1. Introduction
Sweet taste is one of the most preferred sensory modalities in humans. It is detected by taste receptor cells (TRCs) that are found in taste buds across the mouth, including the tongue, palate, and throat [
1]. It is now well-known that sweet taste is primarily detected by the human sweet taste receptor-a heterodimeric family C G protein-coupled receptor (GPCR) Tas1R2/Tas1R3 (taste 1 receptor member 2/taste 1 receptor member 3), which is expressed in taste buds on the tongue [
2]. However, sweet taste perception is not solely dependent on the activation of the sweet taste receptor and it is also influenced by interactions with other sensory systems. Previous research on the activity of sweeteners in inducing taste receptors has mainly focused on the interaction between individual sweeteners and sweet taste receptor, while the relationship between other receptors and sweeteners was relatively less studied. Understanding these interactions is essential for developing food with optimal sensory characteristics and for addressing issues related to overconsumption of sweet substances. Meanwhile, these interactions can modulate the intensity, quality and hedonic value of sweet taste, making it a complex and multifaceted sensory experience.
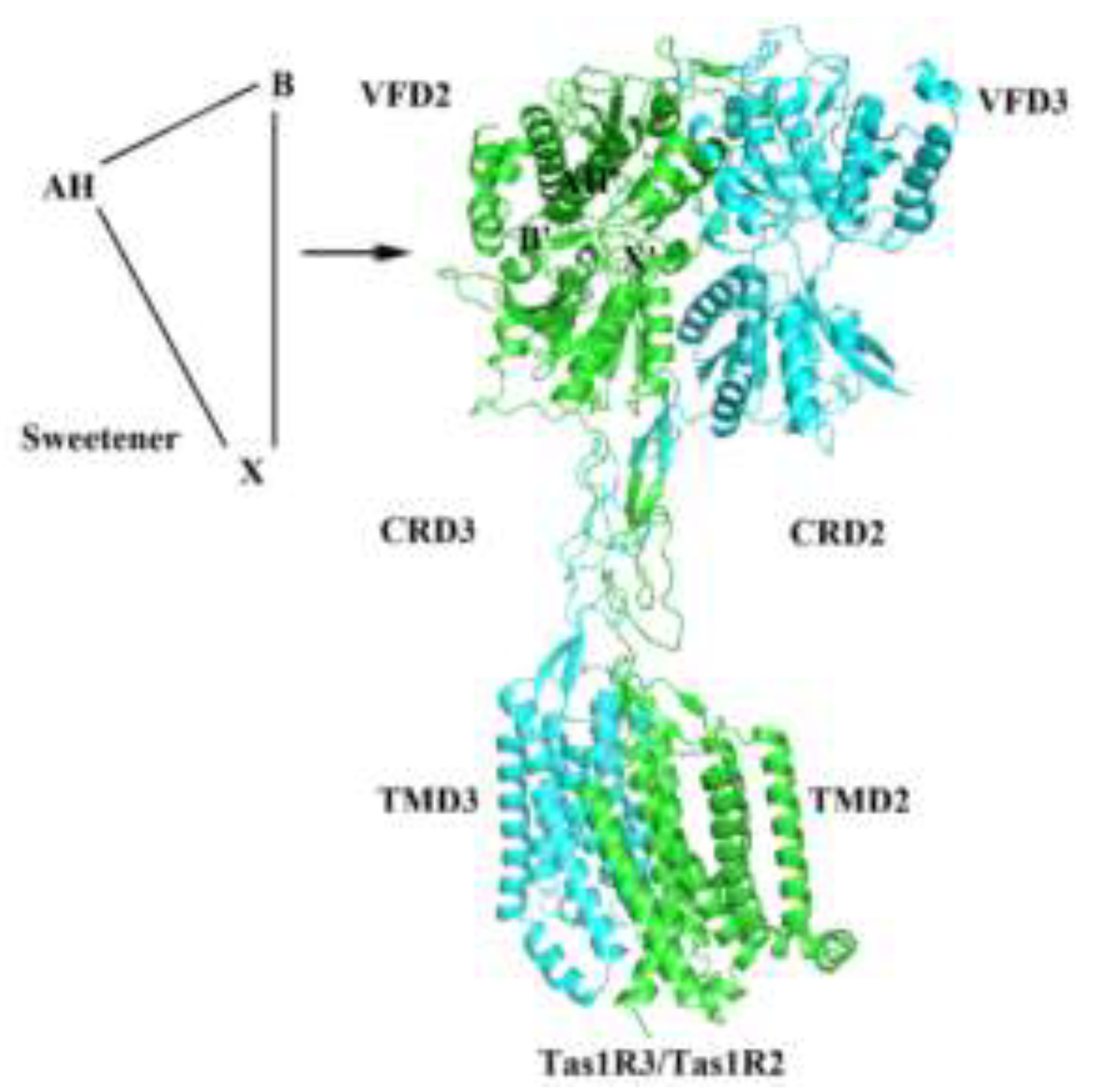
The first partial crystal structure of a sweet taste receptor was determined by Nango et al. [
3], who used X-ray crystallography to analyze the Venus flytrap domain (VFD) of the medaka fish Tas1R2/Tas1R3 sweet receptor and obtained its co-crystal structure, and the ability of the VFD to bind sweet ligands was confirmed. Recently, it was reported that the first cryo-EM structures of the full-length human Tas1R2/Tas1R3 in both their unliganded (apo) and sucralose bound states were elucidated [
4]. The structural characteristics agonist binding mode and potential activation mechanism of the receptor were elucidated, which provided an important molecular basis for understanding sweet taste perception.
First proposed by Shallenberger and Acree in 1967 to explain the interaction mechanism between sweet-tasting molecules and sweet taste receptor, this theory demonstrated that a sweet-tasting molecule must contain both a hydrogen donor (AH) and a proton acceptor (B) within a distance of 2.5–4 Å [
5]. Subsequently, the concept of a third hydrophobic (X) site in proper array with AH and B was supplemented by Kier et al. [
6]. Recently, it was proposed that sweeteners and the sweet taste receptor Tas1R2/Tas1R3 exhibit analogous intramolecular connectivity patterns, including electrostatic complementarity and topochemical compatibility, which mediate their complementary interaction and account for the common molecular feature of sweetness generation (
Figure 1) [
7]. They also highlighted the synergistic role of multiple glucophores in a topological system in eliciting and enhancing sweetness, presenting a framework for the structure-activity relationship-based molecular design and modification of sweeteners (
Figure 2).
2. Interaction of Sweet Taste with Other Taste Modalities
The taste sense of mammals can be divided into five categories: sour, sweet, bitter, salty, and umami, which are crucial for the evaluation of the quality of food [
8]. Therefore, the perception of different tastants can enable humans and other species to select beneficial nutrients or discard potentially detrimental substances. It is currently clear that sweet, bitter, and umami tastes are mediated by the Tas1R and Tas2R receptor families of G protein-coupled receptors (GPCRs) [
9], while saltiness and sourness are mediated by the ion channels ENaC [
10] and Otopetrin 1 [
11,
12,
13], respectively (
Table 1). Results of several research groups [
14,
15,
16,
17,
18] showed that a low concentration of sweet taste had both enhanced and suppressed effects on different taste sensations. At the medium or high concentration level, the sweet taste may mask other taste sensations.
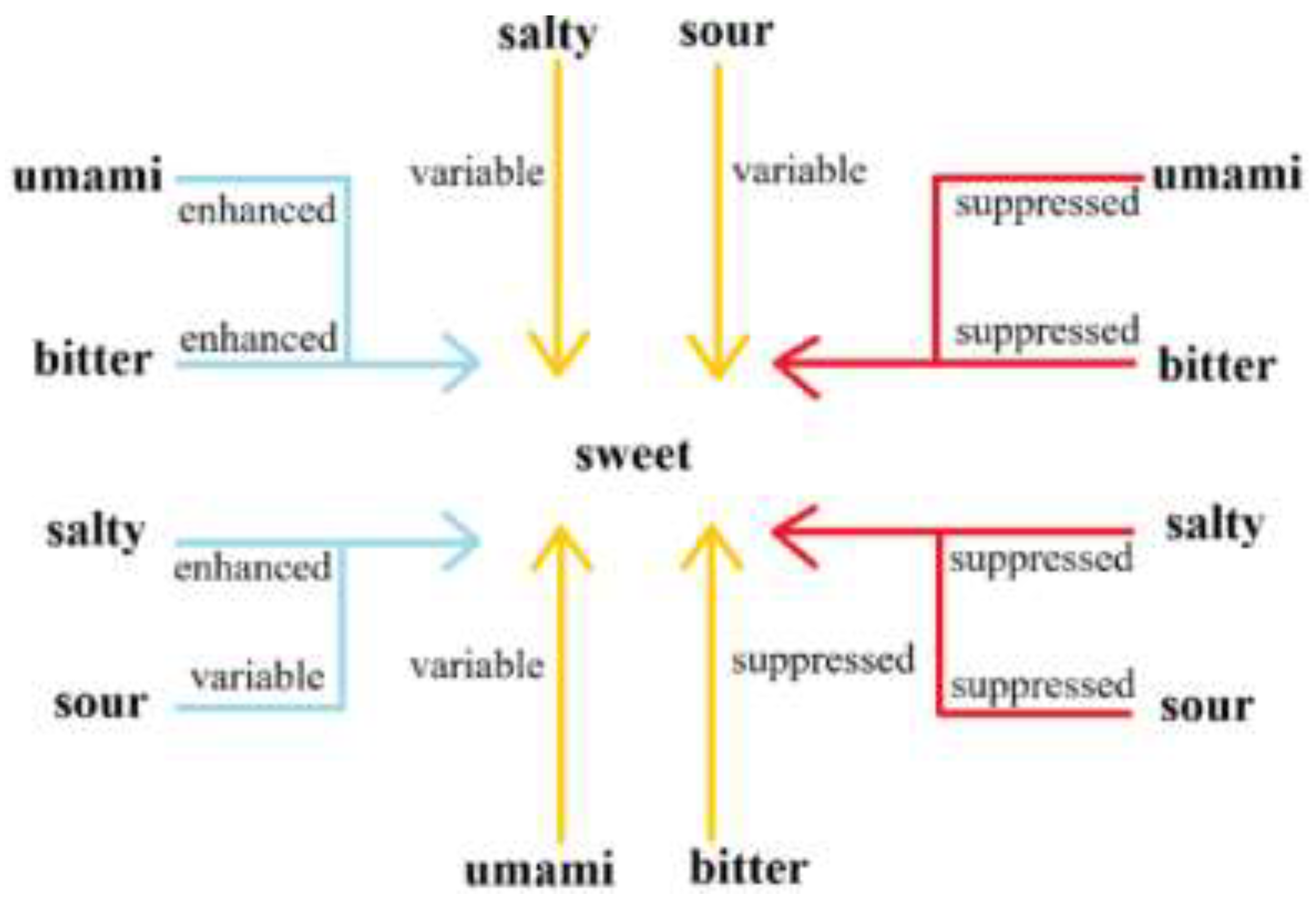
Currently, sweet taste is widely used in food, which can not only provide pleasant taste, but also works with other tastes to mask off-flavors and improve taste. Keast et al. proposed binary taste-taste interactions: at low intensity/concentrations, the perception of sweetness is enhanced by saltiness; at medium intensity/concentrations, sweetness is suppressed by bitterness; at high intensity/concentrations, both bitterness and sourness suppressed the perception of sweetness, while saltiness either suppressed sweetness or has no effect (
Figure 3) [
19]. Therefore, the existence of such taste interactions implies that one taste can change the perceived intensity of one or more other tastes. The effects of sourness, saltiness, umami, and bitterness on the perception of sweetness indicate that taste interactions are mediated at different concentrations. It is important to note that these are only generalizations and the effect will be compound specific.
2.1. Sweet-Bitter Interactions
Bitterness arises from specific bioactive components. In summary, bitter compounds mainly include phenolics, terpenoids, alkaloids, amino acids, nucleosides and purines [
20]. The interaction between sweet and bitter tastes has been extensively studied, especially in the context of low-calorie sweeteners. This interaction is not only involved in taste perception but also reflects the complex relationship between food components. The sweet-bitter balance has long been a focus of research: too much bitterness inhibits sweetness, while too much sweetness masks or diminishes bitterness. When bitter and sweet stimuli are present at the same time, subjects’ sensitivity to sweet decreases significantly with increasing bitter compound concentration. To some extent, bitterness inhibits the perception of sweetness, a phenomenon known as the “bitterness suppression effect”. Jin et al. [
21] found that bitter compounds inhibited the responses of sweet-tuned Calb2
+ neurons in the nucleus tractus solitarius to sweet stimuli. Experiments show that low concentrations of sucralose signal appetitive feeding in Drosophila, while high concentrations signal feeding aversion. Finally, silencing bitter GRNs reduced the aversive signal elicited by high concentrations of sucralose and significantly increased sucralose feeding behaviors [
22]. In another study, the sweet synergy and bitter antagonism by adding sucrose, vanillin, and cocoa mixture to chocolate milk. On the basis of reducing the amount of sucrose, adding vanillin to chocolate milk can keep the sweetness of milk and inhibit the bitterness of milk [
23]. With the exception of sucrose, fructose, and glucose, most sweeteners produce a bitter taste [
24]. The bitter taste of these sweet compounds is due to their ability to activate a family of 25 type 2 taste receptors (Tas2Rs). The cellular-based assay demonstrated that sucralose, acesulfame-K, rebaudioside A, mogroside V, and D-allulose activated at least 2 human bitter taste receptors (Tas2Rs) [
25]. Due to the existence of multiple binding sites in Tas1R2/Tas1R3, it has been shown that mixing sweeteners can reduce bitterness. Recent studies have shown that combining highly potent sweeteners, such as acesulfame-K or saccharin, with natural sugars, such as glucose or fructose, can enhance sweetness while reducing bitterness [
26]. This effect is attributed to the ability of monosaccharides to inhibit the activation of bitter taste receptors, thereby improving the overall sensory experience.
2.2. Sweet-Sour Interactions
Sweet and sour tastes often coexist in many food products and their interaction can significantly influence the perceived flavor balance. The interaction between sweetness and sourness is a complex phenomenon, which is influenced by many factors, including the concentration level used, the evaluation method employed and the specific acid type, and therefore sometimes presents different patterns. Studies have shown that the presence of sour taste can either enhance or inhibit sweetness, depending on concentration and environment [
27]. Similarly, Bonnans et al. [
28] found that sucrose and aspartame had the same inhibitory effect on sour taste. Sucrose was found to selectively elevate sweet intensity alone, while also modulating the sour taste imparted by lemon extract [
29]. Conversely, citric acid was shown to inhibit the sweet taste of sucrose [
30].
Generally, the degree to which sweetness inhibits sourness depends on the concentration of both tastes, and increasing sweeteners has a greater effect on inhibition than decreasing sourness. By contrast, the influence of sourness on sweetness is more complex: high levels of citric acid can suppress perceived sweetness [
31], whereas very low concentrations may actually enhance it [
32]. Mao et al. [
33] explored the sweet-sour interactions in sucrose-citric-acid mixtures by constructing a sweet-sour taste sensory strength variation model (SSTVM). Citric acid increased the absolute and differential thresholds of sucrose, effectively inhibited perceived sweetness, and reduced sensitivity to further sweetening increments. Conversely, sucrose elevated citric acid’s absolute threshold and reduced perceived sourness, while simultaneously lowering its difference threshold and enhanced the ability to detect subtle changes in sourness. Subsequently, the dynamic perceptual interaction between sucrose (sweet) and citric acid (sour) was simulated, demonstrating a significant reduction in citric acid-induced sour intensity with elevated sucrose concentrations [
34].
Sucrose inhibits citric-acid sourness far more strongly than citric acid inhibits sucrose sweetness, which greatly reduces the maximum sour intensity and overall sour sensation in the mouth. These quantitative models can guide the food and beverage industries to develop products with desirable sweet and sour properties. Similarly, in fruit processing, Ikegaya et al. [
35] found that increasing the ratio of sucrose to total sugar in strawberry juice significantly enhanced the overall sweetness, while increasing the ratio of malic acid to total organic acid significantly enhanced the perceived sourness. In tomato juice, sugar significantly masked the effect of increasing sourness and saltiness [
36].
2.3. Sweet-Umami Interactions
In 1908 Kikunae Ikeda discovered the unique taste component of konbu (kelp) as the salt of glutamic acid and coined the term “umami” to describe this taste [
37]. Umami was formally recognized in 2002 as the fifth basic taste after salty, sweet, sour, and bitter, and was characterized by a mouthwatering taste reminiscent of monosodium glutamate (MSG) [
38]. Previous studies have shown that heterodimeric G protein-coupled receptor Tas1R2/Tas1R3 and Tas1R1/Tas1R3 have been identified as sweet and umami receptors, respectively, and these two receptors share the Tas1R3 subunit. By comparing the responses of sweet and umami receptors to compounds that act on the Tas1R3 subunit, it showed that sweet receptor inhibitors can also act on umami receptors [
39]. The research conducted by Galindo-Cuspinera et al. [
40] also confirmed the same results. The binding mode of the Tas1R3 subunit of the sweet taste receptor and the perception of umami are interrelated, but the effect of Tas1R3 TMD on umami taste receptor function is lower than that on sweet taste receptor. At present, umami compounds are mainly classified into amino acids, nucleotides, organic acids and compound umami enhancers. Umami and sweetness interact in a synergistic manner. Research has shown that the combination of sweetness and umami can improve the overall palatability of foods. This interaction is particularly important in the context of enhancing food taste, where the addition of umami substances like glutamate can enhance perceived sweetness without increasing sugar content. Taste interactions between umami and sweet compounds were investigated via a cell-based assay of human Tas1R2/Tas1R3 [
41]. The results showed that MSG and umami peptides significantly reduced the sensitivity of sucrose to human Tas1R2/Tas1R3, but umami active nucleotides did not. In addition, when sweet taste receptors are activated by sweeteners (such as sucrose and acesulfame-K) that target the extracellular domain (ECD) of Tas1R2, MSG and glutamine peptides have a clear inhibitory effect on sweet taste receptors. This suggests that MSG and glutamine peptides may play an important role in the activation of sweet taste receptors. Furthermore, the umami compounds and lactisole (an inhibitor targeting Tas1R3) have more significant inhibitory effects. Inosine-5′-monophosphate (IMP), traditionally regarded as an umami enhancer, was found to bind directly to the Tas1R3 subunit and potentiate the response of the Tas1R2/Tas1R3 receptor to sweeteners including sucralose, neotame, and cyclamate [
42]. Additionally, when L-AP4, a glutamate receptor agonist, was mixed with one of the sweet substances, such as sucrose, glucose, fructose, and maltose, large synergistic responses were observed [
43].
2.4. Sweet-Salty Interactions
Salt is one of the most common seasonings in daily life, and saltiness meets people’s daily needs for delicious food. It has been reported that a small amount of salt could sometimes taste sweet. Current findings have shown that chloride ions can evoke taste by binding to the extracellular ligand-binding domain (LBD) of sweet taste receptors. Atsumi et al. [
44] employed structural biology techniques to characterize the Tas1R2/Tas1R3 sweet-taste heterodimer in medaka fish: at sub-to low-mM concentrations, chloride ions induced conformational changes in the Tas1R2/Tas1R3 LBD, similar to the effects of typical taste substances. Furthermore, oral administration of chloride ions to mice increased the impulse frequency of taste nerves connected to Tas1R-expressing cells and promoted behavioral preferences. These responses were attenuated by Tas1R-specific blockers or Tas1R3 knockout [
45]. In addition, the interactive effect between saltiness and sweetness seemed to be that salty taste inhibited sweet taste, while saltiness enhanced sweetness in dilute solution. Studies have shown that the sweetness and saltiness of sucrose and NaCl solutions are not simply additive. Instead, there is a complex interaction whose main feature is mutual masking. In addition, saltiness enhances the perception of sweetness in certain systems. It was proposed that low concentrations of salty substances can enhance sweetness, while moderate concentrations can inhibit it. It was shown that NaCl slightly increased the sweetness of the sucrose solution when both sucrose and sodium chloride concentrations were low. However, this effect turned into a pronounced inhibition as the concentration increased [
46].
Sweetness and saltiness are one of the most common flavor pairings: At low intensity/concentration, the mixture resulted in an enhancement of sweetness. The effect on sweetness was variable at moderate strength/concentration. High intensity/concentration mixing yielded a suppression of sweetness.
3. Interaction of Sweet Taste with Olfaction
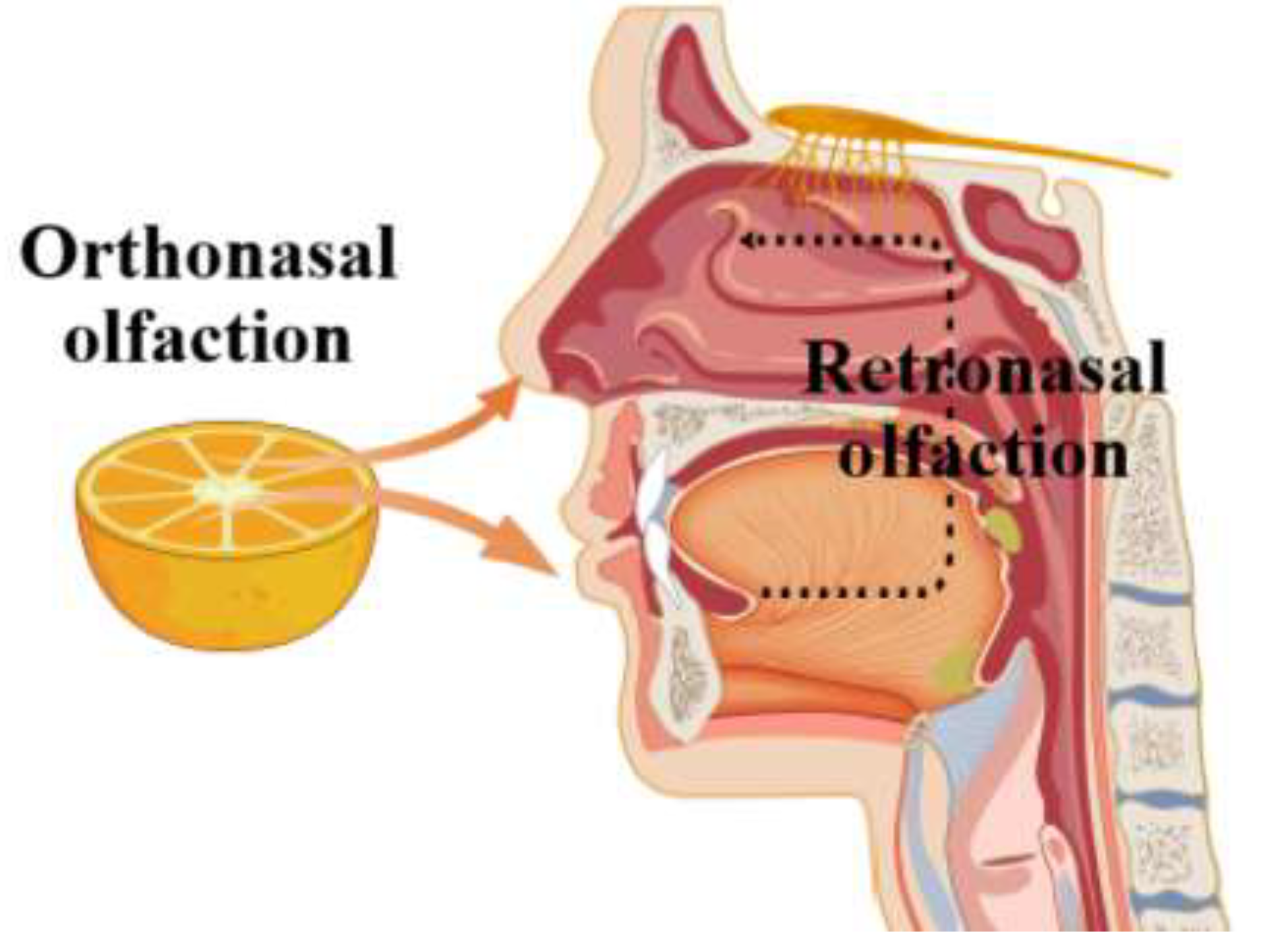
Cross-modal perception refers to the interaction between different sensory modalities, such as taste and smell. Early studies focused on simple sensory interactions, but modern studies have revealed more complex mechanisms. Olfactory perception occurs via two routes: the orthonasal and the retronasal pathways [
47]. In the orthonasal route, odorants are drawn directly into the nasal cavity through the nostrils, generating an olfactory sensation; in the retronasal route, odor compounds are released within the mouth as the food is processed (chewed and swallowed) (
Figure 4). These volatiles enter the nasal cavity through the oropharyngeal channel, where they interact with olfactory receptors on the olfactory epithelium to produce olfactory perception [
48]. Olfaction plays a crucial role in the perception of taste, and recent studies have highlighted the significant interaction between sweet taste and olfactory cues. Odor-induced taste enhancement (OITE) is a phenomenon of integrated processing of taste and odor in the brain [
49]. At present, the study of cross-modal interaction between taste and olfaction is primarily conducted through sensory evaluation combined with flavoromics techniques [
50]. In recent years, the use of odors to enhance the perception of sweetness in food has received extensive attention [
51]. The combination of sweet and aromatic compounds can enhance the overall sensory experience of the food. Existing studies have provided evidence support for this theory. For instance, the presence of vanilla aroma can enhance the perceived sweetness of a food product, even at lower sugar concentrations [
52]. This phenomenon is attributed to the multisensory integration of taste and olfactory signals in the brain, which can regulate the perception of sweetness. Similarly, certain odors can enhance sweetness, while others can inhibit it. For instance, it was proposed that when chocolate and strawberry aromas were co-stimulated with sucrose, the response amplitude of the Insular Cortex (IC) region was positively correlated with odor concentration [
53]. Aromas such as caramel, strawberry and lychee (at low concentrations) tended to enhance the sweetness of the sucrose solution, while aromas such as angelica oil tended to inhibit it [
54].
Enhancing sweetness via aroma is considered a sugar reduction strategy, but its effectiveness depends on the aroma type. Bertelsen et al. [
55] used descriptive sensory analysis to study the enhancement effect of different aroma compounds on sweet perception of sucrose solution. Vanilla, honey and banana significantly enhanced the perceived sweetness of the sucrose solution compared to elderflower and raspberry aromas. A series of studies found that aroma compounds with sweet attributes in brown sugar, such as 2,3-butanedione, β-damascenone, 2-methoxyphenol, dihydro-2-methyl-3(2H)-furanone, 2-furanmethanol and butyrolactone, can significantly improve the sweet perception of sucrose solution [
56]. The effects of red pigment and strawberry aroma on the sweetness of aqueous sucrose solutions were evaluated, the results showed that strawberry aroma could significantly enhance the perception of sweetness in aqueous sucrose solution. These findings suggest that some aroma compounds have the potential to enhance sweetness by acting synergistically with sweet substances to enhance sweetness perception in food [
57].
Fruit aroma can also enhance the perception of sweetness in humans. Dai et al. [
58] identified seven watermelon juice volatiles-ethyl acetate, ethyl propionate, octanal, (E,E)-2,4-hexadienal, (E)-2-octenal, methylheptenone and geranyl acetone—that significantly increased the perceived sweetness of a 2.5% fructose solution while prolonging sweetness duration to >45 s. In addition, aroma is a key quality attribute of citrus fruits. The aroma characteristics of sweet orange are a critical determinant of fruit quality. Through analysis of volatiles in sweet orange juice, identified seven sweetness-linked odorants—(E)-citral, (E)-β-farnesene, β-myrcene, tallo-ocimene, nonanal, citronellyl formate and tallo-ocimene-significantly—that markedly enhanced the sweetness of 5% sucrose solution [
59]. Similarly, nine sweet orange aroma compounds—ethyl propionate, ethyl 2-methylbutanoate, octyl acetate, ethyl butanoate, ethyl hexanoate, butyl hexanoate, γ-terpinene, nootkatone, and acetophenone—were assessed for their sweetness-enhancing effects, and all nine significantly influenced perceived sweetness in a 5% sucrose solution [
60].
4. Interaction of Sweet Taste with Trigeminal Nerve
The trigeminal nerve is the largest cranial nerve—it comprises three branches: ophthalmic (V₁), maxillary (V₂) and mandibular (V₃). Collectively, the trigeminal system transduces nociceptive signals, chemical irritation, temperature, texture and mechanical consistency of food [
61,
62]. Menthol, a naturally occurring compound in peppermint oil, is a trigeminal stimulant [
63]. At low concentrations it can activate olfactory receptors, producing a characteristic minty taste [
64]. Büchner et al. [
65] found that menthol increased the recognition threshold of bitter/metallic taste of acesulfame-K from 21.5 to 36.5 mg/L without changing its own recognition threshold. Trace amounts of menthol can mask the odor of artificial sweeteners. In addition, sensory data showed no significant change in perceived sweetness, but psychophysiology revealed menthol modulated autonomic responses, indicating subtle interaction between the trigeminal nerve and taste pathways. TRPM8 is a TRP family cation channel which can be activated by cold stimuli or l-menthol. The result suggests that TRPM8 protein is present in sensory lingual nerve fibers mainly projected from trigeminal ganglia and might work as cold and l-menthol receptors on tongue. The texture and mouthfeel of food can also influence the perception of sweetness [
66]. Studies have shown that the viscosity and aeration of food can modulate the release and perception of sweet substances [
61]. For instance, creamy food may enhance the perception of sweetness compared to watery food. This interaction may be mediated by the trigeminal system, which detects tactile and thermal sensations in the mouth. Lago et al. [
67] investigated how the texture of the cup held while drinking iced tea influenced perceived sweetness; their results showed that tea sweetness could be increased by changing the surface of the cup. Some research also revealed a centrally mediated suppression of trigeminal sensation by taste inputs [
68]. It was proposed that there was a large decline in taste and trigeminal functioning after COVID-19 infection [
69]. The research indicates that in the paraventricular nucleus of mice, TRPV1-positive somatic sensory fibers communicate with gustatory neurons. The somatic sensory input from the trigeminal nerve can regulate the taste signals, providing evidence for the interaction between sweetness and the trigeminal nerve center [
70]. Other studies show that the greater superficial petrosal (GSP) nerve is important to the rat in reinforcing high lick rates to sucrose [
71].
5. Practical Application
As the health risks posed by the soaring intake of sugar in the diet become increasingly apparent, the interplay of sweetness with other tastes is crucial in beverage formulation. The selection and application of sweeteners are becoming more important in the current boom in low-sugar or diet drinks. Through precise formulation, sweetness can be paired with sourness, such as citric acid to maintain palatability while reducing sugar intake [
30]. The addition of umami enhancers such as monosodium glutamate further enriches the complexity of beverages, and balancing the bitterness of tea polyphenols with sweetness is a key strategy in tea-based products. By reconciling sweetness with other tastes, this approach not only provides a unique sensory experience but also helps to satisfy the different taste preferences of today’s consumers. In addition, in baked goods, proper sweetness not only enhances flavor and mouthfeel but also creates complementary interaction with other taste modalities. In pastry and confectionery, sweetness is often used to balance the salt contributed by added sodium chloride [
46]. When the concentrations of both tastes are moderate, a synergistic effect emerges that makes the overall flavor more pleasant. The combination of bitter cocoa and sweetness reinforces the rich image of chocolate pastries, while the addition of trace amounts of salt makes breads and other foods richer and sweeter. Umami, such as that produced by yeast fermentation, can further enrich the overall taste experience of flour products.
In cooking and seasoning, sweetness is often used to add umami and improve flavor [
43]. In stews, the right amount of sugar can reconcile sour and bitter, making dishes more balanced and harmonious. In addition, natural sweeteners such as honey are widely combined with umami-rich spices such as vanilla. Modern condiments also incorporate sweet elements to add complexity and depth to the taste. This synergy provides inspiration and market potential for new composite seasonings: a low-sodium sauce can be balanced with a touch of sweetness to round out saltiness and enrich overall taste. Such formulations satisfy not only consumers’ health concerns but also their growing demand for diverse and sophisticated tastes.
Beyond sweetness and saltiness, other tastes such as sourness, umami, and bitterness also interact with sweetness. Sourness could enhance the refreshing quality of sweetness, while umami could reduce the cloying effect of excessive sweetness. Sometimes, bitterness can be used as a flavoring to balance out other more prominent tastes [
24], especially when sweetness is evident. Proper addition of salt can enhance the sweetness of desserts while reducing any pungent sourness. The balance between sweetness and saltiness enriches the overall mouthfeel and adds depth to the taste.
6. Future Directions
In practice, many foods such as coffee, tea, and beverages as well as medicines must be carefully balanced for sweetness and bitterness. Adjusting the proportion of ingredients or using new food additives (such as natural sweeteners and bitter masking agents) can significantly improve the sensory quality. Therefore, future research should investigate the interaction between bitter and sweet taste in more depth to provide a theoretical basis for the development of taste modulators. In this regard, the understanding of sweet taste interactions with other sensory systems has significant implications for food science and nutrition. For instance, the use of aroma compounds and umami substances can help reduce sugar content in food products while maintaining palatability. Additionally, the development of novel sweeteners and flavor enhancers can benefit from insights into the multisensory integration of taste and olfaction. Future research should focus on elucidating the molecular and neural mechanisms underlying these interactions, as well as exploring their potential applications in personalized nutrition and health.
7. Conclusion and Outlook
In this article, we have reviewed the interactions between sweetness and the four basic tastes—bitter, sour, umami, and salty—and explored the cross-modal perceptual interactions of sweetness with olfaction and trigeminal nerve, thereby offering new insights into how sweetness interacts with other fundamental sensory qualities. Moreover, under specific conditions, aroma can enhance the perception of sweetness. Building on this, combining sweeteners with selected aromas can be investigated as a sugar-reduction strategy to intensify sweet taste. As for the interaction between sweetness and other basic tastes, existing studies have confirmed the fundamental principles of sweetness interactions with other basic taste modalities across a range of food matrices. Investigating the functional mechanisms of these receptors and their interaction with sweetness will help elucidate the principles underlying flavor formation and provide a theoretical foundation for optimizing food formulations and enhancing product quality.
Author Contributions
Conceptualization, B.L.; writing—original draft preparation, Y.W.; writing—review and editing, B.L. and C.D.; research, C.D. and M.Z.; funding acquisition B.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (31970935).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lee, A.A.; Owyang, C. Sugars, Sweet Taste Receptors, and Brain Responses. Nutrients 2017, 9, 653. [Google Scholar] [CrossRef]
- Li, X.; Staszewski, L.; Xu, H.; Durick, K.; Zoller, M.; Adler, E. Human receptors for sweet and umami taste. Proc Natl Acad Sci U S A 2002, 99, 4692–6. [Google Scholar] [CrossRef]
- Nango, E.; Akiyama, S.; Maki-Yonekura, S.; Ashikawa, Y.; Kusakabe, Y.; Krayukhina, E.; Maruno, T.; Uchiyama, S.; Nuemket, N.; Yonekura, K.; Shimizu, M.; Atsumi, N.; Yasui, N.; Hikima, T.; Yamamoto, M.; Kobayashi, Y.; Yamashita, A. Taste substance binding elicits conformational change of taste receptor T1r heterodimer extracellular domains. Sci Rep. 2016, 6, 25745. [Google Scholar] [CrossRef]
- Shi, Z.; Xu, W.; Wu, L.; Yue, X.; Liu, S.; Ding, W.; Zhang, J.; Meng, B.; Zhao, L.; Liu, X.; Liu, J.; Liu, Z.J.; Hua, T. Structural and functional characterization of human sweet taste receptor. Nature 2025, 645, 801–808. [Google Scholar] [CrossRef]
- Shallenberger, R.S.; Acree, T.E. Molecular theory of sweet taste. Nature 1967, 216, 480–482. [Google Scholar] [CrossRef]
- Kier, L.B. A molecular theory of sweet taste. Pharm Sci. 1972, 61, 1394–7. [Google Scholar] [CrossRef]
- Zheng, H.; Xu, X.; Fang, Y.; Sun, R.; Liu, B. The Molecular Theory of Sweet Taste: Revisit, Update, and Beyond. Med Chem. 2024, 67, 3232–3243. [Google Scholar] [CrossRef]
- Taruno, A.; Nomura, K.; Kusakizako, T.; Ma, Z.; Nureki, O.; Foskett, J.K. Taste transduction and channel synapses in taste buds. Pflugers Arch. 2021, 473, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; Dalziel, J.E. G Protein-Coupled Receptors in Taste Physiology and Pharmacology. Front Pharmacol. 2020, 11, 587664. [Google Scholar] [CrossRef] [PubMed]
- Bigiani, A. Does ENaC Work as Sodium Taste Receptor in Humans? Nutrients 2020, 12, 1195. [Google Scholar] [CrossRef] [PubMed]
- Teng, B.; Wilson, C.E.; Tu, Y.H.; Joshi, N.R.; Kinnamon, S.C.; Liman, E.R. Cellular and Neural Responses to Sour Stimuli Require the Proton Channel Otop1. Curr Biol. 2019, 29, 3647–3656. [Google Scholar] [CrossRef]
- Tu, Y.H.; Cooper, A.J.; Teng, B.; Chang, R.B.; Artiga, D.J.; Turner, H.N.; Mulhall, E.M.; Ye, W.; Smith, A.D.; Liman, E.R. An evolutionarily conserved gene family encodes proton-selective ion channels. Science 2018, 359, 1047–1050. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jin, H.; Zhang, W.; Ding, C.; O’Keeffe, S.; Ye, M.; Zuker, C.S. Sour Sensing from the Tongue to the Brain. Cell. 2019, 179, 392–402. [Google Scholar] [CrossRef]
- Breslin, P.A.; Beauchamp, G.K. Salt enhances flavour by suppressing bitterness. Nature 1997, 387, 563. [Google Scholar] [CrossRef] [PubMed]
- Junge, J.Y.; Andersen, G.H.; Kidmose, U. Suppression of sweetness: evidence for central mechanism for suppression of sweetness from sucrose by citric acid. Chem Senses 2023, 48. [Google Scholar] [CrossRef]
- Calviño, A.M.; Garrido, D. Spatial and temporal suppressive behavior of bitter-sweet mixtures. Percept Mot Skills 1991, 73, 1216. [Google Scholar] [CrossRef]
- Prescott, J.; Ripandelli, N.; Wakeling, I. Binary taste mixture interactions in prop non-tasters, medium-tasters and super-tasters. Chem Senses 2001, 26, 993–1003. [Google Scholar] [CrossRef]
- Schiffman, S.S.; Sattely-Miller, E.A.; Graham, B.G.; Bennett, J.L.; Booth, B.J.; Desai, N.; Bishay, I. Effect of temperature, pH, and ions on sweet taste. Physiol Behav. 2000, 68, 469–481. [Google Scholar] [CrossRef]
- Keast, R.S.; Breslin, P.A. An overview of binary taste–taste interactions. Food Quality and Preference 2003, 14, 111–124. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, H.; Huang, M.; Chen, J.; Chen, J.; Cheng, H.; Liu, D. Role of bitter contributors and bitter taste receptors: a comprehensive review of their sources, functions and future development. Food Science and Human Wellness 2024, 13, 1806–1824. [Google Scholar] [CrossRef]
- Jin, H.; Fishman, Z.H.; Ye, M.; Wang, L.; Zuker, C.S. Top-Down Control of Sweet and Bitter Taste in the Mammalian Brain. Cell. 2021, 184, 257–271. [Google Scholar] [CrossRef] [PubMed]
- Arntsen, C.; Grenon, J.; Chauvel, I.; Fraichard, S.; Dupas, S.; Cortot, J.; Audette, K.; Musso, P.Y.; Stanley, M. Artificial sweeteners differentially activate sweet and bitter gustatory neurons in Drosophila. Scientific reports 2025, 15, 20785. [Google Scholar] [CrossRef]
- Hopfer, H.; Riak, A.C.; Roberts, R.F.; Hayes, J.E.; Ziegler, G.R. Synergistic and antagonistic ingredient interactions as a sugar reduction strategy in chocolate milk. Journal of Sensory Studies 2022, 37, e12770. [Google Scholar] [CrossRef]
- Riera, C.E.; Vogel, H.; Simon, S.A.; Ie Coutre, J. Artificial sweeteners and salts producing a metallic taste sensation activate TRPV1 receptors. Am J Physiol Regul Integr Comp Physiol. 2007, 293, R626–R634. [Google Scholar] [CrossRef] [PubMed]
- Belloir, C.; Jeannin, M.; Karolkowski, A.; Scott, C.; Briand, L. A receptor-based assay to study the sweet and bitter tastes of sweeteners and binary sweet blends: the SWEET project. Chemical senses 2024, 49, bjae041. [Google Scholar] [CrossRef]
- Choi, Y.; Wong, R.R.; Cha, Y.K.; Park, T.H.; Kim, Y.; Chung, S.J. Sweet-bitter taste interactions in binary mixtures of sweeteners: Relationship between taste receptor activities and sensory perception. Food Chem. 2024, 459, 140343. [Google Scholar] [CrossRef]
- Beauchamp, G.K.; Mennella, J.A. Flavor perception in human infants: development and functional significance. Digestion 2011, 83, 1–6. [Google Scholar] [CrossRef]
- Bonnans, S.; Noble, A.C. Effect of sweetener type and of sweetener and acid levels on temporal perception of sweetness, sourness and fruitiness. Chemical Senses 1993, 18, 273–283. [Google Scholar] [CrossRef]
- Veldhuizen, M.G.; Siddique, A.; Rosenthal, S.; Marks, L.E. Interactions of Lemon, Sucrose and Citric Acid in Enhancing Citrus, Sweet and Sour Flavors. Chemical senses 2017, 43, 17–26. [Google Scholar] [CrossRef]
- Junge, J.Y.; Andersen, G.H.; Kidmose, U. Suppression of sweetness: evidence for central mechanism for suppression of sweetness from sucrose by citric acid. Chemical senses 2023, 48. [Google Scholar] [CrossRef] [PubMed]
- McBRIDE, R.L.; Finlay, D.C. Perception of taste mixtures by experienced and novice assessors. Journal of Sensory Studies 1989, 3, 237–248. [Google Scholar] [CrossRef]
- KAMEN, J.M.; PILGRIM, F.J.; GUTMAN, N.J.; KROLL, B.J. Interactions of suprathreshold taste stimuli. Exp Psychol. 1961, 62, 348–356. [Google Scholar] [CrossRef]
- Mao, Y.; Tian, S.; Qin, Y.; Chen, S. Sensory sweetness and sourness interactive response of sucrose-citric acid mixture based on synergy and antagonism. NPJ Sci Food 2022, 6, 33. [Google Scholar] [CrossRef]
- Mao, Y.; Zhang, Z.; Liu, L.; Qin, Y.; Qin, Z.; Cao, Y.; Zou, X.; Shi, J.; Tian, S.; Jiang, G. Dynamic interaction of sweet and sour taste perceptions based on sucrose and citric acid. NPJ Sci Food 2025, 9, 152. [Google Scholar] [CrossRef]
- Ikegaya, A. Composition of free sugars and organic acids in Japanese strawberry cultivars and their influence on the perception of sweetness and sourness. Food Sci. 2024, 89, 614–624. [Google Scholar] [CrossRef] [PubMed]
- Salles, C.; Nicklaus, S.; Septier, C. Determination and gustatory properties of taste-active compounds in tomato juice. Food Chemistry 2003, 81, 395–402. [Google Scholar] [CrossRef]
- Kurihara, K. Glutamate: from discovery as a food flavor to role as a basic taste (umami). Am J Clin Nutr. 2009, 90, 719S–722S. [Google Scholar] [CrossRef] [PubMed]
- ZZhang, J.; Sun-Waterhouse, D.; Su, G.; Zhao, M. New insight into umami receptor, umami/umami-enhancing peptides and their derivatives: A review. Trends in Food Science & Technology 2019, 88, 429–38. [Google Scholar]
- Kawasaki, M.; Kidera, Y.; Goda, R.; Taketani, C.; Ide, M.; Fujii, W.; Nakagita, T.; Misaka, T. Distinct potency of compounds targeting the T1R3 subunit in modulating the response of human sweet and umami taste receptors. Sci Rep. 2025, 15, 27167. [Google Scholar] [CrossRef]
- Galindo-Cuspinera, V.; Breslin, P.A. The liaison of sweet and savory. Chem Senses. 2006, 31, 221–225. [Google Scholar] [CrossRef]
- Shim, J.; Son, H.J.; Kim, Y.; Kim, K.H.; Kim, J.T.; Moon, H.; Kim, M.J.; Misaka, T.; Rhyu, M.R. Modulation of sweet taste by umami compounds via sweet taste receptor subunit hT1R2. PLoS One 2015, 10, e0124030. [Google Scholar] [CrossRef]
- Belloir, C.; Moitrier, L.; Karolkowski, A.; Poirier, N.; Neiers, F.; Briand, L. Inosine-5′-monophosphate interacts with the TAS1R3 subunit to enhance sweet taste detection. Food Chemistry: Molecular Sciences 2025, 10, 100246. [Google Scholar] [CrossRef]
- Sako, N.; Tokita, K.; Sugimura, T.; Yamamoto, T. Synergistic responses of the chorda tympani to mixtures of umami and sweet substances in rats. Chemical senses 2003, 28, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Atsumi, N.; Yasumatsu, K.; Takashina, Y.; Ito, C.; Yasui, N.; Margolskee, R.F.; Yamashita, A. Chloride ions evoke taste sensations by binding to the extracellular ligand-binding domain of sweet/umami taste receptors. Elife 2023, 12, e84291. [Google Scholar] [CrossRef]
- Chandrashekar, J.; Kuhn, C.; Oka, Y.; Yarmolinsky, D.A.; Hummler, E.; Ryba, N.J.; Zuker, C.S. The cells and peripheral representation of sodium taste in mice. Nature 2010, 464, 297–301. [Google Scholar] [CrossRef]
- Stevens, J.C.; Traverzo, A. Detection of a target taste in a complex masker. Chem Senses 1997, 22, 529–534. [Google Scholar] [CrossRef]
- Small, D.M.; Gerber, J.C.; Mak, Y.E.; Hummel, T. Differential neural responses evoked by orthonasal versus retronasal odorant perception in humans. Neuron 2005, 47, 593–605. [Google Scholar] [CrossRef]
- Blankenship, M.L.; Grigorova, M.; Katz, D.B.; Maier, J.X. Retronasal Odor Perception Requires Taste Cortex, but Orthonasal Does Not. Current Biology 2019, 29, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Ai, Y.; Han, P. Neurocognitive mechanisms of odor-induced taste enhancement: A systematic review. International Journal of Gastronomy and Food Science 2022, 28, 100535. [Google Scholar] [CrossRef]
- Tian, H.; Zheng, G.; Yu, H.; Chen, C.; Lou, X. Research Progress on the Effect of the Interaction between Odor and Taste on Food Flavor Perception. Food Science (In Chinese) 2023, 44, 259–269. [Google Scholar]
- Zhang, D.; Lao, F.; Pan, X.; Li, J.; Yuan, L.; Li, M.; Cai, Y.; Wu, J. Enhancement effect of odor and multi-sensory superposition on sweetness. Compr Rev Food Sci Food Saf. 2023, 22, 4871–4889. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, R.J.; Boakes, R.A. Sweet and sour smells: learned synesthesia between the senses of taste and smell; 2004. [Google Scholar]
- Veldhuizen, M.G.; Nachtigal, D.; Teulings, L.; Gitelman, D.R.; Small, D.M. The insular taste cortex contributes to odor quality coding. Front Hum Neurosci. 2010, 4, 58. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, R.J.; Prescott, J.; Boakes, R.A. Confusing tastes and smells: how odours can influence the perception of sweet and sour tastes. Chem Senses 1999, 24, 627–635. [Google Scholar] [CrossRef]
- Bertelsen, A.S.; Mielby, L.A.; Alexi, N.; Byrne, D.V.; Kidmose, U. Sweetness Enhancement by Aromas: Measured by Descriptive Sensory Analysis and Relative to Reference Scaling. Chem Senses 2020, 45, 293–301. [Google Scholar] [CrossRef]
- Liu, J.; Wan, P.; Xie, C.; Chen, D.W. Key aroma-active compounds in brown sugar and their influence on sweetness. Food Chem. 2021, 345, 128826. [Google Scholar] [CrossRef]
- Frank, R.A.; Ducheny, K.; Mize, S.J.S. Strawberry odor, but not red color, enhances the sweetness of sucrose solutions. Chemical Senses 1989, 14, 371–377. [Google Scholar] [CrossRef]
- Dai, Y.; Sun, S.; Yang, F.; Zhen, S.; Xiong, X.; Liu, Y.; Bi, S. Revealing Volatile Odor Compounds in Watermelon Juice to Enhance Fructose Sweetness Perception: Sensory Evaluation and Molecular Docking Techniques. Foods 2025, 14, 1034. [Google Scholar] [CrossRef]
- Niu, Y.; Wei, J.; Zhu, J.; Feng, H.; Ren, Y.; Guo, Z.; Zhang, J.; Zhou, R.; She, Y.; Wang, Z.; Xiao, Z. Study on the mechanism of glucose-lowering and sweetening of key sweet aroma compounds in sweet orange. Food Research International 2024, 196, 114874. [Google Scholar] [CrossRef]
- Xiao, Z.; Gao, J.; Niu, Y.; Wang, Z.; Zhou, R.; Zhang, J.; Zhu, J. Elucidation of the sweetening mechanism of sweet orange fruit aroma compounds on sucrose solution using sensory evaluation, electronic tongue, molecular docking, and molecular dynamics simulation. LWT 2024, 205, 116555. [Google Scholar] [CrossRef]
- Auvray, M.; Spence, C. The multisensory perception of flavor. Conscious Cogn. 2008, 17, 1016–1031. [Google Scholar] [CrossRef]
- Delwiche, J. The impact of perceptual interactions on perceived flavor. Food Quality and Preference 2004, 15, 137–146. [Google Scholar] [CrossRef]
- Viana, F. Chemosensory properties of the trigeminal system. ACS Chem Neurosci. 2011, 2, 38–50. [Google Scholar] [CrossRef]
- Renner, B.; Schreiber, K. Olfactory and trigeminal interaction of menthol and nicotine in humans. Exp Brain Res. 2012, 219, 13–26. [Google Scholar] [CrossRef]
- Büchner, K.; Haagen, J.; Sastrosubroto, A.; Kerpes, R.; Freiherr, J.; Becker, T. Trigeminal Stimulus Menthol Masks Bitter Off-Flavor of Artificial Sweetener Acesulfame-K. Foods 2022, 11, 2734. [Google Scholar] [CrossRef] [PubMed]
- Abe, J.; Hosokawa, H.; Okazawa, M.; Kandachi, M.; Sawada, Y.; Yamanaka, K.; Kobayashi, S. TRPM8 protein localization in trigeminal ganglion and taste papillae. Molecular brain research 2025, 136, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Lago, M.; De la Rosa, J.; Vázquez-Araújo, L. Using tactile stimuli to enhance sweet perception in iced tea samples. Journal of Sensory Studies 2021, 36, e12612. [Google Scholar] [CrossRef]
- Schöbel, N.; Kyereme, J.; Minovi, A.; Dazert, S.; Bartoshuk, L.; Hatt, H. Sweet taste and chorda tympani transection alter capsaicin-induced lingual pain perception in adult human subjects. Physiol Behav. 2012, 107, 368–373. [Google Scholar] [CrossRef]
- Boesveldt, S.; Postma, E.M.; Boek, W.; Kamalski, D.M.; Van Dijk, B. Longitudinal follow-up of taste function and trigeminal perception in COVID-19 patients with olfactory dysfunction–The COVORTS study. Clinical Nutrition Open Science 2024, 55, 249–262. [Google Scholar] [CrossRef]
- Li, J.; Ali, M.S.S.; Lemon, C.H. TRPV1-Lineage Somatosensory Fibers Communicate with Taste Neurons in the Mouse Parabrachial Nucleus. Neurosci. 2022, 42, 1719–1737. [Google Scholar] [CrossRef]
- Krimm, R.F.; Nejad, M.S.; Smith, J.C.; Miller, I.J., Jr.; Beidler, L.M. The effect of bilateral sectioning of the chorda tympani and the greater superficial petrosal nerves on the sweet taste in the rat. Physiol Behav. 1987, 41, 495–501. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).