Submitted:
27 February 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Agronomical Performance
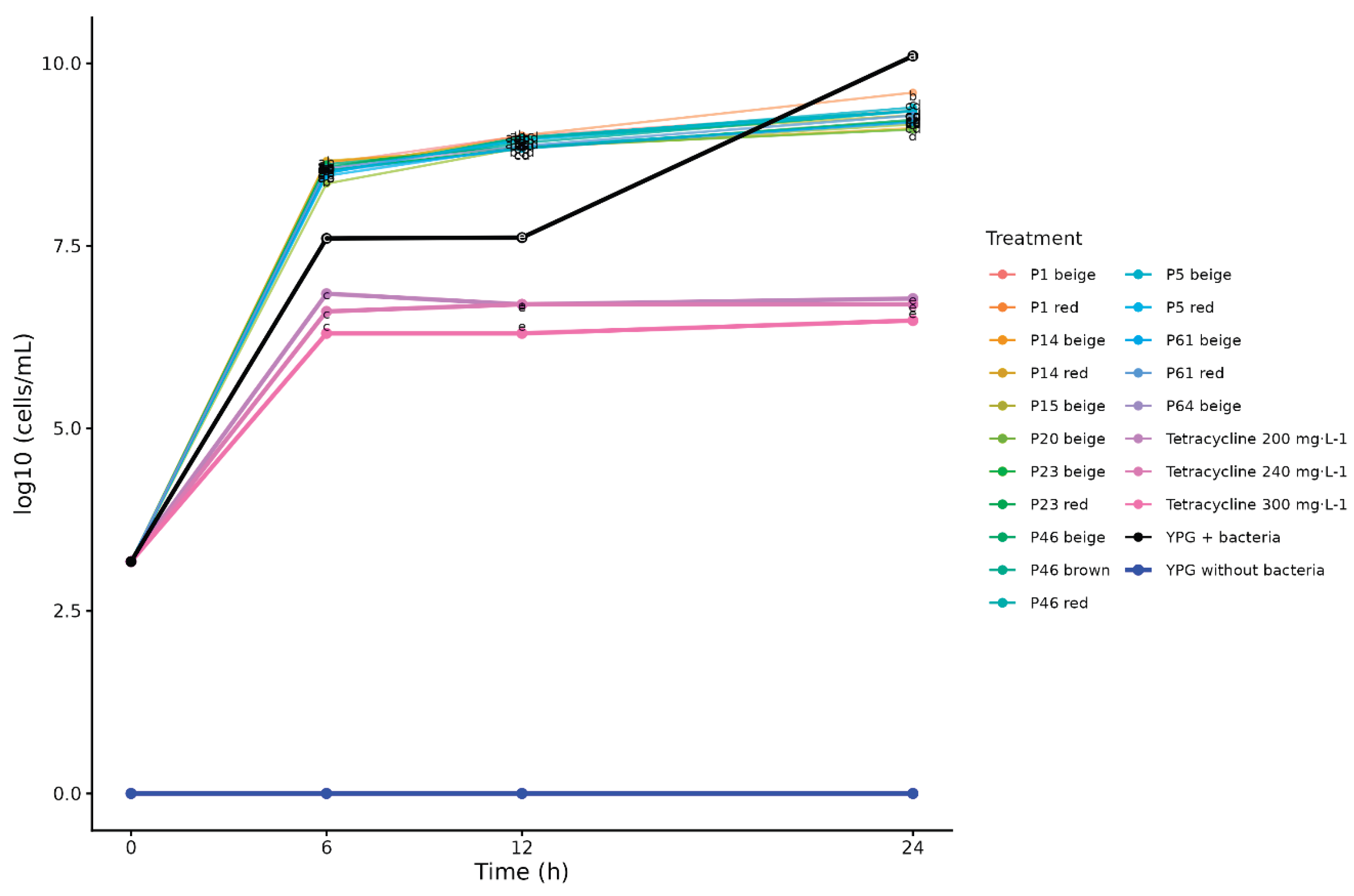
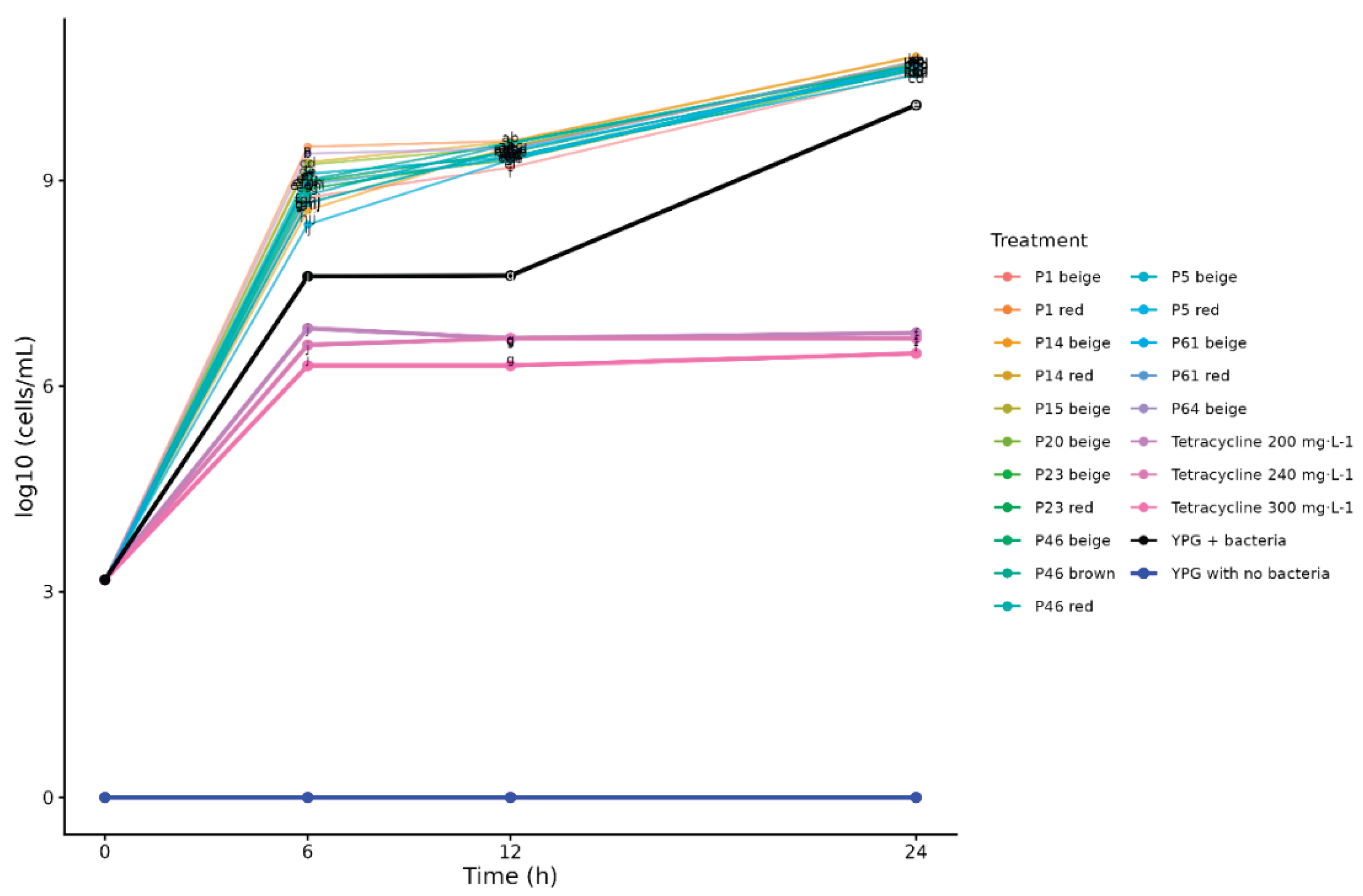
2.2. Effect of 0.5% Yam Bean Extracts
2.3. Effect of 5% Yam Bean Extracts
3. Discussion
3.1. Seed Yield and Agronomical Traits
3.2. Effect of Pachyrhizus Extracts
3.3. Limitations and Implications
4. Materials and Methods
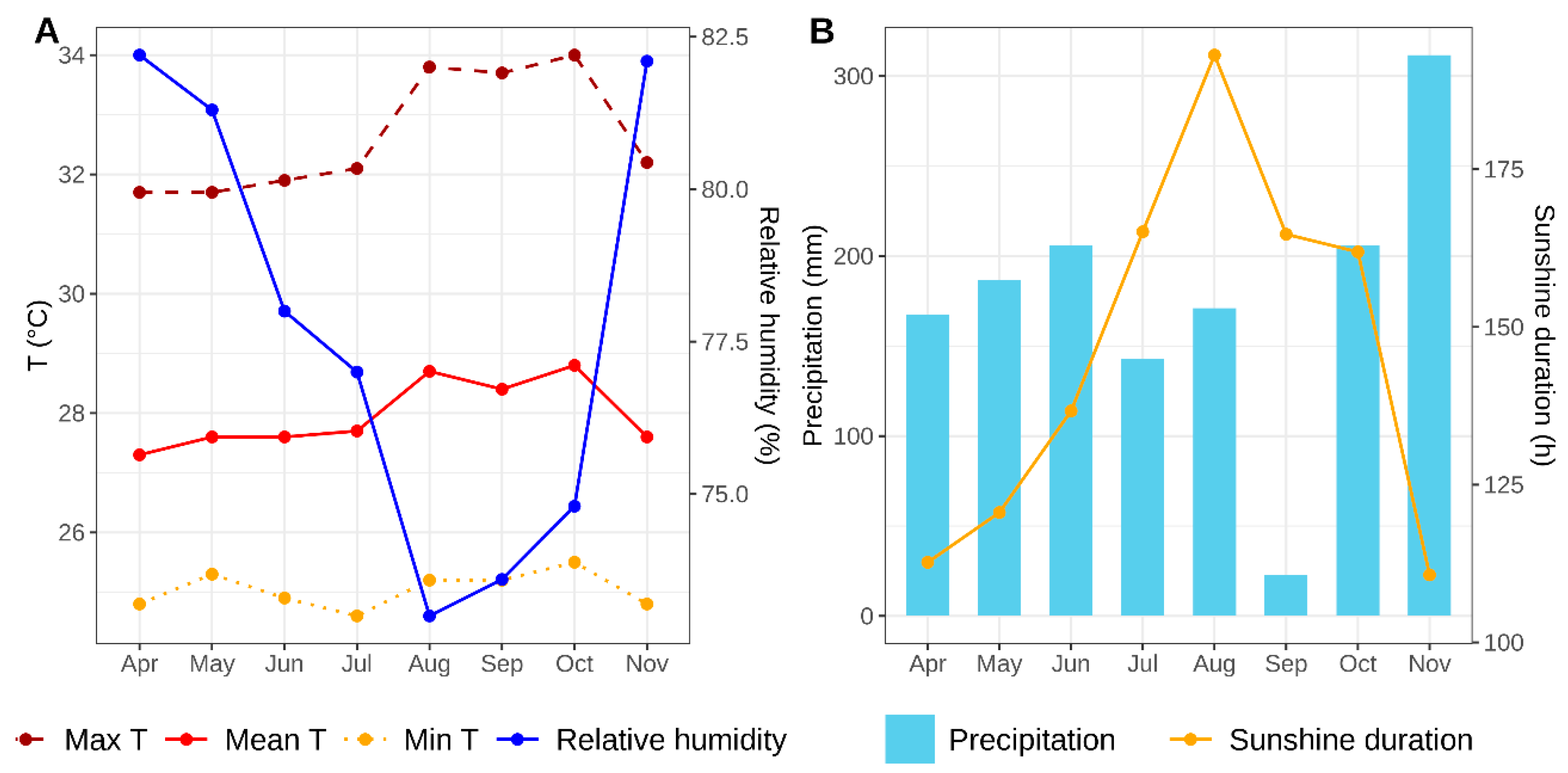
4.1. Field Experiment
4.1.1. Location
4.1.2. Material
4.1.3. Soil Preparation
4.1.4. Seedling Preparation and Field Establishment
4.1.5. Cultivation
4.1.6. Harvest and Evaluation
4.2. In Vitro Experiment
4.2.1. Material
4.2.2. Preparation of Aqueous Seed Extracts
4.2.3. In Vitro Toxicity Assay
4.2.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| RS | Ralstonia solanacearum |
| YPG | yeast-peptone-glucose |
| INPA | National Institute for Amazonian Research |
| LD50 | median lethal dose |
References
- Verma, N.S.; Kuldeep, D.K.; Chouhan, M.; Prajapati, R.; Singh, S.K. A Review on Eco-Friendly Pesticides and Their Rising Importance in Sustainable Plant Protection Practices. Int. J. Plant Soil Sci. 2023, 35, 200–214. [Google Scholar] [CrossRef]
- Gundreddy, R.R.; Bv, J.; M, S.; Darjee, S.; Alekhya, G. Recent Advances in Biopesticide Formulations for Targeted Insect Pest Management: A Review. J. Exp. Agric. Int. 2024, 46, 876–889. [Google Scholar] [CrossRef]
- Tembo, Y.; Mkindi, A.G.; Mkenda, P.A.; Mpumi, N.; Mwanauta, R.; Stevenson, P.C.; Ndakidemi, P.A.; Belmain, S.R. Pesticidal Plant Extracts Improve Yield and Reduce Insect Pests on Legume Crops Without Harming Beneficial Arthropods. Front. Plant Sci. 2018, 9, 1425. [Google Scholar] [CrossRef] [PubMed]
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The Global Burden of Pathogens and Pests on Major Food Crops. Nat. Ecol. Evol. 2019, 3, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, J.; Amin, H. Implication of Biopesticides for Insect Pest Management: An Alternative of Chemical Pesticides. Bangladesh J. Entomol. 2023, 31. [Google Scholar] [CrossRef]
- Sørensen, M. Yam Bean: Pachyrhizus DC.—Promoting the Conservation and Use of Underutilized and Neglected Crops. 4; Bioversity International (IPGRI): Rome, Italy, 1996; ISBN 92-9043-293-8. [Google Scholar]
- Nielsen, P.E.; Sørensen, M.; Halafihi, M. Yield Potential of Yam Bean Pachyrhizus erosus (L.) Urban Accessions in the Kingdom of Tonga, South Pacific. Trop. Agric. 2000, 77, 174–179. [Google Scholar]
- Tapia, C.; Sørensen, M. Morphological Characterization of the Genetic Variation Existing in a Neotropical Collection of Yam Bean, Pachyrhizus tuberosus (Lam.) Spreng. Genet. Resour. Crop Evol. 2003, 50, 681–692. [Google Scholar] [CrossRef]
- Zanklan, A.S.; Ahouangonou, S.; Becker, H.C.; Pawelzik, E.; Grüneberg, W.J. Evaluation of the Storage Root-Forming Legume Yam Bean (Pachyrhizus spp.) under West African Conditions. Crop Sci. 2007, 47, 1934–1946. [Google Scholar] [CrossRef]
- Silva, E.S.D.; Silva Filho, D.F.D.; Ticona-Benavente, C.A. Diversity of Yam Bean (Pachyrhizus spp. Fabaceae) Based on Morphoagronomic Traits in the Brazilian Amazon. Acta Amaz. 2016, 46, 233–240. [Google Scholar] [CrossRef]
- de Lima, R.G.; Ticona-Benavente, C.E.A.; da Silva Filho, D.F.; e Silva, L.S.; Guimarães, R.G.V.; Hanada, R.E. Pachyrhizus Spp. Seed Extracts Control the Sclerotium rolfsii Sacc. Mycelial Growth. J. Med. Plants Res. 2021, 15, 289–296. [Google Scholar] [CrossRef]
- Silva, T.M.; Guimarães, R.G.; Martins, A.K.; Silva Filho, D.F.; Ticona-Benavente, C.A. Pachyrhizus Toxicity: Genetic Variability of Mature and Immature Seeds and Its Effects on Sclerotium rolfsii and Ralstonia solanacearum. J. Med. Plants Res. 2023, 17, 8–15. [Google Scholar] [CrossRef]
- Barrera-Necha, L.; Bautista-Baños, S.; Bravo-Luna, L.; García-Suárez, F.; Alavez-Solano, D.; Reyes-Chilpa, R. Antifungal Activity of Seed Powders, Extracts, and Secondary Metabolites of Pachyrhizus erosus (L.) Urban (Fabaceae) against Three Postharvest Fungi. Rev. Mex. Fitopatol. 2004, 22, 356–361. [Google Scholar]
- Zhang, H.; Xu, D.; Pan, R.; Xu, H. Screening for antimicrobial activities in some plant extracts. Acta Phytopathol. Sin. 2008, 38, 441–444. [Google Scholar]
- Agustini, M.; Sartiami, D. Dadang Effectiveness of soursop leaves, yam bean seeds, and javanese long pepper fruits extracts against larvae of fall armyworm (Spodoptera frugiperda (Smith)) (Lepidoptera: Noctuidae). J. Entomol. Indones. Indones. J. Entomol. 2024, 21, 224–233. [Google Scholar] [CrossRef]
- Ravindranath, N.; Samanta, A.; Samanta, S. Dose-Dependent Toxicity and Field Efficacy of PEG-Emulsified Botanical Formulations against Chickpea Pod Borer (Helicoverpa Armigera). J. Entomol. Res. 2025, 49, 632–637. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, P. Efficacy of Certain Insecticides against Bemisia tabaci (Gennadius) in Okra. Indian J. Entomol. 2022, 84, 348–349. [Google Scholar] [CrossRef]
- Basukriadi, A.; Wilkins, R. Oviposition Deterrent Activities of Pachyrhizus erosus Seed Extract and Other Natural Products on Plutella xylostella (Lepidoptera: Plutellidae). J. Insect Sci. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Phrutivorapongkul, A.; Lipipun, V.; Ruangrungsi, N.; Watanabe, T.; Ishikawa, T. Studies on the Constituents of Seeds of Pachyrrhizus erosus and Their Anti Herpes Simplex Virus (HSV) Activities. Chem. Pharm. Bull. (Tokyo) 2002, 50, 534–537. [Google Scholar] [CrossRef]
- Lautié, E.; Rozet, E.; Hubert, P.; Leclercq, J.Q. Quantification of Rotenone in Seeds of Different Species of Yam Bean (Pachyrhizus sp.) by a SPE HPLC–UV Method. Food Chem. 2012, 131, 1531–1538. [Google Scholar] [CrossRef]
- Lautié, E.; Rozet, E.; Hubert, P.; Vandelaer, N.; Billard, F.; Zum Felde, T.; Grüneberg, W.J.; Quetin-Leclercq, J. Fast Method for the Simultaneous Quantification of Toxic Polyphenols Applied to the Selection of Genotypes of Yam Bean (Pachyrhizus sp.) Seeds. Talanta 2013, 117, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Leuner, O.; Havlik, J.; Budesinsky, M.; Vrkoslav, V.; Chu, J.; Bradshaw, T.D.; Hummelova, J.; Miksatkova, P.; Lapcik, O.; Valterova, I.; et al. Cytotoxic Constituents of Pachyrhizus tuberosus from Peruvian Amazon. Nat. Prod. Commun. 2013, 8, 1423–1426. [Google Scholar] [CrossRef]
- Catteau, L.; Lautié, E.; Koné, O.; Coppée, M.; Hell, K.; Pomalegni, C.; Quetin-Leclercq, J. Degradation of Rotenone in Yam Bean Seeds (Pachyrhizus sp.) through Food Processing. J. Agric. FOOD Chem. 2013, 61, 11173–11179. [Google Scholar] [CrossRef]
- Song, X.; Wang, J.; Wu, F.; Li, X.; Teng, M.; Gong, W. cDNA Cloning, Functional Expression and Antifungal Activities of a Dimeric Plant Defensin SPE10 from Pachyrrhizus erosus Seeds. Plant Mol. Biol. 2005, 57, 13–20. [Google Scholar] [CrossRef]
- Cabras, P.; Caboni, P.; Cabras, M.; Angioni, A.; Russo, M. Rotenone Residues on Olives and in Olive Oil. J. Agric. Food Chem. 2002, 50, 2576–2580. [Google Scholar] [CrossRef] [PubMed]
- Isman, M.B. Botanical Insecticides in the Twenty-First Century—Fulfilling Their Promise? Annu. Rev. Entomol. 2020, 65, 233–249. [Google Scholar] [CrossRef] [PubMed]
- De Wilde, A.R.; Heyndrickx, A.; Carton, D. A Case of Fatal Rotenone Poisoning in a Child. J. Forensic Sci. 1986, 31, 1492–1498. [Google Scholar] [CrossRef] [PubMed]
- Mascaro, U.C.P.; Rodrigues, L.A.; Bastos, J.K.; Santos, E.; Costa, J.P. Valores de DL50 Em Peixes e No Rato Tratados Com Pó de Raízes de Derris spp. e Suas Implicações Ecotoxicológicas. Pesqui. Veterinária Bras. 1998, 18, 53–56. [Google Scholar] [CrossRef]
- Cavoski, I.; Caboni, P.; Sarais, G.; Miano, T. Degradation and Persistence of Rotenone in Soils and Influence of Temperature Variations. J. Agric. Food Chem. 2008, 56, 8066–8073. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhang, N.; Wang, K.; Li, W.; Li, H.; Zhang, Z. Dissipation and Residue of Rotenone in Cabbage and Soil under Field Conditions. Bull. Environ. Contam. Toxicol. 2013, 91, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.S.; Atroch, A.L.; de Jesus, R.S.; Bitencourt, M.A.F.; Ticona-Benavente, C.A. Genotype x Year Interaction for Agronomical Traits and Proximate Composition in Yam Beans (Pachyrhizus spp., Fabaceae) in the Brazilian Amazon. Genet. Mol. Res. 2022, 21, 1–12. [Google Scholar] [CrossRef]
- Netto, R.A.C.; Pereira, B.G.; Noda, H.; Boher, B. Murcha Bacteriana No Estado Do Amazonas, Brasil. Fitopatol. Bras. 2004, 29, 17–23. [Google Scholar] [CrossRef]
- Costa, S.B.; Ferreira, M.A.S.V.; Lopes, C.A. Diversidade Patogênica e Molecular de Ralstonia solanacearum Da Região Amazônica Brasileira. Fitopatol. Bras. 2007, 32, 285–294. [Google Scholar] [CrossRef]
- Cernay, C.; Pelzer, E.; Makowski, D. A Global Experimental Dataset for Assessing Grain Legume Production. Sci. Data 2016, 3, 160084. [Google Scholar] [CrossRef]
- Bawari, M.R.; Narendrappa, T. In-Vitro Evaluation of Bioagents and Antibiotics against Ralstonia solanacearum Causing Brinjal Wilt. J. Pharmacogn. Phytochem. 2019, 8, 2038–2041. [Google Scholar]
- Santás-Miguel, V.; Díaz-Raviña, M.; Martín, A.; García-Campos, E.; Barreiro, A.; Núñez-Delgado, A.; Álvarez-Rodríguez, E.; Arias-Estévez, M.; Fernández-Calviño, D. Soil Enzymatic Activities and Microbial Community Structure in Soils Polluted with Tetracycline Antibiotics. Agronomy 2021, 11, 906. [Google Scholar] [CrossRef]
- Leipold, M.; Tausch, S.; Hirtreiter, M.; Poschlod, P.; Reisch, C. Sampling for Conservation Genetics: How Many Loci and Individuals Are Needed to Determine the Genetic Diversity of Plant Populations Using AFLP? Conserv. Genet. Resour. 2020, 12, 99–108. [Google Scholar] [CrossRef]
- McGlaughlin, M.E.; Riley, L.; Brandsrud, M.; Arcibal, E.; Helenurm, M.K.; Helenurm, K. How Much Is Enough? Minimum Sampling Intensity Required to Capture Extant Genetic Diversity in Ex Situ Seed Collections: Examples from the Endangered Plant Sibara filifolia (Brassicaceae). Conserv. Genet. 2015, 16, 253–266. [Google Scholar] [CrossRef]
- Castillo, J.A.; Greenberg, J.T. Evolutionary Dynamics of Ralstonia solanacearum. Appl. Environ. Microbiol. 2007, 73, 1225–1238. [Google Scholar] [CrossRef]

| Progeny | Stand | Pod yield (t·ha−1) |
Pod length (cm) |
Pod width (cm) |
Pods per plant | 100-seeds mass (g) | Seed yield (t·ha−1) |
|---|---|---|---|---|---|---|---|
| P1 | 3.50† | 1.88† | 11.15 a | 1.41 abc | 52.95† | 21.70 ab | 0.80† |
| P5 | 3.50 | 1.47 | 10.90 a | 1.46 a | 41.12 | 23.20 a | 0.56 |
| P14 | 4.00 | 1.84 | 11.11 a | 1.37 bcd | 55.56 | 19.63 cd | 0.83 |
| P15 | 3.75 | 1.50 | 10.70 a | 1.40 abc | 47.50 | 20.93 bc | 0.66 |
| P20 | 4.00 | 1.98 | 10.90 a | 1.35 cd | 56.31 | 20.90 bc | 0.86 |
| P23 | 3.75 | 2.21 | 11.30 a | 1.46 a | 59.70 | 22.2 ab | 0.98 |
| P46 | 3.50 | 1.49 | 10.80 a | 1.40 abc | 44.81 | 18.99 d | 0.61 |
| P61 | 3.75 | 1.69 | 10.91 a | 1.43 ab | 48.33 | 22.64 a | 0.74 |
| P64 | 3.50 | 1.57 | 10.07 b | 1.32 d | 48.60 | 20.52 bcd | 0.69 |
| Mean | 3.70 | 1.74 | 10.90 | 1.40 | 50.55 | 21.20 | 0.80 |
| CV (%) | 14.21 | 32.20 | 3.10 | 2.90 | 30.83 | 5.00 | 35.42 |
| R2 | 0.31 | 0.36 | 0.61 | 0.65 | 0.35 | 0.74 | 0.34 |
| H2 | 0 | 0 | 0.77 | 0.81 | 0 | 0.85 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




