1. Introduction
Soils are a central component of forest ecosystems, regulating biological activity, nutrient cycling and biogeochemical processes. In forested landscapes, the physicochemical and biochemical properties of soils are shaped by the complex interplay of climate, parent material, topography and vegetation, resulting in pronounced spatial heterogeneity even under relatively uniform environmental conditions [
1]. Although climatic conditions and geological substrates determine the general framework for soil development, vegetation plays a pivotal role in regulating soil physicochemical and biochemical processes by controlling the input of organic matter, nutrient cycling and the conditions of microbial habitats [
2,
3]. Elucidating the drivers of these dynamics is essential for understanding soil processes and predicting how ecosystems will respond to environmental change.
Forest soils represent dynamic systems in which litter quantity and quality, root exudation patterns, nutrient uptake strategies, and belowground biomass allocation vary substantially among tree species [
4]. Especially among biotic drivers, tree species identity influences soil organic carbon accumulation, nutrient availability, and microbial activity, thereby shaping soil biochemical functioning [
5,
6,
7]. Differences in litter chemistry, decomposition rates, and root exudation can modify soil organic carbon dynamics, nutrient availability, and microbial activity, thereby exerting long-term effects on soil development and functioning [
8,
9]. Previous studies have demonstrated that differences in tree species composition can lead to marked variations in soil texture-associated carbon stabilization, microbial biomass, and respiration rates, even under similar climatic conditions [
10,
11,
12]. Such findings highlight vegetation as an environmental filter that selectively modifies soil processes rather than merely responding to pre-existing edaphic conditions.
Soil physicochemical characteristics such as texture, pH, and electrical conductivity are widely regarded as the primary determinants of microbial community structure and activity [
13,
14]. These properties influence the conversion of vegetation-driven inputs into stable soil organic matter or rapid mineralization [
6,
15]. Biochemical indicators as proxies of microbial functioning, including microbial biomass carbon (C
mic), microbial biomass nitrogen (N
mic), and microbial basal respiration (MBR), provide sensitive measures of soil biological activity and reflect short-term responses to changes in substrate availability and environmental conditions [
16]. Variations in these indicators across forest types have been linked to differences in litter chemistry, decomposition rates, and microbial efficiency. However, it is challenging to disentangle vegetation effects from confounding factors such as climate, parent material, and land-use history, particularly in heterogeneous forest landscapes.
Previous studies have shown that different species of broadleaf trees can affect soil organic matter accumulation, enzyme activities, and microbial biomass in different ways [
8,
17]. However, it is often difficult to determine the relative importance of the effects of different tree species when confounding environmental factors such as climate, land-use history, and parent material are present. This is particularly evident in humid montane forest ecosystems, where strong climatic gradients and complex terrain often mask vegetation-driven soil processes. Conducting species-level comparisons under similar site conditions is therefore critical to isolate the influence of tree identity on soil properties and to improve our mechanistic understanding of soil–vegetation interactions.
The Eastern Black Sea region is characterized by humid montane climates and diverse broadleaf forests, providing a natural laboratory in which to study vegetation-driven soil differentiation under relatively uniform climatic conditions [
18,
19]. In this context, Oriental Beech (
Fagus orientalis Lipsky), Sessile Oak (
Quercus petraea (Matt.) Liebl.), and Pontic Oak (
Quercus pontica K. Koch) form distinct forest stands that differ markedly in terms of litter quality, rooting behavior and nutrient cycling strategies. This suggests a strong potential for species-specific soil modification. Despite their ecological importance, there is limited research on how these dominant tree species influence the physical, chemical and biochemical properties of the soil.
This study aims to evaluate the extent to which tree species identity controls soil physicochemical and biochemical properties in temperate montane forests of the Eastern Black Sea region. By focusing on soils that develop under different vegetation types, we isolate the effects of climatic and geological conditions and assess their roles in shaping soil organic carbon dynamics and microbial functions. We hypothesize that (i) soil biochemical properties differ significantly among forest types due to species-specific organic matter inputs, and (ii) soil texture and pH act as key mediators regulating vegetation effects on microbial activity. This process-oriented approach sheds light on the critical interactions between soil and vegetation that are essential for understanding the functioning of forest soils, the stability of ecosystems, and pedo-ecological processes. This has direct implications for the sustainable management of forests and the restoration of ecosystems in temperate montane regions.
2. Materials and Methods
2.1. Study Site Description
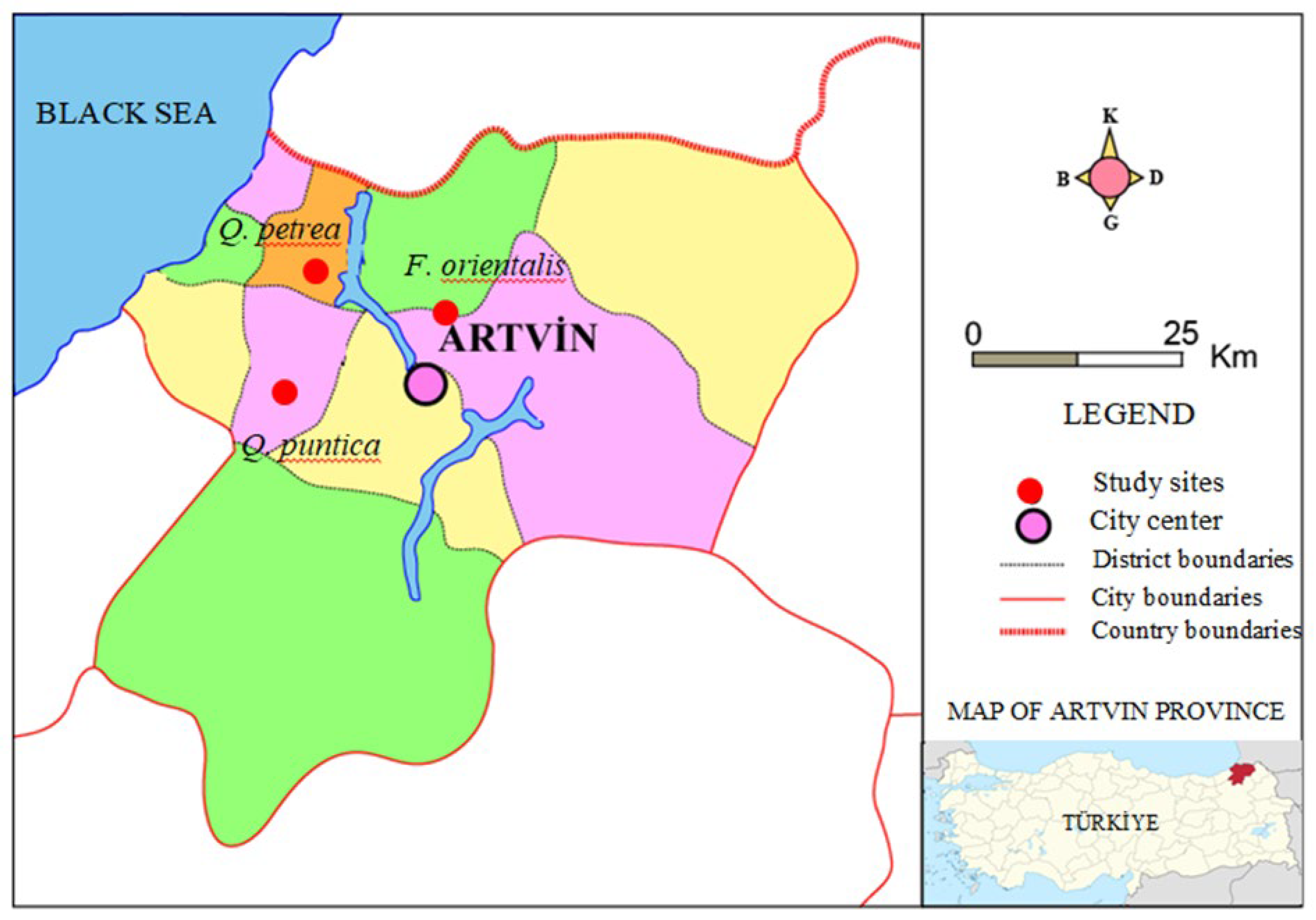
To determine the impact of tree species variation on soil properties, study sites were selected from deciduous forest stands in the Saçinka (Merkez), Başköy (Murgul), and Küçükköy (Borçka) regions within the Artvin province of Türkiye, representing Oriental beech (OB), Sessile oak (SO), and Pontic oak (PO) species, respectively (
Figure 1). Soil samples from the experimental plots established in these regions were collected randomly from areas representing the forests where they were located far apart from each other. The location, topographical characteristics (elevation, slope), stand attributes (stand age and canopy cover), and soil properties (texture and acidity) of the experimental plots are presented in
Table 1.
According to the Köppen climate classification, Cfb and Cfa climate types are seen in the province of Artvin. In the region where the experimental sites are located, the prevailing climate is of the Cfb type, characterized by mild winters, warm summers, and precipitation distributed throughout all seasons. Based on data from the Artvin central meteorological station, the mean annual temperature is 12.4 °C and the mean annual precipitation is 694.1 mm.
Artvin is situated on the North Anatolian orogenic belt in geological terms. The oldest formations of the region are represented by a metamorphic series extending in a northeast direction from the lower sections of the Çoruh Valley through Sirya [
20]. A wide variety of rock types occur within the geological structure of the catchments in which the study areas are located. The experimental plots are situated on basalt (Saçinka and Başköy) and andesite (Küçükköy) parent materials. In the study areas, brown forest soils and non-calcareous brown forest soils are the dominant soil types.
In the Saçinka region, the dominant tree species is Oriental beech (Fagus orientalis Lipsky). Oriental spruce (Picea orientalis (L.) Link) occurs in small admixtures, and the understory vegetation consists locally of blackberry (Rubus spp.), bracken fern (Pteridium aquilinum), and forest ivy (Hedera helix). In the Küçükköy region, the dominant tree species is Sessile oak (Quercus petraea (Matt.) Liebl.), with Scots pine (Pinus sylvestris L.) partially contributing to the stand composition. In the Başköy region, Pontic oak (Quercus pontica K. Koch) communities exist pure forest and mixed forests with oriental beech and oriental spruce species. Similar to the other sites, bracken fern and blackberry form a dense understory in this region
2.2. Soil Sampling
Soil samples were collected in September 2021 from the 0–10 cm depth of the mineral soil layer. In each forest where the three tree species occurred individually, one experimental plot was established, and five soil samples were taken from each plot. Sampling was conducted under homogeneous climatic and geological conditions to minimize environmental variability. To prevent the organic horizon from mixing with the mineral soil, the litter–fermentation–humus layers were carefully removed prior to sampling. In addition, to accurately capture the influence of the tree on soil properties, samples were taken from within the projected area of the tree crown, corresponding to the root-influenced zone.
The collected soil samples were placed in polyethylene bags and transported to the laboratory under cool conditions. A total of 15 fresh soil samples were used as research material for physicochemical and biochemical analyses.
2.3. Soil Physicochemical and Biochemical Analysis
Soil particle-size distribution was determined using the Bouyoucos hydrometer method, and organic carbon content was measured using the Walkley–Black wet oxidation method [
21]. Soil pH was measured with a pH meter, and electrical conductivity (EC) was measured using the EC mode of the same device with an EC-specific probe. For pH measurements, a 1:2.5 soil-to-water suspension was prepared, while a 1:5 suspension was used for EC analysis.
Microbial biomass analyses were performed following the chloroform fumigation–extraction method. For this purpose, 30 g of soil was mixed with K
2SO
4 solution, filtered, and the resulting extract was used for analysis [
22,
23]. A subsample of the same soil was subjected to fumigation, and the extract obtained afterward was analyzed in parallel. For microbial biomass carbon (C
mic), 8 mL of extract was mixed with 2 mL of 0.4 N potassium dichromate solutions and digested with a sulfuric acid–orthophosphoric acid mixture at 150 °C. Following digestion, the mixture was diluted with 20–25 mL of distilled water and titrated with 0.4 N ammonium ferrous sulfate solution using 1,10-phenanthroline as an indicator. Biomass carbon values were calculated separately for fumigated and non-fumigated extracts. C
mic was determined using the following formula [
23]:
where EC is the difference between fumigated and non-fumigated carbon values, and 2.64 is the conversion factor for microbial biomass carbon.
For microbial biomass nitrogen (N
mic), 50 mL of the extract was oxidized after adding 0.2 M copper sulfate and concentrated sulfuric acid, followed by digestion at 380 °C for 240 min. The resulting solution was diluted before and after distillation and titrated with 0.00714 N sulfuric acid using a bromocresol indicator. N
mic was calculated using the following formula [
24]:
where F
N is the difference in nitrogen content between fumigated and non-fumigated samples, and
kN = 0.54 is the conversion coefficient for mineralizable microbial nitrogen [
22].
Microbial basal respiration (MBR) was determined by trapping CO
2 released from soil incubated in sealed chambers for one week in a sodium hydroxide solution [
25]. The metabolic quotient (
qCO
2) was calculated as the ratio of MBR to C
mic [
26,
27], while the microbial quotient (
qMic) was obtained as the ratio of C
mic to soil organic carbon (SOC).
2.4. Statistical Analysis
All statistical analyses were conducted in R Statistical Software (v4.1.2; R Core Team 2021). Data normality was assessed using the Shapiro–Wilk test, and homogeneity of variances was evaluated using Levene’s test. When necessary, data transformations were applied to satisfy model assumptions. Differences in soil biochemical properties among oak species were tested using one-way analysis of variance (ANOVA), followed by Tukey’s HSD test for post hoc comparisons. Statistical significance was accepted at p < 0.05. Spearman’s rank correlation coefficients were calculated to assess relationships among soil physicochemical and microbial biochemical variables. To identify the major gradients structuring soil characteristics, Principal Component Analysis (PCA) was applied to the soil variables (sand, silt, clay, EC, pH, SOC, Cmic, Nmic, MBR, qMic, and qCO2).
3. Results
3.1. Soil Physical and Chemical Properties
Statistically significant differences were observed among tree species for all physical and chemical soil parameters, with the exceptions of EC and SOC (
Table 2). Regarding interspecific variation, significance levels were determined at
p ≤ 0.001 for sand content, clay content, and pH values, and at
p ≤ 0.01 for silt content. The highest F-value was recorded for the pH parameter. Soils of the PO species exhibited the highest sand content alongside the lowest clay, silt, and pH values. In contrast, soils under OB and SO species displayed higher clay, silt, and pH values, but lower sand content compared to PO, with these two species showing similar trends. These findings indicate that soils associated with OB and SO species exhibit a clayey character, and PO soils possess a more pronounced acidic character (
Table 1). Although not statistically significant, the highest EC value was observed in the SO species, and the highest SOC content was recorded in the PO species (
Table 2).
3.2. Biochemical Properties and Stoichiometric Indices
Statistically significant differences were detected among tree species for all examined biochemical soil properties. Significance at the level of
p ≤ 0.01 was found for all parameters, including microbial biomass carbon (C
mic), microbial biomass nitrogen (N
mic), microbial basal respiration (MBR), microbial quotient (
qMic), and C
mic/N
mic. Furthermore, the metabolic quotient (
qCO
2) showed the highest significant difference (
p ≤ 0.001) (
Table 3).
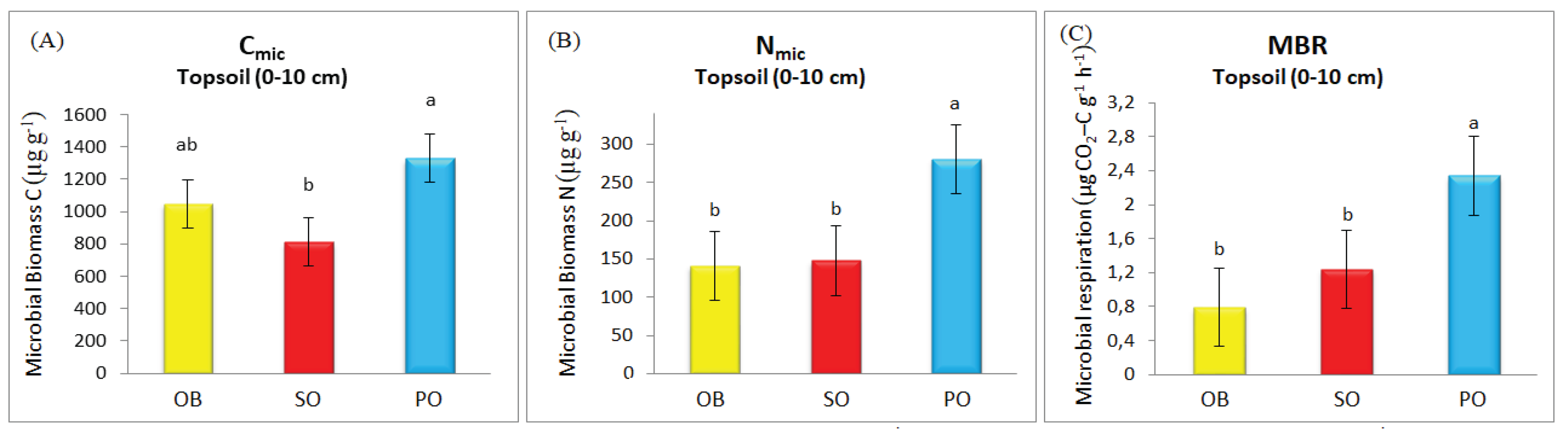
The mean values of C
mic, N
mic, and MBR were highest in soils associated with the OB species, with measured values of 1049.7 μg C g
−1, 141.3 μg N g
−1, and 0.79 g CO
2-C g
−1 h
−1, respectively (see
Figure 2A–C). Although no statistically significant differences were observed between the OB and SO species for N
mic and MBR, the lowest values for both parameters were in OB soils. For C
mic, a partially significant difference was detected between the two species, with the lowest C
mic content observed in SO soils. The C
mic, N
mic, and MBR values in PO soils were 63.8%, 89.7%, and 88.7% higher than in SO soils and 26.6%, 98.6%, and 196.2% higher than in OB soils, respectively.
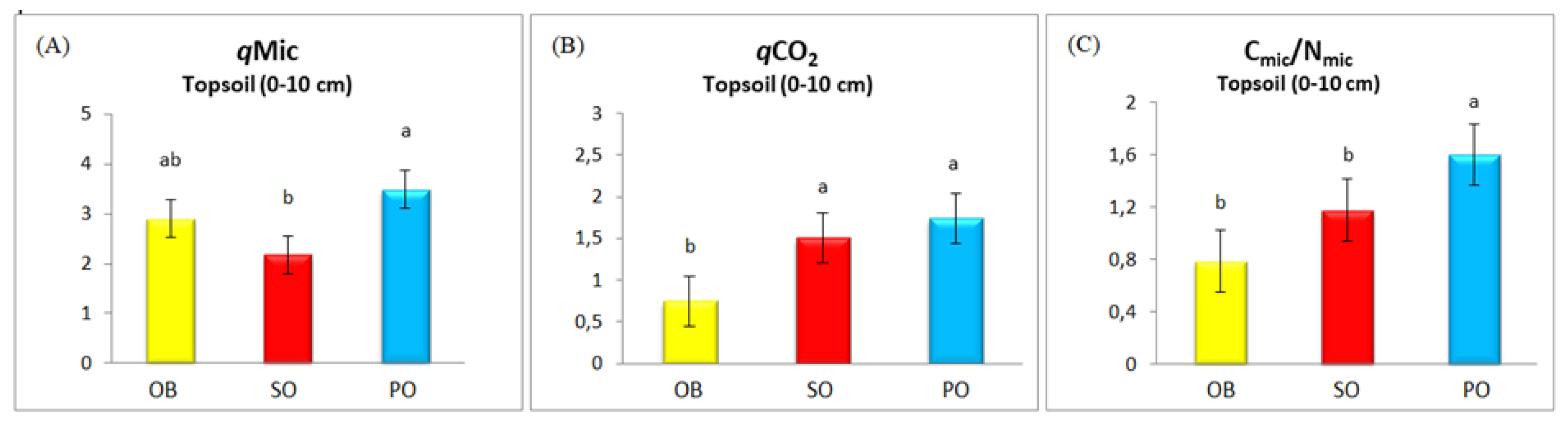
Analysis of stoichiometric indices revealed a pattern consistent with biochemical properties (
Figure 3A–C). The highest values of
qMic,
qCO
2, and the C
mic/N
mic ratio were found in PO soils. No statistically significant difference was detected between OB and SO species in the C
mic/N
mic ratio, with the lowest value observed in OB soils. The lowest
qCO
2 value was recorded in OB soils, and the lowest
qMic value, showing a partially significant difference between PO and SO soils.
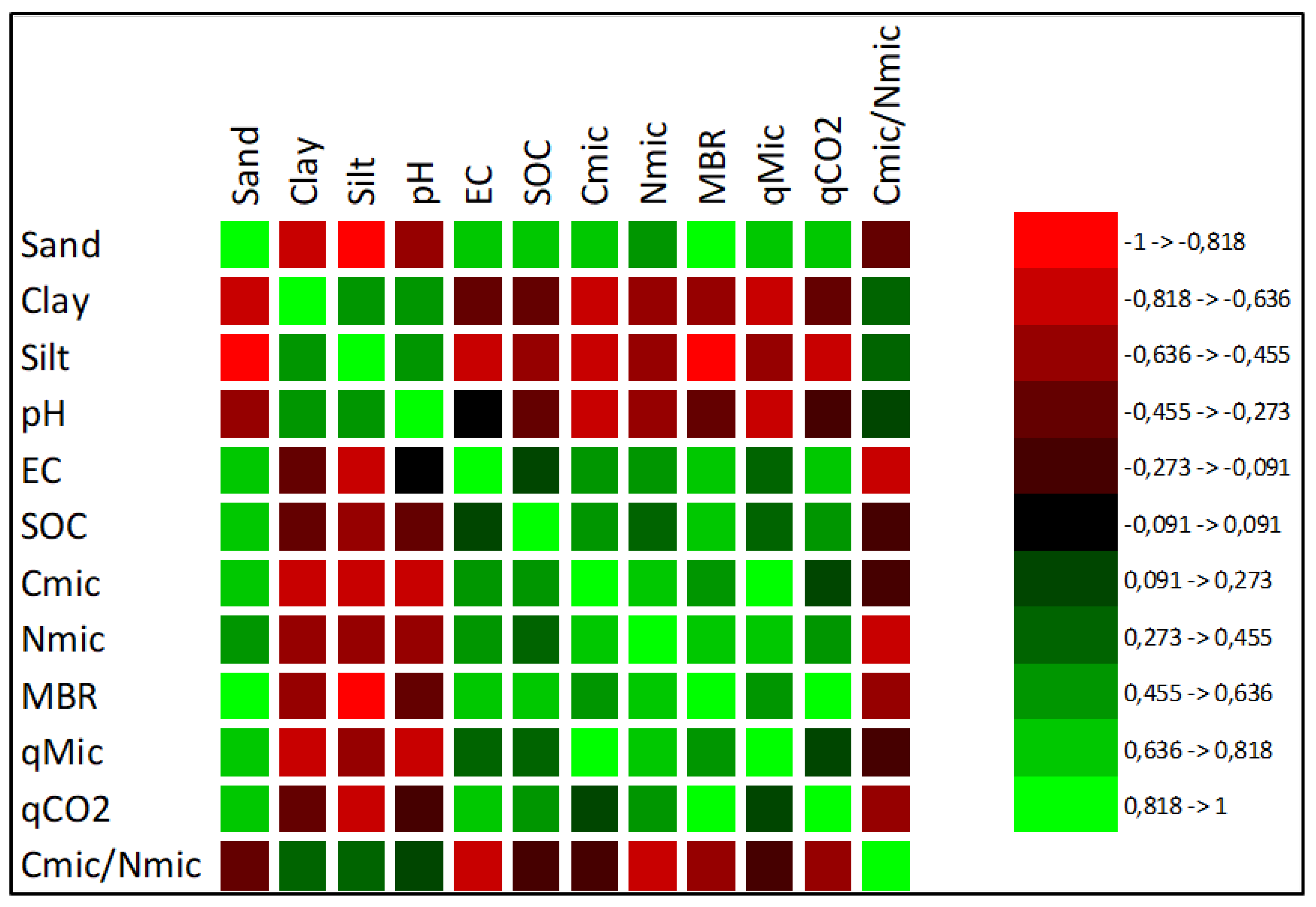
3.3. Correlation Analysis and Principal Component Analysis of Soil Physicochemical and Biochemical Properties
The correlation analysis revealed statistically significant relationships between many soil properties. Silt content and N
mic showed significant correlations with all soil variables, except for the C
mic/N
mic ratio and SOC. Sand content was significantly correlated with all variables except the C
mic/N
mic ratio. Clay content also showed a significant negative correlation with sand, C
mic, N
mic, MBR and
qMic. C
mic showed significant correlations with all soil properties except EC, SOC,
qCO
2 and the C
mic/N
mic ratio. Additionally, a statistically significant relationship was detected between the C
mic/N
mic ratio and
qCO
2.
Figure 4 shows the direction (positive or negative) and strength (based on R
2 values) of these relationships.
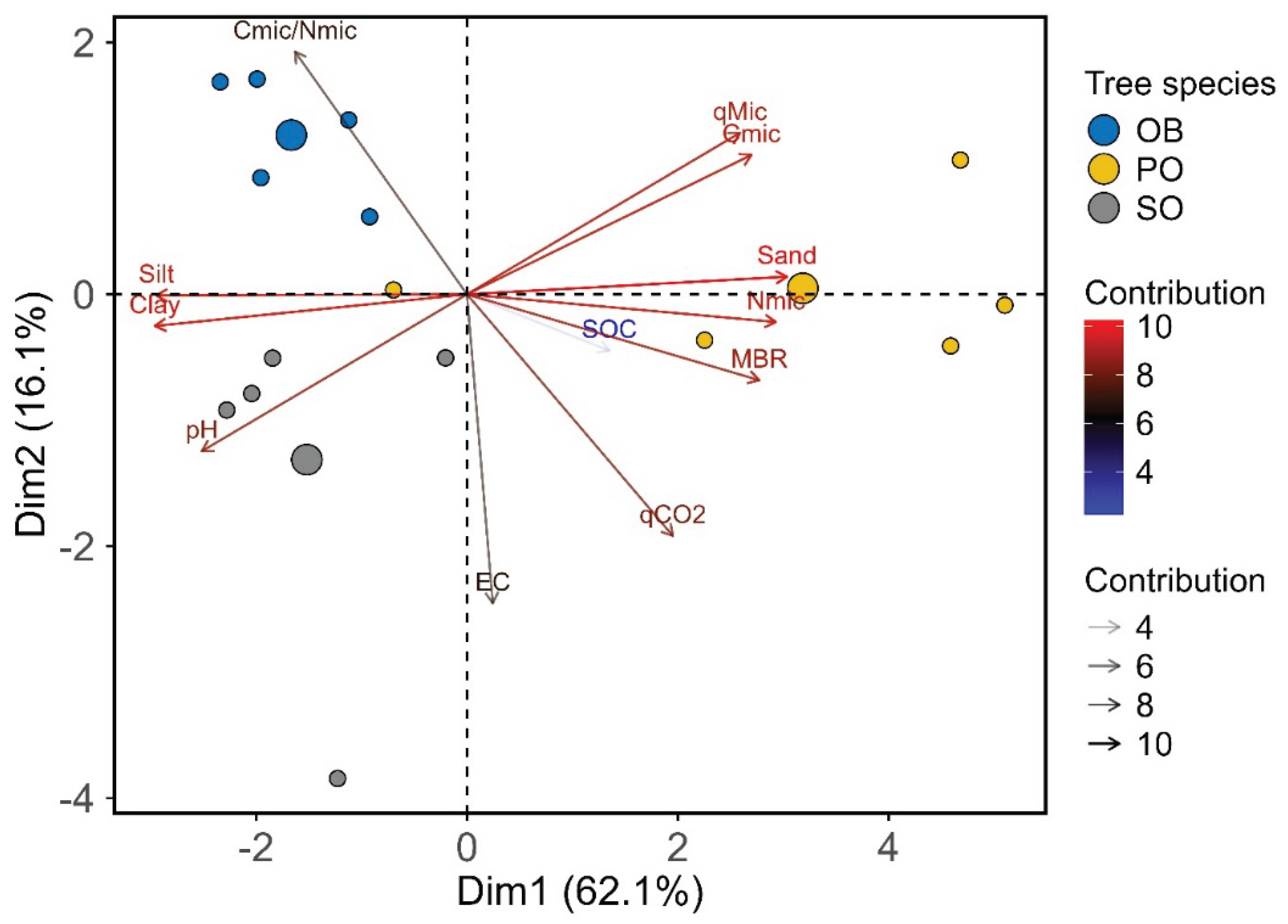
According to the results of the principal component analysis (PCA), the first two principal components explained 78.2% of the total variance in soil properties (dimension 1: 62.1%; dimension 2: 16.1%) (
Figure 5). The analysis revealed a distinct separation of soil samples associated with the three tree species, based on the examined parameters. The group representing the OB species, which was clustered on the positive side of Dim1, was strongly associated with high sand content and several biochemical and stoichiometric variables, including C
mic, N
mic, MBR, and
qMic. Soils from the OB species, which were separated along the positive direction of Dim2, were characterized particularly by high C
mic/N
mic ratios and relatively higher silt and clay contents. In contrast, soils belonging to the SO species were positioned in the lower-left (negative) quadrant of the plot and were associated with higher pH, EC, and
qCO
2 values.
In summary, the PCA results demonstrate that tree species have a significant influence on the physical, chemical, biochemical, and stoichiometric properties of soils. Key driving factors differentiating the study sites belonging to the tree species were variables such as sand content, Nmic, the Cmic/Nmic ratio, and pH.
4. Discussion
4.1. Tree Species Effects on Soil Physicochemical and Biochemical Properties
Our results demonstrate that tree species identity exerts a strong control on soil physicochemical and biochemical properties in temperate montane forests of the Eastern Black Sea region. Despite relatively uniform climatic and geological conditions, soils developed under
Quercus pontica, Quercus petraea, and
Fagus orientalis exhibited marked differences in texture-related attributes, pH, organic carbon dynamic, and microbial activity indicators. This supports the concept that vegetation functions as an environmental propulsive force, shaping soil processes rather than merely responding to pre-existing edaphic conditions [
28,
29].
Higher values of C
mic, N
mic, and MBR observed under
Q. pontica stands suggest that species-specific litter inputs and root-derived carbon play a decisive role in regulating soil biological functioning. Differences in litter chemistry, including lignin content, C:N ratio, and secondary compounds, are known to influence decomposition rates and substrate availability, thereby affecting microbial growth and activity [
11,
30]. Similar species-driven contrasts in microbial biomass and respiration have been reported for temperate forest ecosystems, emphasizing the sensitivity of soil biochemical indicators to vegetation composition [
31]. Tree species significantly affect soil pH at a depth of 0–10 cm [
32], as this layer is directly exposed to the effects of the organic layer and plant root activity. The more acidic nature of soils under PO stands, together with the observed biochemical changes, is likely due to litter chemistry, decomposition dynamics, or accumulation. The humid climate leads to intense leaching [
33] and, consequently, the high precipitation regime of the Artvin region contributes to soil acidification. Furthermore, the higher elevation of PO stands means that the combination of increased precipitation and litter accumulation may enhance soil acidity compared with other species.
4.2. Role of Soil Texture and pH as Mediating Factors
Soil texture emerged as a key mediator of vegetation effects on microbial processes. Variations in sand, silt, and clay fractions influence pore-size distribution, water-holding capacity, and the physical protection of organic matter, thereby regulating microbial access to substrates [
3,
34]. The strong associations observed between texture fractions and biochemical indicators in this study align with previous findings that texture-dependent mechanisms govern soil carbon stabilization and microbial turnover [
35,
36]. Clay soil conditions provide an unfavorable environment for microorganisms in terms of drainage and aeration [
37]. Consequently, due to the limitation of aerobic microbial activity, C
mic, N
mic, and MBR values were lower in the clay soils of SO and OB stands, whereas they were higher in the sandy soils of PO stands. Although clay soils generally possess the capacity to store organic matter [
38], a higher organic carbon content was detected in the sandy soils of PO stands. Furthermore, the high C
mic/SOC ratio (
qMic) observed in sandy soils indicates that the available organic carbon is predominantly labile and thus more accessible and readily utilizable by microorganisms. Consequently, the quality and quantity of PO litter have positively influenced soil carbon dynamics.
Soil pH also plays a central role in the functioning of soil biology. Often described as a master variable, pH integrates multiple soil properties and exerts strong physiological constraints on microbial communities [
39,
40]. The observed pH gradients among forest types likely contribute to differences in microbial activity by influencing nutrient solubility, enzyme activity, and microbial metabolic efficiency. Similar relationships between soil pH and microbial biomass or respiration have been widely reported in forest and grassland ecosystems [
41,
42]. Interestingly, PO soils exhibited the highest C
mic, N
mic, and MBR values despite having a very low pH. Similarly, Xu et al. [
43] reported higher C
mic values in the stand with the lower pH of the two different oak stands. Furthermore, Cheng et al. [
44] found a strong negative correlation between soil pH and C
mic in coniferous and broad-leaved forest soils. In this study, there was a negative correlation between
qCO
2 and soil pH. This relationship may demonstrate that reducing the pH of PO soil led to an increase in
qCO
2 compared to SO soil. According to the findings of Anderson and Domsch [
27], the specific respiration rate of microorganisms inhabiting acidic forest soils is higher than in more circumneutral conditions. Some studies have suggested that litter quality may directly or indirectly affect the quality of organic C and N in mineral soil, thereby influencing ecosystem processes such as soil C and N cycling [
45,
46]. Therefore, substrate availability, driven by the distinct litter characteristics of PO stands, may have maintained C
mic and metabolic activity at elevated levels even under low pH conditions.
4.3. Implications for Soil Organic Carbon Dynamics and Ecosystem Functioning
The close coupling between SOC and microbial indicators observed in this study underscores the role of microbial communities as regulators of carbon cycling in forest soils. Microbial biomass represents the active fraction of SOC and serves as a sensitive indicator of changes in substrate supply and environmental conditions [
47]. Higher microbial biomass and respiration under
Q. pontica suggest more rapid carbon turnover, potentially reflecting higher-quality organic inputs or enhanced microbial efficiency.
However, it is important to acknowledge that vegetation effects on soil properties may also be partially influenced by pre-existing soil conditions that favor the establishment of certain tree species. As noted by Babur et al. [
16] and Khan et al. [
48], soil microbial community assembly and SOC pools are shaped by interacting climatic, geological, and biological drivers. Therefore, while our results highlight strong vegetation-driven patterns, disentangling cause–effect relationships in natural forest ecosystems remains challenging.
4.4. Broader Implications and Limitations
The findings of this study contribute to a growing body of evidence demonstrating that tree species identity can drive substantial differentiation in soil properties at the stand scale. Such differentiation has important implications for nutrient cycling, soil carbon storage, and ecosystem resilience under changing environmental conditions [
6,
49,
50]. Despite the ultra-acidic conditions and sandy texture, the high organic carbon accumulation and biochemical activity promoted by Pontic oak underscore the critical importance of species selection in regional sustainable forest management and ecosystem restoration strategies. The unique ‘biogeochemical signature’ that each broadleaved species imparts to the soil must be considered a fundamental baseline for evaluating the carbon management capacities and nutrient cycling functions of forests. Nevertheless, soil processes operate within complex, multi-factorial systems. Potential influences of microtopography, historical land use, and unmeasured climatic variability cannot be fully excluded and warrant further investigation.
5. Conclusions
This study demonstrates that the identity of tree species plays a decisive role in shaping the physicochemical and biochemical properties of soils in temperate montane forests in the Eastern Black Sea region. Even under comparable climatic and geological conditions, soils under Quercus pontica, Quercus petraea, and Fagus orientalis exhibited distinct texture, pH, organic carbon dynamics, and microbial activity patterns. These differences emphasize the importance of vegetation as a key biotic driver that influences soil functioning through species-specific organic matter inputs and interactions with texture and pH. Soil texture and pH act as primary mediators, regulating microbial biomass and respiration and thus underlining their importance in controlling soil biological processes and carbon dynamics. The strong correlations between soil microbial indicators with the soil texture and pH highlight the role of microbial communities in soil and ecosystem management.
Overall, our findings provide process-based insights into soil–vegetation interactions relevant to soil formation, ecosystem functioning, and sustainable forest management. Understanding how dominant tree species shape soil properties and how vegetation composition links to microbial assembly is essential for predicting carbon cycling and anticipating ecosystem responses to environmental change. This knowledge is also vital for developing effective strategies for forest conservation and restoration. Future studies should extend this approach to mixed and uneven-aged forest stands to further elucidate the mechanisms linking vegetation composition, soil processes, and ecosystem stability.
Author Contributions
Conceptualization, E.B. and A.T.; methodology, M.A. and E.B.; software, M.A. and E.B.; validation, M.A., E.B. and A.T.; investigation, M.A., E.B. and A.T.; resources, M.A., and A.T.; data curation, M.A. and E.B.; writing—original draft preparation, M.A., E.B. and A.T.; writing—review and editing, M.A., E.B. and A.T.; visualization, M.A., E.B. and A.T.; supervision, E.B. and A.T.; project administration, E.B.; All authors have read and agreed to the published version of the manuscript.
Funding
This study was financially supported by the 2219-International Postdoctoral Research Fellowship Program (1059B192000961) from TÜBİTAK.
Acknowledgments
We thank all those who have contributed to this research.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Babur, E. Significant Differences in Microbial Soil Properties, Stoichiometry and Tree Growth Occurred within 15 Years after Afforestation on Different Parent Material. Life 2024, 14, 1139. [Google Scholar] [CrossRef]
- Khan, M.T.; Supronienė, S.; Žvirdauskienė, R.; Aleinikovienė, J. Climate, Soil, and Microbes: Interactions Shaping Organic Matter Decomposition in Croplands. Agronomy 2025, 15, 1928. [Google Scholar] [CrossRef]
- Kara, O.; Babur, E.; Altun, L.; Seyis, M. Effects of afforestation on microbial biomass C and respiration in eroded soils of Turkey. J. Sustain. For. 2016, 35, 385–396. [Google Scholar] [CrossRef]
- Cao, J.; He, X.; Chen, Y.; Chen, Y.; Zhang, Y.; Yu, S.; Zhou, L.; Liu, Z.; Zhang, C.; Fu, S. Leaf Litter Contributes More to Soil Organic Carbon than Fine Roots in Two 10-Year-Old Subtropical Plantations. Sci. Total Environ. 2020, 704, 135341. [Google Scholar]
- Scheibe, A.; Steffens, C.; Seven, J.; Jacob, A.; Hertel, D.; Leuschner, C.; Gleixner, G. Effects of tree identity dominate over tree diversity on the soil microbial community structure. Soil Biol. Biochem. 2015, 81, 219–227. [Google Scholar] [CrossRef]
- Babur, E.; Dindaroglu, T.; Uslu, O.S.; Gozukara, G.; Ozlu, E. Long-term effects of land use conversion on soil microbial biomass and stoichiometric indices in eastern Mediterranean Karst ecosystems (1981–2018). Land Degrad. Dev. 2025, 36, 5666–5680. [Google Scholar] [CrossRef]
- Babur, E.; Dindaroglu, T.; Roy, R.; Seleiman, M.F.; Ozlu, E.; Battaglia, M.L.; Uslu, Ö.S. Relationship between organic matter and microbial biomass in different vegetation types. In Microbial Syntrophy-Mediated Eco-Enterprising; Elsevier: Amsterdam, The Netherlands, 2022; pp. 225–245. [Google Scholar]
- Sariyildiz, T.; Anderson, J.M.; Kucuk, M. Effects of tree species and topography on soil chemistry, litter quality, and decomposition in Northeast Turkey. Soil Biol. Biochem. 2005, 37, 1695–1706. [Google Scholar] [CrossRef]
- Babur, E.; Dindaroglu, T.; Riaz, M.; Uslu, O.S. Seasonal Variations in Litter Layers’ Characteristics Control Microbial Respiration and Microbial Carbon Utilization under Mature Pine, Cedar, and Beech Forest Stands in the Eastern Mediterranean Karstic Ecosystems. Microb. Ecol. 2022, 84, 153–167. [Google Scholar]
- Eisenhauer, N.; Beßler, H.; Engels, C.; Gleixner, G.; Habekost, M.; Milcu, A.; Partsch, S.; Sabais, A.C.W.; Scherber, C.; Steinbeiss, S.; et al. Plant diversity effects on soil microorganisms support the singular hypothesis. Ecology 2010, 91, 485–496. [Google Scholar] [CrossRef]
- Prescott, C.E.; Grayston, S.J. Tree species influence on microbial communities in litter and soil: Current knowledge and research needs. For. Ecol. Manage. 2013, 309, 19–27. [Google Scholar]
- Bardgett, R.D.; Caruso, T. Soil microbial community responses to climate extremes: Resistance, resilience and transitions to alternative states. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2020, 375, 20190112. [Google Scholar] [CrossRef]
- Pan, J.; Guo, Q.; Li, H.; Luo, S.; Zhang, Y.; Yao, S.; Fan, X.; Sun, X.; Qi, Y. Dynamics of Soil Nutrients, Microbial Community Structure, Enzymatic Activity, and Their Relationships along a Chronosequence of Pinus massoniana Plantations. Forests 2021, 12, 376. [Google Scholar] [CrossRef]
- Sheshnitsan, S.; Odnoralov, G.; Tikhonova, E.; Gorbunova, N.; Sheshnitsan, T.; Murariu, O.C.; Caruso, G. Influence of Soil Texture on Carbon Stocks in Deciduous and Coniferous Forest Biomass in the Forest-Steppe Zone of Oka–Don Plain. Soil Syst. 2024, 8, 118. [Google Scholar]
- Kuśmierz, S.; Skowrońska, M.; Tkaczyk, P.; Lipiński, W.; Mielniczuk, J. Soil Organic Carbon and Mineral Nitrogen Contents in Soils as Affected by Their pH, Texture and Fertilization. Agronomy 2023, 13, 267. [Google Scholar] [CrossRef]
- Babur, E.; Ozlu, E.; Uslu, O.S. Soil respiration, microbial biomass, and stoichiometry within riparian buffers and adjacent land use. Sci. Rep. 2025, 15, 40445. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y.; Guo, K.; Zhao, H.; Qiao, X.; Wang, S.; Zhang, L.; Cai, X. Mixing litter from deciduous and evergreen trees enhances decomposition in a subtropical karst forest in southwestern China. Soil Biol. Biochem. 2016, 101, 44–54. [Google Scholar] [CrossRef]
- Atalay, İ.; Efe, R. Structural and distributional evaluation of forest ecosystems in Turkey. J. Environ. Biol. 2010, 31, 61–70. [Google Scholar]
- Olson, D.M.; Dinerstein, E.; Wikramanayake, E.D.; Burgess, N.D.; Powell, G.V.N.; Underwood, E.C.; D’amico, J.A.; Itoua, I.; Strand, H.E.; Morrison, J.C.; et al. Terrestrial Ecoregions of the World: A New Map of Life on Earth: A new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. BioScience 2001, 51, 933–938. [Google Scholar]
- Gattinger, T. Explonatory Text of Geological Map of Turkey; Volume Yayın No 36, Maden Tetkik ve Arama Enstitüsü: Ankara, Turkey, 1962; 75 s. [Google Scholar]
- Gülçur, F. Toprağın Fiziksel ve Kimyasal Analiz Metodları; İstanbul Üniversitesi Orman Fakültesi Yayınları: İstanbul, Turkey, 1974. Yayın No 1970 (In Turkish) [Google Scholar]
- Brookes, P.C.; Landman, A.; Pruden, G.; Jenkinson, D.S. Chloroform fumigation and the release of soil nitrogen: A rapid extraction method to measure microbial biomass nitrogen in soil. Soil Biol. Biochem. 1985, 17, 837–842. [Google Scholar] [CrossRef]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Baillie, I.C.; Anderson, J.M.; Ingram, J.S.I. Tropical soil biology and fertility: A handbook of methods. J. Ecol. 1990, 78, 547. [Google Scholar] [CrossRef]
- Alef, K. Soil respiration. In Methods in Applied Soil Microbiology and Biochemistry; Alef, K., Nannipieri, P., Eds.; Academic Press: San Diego, CA, USA, 1995; pp. 214–219. [Google Scholar]
- Anderson, T.H.; Domsch, K.H. Application of eco-physiological quotients (qCO2, and qD) on microbial biomasses from soils of different cropping histories. Soil Biol. Biochem. 1990, 22, 251–255. [Google Scholar]
- Anderson, T.H.; Domsch, K.H. The metabolic quotient for CO2 (qCO2) as a specific activity parameter to assess the effects of environmental conditions, such as pH, on the microbial biomass of forest soils. Soil Biol. Biochem. 1993, 25, 393–395. [Google Scholar] [CrossRef]
- Wardle, D.A.; Bardgett, R.D.; Klironomos, J.N.; Setälä, H.; van der Putten, W.H.; Wall, D.H. Ecological linkages between aboveground and belowground biota. Science 2004, 304, 1629–1633. [Google Scholar] [CrossRef]
- Augusto, L.; De Schrijver, A.; Vesterdal, L.; Smolander, A.; Prescott, C.; Ranger, J. Influences of evergreen gymnosperm and deciduous angiosperm tree species on the functioning of temperate and boreal forests. Biol. Rev. 2015, 90, 444–466. [Google Scholar]
- Cornwell, W.K.; Cornelissen, J.H.; Amatangelo, K.; Dorrepaal, E.; Eviner, V.T.; Godoy, O.; Hobbie, S.E.; Hoorens, B.; Kurokawa, H.; Pérez-Harguindeguy, N.; et al. Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol. Lett. 2008, 11, 1065–1071. [Google Scholar] [CrossRef]
- Babur, E.; Dindaroğlu, T.; Solaiman, Z.M.; Battaglia, M.L. Microbial respiration, microbial biomass and activity are highly sensitive to forest tree species and seasonal patterns in the Eastern Mediterranean Karst Ecosystems. Sci. Total Environ. 2021, 775, 145868. [Google Scholar]
- Augusto, L.; Ranger, J.; Binkley, D.; Rothe, A. Impact of several common tree species of European temperature forests on soil fertility. Ann. For. Sci. 2002, 59, 233–253. [Google Scholar] [CrossRef]
- Macías, F. Materias Orixinais E Solos De Galiza. Cuad. Semin. Sargadelos 1986, 47, 47–79. [Google Scholar]
- Six, J.; Conant, R.T.; Paul, E.A.; Paustian, K. Stabilization mechanisms of soil organic matter: Implications for C-saturation of soils. Plant Soil 2002, 241, 155–176. [Google Scholar] [CrossRef]
- Zhang, Y.; Dong, S.K.; Gao, Q.Z.; Liu, S.L.; Zhou, H.K.; Ganjurjav, H.; Wang, X. Climate Change and Human Activities Altered the Diversity and Composition of Soil Microbial Community in Alpine Grasslands of the Qinghai Tibetan Plateau. Sci. Total Environ. 2016, 562, 353–363. [Google Scholar] [CrossRef]
- Gu, Y.F.; Bai, Y.; Xiang, Q.J.; Yu, X.M.; Zhao, K.; Zhang, X.P.; Li, C.; Liu, S.; Chen, Q. Degradation Shaped Bacterial and Archaeal Communities with Predictable Taxa and Their Association Patterns in Zoige Wetland at Tibet Plateau. Sci. Rep. 2018, 8, 3884. [Google Scholar] [CrossRef] [PubMed]
- Bauhus, J.; Pare, D. Effects of tree species, stand age and soil type on soil microbial biomass and its activity in a southern boreal forest. Soil Biol. Biochem. 1998, 30, 1077–1089. [Google Scholar] [CrossRef]
- Lutzow, M.; Kogel-Knabner, I.; Ekschmitt, K.; Matzner, E.; Guggenberger, G.; Marschner, B.; Flessa, H. Stabilization of organic matter in temperate soils: Mechanisms and their relevance under different soil conditions—A review. Eur. J. Soil Sci. 2006, 57, 426–445. [Google Scholar] [CrossRef]
- Lauber, C.L.; Hamady, M.; Knight, R.; Fierer, N. Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl. Environ. Microbiol. 2009, 75, 5111–5120. [Google Scholar] [CrossRef]
- Li, H.; Ye, D.; Wang, X.; Settles, M.L.; Wang, J.; Hao, Z.; Zhou, L.; Dong, P.; Jiang, Y.; Ma, Z. Soil bacterial communities of different natural forest types in Northeast China. Plant Soil 2014, 383, 203–216. [Google Scholar] [CrossRef]
- Ramirez, K.S.; Lauber, C.L.; Knight, R.; Bradford, M.A.; Fierer, N. Consistent effects of nitrogen fertilization on soil bacterial communities in contrasting systems. Ecology 2010, 91, 3463–3470. [Google Scholar] [CrossRef]
- Davinic, M.; Fultz, L.M.; Acosta-Martinez, V.; Calderón, F.J.; Cox, S.B.; Dowd, S.E.; Allen, V.G.; Zak, J.C.; Moore-Kucera, J. Pyrosequencing and mid-infrared spectroscopy reveal distinct aggregate stratification of soil bacterial communities and organic matter composition. Soil Biol. Biochem. 2012, 46, 63–72. [Google Scholar] [CrossRef]
- Xu, X.; Inubushi, K.; Sakamoto, K. Effect of vegetations and temperature on microbial biomass carbon and metabolic quotients of temperate volcanic forest soils. Geoderma 2006, 136, 310–319. [Google Scholar] [CrossRef]
- Cheng, F.; Peng, X.; Zhao, P.; Yuan, J.; Zhong, C.; Cheng, Y.; Cui, C.; Zhang, S. Soil microbial biomass, basal respiration and enzyme activity of main forest types in the Qinling Mountains. PLoS ONE 2013, 8, 67353. [Google Scholar] [CrossRef]
- Finzi, A.C.; Van Breemen, N.; Canham, C.D. Canopy tree-soil interactions within temperate forests: Species effects on soil carbon and nitrogen. Ecol. Appl. 1998, 8, 440–446. [Google Scholar] [CrossRef]
- Lovett, G.; Weathers, K.; Arthur, M.; Schultz, J. Nitrogen cycling in a northern hardwood forest: Do species matter? Biogeochemistry 2004, 67, 289–308. [Google Scholar] [CrossRef]
- Chen, T.H.; Chiu, C.Y.; Tian, G.L. Seasonal dynamics of soil microbial biomass in coastal sand dune forest. Pedobiologia 2005, 49, 645–653. [Google Scholar] [CrossRef]
- Khan, M.T.; Supronienė, S.; Žvirdauskienė, R.; Aleinikovienė, J. Climate, Soil, and Microbes: Interactions Shaping Organic Matter Decomposition in Croplands. Agronomy 2025, 15, 1928. [Google Scholar] [CrossRef]
- Liu, K.; Xue, Y.Q.; Zhu, L.; Xu, F.; Zhu, Z.; Zhang, T. Effect of Different Land Use Types on the Diversity of Soil Bacterial Community in the Coastal Zone of Jialing River. Environ. Sci. 2021, 43, 1620–1629. [Google Scholar]
- Li, Y.; Han, C.; Sun, S.; Zhao, C. Effects of Tree Species and Soil Enzyme Activities on Soil Nutrients in Dryland Plantations. Forests 2021, 12, 1153. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).