1. Introduction
The synthetic chemicals known as per- and polyfluoroalkyl substances (PFAS) were discovered in the 1930s and have since been utilized in various industries due to their advantageous properties, including their resistance to high temperatures, oil, and water [
1]. Many studies to date have analyzed the biological impacts of PFAS on invertebrates and vertebrates, such as algae, fish, rodents, and humans [
2,
3,
4]. Several PFAS, including perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), are reported to bioaccumulate within species and can contribute to a wide range of biological and physiological effects (i.e., altered metabolism, endocrine disruption, oxidative stress, reproduction disruption) [
5,
6,
7,
8]. PFAS can be classified by their carbon chain lengths whereby longer carbon chains have been associated with greater toxicity than shorter carbon chains [
9] and certain PFAS have already been phased out of production due to their toxicity. For example, PFOS began to be phased out of production in the early 2000s due to its harmful impacts on humans, which include birth defects, hormone disruption, and possible carcinogenicity [
1]. Various PFAS, including PFOA and PFOS, are classified as environmental contaminants due to their documented accumulation in environmental matrices (i.e., water, soil, sludge) and organisms, which may contribute to a wide range of biological and physiological impacts. PFAS resistance to degradation contributes to their environmental presence; however, it is also proposed that environmental concentrations of PFAS are positively correlated to population density [
10]. PFAS have been found to range from varying concentrations (ng/L - µg/L) in aquatic ecosystems and have been reported in ng/g in various organisms, including humans, fish, and rodents. For instance, Hoff et al. [
11] measured the concentrations of PFOS and organohalogen pollutants in the liver of gibel carp (
Carassius auratus gibelio), carp (
Cyprinus carpio), and eel (
Anguilla anguilla) collected from Belgium and PFOS was found to range from 11.2-781, 11.3-1822, and 17.3-9031 ng/g wet weight in the species, respectively. These chemicals have already been found in different fish species, such as carp, sea bass and tilapia, and the data suggests different toxicity targets (such as the heart, liver, immunity and reproductive system) [
12]. In aquatic species, perfluorohexanoic acid (PFHxA) and perfluorohexanesulfonic acid (PFHxS) exert toxicity resulting in oxidative stress and endocrine disruption [
13]. Currently, there are hundreds of PFAS in the environment with little data on their toxicity.
Similarly, many pesticides have been banned or phased out of production due to their toxicity on non-target organisms. Pesticides are utilized worldwide to increase the harvest yield in the agricultural sector and to mitigate pests in residential areas; thus, thousands to millions of pounds of herbicidal products can be applied yearly. For example, about 240 million pounds of glyphosate were reportedly applied by ranchers and farmers in 2014 [
14]. Many herbicides in the environment are persistent due to their resistance to degradation, which can compromise soil health and cause water contamination. Consequently, numerous studies to date have assessed the impacts of pesticide application to non-target organisms. For example, juvenile
Labeo rohita fish exposure to bifenthrin and chlorpyrifos revealed DNA damage and genotoxic effects [
15]. Additionally, organophosphate insecticides have been found to have pronounced effects on fish cholinesterases (ChEs) followed by carbamates > organochlorines and pyrethroids where the herbicides organophosphates and oxazolidinones are noted to decrease the average activity of ChEs [
16]. As another example, Barbosa et al. [
17] detected azosxystrobin, epoxiconazol and chlorpyrifos in tambaqui fish muscle tissue samples collected in Rondonia State, Brazil, with values above those permitted by Brazilian legislation. Indiscriminate pesticide use continues to be a persistent issue globally.
Another pressing global issue is that of micro- and nanoplastics and their toxicity to aquatic wildlife. Significant portions of plastic waste are scattered throughout our environment. According to the United States Environmental Protection Agency [
18], plastic particles that range from 5 millimeters (mm) to 1 nanometer (nm) in size are known as microplastics (MPs). MPs can be categorized as primary or secondary MPs. Primary MPs are purposefully manufactured to be small to be incorporated in consumer products, whereas secondary MPs result from the degradation of larger plastic particles. MPs can penetrate organisms leading to their accumulation and potential toxicity mechanisms. For example, microplastics have been reported to bioaccumulate in zebrafish organs following exposure, causing toxic effects like structural deterioration of the gonads, increased oxidative stress, developmental disorders, incomplete functioning of the digestive system and decreased locomotor activity [
19].
Similar to PFAS and MPs, heavy metals (i.e., cadmium (Cd), copper (Cu), lead (Pb), mercury (Hg), nickel (Ni), and zinc (Zn)) also have the potential to accumulate within organisms, which can lead to chronic poisoning [
20]. Exposure to such metals in various fish species (i.e., goldfish (
Carassius auratus), rainbow trout (
Oncorhynchus mykiss), common carp (
Cyprinus carpio)) has been found to contribute to oxidative stress through excessive ROS production [
21,
22,
23], damage tissues, such as the intestines, liver, and gills [
22,
24], which can induce DNA damage/modification [
22,
23,
25].
Despite a wealth of knowledge regarding the individual effects of chemicals on biological systems, their combined sub-lethal toxicity effects are not well studied. Mixture toxicity will influence dosage and potency of individual compounds; however, the interaction of these compounds can also antagonistic or synergistic effects [
26]. Furthermore, there is significant concern regarding the bioaccumulation of these species in fish muscles, as this is an important nutritional source [
27]. Given the widespread utilization and persistence of PFAS, we surveyed the available studies from literature examining the effects of PFAS in combination with pesticides, MPs, or heavy metals in vertebrates and invertebrates. As a result, a more accurate representation of the environmental and health risks related to these compounds may be obtained by examining cumulative exposures rather than individual ones.
2. Materials and Methods
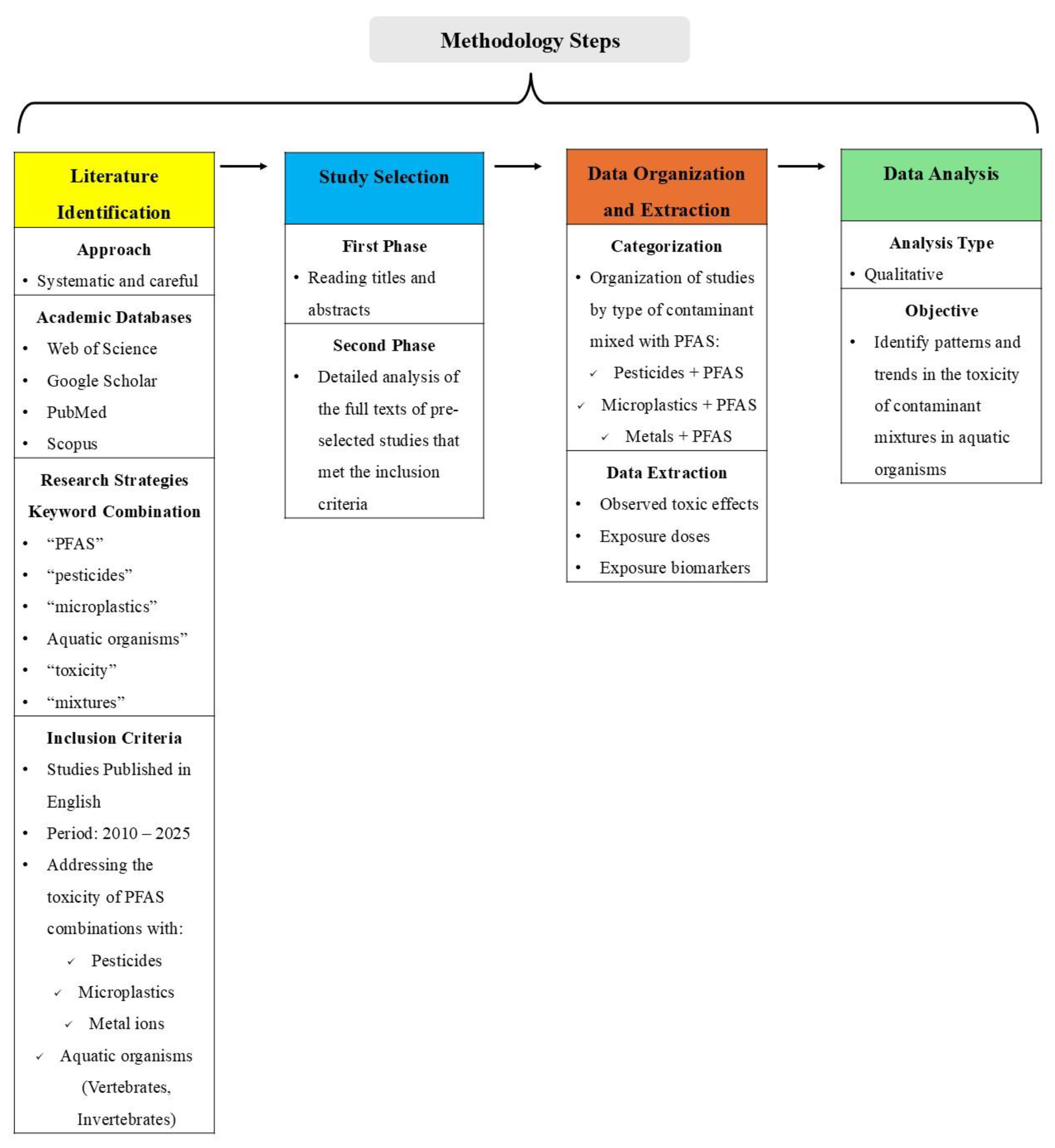
The aim of this review was to compile toxicity data for co-exposures to PFAS with either pesticides or metals or microplastics. We focused on aquatic organisms such as fish, algae, microorganisms at different endpoints. We collected and analyzed the available literature based on the recognized academic databases, such as PubMed, Scopus, Web of Science and Google Scholar, using search strategies combining related keywords, such as: "PFAS", "pesticides", "microplastics", "metal ions", "aquatic organisms", "toxicity", "mixtures", among others. After this first screening, specific pollutants were used for the search, such as “PFOS and Pesticides and fish” or “PFAS and glyphosate and toxicity”, to expand the search. To ensure comprehensive searches, the most well-studied PFAS, pesticides, and metal ions were also used (i.e., PFOA, PFOS, PFOSA, glyphosate, chlorpyrifos, Cu, iron (Fe), etc.). Searches were conducted between May 2024 to May 2025. Studies were published in English, between 2010 and 2025, that addressed the toxicity of combinations of PFAS with either pesticides, microplastics or metal ions on aquatic organisms (both vertebrates and invertebrates,). The inclusion criteria were: (1) The study involved an exposure experiment with at least one PFAS and at least one pesticide, microplastic, or metal, in environmental samples (water/sediment) and (2) measured at least one biological outcome in an aquatic species.
Figure 1.
Overview of the methodology for the literature review, including literature identification, study selection, data organization and extraction, and data analysis of PFAS-containing contaminant mixtures in aquatic organisms.
Figure 1.
Overview of the methodology for the literature review, including literature identification, study selection, data organization and extraction, and data analysis of PFAS-containing contaminant mixtures in aquatic organisms.
Methods for chemical interactions: Ground state geometry optimization for the molecules was performed by density functional theory (DFT) [
28] using the B3LYP functional with the GD3 term for Grimme’s dispersion correction [
29] and the 6-311G(d,p) basis set [
30]. In the optimization procedure, the solvent effect was also introduced using polarizable continuous model of integral equation formalism (IEFPCM) [
31] with water as solvent. First, the separated molecules had their geometry optimized and then they were combined with PFOS in different positions, based on their MEP and potential h-bond interactions. Then, optimized to see the interactions with the PFASs species. Calculations were performed with the Gaussian 09 program package [
32].
3. Results and Discussion
3.1. PFAS and Pesticides
Like PFAS, pesticides are also classified as environmental contaminants. Globally, millions of tons of pesticides are used annually for agricultural purposes in which about 3.70 million tons of pesticides were applied in 2022 alone [
33]. Pesticides can accumulate in the environment and organisms from runoff, aerial applications, and improper disposal [
34]. Additionally, pesticides may contaminate aquatic environments, potentially causing toxic effects on non-target organisms, which may contribute to declines in biodiversity over time [
35]. Despite evidence of environmental accumulation of PFAS and pesticides, limited studies evaluate the impact of these compounds simultaneously.
3.1.1. Co-Occurrence of PFAS and Pesticides in Aquatic Species
The coexistence of PFAS and pesticides has been reported in environmental matrices (i.e., surface water, sediment, vegetation) in Saudia Arabia [
36]. In addition to 12 organophosphates and 34 pharmaceutical and personal care products (PPCPs), 64 pesticides and 21 PFASs were detected in the samples analyzed. The median concentrations of PFAS in water, sediment, vegetation for human consumption, agricultural crops, and natural vegetation were 29.7 ng/L, 5.66 ng/g, 0.46 ng/g, 3.2 ng/g, and 1.88 ng/g, respectively, while pesticides had median concentrations of 231 ng/L, 40.4 ng/g, 42.0 ng/g, 57.5 ng/g, and 10.3 ng/g, respectively [
36]. Additionally, in an assessment of sediments collected from a sheltered bay on the west coast of Norway that was used as waste disposal, Dale et al. [
37] reports up to 26 μg/kg polychlorinated biphenyls (PCBs), which exceeds the environmental quality standards established by European Union legislation. PFOS, PFOA and organochlorine pesticides, including dichlorodiphenyltrichloroethane (DDT) and 2,4-dichlorophenoxyacetic acid (2,4-D), were also detected in all the sediments analyzed.
Varies studies report on the coexistence of these two chemicals within fish. Atlantic cod (
Gadus morhua) were exposed for six weeks in cages submerged in various basins of a bay in Norway that was used as a waste disposal and that has a known presence of various PFAS and pesticides [
37]. The Hepatosomatic Index and Condition Factor were significantly lower in cod kept in cages near the waste disposal site, suggesting a compromised state of health. In addition, protein biomarkers such as vitellogenin, metallothionein, and oxidative stress enzymes were measured, with significant reductions in catalase (CAT) and glutathione S-transferase (GST) activities in the liver, indicating oxidative stress. Gene expression also showed an increase in the transcriptions of genes related to lipid metabolism and steroidogenic enzymes, suggesting possible impacts on the reproductive system. Finally, high levels of 17β-estradiol (E2) were observed in the plasma of cod from the innermost season, pointing to a possible endocrine dysregulation [
37]. However, with such field studies, it is not possible to attribute these biological responses to any one class of chemical.
Other studies have measured PFAS and pesticides in the same tissues of fish. For example, tissues of aquatic species from The Netherlands, including salmon (
Salmo salar), pangasius (
Pangasius hypophthalmus), tilapia (
Oreochromis mossambicus, Oreochromis niloticus), trout (
Oncorhynchus mykiss, Salmo trutta), and shrimp (
Penaeus monodon, Penaeus vannamei, Litopenaeus vannamei), were analyzed by Van Leeuwen et al. [
38]. Samples revealed variation in the degree of contamination between species, with salmon being the most contaminated species, followed by trout, while tilapia, pangasius, and shrimp. Salmon, which have the highest lipid content, accumulated the highest concentrations of polychlorinated biphenyls (PCBs), the pesticide DDT, and PFOS, with 10,860 pg/g wet weight for PCBs, more than 1 ng/g for DDT and up to 600 pg/g for PFOS. Species with plant-based diets, such as tilapia and pangasius, tended to accumulate fewer contaminants, which reflects the influence of diet and lipid content on the bioaccumulation of these compounds. Among the organochlorine pesticides (OCPs), concentrations varied according to species, but all were below the maximum limits allowed by European Union legislation. Tilapia and pangasius showed lower levels, while salmon showed significantly higher concentrations. In relation to PFAS, PFOS was the most prevalent contaminant in shrimp, while the other PFAS samples were at low or non-detectable levels [
38]. Lastly, the presence of perfluorooctane sulfonic acid (PFOS) was assessed in liver samples of gibel carp (
Carassius auratus gibelio), carp (
Cyprinus carpio), and eel (
Anguilla anguilla) collected in the Flanders region of Belgium. The same study also investigated the presence of 13 organochlorine pesticides, as well as other organohalogenated compounds. Among the pesticides detected,
p,p’-dichlorodiphenylethylene (
p,p’-DDE) and
p,p’-dichlorodiphenyldichloroethane (
p,p’-DDD) were identified, with mean concentrations of 14.6 ng/g and 13.8 ng/g of
p,p’-DDE, and 6.2 ng/g and 4.8 ng/g of
p,p’-DDD in carp and eel, respectively. Serological alterations observed in eel liver were attributed to the presence of PFOS, with no apparent contribution from the detected pesticides, suggesting that other organohalogenated compounds present in the samples may have contributed to the observed effects [
11]. Nevertheless, it is difficult to definitively conclude that the presence of PFOS exacerbated toxicity in natural populations of fish.
3.1.2. Combined Toxicity of PFAS and Pesticides in Aquatic Species
Few laboratory studies evaluate the combined toxicity of PFAS and pesticides. One study exposed zebrafish (
Danio rerio) from 5-800 mg/L PFOA and 5-15 mg/L atrazine for 5 days to determine if co-exposure contributed to stronger compound toxicity [
39]. Co-exposure resulted in more than a 2-fold higher incidence of malformations (i.e., yolk sac abnormalities, liver abnormalities, spinal curvature) in a dose-dependent manner compared to single compound exposure. Additionally, co-exposure significantly inhibited embryo hatchability whereas single exposure to atrazine did not significantly affect hatching rate. Amino acid metabolism was analyzed, and more complex alterations were recorded in the co-exposure, including cysteine, methionine, and serine being more activated. Regarding reactive oxygen species (ROS), ROS were significantly increased by 56.5% in co-exposure compared to 29.3% and 38.6% in the single PFOA and atrazine exposure, respectively. Overall, combined exposure exerted stronger toxicity. In another study conducted by Zoupa et al. [
26], zebrafish were exposed to up to 300 µM cyproconazole or triadimefon, two fungicides, with up to 30 µM PFOS over a course of 5 days. The maximum response in fish treated with PFOS and triadimefon was higher than when combined with cyproconazole; however, this variation was proposed to be due to the lethality observed with PFOS. In the study by Valle et al. [
40], developing zebrafish were exposed to 0.62/10, 6.2/10, 62/10, and 620/10 μg/L chlorpyrifos/PFHxA for 7 days. At the highest concentration, survival and locomotor activity were reduced significantly. Additionally, co-exposure caused genes associated with neurotoxicity and oxidative stress to be upregulated and ROS levels were reduced at lower chlorpyrifos concentrations, indicating a potential modulatory interaction on oxidative stress pathways.
Other mixture experiments have been conducted in the laboratory. Bizarro et al. [
41] exposed Atlantic cod to a mixture containing PFOA, chlorpyrifos, bis-(2-ethylhexyl)-phthalate (DEHP), and 17α-ethinylestradiol (EE2) for 48 hours. The lowest concentration mixture used contained 0.01 µM EE2 and 0.1 µM chlorpyrifos, DEHP and PFOA. The other mixture tested included environmentally relevant concentrations (0.01 µM EE2, 0.1 µM chlorpyrifos, and 1 µM DEHP and PFOA). Significant effects on liver metabolism were detected following exposure to both mixtures in which transcripts related to lipid/cholesterol metabolism (
fabp and
hmg-CoA), vitamin D metabolism (
cyp24a1), and xenobiotic metabolism (
cyp3a) were altered only in mixture scenarios. It should be noted that DEHP is a plasticizer and EE2 is a pharmaceutical estrogen, which may have contributed to the biological impacts recorded.
The experiments conducted by Rodea-Palomares et al. [
42] with recombinant bioluminescent cyanobacterium Anabaena CPB4337 analyzed the interaction between PFOA and PFOS with 2,4-dichlorophenoxyacetic acid (2,4-D). The results obtained were based on the inhibition of constitutive luminescence caused by the presence of any toxic substance. Binary combinations of PFOA with 2,4-D showed antagonistic behavior throughout the range of effects analyzed. In contrast, combinations of PFOS with 2,4-D showed synergistic interactions over most of the effect range. These variations in the behavior of the mixtures may be related to the relative hydrophobicity of the compounds, since PFOA and PFOS have different cell permeability properties, affecting their interactions with the compounds. Lastly, Rodea-Palomares et al. [
43] pre-exposed cyanobacterium Anabaena CPB4337 cells to 5 mg/L PFOA or PFOS for 72 hours before being exposed to one of four herbicides: 2,4-D, atrazine, diuron, or paraquat. Cells were pre-exposed to PFAS to determine their impact on cell toxicity and both PFAS were found to significantly alter herbicide toxicity. PFOA pre-exposure significantly increased the toxicity of all herbicides, except for atrazine. For example, the toxicity of paraquat on cells doubled with PFOA pre-exposure. Regarding PFOS pre-exposure, toxicity of paraquat and diuron increased, while the toxicity of atrazine decreased.
Overall, limited data are available to examine the potential biological and physiological consequences of PFAS and pesticides in mixture. Additionally, to our knowledge, no studies to date have examined the impact of both PFAS and pesticides on mammalian models. Considering the synergistic effect reported in aquatic organisms exposed to these chemical classes, further research on mammalian data is worthy of completion.
Table 1 summarizes studies of the toxicity of PFASs and pesticides in different aquatic organisms.
3.2. PFAS and Microplastics
Plastic production continues to increase annually. According to the United Nations Environment Programme [
44], about 400 million tons of plastic is produced annually. Additionally, it is estimated that 9-14 million tons of plastic entered aquatic ecosystems annually in 2016; however, this range is projected to increase to 23-27 million tons by 2040. Overall, the projected increase in plastic production is associated with plastic accumulation in aquatic ecosystems. Overtime, plastic degrades into MPs, which allows for easy ingestion or absorption due to their small size. Various studies have elaborated on the impacts of MP presence in aquatic organisms (
Table 1) [
45,
46,
47].
3.2.1. Co-Occurrence of PFAS and Microplastics in Aquatic Species
Very few studies elaborate on the co-occurrence of PFAS and microplastics in fish tissue and water. In terms of fish, Espinosa-Ruiz et al. [
48] analyzed accumulation of PFOS and polyethylene MPs (PE-MPs) in European seabass (
Dicentrarchus labrax L.) liver after being fed 100 mg/kg PE, 4.83 μg/kg PFOS, or a combination of the two compounds (4.83 μg/kg PFOS + 100 mg/kg PE) for 21 days. No accumulation was noted in fish fed PE or PE-PFOS; however, bioaccumulation data completed by Islam et al. [
49] contradict this data as combined exposure to PE-MPs and PFOS enhanced PFOS accumulation in all soft tissues of clams (
Scrobicularia plana) after being exposed for 14 days to PE-MPs (1 mg/L) and PFOS (55.7 µg/g or 46.1 µg/g). Cocci et al. [
50] described the presence and effects of MP and PFAS accumulation in the gastrointestinal tract and coelomic fluid of sea cucumber (
Holothuria tubulosa) specimens. The types of polymers found were mainly polyethylene (PE) and polypropylene (PP). Seven PFAS were also detected, including PFBS, PFHpA, PFOA and PFHxA. In this study, however, no correlation was observed between PFAS and MP bioaccumulation.
3.2.2. Combined Toxicity of PFAS and Microplastics in Aquatic Species
Regarding combined toxicity of PFAS and microplastics on aquatic organisms, Espinosa-Ruiz et al. [
48] found that immunological effects by PFOS are reduced in European seabass (
Dicentrarchus labrax L.) when fed a diet with PFOS and polyethylene MPs (PE-MPs). Fish were fed 100 mg/kg PE, 4.83 μg/kg PFOS, or a combination of the two compounds (4.83 μg/kg PFOS + 100 mg/kg PE) for 21 days. Several immune and stress-related genes were also analyzed, and fish fed the PE-PFOS diet had decreased their regulation compared to the single compound diets. Additionally, the combined diet increased bactericidal activity by 93%. Principal component analysis was used to examine the relationships between the parameters assessed and it was found that seabass fed PE-PFOS clustered closer to those fed PE alone than when data were grouped by treatment group, confirming that PE lessens the impact of PFOS. Islam et al. [
49] study notes increased superoxide dismutase (SOD) activity around 1.7 and 1.8 times (p < 0.05) in clam gills and digestive gland after a 14-day exposure to PE-MPs (1 mg/L) and PFOS (55.7 µg/g or 46.1 µg/g). In algae, Zhao et al. [
51] examined single or co-exposure of PFOA (0.05, 0.5, 5 mg/L) and polystyrene MPs (PS-MPs; 10 mg/L) for 96 hours in the microalga
Chlorella sorokiniana where it was reported that co-exposure induced greater biotoxicity. The half maximal effective concentration (EC
50) was significantly reduced in co-exposures compared to other treatments and cell morphology (i.e., cell membrane and wall destruction) was more severely impacted with co-exposure. The ROS ratio was 209.35% following co-exposure compared to 160.01% following single exposure to PFOA. Additionally, photosynthetic inhibition of cells was also ranked as the following: PS-MPs+PFOA > PS-MPs > PFOA. The effects of co-exposure to polyvinyl chloride (PVC) and PFOA on the algae
Microcystis aeruginosa for 15 days to 50.0 mg/L PVC + 100.0 ng/L, 10.0 μg/L, 1.0 mg/L, 20.0 mg/L, and 100.0 mg/L PFOA promoted the synthesis and release of Microcystin-LR, had a synergistic effect on inhibiting algal growth, and antagonistic effects on CAT activity and malondialdehyde content were observed [
52]. Lastly, Soltanighias et al. [
53] exposed
D. magna to chronic exposures of 70 ng/L PFOS, 7 ng/L PFOA, and 50 mg/L polyethylene terephthalate (PET). Mixtures of two or all three variables were tested, and the exposure continued until each
Daphnia had released its second brood. Combined toxicity was more severe with joint exposure than single exposure in which about 59% of the effect was additive and 41% synergistic. Joint exposure also delayed sexual maturity, suppressed reproduction, triggered developmental failures, and reduced somatic growth. These studies indicate that the presence of microplastics and PFAS should concern the environment and, consequently, human health. In this sense, studies that evaluate the toxicity of these compounds in different environments and conditions are increasingly necessary.
3.3. PFAS and Metals
Metal compounds can be introduced into the environment in various ways, such as by agricultural irrigation, fertilizer consumption, pharmaceutical production, and soil erosion [
54]. Additionally, environmental levels of metals are increasing due to industrialization and urbanization [
55] and, though treatment methods to reduce levels in the environment have been created, many are costly and time-consuming. Heavy metal exposure has been noted to have consequences in both humans and animals, including disruptions to the gastrointestinal, reproductive, respiratory, and nervous systems [
56,
57]. Additionally, some heavy metals can transform into a more toxic form. For example, Hg is capable in transforming into methylmercury (MeHg), which can be readily absorbed by organisms and biomagnified in the food chain [
58].
3.3.1. Co-Occurrence of PFAS and Metals in Aquatic Species
Both metals and PFAS are present in the environment and can bioaccumulate in different aquatic organisms. The co-occurrence of each type of pollutant in tissues of aquatic species has been documented. An analysis carried out on a deceased and pregnant adult female bluntnose sixgill shark (
Hexanchus griseus) recovered in Coles Bay, Vancouver Island in 2019, detected the presence of perfluorinated compounds (such as PFOS, PFUnDA, and PFTeDA) and metal ions (such as Cr, Cd, Cu, and Pb) in the liver of the mother and offspring [
59]. The study shows that these chemicals are transferred from mother to offspring, with PFAS being deposited alongside metals. In another study conducted in Haizhou Bay, eastern China, the presence of metal ions and PFAS in the local biota was observed following analysis of various marine organisms including fish, shrimp, and gastropods [
60]. Among the metals, Zn was the most abundant, followed by Cu and chromium (Cr), while Cd and Hg were present at relatively low levels. Twelve PFAS were detected, with PFOS found in over 50% of the samples, followed by perfluorooctanoic acid (PFOA). Although some compounds were detected at concentrations considered safe limited to human consumption, their co-occurrence highlights the need for further studies on the combined effects of these pollutants on aquatic species.
3.3.2. Combined Toxicity of PFAS and Metals in Aquatic Species
Metal ions occur naturally in the environment, such as Cu and Fe, but can also result from anthropogenic sources, particularly industrial discharges, as is the case with Pb and Cd. Likewise, PFAS compounds have been increasingly reported in environmental matrices, as previously discussed. Given the frequent co-occurrence of these contaminants in aquatic ecosystems, recent studies have focused on evaluating their combined toxicological effects. Some toxicity studies have assessed the interactions and impacts caused by transition metal dichalcogenides (TMDCs – molybdenum disulfide (MoS
2) and tungsten disulfide (WS
2), and seven PFAS in cell lines and zebrafish. The studies suggested that TMDCs-PFAS interactions in aqueous media significantly increased the bioaccumulation of the species in zebrafish, which consequently increased oxidative stress in the liver and intestine of zebrafish, demonstrated by increased levels of ROS and other enzymatic activities [
61]. In goldfish, exposure to PFOA (1.21 and 12.10 µmol/L), PFOS (1 and 10 µmol/L), Cu (0.79 and 3.15 µmol/L), PFOA + Cu (1.21 + 0.79 µmol/L and 12.10 + 3.15 µmol/L), or PFOS + Cu (1 + 0.79 µmol/L and 10 + 3.15 µmol/L) for 4 days caused significant inhibition of antioxidant defense enzymes, such as CAT and SOD enzyme activities, with mixtures compared to single exposures [
62]. Toxicity of the treatment groups were also ranked: PFOA < PFOS < Cu < PFOA+Cu < PFOS+Cu. Thus, in this study, combined exposure of PFOS with a metal led to higher levels of toxicity in the goldfish.
The effect of Cd and PFOS exposure on aquatic oligochaete
Limnodrilus hoffmeisteri at various pH values was investigated and it was observed that pH values interfere with exposure impacts where combined Cd/PFOS exposure may increase acute toxicity but decrease internal concentrations of Cd [
63]. Lastly, in cyanobacterium Anabaena CPB4337 exposed to binary and tertiary mixtures containing PFOA or PFOS with Hg and Cd were completed by Rodea-Palomares et al. [
42]. Regarding PFOA mixtures, a combined exposure of PFOA and Hg showed strong antagonism where PFOA, Hg, and Cd led to dual synergistic/antagonistic behavior. Regarding PFOS mixtures, both the binary and tertiary mixtures showed strong antagonism. Such interaction towards heavy metals may be due to the ability of PFAS to stabilize these metals through complexation or ion exchange. A study investigated the response of
Vallisneria natans (
V. natans) to combined exposure to PFOS and Cu at concentrations of 1.0, 10.0, and 100.0 μg/L [
64]. The results indicated that both individual and combined treatments triggered the metabolism of reactive oxygen species (ROS), as evidenced by increased activities of glutathione (GSH), malondialdehyde (MDA), catalase (CAT), total superoxide dismutase (TSOD), and an overall antagonistic joint toxicity. Transcriptomic analyses further revealed that the expression levels of detoxification-related genes, such as peroxidase (PER), glutaredoxin 9 (GRX9), and glutathione S-transferase 3 (GST3) were upregulated in response to exposure. Moreover, both single and combined exposures induced the upregulation of transporter-related genes encoding aquaporins, rapid-type anion channels, and P-type ATPase copper exporters, thereby enhancing the uptake of PFOS and Cu in
V. natans and promoting detoxification processes. This study confirmed that the individual toxicity of PFOS and Cu was greater than that observed under co-exposure conditions, suggesting an antagonistic interaction between PFOS and Cu in submerged macrophytes.
3.4. Theoretical Calculations
We also performed theoretical calculations using PFOS as a model compound to determine whether this molecule could interact with common environmental contaminants such as pesticides, microplastics, and metals. For this purpose, three herbicides from different chemical classes were selected: atrazine, a triazine herbicide; azamethiphos and glyphosate, both organophosphorus compounds with distinct structures. In addition, a PET monomer was included to represent a microplastic particle.
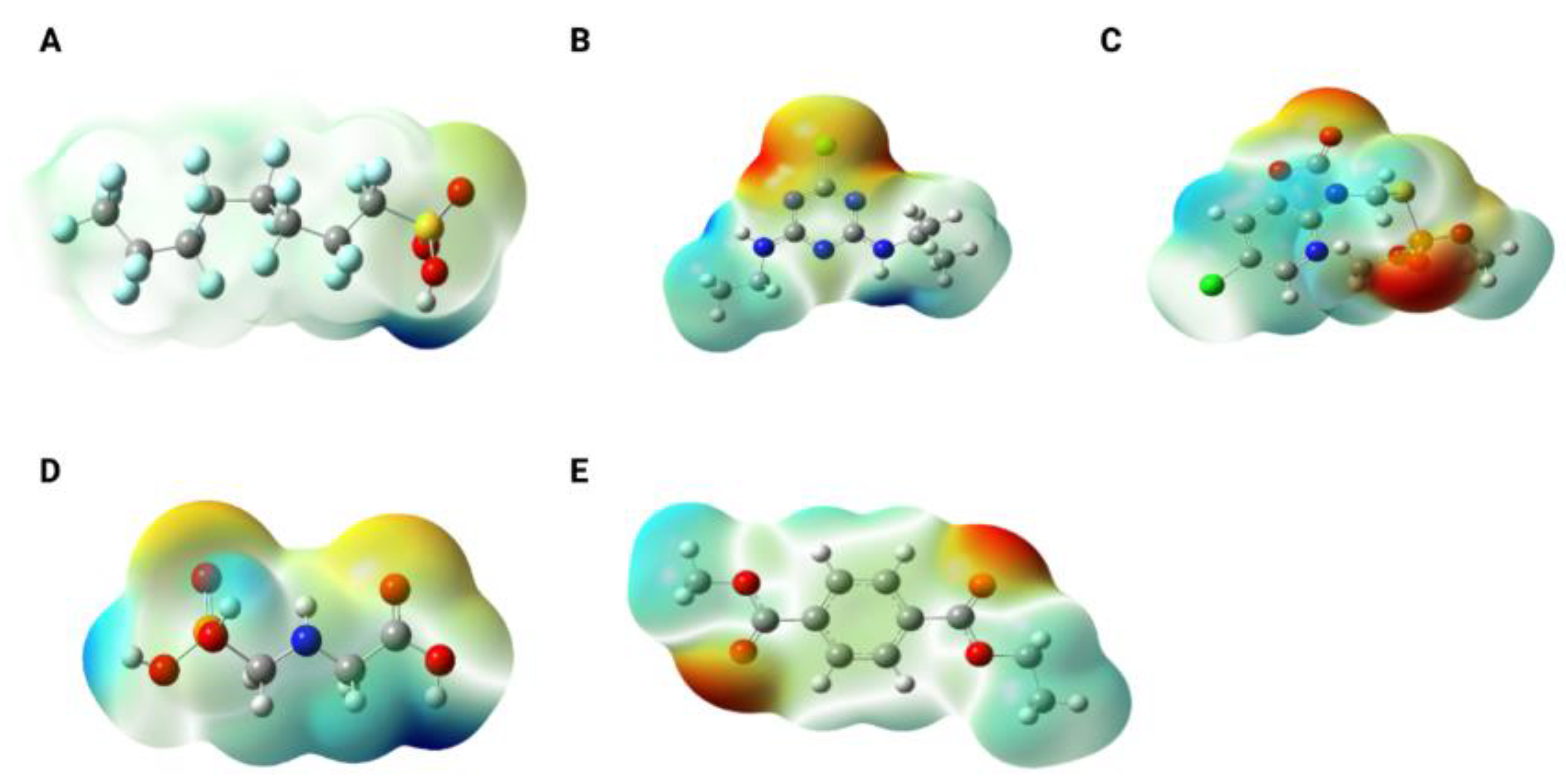
Figure 2 shows the electrostatic potential maps of the molecules studied. As observed, PFOS (
Figure 2A) exhibits a slight negative charge density over the oxygen atoms, while the remainder of the chain behaves as a traditional alkyl chain, displaying overall neutrality. This neutrality arises from the cancellation of the C–F bond polarity vectors. Atrazine (
Figure 2B) presents a high negative charge density around the chlorine atom, whereas the amine groups in the lateral branches exhibit regions of positive charge density. Azamethiphos (
Figure 2C) displays a pronounced negative charge density over the carbonyl oxygen of the ring and the phosphoryl oxygen (P=O), along with a slight positive charge density on the ring carbons adjacent to the chlorine atom. Glyphosate (
Figure 2D) shows strong negative charge densities over the oxygens of the carboxylic acid and phosphate groups, and high positive charge densities on the hydrogen atoms of the terminal –OH groups (phosphate and carboxylic acid). Finally, the PET monomer also exhibits negative charge density localized over the carbonyl oxygens of the ester groups.
After evaluating the charge density distribution of the individual molecules, additional analyses were performed to investigate possible interactions between the species. All molecules were evaluated in combination with PFOS. Supplemental
Figure S1 presents the electrostatic potential maps for all assessed interactions.
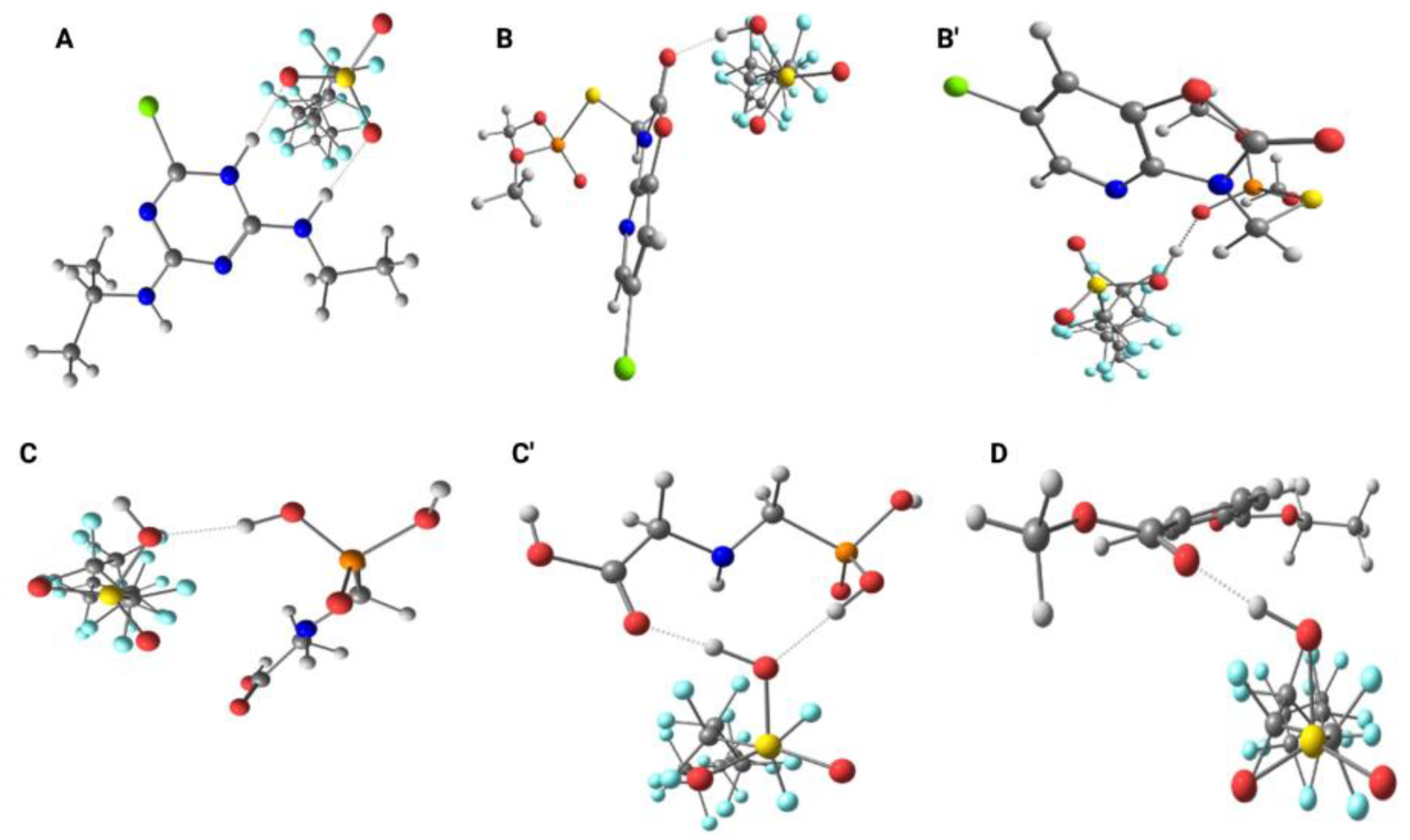
The possible molecular interactions can be better visualized in
Figure 3. The interaction between PFOS and atrazine (
Figure 3A) showed that the charge densities on the chlorine atom and on the atoms of the triazine ring became more positive in the presence of PFOS, probable due to the influence of the fluorine atoms. The calculations indicated that the proximity between the molecules may favor the formation of a hydrogen bond between the –S=O group of PFOS and the nitrogen atom of the –NHCH₂CH₃ amine group. Furthermore, the data suggest proton donation from the –SOH group of PFOS to the nitrogen atom of the atrazine ring, followed by the formation of an additional hydrogen bond.
In the evaluation of the interactions between PFOS and azamethiphos, and between PFOS and glyphosate, the data suggests two possible sites for hydrogen bond formation in these herbicides. For azamethiphos, possible interactions can occur at the carbonyl group of the ring (
Figure 3B) and at the phosphate group (
Figure 3B’). In both cases, the hydrogen atom from the –SOH group of PFOS acts as the donor in the hydrogen bonding interaction with azamethiphos. In the case of glyphosate, the results indicate the possible formation of a hydrogen bond between the phosphate group of glyphosate and the –SOH group of PFOS (
Figure 3C). Additionally, the calculations showed the potential for simultaneous interactions, in which the same –SOH group of PFOS can forms one hydrogen bond with the phosphate group and another with the carbonyl oxygen of the carboxylic acid group in glyphosate (
Figure 3C’). Finally, for the PET monomer (
Figure 3D), the results indicate a potential interaction between the oxygen atom of the C=O group in the PET ester and the –SOH group of PFOS.
All molecules exhibit regions of positive and negative charge, as well as oxygen atoms that can facilitate intermolecular interactions in solution. These interactions can modify the behavior of these molecules on the environment. Based on the obtained data, it can be suggested that, in the presence of metal ions, these molecules may further interact through electrostatic attraction, forming ion–permanent dipole interactions and possibly metal complexes, owing to the availability of lone electron pairs on the oxygen atoms.
4. Conclusion
Studies in the literature have demonstrated that PFAS co-occurring with other pollutants, such as pesticides, microplastics, and metal ions contribute to increased toxicity and harm aquatic organisms. Available evidence suggests that the coexistence of PFAS and other contaminants in environmental matrices is a reality, raising concerns about ecological and health risks. In vivo and in vitro studies in several aquatic organisms indicate that co-exposure can exacerbate toxicity, leading to a higher incidence of malformations, inhibition of hatching, increased oxidative stress and alterations in metabolism and gene expression. However, some studies also point to antagonistic interactions, where the presence of one contaminant can mitigate the effects of the other, highlighting the complexity of these interactions. Notably, research on the combined effects of PFAS and pesticides is particularly scarce, despite evidence of synergism observed in aquatic organisms. Similarly, although the ubiquitous presence of microplastics and PFAS in aquatic ecosystems is concerning, studies investigating their combined effects are still in int infancy. The interaction of PFAS with metals ions demonstrates the potential to alter bioavailability and exacerbate toxic effects. Thus, the complexity of interactions between PFAS and other environmental contaminants highlights the urgent need for further research.
This review highlights a significant gap in our understanding of how PFAS interact with other common aquatic contaminants, and how these compounds can influence toxicity in aquatic organisms. Although numerous studies have examined the individual effects of these pollutants, research on their combined effects, particularly at the sub-lethal levels, and studies reporting on synergistic or antagonistic interactions, remains limited. Our computational chemical analysis demonstrates that these chemicals can indeed interact, which can lead to differences in receptor binding affinities and novel mechanisms of toxicity. Future studies should focus on elucidating the mechanisms underlying these complex interactions, investigating effects at different trophic levels and in a broader range of species, including mammalian models, and considering chronic exposures and environmentally relevant mixtures. Understanding the effects of cumulative exposure is crucial for more accurate environmental and human health risk assessment and for developing effective management and remediation strategies to protect the health of aquatic ecosystems and public health.
Supplementary Materials
The following supporting information can be downloaded at: Preprints.org, Figure S1: Electrostatic Potential Map
Author Contributions
Conceptualization, C.J.M.; investigation, C.Q.C., E.M.A.V., E.I., H.P.M.O., L.C., and M.L.P.; writing—original draft preparation, E.M.A.V. and E.I.; writing—review and editing, C.J.M., E.M.A.V. and E.I.; supervision, C.J.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the São Paulo Research Foundation (FAPESP, Portuguese: Fundação de Amparo à Pesquisa do Estado de São Paulo) (Grant # 2022/12776-2).
Institutional Review Board Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| 2,4-D |
2,4-dichlorophenoxyacetic acid |
| ATPase |
Adenosine triphosphatase |
| B3LYP |
Becke three-parameter Lee–Yang–Parr functional |
| CAT |
Catalase |
| Cd |
Cadmium |
| ChEs |
Cholinesterases |
| Cr |
Chromium |
| Cu |
Copper |
| DDT |
Dichlorodiphenyltrichloroethane |
| DEHP |
Bis-(2-ethylhexyl)-phthalate |
| DFT |
Density functional theory |
| DNA |
Deoxyribonucleic acid |
| E2 |
17β-estradiol |
| EC50 |
Half maximal effective concentration |
| EE2 |
17α-ethinylestradiol |
| Fe |
Iron |
| GD3 |
Grimme’s dispersion correction (D3) |
| GSH |
Glutathione |
| GST |
Glutathione S-transferase |
| GST3 |
Glutathione S-transferase 3 |
| Hg |
Mercury |
| hmg-CoA |
3-hydroxy-3-methylglutaryl–coenzyme A |
| IEFPCM |
Integral equation formalism polarizable continuum model |
| MeHg |
Methylmercury |
| MDA |
Malondialdehyde |
| MEP |
Molecular electrostatic potential |
| MPs |
Microplastics |
| MoS₂ |
Molybdenum disulfide |
| Ni |
Nickel |
| OCPs |
Organochlorine pesticides |
| Pb |
Lead |
| PCBs |
Polychlorinated biphenyls |
| PE |
Polyethylene |
| PE-MPs |
Polyethylene microplastics |
| PER |
Peroxidase |
| PET |
Polyethylene terephthalate |
| PFAS |
Per- and polyfluoroalkyl substances |
| PFBS |
Perfluorobutanesulfonic acid |
| PFHpA |
Perfluoroheptanoic acid |
| PFHxA |
Perfluorohexanoic acid |
| PFHxS |
Perfluorohexanesulfonic acid |
| PFOA |
Perfluorooctanoic acid |
| PFOS |
Perfluorooctane sulfonic acid |
| PFOSA |
Perfluorooctanesulfonamide |
| PFTeDA |
Perfluorotetradecanoic acid |
| PFUnDA |
Perfluoroundecanoic acid |
| PP |
Polypropylene |
| PPCPs |
Pharmaceutical and personal care products |
| PS-MPs |
Polystyrene microplastics |
| PVC |
Polyvinyl chloride |
| ROS |
Reactive oxygen species |
| SOD |
Superoxide dismutase |
| TMDCs |
Transition metal dichalcogenides |
| TSOD |
Total superoxide dismutase |
| Zn |
Zinc |
References
- Singh, K. Per-and polyfluoroalkyl substances (PFAS) as a health hazard: current state of knowledge and strategies in environmental settings across Asia and future perspectives. Chemical Engineering Journal 2023, 145064. [Google Scholar] [CrossRef]
- Liu, X. Joint toxicity mechanisms of binary emerging PFAS mixture on algae (Chlorella pyrenoidosa) at environmental concentration. Journal of Hazardous Materials 2022, 437, 129355. [Google Scholar] [CrossRef]
- Rericha, Y. Diverse PFAS produce unique transcriptomic changes linked to developmental toxicity in zebrafish. Frontiers in Toxicology 2024, 6, 1425537. [Google Scholar] [CrossRef] [PubMed]
- Solan, M.E. Comparative cytotoxicity of seven per-and polyfluoroalkyl substances (PFAS) in six human cell lines. Toxicology 2022, 477, 153281. [Google Scholar] [CrossRef] [PubMed]
- Behr, A.-C. Activation of human nuclear receptors by perfluoroalkylated substances (PFAS). Toxicology in Vitro 2020, 62, 104700. [Google Scholar] [CrossRef]
- Grønnestad, R. Effects of an environmentally relevant PFAS mixture on dopamine and steroid hormone levels in exposed mice. Toxicology and Applied Pharmacology 2021, 428, 115670. [Google Scholar] [CrossRef]
- Piva, E. Antioxidant responses induced by PFAS exposure in freshwater fish in the veneto region. Antioxidants 2022, 11, 1115. [Google Scholar] [CrossRef]
- Taibl, K.R. Associations of per-and polyfluoroalkyl substances (PFAS) and their mixture with oxidative stress biomarkers during pregnancy. Environment international 2022, 169, 107541. [Google Scholar] [CrossRef]
- Sodani, K. Toxicological mode-of-action and developmental toxicity of different carbon chain length PFAS. Toxicology Letters 2025, 405, 59–66. [Google Scholar] [CrossRef]
- Jarvis, A.L. Perfluorooctane sulfonate in US ambient surface waters: A review of occurrence in aquatic environments and comparison to global concentrations. Environmental toxicology and chemistry 2021, 40, 2425–2442. [Google Scholar] [CrossRef]
- Hoff, P.T. Perfluorooctane sulfonic acid and organohalogen pollutants in liver of three freshwater fish species in Flanders (Belgium): relationships with biochemical and organismal effects. Environmental pollution 2005, 137, 324–333. [Google Scholar] [CrossRef]
- Ivantsova, E.; Lu, A.; Martyniuk, C.J. Occurrence and toxicity mechanisms of perfluorobutanoic acid (PFBA) and perfluorobutane sulfonic acid (PFBS) in fish. Chemosphere 2024, 349, 140815. [Google Scholar] [CrossRef] [PubMed]
- Kreychman, M. A comparative review of the toxicity mechanisms of perfluorohexanoic acid (PFHxA) and perfluorohexanesulphonic acid (PFHxS) in fish. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 2024, 109874. [Google Scholar] [CrossRef] [PubMed]
- Benbrook, C.M. Trends in glyphosate herbicide use in the United States and globally. Environmental Sciences Europe 2016, 28, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bano, N. Effect of pesticides on erythrocytes of indigenous fish Labeo rohita. Journal of King Saud University-Science 2021, 33, 101586. [Google Scholar] [CrossRef]
- Santana, M.S. Pesticide effects on fish cholinesterase variability and mean activity: A meta-analytic review. Science of the Total Environment 2021, 757, 143829. [Google Scholar] [CrossRef]
- Barbosa, E.F. Pesticide residues detected in Colossoma macropomum by the modified QuEChERS and GC-MS/MS methods. Acta Scientiarum. Technology 2023, 45, e63831. [Google Scholar] [CrossRef]
- Programme, U.N.E. n.d. Available online: https://www.unep.org/interactives/beat-plastic-pollution/.
- Yön Ertuğ, N. Recent advances of microplastics toxicity and fate on zebrafish-a review. International Journal of Environmental Science and Technology 2024, 1–16. [Google Scholar] [CrossRef]
- Ali, M.M. Environmental pollution with heavy metals: A public health concern. Heavy metals-their environmental impacts and mitigation 2021, 771–783. [Google Scholar]
- Lee, J.-W. Review of cadmium toxicity effects on fish: Oxidative stress and immune responses. Environmental research 2023, 236, 116600. [Google Scholar] [CrossRef]
- Nedzvetsky, V. Indices of the DNA repair system in the brain of fish as a biomarker of inorganic mercury burden. Ecology and Noospherology 2021, 32, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Mercury exposure induces the release of neutrophil extracellular traps and apoptosis in carp neutrophils. Aquaculture 2021, 533, 736103. [Google Scholar] [CrossRef]
- Yu, Y. Bioaccumulation, histopathological and apoptotic effects of waterborne cadmium in the intestine of crucian carp Carassius auratus gibelio. Aquaculture Reports 2021, 20, 100669. [Google Scholar] [CrossRef]
- Chouke, P.B. Bioinspired NiO nanospheres: Exploring in vitro toxicity using Bm-17 and L. rohita liver cells, DNA degradation, docking, and proposed vacuolization mechanism. Acs Omega 2022, 7, 6869–6884. [Google Scholar] [CrossRef]
- Zoupa, M. Dose addition in chemical mixtures inducing craniofacial malformations in zebrafish (Danio rerio) embryos. Food and chemical toxicology 2020, 137, 111117. [Google Scholar] [CrossRef] [PubMed]
- Wu, P. Chemical mixtures of mercury, PCBs, PFAS, and pesticides in freshwater fish in the US and the risks they pose for fish consumption. Environmental Research 2025, 266, 120381. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L.J. Self-consistent equations including exchange and correlation effects. Physical review 1965, 140, A1133. [Google Scholar] [CrossRef]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. Journal of computational chemistry 2011, 32, 1456–1465. [Google Scholar] [CrossRef]
- Proppe, J.; Gugler, S.; Reiher, M. Gaussian process-based refinement of dispersion corrections. Journal of chemical theory and computation 2019, 15, 6046–6060. [Google Scholar] [CrossRef]
- Cancès, E.; Mennucci, B.; Tomasi, J. A new integral equation formalism for the polarizable continuum model: Theoretical background and applications to isotropic and anisotropic dielectrics. The Journal of chemical physics 1997, 107, 3032–3041. [Google Scholar] [CrossRef]
- Frisch, M.J. Gaussian 09, Revision A; Gaussian, Inc.: Wallingford CT, 2016. [Google Scholar]
- FAO, Pesticides use and trade - 1990-2022. FAOSTAT Analytical Briefs. Rome, 2024; No. 89.
- Kaur, R. Pesticides: An alarming detrimental to health and environment. Science of The Total Environment 2024, 170113. [CrossRef] [PubMed]
- Wan, N.-F. Pesticides have negative effects on non-target organisms. Nature communications 2025, 16, 1360. [Google Scholar] [CrossRef]
- Picó, Y. A reconnaissance study of pharmaceuticals, pesticides, perfluoroalkyl substances and organophosphorus flame retardants in the aquatic environment, wild plants and vegetables of two Saudi Arabia urban areas: Environmental and human health risk assessment. Science of the Total Environment 2021, 776, 145843. [Google Scholar] [CrossRef] [PubMed]
- Dale, K. Contaminant accumulation and biological responses in Atlantic cod (Gadus morhua) caged at a capped waste disposal site in Kollevåg, Western Norway. Marine environmental research 2019, 145, 39–51. [Google Scholar] [CrossRef]
- Van Leeuwen, S. Halogenated contaminants in farmed salmon, trout, tilapia, pangasius, and shrimp. Environmental science & technology 2009, 43, 4009–4015. [Google Scholar]
- Zhao, X. Synergistic developmental effects of zebrafish exposed to combined perfluorooctanoic acid and atrazine. Chemosphere 2024, 358, 142080. [Google Scholar] [CrossRef]
- Valle, E.M.A. Toxicity Assessment of Mixture Effects of Insecticides and Perfluorinated Chemicals (PFAS) in Zebrafish (Danio rerio): A Case Study With Chlorpyrifos and Perfluorohexanoic Acid (PFHxA). Journal of Applied Toxicology 2025. [Google Scholar] [CrossRef] [PubMed]
- Bizarro, C. Single and mixture effects of aquatic micropollutants studied in precision-cut liver slices of Atlantic cod (Gadus morhua). Aquatic Toxicology 2016, 177, 395–404. [Google Scholar] [CrossRef]
- Rodea-Palomares, I. Toxicological interactions of perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) with selected pollutants. Journal of hazardous materials 2012, 201, 209–218. [Google Scholar] [CrossRef]
- Rodea-Palomares, I. Effect of PFOA/PFOS pre-exposure on the toxicity of the herbicides 2, 4-D, Atrazine, Diuron and Paraquat to a model aquatic photosynthetic microorganism. Chemosphere 2015, 139, 65–72. [Google Scholar] [CrossRef]
- Nations, U. World Environment Day: Unite to Beat Plastic Pollution. 2025. Available online: https://www.un.org/it/node/71013.
- Barboza, L.G.A. Microplastics in wild fish from North East Atlantic Ocean and its potential for causing neurotoxic effects, lipid oxidative damage, and human health risks associated with ingestion exposure. Science of the total environment 2020, 717, 134625. [Google Scholar] [CrossRef]
- Qiao, R. Microplastics induce intestinal inflammation, oxidative stress, and disorders of metabolome and microbiome in zebrafish. Science of the Total Environment 2019, 662, 246–253. [Google Scholar] [CrossRef]
- Yong, C.Q.Y.; Valiyaveettil, S.; Tang, B.L. Toxicity of microplastics and nanoplastics in mammalian systems. International journal of environmental research and public health 2020, 17, 1509. [Google Scholar] [CrossRef]
- Espinosa-Ruiz, C. Immunotoxicological effects of perfluorooctanesulfonic acid on European seabass are reduced by polyethylene microplastics. Fish & Shellfish Immunology 2023, 137, 108793. [Google Scholar] [CrossRef]
- Islam, N. Perfluorooctane sulfonic acid (PFOS) adsorbed to polyethylene microplastics: accumulation and ecotoxicological effects in the clam Scrobicularia plana. Marine Environmental Research 2021, 164, 105249. [Google Scholar] [CrossRef] [PubMed]
- Cocci, P. Levels and oxidative toxicity of microplastics and perfluoroalkyl substances (PFASs) in different tissues of sea cucumber (Holothuria tubulosa). Science of The Total Environment 2025(962), 178472. [CrossRef] [PubMed]
- Zhao, Z. Response mechanisms of Chlorella sorokiniana to microplastics and PFOA stress: Photosynthesis, oxidative stress, extracellular polymeric substances and antioxidant system. Chemosphere 2023, 323, 138256. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W. Effects of combined exposure of PVC and PFOA on the physiology and biochemistry of Microcystis aeruginosa. Chemosphere 2023, 338, 139476. [Google Scholar] [CrossRef]
- Soltanighias, T. Combined toxicity of perfluoroalkyl substances and microplastics on the sentinel species Daphnia magna: Implications for freshwater ecosystems. Environmental Pollution 2024, 363, 125133. [Google Scholar] [CrossRef]
- Zaynab, M. Health and environmental effects of heavy metals. Journal of King Saud University-Science 2022, 34, 101653. [Google Scholar] [CrossRef]
- Qu, L. Risk analysis of heavy metal concentration in surface waters across the rural-urban interface of the Wen-Rui Tang River, China. Environmental pollution 2018, 237, 639–649. [Google Scholar] [CrossRef]
- Massányi, P. Effects of cadmium, lead, and mercury on the structure and function of reproductive organs. Toxics 2020, 8, 94. [Google Scholar] [CrossRef]
- Witkowska, D.; Słowik, J.; Chilicka, K. Heavy metals and human health: Possible exposure pathways and the competition for protein binding sites. Molecules 2021, 26, 6060. [Google Scholar] [CrossRef]
- Al-Sulaiti, M.M.; Soubra, L.; Al-Ghouti, M.A. The causes and effects of mercury and methylmercury contamination in the marine environment: A review. Current Pollution Reports 2022, 8, 249–272. [Google Scholar] [CrossRef]
- Zvekic, M. Unique hepatic maternal transfer pattern of trace metals and perfluoroalkyl substances (PFAS) in a bluntnose sixgill shark (Hexanchus griseus). Chemosphere 2024, 359, 142315. [Google Scholar] [CrossRef]
- Chen, J. Distribution, trophic magnification and risk of trace metals and perfluoroalkyl acids in marine organisms from Haizhou Bay. Environmental Research 2024, 261, 119746. [Google Scholar] [CrossRef]
- Liu, G. Unraveling the joint toxicity of transition-metal dichalcogenides and per-and polyfluoroalkyl substances in aqueous mediums by experimentation, machine learning and molecular dynamics. Journal of Hazardous Materials 2023, 443, 130303. [Google Scholar] [CrossRef] [PubMed]
- Feng, M. Evaluation of single and joint toxicity of perfluorooctane sulfonate, perfluorooctanoic acid, and copper to Carassius auratus using oxidative stress biomarkers. Aquatic Toxicology 2015, 161, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Qu, R. The toxic effect and bioaccumulation in aquatic oligochaete Limnodrilus hoffmeisteri after combined exposure to cadmium and perfluorooctane sulfonate at different pH values. Chemosphere 2016, 152, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y. Ecological effects and molecular mechanisms of single and coexisting PFOS and Cu exposure on submerged macrophytes and periphytic biofilms in aquatic environments. Environmental and Experimental Botany 2023, 213, 105435. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).