Submitted:
02 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
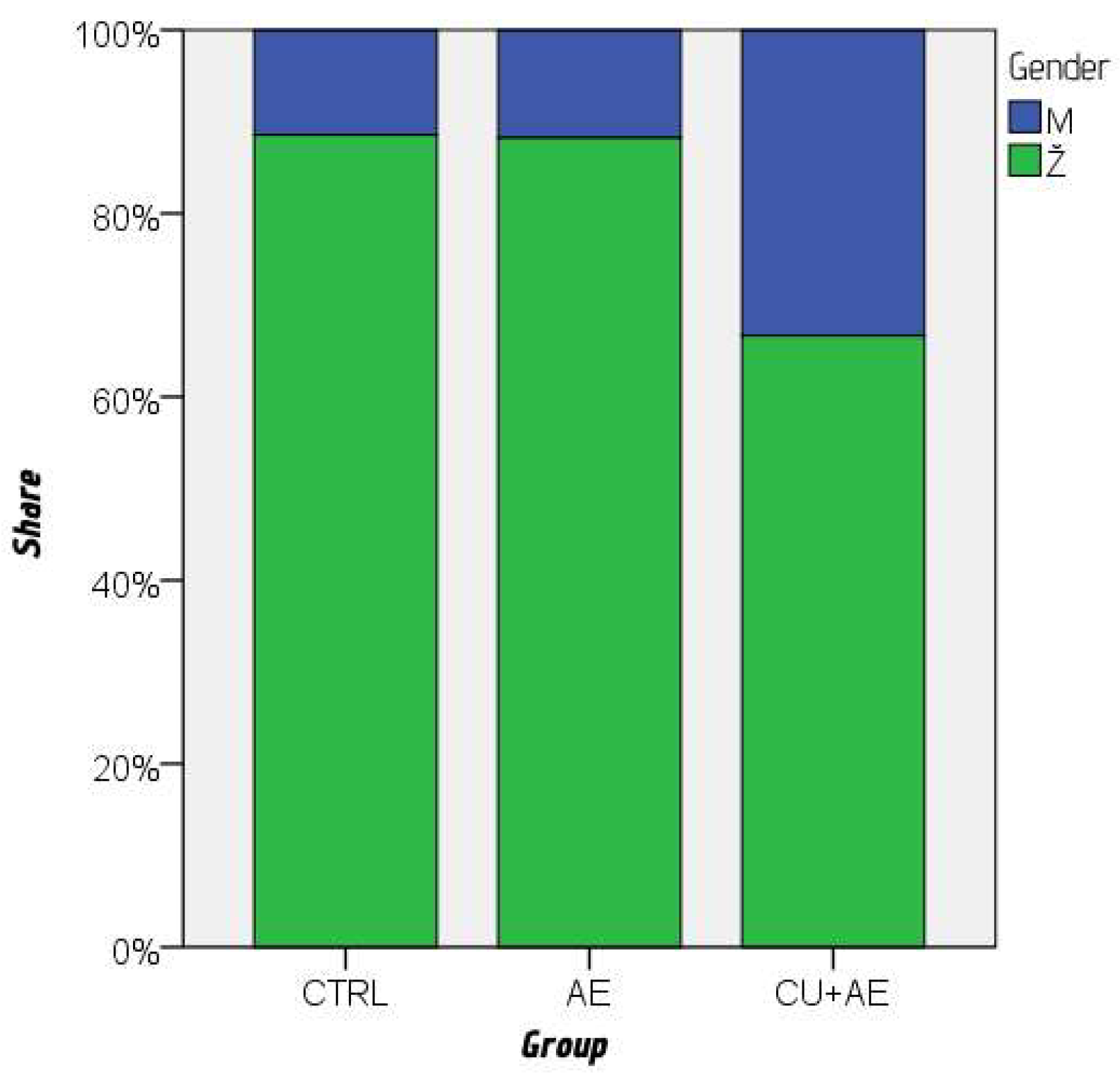
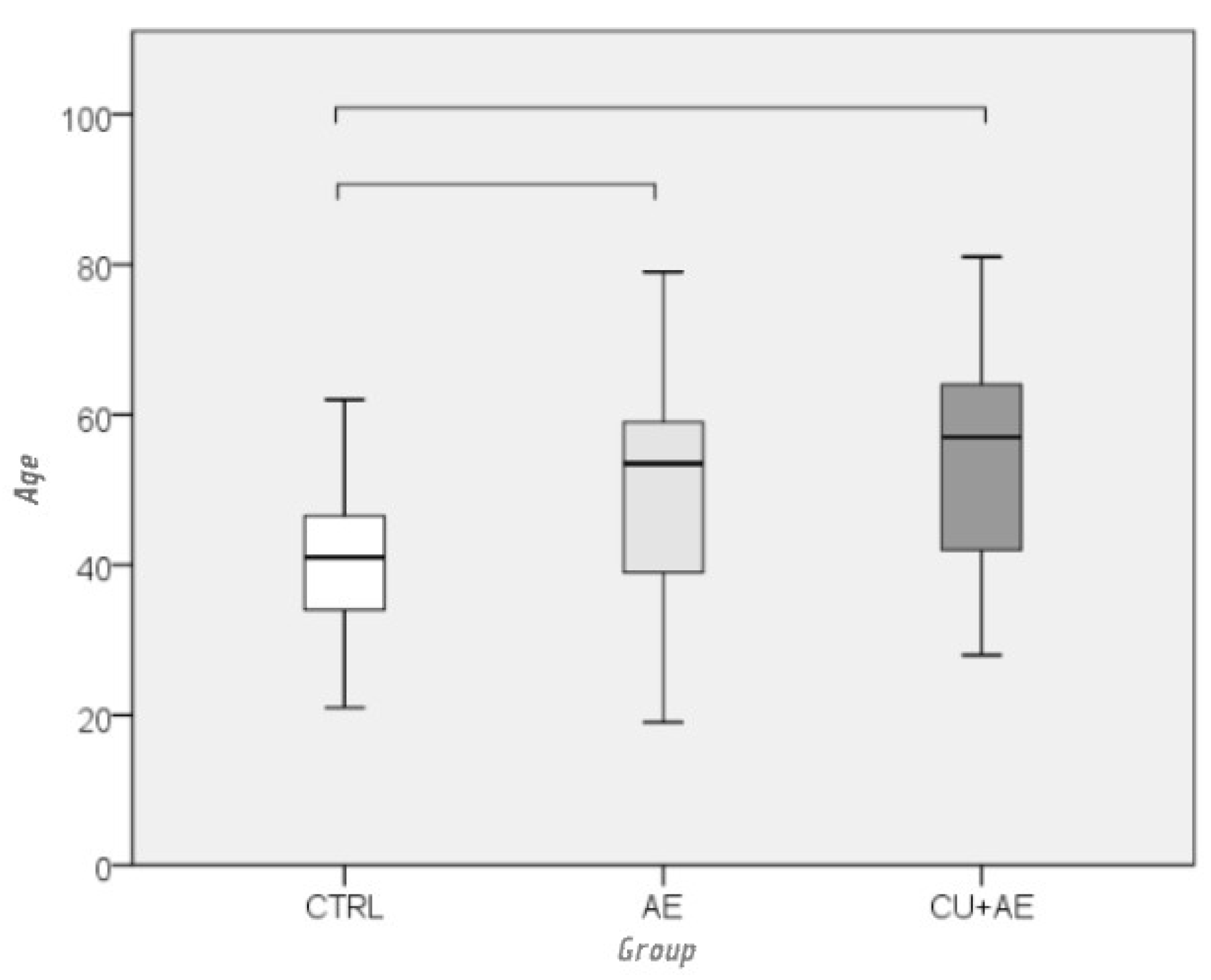
2.1. Patient Population
2.2. Assessment of AE severity - Angioedema Control Test (AECT)
2.3. Assessment of serum/ blood cells
2.4. Assessment of Serum Coagulation Factors
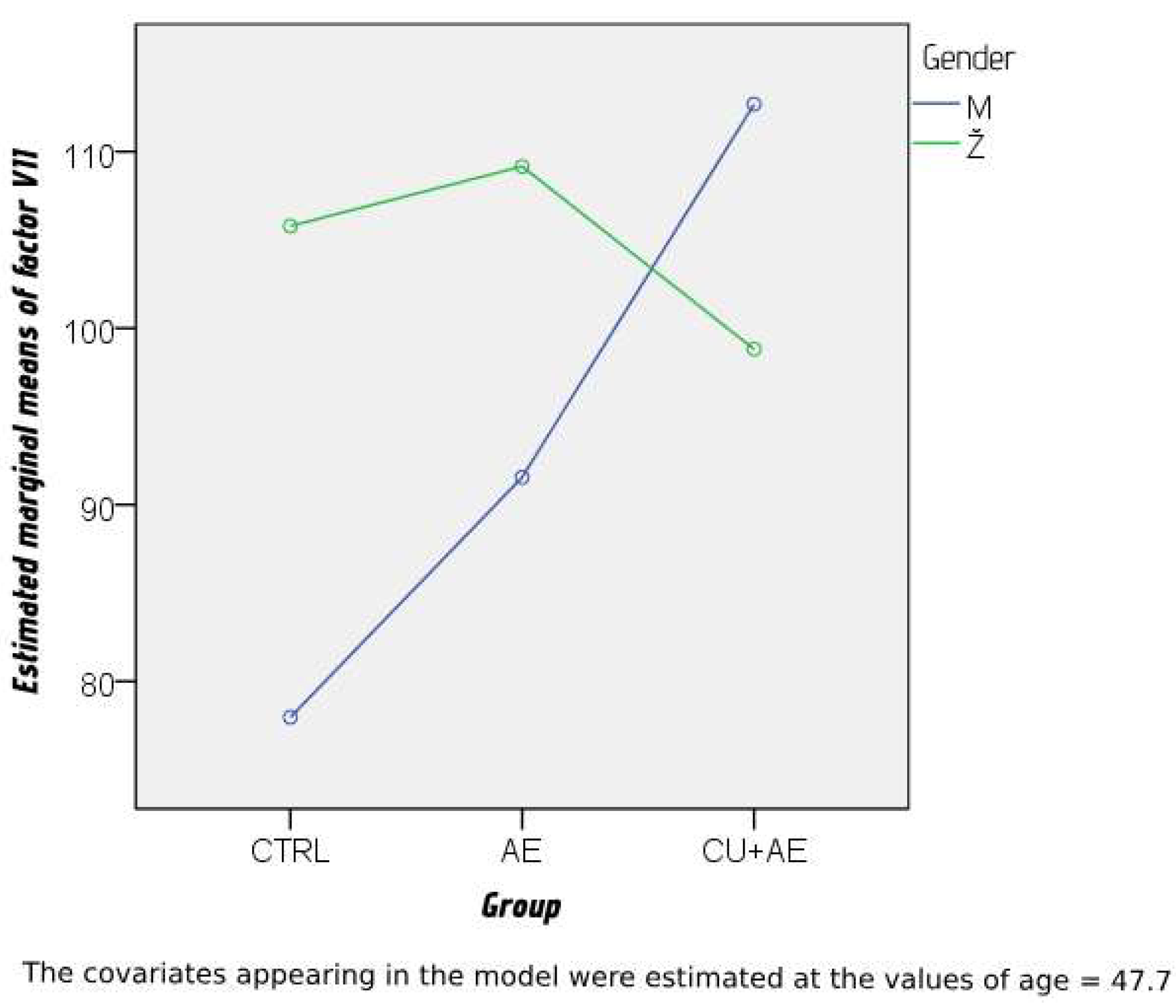
3. Results
3.1. Analysis of the Tested Sample
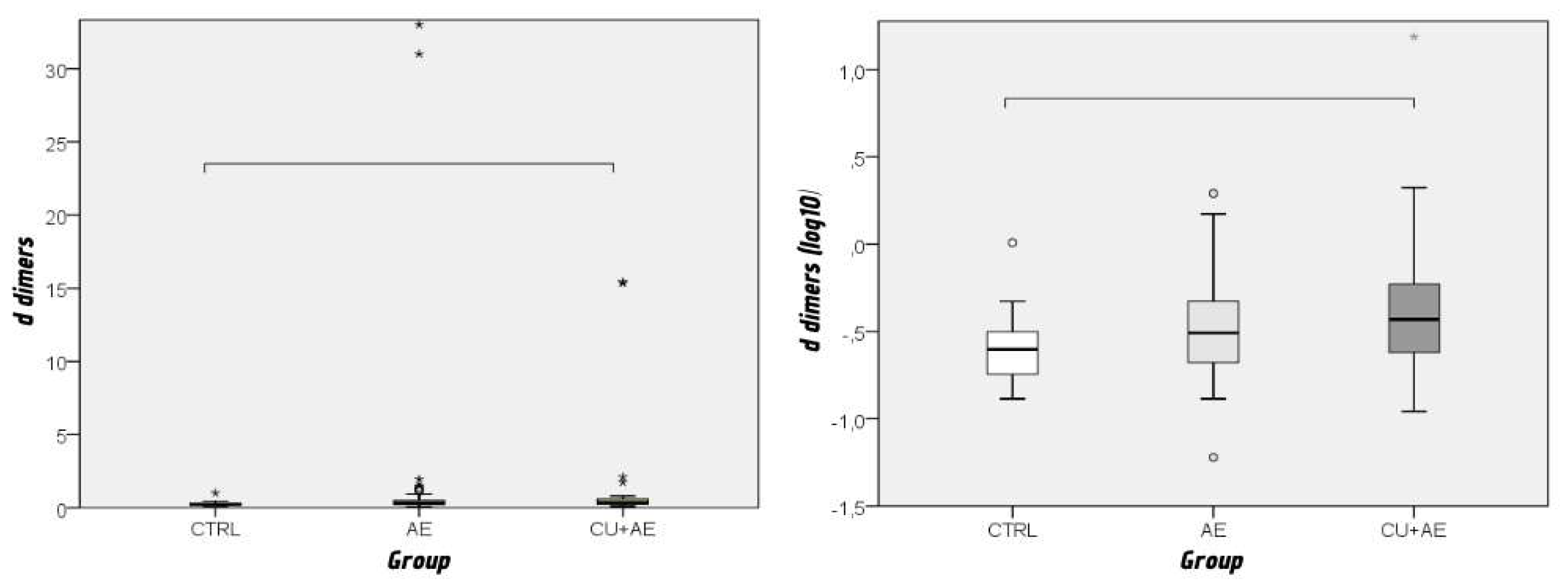
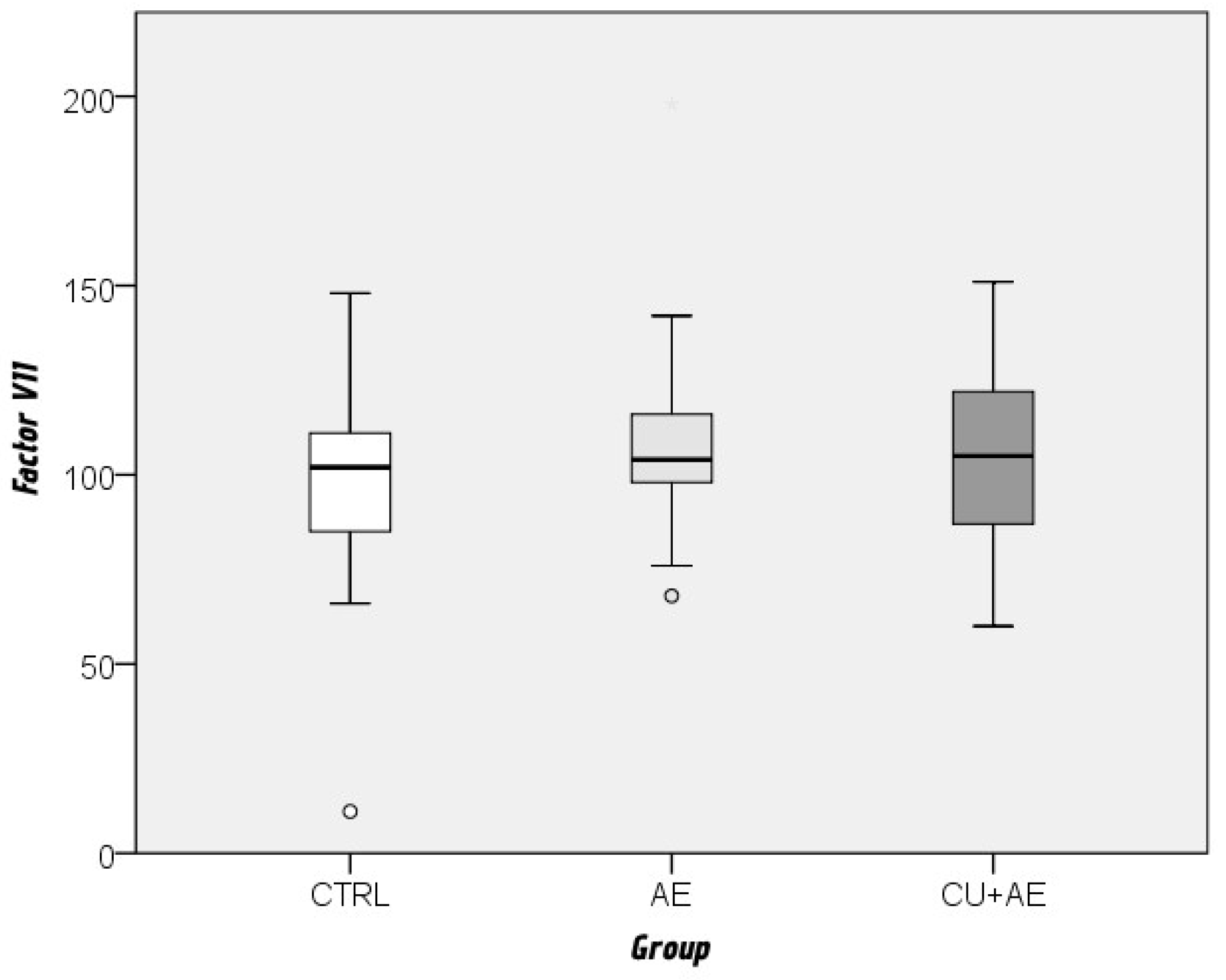
3.2. Results of Coagulation Factors Analysis
3.3. Results of Analysis of Other Tested Serum Factors
3.4. Results of the extended statistical analysis of the factors surveyed
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yanase, Y; Takahagi, S; Ozawa, K; Hide, M. The role of coagulation and complement factors for mast cell activation in the pathogenesis of chronic spontaneous urticaria. Cells 2021, 10(7), 3. [Google Scholar] [CrossRef]
- Zhou, B; Li, J; Liu, R; Zhu, L; Peng, C. The role of crosstalk of immune cells in pathogenesis of chronic spontaneous urticaria. Front Immunol 2022, 31(13), 879754. [Google Scholar] [CrossRef]
- Lugović-Mihić, L; Bukvić, I; Bulat, V; Japundžić, I. Factors contributing to chronic urticaria/angioedema and nummular eczema resolution–which findings are crucial? Acta Clin Croat. 2019, 58, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Porebski, G; Kwitniewski, M; Reshef, A. Biomarkers in hereditary angioedema. Clin Rev Allergy Immunol. 2021, 60(3), 404–415. [Google Scholar] [CrossRef]
- Baek, YS; Jeon, J; Kim, JH; Oh, CH. Severity of acute and chronic urticaria correlates with D-dimer level, but not C-reactive protein or total IgE. Clin Exp Dermatol 2014, 39, 795–800. [Google Scholar] [CrossRef]
- Kolkhir, P; André, F; Church, MK; Maurer, M; Metz, M. Potential blood biomarkers in chronic spontaneous urticaria. Clin. Exp. Allergy 2017, 1, 19–36. [Google Scholar] [CrossRef]
- Yanase, Y; Takahagi, S; Hide, M. Chronic spontaneous urticaria and the extrinsic coagulation system. Allergol Int. 2019, 67(2), 191–4. [Google Scholar] [CrossRef] [PubMed]
- Yanase, Y; Takahagi, S; Ozawa, K; Hide, M. The role of coagulation and complement factors for mast cell activation in the pathogenesis of chronic spontaneous urticaria. Cells 2021, 10(7), 3. [Google Scholar] [CrossRef] [PubMed]
- Wang, D; Tang, H; Shen, Y; Wang, F; Lin, J; Xu, J. Activation of the blood coagulation system in patients with chronic spontaneous urticaria. Clin. Lab. 2015, 61(9), 1283–1288. [Google Scholar] [CrossRef]
- D-Dimer, Asero R. A biomarker for antihistamine-resistant chronic urticaria. J. Allergy Clin. Immunol. 2013, 132(4), 983–986. [Google Scholar] [CrossRef]
- Confino-Cohen, R; Chodick, G; Shalev, V; Leshno, M; Kimhi, O; Goldberg, A. A chronic urticaria and autoimmunity: associations found in a large population study. J. Allergy Clin. Immunol. 2012, 129(5), 1307–1313. [Google Scholar] [CrossRef]
- Guilarte, M; Sala-Cunill, A; Luengo, O; Labrador-Horrillo, M; Cardona, V. The Mast Cell, Contact, and Coagulation System Connection in Anaphylaxis. Front Immunol. 2017, 8, 846. [Google Scholar] [CrossRef]
- Zhu, H; Liang, B; Li, R; Li, J; Lin, L; Ma, S; et al. Activation of coagulation, anti-coagulation, fibrinolysis and the complement system in patients with urticaria. Asian Pac. J. Allergy Immunol. 2013, 31(1), 43–50. [Google Scholar]
- Štrajtenberger, M; Lugović-Mihić, L; Stipić-Marković, A; Artuković, M; Mihić, R; Dolački, L; et al. Analysis of coagulation factors in angioedema/urticaria: increased values of D-dimer and fibrinogen in isolated angioedema. Acta Dermatovenerol Alp Pannonica Adriat. 2024, 33(2), 63–68. [Google Scholar] [CrossRef]
- Levi, M; van der Poll, T. Two-way interactions between inflammation and coagulation. Trends Cardiovasc. Med. 2005, 15(7), 254–259. [Google Scholar] [CrossRef]
- Vaswani, S. K.; Plack, R. H.; Norman, P.S. Acute severe urticaria and angioedema leading to myocardial infarction. Ann Allergy Asthma Immunol. 1996, 77(2), 101–104. [Google Scholar] [CrossRef]
- Pontillo, D; Capezzuto, A; Castellani, R. Acute generalized urticaria leading to acute myocardial infarction. Angiology 2000, 51(1), 89–90. [Google Scholar] [CrossRef]
- Yaegashi, T; Nakamura, Y; Sakagami, S; Saeki, T; Omi, W; Ikeda, K. Acute myocardial infarction following food-dependent exercise-induced anaphylaxis. Intern. Med. 2011, 50(5), 451–454. [Google Scholar] [CrossRef] [PubMed]
- Connor, S; Child, N; Burdon-Jones, D; Connor, A. Cardiac arrest secondary to type 2 Kounis syndrome resulting from urticaria and angioedema. Emerg. Med. J. 2010, 27(7), 568–569. [Google Scholar] [CrossRef] [PubMed]
- Zuberbier, T; Abdul Latiff, AH; Abuzakouk, M; Aquilina, S; Asero, R; Baker, D; et al. The international EAACI/GA²LEN/EuroGuiDerm/APAAACI guideline for the definition, classification, diagnosis, and management of urticaria. Allergy 2022, 77(3), 734–766. [Google Scholar] [CrossRef] [PubMed]
- Maurer, M; Aygören-Pürsün, E; Banerji, A; Bernstein, JA; Balle Boysen, H; Busse, PJ; et al. Consensus on treatment goals in hereditary angioedema: A global Delphi initiative. J Allergy Clin Immunol. 2021, 148(6), 1526–1532. [Google Scholar] [CrossRef] [PubMed]
- Prior, N; Remor, E; Gómez-Traseira, C; López-Serrano, C; Cabañas, R; Contreras, J; et al. Development of a disease-specific quality of life questionnaire for adult patients with hereditary angioedema due to C1 inhibitor deficiency (HAe-QoL): Spanish multi-centre research project. Health Qual Life Outcomes 2012, 10, 82. [Google Scholar] [CrossRef]
- Weller, K; Groffik, A; Magerl, M; Tohme, N; Martus, P; Krause, K; et al. Development and construct validation of the angioedema quality of life questionnaire. Allergy 2012, 67, 1289–1298. [Google Scholar] [CrossRef]
- Weller, K; Magerl, M; Peveling-Oberhag, A; Martus, P; Staubach, P; Maurer, M. The Angioedema Quality Questionnaire (AE-QoL) – assessment of sensitivity to change and minimal clinically important difference. Allergy 2016, 71, 1203–1209. [Google Scholar] [CrossRef]
- Weller, K; Donoso, T; Magerl, M; Aygören-Pürsün, E; Staubach, P; Martinez-Saguer, I; et al. Validation of the angioedema control test (AECT)- a patient-reported outcome instrument for assessing angioedema control. J Allergy Clin Immunol Pract. 2020, 8(6), 2050–2057. [Google Scholar] [CrossRef] [PubMed]
- Weller, K; Groffik, A; Church, MK; Hawro, T; Krause, K; Metz, M; et al. Development and validation of the urticaria control test: a patient-reported outcome instrument for assessing urticaria control. J Allergy Clin Immunol. 2014, 133, 1365-1372.e1-e6. [Google Scholar] [CrossRef]
- Nathan, RA; Sorkness, CA; Kosinski, M; Schatz, M; Li, JT; Marcus, P; et al. Development of the asthma control test: a survey for assessing asthma control. J Allergy Clin Immunol. 2004, 113, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Kocatürk, E; Kızıltaç, U; Can, P; Öztaş Kara, R; Erdem, T; Kızıltaç, K; et al. Validation of the Turkish version of the urticaria control test: correlation with other tools and comparison between spontaneous and inducible chronic urticaria. World Allergy Organ J 2019, 12, 100009. [Google Scholar] [CrossRef]
- Obtułowicz, A; Migacz-Gruszka, K; Pirowska, M; Basta-Klonowska, K; Wojas-Pelc, A. Participation of the coagulation system and fibrinolysis as well as selected biomarkers in pathogenesis of chronic urticaria with various activity degree. Postepy Dermatol. Alergol. 2020, 37(4), 608–612. [Google Scholar] [CrossRef]
- Farres, MN; Refaat, M; Melek, NA; Ahmed, EE; Shamseldine, MG; Arafa, NA. Activation of coagulation in chronic urticaria in relation to disease severity and activity. Allergol Immunopathol (Madr) 2015, 43(2), 162–7. [Google Scholar] [CrossRef]
- Baek, YS; Jeon, J; Kim, JH; Oh, CH. Severity of acute and chronic urticaria correlates with D-dimer level, but not C-reactive protein or total IgE. Clin Exp Dermatol 2014, 39(7), 795–800. [Google Scholar] [CrossRef] [PubMed]
- Triwongwaranat, D; Kulthanan, K; Chularojanamontri, L. Correlation between plasma D-dimer levels and the severity of patients with chronic urticaria. Asia Pac Allergy 2013, 3(2), 100–5. [Google Scholar] [CrossRef]
- Criado, PR; Antinori, LC; Maruta, CW; Reis, VM. Evaluation of D-dimer serum levels among patients with chronic urticaria, psoriasis and urticarial vasculitis. An Bras Dermatol 2013, 88(3), 355–60. [Google Scholar] [CrossRef]
- Zhu, H; Liang, B; Li, R. Activation of coagulation, anti-coagulation, fibrinolysis and the complement system in patients with urticaria. Asian Pac J Allergy Immunol. 2013, 31(1), 43–50. [Google Scholar]
- Takeda, T; Sakurai, Y; Takahagi, S; Kato, J; Yoshida, K; Yoshioka, A; et al. Increase of coagulation potential in chronic spontaneous urticaria. Allergy 2011, 66, 428–433. [Google Scholar] [CrossRef]
- Takahagi, S; Mihara, S; Iwamoto, K; Morioke, S; Okabe, T; Kameyoshi, Y; et al. Coagulation/fibrinolysis and inflammation markers are associated with disease activity in patients with chronic urticaria. Allergy 2010, 65(5), 649–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, F; Tang, H; Xu, JH. Activation of the blood coagulation cascade is involved in patients with chronic urticaria. J Allergy Clin Immunol. 2009, 123(4), 972–3. [Google Scholar] [CrossRef]
- Asero, R; Tedeschi, A; Riboldi, P; Griffini, S; Bonanni, E; Cugno, M. Severe chronic urticaria is associated with elevated plasma levels of D-dimer. Allergy 2008, 63(2), 176–80. [Google Scholar] [CrossRef]
- Asero, R; Tedeschi, A; Coppola, R; Griffini, S; Paparella, P; Riboldi, P; et al. Activation of the tissue factor pathway of blood coagulation in patients with chronic urticaria. J Allergy Clin Immunol. 2007, 119(3), 705–10. [Google Scholar] [CrossRef]
- Asero, R; Cugno, M; Tedeschi, A. Activation of blood coagulation in plasma from chronic urticaria patients with negative autologous plasma skin test. J Eur Acad Dermatol Venereol. 2011, 25(2), 201–5. [Google Scholar] [CrossRef] [PubMed]
- Baskurt, D; Sarac, E; Asero, R; Kocatürk, E. D-dimer levels decline after immunosuppressive treatment rather than anticoagulant treatment in severe autoimmune chronic spontaneous urticaria. Eur Ann Allergy Clin Immunol. 2024, 56(1), 42–44. [Google Scholar] [CrossRef]
- Takahashi, T; Minami, S; Teramura, K. Four cases of acute infectious urticaria showing significant elevation of plasma D-dimer level. J Dermatol 2018, 45(8), 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Asero, R; Marzano, AV; Ferrucci, S; Cugno, M. D-dimer plasma levels parallel the clinical response to omalizumab in patients with severe chronic spontaneous urticaria. Int Arch Allergy Immunol. 2017, 172(1), 40–44. [Google Scholar] [CrossRef]
- Reshef, A; Zanichelli, A; Longhurst, H; Relan, A; Hack, CE. Elevated D-dimers in attacks of hereditary angioedema are not associated with increased thrombotic risk. Allergy 2015, 70(5), 506–13. [Google Scholar] [CrossRef]
- Wang, D; Tang, H; Shen, Y; Wang, F; Lin, J; Xu, J. Activation of the blood coagulation system in patients with chronic spontaneous urticaria. Clin Lab. 2015, 61(9), 1283–8. [Google Scholar] [CrossRef]
- Lu, T; Jiao, X; Si, M; He, P; Zou, J; Zhang, S; et al. The Correlation of serums CCL11, CCL17, CCL26, and CCL27 and disease severity in patients with urticaria. Dis Markers 2016, 2016, 1381760. [Google Scholar] [CrossRef]
- Di Pino, M; Ruberto, MF; Costanzo, G; Firinu, D; Piras, MS; Mura, MN; et al. Chronic spontaneous urticaria: a low-grade disseminated intravascular coagulation only partially reversed by Omalizumab. Clin Exp Med. 2023, 2(2), 495–502. [Google Scholar] [CrossRef] [PubMed]
- Favaloro, EJ; Franchini, M; Lippi, G. Aging hemostasis: changes to laboratory markers of hemostasis as we age - a narrative review. Semin Thromb Hemost. 2014, 40(6), 621–33. [Google Scholar] [CrossRef] [PubMed]
- Donkin, R; Fung, YL; Singh, I. Fibrinogen, coagulation, and ageing. Subcell Biochem. 2023, 102, 313–342. [Google Scholar] [CrossRef]
- Amin, H; Mohsin, S; Aslam, M; Hussain, S; Saeed, T; Ullah, MI; et al. Coagulation factors and antithrombin levels in young and elderly subjects in Pakistani population. Blood Coagul Fibrinolysis 2012, 23(8), 745–50. [Google Scholar] [CrossRef]
- Akrivou, D; Perlepe, G; Kirgou, P; Gourgoulianis, KI; Malli, F. Pathophysiological aspects of aging in venous thromboembolism: An Update. Medicina (Kaunas) 2022, 58(8), 1078. [Google Scholar] [CrossRef]
- Metin, Z; Akca, HM; Tur, K; Akogul, S. Intersecting pathways: evaluating inflammatory markers and metabolism in chronic spontaneous urticaria with a multi-marker approach. Int J Dermatol 2024, 63(5), 604–610. [Google Scholar] [CrossRef]
- Zhang, L; Jiang, W; Gebreab, YB; Ye, X. Increased D-dimer and fibrin degradation product levels as potential indicators for evaluating infection-related acute urticaria: a case-case-control study. Arch Dermatol Res. 2023, 315(10), 2871–2876. [Google Scholar] [CrossRef]
- Criado, RF; Bensi, CG; Criado, PR; Henriques, MT; de Espindola, BAR; Machado Filho, CD. Evaluation of serum levels of C-reactive protein, D-dimer and autologous serum skin test in patients with chronic spontaneous urticaria in a Brazilian tertiary center: a cross-sectional study. An Bras Dermatol 2021, 96(2), 148–154. [Google Scholar] [CrossRef] [PubMed]
- Kasperska-Zajac, A; Sztylc, J; Machura, E; Jop, G. Plasma IL-6 concentration correlates with clinical disease activity and serum C-reactive protein concentration in chronic urticaria patients. Clin Exp Allergy 2011, 41(10), 1386–91. [Google Scholar] [CrossRef]
- de Montjoye, L; Darrigade, AS; Giménez-Arnau, A; Herman, A; Dumoutier, L; Baeck, M. Correlations between disease activity, autoimmunity and biological parameters in patients with chronic spontaneous urticaria. Eur Ann Allergy Clin Immunol. 2021, 53(2), 55–66. [Google Scholar] [CrossRef] [PubMed]
- Sabaté-Brescó, M; Rodriguez-Garijo, N; Azofra, J; Baeza, ML; Donado, CD; Gaig, P; et al. A comparative study of sex distribution, autoimmunity, blood, and inflammatory parameters in chronic spontaneous urticaria with angioedema and chronic histaminergic angioedema. J Allergy Clin Immunol Pract. 2021, 9(6), 2284–2292. [Google Scholar] [CrossRef] [PubMed]
- Tamer, F. Omalizumab does not lead to a distinct alteration in hematological parameters and complete blood count-derived inflammation biomarkers except for basophil count. Cutan Ocul Toxicol. 2020, 39(3), 229–232. [Google Scholar] [CrossRef]
- Puzianowska-Kuźnicka, M; Owczarz, M; Wieczorowska-Tobis, K; Nadrowski, P; Chudek, J; Slusarczyk, P; et al. Interleukin-6 and C-reactive protein, successful aging, and mortality: The PolSenior study. Immun Ageing 2016, 13, 21. [Google Scholar] [CrossRef]
- McCabe, EL; Larson, MG; Lunetta, KL; Newman, AB; Cheng, S; Murabito, JM. Association of an index of healthy aging with incident cardiovascular disease and mortality in a community-based sample of older adults. J Gerontol A Biol Sci Med Sci. 2016, 71, 1695–1701. [Google Scholar] [CrossRef]
- Paik, JK; Chae, JS; Kang, R; Kwon, N; Lee, SH; Lee, JH. Effect of age on atherogenicity of LDL and inflammatory markers in healthy women. Nutr Metab Cardiovasc Dis. 2013, 23, 967–972. [Google Scholar] [CrossRef]
- Maurer, M; Fluhr, JW; Khan, DA. How to approach chronic inducible urticaria. J Allergy Clin Immunol Pract. 2018, 6(4), 1119–1130. [Google Scholar] [CrossRef]
- Larenas-Linnemann, D. Biomarkers of autoimmune chronic spontaneous urticaria. Curr Allergy Asthma Rep. 2023, 23(12), 655–664. [Google Scholar] [CrossRef]
- Saini, SS. Urticaria and basophils. Allergol Int. 2023, 72(3), 369–374. [Google Scholar] [CrossRef]
- Vena, GA; Cassano, N; Marzano, AV; Asero, R. The role of platelets in chronic urticaria. Int Arch Allergy Immunol. 2016, 169(2), 71–9. [Google Scholar] [CrossRef]
- Berth-Jones, J; Hutchinson, PE; Wicks, AC; Mitchell, VE. Chronic urticaria with angio-oedema controlled by warfarin. BMJ 1988, 297(6660), 1382–1383. [Google Scholar] [CrossRef]
- Barlow, R.J; Greaves, M.W. Warfarin in the treatment of chronic urticaria/angio-edema. Br. J. Dermatol. 1992, 126(4), 415–416. [Google Scholar] [CrossRef] [PubMed]
- Chua, S.L.; Gibbs, S. Chronic urticaria responding to subcutaneous heparin sodium. Br. J. Dermatol. 2005, 153(1), 216–217. [Google Scholar] [CrossRef] [PubMed]
- Takahagi, S; Shindo, H; Watanabe, M; Kameyoshi, Y; Hide, M. Refractory chronic urticaria treated effectively with the protease inhibitors, nafamostat mesilate and camostat mesilate. Acta Derm Venereol. 2010, 90(4), 425–426. [Google Scholar] [CrossRef] [PubMed]
- Meyer-De Schmid, J. J.; Neuman, A. Treatment of chronic urticaria with heparin. Bull. Soc. Fr. Dermatol. Syphiligr. 1952, 59(3), 286–287. [Google Scholar]
- Chua, S.L.; Gibbs, S. Chronic urticaria responding to subcutaneous heparin sodium. Br. J. Dermatol. 2005, 153(1), 216–217. [Google Scholar] [CrossRef] [PubMed]
- Parslew, R; Pryce, D; Ashworth, J; Friedmann, PS. Treatment of chronic idiopathic urticaria and angio-oedema. Clin. Exp. Allergy 2000, 30(8), 1161–1165. [Google Scholar] [CrossRef]
- Mahesh, PA; Pudupakkam, VK; Holla, AD; Dande, T. Effect of warfarin on chronic idiopathic urticaria. Indian J. Dermatol. Venereol. Leprol. 2009, 75(2), 187–189. [Google Scholar] [CrossRef] [PubMed]
- Asero, R; Tedeschi, A; Cugno, M. Heparin and tranexamic acid therapy may be effective in treatment-resistant chronic urticaria with elevated D-dimer. A Pilot Study. Int. Arch. Allergy Immunol. 2010, 152(4), 384–389. [Google Scholar] [CrossRef] [PubMed]
| Variable | Average | Std. deviation | Minimum | Maximum | Median | IQR |
| Age | 47,7 | 14,3 | 19 | 81 | 49 | 36 – 58 |
| Factor VII | 104,4 | 24,3 | 11 | 198 | 104 | 88,8 – 118,3 |
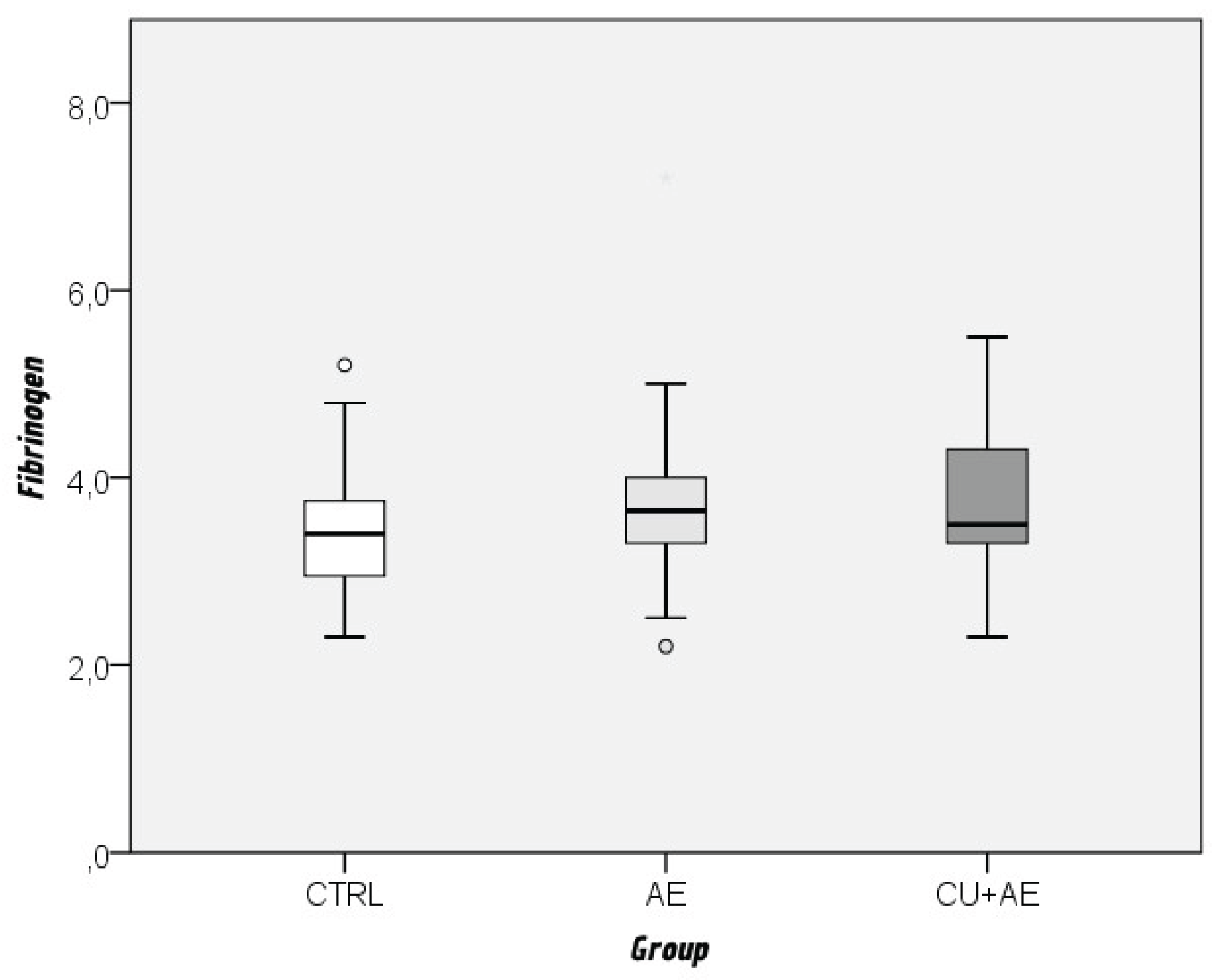
| Fibrinogen | 3,6 | 0,8 | 2,2 | 7,2 | 3,5 | 3,2 – 4,0 |
| D-dimers | 1,31 | 4,85 | 0,06 | 33,00 | 0,29 | 0,3 – 0,44 |
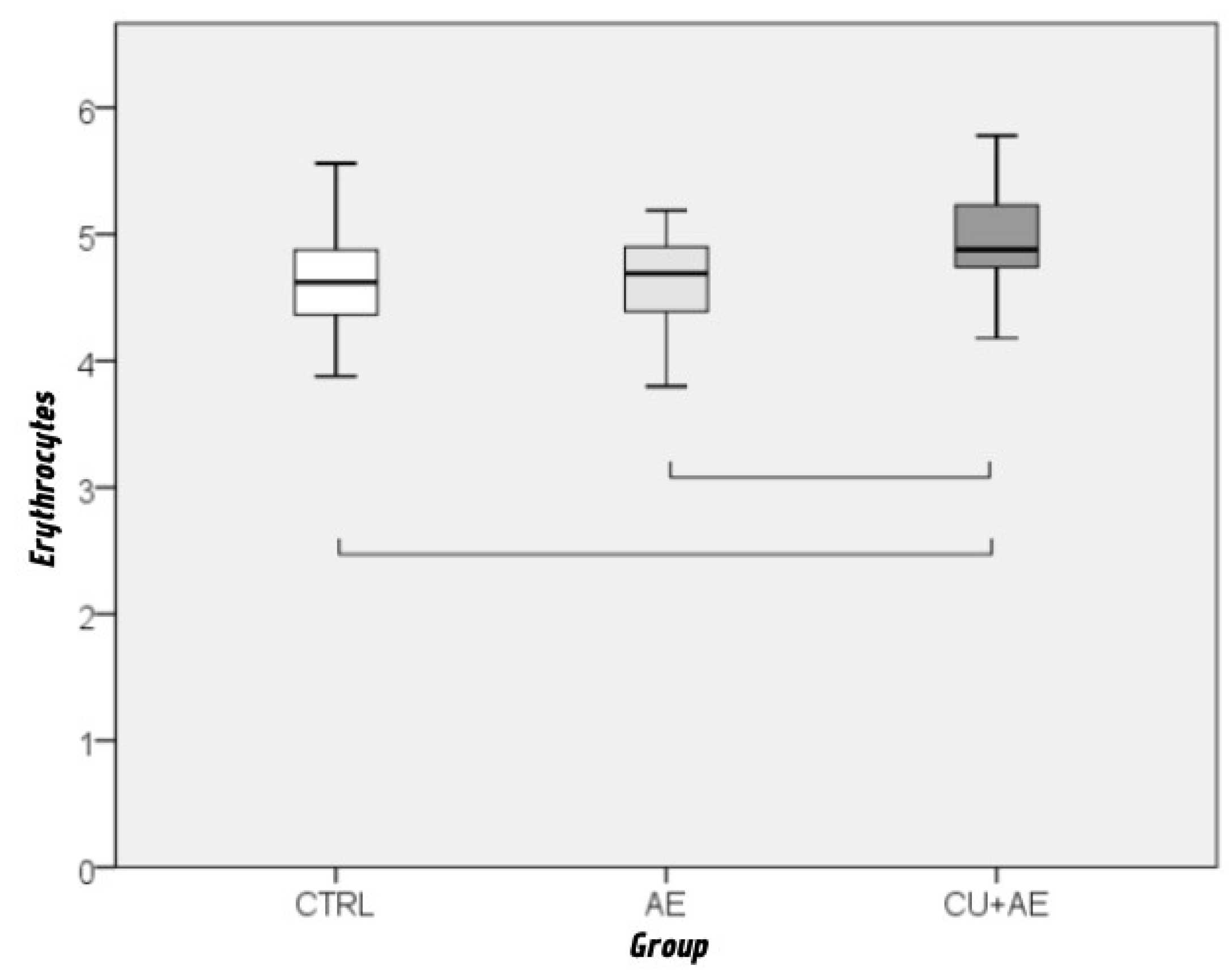
| Erythrocytes | 4,8 | 0,4 | 3,8 | 5,8 | 4,8 | 4,5 – 5,0 |
| Platelets | 255,5 | 57,5 | 145 | 492 | 258,5 | 220,0 – 2 84,3 |
| Neutrophils | 4,1 | 2,2 | 1,8 | 18,6 | 3,6 | 2,9 – 4,7 |
| Basophils | 0,04 | 0,05 | 0,00 | 0,39 | 0,03 | 0,02 – 0,04 |
| Leucocytes | 6,5 | 2,5 | 3,2 | 22,3 | 6,2 | 4,9 – 7,1 |
| ESR | 10,2 | 8,0 | 2 | 40 | 7,0 | 4,8 – 13,3 |
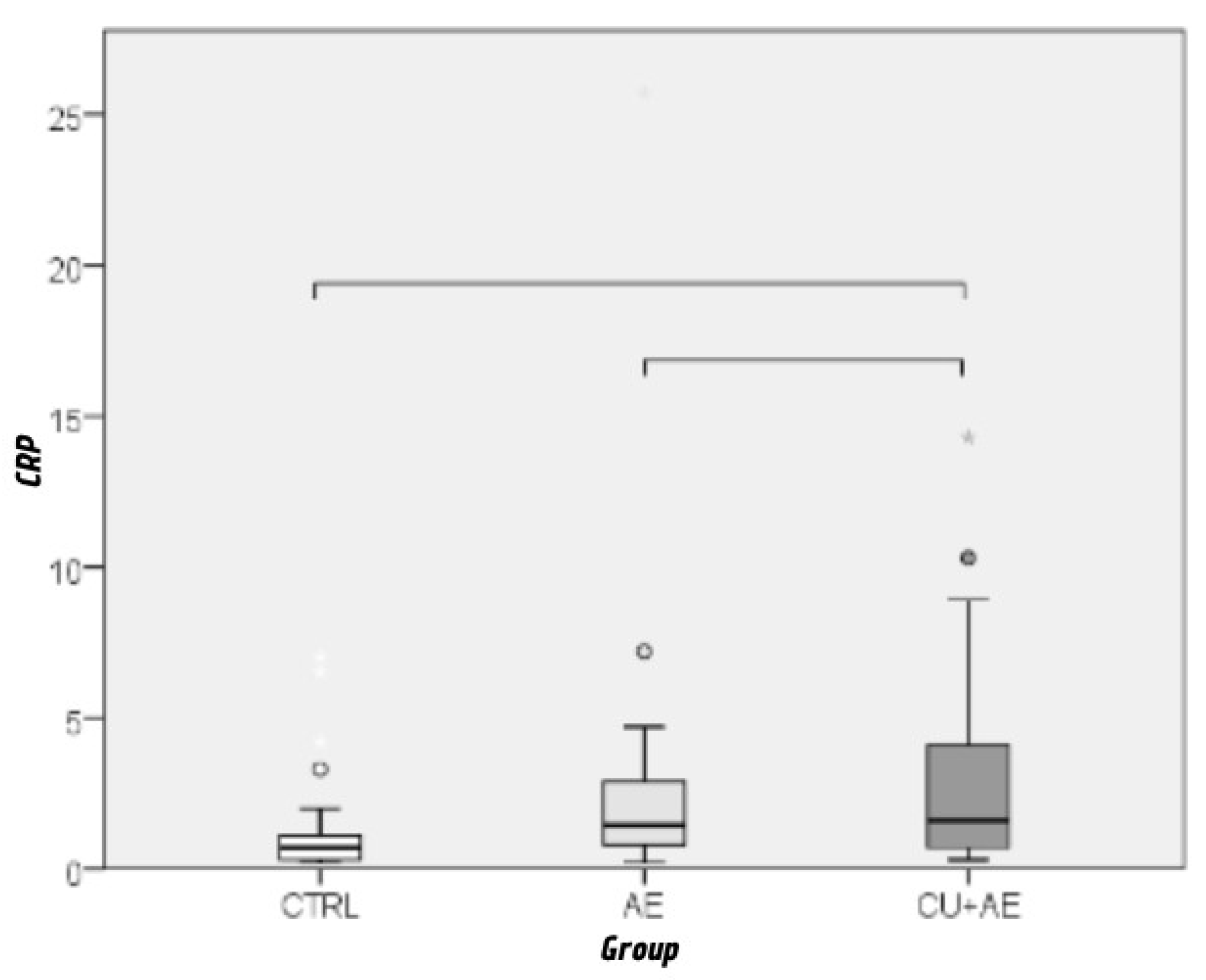
| CRP | 2,4 | 3,7 | 0,2 | 25,7 | 1,1 | 0,6 – 2,7 |
| AECT score | 6,4 | 4,5 | 0 | 16 | 5 | 3 – 8 |
| CTRL (N = 35) |
CU + AE (N = 34) |
AE (N = 33) |
Sum | P | V | ||
| Factor VII | In reference range or decreased | 10 (28,6 %) | 8 (24,2 %) | 3 (8,8 %) | 21 (20,6 %) | ||
| (N(%)) | Increased | 25 (71,4 %) | 25 (75,8 %) | 31 (91,2 %) | 81 (79,4 %) | 0,105 | 0,210 |
| Fibrinogen | In reference range | 32 (91,4 %) | 28 (84,8 %) | 31 (91,2 %) | 91 (89,2 %) | ||
| (N(%)) | elevated | 3 (8,6 %) | 5 (15,2%) | 3 (8,8 %) | 11 (10,8 %) | 0,616 | 0,097 |
| D-dimers | In reference range | 34 (97,1 %) | 24 (72,7 %) | 25 (73,5 %) | 83 (81,4 %) | ||
| (N(%)) | elevated | 1a (2,9 %) | 9b (27,3 %) | 9b (26,5 %) | 19 (18,6 %) | 0,013 | 0,293 |
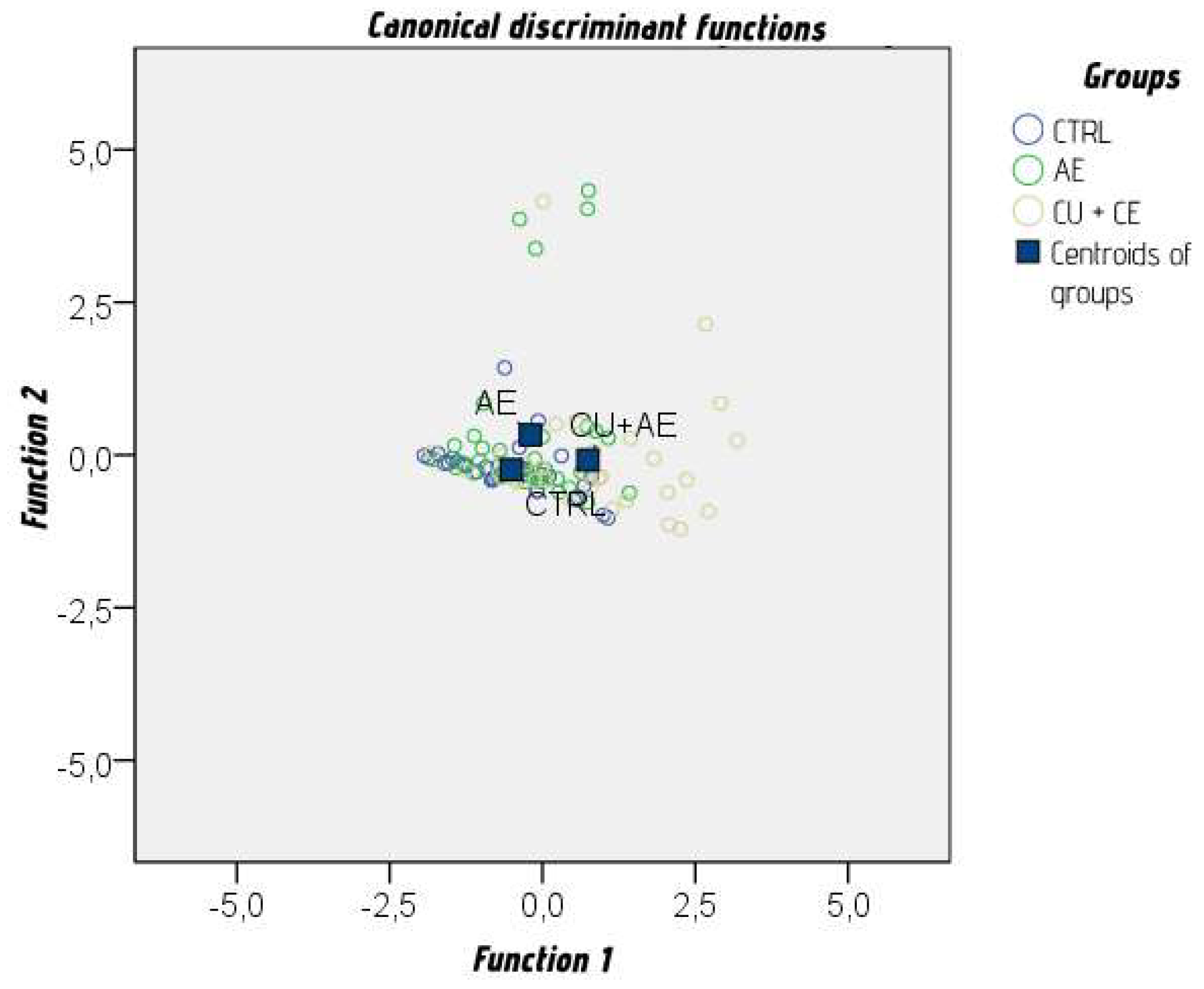
| Function | Unit value | % of variance | Cumulative % | Canonical correlation | Wilks' lambda | P |
| 1 | 0,385a | 83,8 | 83,8 | 0,527 | 0,672 | <0,001 |
| 2 | 0,075a | 16,2 | 100,0 | 0,263 | 0,931 | 0,137 |
| Variables | Functions | |
| 1 | 2 | |
| Age | 0,630* | 0,476 |
| Erythrocyte levels | 0,588* | –0,453 |
| D-dimer levels | 0,118 | 0,597* |
| CRP levels | 0,282 | 0,584* |
| Group | Predicted group classification/membership* | Total | ||||
| CTRL | AE | CU+AE | ||||
| Original group classification/membership | N | CTRL | 25 | 5 | 5 | 35 |
| AE | 12 | 12 | 10 | 34 | ||
| CU+AE | 10 | 5 | 18 | 33 | ||
| % | CTRL | 71,4 | 14,3 | 14,3 | 100,0 | |
| AE | 35,3 | 35,3 | 29,4 | 100,0 | ||
| CU+AE | 30,3 | 15,2 | 54,5 | 100,0 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





