1. Introduction
Healthcare-associated infections (HAIs) remain a major global public health challenge and an important indicator of health system performance. Despite advances in antimicrobial therapy, intensive care technologies, and infection prevention and control (IPC) practices, HAIs continue to contribute substantially to preventable morbidity, mortality, prolonged hospitalisation, and escalating healthcare costs worldwide [
1,
2]. The World Health Organization (WHO) defines HAIs as infections acquired during the provision of healthcare that were neither present nor incubating at the time of admission, including those that become apparent after discharge but were contracted during hospitalisation [
1].
Globally, pooled prevalence estimates indicate that HAIs remain particularly high in low- and middle-income countries (LMICs), especially within intensive care units (ICUs) [
1,
3]. A recent meta-analysis of ICU populations in sub-Saharan Africa reported a pooled HAI prevalence of approximately 28.2%, identifying intubation, catheterisation, comorbidities, prolonged hospital stay, neonatal age, and advanced age (>50 years) as significant risk factors [
3]. However, heterogeneity in surveillance methodologies and diagnostic criteria across settings continues to limit direct comparisons and benchmarking.
ICUs represent high-risk environments for HAI acquisition because of the convergence of critical illness, immunological vulnerability, invasive device use, broad-spectrum antimicrobial exposure, and extended lengths of stay [
2,
3]. Device-associated infections—particularly ventilator-associated pneumonia (VAP), central line-associated bloodstream infection (CLABSI), and catheter-associated urinary tract infection (CAUTI)—account for a substantial proportion of ICU-related HAIs globally [
2]. Although evidence-based IPC bundles and structured surveillance systems have reduced device-associated infections in well-resourced settings, their implementation remains inconsistent in many resource-limited environments [
2,
3].
HAIs are also closely intertwined with antimicrobial resistance (AMR), an escalating environmental and public health threat [
4]. The 2025 WHO Global Antibiotic Resistance Surveillance Report highlights increasing resistance among priority bacterial pathogens, underscoring the challenge of treating ICU-acquired infections effectively [
4]. ICUs function as ecological niches for multidrug-resistant organisms due to intense antimicrobial selective pressures and frequent invasive procedures. The relationship is bidirectional: HAIs drive antimicrobial consumption, while antimicrobial misuse accelerates resistance, further increasing the risk and severity of HAIs [
4].
In South Africa, HAI epidemiology is shaped by a complex disease burden characterised by high prevalence of HIV and tuberculosis alongside rising non-communicable diseases. Although tertiary centres in more resourced provinces report HAI prevalence estimates ranging from 14% to 22% in general wards [
5], rural provinces such as the Eastern Cape remain underrepresented in surveillance literature, particularly for ICU populations. Context-specific epidemiological data are essential to guide IPC implementation and antimicrobial stewardship in resource-constrained settings.
This study aimed to determine the prevalence, risk factors, and mortality impact of HAIs among adult and paediatric ICU patients at Nelson Mandela Academic Hospital. We demonstrate a high HAI burden with prolonged ICU stay as an independent predictor and a significant association between HAI occurrence and mortality, underscoring the urgent need for strengthened infection prevention strategies in rural tertiary ICUs.
2. Materials and Methods
2.1. Study Design and Setting
A retrospective cross-sectional study was conducted in the adult and paediatric intensive care units (ICUs) of Nelson Mandela Academic Hospital, a tertiary referral centre serving a predominantly rural population in the Eastern Cape Province, South Africa. The ICUs provide advanced organ support, including mechanical ventilation and central venous access. The study period extended from 1 January 2022 to 31 December 2023.
2.2. Study Population and Eligibility Criteria
All patients admitted to the adult and paediatric ICUs during the study period were screened for eligibility. Inclusion criteria were ICU admission for ≥48 hours and availability of complete clinical records. Patients with documented infection at the time of ICU admission or with incomplete records were excluded. For patients with multiple ICU admissions during the same hospitalisation, only the first admission was included to avoid duplication.
2.3. Data Collection and Variables
Data were extracted from ICU admission registers, individual patient medical records, nursing charts, ICU flow sheets, the hospital laboratory information system, microbiology culture and susceptibility reports, medication prescription charts, and pharmacy records. A predesigned and pilot-tested structured data abstraction form was developed specifically for this study. The tool was reviewed by two senior clinicians and a microbiologist to ensure content validity and alignment with study objectives. A pilot extraction of 10 randomly selected ICU records was undertaken to refine variable definitions and standardise interpretation across data collectors.
Sociodemographic variables included age at admission and sex. Clinical variables included documented comorbidities. ICU-related exposure variables comprised total ICU length of stay, mechanical ventilation use and duration, central venous catheterisation, and indwelling urinary catheterisation. Device exposure was recorded if the device had been in situ for ≥48 hours.
Healthcare-associated infections (HAIs) were defined according to standard ≥48-hour post-admission criteria consistent with international surveillance definitions. For each suspected HAI episode, the date of specimen collection, specimen type, isolated organism(s), and antimicrobial susceptibility profiles were recorded. Microorganisms were classified using standard microbiological nomenclature, and susceptibility interpretations (susceptible, intermediate, resistant) followed the laboratory’s Clinical and Laboratory Standards Institute (CLSI)-aligned reporting system in use during the study period.
Antibiotic utilisation data included all systemic antibiotics administered during ICU stay, specifying agent, class, start date, and whether therapy was empirical (initiated prior to culture results) or culture-directed (adjusted according to documented susceptibility findings).
Data extraction was performed by two trained research assistants with clinical backgrounds. To ensure reliability, 10% of records were independently re-abstracted by a second reviewer. Discrepancies were resolved through discussion and consultation with a senior investigator. Completed data forms were checked daily for completeness and consistency.
2.4. Statistical Analysis
Data were analysed using IBM SPSS Statistics (Version 27.0; IBM Corp., Armonk, NY, USA). Continuous variables were assessed for normality and summarised as means ± standard deviation (SD) or medians with interquartile ranges (IQR), as appropriate. Categorical variables were summarised as frequencies and percentages.
HAI prevalence was calculated as the proportion of ICU patients who developed at least one HAI during admission. Associations between categorical variables were assessed using Chi-square or Fisher’s exact tests, while continuous variables were compared using independent t-tests or Mann–Whitney U tests as appropriate.
Multivariable logistic regression analysis was performed to identify independent predictors of HAIs. Variables with p < 0.20 in univariable analysis were entered into the multivariable model. Adjusted odds ratios (aORs) with 95% confidence intervals (CIs) were reported. Statistical significance was defined as p < 0.05.
2.5. Ethical Considerations
Ethical approval was obtained from the Walter Sisulu University Health Sciences Research Ethics Committee (approval number: 058/2025) and the Eastern Cape Department of Health. Institutional permission was granted by hospital management.
As this was a retrospective study using routinely collected data, the ethics committee waived the requirement for informed consent. Patient confidentiality was maintained through anonymisation and assignment of unique study identifiers. Data were stored in password-protected electronic files accessible only to the research team.
2.6. Data Availability and Use of Generative Artificial Intelligence
The dataset analysed in the current study is not publicly available due to institutional and ethical restrictions on patient confidentiality but may be made available from the corresponding author upon reasonable request and with permission from the relevant authorities.
No generative artificial intelligence tools were used in study design, data collection, statistical analysis, or interpretation of findings. Any language editing performed did not influence the scientific content of the manuscript.
3. Results
3.1. Patient Characteristics
A total of 266 ICU patients met the inclusion criteria. The median age was 6.5 years (IQR, 0–32), and infants aged <1 year constituted the largest subgroup (33.8%). Males accounted for 53.4% of admissions. Patients were distributed across adult (41.4%), paediatric (34.6%), and neonatal (24.1%) ICUs. Overall ICU mortality was 27.1%.
Table 1.
Demographic and Clinical Characteristics of ICU Patients (n = 266).
Table 1.
Demographic and Clinical Characteristics of ICU Patients (n = 266).
| Characteristic |
n (%) |
| Median age (IQR) |
6.5 years (0–32) |
| Sex |
|
| Male |
142 (53.4) |
| Female |
124 (46.6) |
| Age group (years) |
|
| <1 |
90 (33.8) |
| 1–14 |
65 (24.4) |
| 15–34 |
50 (18.8) |
| 35–55 |
36 (13.5) |
| ≥56 |
25 (9.4) |
| ICU type |
|
| Adult |
110 (41.4) |
| Paediatric |
92 (34.6) |
| Neonatal |
64 (24.1) |
| Urinary catheter present |
132 (49.6) |
| Mortality |
72 (27.1) |
3.2. Prevalence and Spectrum of Healthcare-Associated Infections
Seventy-seven patients developed at least one healthcare-associated infection (HAI), yielding a prevalence of 28.95%. Most affected patients experienced a single HAI episode (80.5%). Ventilator-associated pneumonia (VAP) was the predominant infection (40.2%), followed by central line-associated bloodstream infection (CLABSI) (28.3%) and catheter-associated urinary tract infection (CAUTI) (25.0%). Surgical site infections (SSI) were uncommon (6.5%). Infants were disproportionately affected compared with older age groups.
Table 2.
Distribution of Healthcare-Associated Infections (n = 77).
Table 2.
Distribution of Healthcare-Associated Infections (n = 77).
| HAI Type |
n (%) |
| Ventilator-associated pneumonia (VAP) |
37 (40.2) |
| Central line-associated bloodstream infection (CLABSI) |
26 (28.3) |
| Catheter-associated urinary tract infection (CAUTI) |
23 (25.0) |
| Surgical site infection (SSI) |
6 (6.5) |
3.3. Microbiological Profile and Antibiotic Utilisation
Gram-negative organisms predominated, particularly Acinetobacter baumannii, followed by Escherichia coli and Klebsiella pneumoniae. Fungal isolates included Candida species, while Gram-positive organisms included coagulase-negative staphylococci and Staphylococcus aureus.
Antibiotic utilisation varied by ICU type. Adult and paediatric ICUs more frequently prescribed broader-spectrum agents, whereas neonatal prescribing patterns were dominated by ampicillin–gentamicin–based empirical regimens.
Table 3.
Microbiological Profile and Antibiotic Utilisation Patterns in ICU Patients.
Table 3.
Microbiological Profile and Antibiotic Utilisation Patterns in ICU Patients.
| Domain |
Category |
Variable |
n |
% |
| Microbiological Profile |
Gram-negative isolates |
Acinetobacter baumannii |
32 |
21.1 |
| |
|
Escherichia coli |
21 |
13.8 |
| |
|
Klebsiella pneumoniae |
17 |
11.2 |
| |
|
Pseudomonas aeruginosa |
16 |
10.5 |
| |
Fungal isolates |
Candida species |
16 |
10.5 |
| |
Gram-positive isolates |
Coagulase-negative staphylococci |
17 |
11.2 |
| |
|
Staphylococcus aureus |
12 |
7.9 |
| |
|
Enterococcus faecalis |
6 |
3.9 |
| |
|
Streptococcus group D |
5 |
3.3 |
| Antibiotic Utilisation |
Adult ICU (most prescribed) |
Ceftriaxone |
27 |
21.4* |
| |
|
Amoxicillin–clavulanate |
22 |
17.5* |
| |
|
Meropenem |
21 |
16.7* |
| |
Paediatric ICU (most prescribed) |
Azithromycin |
32 |
22.5* |
| |
|
Ampicillin |
27 |
19.0* |
| |
|
Meropenem |
21 |
14.8* |
| |
Neonatal ICU (most prescribed) |
Ampicillin |
61 |
31.8* |
| |
|
Gentamicin |
57 |
29.7* |
| |
|
Meropenem |
20 |
10.4* |
3.4. Risk Factors for Healthcare-Associated Infection
In multivariable logistic regression analysis, prolonged ICU stay (>8 days) independently predicted HAI (adjusted odds ratio (AOR) 4.51; 95% CI 2.39–8.50; p < 0.001). Age 1–14 years (AOR 0.32; p = 0.006) and 15–34 years (AOR 0.35; p = 0.021) were protective compared with infants. Sex, urinary catheterisation, and invasive device presence were not independently associated with HAI.
Table 4.
Multivariate Analysis of Risk Factors for Healthcare-Associated Infection.
Table 4.
Multivariate Analysis of Risk Factors for Healthcare-Associated Infection.
| Variable |
AOR |
95% CI |
p-value |
Interpretation |
| Prolonged ICU stay (>8 days) |
4.51 |
2.39–8.50 |
<0.001 |
Independent risk factor |
| Age 1–14 years |
0.32 |
Not reported |
0.006 |
Protective vs infants |
| Age 15–34 years |
0.35 |
Not reported |
0.021 |
Protective vs infants |
| Sex |
— |
— |
>0.05 |
Not independently associated |
| Urinary catheterisation |
— |
— |
>0.05 |
Not independently associated |
| Invasive device presence |
— |
— |
>0.05 |
Not independently associated |
3.5. Healthcare-Associated Infection and Mortality
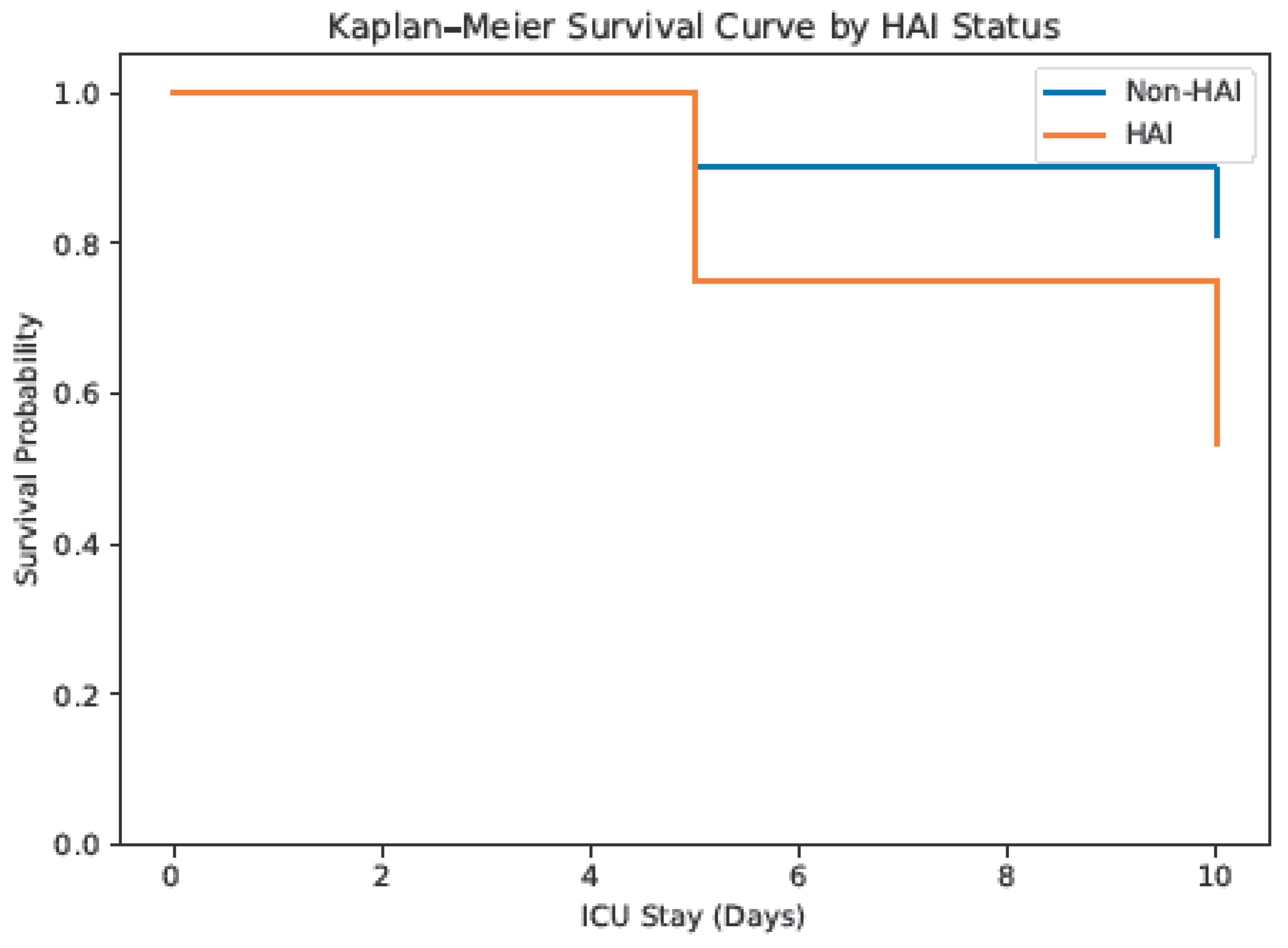
Mortality among patients with HAI was significantly higher than among those without HAI (46.8% vs. 19.0%; χ² p < 0.001). Kaplan–Meier survival analysis demonstrated significantly reduced survival probability in patients who developed HAIs (log-rank p < 0.001). Among infection types, CAUTI and VAP were significantly associated with mortality, whereas CLABSI and SSI were not.
4. Discussion
This study demonstrates a high prevalence of healthcare-associated infections (HAIs) (28.95%) among critically ill patients in a rural tertiary ICU setting at Nelson Mandela Academic Hospital. Ventilator-associated pneumonia (VAP) was the most frequent infection, followed by central line-associated bloodstream infection (CLABSI) and catheter-associated urinary tract infection (CAUTI). Prolonged ICU stay (>8 days) and infant age independently predicted HAI occurrence, and HAIs were significantly associated with increased mortality, particularly among patients with VAP and CAUTI.
4.1. Interpretation in the Context of Previous Evidence
The observed HAI prevalence aligns with pooled estimates from sub-Saharan Africa, which report ICU HAI rates of approximately 28% [
6,
7]. In contrast, high-income countries generally report lower overall ICU HAI prevalence, often around 10%, reflecting stronger infection prevention and control (IPC) systems, structured surveillance networks, and more consistent adherence to device-care bundles [
8]. These disparities highlight the influence of health system infrastructure, staffing ratios, access to consumables, and laboratory capacity on infection rates.
Consistent with global ICU epidemiology, device-associated infections predominated in this cohort. VAP was the most common HAI, reflecting prolonged mechanical ventilation and impaired host defences among critically ill patients [
9]. CAUTI and CLABSI were also frequent, underscoring the central role of invasive devices in HAI pathogenesis. The predominance of Gram-negative organisms—particularly
Acinetobacter baumannii,
Escherichia coli, and
Klebsiella pneumoniae—mirrors findings from other African ICU studies and reinforces global concerns regarding multidrug-resistant Gram-negative pathogens in critical care environments [
10,
11].
A prolonged ICU stay emerged as the strongest independent predictor of HAI, supporting existing evidence that prolonged exposure increases the risk of colonisation and infection, especially in the presence of invasive devices [
6,
12]. Infant age was also independently associated with HAI risk, likely reflecting immunological immaturity and greater dependence on invasive organ support [
7,
13]. Although urinary catheterisation and sex were not independently associated with HAI after adjustment, the biological plausibility of device-related infection remains well established, suggesting that duration and care practices may be more influential than mere device presence.
4.2. Clinical Outcomes and Broader Implications
The strong association between HAI and mortality (46.8% vs. 19.0%) underscores the clinical significance of these infections. VAP and CAUTI were particularly associated with death, consistent with multicentre ICU data demonstrating that device-associated infections increase mortality risk and prolong ICU stay [
9,
12]. These findings reinforce the hypothesis that HAIs are not merely markers of severe illness but active contributors to adverse outcomes in resource-limited ICUs.
From a health systems perspective, the high HAI burden reflects both patient-level vulnerability and structural constraints. Heavy reliance on broad-spectrum antibiotics, as observed in this study, raises concerns about antimicrobial resistance and highlights the need for integrated antimicrobial stewardship. Tailoring empirical therapy to local antibiograms and strengthening microbiological diagnostics are critical steps toward improving outcomes while limiting the selection pressure for resistance.
Reducing HAIs would likely yield multiple benefits: improved patient survival, shorter ICU stays, lower antimicrobial consumption, and better allocation of scarce critical care resources. Evidence-based strategies include:
Implementation of device-care bundles for VAP, CAUTI, and CLABSI, with daily device assessment and prompt removal when clinically feasible.
Continuous surveillance and feedback mechanisms using standardised definitions.
Structured IPC training programs, including paediatric- and neonatal-specific modules.
Strengthened antimicrobial stewardship programs, integrated into routine ICU practice.
Policy-level investment in IPC supplies, staffing, and laboratory capacity.
4.3. Limitations and Future Research
This retrospective, single-centre study cannot establish causality and may be subject to documentation bias. Culture-based diagnostics may underestimate the infection burden, particularly for fastidious organisms or in the presence of prior antibiotic exposure. Additionally, findings may not be fully generalisable to other South African or regional ICUs with different resource profiles.
Future research should prioritise multicentre prospective surveillance using standardised definitions to enhance comparability. Interventional studies evaluating the effectiveness and cost-effectiveness of IPC bundles and antimicrobial stewardship interventions in rural, resource-constrained ICUs are warranted. Integration of molecular resistance profiling may further elucidate transmission dynamics and guide targeted prevention strategies.
5. Conclusions
Healthcare-associated infections represent a substantial burden among ICU patients at Nelson Mandela Academic Hospital, with a prevalence of 28.95% and a strong association with mortality. Prolonged ICU stay and infant age were independent predictors, and device-associated infections—particularly VAP—predominated. Strengthening infection prevention and control practices, implementing structured surveillance systems, and optimising antimicrobial stewardship are essential to reduce HAI incidence, improve patient outcomes, and enhance the utilisation of critical care resources in resource-limited settings.
Author Contributions
Conceptualization, D.A.; methodology, D.A.; formal analysis, D.A. and S.D.; investigation, D.A. and S.D.; resources, D.A.; data curation, D.A. and S.D.; writing—original draft preparation, D.A.; writing—review and editing, D.A.; visualization, D.A. and S.D.; supervision, D.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Ethics Committee of Walter Sisulu University Health Sciences Research Ethics Committee on 26 May 2025 (approval number: 058/2025) and the Eastern Cape Department of Health. Institutional permission was granted by hospital management.
Informed Consent Statement
Patient consent was waived because this study was retrospective and relied solely on routinely collected patient management and medical record data. No direct patient contact occurred during the conduct of the research.
Data Availability Statement
The datasets generated and/or analysed during this study are not publicly available because they form part of routine patient management records in the ICU of Nelson Mandela Academic Hospital. Public disclosure would constitute an ethical breach of patient confidentiality. Data may, however, be made available upon reasonable request, subject to approval from the institutional gatekeeper and relevant regulatory authorities.
Acknowledgments
The authors would like to acknowledge the staff of the Intensive Care Units at Nelson Mandela Academic Hospital for their support and cooperation during data collection. The Head of Department, colleagues, and support staff of the Department of Laboratory Medicine and Pathology are also gratefully acknowledged for their assistance.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript
| HAIs |
Healthcare-associated infections |
| ICU |
intensive care unit |
| LMIC |
low- and middle-income countries |
| IPC |
Infection prevention and control |
| WHO |
World Health Organisation |
| VAP |
ventilator-associated pneumonia |
| CLABSI |
central line-associated bloodstream infection |
| CAUTI |
catheter-associated urinary tract infection |
| AMR |
antimicrobial resistance |
| CLSI |
Clinical and Laboratory Standards Institute |
| SD |
standard deviation |
| IQR |
interquartile ranges |
| aOR |
Adjusted odds ratios |
| SSI |
Surgical site infections |
References
- World Health Organization. Global Report on the Burden of Endemic Health Care-Associated Infection Worldwide; WHO: Geneva, 2022. [Google Scholar]
- Centers for Disease Control and Prevention. National and State Healthcare-Associated Infections Progress Report; CDC: Atlanta, 2023. [Google Scholar]
- Adal, O; Tsehay, YT; Ayenew, B; et al. The burden and predictors of hospital-acquired infection in intensive care units across sub-Sahara Africa: systematic review and meta-analysis. BMC Infect Dis. 2025, 25, 634. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Antibiotic Resistance Surveillance Report 2025; WHO: Geneva, 2025. [Google Scholar]
- Dramowski, A; Whitelaw, A; et al. Healthcare-associated infections in South Africa: prevalence and implications. S Afr Med J. 2024; Published online ahead of print. [Google Scholar]
- Muhindo, R; et al. Healthcare-associated infections in Sub-Saharan African ICUs: a systematic review. BMC Infect Dis. 2025, 25, 38. [Google Scholar]
- Chigangaidze, R; et al. ICU-acquired infections among neonates and infants in resource-limited settings. BMC Infect Dis. 2025, 25, 110. [Google Scholar]
- European Centre for Disease Prevention and Control. Point prevalence survey of healthcare-associated infections in European acute care hospitals. 2023. [Google Scholar]
- Silva, E; et al. Device-associated infections in Brazilian ICUs: incidence and mortality. Crit Care 2025, 29, 203. [Google Scholar]
- Adegbite, BR; et al. Microbiological profile of ICU-acquired infections in African hospitals. One Health Bull. 2024, 12, 51–62. [Google Scholar]
- Dramowski, A; et al. Multidrug-resistant Gram-negative pathogens in African ICUs. BMC Infect Dis. 2022, 22, 387. [Google Scholar]
- Zingg, W; et al. Risk factors for ICU-acquired infections: a multicenter cohort study. Crit Care 2024, 28, 105. [Google Scholar]
- Rosenthal, VD; et al. Device-associated infection risk in pediatric and neonatal ICUs. BMC Infect Dis. 2022, 22, 122. [Google Scholar]
- Ghosh, R; et al. Effectiveness of infection prevention and control bundles in ICUs. RDS J. 2024, 5, 1783–1795. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).