Background
An intellectual disability (ID) is a lifelong neurodevelopmental condition characterised by significant limitations in cognitive functioning and adaptive behaviours, such as social and practical skills, with onset during childhood (1). Globally, approximately 1-3% of the population, equating to around 200 million individuals, live with an ID, encountering unique challenges that impact daily life (2, 3). This group frequently faces systemic barriers within healthcare services, including communication difficulties, inadequate provision of reasonable adjustments, and diagnostic overshadowing, where health issues are mistakenly attributed to the ID (3). These challenges contribute to health inequalities, with adults with an ID experiencing a life expectancy reduction of 19-23 years compared to the general population, and nearly half of deaths amongst people with an ID deemed preventable through better care and timely interventions (4). To tackle these disparities, initiatives such as the UK’s National Health Service (NHS) Long Term Plan (2019) and guidance from the National Institute for Health and Care Excellence (NICE) have prioritised improving equitable access to healthcare and enhancing health outcomes for individuals with an ID (5, 6).
ID is increasingly recognised as a hidden driver of cancer mortality (7-9). While the relationship between ID and cancer varies by cancer type, studies consistently report a higher risk of bowel cancer and related deaths (10-12). In addition, participation in the UK’s national bowel cancer screening programme is significantly lower among adults with an ID, with only 50.3% of eligible individuals taking part compared to 66.8% in the general population (13). Reduced screening uptake may contribute to late-stage diagnoses and emergency presentations, though data on these outcomes are limited (14). Further uncertainties exist, particularly regarding the risk of bowel cancer in younger individuals with an intellectual disability below 50 years of age, which is the age bowel cancer screening programmes begin in several countries. It also remains unclear how frequently symptoms associated with bowel cancer occur in this group, whether such symptoms are appropriately investigated in primary and secondary care settings, and how treatment decisions and survival outcomes compare following diagnosis.
The aim of this study was to examine the risk of bowel cancer in individuals with an ID, including those under the age of 50, and to assess the effect of an ID on each stage of the bowel cancer care pathway. This included the frequency of symptoms and patterns of investigation, such as faecal occult blood (FOB) or faecal immunochemical (FIT) testing, urgent suspected cancer referrals, endoscopy, surgery for stage I-III disease, systemic anticancer therapy for stage IV disease, and bowel cancer-specific survival, using a large population-based matched cohort.
Methods
Study population
The study cohort was from anonymised data held in the Clinical Practice Research Datalink (CPRD) Aurum database (15). CPRD Aurum includes routinely recorded clinical information from general practices in England that use the EMIS Web system and is representative of the broader UK population in terms of age, gender and ethnicity (15). General practice registration is necessary to access NHS healthcare and, as a result, almost all people in the UK are registered. On the date that data were extracted, CPRD Aurum contained information on approximately 50 million patients (including ~ 16 million currently contributing patients), representing around one-quarter of the UK population and one-fifth of general practices. In this study, primary care, ethnicity (16) and deprivation data were linked with mortality records from the Office for National Statistics (ONS), cancer registration data from the National Cancer Registration and Analysis Service (NCRAS), and secondary care data from Hospital Episode Statistics (HES).
Inclusion and exclusion criteria
Participants eligible for inclusion were those with a documented diagnosis of an ID between 1 January 2000 and 31 December 2018. The date of the first recorded ID diagnosis within this time window served as the index date for matching purposes (further described below). A list of Read codes, SNOMED CT, and EMIS codes for intellectual disability were compiled based on previous research together with keyword searches of the CPRD data dictionary (17-26). The list of codes comprised specific diagnostic codes for ID and codes related to conditions linked to ID (Supplementary Code List 1). This was because of known under-recording of ID in primary care, where only around 25% of individuals have a recorded diagnosis (27). Codes relating to “learning difficulty” were considered equivalent to “learning disability” (another term for intellectual disability) in view of both terms being used interchangeably in UK legislation and NHS guidance (28). The final list of codes was agreed on by two clinicians (OJK and UC), following previously applied methods (29).
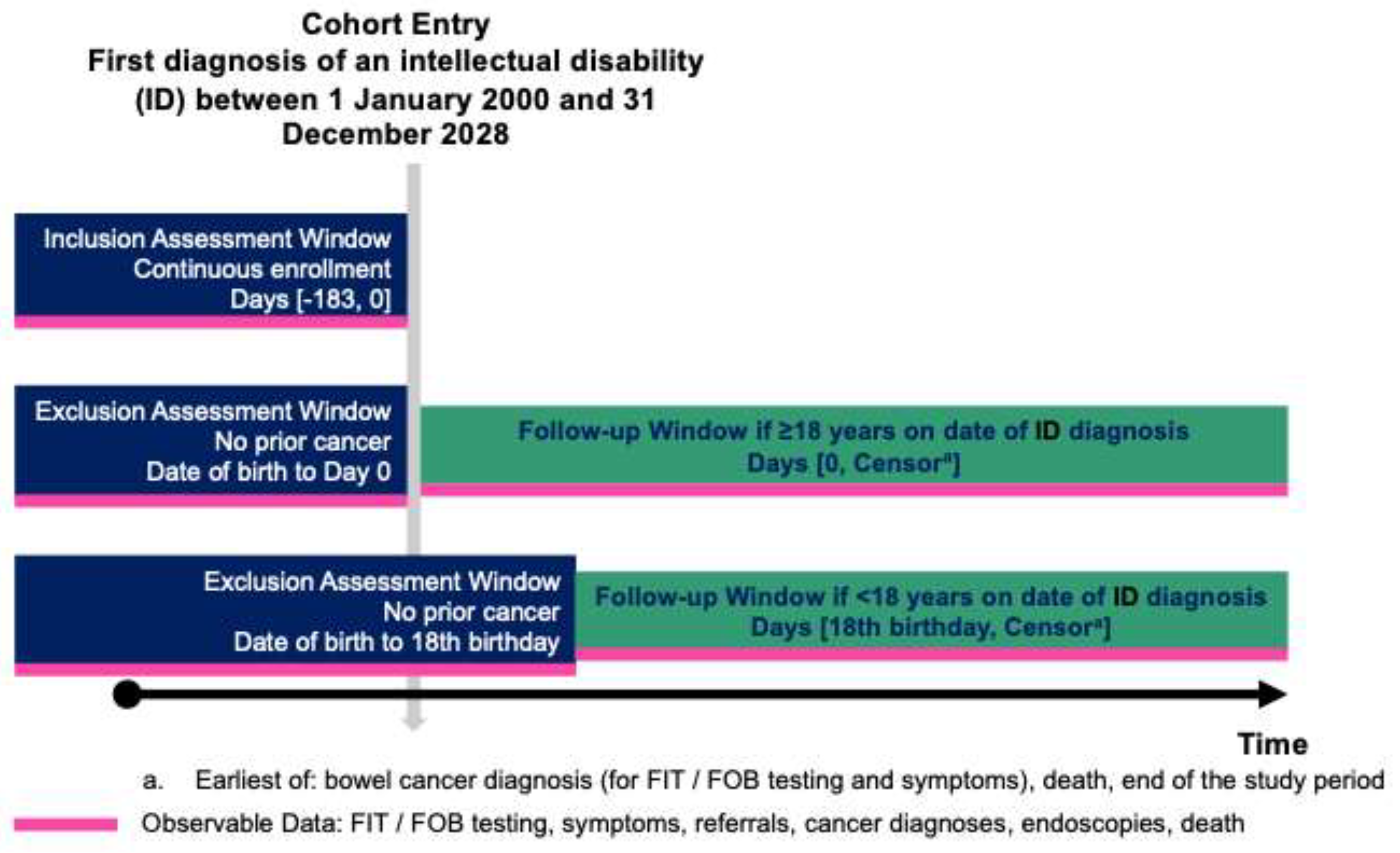
Each person with an ID was matched with up to 20 comparators using incidence density sampling, matched by age (within a two-year range), gender, general practice and index date. Comparators had no recorded ID diagnosis before the index date and were given the same follow-up start date as their matched individual with an ID. Follow-up started on the later of the ID diagnosis date or the individual’s 18th birthday. This age was selected to balance the lower risk of bowel cancer in younger people with the need to capture early-onset disease. Early-onset bowel cancer is an emerging public health concern (30), and this study investigated whether it disproportionately affects people with an ID. Exclusion criteria included cancer diagnosis before follow-up, less than six months of continuous general practice registration before the index date, non-eligibility for linkage to the NCRAS, ONS, and HES datasets. Follow-up ended on 31 December 2018, the latest date with complete linked data. A standardised graphical overview of cohort entry and follow-up timelines is shown in
Figure 1 (31).
Outcomes and statistical analysis
Pre-diagnostic outcomes of interest included symptoms potentially indicative of bowel cancer (
Table 2) and faecal occult blood (FOB) or faecal immunochemical (FIT) testing. For these outcomes, each comparator’s follow-up was limited to the observation period of their matched case to allow for comparisons over equivalent timeframes. Outcomes were assessed following the first presentation with symptoms: within 28 days, these included completion of a FIT/FOB test (in the absence of a prior test in the preceding year) and urgent referral for suspected cancer; within 56 days, outcomes included endoscopy (e.g., colonoscopy or flexible sigmoidoscopy) and a confirmed diagnosis. Diagnoses within six-month, 1 and 3-year windows were additionally used to capture potential delays or missed diagnoses, including malignancy of undefined origin (MUO) and cancer of unknown primary (CUP), which may indicate late presentation or other barriers to investigation leading to undiagnosed bowel cancer. Among individuals diagnosed with bowel cancer, outcomes included stage at diagnosis (I, II, III, IV and unknown), route to diagnosis (screening programme, urgent suspected cancer referral and emergency presentation), receipt of curative surgery within six months for those with stages I-III disease and receipt of SACT within six months for those with stages IV disease. Associations between ID and diagnosis of bowel cancer at any age and before 50 years of age were also estimated.
Table 2.
Associations of intellectual disability with bowel cancer, symptoms suggestive of bowel cancer and FIT / FOB testing.
Table 2.
Associations of intellectual disability with bowel cancer, symptoms suggestive of bowel cancer and FIT / FOB testing.
| |
No ID |
ID |
Estimate |
| Bowel cancer risk |
Diagnoses / total |
Diagnoses / total |
HR (95% CI) |
| |
6,936 / 1,964,420 |
381 / 111,034 |
1.30 (1.18-1.44) |
| Bowel cancer risk age <50 years |
Diagnoses <50 years / total <50 years |
Diagnoses <50 years / total <50 years |
RR (95% CI) |
| |
489 / 1,405,445 |
58 / 81,240 |
2.19 (1.68-2.85) |
| Symptoms suggestive of bowel cancer |
Patients with symptoms† / total patients |
Patients with symptoms / total patients |
IRR (95% CI) |
| |
307,560 / 1,964,420 |
35,247 / 111,034 |
2.59 (2.53-2.65) |
| FIT / FOB test |
Patients tested / total patients |
Patients tested / total patients |
IRR (95% CI) |
| |
208,723 / 1,964,420 |
12,793 / 111,034 |
0.92 (0.91-0.93) |
Crude proportions for each outcome were summarised for individuals with and without an ID. Age, gender, ethnicity and deprivation-adjusted incidence rate ratios (aIRRs), risk ratios (aRRs) and hazard ratios (aHRs), along with 95% confidence intervals (CIs), were estimated using Poisson regression (with follow-up time as an offset in the model), modified Poisson regression with inverse probability of censoring weights, and Cox proportional hazards regression. Log-log (survival) versus log-time plots were used to check proportionality assumptions. Analyses of outcomes with follow-up starting at baseline (e.g., bowel cancer risk, rates of symptoms) used matched-set fixed effects or stratification with robust standard errors. Analyses with follow-up starting after a post-baseline event (e.g., bowel cancer-specific survival or outcomes following symptoms) used unmatched, event-anchored models to mitigate collider bias. Kaplan-Meier estimates were also used to estimate bowel cancer-specific survival. All statistical analyses were performed using R version 4.0.0. Missing data (<5% for all variables) were handled using multiple imputation by chained equations. The study was conducted in accordance with the RECORD (REporting of studies Conducted using Observational Routinely-collected health Data) guidelines (32).
Results
The study population comprised 111,034 individuals with an ID and 1,964,420 matched comparators (
Table 1). The median ages at the start of follow-up were similar for those with and without an ID (34.0 vs. 35.8 years), as were the proportions of males (57.9% vs. 56.9%). A higher proportion of individuals with an ID were of White ethnicity compared to those without (86.3% vs. 82.6%), with lower proportions of unknown ethnicity (2.6% vs. 5.3%) and broadly similar proportions of Asian (5.6% vs. 6.6%) and Black ethnicities (4.0% vs. 3.8%). Individuals with an ID were more likely to live in areas of greater socioeconomic deprivation (27.2% .vs 22.8% in the most deprived quintile). The median follow-up was 4.8 years.
Table 1.
Baseline characteristics of the included population.
Table 1.
Baseline characteristics of the included population.
| |
No intellectual disability |
Intellectual disability |
| Total |
1,964,420 |
111,034 |
| Gender |
|
|
| Male |
1,117,680 (56.9%) |
64,268 (57.9%) |
| Female |
846,740 (43.1%) |
46,766 (42.1%) |
| Age (years) |
|
|
| 40–50 |
589,880 (30%) |
32,028 (28.8%) |
| 50–60 |
257,704 (13.1%) |
13,732 (12.4%) |
| 60–70 |
158,617 (8.1%) |
8,406 (7.6%) |
| >70 |
142,522 (7.3%) |
7,649 (6.9%) |
| Ethnicity |
|
|
| White |
1,622,952 (82.6%) |
95,798 (86.3%) |
| Asian |
129,220 (6.6%) |
6,169 (5.6%) |
| Black |
75,192 (3.8%) |
4,479 (4%) |
| Mixed/Multiple |
25,567 (1.3%) |
1,566 (1.4%) |
| Other |
7,630 (0.4%) |
182 (0.2%) |
| Unknown |
103,859 (5.3%) |
2,840 (2.6%) |
| Deprivation (IMD quintile) |
|
|
| 1 - Least deprived |
347,257 (17.7%) |
14,824 (13.4%) |
| 2 |
370,388 (18.9%) |
18,721 (16.9%) |
| 3 |
380,329 (19.4%) |
21,651 (19.5%) |
| 4 |
415,110 (21.1%) |
25,506 (23%) |
| 5 - Most deprived |
448,408 (22.8%) |
30,169 (27.2%) |
| Unknown |
2,928 (0.1%) |
163 (0.1%) |
As shown in
Table 2, ID was associated with a significantly higher incidence rate of symptoms (IRR 2.59, 95% CI 2.53-2.64) but a reduced incidence rate of FIT/FOB testing (IRR 0.92, 95% CI 0.91-0.93). Following their first symptom, individuals with an ID were markedly less likely to have a FIT or FOB test (RR 0.74, 95% CI 0.67-0.83) or an urgent suspected cancer referral (RR 0.57, 95% CI 0.52-0.62) within 28 days. They were also less likely to have an endoscopy (RR 0.45, 95% CI 0.42-0.49) or receive a diagnosis within 56 days (RR 0.52, 95% CI 0.41-0.67). Differences in diagnosis decreased over time: at six months (RR 0.64, 95% CI 0.53-0.77), one year (RR 0.67, 95% CI 0.56-0.79), and three years (RR 0.78, 95% CI 0.68-0.90) after the first symptom. When MUO and CUP cases were included, the difference at three years was not statistically significant (RR 0.89, 95% CI 0.78-1.01).
During follow-up, 381 individuals with an ID and 6,936 without were diagnosed with bowel cancer, including 58 and 489 cases, respectively, under the age of 50. ID was strongly associated with a higher overall risk of bowel cancer (HR 1.30, 95% CI 1.18-1.44,
Table 2) and a twofold increase in risk before age 50 (RR 2.19, 95% CI 1.68-2.85). Diagnoses through screening programmes were less common among individuals with an ID (n=10 [2.9%] vs. n=580 [10.4%]; RR 0.27, 95% CI 0.14-0.50), as were diagnoses via urgent suspected cancer referrals (n=72 [20.6%] vs. n=1,896 [34.0%]; RR 0.62, 95% CI 0.50-0.76) (
Table 4). In contrast, emergency presentations were more common in the ID group (n=136 [39.0%] vs. n=1,240 [22.3%]; RR 1.76, 95% CI 1.52-2.02), as was having the diagnosis first recorded on the date of death (n=16 [4.2%] vs. n=60 [1.0%]; RR 5.08, 95% CI 2.92-8.84) and stage IV disease at diagnosis (n=89 [23.4%] vs. n=1,178 [19.4%]; RR 1.25, 95% CI 1.01-1.56).
Table 3.
Associations between intellectual disability and outcomes following a first symptom suggestive of bowel cancer.
Table 3.
Associations between intellectual disability and outcomes following a first symptom suggestive of bowel cancer.
| |
No ID (N) |
ID (N) |
RR (95% CI) |
| Total patients with ≥1 symptom |
405,891 |
35,427 |
|
| FIT / FOB test within 28 days of first symptom |
5,070 |
333 |
0.74 (0.67-0.83) |
| Referral within 28 days of first symptom |
9,513 |
479 |
0.57 (0.52-0.63) |
| Endoscopy within 56 days of first symptom |
17,324 |
731 |
0.45 (0.42-0.49) |
| Diagnosis within 56 days of first symptom |
1,448 |
65 |
0.52 (0.41-0.67) |
| Diagnosis within 6 months of first symptom |
2,154 |
117 |
0.64 (0.53-0.77) |
| Diagnosis within 1 year of first symptom |
2,462 |
137 |
0.67 (0.56-0.79) |
| Diagnosis within 3 years of first symptom |
3,035 |
192 |
0.78 (0.68-0.90) |
| Diagnosis within 3 years (inc. MUO / CUP) |
3,411 |
244 |
0.89 (0.78-1.01) |
A total of 95 (80.5%) individuals with an ID underwent curative surgery for stage I-III disease within six months (
Table 4), compared with 1,813 (81.7%) without an ID (RR 0.98, 95% CI 0.79-1.19). In comparison, fewer than five individuals with an ID and 241 (50.6%) without an ID received SACT within six months of diagnosis for stage IV disease (RR 0.15, 95% CI 0.05-0.46). Among all those diagnosed, ID was associated with shorter bowel cancer-specific survival (
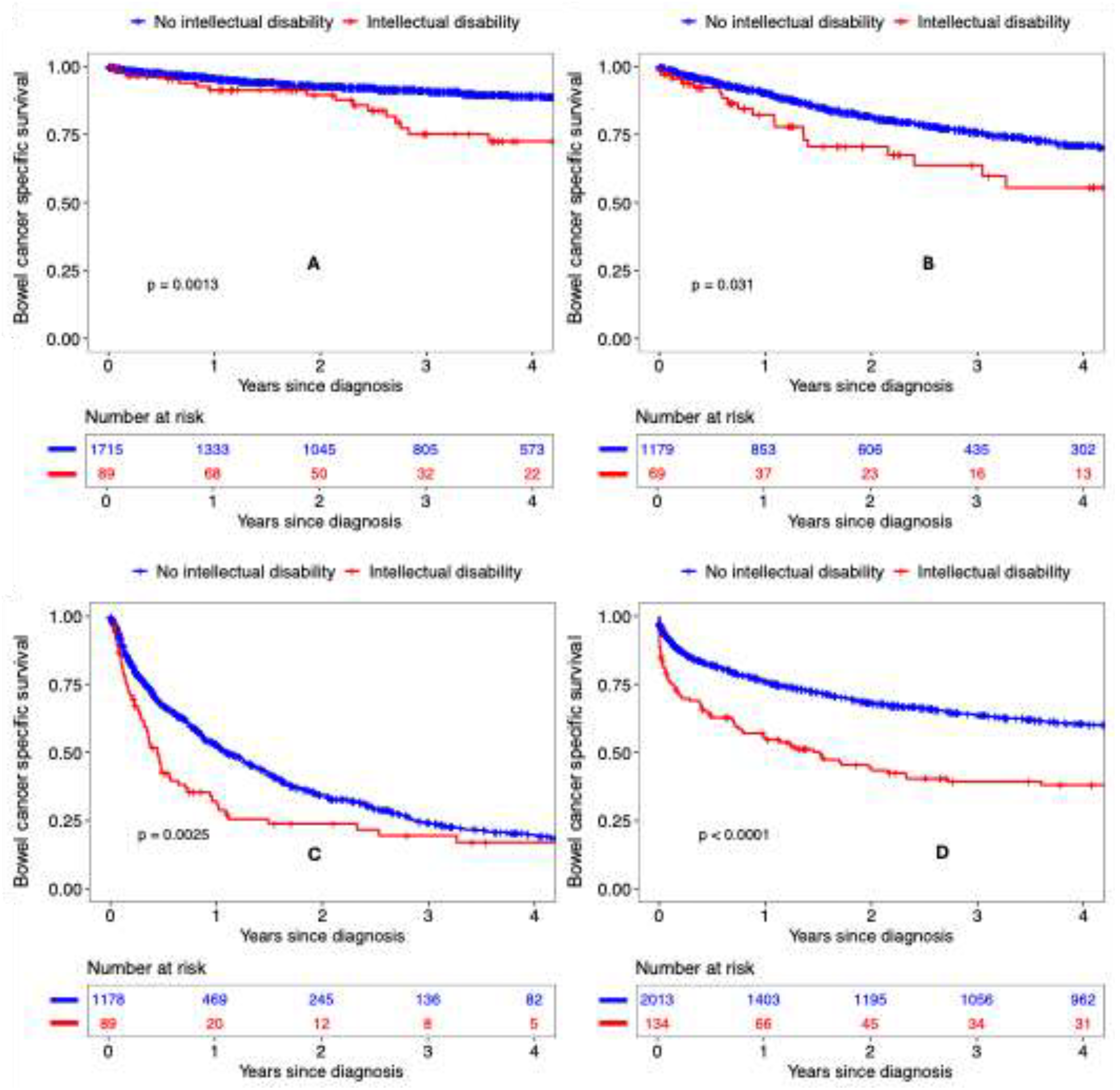
Table 5; HR 2.00, 95% CI 1.71-2.33). This pattern was consistent across all stages (
Figure 2), with particularly pronounced differences in three-year survival for stage I-II disease (75.1% [95% CI 61.4-84.5] for people with an ID vs. 91.1% [95% CI 89.5-92.5] for those without) and one-year survival for stage IV disease (32.0%, 95% CI 21.8-42.7 vs. 52.9%, 95% CI 49.8-56.0).
Table 4.
Associations between intellectual disability and outcomes among those diagnosed with bowel cancer.
Table 4.
Associations between intellectual disability and outcomes among those diagnosed with bowel cancer.
| |
No ID |
ID |
RR (95% CI) |
| Route to diagnosis |
N / total diagnoses* |
N / total diagnoses* |
|
| Screening |
10 / 349 (2.9%) |
580 / 5,571 (10.4%) |
0.27 (0.14-0.50) |
| Urgent suspected cancer referral |
72 / 349 (20.6%) |
1,896 / 5,571 (34.0%) |
0.62 (0.50-0.76) |
| Emergency presentation |
136 / 349 (39.0%) |
1,240 / 5,571 (22.3%) |
1.76 (1.52-2.02) |
| Diagnosis recorded on date of death |
N / total diagnoses |
N / total diagnoses |
|
| |
60 / 6,085 (1.0%) |
16 / 381 (4.2%) |
5.08 (2.92-8.84) |
| Curative surgery within 6 months |
N / total with non-metastatic disease† |
N / total with non-metastatic disease† |
|
| |
1,813 / 2,218 (81.7%) |
95 / 118 (80.5%) |
0.98 (0.79-1.19) |
| SACT within 6 months for metastatic disease |
N / total with metastatic disease†¶ |
N / total with metastatic disease†¶ |
|
| |
241 / 476 (50.6%) |
<5 / 34 |
0.15 (0.05-0.46) |
| Stage at diagnosis |
N |
N |
|
| Stage I |
736 (12.1%) |
36 (9.4%) |
|
| Stage II |
979 (16.1%) |
53 (13.9%) |
|
| Stage III |
1,179 (19.4%) |
69 (18.1%) |
|
| Stage IV |
1,178 (19.4%) |
89 (23.4%) |
1.25 (1.01-1.56)§ |
| Unknown |
2,013 (33.1%) |
134 (35.2%) |
1.08 (0.91-1.29)‖ |
Table 5.
Association between intellectual disability and bowel cancer specific survival.
Table 5.
Association between intellectual disability and bowel cancer specific survival.
| |
No ID (deaths / total) |
ID (deaths / total) |
1-year (no ID) |
1-year (ID) |
3-year (no ID) |
3-year (ID) |
HR (95% CI) |
| All |
2,015 / 6,085 |
176 / 381 |
80.1% (79.1–81.1) |
63.1% (57.8–68.0) |
66.6% (65.3–67.9) |
47.6% (41.7–53.3) |
2.00 (1.71-2.33) |
| Stages 1–2 |
162 / 1,715 |
17 / 89 |
95.5% (94.4–96.5) |
91.3% (82.5–95.8) |
91.1% (89.5–92.5) |
75.1% (61.4–84.5) |
2.34 (1.40-3.90) |
| Stage 3 |
270 / 1,179 |
19 / 69 |
90.3% (88.4–91.9) |
82.3% (69.3–90.1) |
75.6% (72.5–78.4) |
63.7% (47.1–76.4) |
2.13 (1.33-3.41) |
| Stage 4 |
748 / 1,178 |
61 / 89 |
52.9% (49.8–56.0) |
32.0% (21.8–42.7) |
24.2% (21.2–27.3) |
19.5% (10.8–30.1) |
1.63 (1.25-2.13) |
| Unknown |
835 / 2,013 |
79 / 134 |
76.1% (74.1–77.9) |
54.6% (45.6–62.7) |
63.7% (61.5–65.9) |
39.2% (30.3–48.0) |
2.27 (1.79-2.86) |
Figure 2.
Bowel cancer specific survival among individuals with and without an intellectual disability with stage 1-2 (A), 3 (B), 4 (C) and unknown (D) stage disease.
Figure 2.
Bowel cancer specific survival among individuals with and without an intellectual disability with stage 1-2 (A), 3 (B), 4 (C) and unknown (D) stage disease.
Discussion
In this large population-based matched cohort study of over two million individuals, the impact of an ID on the bowel cancer care pathway was assessed. Individuals with an ID were more likely to develop bowel cancer, particularly those under the age of 50. They were also more likely to present to primary care with symptoms associated with bowel cancer but, following such symptoms, were less likely to receive timely investigations, including FIT/FOB testing, referral, and endoscopy. They were less likely to receive a diagnosis of bowel cancer within 56 days, though the total proportions diagnosed with either bowel cancer or MUO/CUP were similar after three years. Bowel cancer was less often diagnosed through screening or urgent suspected cancer referral, and more often diagnosed following an emergency presentation, with stage IV disease, or first recorded on the date of death. ID was associated with reduced likelihood of SACT for stage IV disease, and bowel cancer-specific survival was significantly poorer across all stages.
Considering growing concern about the rising incidence of bowel cancer in younger people, the finding of a twofold higher risk of bowel cancer in individuals with an ID under the age of 50 warrants further investigation. Early-onset bowel cancer is poorly understood, but proposed risk factors include obesity, processed diet, and physical inactivity (33), which may be more common in individuals with an ID (34, 35). As such, individuals with an ID may be disproportionately affected by this trend of increasing early-onset bowel cancer as well as being an under-recognised contributor to it. The elevated risk in those under 50 also indicates that current bowel cancer screening programmes, which in many countries do not begin until age 50, may not be providing equitable benefit for people with an ID, and earlier screening in this population may be merited.
This study identifies specific stages within the bowel cancer diagnostic pathway where disparities may contribute to poorer outcomes for individuals with an ID. Lower rates of FIT/FOB testing, urgent referrals, and endoscopic procedures may result in underdiagnosis and explain the greater likelihood of bowel cancer being identified during emergency presentations, at stage IV disease, or only on the date of death. The reasons for these missed opportunities for investigation remain unclear; however, clinicians may face challenges due to the higher prevalence of bowel-related symptoms in individuals with ID, making it harder to distinguish potentially concerning signs. Additional concerns may include issues around consent or perceived fitness for endoscopy or treatment if cancer is diagnosed. Nonetheless, the findings suggest that improving the investigation of symptoms in this population could facilitate earlier detection and lead to better outcomes.
It should also be noted that while differences in the proportion diagnosed after symptom presentation may indicate under-investigation, they may also reflect reduced specificity of guideline-listed bowel cancer symptoms in individuals with an intellectual disability. In the current study, the proportions diagnosed with bowel cancer in the two groups became more similar over time and were comparable by three years when MUO and CUP cases were included. These may represent bowel cancers that could not be fully investigated. This suggests that although reduced symptom specificity may play a role, under-investigation is likely a major contributing factor.
Among individuals diagnosed with stages I-III disease, receipt of curative surgery within six months was similar by ID status. However, bowel cancer-specific survival was substantially worse in individuals with an ID. Given similar curative surgery rates, this survival gap may be driven by delays in diagnosis and a higher frequency of emergency presentations, which can limit the time available for complete diagnostic workup (e.g. full staging) and coordinated treatment planning. Surgical outcomes may also differ in patients with and without an ID, for example if there were differences in procedure selection (e.g., total mesorectal excision vs transanal approaches due to concerns about morbidity), peri-operative SACT use, complication rates, or postoperative care. For patients with stage IV disease, those with an ID were more than 80% less likely to receive SACT and, accordingly, had markedly shorter survival. The reasons for this are unclear but may include concerns about fitness, capacity, or the ability to self-report adverse events such as fever due to neutropenic sepsis, which carries a high risk of mortality if not promptly treated.
Several earlier studies have investigated bowel cancer outcomes in individuals with an ID, although information on presentation, investigations, referrals, and treatment remains very limited. Most previous studies, similar to the present study, have reported worse bowel cancer-related outcomes in this population. In a cross-sectional study in the Netherlands involving around 100,000 individuals with an ID, Banda et al. (36) reported lower participation in bowel cancer screening (51.7% vs. 72.7%; odds ratio [OR] 0.40, 95% CI 0.41-0.42). In the UK, Heslop et al. (14) analysed the death records of 1,096 adults and found that 36% of cancer deaths in people with ID were due to digestive system cancers, with bowel cancer accounting for roughly half of these. A higher burden of bowel cancer relative to other cancer types in this population, compared with the general population, may be expected given higher rates of obesity but lower rates of smoking and alcohol use (34, 35). A Scottish study found similar incidence rates of bowel cancer in people with and without ID, but higher mortality in the ID group (standardised mortality ratio 1.54) (37). A Canadian study by Mahar et al. (11) found that individuals with intellectual and developmental disabilities diagnosed with bowel cancer were 1.44 times more likely to have stage IV disease at diagnosis compared to those without. Another study in Canada (10) found that ID was associated with a 2.42-fold higher risk of death in patients with bowel cancer. In the United States, an analysis of over 722,000 patients undergoing bowel cancer surgery showed that those with an ID had significantly worse outcomes, including a twofold higher risk of in-hospital mortality, more complications, and substantially greater odds of non-home discharge (38).
Several limitations of this study should be acknowledged. The observational design precludes conclusions about causality. While the large sample size enabled robust overall comparisons, subgroup analyses (e.g., by ethnicity, deprivation, or severity of intellectual disability) were limited by small numbers in some strata. Although matching and adjustment for age and gender reduced confounding, residual confounding cannot be ruled out. The dataset lacked clinical context necessary to assess the appropriateness of individual decisions along the care pathway. For instance, although individuals with an ID were less likely to receive FIT/FOB testing after presenting with symptoms suggestive of bowel cancer, we could not determine whether this reflected appropriate clinical judgment based on the nature, severity, or persistence of symptoms. Similarly, we were unable to assess whether lower rates of investigation or diagnosis were due to clinical ineligibility (e.g., fitness for endoscopy) or other system- or patient-level barriers. Our analysis including MUO/CUP cases, which aimed to capture potentially missed bowel cancers, was limited by the possibility that it also encompassed malignancies not of bowel origin. We were unable to investigate the role of perioperative SACT or chemoradiotherapy in stages I-III disease. Future research should explore these factors in greater depth, including how clinical decision-making, patient characteristics, and organisational barriers may contribute to observed differences. Nonetheless, this study offers the most comprehensive examination to date of bowel cancer diagnosis and outcomes in individuals with ID and identifies key stages in the diagnostic pathway where disparities may emerge, and where targeted interventions could have the greatest impact.
Conclusions
This large, population-based cohort study reveals marked disparities across the bowel cancer diagnostic and care pathway for individuals with an ID. Despite being more likely to report symptoms suggestive of bowel cancer, they were less likely to receive timely investigations, such as FIT/FOB testing, referrals, or endoscopy, and more likely to be diagnosed during emergency presentations or at the time of death. Key diagnostic steps were often missed or delayed, potentially contributing to poorer outcomes. Focused efforts to investigate the causes of these gaps and to develop tailored strategies to improve early detection and care are urgently needed to reduce inequities for people with an ID.
Ethics approval and consent to participate
This study was approved by the Clinical Practice Research Datalink’s (CPRD) independent scientific advisory committee (23_003009). CPRD also has ethical approval from the Health Research Authority to support research using anonymised patient data (research ethics committee reference 21/EM/0265). Individual patient consent was not required as all data were deidentified.
Consent for publication
Not applicable.
Availability of data and materials
Electronic health records are, by definition, considered sensitive data in the UK by the Data Protection Act and cannot be shared via public deposition because of information governance restriction in place to protect patient confidentiality. Access to Clinical Practice Research Datalink (CPRD) data is subject to protocol approval via CPRD’s research data governance process. For more information see
https://cprd.com/data-access. Linked secondary care data from Hospital Episodes Statistics, mortality data from the Office for National Statistics, cancer data from the National Cancer Registration and Analysis Service and index of multiple deprivation data can also be requested from CPRD.
Competing interests
DA and TVS report receiving support for the present work from National Institute for Health and Care Research (NIHR). PL reports grants or contracts from Pierre Fabre, consulting fees from Regeneron, payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from BMS, Pierre Fabre and Merck, and Support for attending meetings and/or travel from Merck.
Author Contributions
Oliver John Kennedy: Conceptualization, Literature Search, Formal analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing. Umesh Chauhan: Conceptualization, Investigation, Methodology, Writing – review & editing. Louise Gorman: Conceptualization, Investigation, Writing – review & editing. Paul Lorigan: Conceptualization, Investigation, Methodology, Writing – review & editing. Supervision. Samuel Merriel: Conceptualization, Investigation, Methodology, Writing – review & editing. Supervision. Antonia Perumal: Data collection & curation. Tjeerd Van Staa: Conceptualization, Investigation, Methodology, Writing – review & editing. Alison Wright: Conceptualization, Data collection & curation, Investigation, Methodology, Writing – review & editing. Darren Mark Ashcroft: Conceptualization, Investigation, Methodology, Writing – review & editing. Supervision, Project administration. Oliver John Kennedy and Alison Wright had full access to and verified the underlying data. Darren Mark Ashcroft had final responsibility for the decision to submit for publication.
Funding
NIHR Greater Manchester Patient Safety Research Collaboration (NIHR204295).
Acknowledgments
Oliver John Kennedy is funded by the National Institute for Health and Care Research (NIHR) Manchester Biomedical Research Centre (BRC) (NIHR203308). This study is based on data from the Clinical Practice Research Datalink obtained under licence from the UK Medicines and Healthcare Products Regulatory Agency (MHRA). The data are provided by patients and collected by the NHS as part of its care and support. Hospital Episode Statistics, National Cancer Registration and Analysis Service (NCRAS) data and Office for National Statistics mortality data are subject to Crown copyright (2025) protection, reused with the permission of The Health and Social Care Information Centre, all rights reserved. The interpretation and conclusions contained in this study are those of the authors alone, and not necessarily those of the National Institute of Health and Care Research, NHS, or Department of Health and Social Care. We would like to acknowledge all the data providers and general practices who make anonymised data available for research.
References
- National Institute of Health and Care Excellence. Clinical Knowledge Summaries. Learning Disabilities. 2023. Available at: https://cks.nice.org.uk/topics/learning-disabilities/background-information/definition/ (accessed 27 May 2025). .
- Nair R, Chen M, Dutt AS, Hagopian L, Singh A, Du M. Significant regional inequalities in the prevalence of intellectual disability and trends from 1990 to 2019: a systematic analysis of GBD 2019. Epidemiol Psychiatr Sci. 2022;31:e91.
- Parkin, E. House of Commons Library. Research Briefing. Learning disabilities: health policies. Available at: https://researchbriefings.files.parliament.uk/documents/SN07058/SN07058.pdf (accessed 1 May 2025).
- NHS England. Learning from lives and deaths - People with a learning disability and autistic people (LeDeR). Action from learning report 2022/23. Available at: https://leder.nhs.uk/images/resources/action-from-learning-report-22-23/20231019_LeDeR_action_from_learning_report_FINAL.pdf (accessed 1 May 2025).
- NHS England. The NHS Long Term Plan. 2019. Available at: www.longtermplan.nhs.uk/publication/nhs-long-term-plan (accessed 21 May 2025).
- National Institute for Health and Care Excellence. NICE guideline: NG96. Care and support of people growing older with learning disabilities. 2018. Available at: https://www.nice.org.uk/guidance/ng96/chapter/recommendations (accessed 1 May 2025).
- Glover G, Williams R, Heslop P, Oyinlola J, Grey J. Mortality in people with intellectual disabilities in England. J Intellect Disabil Res. 2017;61(1):62-74.
- Banda A, Naaldenberg J, Timen A, van Eeghen A, Leusink G, Cuypers M. Cancer risks related to intellectual disabilities: A systematic review. Cancer Med. 2024;13(9):e7210.
- The Lancet O. Intellectual and developmental disabilities-an under-recognised driver of cancer mortality. Lancet Oncol. 2024;25(4):411.
- Hansford RL, Ouellette-Kuntz H, Griffiths R, Hallet J, Decker K, Dawe DE, et al. Breast (female), colorectal, and lung cancer survival in people with intellectual or developmental disabilities: A population-based retrospective cohort study. Can J Public Health. 2024;115(2):332-42.
- Mahar AL, Biggs K, Hansford RL, Derksen S, Griffiths R, Enns JE, et al. Stage IV breast, colorectal, and lung cancer at diagnosis in adults living with intellectual or developmental disabilities: A population-based cross-sectional study. Cancer. 2024;130(5):740-9.
- Liu Q, Adami HO, Reichenberg A, Kolevzon A, Fang F, Sandin S. Cancer risk in individuals with intellectual disability in Sweden: A population-based cohort study. PLoS Med. 2021;18(10):e1003840.
- NHS England. Health and Care of People with Learning Disabilities, Experimental Statistics 2021 to 2022. Available at https://digital.nhs.uk/data-and-information/publications/statistical/health-and-care-of-people-with-learning-disabilities/experimental-statistics-2021-to-2022 (accessed 12 July 2025).
- Heslop P, Cook A, Sullivan B, Calkin R, Pollard J, Byrne V. Cancer in deceased adults with intellectual disabilities: English population-based study using linked data from three sources. BMJ Open. 2022;12(3):e056974.
- Wolf A, Dedman D, Campbell J, Booth H, Lunn D, Chapman J, et al. Data resource profile: Clinical Practice Research Datalink (CPRD) Aurum. Int J Epidemiol. 2019;48(6):1740-g.
- Medicines and Healthcare products Regulatory Agency. Clinical Practice Research Datalink. CPRD Ethnicity Records. Avilable at: https://www.cprd.com/data/algorithm-derived-data/cprd-ethnicity-records (accessed 2 October 2025).
- Eggermont AMM, Blank CU, Mandala M, Long GV, Atkinson VG, Dalle S, et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma (EORTC 1325-MG/KEYNOTE-054): distant metastasis-free survival results from a double-blind, randomised, controlled, phase 3 trial. Lancet Oncol. 2021;22(5):643-54.
- Davidson, J and Warren-Gash, C (2022). Clinical codelist - CPRD Aurum - learning and intellectual disability. [Data Collection]. London School of Hygiene & Tropical Medicine, London, United Kingdom. (accessed 4 December 2024). [CrossRef]
- Root, A (2019). Clinical Codelist - Intellectual Disability Codes. [Data Collection]. London School of Hygiene & Tropical Medicine, London, United Kingdom. (accessed 9 November 2024). [CrossRef]
- Tazare J. OpenSAFELY. OpenCodelists. Intellectual disability. Codelist ID: opensafely/intellectual-disability. 2020. Available at: https://www.opencodelists.org/codelist/opensafely/intellectual-disability/2020-08-27/ (accessed 30 April 2025).
- Kontopantelis E, Olier I, Planner C, Reeves D, Ashcroft DM, Gask L, et al. Primary care consultation rates among people with and without severe mental illness: a UK cohort study using the Clinical Practice Research Datalink. BMJ Open. 2015;5(12):e008650.
- Reilly S, Olier I, Planner C, Doran T, Reeves D, Ashcroft DM, et al. Inequalities in physical comorbidity: a longitudinal comparative cohort study of people with severe mental illness in the UK. BMJ Open. 2015;5(12):e009010.
- Carey IM, Hosking FJ, Harris T, DeWilde S, Beighton C, Cook DG. An evaluation of the effectiveness of annual health checks and quality of health care for adults with intellectual disability: an observational study using a primary care database. Health Services and Delivery Research. Southampton (UK)2017.
- Harvey PR, Thomas T, Chandan JS, Bhala N, Nirantharakumar K, Trudgill NJ. Outcomes following feeding gastrostomy (FG) insertion in patients with learning disability: a retrospective cohort study using the health improvement network (THIN) database. BMJ Open. 2019;9(6):e026714.
- OpenSAFELY. OpenCodelists. Learning disabilities. Codelist ID: opensafely/learning-disabilities. Available at: https://www.opencodelists.org/codelist/opensafely/learning-disabilities/2020-07-06/ (accessed 30th April 2025).
- Hippisley-Cox, J. QResearch. QCode Group Library. Learning disability (LD) codes from QOF excl downs. Available at: https://www.qresearch.org/data/qcode-group-library/ (accessed 30 April 2025). .
- . Public Health England. Learning Disabilities Observatory. People with learning disabilities in England 2015: Main report. Version 1.0. November 2016.
- Department for Education. Special Educational Needs Information Act. An Analysis 2011. Available at: https://assets.publishing.service.gov.uk/media/5a7c1419e5274a25a9140514/main_20text_20osr202011.pdf (accessed 1 May 2025).
- Watson J, Nicholson BD, Hamilton W, Price S. Identifying clinical features in primary care electronic health record studies: methods for codelist development. BMJ Open. 2017;7(11):e019637.
- Sung H, Siegel RL, Laversanne M, Jiang C, Morgan E, Zahwe M, et al. Colorectal cancer incidence trends in younger versus older adults: an analysis of population-based cancer registry data. Lancet Oncol. 2025;26(1):51-63.
- Wang SV, Schneeweiss S. A Framework for Visualizing Study Designs and Data Observability in Electronic Health Record Data. Clin Epidemiol. 2022;14:601-8.
- Benchimol EI, Smeeth L, Guttmann A, Harron K, Moher D, Petersen I, et al. The REporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement. PLoS Med. 2015;12(10):e1001885.
- Patel SG, Karlitz JJ, Yen T, Lieu CH, Boland CR. The rising tide of early-onset colorectal cancer: a comprehensive review of epidemiology, clinical features, biology, risk factors, prevention, and early detection. Lancet Gastroenterol Hepatol. 2022;7(3):262-74.
- Public Health England. Guidance. Obesity and weight management for people with learning disabilities. 2020.
- Public Health England. Guidance. Substance misuse in people with learning disabilities: reasonable adjustments guidance. 2016.
- Banda A, Cuypers M, Naaldenberg J, Timen A, Leusink G. Cancer screening participation and outcomes among people with an intellectual disability in the Netherlands: a cross-sectional population-based study. Lancet Public Health. 2025;10(3):e237-e45.
- Ward LM, Cooper SA, Sosenko F, Morrison D, Fleming M, McCowan C, et al. Population-based cancer incidence and mortality rates and ratios among adults with intellectual disabilities in Scotland: a retrospective cohort study with record linkage. BMJ Open. 2024;14(8):e084421.
- Ng AP, Kim S, Chervu N, Gao Z, Mallick S, Benharash P, et al. Disparities in outcomes of colorectal cancer surgery among adults with intellectual and developmental disabilities. PLoS One. 2024;19(8):e0308938.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).