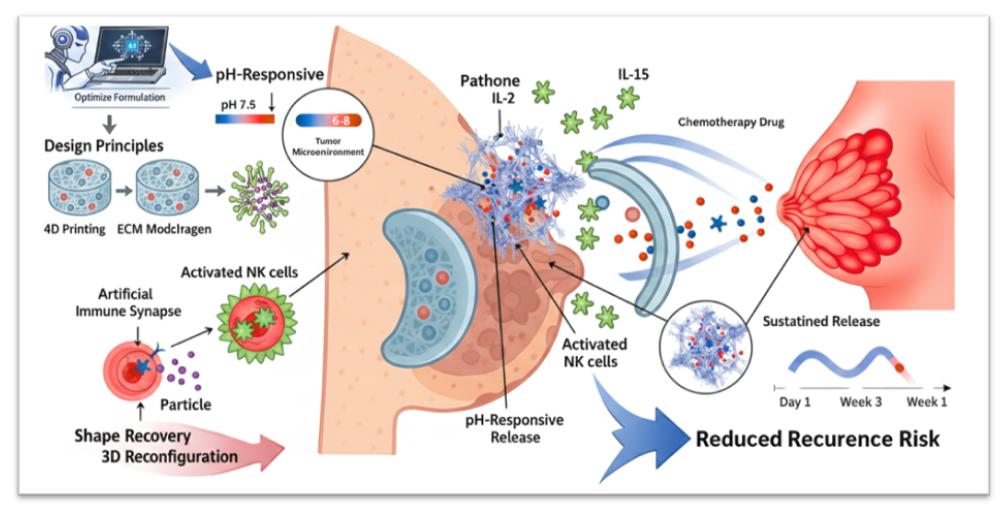
Breast cancer remains one of the leading causes of cancer-related morbidity and mortality worldwide, with tumour recurrence and therapeutic resistance largely driven by the immunosuppressive tumour microenvironment (TME). Conventional systemic chemotherapy and immunotherapy often suffer from poor tumour selectivity, systemic toxicity, and limited immune activation within the acidic and hypoxic TME. In this context, 4D-printed pH-responsive nanofiber implants have emerged as a next-generation platform capable of delivering spatiotemporally controlled therapy tailored to dynamic tumour conditions. Unlike static 3D constructs, 4D systems incorporate stimuli-responsive polymers that undergo programmed structural or functional transformations in response to environmental triggers such as acidic pH, enabling site-specific drug release. This review critically examines the design principles of pH-responsive nanofiber implants, including polymer selection, fabrication strategies, cytokine nano-assembly, and controlled release kinetics. Special emphasis is placed on TME modulation, highlighting how localised delivery of immune-stimulatory agents such as interleukin-15 and interleukin-2 can enhance natural killer cell activation, promote artificial immune synapse formation, and induce tumour apoptosis while minimising systemic toxicity. Furthermore, we analyse the translational challenges associated with manufacturing scalability, sterilisation, regulatory classification, and long-term implant safety. By integrating smart biomaterials engineering with immunotherapeutic strategies, 4D-printed nanofiber implants represent a transformative approach for localised breast cancer treatment. However, successful clinical translation will require interdisciplinary optimisation across materials science, pharmaceutical engineering, and regulatory frameworks. This review outlines future directions toward personalised, microenvironment-responsive cancer immunotherapy platforms.