1. Introduction
Cancer progression is supported by the crosstalk between tumor cells and the surrounding stroma. In this context, it is known that senescent stromal cells contribute to the development of a pro-inflammatory milieu and the acquisition of aggressive traits by cancer cells [
1]. In addition to classic senescence inducers, such as telomere shortening, oncogene activation, DNA damage, epigenetic changes, or oxidative stress [
2], it has been shown that anticancer treatments can also induce cellular senescence (known as therapy-induced senescence, TIS) in both tumor and non-cancerous cells, thus contributing to many detrimental side effects of therapies [
3]. In this context, the senescence-associated secretory phenotype (SASP), which comprises a broad spectrum of cytokines, chemokines, growth factors, proteases, and metabolites, profoundly influences the tumor microenvironment (TME) by modulating inflammation, remodeling the extracellular matrix, and altering the behavior of neighboring cells [
4]. While initially senescence acts as a tumor-suppressive mechanism by halting the proliferation of damaged cells, the SASP can paradoxically promote tumor progression at later stages of the disease. In particular, factors released by senescent fibroblasts promote cancer cell proliferation, angiogenesis, immune evasion, enhance phenotypic plasticity, by fostering traits such as epithelial-to-mesenchymal transition (EMT) and stemness, which are associated with metastasis and therapy resistance [
5,
6].
Previously, we investigated the effects of chemotherapy on the stromal compartment of prostate and ovarian cancers, and demonstrated that anticancer chemotherapeutics, regardless of their specific mechanism of action, induce a senescent phenotype in patient-derived stromal prostate and ovarian fibroblasts sustaining the invasive potential of tumor cells through the SASP [
7]. Given that senescent cells undergo intense metabolic reprogramming to support survival and increased secretion of SASP factors [
8], here we focused our attention on a possible involvement of SASP-associated metabolic components in regulating tumor progression, which to date has not been extensively investigated. Beyond soluble factors and cytokines, extracellular metabolites released by the tumor stroma are increasingly recognized as key regulators of cancer progression, promoting proliferation, immune escape, and metastases [
9,
10]. In this context, we found that the amino acid Glutamine (Gln) is enriched in the conditioned media (CM) of TIS senescent stromal cells. Given the established role of Gln in fueling cancer metabolism and supporting anabolic processes [
11], we hypothesized that stromal-derived Gln might contribute to the acquisition of aggressive traits in cancer cells. In accordance, we demonstrated that the utilization of senescent-stromal-derived Gln by ovarian and prostate cancer cells plays a pivotal role in supporting increased metastatic potential and stem-like traits. Collectively, our findings uncover a previously uncharacterized role of the metabolic component of the SASP, specifically Gln, in driving cancer cell invasion and stemness. Collectively, these results identify TIS-derived Gln as a critical factor in tumor progression and point to metabolic crosstalk between tumor and stroma as a potential therapeutic vulnerability.
2. Materials and Methods
Cell Lines
Human prostate (PC3) and ovarian (SKOV3) cancer cell lines were obtained from ATCC. Human Prostate Fibroblasts (HPFs) were obtained from tissue samples isolated during surgery from patients (average age 70) who underwent surgical treatment for lower urinary tract symptoms caused by benign prostatic hyperplasia (BPH). Human Ovarian Fibroblasts (HOFs) were extracted from healthy peritoneal tissue samples collected during cytoreductive surgery performed on patients (average age 66) with advanced ovarian cancer. Surgical explants were obtained in accordance with the Ethics Committee of the Azienda Ospedaliera Universitaria Careggi (Florence, Italy), for prostate tissues: num 16583_bio; for ovarian tissues: num 14780.
All cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) high glucose (4.5 g/L) (Euroclone), supplemented with 10% Fetal Bovine Serum (Euroclone), 2 mM L-Gln (Euroclone), 1% penicillin and streptomycin (Euroclone). Cells were routinely grown at 37°C in humidified atmosphere with 5% CO2.
To obtain HPFs and HOFs, a fragment of surgical explant was transferred into a Petri dish and cut into small pieces about 0.2 cm. Then, tissue fragments were transferred into a new cell culture dish and placed in a central stripe under the pressure of a sterilized slide. DMEM high glucose supplemented with 20% FBS, 2 mM L-Gln, 2% Penicillin/Streptomycin, 100 μg/mL Kanamycin (Merck Sigma) and 2.5 μg/mL Amphotericin B (Euroclone) was added. After 20 days fibroblasts that had formed a monolayer were detached by trypsinization and routinely cultured.
Cell Treatments and Preparation of CM
To induce senescence, HPFs and HOFs were exposed to either 5 nM Docetaxel (MedChemExpress HY-B0011) in dimethyl sulfoxide (DMSO) or 20 μM Cisplatin (Merck Sigma #5663-27-1) in DMSO, respectively for 24h, while DMSO was added in control samples. Then, culture medium was changed, and cells were maintained in complete medium for additional 6 days before experiments were conducted, unless noted otherwise. To collect CM, senescent and control cells obtained as previously described, were incubated for 24h in starvation medium. CM was collected, clarified by centrifugation for 10 min at 1000 rpm and used freshly or stored at -80°C until use. Prostate and ovarian cancer cells were then incubated for 72h with CM from HPFs or HOFs, respectively.
Senescence-Associated β-Galactosidase Staining
Fibroblasts were fixed with 3% paraformaldehyde in PBS for 5 min and washed three times in PBS. Then cells were incubated with Senescence-Associated β-Galactosidase (SA-β-Gal) staining solution (5 mM potassium ferrocyanide, 5 nM potassium ferricyanide, 150 mM NaCl, 2 mM MgCl2, 40 mM citric acid monohydrate, 1 mg/mL 5-bromo-4-chloro-3-indolyl β-D-galactopyranoside, (Merck Sigma #B4252) pH 6.0) for 12 to 18h at 37°C in a non-humidified incubator under atmospheric CO2 conditions. Photos were taken at five randomly chosen fields and cells positive to (SA-β-Gal) staining were detected by the presence of an insoluble blue intracellular precipitate. Total and positive cells were counted using ImageJ imaging system.
Western Blotting
Cells were lysed at 4°C with RIPA buffer (Thermo Fisher Scientific, #89900) and supplemented with protease and phosphatase cocktail inhibitors (Merck Sigma). After 20 min cellular extracts were centrifuged for 10 min at 14000 rpm and protein concentration were quantified using Bicinchoninic Acid (BCA) assay (Euroclone). Then, 20 to 30 μg of total proteins was loaded on SDS-PAGE gels (BioRad) and transferred to PVDF membranes (BioRad). Membranes were blocked for 1h at room temperature in 5% non-fat dry milk (Santa Cruz Biotechnology) in PBS-Tween 0.1% and then incubated overnight at 4°C with primary antibody against p21 (Santa Cruz Biotechnology, sc-271610), p16 (Santa Cruz Biotechnology, sc-56330), Gln synthetase (GS) (Cell Signaling, D203F), NRF2 (Santa Cruz Biotechnology, sc-365949), ETS1 (Cell Signaling, B808A), HSP90 (Santa Cruz Biotechnology, sc-69703), β-Actin (Santa Cruz Biotechnology, sc-47778), Vinculin (Merck Sigma V9264). Then, membranes were incubated for 1h at room temperature with anti-rabbit horseradish peroxidase-conjugated (Santa Cruz Biotechnology #2357) or anti-mouse horseradish peroxidase-conjugated (Santa Cruz Biotechnology #516102). Proteins were visualized using Clarity Western ECL Substrate (BioRad) and images acquired using Amersham Imager 600 (GE Healthcare). HSP90 or β-Actin was used to ensure equal loading. All western blot images are representative of at least three independent experiments.
Invasion Assay
Prostate cancer cells (8x104) and ovarian cancer cells (10x104) were seeded in 200 μL of starvation medium in the upper compartment of 8 μm pore Transwell (Greiner Bio-One) coated with 50 μg/cm2 Matrigel (Corning). In the lower compartment, complete medium containing 10% FBS was added as chemoattractant. After 16h, cells that have invaded toward the lower surface of the filters were fixed and stained with Crystal Violet (Merck Sigma). The values for invasion were expressed as the average number of invading cells per microscopic field over five randomly chosen fields.
Real-Time PCR
Total RNA was extracted from cells using RNeasy Plus Mini Kit (Qiagen #74134) according to the manufacturer’s instruction and quantified with NanoDrop Microvolume Spectrophotometer and Fluorometer (Thermo Fisher Scientific). cDNA synthesis was obtained by incubating 1 μg of total RNA with High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific) according to the manufacturer’s instructions. mRNA expression levels were quantified by Real-Time PCR using Luna Universal qPCR Master Mix (New England Biolabs).
The nucleotide sequences of the specific primers (Thermo Fisher Scientific) used were: EpCAM-FW 5’-TGTGGTGATAGCAGTTGTTGC-3’, EpCAM-REV 5’-CTATGCATCTCACCCATCTCC-3’; ECAD-FW 5’-AGGCCAAGCAGCAGTACATT-3’, ECAD-REV 5’-ATTCACATCCAGCACATCCA-3’; NCAD-FW 5’-CCTCCAGAGTTTACTGCCATGAC-3’, NCAD-REV 5’-GTAGGATCTCCGCCACTGATTC-3’; VIM-FW 5’-ACACCCTGCAATCTTTCAGACA-3’, VIM-REV 5’-GATTCCACTTTGCGTTCAAGGT-3’; ZEB1-FW 5’-AAGAAAGTGTTACAGATGCAGCTG-3’, ZEB1-REV 5’-CCCTGGTAACACTGTCTGGTC-3’; ZEB2-FW 5’-AGGGACAGA TCAGCACCAAA-3’; ZEB2-REV 5’-GTGCGAACTGTAGGAACCAG-3’; SNAIL-FW 5’-CCTCCCTGTCAGATGAGGAC-3’, SNAIL-REV 5’-CAAGGAATACCTCAGCCTGG-3’; SLUG-FW 5’-ACAGCGAACTGGACACACAT-3’, SLUG-REV 5’-GATGGGGCTGTATGCTCCT-3’; B2M-FW 5’-AGTATGCCTGCCGTGTGAAC-3’, B2M-REV 5’-GCGGCATCTTCAAACCTCCA-3’.
qRT-PCR was performed using CFX96 Real-Time PCR System (BioRad). Data were reported as relative quantity with respect to the reference samples using 2-ΔΔCt. Data were normalized on β2-microglobulin.
Total ROS Quantification
Intracellular reactive oxygen species (ROS) levels were assessed using the fluorescent probe 2′,7′–dichlorofluorescin diacetate (DCFDA, Merck Sigma, #287810). Cells were detached, pelleted by centrifugation, and resuspended in a staining solution containing 5 µM DCFDA. Samples were incubated for 30 min at 37°C protected from light. Following incubation, cells were washed to remove excess dye and resuspended in PBS for recovery. Flow cytometry analysis was performed using a BD FACS Canto II cytometer (BD Biosciences). The viable cell population was gated based on morphological properties using Forward Scatter (FSC) and Side Scatter (SSC). Fluorescence was detected on the FITC channel (488 nm excitation laser; 530/30 nm bandpass filter). Background fluorescence was established using unstained controls to define the threshold for positivity. Data were recorded and analyzed as both the percentage of positive cells and the Mean Fluorescence Intensity (MFI).
Cell Transfection
Control siRNA (SIC001) and GS siRNA (#HS02_00307974) were purchased from Merck Sigma. Senescent cells were transfected with RNAiMAX (Thermo Fisher Scientific) according to manufacturer’s instructions. GS expression was assessed by western blotting 48h after transfection.
Cell Viability
Cells (6x103) were seeded in 100 μL of complete culture medium in 96-well plates and treated 24h after the seeding. Then, cells were washed with PBS and 5 mmol/L MTT (3-(4,5-Dimethylthiazol 2-yl)-2,5-diphenyltetrazolium bromide, Merck Sigma) was added for 1h at 37°C. The resulting formazan crystals were dissolved in 200 µL of DMSO, and absorbance was measured at 595 nm using a spectrophotometer MULTISKAN FC (Thermo Scientific).
To test the effect of Gln deprivation, 15x104 cells were seeded in 35 mm dishes and incubated with or without 1% Gln (Euroclone) for 72h. Then cells were detached and incubated with LIVE/DEAD Violet Kit (Thermo Fisher Scientific, #L34964A) for 30 min at RT according to the manufacturer’s protocol. The dye was reconstituted in 50 µL of DMSO and diluted 1:500 in PBS to create a working solution. Following a single wash with PBS, samples were analyzed via flow cytometry using a BD FACSCanto II. The violet dye (excitation 405 nm; emission 450 nm) was detected using a BV421/DAPI filter, allowing for clear discrimination between unstained live cells and stained dead cells.
Determination of GSH/GSSG
The intracellular reduced (GSH) and oxidized (GSSG) glutathione levels were measured using the GSH/GSSG-Glo™ Assay (Promega, V6611), according to the manufacturer’s instructions. Briefly, cells were lysed and total glutathione (GSH + GSSG) was quantified through the conversion of a GSH probe, Luciferin-NT, to luciferin by a glutathione S-transferase enzyme. To selectively measure GSSG, GSH was first derivatized using the provided masking reagent, allowing specific detection of GSSG. Luminescence was measured using a Sinergy H1 plate reader (BioTek Winooski, Vermont, USA), and GSH concentrations were calculated from standard curves. Values were normalized to protein content.
Determination of Gln and Ammonium Levels
Gln and ammonium levels were determined using the L-Glutamine/Ammonia Assay Kit (Rapid) (Megazyme, K-GLNAM), according to the manufacturer’s instructions. Briefly, the Gln levels were measured in culture media through an enzymatic conversion of Gln to glutamate (Glu) and ammonium by glutaminase (GLS). Ammonium was subsequently quantified through a glutamate dehydrogenase–coupled reaction by monitoring NADPH consumption as a decrease in absorbance at 340 nm with an uv-1800 shimadzu spectrophotometer.
Gas Chromatography–Mass Spectrometry (GC-MS) Analysis
For total metabolites quantification, media from HPFs and HOFs were collected and 50 μL were mixed with 50 μL of cold 80% methanol in HPLC water containing 1 µg/ml norvaline and 1.25 µg/ml glutaric acid as internal standards.
For tracing experiments, fibroblasts were cultured in MEM (Gibco) supplemented with 1% MEM vitamins (Merck Sigma), 1% penicillin-streptomycin (Euroclone), 0.4 mM glycine (Merck Sigma), 0.4 mM serine (Merck Sigma), and 17 mM [U-13C] glucose (Cambridge Isotope Laboratories, Inc). After 24h, CM was collected. One aliquot of CM was used for metabolites extraction and GC–MS analysis, as described above. Metabolites were also extracted from fibroblast cell lysates. The remaining portion of the labelled CM was used to treat PC3 cells for 24h. Then, metabolites were extracted from PC3 cells for subsequent analysis. To extract intracellular metabolites, fibroblasts and PC3 cells were washed twice with 0.9% NaCl at 4°C and scraped in 400 µL of cold 80% methanol in HPLC water containing 1 µg/ml norvaline and 1.25 µg/ml glutaric acid as internal standards. Samples were sonicated on ice for 5s for three times with a 5s interval at 70% amplitude, centrifugated at 14000 rpm, 4°C for 10 min, and supernatants were collected. Samples were dried by using a vacuum concentrator (Labconco). Dried samples were derivatized with 10 µl of 40 mg/ml methoxyamine hydrochloride (Merck #226904) in pyridine (Merck #270970) for 90 min at 37 °C. Then, 50 µl of N-(tert-butyldimethylsilyl)-N-methyl-trifluoroacetamide, with 1% tert-butyldimethylchlorosilane (Merck Sigma #375934) were added in each sample and incubated for 30 min at 60°C. GC-MS runs were performed by using an Intuvo 9000 GC / 5977B MS System (Agilent Technologies) equipped with an HP-5MS capillary column (30 m x 0.25 mm x 0.25 µm). 1 µl of each sample was injected in splitless mode using an inlet liner temperature of 240°C. GC runs were performed with helium as carrier gas at 1 ml/min. The GC oven temperature ramp was from 70°C to 280°C. The temperature of 70°C was held for 2min. Then, the first temperature ramp was from 70°C to 140°C at 3°C/min. The second ramp was from 140°C to 150°C at 1°C/min. The third temperature ramp was from 150°C to 280°C at 3°C/min. The measurement of metabolites was performed under electron impact ionization at 70 eV using a SIM mode. The ion source and transfer line temperature were set to 230°C and 290°C, respectively.
MS Quantitative Analysis software (Agilent Technologies, version 10.2) was used for determination of relative metabolite abundance: the integrated signal of selected ion for each metabolite was normalized by the signal of the norvaline or glutaric acid and by protein amount. For isotopic labelling experiments, all the measured values were corrected for
13C natural abundance by using IsoCorrectoR [
13].
Confocal Immunofluorescence
Sub-confluent PC3 and SKOV3 cells grown on glass coverslips were incubated for 72h with CM from HPFs or HOFs; Bis-2-(5-phenylacetamido-1,2,4-thiadiazol-2-yl) ethyl sulfide (BPTES) 1 µM was added during the final 16h of incubation. Nuclei were stained with DAPI (Sigma Aldrich, D9542) for 20 min at 37 °C. ETS1 protein was stained with rabbit monoclonal anti-ETS1 (1:1000, Cell Signaling, #14069), followed by anti-rabbit Alexa Fluor 568-conjugated secondary antibodies (Thermo Fisher Scientific, #A-11011, red channel). Cell fluorescence was imaged using a Leica TCS SP8 confocal scanning microscope (Leica, Mannheim, DE, USA). Observations were made with a Leica HC PL Apo CS2 X63 oil immersion objective. Images reconstruction and signal fluorescence quantification was obtained using Image J Fiji software [
14].
Statistical Analysis
Statistical analysis of the data was performed by unpaired Student t-test or One-way ANOVA for pairwise comparison of groups with GraphPad Prism version 8.0 (GraphPad Software). All data were expressed as the mean ±SEM. A p value ≤ 0.05 was considered statistically significant. All the statistical analyses were carried out on three biological replicates. Statistical analysis of gene expression datasets from patients with ovarian cancer was performed by Wilcoxon paired test.
4. Discussion
Senescent stroma is increasingly recognized as a key player in modulating cancer progression in several tumor types [
25]. Although anticancer therapies can initially restrain tumor growth, relapse is unfortunately common, and recurrent tumors often exhibit a more aggressive phenotype characterized by increased invasiveness, therapy resistance, and metastatic potential. Mounting evidence suggests that TIS-fibroblasts significantly contribute to this unfavourable evolution, as they persist in the tumor microenvironment and, through the acquisition of a SASP, create a permissive niche that fosters tumor cell survival and disease progression. While the SASP was originally defined as a complex mixture of secreted proteins, recent evidence indicates that it also includes non-protein factors, such as metabolites and extracellular vesicles, which collectively broaden its impact on the tumor microenvironment.
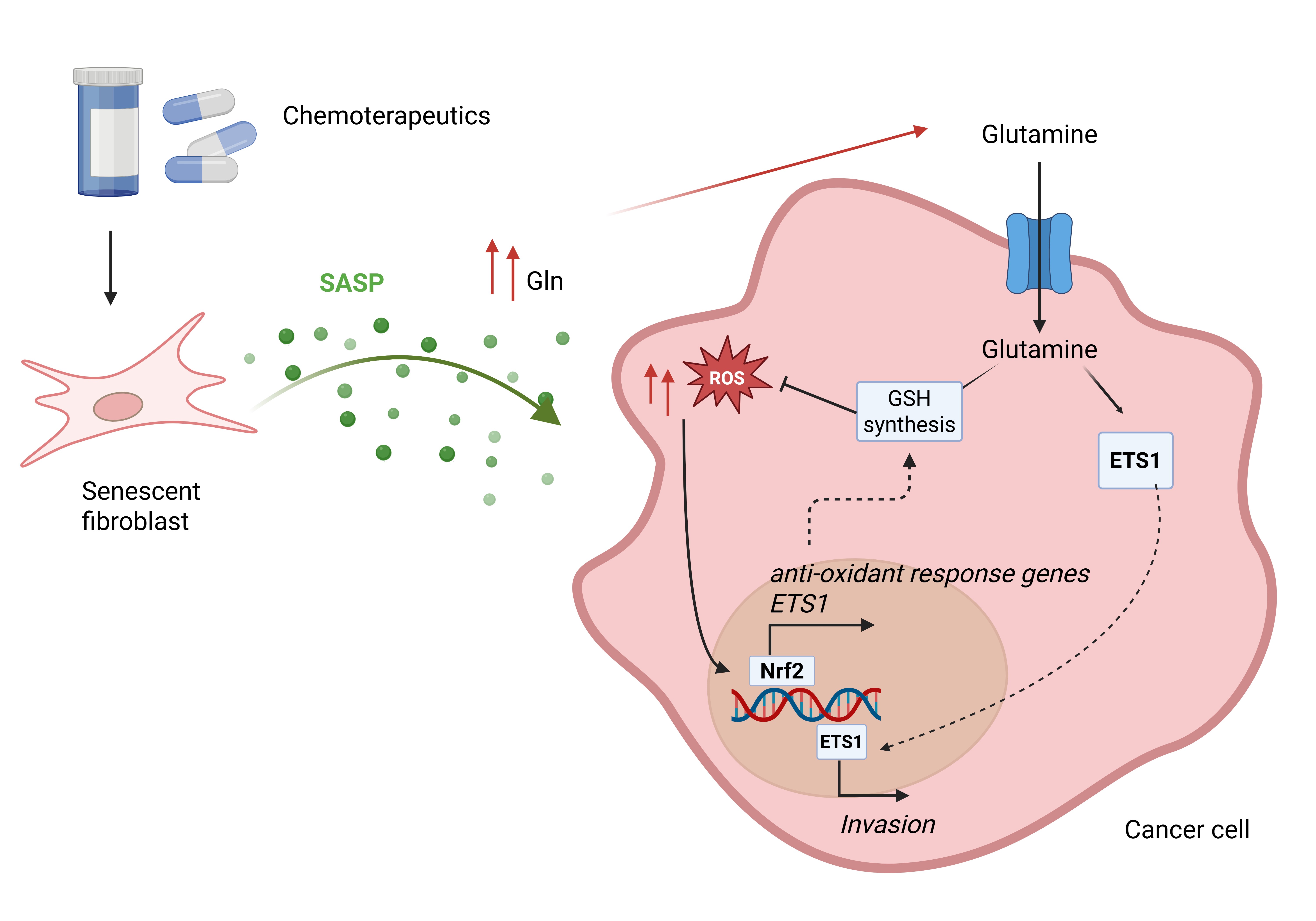
Here, we identified Gln secreted by TIS fibroblasts as a central non-protein component of the SASP able to enhance invasive and stem-like features in prostate and ovarian cancer cells. Gln is a well-established metabolic driver of tumor progression, with many cancer types described as "Gln-addicted" due to their reliance on this amino acid for energy production, redox balance, and biosynthetic processes [
17,
26,
27]. In addition, Gln metabolism can help cancer cells resist to apoptosis [
28] and contribute to generate an immunosuppressive tumor microenvironment [
11]. While previous studies have largely focused on tumor-intrinsic Gln metabolism [
15,
23,
29] and stroma-derived Gln in supporting tumor progression [
16,
30,
31], no data are available regarding the contribution of TIS fibroblasts to Gln availability within the TME. Our results highlight TIS fibroblasts as a previously underappreciated source of extracellular Gln in the TME. In this context, Gln may represent a paracrine metabolic determinant originating from a therapy-altered microenvironment. Previous data from our lab disclosed the role of CM from senescent stroma in driving aggressiveness of tumor cells through cytokine-mediated signalling, highlighting the role of canonical SASP components in modulating cancer cell behaviour [
7]. In the current study, we extend these findings by uncovering a previously underexplored metabolic dimension of the SASP. Using metabolomic profiling, we identified several metabolites enriched in the CM of senescent fibroblasts, underscoring the multifactorial nature of the senescent secretome, covering both cytokine and metabolic signals which may contribute to the pro-tumorigenic effects of the senescent microenvironment. Among these, Gln emerged as a metabolite consistently produced across both prostate and ovarian senescent stromal models, pointing to its potential as a shared and functionally relevant metabolic mediator of pro-tumoral roles of TIS fibroblasts. Cancer cell dependency from senescent stroma-derived Gln was confirmed by results obtained upon GS silencing in fibroblasts, Gln depletion in CM or pharmacological inhibition of GLS1 in cancer cells which significantly reduce invasive capacity without affecting viability. Mechanistically, senescent CM promotes the expression of the Gln transporter SLC1A5, Gln uptake, and intracellular availability, thereby triggering metabolic reprogramming and inducing EMT and stem-like traits—effects. Moreover, Gln may act as a precursor of GSH: indeed, we observed an increased level of the GSH/GSSG ratio following administration of senescent CM to cancer cells. Thus, in this context, senescent stroma-derived Gln may contribute to maintaining the redox balance in tumor cells. Finally, we identify the NRF2/ETS1 axis [
21,
32] as a downstream effector pathway that mediates Gln-driven invasion. Senescent CM elevates ROS levels in cancer cells, triggering an antioxidant response via NRF2 activation, which, in turn, promotes ETS1 expression and its nuclear translocation in a Gln-dependent manner [
22]. Given that ETS1 is a key driver of EMT, migration, and invasion [
33,
34], our data suggest that Gln serves as a metabolic regulator of redox-sensitive transcriptional programs that trigger cancer progression. In keeping, BPTES treatment prevents ETS1 nuclear translocation, sustaining the importance of senescent stroma-derived Gln in cancer cell invasion mediated by the NRF2/ETS1 axis. Finally, supporting the clinical relevance of our findings, we show that post-chemotherapy patient tissues from publicly available datasets of ovarian cancers display increased expression of GLS1 and ETS1.
Given the emerging interest in targeting Gln metabolism to halt cancer progression (particularly through glutaminase inhibitors currently in clinical trials) [
35], our findings suggest that strategically timed post-chemotherapy interventions aimed at limiting the availability or utilization of stroma-derived Gln may offer a promising therapeutic approach. Such interventions could counteract the pro-tumorigenic effects of therapy-induced senescent stroma and ultimately reduce the risk of tumor relapse.
In this light, future studies should investigate whether post-therapy treatment with GLS1 inhibitors may attenuate the pro-tumorigenic consequences of TIS. Exploring this approach in co-culture or xenograft models and identifying metabolic biomarkers of senescent stroma in patient samples, could pave the way for more effective and targeted therapeutic strategies.
Collectively, these findings provide strong rationale for targeting Gln metabolism—particularly in the context of senescent stromal niches—as a therapeutic strategy to counteract chemotherapy-induced tumor aggressiveness.
Author Contributions
Conceptualization, M.L.T. and F.M.; Methodology, G.L., C.M., C.P., D.D.; E.P., A.S., M.L., A.B., A.L.; Formal Analysis, M.B.; Investigation, G.L., C.M., C.P., D.D.; E.P., A.S., M.L., A.B., A.L.; Resources, P.S., F.M.M., F.S., M.F., S.S.; Writing-Original Draft Preparation, M.L.T.; Supervision, M.L.T. and F.M.; Funding Acquisition, M.L.T. G.L. and C.M. contributed equally to this work. All authors read and approved the final manuscript.
Figure 1.
A metabolic component secreted by senescent HPFs and HOFs is involved in the acquisition of a pro-invasive phenotype in prostate and ovarian cancer cells. A) Quantification of SA-β-Gal staining of human prostate fibroblasts (HPFs) and human ovarian fibroblasts (HOFs) treated for 24h with 5 nM Docetaxel (DTX) and 20 μM cisplatin (CPT) respectively and then incubated in culture media without drugs for further 6 days. For each condition, images were taken from 5 randomly selected fields, and both the total number of cells and the number of blue (SA-β-Gal positive) cells were counted. Bar-graphs represent the average ratio of positive cells to the total cell count. Representative images of the stained cells are shown below the bargraphs (magnification 20X, scale bar 100μm). Data are represented as mean ± SEM of three independent experiments; t-test ** p≤ 0.01). B) Representative immunoblots of p16 and p21 protein levels in DTX-treated HPFs and CPT-treated HOFs. β-actin was used as loading control. C) PC3 and SKOV3 cells were incubated with conditioned media (CM) from senescent and non-senescent (CTRL) fibroblasts for 72h. Then cells were seeded in Boyden chambers pre-coated with Matrigel. Cells were let to invade for 16h. Invading cells were stained with Crystal Violet and counted. Representative images of the filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test ** p≤ 0.01, *** p≤ 0.001). D) mRNA expression level of EMT key genes in PC3 and SKOV3 cells incubated with CM from senescent or CTRL fibroblasts for 72h. Data are represented as mean ± SEM of three independent experiments, t-test *≤ 0.05, ** p≤ 0.01). E) Prostate and ovarian cancer cells were incubated for 72h with boiled and not boiled CM from DTX-treated HPFs and CPT-treated HOFs. Invasion ability was measured with Boyden chamber assay. Representative images of the filters are shown upper the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test * ≤ 0.05, ** p≤ 0.01, φφφ ≤ 0.01, φφφφ ≤ 0.001, $$ ≤ 0.01, $$$ ≤ 0.001. * symbol indicates CM SEN boiled vs SEN non-boiled; φ symbol indicates CM CTRL boiled vs CM CTRL non-boiled; $ symbol indicates CM SEN vs CM CTRL. F) CM from senescent and control HPFs and HOFs were collected and GC-MS analysis were performed to detect secreted metabolites. Data reported are normalized on CM from CTRL fibroblasts. * symbol indicates metabolite levels significantly increased both in senescent CM from HPFs and HOFs. * p≤0.05.
Figure 1.
A metabolic component secreted by senescent HPFs and HOFs is involved in the acquisition of a pro-invasive phenotype in prostate and ovarian cancer cells. A) Quantification of SA-β-Gal staining of human prostate fibroblasts (HPFs) and human ovarian fibroblasts (HOFs) treated for 24h with 5 nM Docetaxel (DTX) and 20 μM cisplatin (CPT) respectively and then incubated in culture media without drugs for further 6 days. For each condition, images were taken from 5 randomly selected fields, and both the total number of cells and the number of blue (SA-β-Gal positive) cells were counted. Bar-graphs represent the average ratio of positive cells to the total cell count. Representative images of the stained cells are shown below the bargraphs (magnification 20X, scale bar 100μm). Data are represented as mean ± SEM of three independent experiments; t-test ** p≤ 0.01). B) Representative immunoblots of p16 and p21 protein levels in DTX-treated HPFs and CPT-treated HOFs. β-actin was used as loading control. C) PC3 and SKOV3 cells were incubated with conditioned media (CM) from senescent and non-senescent (CTRL) fibroblasts for 72h. Then cells were seeded in Boyden chambers pre-coated with Matrigel. Cells were let to invade for 16h. Invading cells were stained with Crystal Violet and counted. Representative images of the filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test ** p≤ 0.01, *** p≤ 0.001). D) mRNA expression level of EMT key genes in PC3 and SKOV3 cells incubated with CM from senescent or CTRL fibroblasts for 72h. Data are represented as mean ± SEM of three independent experiments, t-test *≤ 0.05, ** p≤ 0.01). E) Prostate and ovarian cancer cells were incubated for 72h with boiled and not boiled CM from DTX-treated HPFs and CPT-treated HOFs. Invasion ability was measured with Boyden chamber assay. Representative images of the filters are shown upper the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test * ≤ 0.05, ** p≤ 0.01, φφφ ≤ 0.01, φφφφ ≤ 0.001, $$ ≤ 0.01, $$$ ≤ 0.001. * symbol indicates CM SEN boiled vs SEN non-boiled; φ symbol indicates CM CTRL boiled vs CM CTRL non-boiled; $ symbol indicates CM SEN vs CM CTRL. F) CM from senescent and control HPFs and HOFs were collected and GC-MS analysis were performed to detect secreted metabolites. Data reported are normalized on CM from CTRL fibroblasts. * symbol indicates metabolite levels significantly increased both in senescent CM from HPFs and HOFs. * p≤0.05.
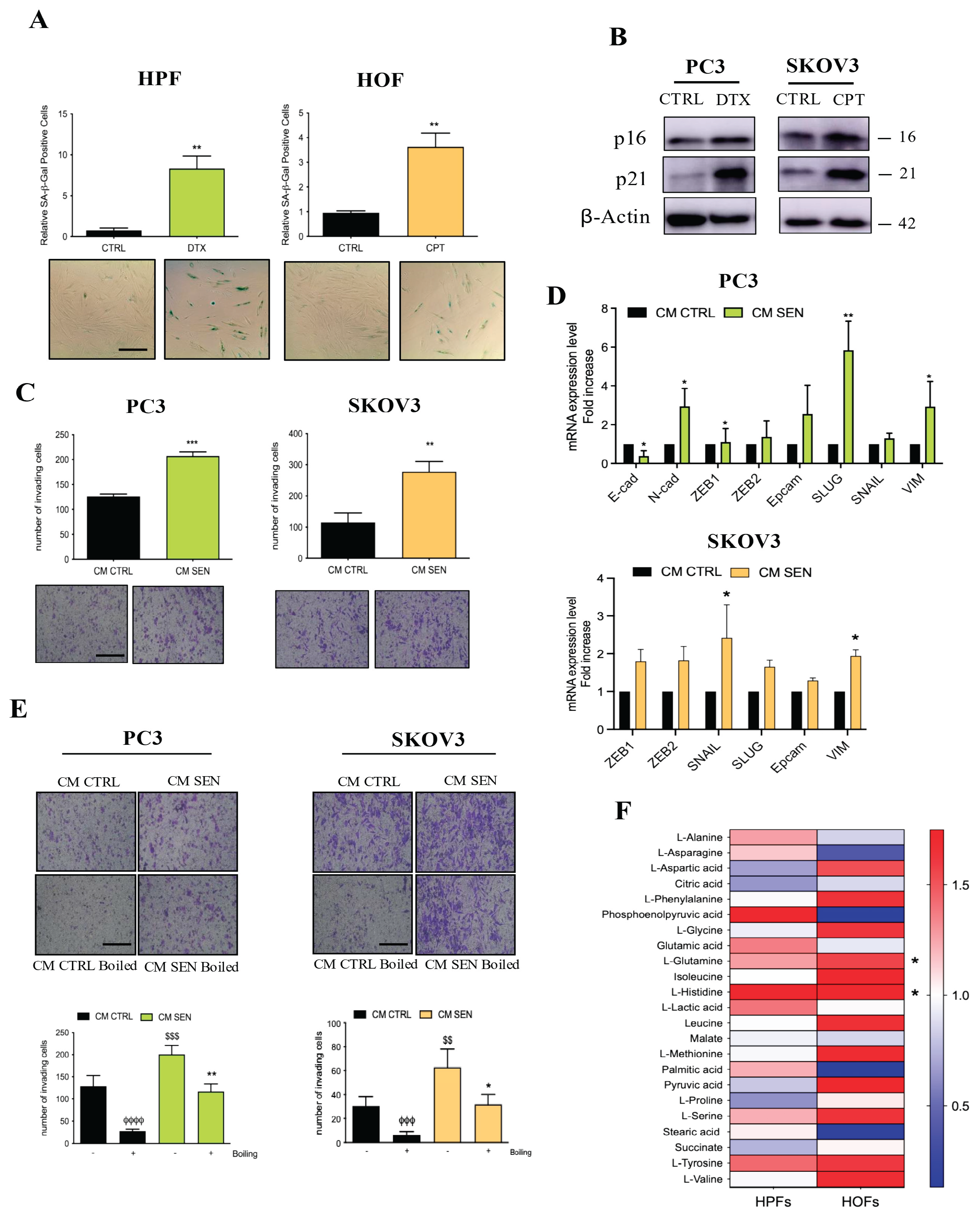
Figure 2.
Gln availability drives invasive ability in PC3 and SKOV3 cells. A) PC3 and SKOV3 cells were incubated for 72h with DMEM in presence or absence of 1% Glutamine (Gln). Invasion ability was measured with Boyden chamber assay. Representative images of the filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01, **** ≤0.0001. B) PC3 and SKOV3 were incubated with CM from DTX-treated HPFs and CPT-treated HOFs for 72h and with ASNase 1U/mL during the last 48h. Then cells were seeded in Boyden chambers pre-coated with Matrigel. Data are represented as mean ± SEM of three independent experiments; t-test * ≤0.05, *** ≤0.001, φ ≤0.05, φφ ≤0.01, $ ≤0.05, $$ ≤0.01. * symbol indicates CM SEN + ASNase vs CM SEN; φ symbol indicates CM CTRL + ASNase vs CM CTRL; $ symbol indicates CM SEN vs CM CTRL. C) mRNA expression level of Gln transporter SLC1A5 in prostate and ovarian cancer cells incubated with CM from senescent and CTRL fibroblasts for 72h. Data are represented as mean ± SEM of three independent experiments; t-test * ≤0.05. D) PC3 and SKOV3 cells were incubated for 72h with CM from senescent and CTRL HPFs and HOFs. Then GC-MS analysis was performed to measure Glu acid and Gln intracellular content. Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01. E) PC3 and SKOV3 cells were incubated for 72h with DMEM and then detached and seeded in a Boyden chamber in presence or absence of BPTES 1μM for 16h. Invaded cells were stained with Crystal Violet and counted. Representative images of the filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01, **** ≤0.0001. F) Prostate and ovarian cancer cells were incubated for 72h with boiled and not boiled CM; BPTES 1 μM was added during the final 16h of incubation. Invasive ability was measured with Boyden chamber assay. Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01, *** ≤0.001, **** ≤0.0001; φ ≤0.05, φφ ≤0.01, φφφ ≤0.001; $ ≤0.05, $$$ ≤0.001. * symbol indicates treated CM SEN vs CM SEN, φ symbol indicates treated CM CTRL vs CM CTRL, $ symbol indicates CM SEN vs CM CTRL.
Figure 2.
Gln availability drives invasive ability in PC3 and SKOV3 cells. A) PC3 and SKOV3 cells were incubated for 72h with DMEM in presence or absence of 1% Glutamine (Gln). Invasion ability was measured with Boyden chamber assay. Representative images of the filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01, **** ≤0.0001. B) PC3 and SKOV3 were incubated with CM from DTX-treated HPFs and CPT-treated HOFs for 72h and with ASNase 1U/mL during the last 48h. Then cells were seeded in Boyden chambers pre-coated with Matrigel. Data are represented as mean ± SEM of three independent experiments; t-test * ≤0.05, *** ≤0.001, φ ≤0.05, φφ ≤0.01, $ ≤0.05, $$ ≤0.01. * symbol indicates CM SEN + ASNase vs CM SEN; φ symbol indicates CM CTRL + ASNase vs CM CTRL; $ symbol indicates CM SEN vs CM CTRL. C) mRNA expression level of Gln transporter SLC1A5 in prostate and ovarian cancer cells incubated with CM from senescent and CTRL fibroblasts for 72h. Data are represented as mean ± SEM of three independent experiments; t-test * ≤0.05. D) PC3 and SKOV3 cells were incubated for 72h with CM from senescent and CTRL HPFs and HOFs. Then GC-MS analysis was performed to measure Glu acid and Gln intracellular content. Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01. E) PC3 and SKOV3 cells were incubated for 72h with DMEM and then detached and seeded in a Boyden chamber in presence or absence of BPTES 1μM for 16h. Invaded cells were stained with Crystal Violet and counted. Representative images of the filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01, **** ≤0.0001. F) Prostate and ovarian cancer cells were incubated for 72h with boiled and not boiled CM; BPTES 1 μM was added during the final 16h of incubation. Invasive ability was measured with Boyden chamber assay. Data are represented as mean ± SEM of three independent experiments; t-test ** ≤0.01, *** ≤0.001, **** ≤0.0001; φ ≤0.05, φφ ≤0.01, φφφ ≤0.001; $ ≤0.05, $$$ ≤0.001. * symbol indicates treated CM SEN vs CM SEN, φ symbol indicates treated CM CTRL vs CM CTRL, $ symbol indicates CM SEN vs CM CTRL.
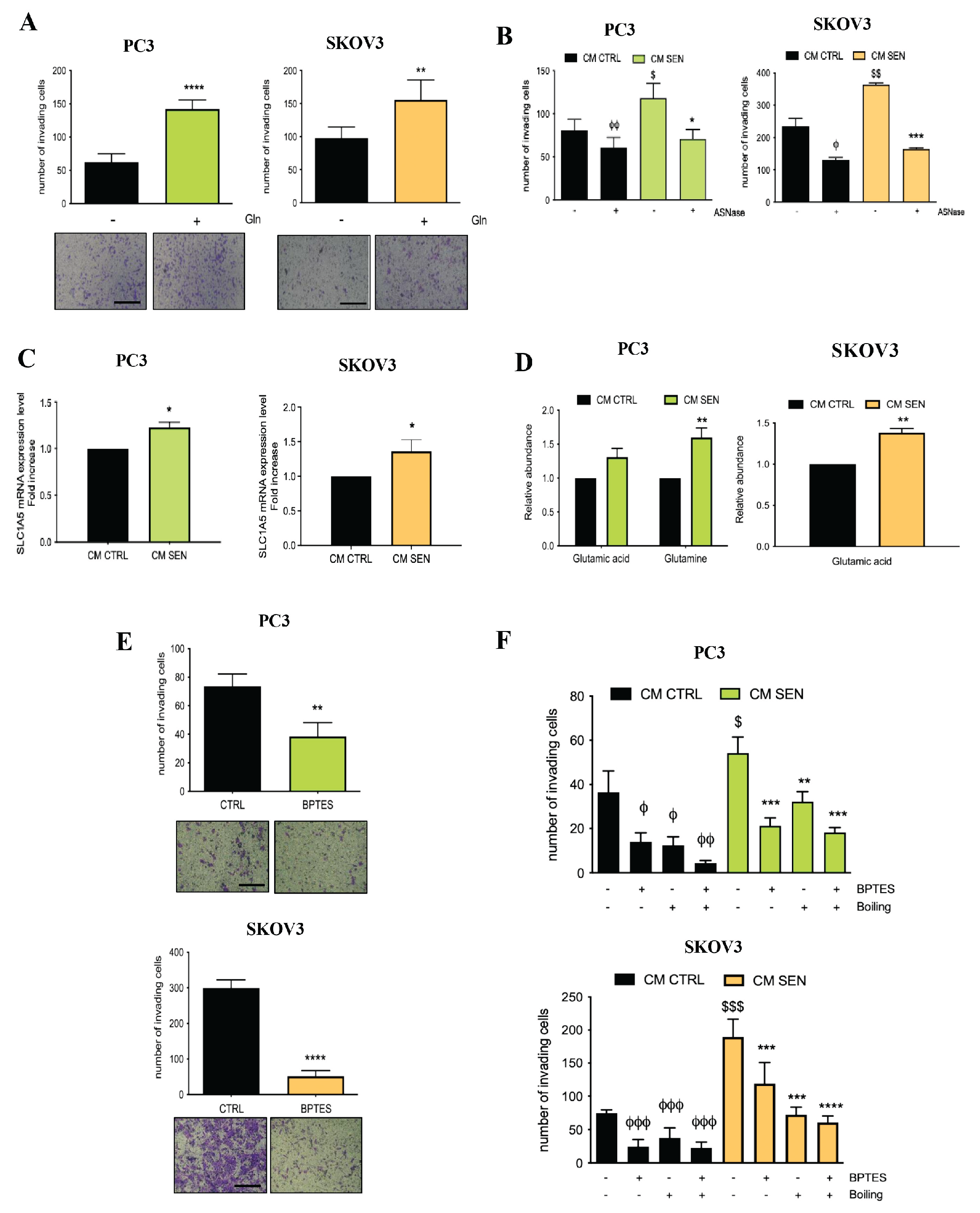
Figure 3.
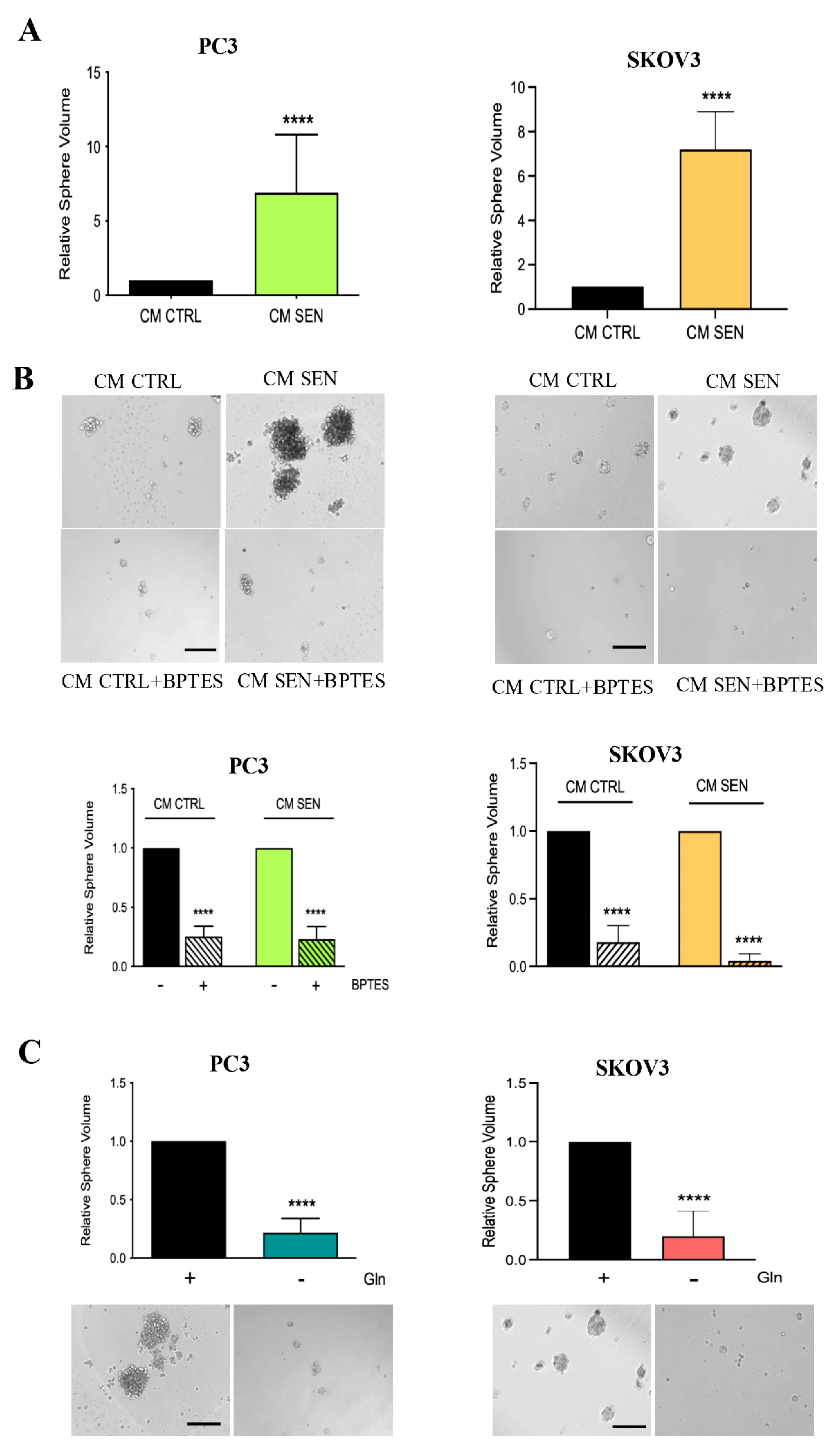
Gln confers aggressiveness to prostate and ovarian cancer cells. A-B) PC3 and SKOV3 cells were conditioned for 72h with CM from HPFs and HOFs; BPTES 1 μM was added during the final 16h of incubation. Then cells were detached and grown as spheroids for 7 days. Volumes of prostatic and ovarian spheroids were calculated as described in M&M. Representative images of spheroids are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test **** ≤0.0001. C) Prostate and ovarian cancer cells were incubated for 72h with or without 1% Gln. Then cells were detached and grown as spheroids for 7 days. Volumes of prostatic and ovarian spheroids were calculated as described in M&M. Representative images of spheroids are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test **** ≤0.0001.
Figure 3.
Gln confers aggressiveness to prostate and ovarian cancer cells. A-B) PC3 and SKOV3 cells were conditioned for 72h with CM from HPFs and HOFs; BPTES 1 μM was added during the final 16h of incubation. Then cells were detached and grown as spheroids for 7 days. Volumes of prostatic and ovarian spheroids were calculated as described in M&M. Representative images of spheroids are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test **** ≤0.0001. C) Prostate and ovarian cancer cells were incubated for 72h with or without 1% Gln. Then cells were detached and grown as spheroids for 7 days. Volumes of prostatic and ovarian spheroids were calculated as described in M&M. Representative images of spheroids are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments; t-test **** ≤0.0001.
Figure 4.
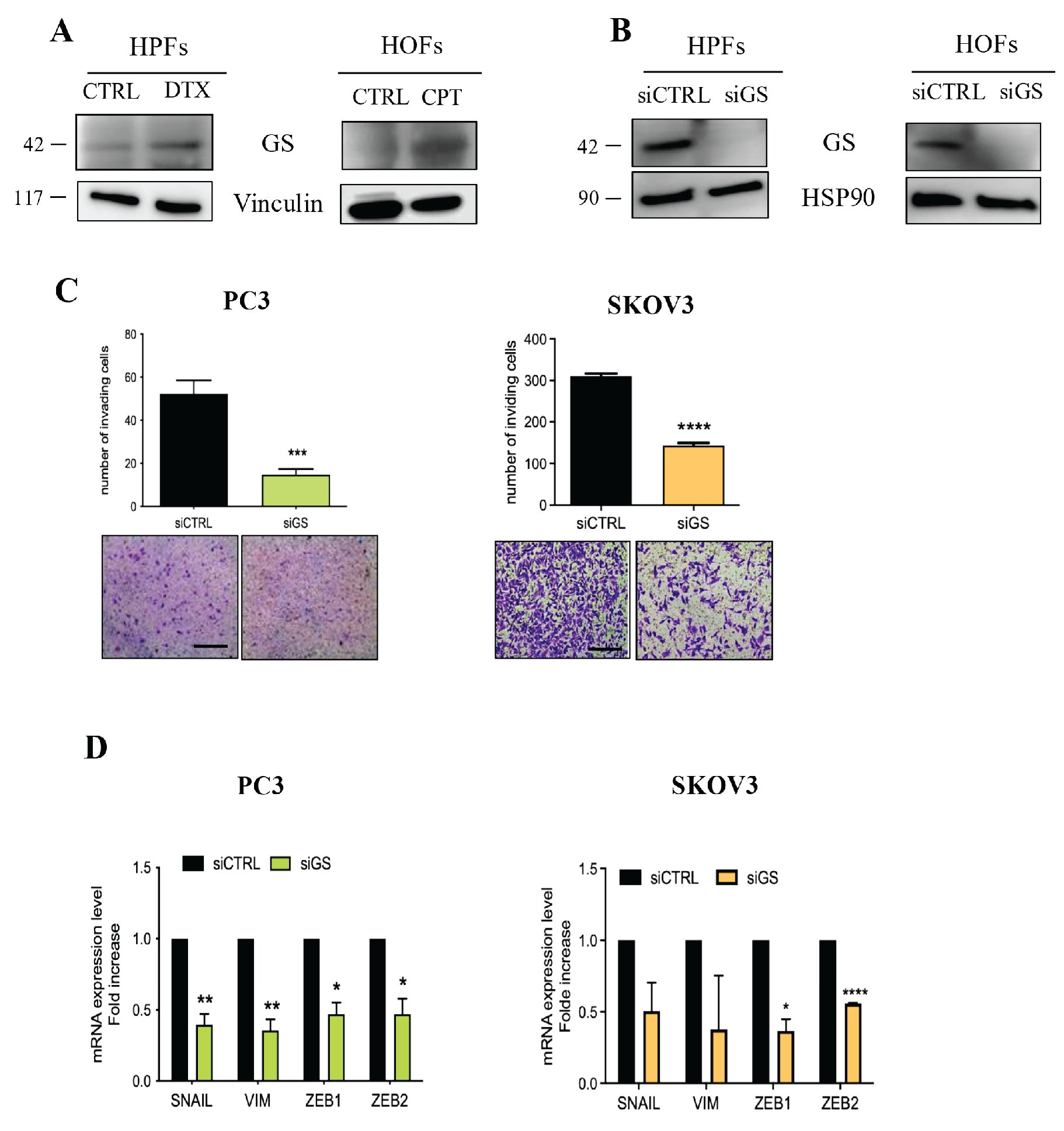
GS silencing in senescent fibroblasts affects EMT program in prostate and ovarian cancer cells. A) Immunoblot of GS protein levels in senescent and CTRL fibroblasts. Vinculin was used as loading control. B) GS protein levels in senescent HPFs and HOFs following 48h gene silencing. HSP90 immunoblot was performed to ensure equal loading. C) PC3 and SKOV3 cells were incubated for 72h with CM from GS silenced HPFs and HOFs. Then cell invasion was assessed using a Boyden chamber assay. Representative images of filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments. t-test *** ≤0.001, **** ≤0.0001. D) mRNA expression level of EMT markers in prostate and ovarian cancer cells after 72h incubation with CM from GS-silenced fibroblasts. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05, ** ≤0.01, **** ≤0.0001.
Figure 4.
GS silencing in senescent fibroblasts affects EMT program in prostate and ovarian cancer cells. A) Immunoblot of GS protein levels in senescent and CTRL fibroblasts. Vinculin was used as loading control. B) GS protein levels in senescent HPFs and HOFs following 48h gene silencing. HSP90 immunoblot was performed to ensure equal loading. C) PC3 and SKOV3 cells were incubated for 72h with CM from GS silenced HPFs and HOFs. Then cell invasion was assessed using a Boyden chamber assay. Representative images of filters are shown below the bargraphs (magnification 20X, scale bar 100 μm). Data are represented as mean ± SEM of three independent experiments. t-test *** ≤0.001, **** ≤0.0001. D) mRNA expression level of EMT markers in prostate and ovarian cancer cells after 72h incubation with CM from GS-silenced fibroblasts. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05, ** ≤0.01, **** ≤0.0001.
Figure 5.
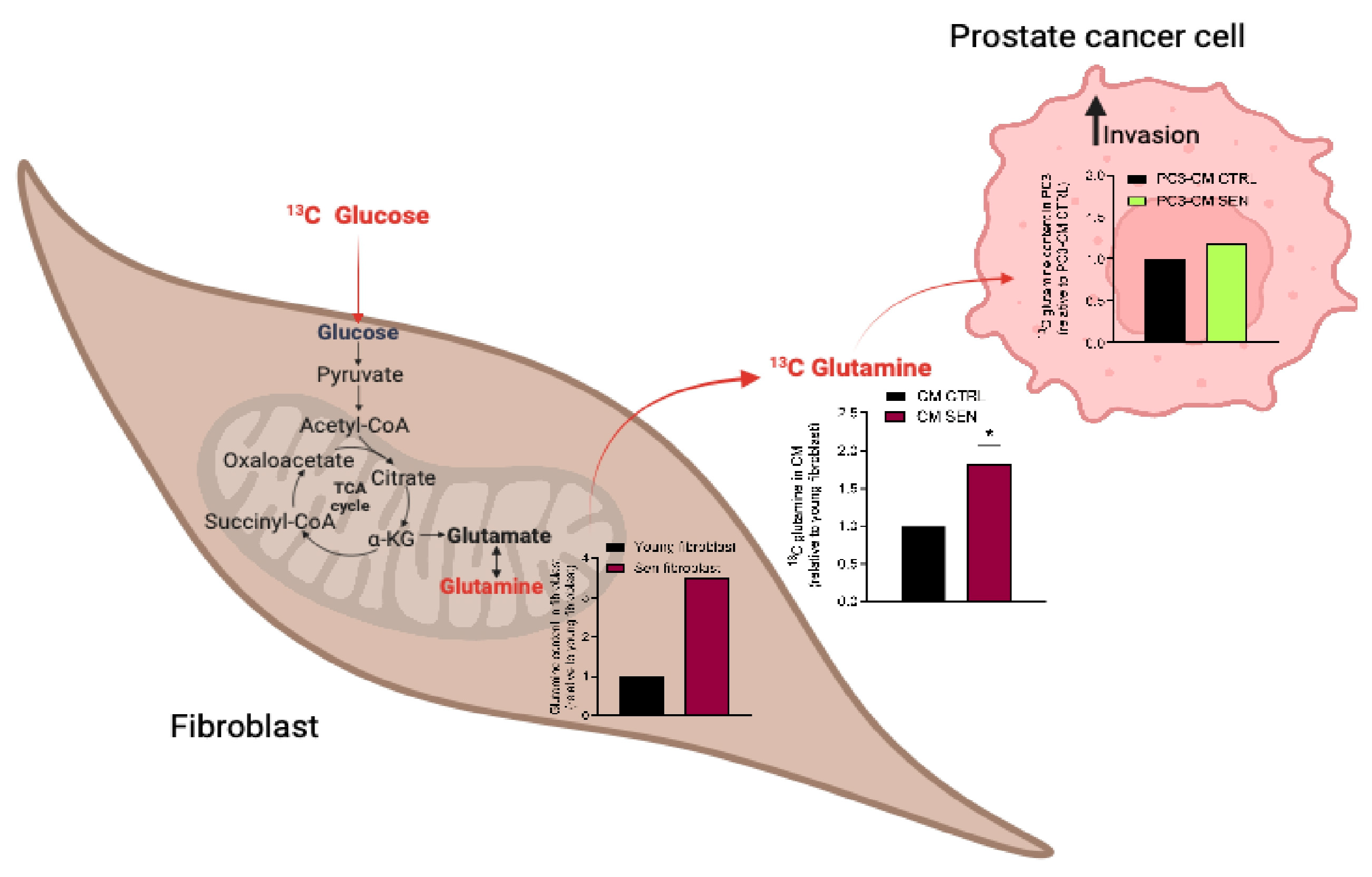
Senescent stroma-derived Gln is uploaded by PC3. CTR and senescent HPFs were incubated for 24h with 13C glucose, then cell lysates and CM were collected for GC-MS analysis. PC3 cells were incubated with CM for further 24h and the amount of 13C Gln inside cells was measured. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05.
Figure 5.
Senescent stroma-derived Gln is uploaded by PC3. CTR and senescent HPFs were incubated for 24h with 13C glucose, then cell lysates and CM were collected for GC-MS analysis. PC3 cells were incubated with CM for further 24h and the amount of 13C Gln inside cells was measured. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05.
Figure 6.
Gln-dependent NRF2/ETS1 pathway drives the invasive abilities of cancer cells. A) Total ROS production was measured with DCFDA probe in PC3 and SKOV3 cells after 72h incubation with CM from senescent and CTRL fibroblasts. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05. B) Total ROS production measured with DCFDA probe in PC3 e SKOV3 cells after incubation with boiled and not boiled CM CTRL for 72h in the presence or absence of ASNase 1U/mL during the last 48h. Data are represented as mean ± SEM of three independent experiments. t-test * ≤0.05, ** ≤0.001. C) Total ROS measured as described above, cells were conditioned for 72h, BPTES 1 μM was added during the final 16h of incubation. Data are represented as mean ± SEM of three independent experiments. t-test. t-test * ≤0.05. D) NRF2 protein level in PC3 and SKOV3 cells incubated either with DMEM in presence or absence of 1% Gln or with senescent or CTRL CM. HSP90 immunoblot was performed to ensure equal loading. E) GSH/GSSG ratio in prostate and ovarian cancer cells incubated for 72h with CM from senescent and CTRL fibroblasts. Data are normalized on protein content. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05, ** ≤0.01. F) ETS1 protein level in PC3 and SKOV3 cells incubated either with DMEM in presence or absence of 1% Gln or with senescent or CTRL CM. Actin was used as loading control. G) Tumor cells were incubated for 72h with CM from HPFs or HOFs; BPTES 1 μM was added during the final 16h of incubation. Representative confocal microscopy images show ETS1 nuclear translocation. Bargraphs on the right show the nuclear fluorescence intensity of ETS1 signal. Fluorescence intensity was quantified with ImageJ software (red: ETS1, blue: DAPI; scale bar 44 μm). Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05, $$ ≤0.01; ϕ≤0.05. * symbol indicates CM CTRL + BPTES vs CM CTRL, $ symbol indicates CM SEN vs CM CTRL, ϕ symbol indicates CM SEN + BPTES vs CM SEN.
Figure 6.
Gln-dependent NRF2/ETS1 pathway drives the invasive abilities of cancer cells. A) Total ROS production was measured with DCFDA probe in PC3 and SKOV3 cells after 72h incubation with CM from senescent and CTRL fibroblasts. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05. B) Total ROS production measured with DCFDA probe in PC3 e SKOV3 cells after incubation with boiled and not boiled CM CTRL for 72h in the presence or absence of ASNase 1U/mL during the last 48h. Data are represented as mean ± SEM of three independent experiments. t-test * ≤0.05, ** ≤0.001. C) Total ROS measured as described above, cells were conditioned for 72h, BPTES 1 μM was added during the final 16h of incubation. Data are represented as mean ± SEM of three independent experiments. t-test. t-test * ≤0.05. D) NRF2 protein level in PC3 and SKOV3 cells incubated either with DMEM in presence or absence of 1% Gln or with senescent or CTRL CM. HSP90 immunoblot was performed to ensure equal loading. E) GSH/GSSG ratio in prostate and ovarian cancer cells incubated for 72h with CM from senescent and CTRL fibroblasts. Data are normalized on protein content. Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05, ** ≤0.01. F) ETS1 protein level in PC3 and SKOV3 cells incubated either with DMEM in presence or absence of 1% Gln or with senescent or CTRL CM. Actin was used as loading control. G) Tumor cells were incubated for 72h with CM from HPFs or HOFs; BPTES 1 μM was added during the final 16h of incubation. Representative confocal microscopy images show ETS1 nuclear translocation. Bargraphs on the right show the nuclear fluorescence intensity of ETS1 signal. Fluorescence intensity was quantified with ImageJ software (red: ETS1, blue: DAPI; scale bar 44 μm). Data are represented as mean ± SEM of three independent experiments. t-test, * ≤0.05, $$ ≤0.01; ϕ≤0.05. * symbol indicates CM CTRL + BPTES vs CM CTRL, $ symbol indicates CM SEN vs CM CTRL, ϕ symbol indicates CM SEN + BPTES vs CM SEN.
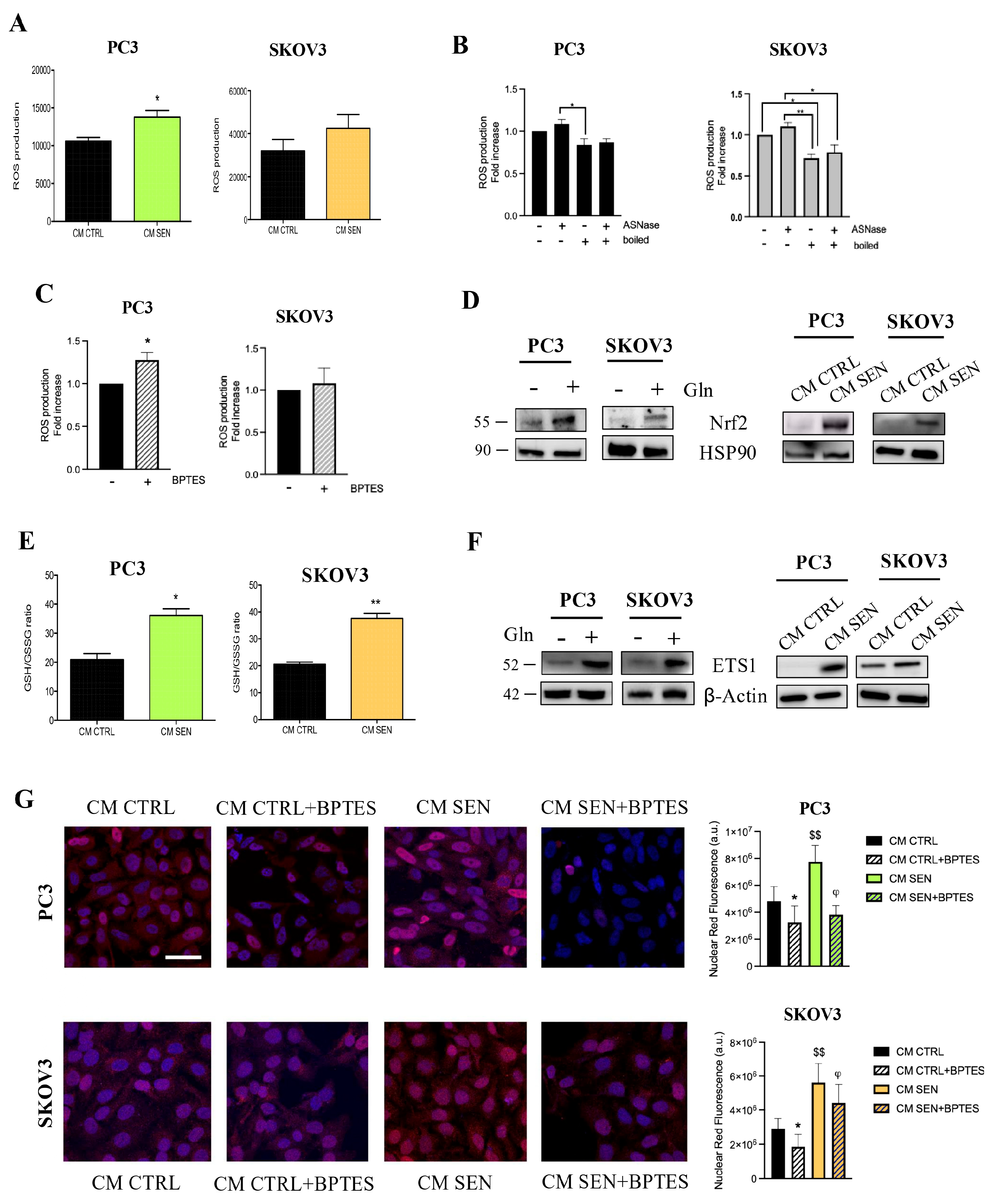
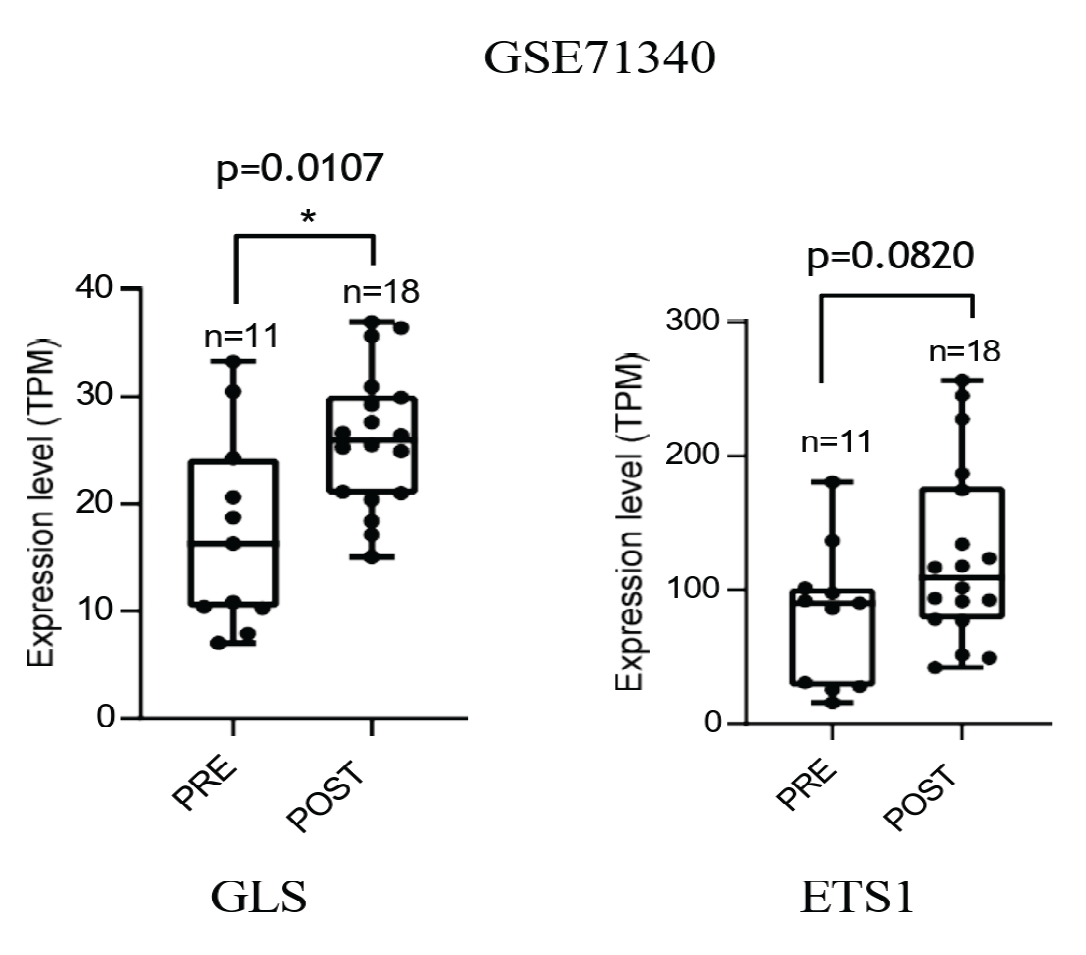
Figure 7.
Platinum-based chemotherapy promotes increased expression of GLS and ETS1 in ovarian cancer patients. Gene expression levels of GLS and ETS1 in ovarian cancer tissues pre and post platinum-based therapy from the public RNAseq dataset (GSE71340). P-values were estimated by Wilcoxon paired test.
Figure 7.
Platinum-based chemotherapy promotes increased expression of GLS and ETS1 in ovarian cancer patients. Gene expression levels of GLS and ETS1 in ovarian cancer tissues pre and post platinum-based therapy from the public RNAseq dataset (GSE71340). P-values were estimated by Wilcoxon paired test.