Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
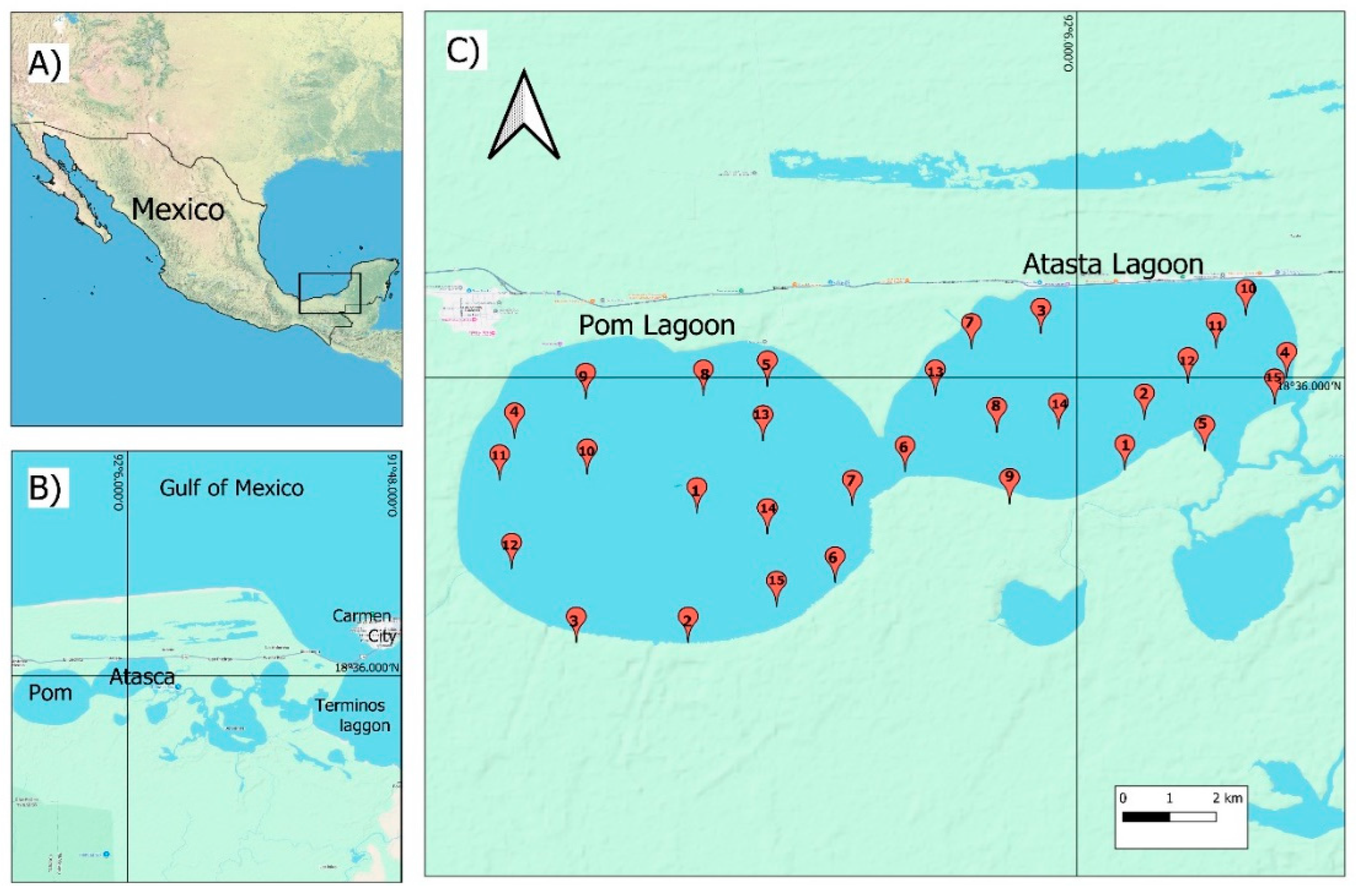
2.1. Study Area and Sampling
2.2. Sediment Analysis
2.3. Metal Analysis
2.4. Risk Assessment
2.5. Biota-Sediment Accumulation Factor (BSAF)
2.6. Statistical Analysis
3. Results and Discussions
3.1. Sediment Texture and Organic Content
3.2. Heavy Metal Concentrations in Sediment and Bioaccumulation in Mollusk
| Location | Ni (µg g-1) | V (µg g-1) | Rate Ni /V | Reference |
|---|---|---|---|---|
| Jinjiang River, China | 0.51-2.85 Media: 1.70 |
0.53 -1.91 Media: 0.97 |
1.75 | [27] |
| Terminos Lagoon, México | 13.5 – 164 Media: 88.75 |
9 – 61 Media: 35 |
2.53 | [25] |
| Terminos Lagoon, México | 16.29 – 56.45 Media: 20.08 |
Nd | 0 | [30] |
| Campeche Coast, Mexico | 0.56 -76.9 Media: 38.73 |
15.6 – 117.5 Media: 66.55 |
0.58 | [31] |
| Campeche and Tabasco coast, Mexico | 1.54 – 211 Media: 106.27 |
17.7 -59.6 Media: 38.65 |
2.65 | [29] |
| Atasta Lagoon, Mexico | 59.56 – 78.39 Media: 68.97 |
32.15 – 40.67 Media: 36.41 |
1.89 | This study |
| Pom Lagoon, Mexico | 61.37 – 73.43 Media: 67.4 |
30.70 – 33.1 Media: 31.9 |
1.05 | This study |
3.3. Heavy Metal Toxicity Assessment
| Stations | Dry season | |||||
| As | Cr | Ni | Pb | V | Cd | |
| A1 | 22.96(1.53) | 165.24(1.84) | 68.89(1.21) | 10.48(0.15) | 37.84(1.15) | 5.11 |
| A2 | 23.77(1.58) | 121.96(1.36) | 64.58(1.13) | 10.58(0.15) | 33.41(1.29) | 4.56 |
| A3 | 5.79(0.39) | 118.41(1.32) | 62.8(1.10) | 7.59(0.11) | 34.64(0.69) | 3.26 |
| A4 | 15.64(1.04) | 116.24(1.29) | 56.67(0.99) | 10.76(0.15) | 33.76(0.52) | 3.82 |
| A5 | 0 | 133.17(1.48) | 57.68(1.01) | 8.56(0.12) | 38.81(1.57) | 3.01 |
| A6 | 13.97(0.93) | 114.13(1.27) | 55.97(0.98) | 12.17(0.17) | 34.82(1.16) | 3.71 |
| A7 | 2.69(0.18) | 128.24(1.42) | 73.34(129) | 11.16(0.16) | 36.87(0.0) | 3.42 |
| A8 | 13.09(0.87) | 112.46(1.25) | 63.76(1.12) | 13.29(0.19) | 33.82(1.07) | 3.77 |
| A9 | 10.69(0.71) | 137.15(1.52) | 60.17(1.06) | 8.16(0.12) | 35.77(0.80) | 3.77 |
| A10 | 26.95(1.8) | 108.51(1.21) | 58.16(1.02) | 7.9(0.11) | 32.39(0.22) | 4.46 |
| A11 | 38.77(2.58) | 122.9(1.37) | 49.36(0.87) | 11.09(0.16) | 31.27(2.81) | 5.29 |
| A12 | 9.15(0.61) | 104.92(1.17) | 48.43(0.85) | 9.55(0.14) | 28.84(1.90) | 3.05 |
| A13 | 12.38(0.83) | 113.02(1.26) | 65.87(1.16) | 9.11(0.13) | 35.26(2.29) | 3.72 |
| A14 | 8.18(0.55) | 116.07(1.29) | 47.27(0.83) | 6.68(0.10) | 27.32(0.0) | 3.04 |
| A15 | 18.9(1.26) | 123.35(1.37) | 60.56(1.06) | 4.94(0.07) | 37.89(0.63) | 4.15 |
| P1 | 22.75(1.52) | 131.15(1.46) | 72.95(1.28) | 9.28(0.13) | 40.04(1.34) | 4.79 |
| P2 | 29.27(1.95) | 212.42(2.36) | 109.87(1.93) | 6.23(0.09) | 54.19(1.37) | 6.88 |
| P3 | 2.08(0.14) | 110.5(1.23) | 47.43(0.83) | 10.4(0.15) | 26.73(0.26) | 2.62 |
| P4 | 11.38(0.76) | 482.4(5.36) | 34.42(0.60) | 5.15(0.07) | 36.27(1.13) | 7.16 |
| P5 | 18.3(1.22) | 120.65(1.34) | 73.77(1.29) | 10.78(0.15) | 39.66(1.40) | 4.41 |
| P6 | 5.32(0.35) | 110.96(1.23) | 50.35(0.88) | 6.7(0.10) | 30.45(2.73) | 2.87 |
| P7 | 24.76(1.65) | 93.08(1.03) | 50.91(0.89) | 8.25(0.12) | 29.14(1.07) | 3.99 |
| P8 | 21.9(1.46) | 119.84(1.33) | 78.47(1.38) | 3.31(0.05) | 40.4(0.77) | 4.62 |
| P9 | 12.49(0.83) | 83.86(0.93) | 31.32(0.55) | 8.23(0.12) | 28.35(0.53) | 2.72 |
| P10 | 14.14(0.94) | 94.05(1.05) | 70.81(1.24) | 3.86(0.06) | 32.93(0.81) | 3.62 |
| P11 | 4.85(0.32) | 55.01(0.61) | 22.31(0.39) | 4.66(0.07) | 13.78(1.34) | 1.53 |
| P12 | 18.76(1.25) | 62.18(0.69) | 16.37(0.29) | 1.6(0.02) | 14.07(0.94) | 2.39 |
| P13 | 21.8(1.45) | 101.86(1.13) | 59.16(1.04) | 7.13(0.10) | 33.59(1.33) | 4.06 |
| P14 | 12.88(0.86) | 103.94(1.15) | 70.15(1.23) | 11.4(0.16) | 33.59(0.10) | 3.75 |
| P15 | 17.4(1.16) | 131.96(1.47) | 132.35(2.32) | 11.21(0.16) | 43.02(0.79) | 5.54 |
| Mean (µg g-1) |
15.4 | 128.3 | 60.5 | 8.3 | 33.6 | 3.97 |
| Min | 0.0 | 55.0 | 16.4 | 1.6 | 13.8 | 1.53 |
| Max | 38.8 | 482.4 | 132.4 | 13.3 | 54.2 | 7.16 |
| Stations | Rainy season | |||||
| As | Cr | Ni | Pb | V | Cd | |
| A1 | 17.31(1.15) | 108.63(1.21) | 61.54(1.08) | 11.97(0.17) | 27.8(0.28) | 3.89 |
| A2 | 19.31(1.29) | 117.35(1.30) | 89.9(1.58) | 9.12(0.13) | 37.65(0.38) | 4.68 |
| A3 | 10.29(0.69) | 165.72(1.84) | 65.76(1.15) | 11.27(0.16) | 29.89(0.30) | 4.14 |
| A4 | 7.83(0.52) | 109.2(1.21) | 71.54(1.26) | 7.83(0.11) | 32.2(0.33) | 3.43 |
| A5 | 23.48(1.57) | 104.05(1.16) | 70.83(1.24) | 4.58(0.07) | 29.81(0.30) | 4.33 |
| A6 | 17.37(1.16) | 103.49(1.15) | 70.91(1.24) | 5.82(0.08) | 34.37(0.35) | 3.98 |
| A7 | 0 | 126.13(1.40) | 61.74(1.08) | 8.12(0.12) | 32.88(0.33) | 2.93 |
| A8 | 15.98(1.07) | 122.49(1.36) | 114.69(2.01) | 8.78(0.13) | 36.79(0.37) | 4.94 |
| A9 | 12(0.80) | 115.5(1.28) | 80.72(1.42) | 1.42(0.02) | 36.96(0.37) | 3.89 |
| A10 | 3.31(0.22) | 60.28(0.67) | 22.01(0.39) | 4.77(0.07) | 14.51(0.15) | 1.49 |
| A11 | 42.16(2.81) | 135.02(1.50) | 86.18(1.51) | 4.23(0.06) | 29.97(0.30) | 6.19 |
| A12 | 28.48(1.90) | 125.1(1.39) | 71.64(1.26) | 16.59(0.24) | 36.37(0.37) | 5.15 |
| A13 | 34.34(2.29) | 111.34(1.24) | 72.25(1.27) | 10.79(0.15) | 36.03(0.36) | 5.31 |
| A14 | 0 | 107.77(1.20) | 90.92(1.60) | 3.49(0.05) | 32.92(0.33) | 3.17 |
| A15 | 9.38(0.63) | 111.28(1.24) | 80.34(1.41) | 9.88(0.14) | 34.13(0.34) | 3.76 |
| P1 | 20.16(1.34) | 156.13(1.73) | 147.42(2.59) | 6.56(0.09) | 54.42(0.55) | 6.31 |
| P2 | 20.62(1.37) | 129.34(1.44) | 82.5(1.45) | 1.83(0.03) | 34.7(0.35) | 4.64 |
| P3 | 3.87(0.26) | 81.82(0.91) | 32.34(0.57) | 1.94(0.03) | 17.14(0.17) | 1.94 |
| P4 | 16.97(1.13) | 52.63(0.58) | 13.86(0.24) | 2.91(0.04) | 12.56(0.13) | 2.13 |
| P5 | 20.97(1.40) | 106.43(1.18) | 69.39(1.22) | 17.87(0.26) | 36.54(0.37) | 4.42 |
| P6 | 40.95(2.73) | 85.99(0.96) | 60.58(1.06) | 10.46(0.15) | 26.5(0.27) | 5.17 |
| P7 | 16.03(1.07) | 119.36(1.33) | 81.65(1.43) | 17.72(0.25) | 40.38(0.41) | 4.49 |
| P8 | 11.62(0.77) | 126.69(1.41) | 77.14(1.35) | 19.36(0.28) | 44.28(0.45) | 4.26 |
| P9 | 7.98(0.53) | 142.3(1.58) | 17.45(0.31) | 11.57(0.17) | 18.45(0.19) | 2.77 |
| P10 | 12.2(0.81) | 205.44(2.28) | 62.48(1.10) | 14.14(0.20) | 40.0(0.40) | 4.80 |
| P11 | 20.1(1.34) | 97.63(1.08) | 55.93(0.98) | 9.36(0.13) | 29.26(0.30) | 3.84 |
| P12 | 14.14(0.94) | 84.54(0.94) | 25.94(0.46) | 4.68(0.07) | 19.11(0.19) | 2.60 |
| P13 | 19.94(1.33) | 128.74(1.43) | 77.18(1.35) | 17.85(0.26) | 38.69(0.39) | 4.76 |
| P14 | 1.48(1.10) | 99.98(1.11) | 67.11(1.18) | 14.8(0.21) | 33.16(0.33) | 2.93 |
| P15 | 11.81(0.79) | 109.61(1.22) | 63.24(1.11) | 10.42(0.15) | 32.86 | 3.60 |
| Mean (µg g-1) |
16.0 | 115.0 | 68.2 | 9.3 | 32.0 | 3.99 |
| Min | 0.0 | 52.6 | 13.9 | 1.4 | 12.6 | 1.49 |
| Max | 42.2 | 205.4 | 147.4 | 19.4 | 54.4 | 6.31 |
| Station | Northerly season | |||||
| As | Cr | Ni | Pb | V | Cd | |
| A1 | 12.39(0.83) | 146.42(1.63) | 59.43(1.04) | 15.98(0.23) | 35.06(0.35) | 4.08 |
| A2 | 0 | 133.12(1.48) | 72.55(1.27) | 16.13(0.23) | 41.67(0.42) | 3.40 |
| A3 | 20.73(1.38) | 201.71(2.24) | 86.77(1.52) | 20.03(0.29) | 50.18(0.51) | 5.94 |
| A4 | 6.32(0.42) | 127.02(1.41) | 69.19(1.21) | 18.95(0.27) | 40.76(0.41) | 3.73 |
| A5 | 17.35(1.16) | 158.62(1.76) | 76.4(1.34) | 18.34(0.26) | 41.9(0.42) | 4.94 |
| A6 | 11.86(0.79) | 112.59(1.25) | 69.45(1.22) | 18.23(0.26) | 37.96(0.38) | 3.90 |
| A7 | 10.65(0.71) | 124.7(1.39) | 83.5(1.46) | 20.21(0.29) | 42.49(0.43) | 4.28 |
| A8 | 0 | 122.71(1.36) | 82.04(1.44) | 20.29(0.29) | 39.78(0.40) | 3.49 |
| A9 | 1.09(0.07) | 136.22(1.51) | 73.17(1.28) | 16.48(0.24) | 39.22(0.40) | 3.50 |
| A10 | 14.72(0.98) | 168.71(1.87) | 84.55(1.48) | 19.66(0.28) | 46.72(0.47) | 5.09 |
| A11 | 35.67(2.38) | 154.91(1.72) | 84.63(1.48) | 15.61(0.22) | 38.1(0.38) | 6.19 |
| A12 | 5.67(0.38) | 144.43(1.60) | 69.18(1.21) | 13.32(0.19) | 34.59(0.35) | 3.74 |
| A13 | 25.57(1.70) | 124.25(1.38) | 91.39(1.60) | 19.38(0.28) | 41.55(0.42) | 5.39 |
| A14 | 6.55(0.44) | 124.85(1.39) | 87.17(1.53) | 17.35(0.25) | 41.35(0.42) | 4.02 |
| A15 | 32.26(2.15) | 122.43(1.36) | 86.52(1.52) | 16.57(0.24) | 38.77(0.39) | 5.66 |
| P1 | 19.66(1.31) | 131.35(1.46) | 126.49(2.22) | 17.77(0.25) | 47.86(0.48) | 5.73 |
| P2 | 9.07(0.60) | 156.41(1.74) | 103.65(1.82) | 7.1(0.10) | 39.64(0.40) | 4.66 |
| P3 | 12.87(0.86) | 95.27(1.06) | 52.47(0.92) | 4.29(0.06) | 23.04(0.23) | 3.13 |
| P4 | 0 | 272.74(3.03) | 28.33(0.50) | 7.9(0.11) | 28.72(0.29) | 3.93 |
| P5 | 10.23(0.68) | 111.64(1.24) | 106.38(1.87) | 16.98(0.24) | 38.44(0.39) | 4.42 |
| P6 | 27.44(1.83) | 96.53(1.07) | 68.0(1.19) | 11.15(0.16) | 29.12(0.29) | 4.55 |
| P7 | 25.26(1.68) | 104.17(1.16) | 100.66(1.77) | 13.95(0.20) | 32.68(0.33) | 5.14 |
| P8 | 6.85(0.46) | 124.18(1.38) | 89.44(1.57) | 18.53(0.26) | 43.71(0.44) | 4.11 |
| P9 | 0 | 104.86(1.17) | 56.77(1.0) | 17.87(0.26) | 23.4(0.24) | 2.65 |
| P10 | 9.34(0.62) | 84.46(0.94) | 51.17(0.90) | 10.23(0.15) | 26.13(0.26) | 2.87 |
| P11 | 0 | 101.06(1.12) | 27.51(0.48) | 4.34(0.06) | 15.64(0.16) | 1.83 |
| P12 | 11.29(0.75) | 100.2(1.11) | 33.76(0.59) | 2.18(0.03) | 15.15(0.15) | 2.64 |
| P13 | 20.11(1.34) | 96.26(1.07) | 69.25(1.21) | 15.61(0.22) | 30.65(0.31) | 4.16 |
| P14 | 12.74(0.85) | 121.89(1.35) | 101.95(1.79) | 16.73(0.24) | 36.96(0.37) | 4.60 |
| P15 | 16.8(1.12) | 107.32(1.19) | 85.66(1.50) | 10.68(0.15) | 29.43(0.3) | 4.27 |
| Mean (µg g-1) |
12.7 | 130.4 | 75.9 | 14.7 | 35.7 | 4.20 |
| Min | 0.0 | 84.5 | 27.5 | 2.2 | 15.2 | 1.83 |
| Max | 35.7 | 272.7 | 126.5 | 20.3 | 50.2 | 6.19 |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AEI | Adverse effects index |
| TRI | toxic risk index |
| ISQG-TEL | Reference values for the quality of marine sediments and threshold effect |
| BSAF | Biota-sediment accumulation factor |
References
- Bakker, A.K.; Dutton, J.; Sdafani, M.; Santangelo, N. Environmental exposure of Atlantic horseshoe crab (Limulus polyphemus) early life stages to essential trace elements. Science of the Total Environment 572 2016, 804–812. [Google Scholar] [CrossRef]
- Saher, N.U.; Siddiqui, A.S. Occurrence of heavy metals in sediment and their bioaccumulation in sentinel crab (Macrophthalmus depressus) from highly impacted coastal areas. Chemosphere 221 2019, 89–98. [Google Scholar] [CrossRef]
- Omar, C.H.; Leticia, R.H.; Andrew, C.B.; Arturo, C.E.; Ian, C.W.; Hector, H.H. Historical trace element accumulation in marine sediment from the Tamaulipas shelf, Gulf of Mexico: An assessment of natural vs anthropogenic inputs. Science of the Total Environment 622–623 2018, 325–336. [Google Scholar] [CrossRef]
- Jahan, S.; Strezov, V. Comparison of pollution indices for the assessment of heavy metals in sediments of seaports of NSW, Australia. Marine Pollution Bulletin 128 2018, 295–306. [Google Scholar] [CrossRef]
- Onder, S.; Dursun, S. Air borne heavy metal pollution of Cedrus libani (A. Rich.) in te city centre of Konya (Turkey). Atmos. Environ 2006, 40, 1122–1133. [Google Scholar] [CrossRef]
- Das, P.; Samantaray, S.; Rout, G.R. Studies on cadmium toxicity in plants: A review. Environmental Pollution 98 1997, 29–36. [Google Scholar] [CrossRef]
- Pinheiro, M.A.A.; Silva, P.P.G.; Duarte, L.F.A.; Almeda, A.A.; Zanotto, F.P. Accumulation of six metals in the mangrove crab Ucides cordatus (Crustacea: Ucididae) and its food source, the red mangrove Rhizophora mangle (Angiospermae: Rhizophoraceae). Ecotoxicology and Environmental Safety 81 2012, 114–121. [Google Scholar] [CrossRef]
- Fan, W.; Xu, Z.; Wang, W.X. Metal pollution in a contaminated bay: Relationship between metal geochemical fractionation in sediment and accumulation in a polychaete. Environmental Pollution 197 2014, 50–57. [Google Scholar] [CrossRef]
- Alvaro, N.V.; Neto, A.I.; Couto, R.P.; Azevedo, J.M.N.; Rodrigues, A. S. Crabs tell the difference: Relating trace metal content with land use and landscape attributes. Chemosphere 144 2016, 1377–1383. [Google Scholar] [CrossRef]
- Ruiz-Marin, A.; Campos-García, S.; Zavala-Loria, J.; Canedo-López, Y. Hydrological aspects of the lagoons of Atasta and Pom, Mexico. Tropical and Subtropical Agroecosystems 10 2009, 63–74. [Google Scholar]
- Romero, C.M.; Ucán, C.A.; Peralta, A.S.; Reyes, J.T.; López, Y.C.; Quiroz, V.C.; Marin, A R. Health risk assessment of heavy metals: Cu, Cd, Pb, Ni and Hg, in catfish Ariopsis felis in southern Mexico. Iranian Journal of Toxicology 2022, 16(3), 163–174. [Google Scholar] [CrossRef]
- De la Lanza-Espino, G.; Ramírez-García, P.; Thomas, F.; Alcántara, A.R. La vegetación de manglar de la Laguna de Términos, Campeche: Evaluación preliminar a través de imágenes LandSat. Hidrobiológica 3 1993, 29–40. [Google Scholar]
- Yañez-Arancibia, A.; Day, J.W. Ecological characterization of the Términos Lagoon. In Ecología de los ecosistemas costeros en el sur del Golfo de México: La región de la laguna de Términos 1988; Yañez-Arancibia., A., Day, J. W., Eds.; Universidad Nacional Autónoma de México / Organización de Estados Americanos, 1988; pp. 1–27. [Google Scholar]
- Norma Oficial Mexicana NOM-021-RECNAT-2000; Fertility specifications, salinity and soil classification. 2000.
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Science 1934. 37 29–38. [CrossRef]
- Long, E.R.; MacDonald, D.D.; Smith, S.L.; Calder, F.D. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environmental Management 1995, 19(1), 81–97. [Google Scholar] [CrossRef]
- Zhang, C.; Bai, J.; Zhao, Q.; Lu, Q.; Jia, J.; Wen, X. Heavy metals in wetland soils along a wetland-forming chronosequence in the Yellow River Delta of China: Levels, sources and toxic risks. Ecological Indicators 69 2016, 331–339. [Google Scholar] [CrossRef]
- Gao, L.; Wang, Z.; Li, S.; Chen, J. Bioavailability and toxicity of trace metals (Cd, Cr, Cu, Ni, and Zn) in sediment cores from the Shima River, South China. Chemosphere 192 2018, 31–42. [Google Scholar] [CrossRef]
- Håkanson, L. An ecological risk index for aquatic pollution control. a sedimentological approach. Water Res 1980, 14, 975e1001. [Google Scholar] [CrossRef]
- Gupta, S.K.; Chabukdhara, M.; Kumar, P.; Singh, J.; Bux, F. Evaluation of ecological risk of metal contamination in River Gomti, India: A biomonitoring approach. Ecotoxicology and Environmental Safety 110 2014, 49–55. [Google Scholar] [CrossRef]
- Flemming, B.W. A revised textural classification of gravel-free muddy sediments on the basis of ternary diagrams. Continental Shelf Research 20 2000, 1125–1137. [Google Scholar] [CrossRef]
- Wedepohl, K.H. The composition of the continental crust. Geochimica et Cosmochimica Acta 1995, 59(7), 1217–1232. [Google Scholar] [CrossRef]
- Osuji, L.C.; Onojake, C.M. Trace heavy metals associated with crude oil: A case study of Ebocha-8 oil-spill-polluted site in the Niger Delta, Nigeria. Chemical Biodiversity 2004, 1(11), 1708–1715. [Google Scholar] [CrossRef]
- Magallanes-Ordoñez, V.R.; Marmolejo-Rodríguez, A.J.; Rodríguez-Figueroa, G.M.; Sánchez-González, A.; Aguiñiga-García, S.; Arreguín-Sánchez, F.; Zetina-Rejón, M.; Tripp-Valdez, A.; Romo-Ríos, J.A. Characterization of lithogenic and biogenic zones and natural enrichment of nickel in sediments of the Términos Lagoon, Campeche, México. Estuarine, Coastal and Shelf Science 156 2015, 116–123. [Google Scholar] [CrossRef]
- Barwise, A.J.G. Role of nickel and vanadium in petroleum classification. Energy Fuels 1990, 4(6), 647–652. [Google Scholar] [CrossRef]
- Liu, X.; Jiang, J.; Yan, Y.; Dai, Y.; Deng, B.; Ding, S.; Su, S.; Sun, W.; Li, Z.; Gan, Z. Distribution and risk assessment of metals in water, sediment, and wild fish from Jinjiang River in Chengdu, China. Chemosphere 196 2018, 45–52. [Google Scholar] [CrossRef]
- Yakubov, M. R.; Milordov, D. V.; Yakubova, S. G.; Borisov, D. N.; Ivanov, V. T.; Sinyashin, K. O. Concentrations of vanadium and nickel and their ratio in heavy oil asphaltenes. Petroleum Chemistry 2016, 56(1), 16–20. [Google Scholar] [CrossRef]
- Vázquez, F.G.; Sharma, V.K.; Pérez-Cruz, L. Concentrations of elements and metals in sediments of the southeastern Gulf of Mexico. Environmental Geology 42 2002, 41–46. [Google Scholar] [CrossRef]
- Canedo-López, Y.; Ruiz-Marin, A.; Chi-Ayil, J.C. Diagnóstico de la contaminación por metales pesados en sedimento superficial de la laguna de Términos, Campeche: Una aproximación estadística. Revista AIDIS de Ingeniería y Ciencias Ambientales 2014, 7(2), 95–104. Available online: https://repositorio.unam.mx/contenidos/40593.
- Macias-Zamora, J.V.; Villaescusa-Celaya, J.A.; Muñoz-Barbosa, A.; Gold-Bouchot, G. Trace metals in sediment cores from the Campeche shelf, Gulf of Mexico. Environmental Pollution 1999, 104(1), 69–77. [Google Scholar] [CrossRef]
- Hao, Z.; Chen, L.; Wang, C.; Zou, X.; Zheng, F.; Feng, W.; Zhang, D.; Peng, L. Heavy metal distribution and bioaccumulation ability in marine organisms from coastal regions of Hainan and Zhoushan, China. Chemosphere 226 2019, 340–350. [Google Scholar] [CrossRef]
- Zang, L.; Liu, J. In situ relationships between spatial-temporal variations in potential ecological risk indexes for metals and the short-term effects on periphyton in a macrophyte-dominated lake. Ecotoxicology 23 2014, 553–566. [Google Scholar] [CrossRef]
- Norville, W. Spatial distribution of heavy metals in sediments from the Gulf of Paria, Trinidad. Revista de Biología Tropical 53 2005, 33–40. [Google Scholar]
- Zhang, S.; Fu, K.; Gao, S.; Liang, B.; Lu, J.; Fu, G. Bioaccumulation of Heavy Metals in the Water, Sediment, and Organisms from the Sea Ranching Areas of Haizhou Bay in China. Water 2023, 15(12), 2218. [Google Scholar] [CrossRef]
- Oliveira, L.F; Martinez, C.B. Chromium accumulation in the Asian clam, Corbicula fluminea (Müller, 1774), as an indicative of landfill leachate contamination. Bull Environ Contam Toxicol. 2014, 93(2), 149–53. [Google Scholar] [CrossRef] [PubMed]
- Pasinszki, T.; Prasad, S.S.; Krebsz, M. Quantitative determination of heavy metal contaminants in edible soft tissue of clams, mussels, and oysters. Environ Monit Asses 2023, 195, 1066. [Google Scholar] [CrossRef]
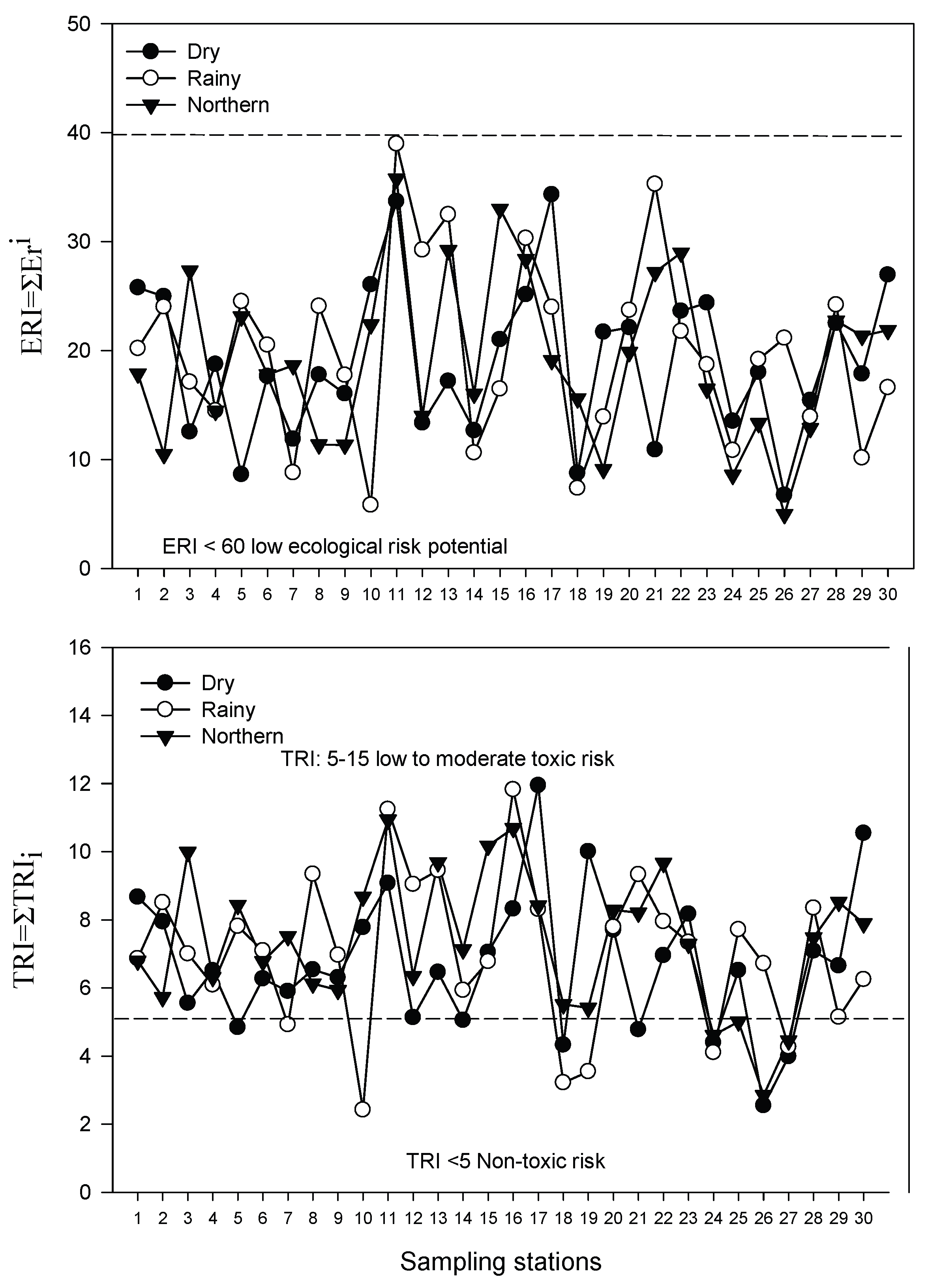
| TRI | Toxic risk | Cd | Degree of contamination |
| <5 | No | Cd < 8 | Low |
| 5-10 | Low | 8 ≤ Cd < 16 | Moderate |
| 10-15 | Moderate | 16 ≤ Cd < 32 | Considerable |
| 15-20 | Considerable | Cd ≥ 32 | Very high |
| >20 | Very high |
| Climate season | Sand (%) | Silt (%) | Clay (%) | OM (%) | TOC (%) |
| Dry | 76.47±8.89a | 19.73±8.55a | 3.79±1.56a | 3.94±2.13a | 2.28±1.24a |
| Rains | 73.96±9.81a | 22.73±10.02a | 3.31±1.65a | 2.97±2.21a | 1.72±1.28a |
| Northerly | 67.27±16.74b | 29.59±16.54b | 3.14±1.14a | 3.03±2.33a | 1.76±1.35a |
| Metal (µg g-1) |
Clam | Sediment Climatic seasons |
UC | ||||
|
Rangia cuneata |
SBAF | Dry | Rains | Northern | ISQG-TEL | Wedepohl [22] | |
| As | < 0.3 | 0.14 | 15.4± 8.9a | 16.0± 10.6 a | 12.7± 9.8 a | 5.9 | 2.0 |
| Cr | 9.48± 3.36 | 0.37 | 128.3± 72.5 b | 115.0± 29.5 b | 130.4± 37 b | 52.3 | 35 |
| Ni | 21.36± 6.55 | 1.51 | 60.5± 22.4 c | 68.2± 27.4 c | 75.9± 22.8 d | 15.9 | 18.6 |
| Pb | <1 | 0.65 | 8.3± 2.9 e | 9.3± 5.3 e | 14.7± 5.2 f | 30.2 | 17 |
| V | nd | - | 33.6± 7.6 g | 32.0± 8.9 g | 35.7± 8.8 g | - | 53 |
| Cd | 0.44 | - | nd | Nd | Nd | 0.68 | 0.102 |
| Hg | 0.51 | - | nd | Nd | Nd | - | - |
| 1As | 1Cr | 1Ni | 1Pb | 1V | 2As | 2Cr | 2Ni | 2Pb | 2V | 3As | 3Cr | 3Ni | 3Pb | 3V | ||
| Dry | 1As | 1.0 | ||||||||||||||
| 1Cr | 0.03 | 1.0 | ||||||||||||||
| 1Ni | 0.26 | 0.06 | 1.0 | |||||||||||||
| 1Pb | 0.00 | -0.09 | 0.32 | 1.0 | ||||||||||||
| 1V | 0.28 | 0.39 | 0.82 | 0.27 | 1.0 | |||||||||||
| Rain | 2As | 0.23 | 0.04 | -0.07 | 0.03 | 0.03 | 1.0 | |||||||||
| 2Cr | 0.12 | -0.33 | 0.28 | -0.01 | 0.21 | 0.04 | 1.0 | |||||||||
| 2Ni | 0.24 | -0.22 | 0.38 | 0.31 | 0.35 | 0.23 | 0.47 | 1.0 | ||||||||
| 2Pb | 0.09 | -0.32 | 0.14 | -0.02 | 0.01 | 0.08 | 0.34 | 0.11 | 1.0 | |||||||
| 2V | 0.21 | -0.30 | 0.45 | 0.14 | 0.37 | 0.16 | 0.62 | 0.88 | 0.42 | 1.0 | ||||||
| Northerly | 3As | 0.29 | -0.19 | 0.14 | 0.02 | 0.13 | 0.40 | 0.09 | 0.20 | 0.10 | 0.13 | 1.0 | ||||
| 3Cr | 0.05 | 0.80 | -0.04 | -0.03 | 0.30 | 0.04 | -0.25 | -0.18 | -0.34 | -0.33 | -0.10 | 1.0 | ||||
| 3Ni | 0.34 | -0.18 | 0.60 | 0.38 | 0.61 | 0.01 | 0.29 | 0.67 | 0.20 | 0.63 | 0.40 | -0.06 | 1.0 | |||
| 3Pb | 0.06 | -0.15 | 0.21 | 0.48 | 0.37 | -0.11 | 0.25 | 0.40 | 0.23 | 0.36 | 0.13 | 0.11 | 0.58 | 1.0 | ||
| 3V | 0.19 | 0.07 | 0.44 | 0.37 | 0.62 | -0.06 | 0.20 | 0.54 | -0.02 | 0.43 | 0.20 | 0.37 | 0.74 | 0.80 | 1.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





