noted that neither these nor the other 5 EDS patients in Table 1 had the keratoconus, keratoglobus, or corneal ruptures typical of brittle cornea syndrome [16, 17--see Table 2]. Diagnoses of EDS provided benefits of understanding and therapy [wise exercise to build muscle around joints, bracing, hydration-salt to increase blood volume and decrease venous pooling/adrenergic stimulation, gluten-dairy restriction, 12], follow-up care and counseling not described here.
Patient 1
A 13-year-old female was referred for evaluation of connective tissue laxity after Chiari deformation and other typical symptoms caused her mother to suspect EDS. They reported 35 of 66 applicable history-physical findings (Table 2), significant impairment beginning at age 11 when activities such as dance were limited by joint pain, fatigue, and fractures (over 10 of her hands and ankles). She had infantile colic and was recognized to be hypermobile with ability to perform double-jointed tricks and clumsiness, developing knee subluxations and arthralgia in her shoulders, knees, and ankles. Other findings included need for orthodontics, slow healing, neurologic issues (migraines, chronic daily headaches, and neuralgia along with a 6-7 mm Chiari herniation), POTS findings (dizziness, chronic fatigue, tachycardia, brain fog, heat-cold sensitivity, salt fancy) with the asthma seen in MCAS and difficulty swallowing typical of IBS. Worsening headaches and fatigue plus trouble breathing with choking prompted a visit to Mayo Clinic where diagnoses of Chiari and POTS were made, the latter favored by a sleep study showing nonspecific arousals and poor latency despite a negative tilt-table testing. Perhaps unrelated were prior diagnoses of psoriasis and, at age 12, splenomegaly with mononucleosis infection.
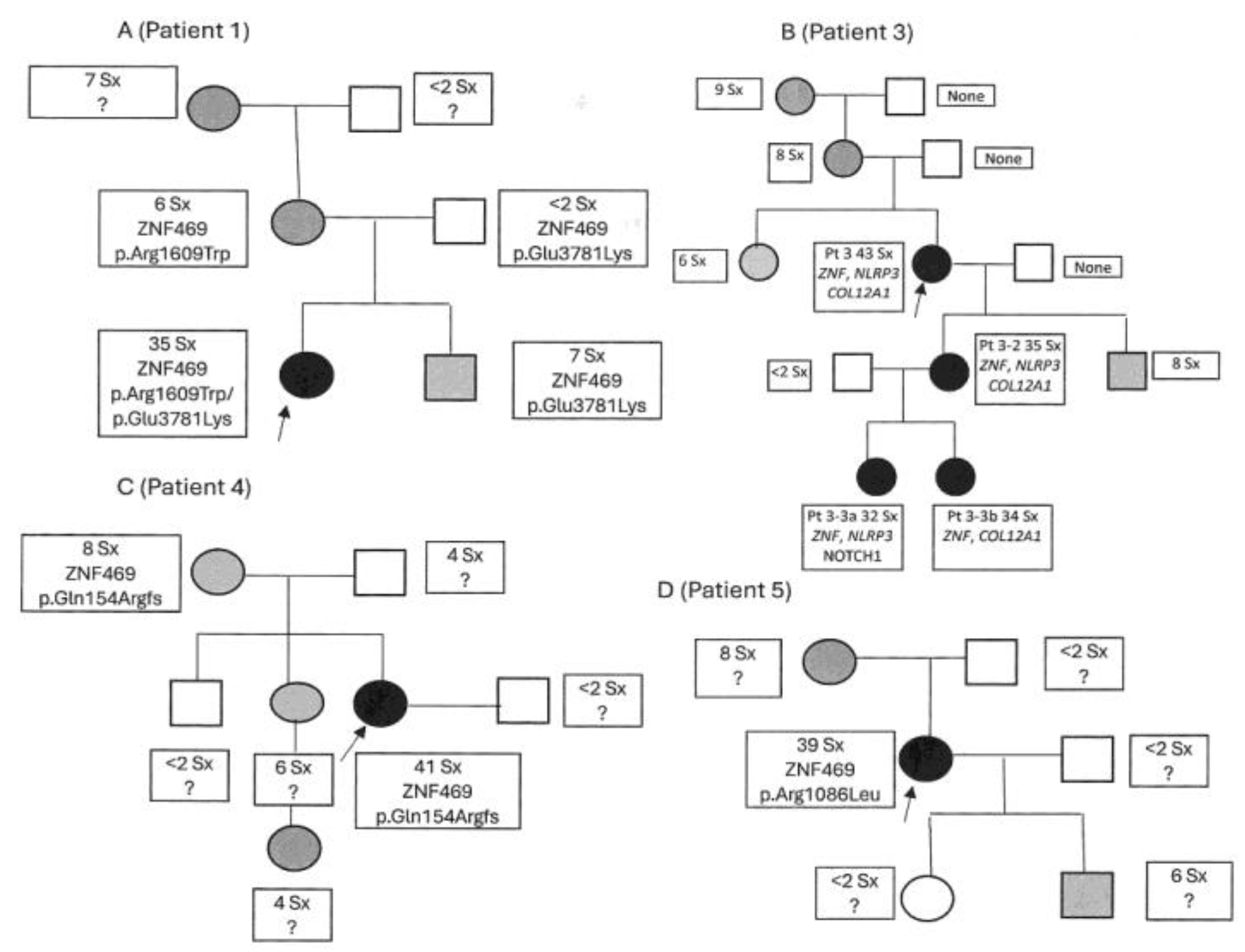
Family history, partially diagrammed in
Figure 1A, included a 10-year-old brother with early feeding problems and clumsiness, increased flexibility, joint injuries, easy bruising, and IBS symptoms. Mother had many suggestive symptoms including flexibility, several fractures, neck and back pain, ruptured tendon, easy bruising with scars, migraines with posterior headaches, menorrhagia, IBS with gastroparesis and cholecystectomy, and frequent rashes and hives with psoriasis. The maternal grandmother had similar symptoms with crippling rheumatoid arthritis, osteopenia, and migraines.
Physical examination showed tall stature (87
th centile) with proportionate weight and without an asthenic or angular build. Her facial appearance was normal without chiseled features or tight skin. She had a high palate that correlated with her orthodontic treatment for crowded teeth, soft and translucent peripheral skin, ability to perform 7 of 9 Beighton maneuvers [
31]. She could join hands, one over shoulder and the other around back and perform the reverse prayer (Namaskar) sign as a sign of large joint maneuvers but had normal strength and balance.
Clinical impression was EDS hypermobile type with dysautonomia and associated . Whole exome sequencing revealed the compound heterozygous ZNF469 mutation in Table 1, testing of the parents confirming the trans orientation with the more disruptive arginine to tryptophan amino acid change in mother.
Patient 2
A 42-year-old female was referred for a second opinion regarding her diagnosis of EDS and interpretation of DNA results, reporting 34 of 66 applicable findings in Table 2. She recognized increased flexibility with ability to perform double-jointed tricks when she was 11 or 12, having a knee ligament tear that limited her sports activities in high school. Diffuse joint pain began in later adolescence along with painful rib subluxations, clumsiness and flat feet contributing to poor balance. Posterior headaches and neuropathy symptoms in her 30s were explained in part by craniocervical instability and Chiari deformation demonstrated by upright head MRI studies. Dysautonomia symptoms included dizziness, syncope, tachycardia, and anxiety typical of POTS, hives-rashes and food-medicine intolerances typical of MCAS, and irregularity-nausea typical of IBS. She recognized color changes and swelling in her feet after standing that along with menorrhagia and bladder symptoms were signs of blood pooling in her lower limbs and pelvis.
Family history (not diagrammed) showed that only her 19-year-old son with flexibility, joint pain, easy bruising, and flat feet had some typical findings of EDS.
Physical findings included average stature with long fingers and ability to perform the Walker-Murdoch and Steinberg signs [
32], soft and velvety skin without unusual scars, flat feet with toeing-in and poor balance while walking. Her Beighton score [
31] was just above average (5/9) although it was probably higher when young. She had large joint flexibility shown by joining hands, one over shoulder and the other around back, and performing the reverse prayer sign.
Clinical impression was hypermobile EDS, DNA testing showed the disruptive ZNF469 proline to arginine replacement in Table 1 along with a solute carrier family SLC26A2 (606718) mutation that caused a serine to cystine amino acid replacement. The additional variant was qualified as having strong diagnostic utility with synergistic action (2+syn, Table 1) because its associated diseases, e. g., 226900, affected skeletal connective tissue.
Patient 3
A 68-year-old woman requested evaluation for chronic pain since childhood and later diagnoses of carotid artery aneurysms with fibromuscular dysplasia. She reported 43 of 70 applicable findings (Table 2), more severe symptoms beginning at age 12 with menorrhagia and fluctuating blood pressure. Infantile findings of colic-poor feeding and low muscle tone were recalled, the latter interesting in view of her collagen type XII alpha-1 chain (
COL12A1, 120320) defect [
10,
11]. She recognized her flexibility and had early joint pain with 3 fractures involving her back and ankles that limited her adolescent activities. Later popping joints with TMJ issues and multiple joint surgeries were accompanied by back fractures, spinal arachnoid cyst and disc degeneration that needed orthopedic and neurosurgeries. Other issues included skin fragility (easy bruising, elasticity), menorrhagia with bladder issues (frequent infections), mitral valve prolapse, and inguinal hernia. Neuromuscular complaints ranged from migraines numbness and tingling, poor balance to neuropathy and muscle aches. She has constipation/diarrhea, bloating/reflux/stomach pain, gall bladder issues, difficulty swallowing, nausea as symptoms of IBS, several of POTS (orthostatic dizziness, syncope, sleep problems, and brain fog plus the the food-medication intolerances suggestive of MCAD. The carotid aneurysms were being monitored, one calcified and the other still at risk for bleeding, other diagnoses being a bowel perforation, Hirschsprung disease, ankylosing spondylitis, early hearing loss, and varicosities needing venous ablation.
Family history diagrammed in
Figure 1B shows that her mother, grandmother, and sister had many symptoms of EDS-dysautonomia, findings in her daughter (patient 3-2) and two grandaughters (patients 3-3a, 3-3b) detailed in Table 2.
Physical exam showed an angular build of average height (wide wing span; long fingers, ability to perform the Walker-Murdoch and Steinberg signs) [
32]; soft, translucent, and elastic skin; lordosis; and poor balance by tandem walk. Beighton score [
31] was 9 of 9 even at her age and she could do the joining hand and reverse prayer signs of shoulder flexibility that were present in patients 1 and 2.
Clinical impression was hypermobile EDS with remarkable flexibility, multiple joint injuries, and significant neuro-autonomic complications. DNA testing documented heterozygous variation in the
COL12A1,
NLRP3, and
ZNF469 genes, all causing disruptive amino acid changes. All three were tranmitted to the daughter (patient 3-2), the
ZNF469 variant to both granddaughters. The
ZNF469 variant was qualified as an additional variant with synergistic action since many EDS patients with
COL12A1 variation had been reported, [
10,
11] including 22 in addition to patient 3 in reference [
12]. Synergistic action of the
NLRP3 gene (606416) variant was designated because among its associated disorders were those with joint pain and mast cell-like inflammation (e. g., 191190) .
Clinical and DNA Findings
Table 1 lists 8 EDS patients, 4 in the same family, and 14 from the literature with
ZNF469 gene changes, all heterozygous except for patients 1 (compound heterozygous) and L7 (three heterozygous variants, cis or trans phase not determined). Clinical findings of the EDS patients are shown in the last column and detailed in Table 2, patients L1-L9 [
18] and L10-L14 [
19,
20,
21] the only
ZNF469 heterozygotes with EDS-like symptoms found in the open access literature. Four patients (1, 3) had whole exome sequencing, others (2, 3a, 4-5, L1-9) 35 to 37-gene panels identified as TAAD (thoracic aortic aneurysm and dissection) by commercial companies, the rest testing for alleles from by exome-panel testing. These TAAD panels screen most genes commonly associated with connective tissue dysplasias including those causing Marfan (154700), forms of Loeys-Dietz (e. g., 609192), and the recognized types of Ehlers-Danlos syndrome [
3].
Patients 1-2, 4-5, and the 3, 3-2, 3-3a, 3-3b family all had systematic evaluations for 120 findings (see Methods), patients 4-5 having similar finding patterns and diagnoses of EDS, hypermobile type, as those detailed in the patient presentations. All of their
ZNF469 variants were qualified as variants of uncertain significance (VUS) except for patient 4’s frame-shift mutation judged as likely pathogenic. Based on the presence of
ZNF469 variation in more than 3 EDS patients [
27] and its homology with collagen type I [
17], pluses were given for association with EDS-connective tissue dysplasia mechanisms [
12] that converted VUS to 4+ diagnostic utility qualifications for the primary
ZNF469 variants in patients 1, 2, 4, and 5 (column 6, Table 1). The additional
ZNF469 variants in patient 3 and her family members contributed synergistically to 4+ diagnostic utility [
12,
26], their
COL12A1 variant judged primary because of longer association with EDS.
The ZNF469 DNA variant is qualified as having synergistic contribution to the EDS-dysautonomia symptoms of patients 3, 3-2, and 3-3b as was the NLR family pyrin-containing NLRP3 (606416) DNA variant associated with inflammatory arthralgia-myalgia and MCAS findings (e. g., 191900) in patients 3, 3-2, and 3-3a. The sulfate-transporting solute carrer family SLC26A2 gene (606718) variant in patient 2 was also qualified as synergistic because of its association with connective tissue dysplasia (bone, i. e., 222600), as was the NOTCH1 transmembrane receptor (190198) variant in patient 3-3a associated with skin defects (616028) or aortic valve disease (109730).
Note the similar profiles of early childhood, facial, joint-skeletal, skin, neuromuscular, cardiac, dysautonomia, and genitourinary findings among the EDS patients with
ZNF469 DNA variants in Table 2, equally resembling the profile of all female EDS patients with a higher frequency of infantile clumsiness (100 versus 55%) as the only significant difference. Some overlap of these EDS findings with those of brittle cornea syndrome is shown in the last column of Table 2, the qualitative descriptions of brittle cornea patients in the literature [
17,
18,
19,
20,
21,
33,
34] requiring 1-4+ grades rather than quantitative proportions. Nevertheless, requisite findings of brittle cornea syndrome like corneal fragility and more consistent ones like myopia, blue sclerae, high palate, joint hypermobility, scoliosis, skin fragility, and mitral valve prolapse (3-4+ in Table 2) confirm its classification as a connective tissue dysplasia [
35] that needed differentiation from the kyphoscoliotic type of EDS [
16]. Overlap of findings like joint hypermobility, velvety skin, long fingers, and kyphoscoliosis is emphasized by the 5 brittle cornea patients with a 12 nucleotide deletion beginning at codon 2939 of the ZNF469 protein [
36].
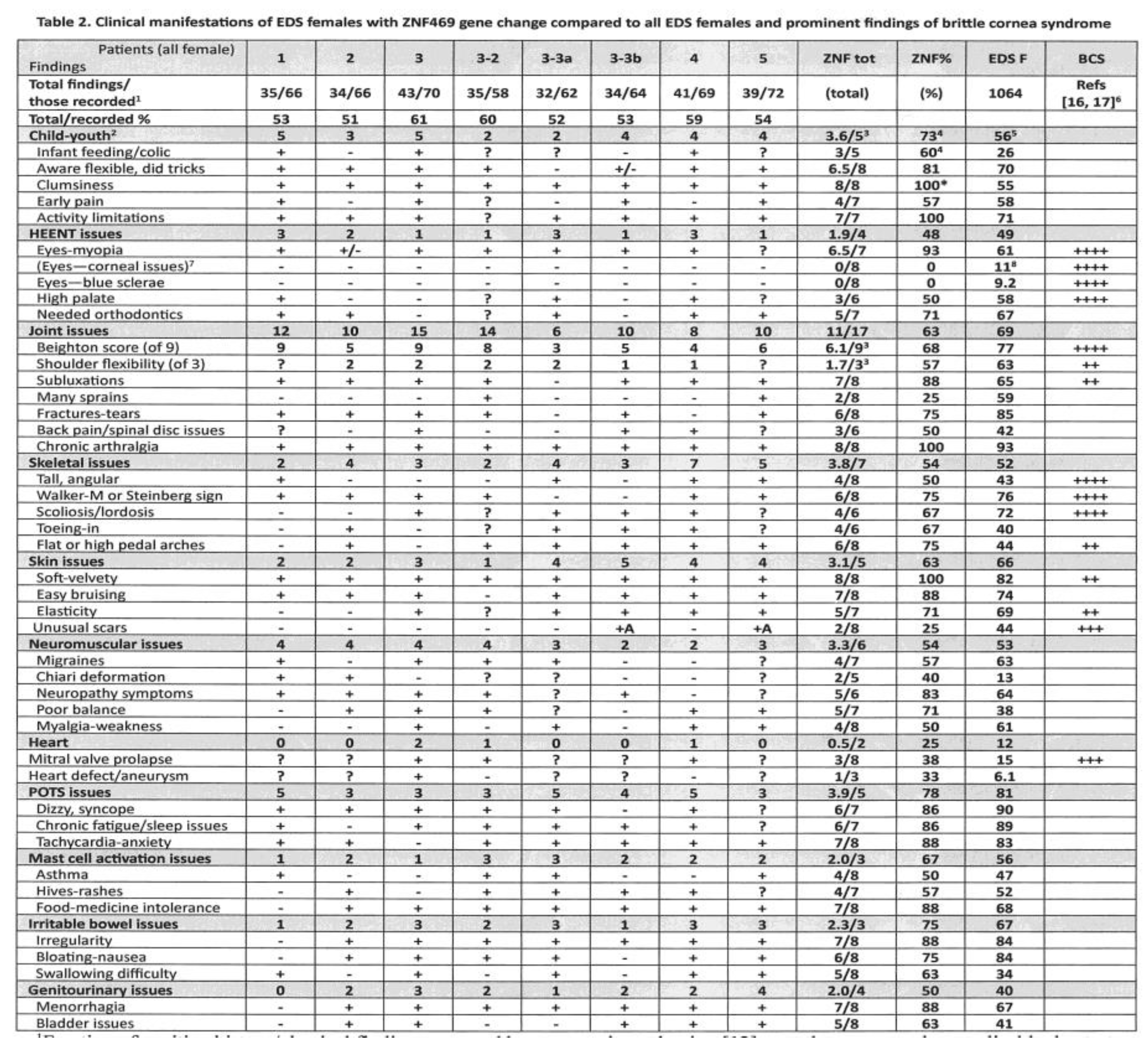
1Fraction of positive history/physical findings reported by systematic evaluation [
12]
over those assessed or applicable due to type of evaluation and age of ZNF patient; 2history-physical findings are combined into groups (shaded), selected ones listed below each group;
3average number of positive findings in ZNF patients/total findings in a category are given in the shaded lines (also for Beighton and shoulder flexibility scores);
4percentages of average or individual findings over the totals, e. g., 3.6 child-youth findings/5 total = 73%, 3 of 5 patients for whom infant/feeding issues registered = 60%);
5equivalent percentage of that finding in 1064 EDS females having systematic evaluations;
61-4+ symptom severity provided for brittle cornea syndrome (BCS) patients since systematic examinations were not reported [
16,
17,
18,
19,
20,
21,
33,
34];
7corneal issues were not included on the systematic evaluation forms;
8dry eyes were the only corneal issue for EDS females, a finding volunteered rather than systematically evaluated; *significantly different p<0.05 [
30]; A, atrophic white scars typical of hypermobile EDS [
4] rather than papyraceous (knurled, discolored) scars typical of classical EDS [
7]; BCS, brittle cornea syndrome; EDS, Ehlers-Danlos syndrome; HEENT, head-eyes-ears-nose-throat; Walker-M, Walker-Murdoch sign; ZNF, zinc finger 469 gene.
Multigenic contribution to EDS-dysautonomia symptoms is supported by the family histories in
Figure 1, the probands having systematic evaluations and their relatives anecdotal descriptions from a checklist form listing 14 EDS-dysautonomia findings. Note the parental heterozygotes of patient 1, mother with the ZNF469 arginine to tryptophan substitution having 6 characteristic symptoms of EDS but father with the similarly disruptive glutamate to lysine substitution described by the historian mother as having less than two (in his case, chronic joint pain). Variable expression is also indicated by patient 1’s brother with the same mutation as their father but reported as having 7 EDS symptoms, another example perhaps being the mother of patient 4 who was less affected (8 symptoms) by the same
ZNF469 frame-shift mutation although not systematically evaluated. The presence of several affected relatives in each family and the similarity of patient 3, 3-2, 3-3a/b profiles impacted by 2-3 DNA variants further suggests multigenic contribution to this more common (hypermobile) EDS phenotype.
A return to Table 1 further emphasizes the variable phenotypes of
ZNF469 heterozygotes and the limitations of this study by listing the findings of 14 such patients from the literature [L1-L14, 18-21]. Patients L1-L9 [
18], predominantly (7 of 9) female like the 8 with EDS, also had variants qualified as VUS with the exception of L4’s frame-shift mutation. The 9 patients with
ZNF469 gene changes were drawn from 135 diagnosed with arterial aneurysms or dissections in a cardiovascular genetic practice [
18], their brief clinical presentations understandably emphasizing vascular findings. Familial predisposition is again indicated by 5 of the 9 having relatives with aneurysms, patient L3 having a son diagnosed with EDS. Patient L4 also has findings like hypermobility with a tall, angular build and pectus that are suggestive of EDS as are the blue sclerae and flat feet of L10 and L11 [
19], the blue sclerae, joint laxity or pain of L12-L14 [
20,
21]. The latter heterozygotes were ascertained as relatives of patients with brittle cornea syndrome, their DNA frame-shift and terminating
ZNF469 mutations qualified as pathogenic because of that relationship. Variable expression in heterozygoes is again emphasized by the 3 asymptomatic relatives of patient L12 and by patient L13 with the same variant as L12.
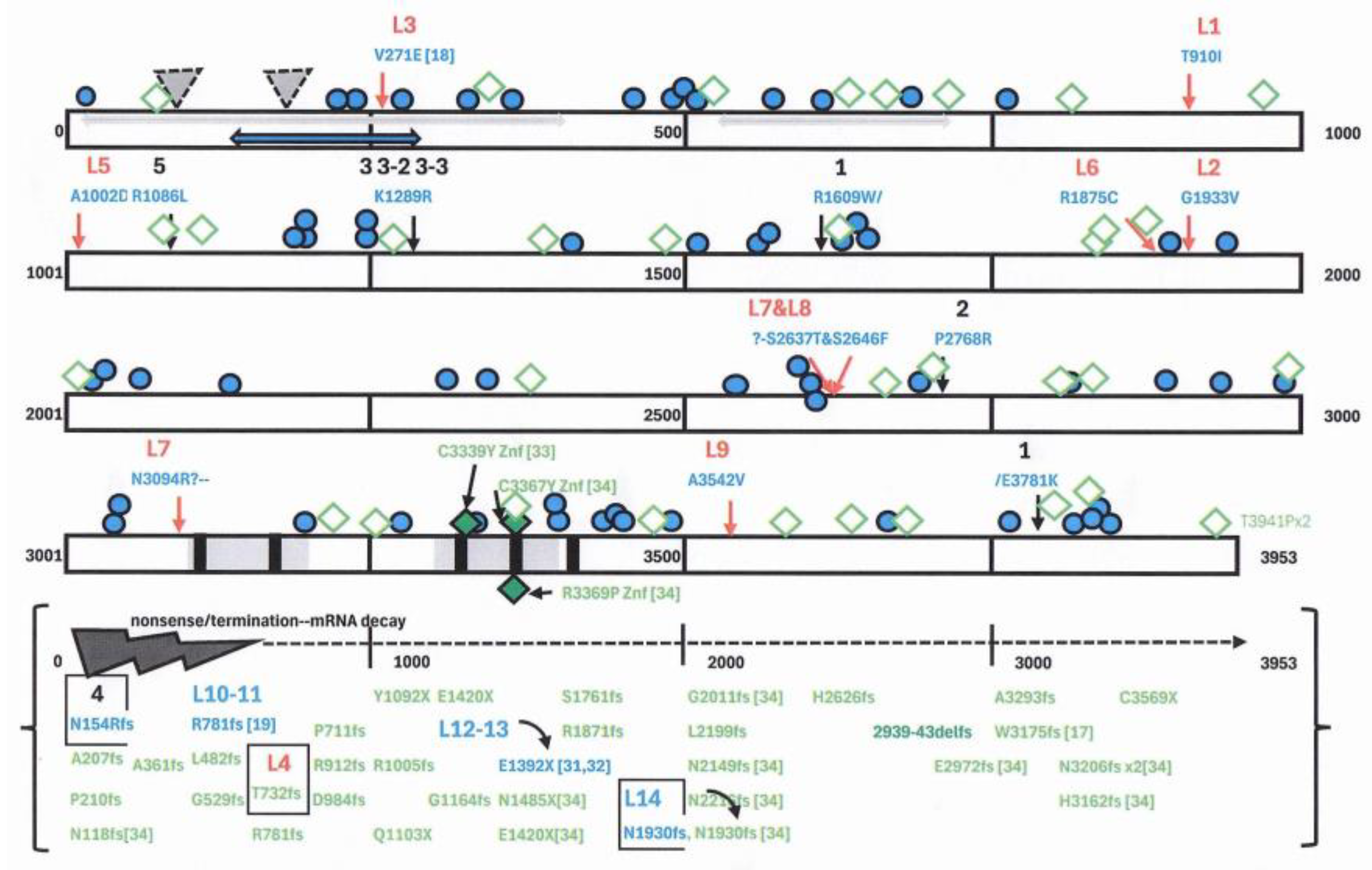
Locations of Table 1 patient and other selected literature mutations within the ZNF469 protein sequence are shown in
Figure 2, missense mutations in the upper 4 rows (each representing 1000 amino acids) and those causing messenger RNA decay (frame-shift, terminating) in the lowest fifth (all 3953 amino acids graphed). Data conflict on the number of exons but the two introns between three are shown by inverted triangles as represented by MANE select [
37], amino-terminal regions homologous to a herpes virus glycoprotein with deubiquinating activity [thin gray line, 38] or collagen type I [blue arrows, 17] and more carboxy-terminal zinc finger (black bars) or putative nucleotide binding sites (wide gray line) shown by reference to Uniprot [
39]. EDS patients 1-5 are numbered in black, L1-L9 ascertained by aneurysm-dissection numbered in red [
18], L10-L14 designated as brittle cornea carriers in blue [
19,
20,
21], all DNA variants associated with EDS findings in blue and those with brittle cornea in green. Patient 1’s two missense variants are connected by / to show their trans conformation, the two in patient L7 accompanied by ‘-?’ to show that their cis or trans phase was not determined (the third benign
ZNF469 variant of patient L7 in Table 1 is not shown).
Selected from the ClinVar database [
29] are missense variants from 60 patients with EDS mentioned as the testing indication (blue circles) and 36 from patients with brittle cornea syndrome as the indication (green triangles), all of them having VUS or conflicting (benign-VUS- occasionally pathogenic) qualifications except for three in the zinc finger regions qualified as pathogenic [green-filled triangles, 33,34]. Two of the latter variants had very disruptive amino acid changes (C3339Y and C3367Y) that affected crucial cysteine residues in the consensus zinc finger sequence [
40]. Another 32 frame-shift or chain-terminating DNA variants associated with brittle cornea syndrome are listed by codon position in the bottow row along with those of patients 4 and L10-L14 that would be associated with decay of messenger RNA from that allele.
The missense ZNF469 gene variants above and its message-decaying variants below are distributed across the coding region, the three zinc finger mutations providing the clearest genotype-phenotype correlation. Mutational hotspots may be emerging for regions around the family 3 and patient L3 variants within the collagen I homology region and for the distal regions around the patient L2-L6, L7-8, and patient 1 E3781K variants. It seems clear that variation in one
ZNF469 allele likely contributes to an EDS phenotype, excepting those in zinc fingers [
33,
34], or the frame-shift in patient L4 [
18], while those in both (excepting those in patients 1 and possibly L7) likely contribute to a brittle cornea syndrome phenotype. Even this conclusion is hampered by a paucity of clinical information and the fact that the 125 variants selected from ClinVar are characterized individually without reference to the presence or identity of a partner allele.
Discussion
The
ZNF469 gene was first identified among DNA segments encoding large proteins in human brain [
41], its mapping to chromosome 16q24.2 near the melanocyte-stimulating hormone receptor MSHR gene (155555); this may explain why some patients with
ZNF469-related brittle cornea syndrome have red hair [
42]. The latter syndrome was shown to have multiple findings of connective tissue dysplasia [
16,
17,
18,
19,
20,
21,
33,
34], correlating with
ZNF469 as a transcription factor regulator of extracellular matrix structure [
35] and its homology to the helical regions of COL1A2 (120160), COL4A1 (120130), and COL1A1 (120150) that are highly expressed in cornea [
17]. It was not surprising, then, that brittle cornea syndrome was categorized as a type of EDS that had to be distinguished from other types with corneal fragility [
16] or that the ZNF469 gene was found variant in 6 of 906 EDS patients having whole exome sequencing [
12].
Seven heterozygous EDS patients (excluding patient 1) and 13 from the literature [18–21,?excluding patient L7] with connective dysplasia findings [
18] plus 60 EDS patients from the ClinVar database [
29] had low-frequency, disruptive, and heterozygous
ZNF469 gene changes, providing strong evidence for correlation rather than coincidental association. The cases in Table 1 and Table 2 further correlate one-copy
ZNF469 gene changes with EDS-like findings and two-copy changes with brittle cornea syndrome, exceptions being 3 missense one-copy mutations in the zinc finger region [
32,
33,
34], patient L4, or the two-copy mutation in EDS patient 1. The predominance of female sex in all the EDS and in 9 of 14 literature cases may indicate enhanced expression of connective tissue findings in ZNF469 heterozygous females, correlating with the 85% of 2000-plus EDS patients who were female [
9,
12] as a reflexion of their greater flexibility [20% of women as opposed to 10% of men can perform over 4 Beighton maneuvers, 4] . teYet a conclusion has the same difficulties as the majority of medical insights from all-gene (genomic) testing.
Complicating the conclusion that heterozygous
ZNF469 change can contribute to EDS is the fragmentary documentation of patient findings that accompany DNA results in the literature and in databases like ClinVar [
29]. Too often extreme findings like corneal ruptures [
16,
17], aneurysms and dissections [
18], or kyphoscoliosis [
16] are mentioned rather than systematic documentation [
12,
13] that would determine if the extreme was an exceptional presentation of a condition or a different condition. Second is the application of biased perspectives from targeting of candidate genes [
23,
24], mentioned examples being the assumptions that all variations in collagen type V genes cause a classical type of EDS [
7] and all collagen type XII gene variations cause Bethlem myopathy [
10,
11]. Network interactions and influence of background genes explain why the majority of 53 patients with collagen type V mutations were shown by systematic evaluation to have hypermobile rather than classical EDS [
12]; even the classical homozygous sickle cell anemia mutation may cause milder symptoms in Jamaicans [
43].
These facts lead to a key concept that will facilitate the use of genome sequencing for scientific investigation and precision medicine: Genetically influenced diseases, particularly the more common multisystemic, multifactorial ones like EDS, arise from alterations of mechanisms involving gene networks. Matching of clinical phenotypes with variant DNA sequence genotypes must therefore proceed by:
1) Delineating the patient’s specific type of disease by quantitative percentages of findings
2) Relating those finding profiles to underlying pathogenic mechanisms
3) Qualifying DNA variants hierarchically first by their relation to disease mechanisms (tissue laxity and dysautonomia-inflammation for EDS) and then, if sufficient variant matches have been found [
27], to the patient’s specific disease
4) Recognizing that multiple gene changes will be involved in most diseases, many not recognized by commercial interpreters while others, as predicted by DNA sites that transcription factors like ZNF469 must bind to, remain undiscovered until the “dark” genome is characterized [
44]
5) Scaled rather than dyadic variant qualification [e. g, with 1-4+ diagnostic utility--12, 13] to minimize the ubiquitous and unhelpful VUS qualifications that detering physician and insurance acceptance of genomic testing [
45].
Further evidence linking ZNF469 with connective tissue dyplasias besides brittle cornea syndrome must come from systematic analysis of findings in families with keratoconus, brittle cornea syndrome, or disorders like EDS. The study documenting select symptoms in two ZNF469 heterozygotes [L13-14, 21] screened a total of 132 individuals in 11 familes with keratoconus and found 35 were heterozygous for 11 ZNF469 variants counting the 2 in Table 1. Of 54 family members with keratoconus, 16 (30%) had a variant compared to 19 of 78 (24%) of asymptomatic relatives, the insignificant difference suggesting a low penetrance for heterozygous ZNF469 gene change. Quantitative profiles rather than selected findings (aneurysms, keratoconus) or pattern diagnoses (fibromyalgia, chronic fatigue syndrome, premature assignment of EDS types) must complement ZNF469 heterozygous DNA variations if EDS or other associations are to be proven.
The participation of ZNF469 protein in tissue fiber formation suggested by collagen homology [
17] and its ability as a transcription factor [
46] to regulate connective tissue formation [
35,
46] supports a causal relationship between its DNA variations and the EDS-like findings in Table 1 [
1,
2,
3,
12,
13], extending to the particular hypermobile type [
4] in patients 1-8 as justified by their finding profiles in Table 2 [
12]. Proof of this causal relationship, as for any of the myriad genomic changes being found in complex diseases, will require more insightful matching of finding-DNA variant combinations, an enterprise ideally suited to large-language models of artificial intelligence [
47,
48].