Submitted:
28 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
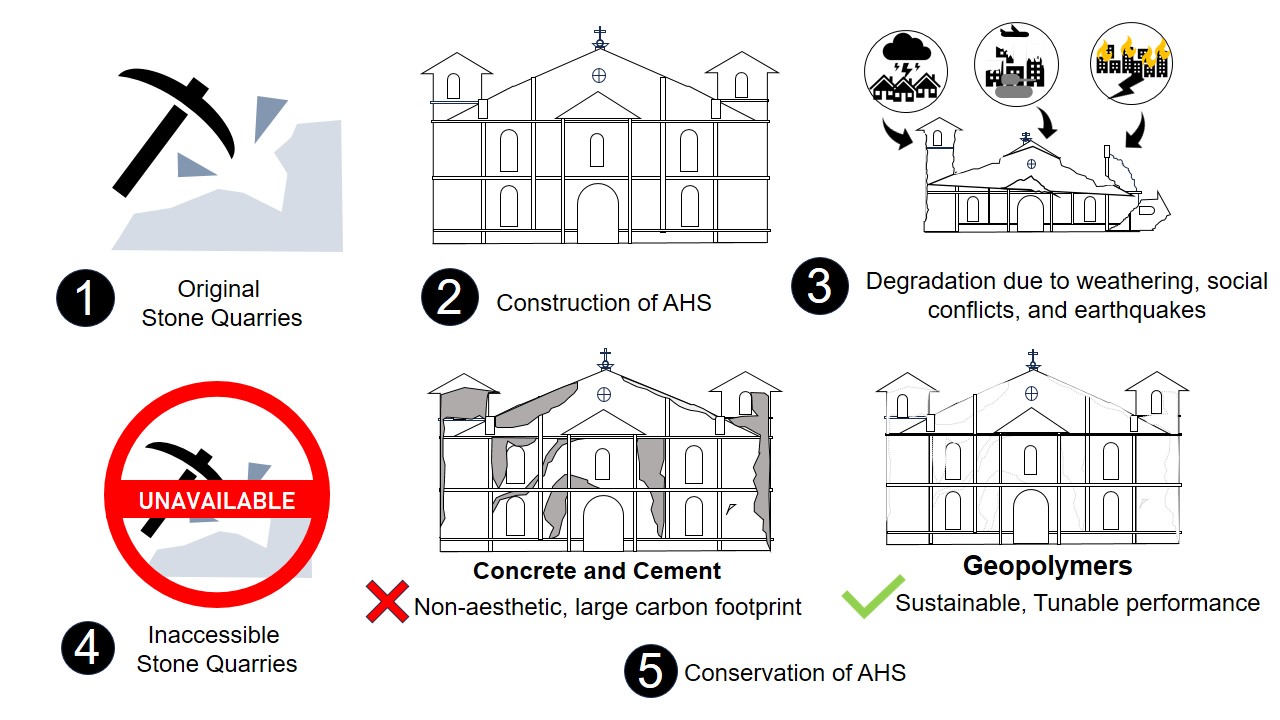
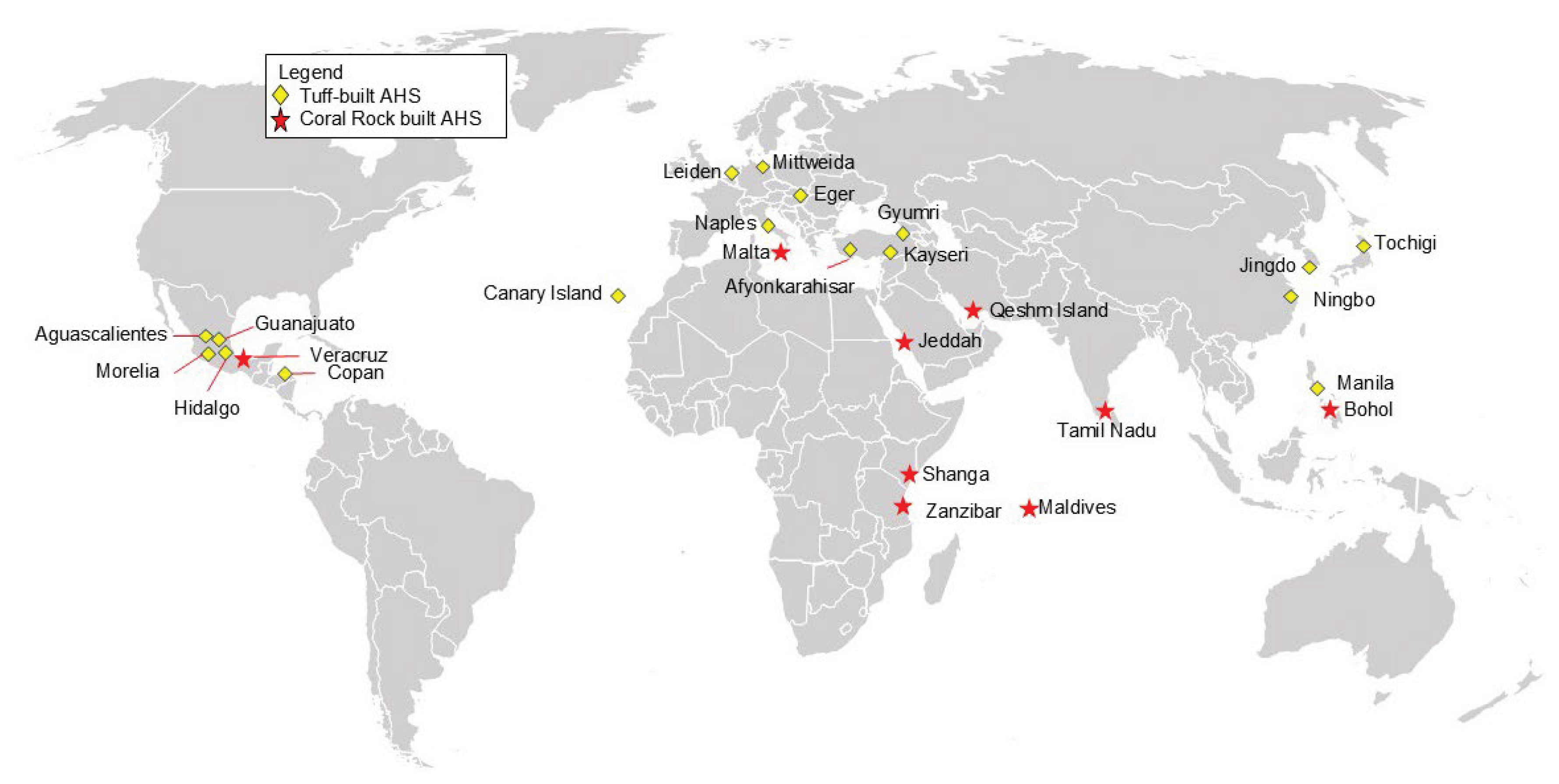
1. Introduction






2. Review Methodology
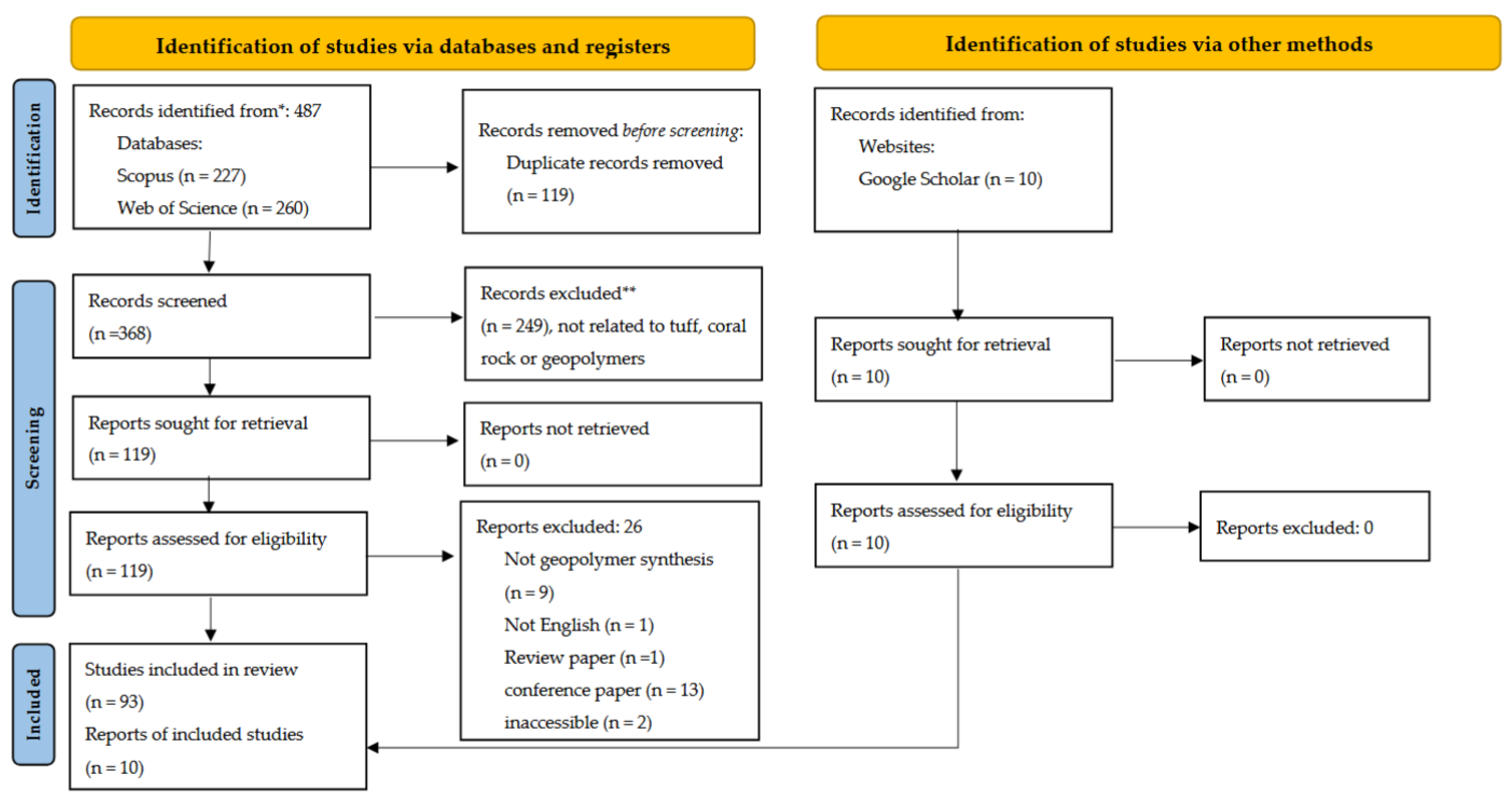
2.1. Search and Identification
2.2. Screening and Inclusion Criteria
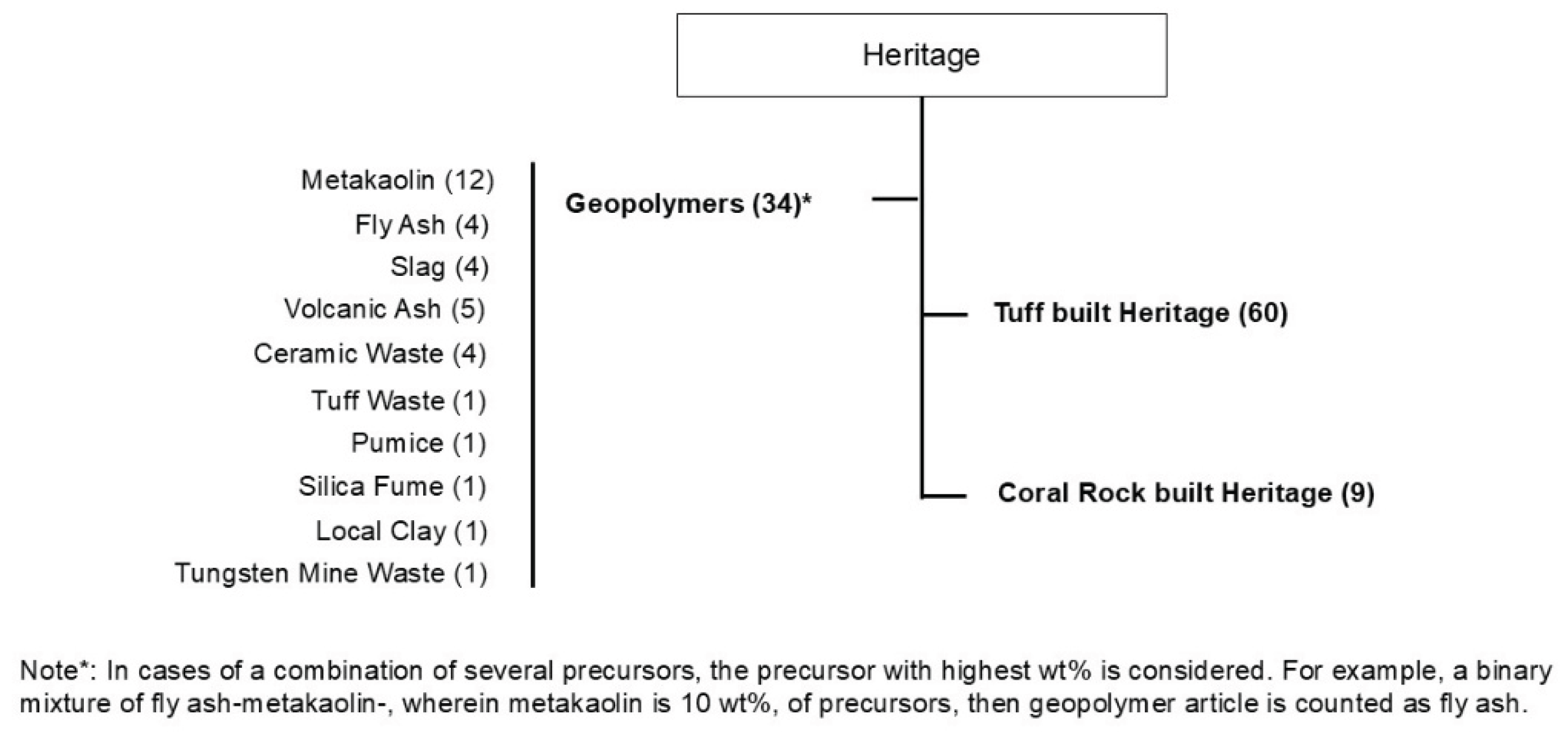
2.3. Publication Trends
2.4. Active Countries, Journals, and Institutions
2.5. Active Authors
3. Geopolymers Used for Stone Architectural Heritage Structure Conservation
3.1. Definition and Formation Mechanism
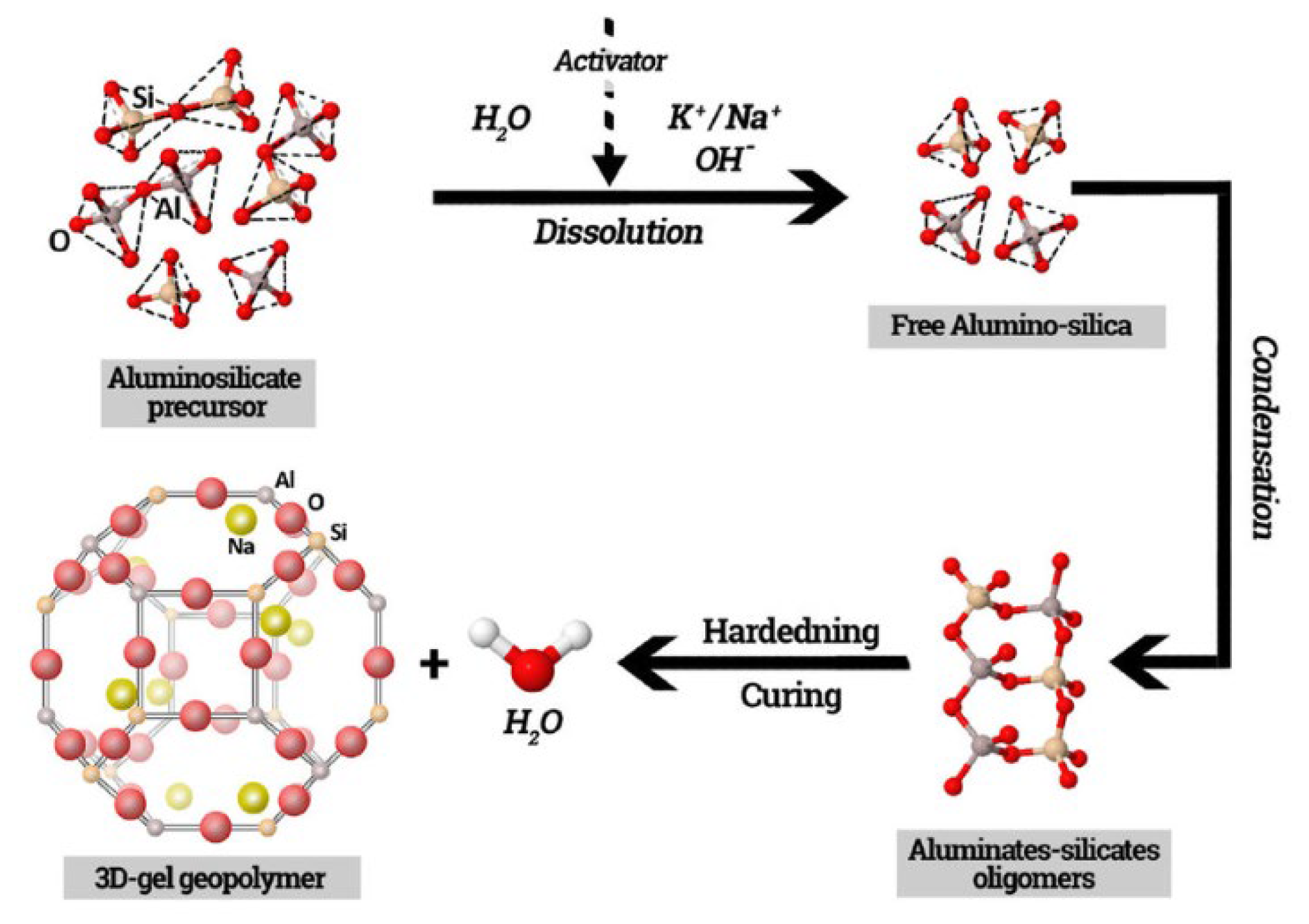
3.2. Metakaolin-Based Geopolymers
| Country | Precursors SiO2=50-67%, Al2O3=31-42%, Fe2O3=0.3-2% K2O+Na2O=0.3-1.3%, CaO+MgO=0.2-1.0% TiO2=0.2-2.2% |
Activators | Admixture | Aggregate | 28-day compressive strength (MPa) | Ref. |
|---|---|---|---|---|---|---|
| Morrocco | Metakaolin | KS, KOH | Calcareous sand (CaCO3) | – | 16–41 (7d) | [68] |
| SS, NaOH | 63–77 (7d) | |||||
| KS, KOH | Ca(OH)2 | 7–10 (7d) | ||||
| SS, NaOH | 2–40 (7d) | |||||
| Italy | Metakaolin | SS, NaOH | - | Dolostone | 18 | [69] |
| Sandstone | 21 | |||||
| Quartz Sand | 75 | |||||
| Italy | Metakaolin | SS, NaOH | None | – | 41 | [70] |
| Epoxy Resin | 49 | |||||
| Portugal | Metakaolin | SS, NaOH | Zeolite | – | 4.6 | [71] |
| NaOH | Ca (OH)2 | 1.0 | ||||
| Portugal | Metakaolin | SS, NaOH | – | – | 0.6–1.0 | [72] |
| Czech Republic | Metakaolin | SS, NaOH | Waste Foam Glass | – | 2.7–8.3 | [73] |
| Czech Republic | Metakaolin (LO5) | KS, KOH | None | 86.6 | [74] | |
| Dolostone | Quartz Sand | 72.7–87.7 | ||||
| Marble | 73.8–75.7 | |||||
| Marlstone | 78–80 | |||||
| Limestone | 75–78 | |||||
| Feldspar | 94–96 | |||||
| Italy | Metakaolin | SS, NaOH | None | – | 40 | [75] |
| Polyvinyl Acetate | 38–42 | |||||
| Polydimethylsiloxane | 37–42 | |||||
| Italy | Metakaolin | SS, NaOH | Bromothymol Blue (Organic Dye) | – | – | [76] |
| Phenolphthalein | ||||||
| Cresol red | ||||||
| Methyl orange | ||||||
| Czech Republic | Metakaolin (LO5) | KS, KOH | Slag, Colored Pigments | Quartz sand | [77] | |
| Czech Republic | Metakaolin (LO5) | KS, KOH | None | Quartz sand | 75.5 | [78] |
| China | Metakaolin, | SS, KS | Silica Fume | – | 40–70 | [79] |
3.3. Fly Ash-Based Geopolymers
| Plant | Region, Country | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | K2O | TiO2 | Mineral Phases | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Neyveli thermal power plant | Tamil Nadu, India | 52.2 | 25.8 | 3.6 | 3.0 | 2.2 | 0.6 | 1.3 | Quartz (SiO2), Calcite (CaCO3), Hematite (Fe2O3) | [81] |
| Calaca Coal-fired power plant | Batangas, Philippines | 79.3 | − | 4.0 | 5.6 | 4.4 | 1.6 | 0.08 | Anorthite (CaAl2Si2O8), Quartz (SiO2), Goethite (α-FeOOH), Mullite (3Al2O3·2SiO2) | [82] |
| CFBC, coal-fired power plant | Philippines | 27.4 | 23.2 | 21.7 | 16.5 | 6.2 | 0.9 | 0.9 | Quartz (SiO2), Calcite (CaCO3), Magnetite (Fe3O4), Hematite (Fe2O3), Maghemite (γ-Fe2O3), Illite, Biotite | [118,119] |
| − | Italy | 37.3 | 16.1 | 30.8 | 6.13 | − | 5.28 | − | − | [83] |
| − | Italy | 59.5 | 27.3 | 2.9 | 1.8 | − | 2.7 | 0.6 | − | [84] |
| Country | Precursors | Activators | Admixture | Aggregate | 28-day compressive strength (MPa) | Ref. |
|---|---|---|---|---|---|---|
| Italy | Fly ash |
SS, NaOH | Metakaolin, Printed circuit board (nonmetallic) |
Sand | 15.0−35.0 | [81] |
| Philippines | Fly ash | SS, NaOH | Silica fume | Fine aggregate | 12.6 | [82] |
| Italy | Fly ash |
SS, NaOH | Metakaolin, | Expanded glass | 6.0 | [83] |
| Italy | Fly ash | SS, NaOH | Metakaolin, | Expanded waste glass | 7−10 | [84] |
3.4. Slag-Based Geopolymers
| Country | Precursors SiO2 =32.5-35.7%, Al2O3=9.94-11.6%, Fe2O3=0.32-1.25% CaO=32.5-42.1, MgO=7-9.3% |
Activators | Admixture | Aggregate | 28-day Compressive (MPa) | Ref. |
|---|---|---|---|---|---|---|
| Italy | Slag | SS, NaOH | Metakaolin, Wollastonite | River Sand | 34.5 | [85] |
| Turkey | Slag | NaOH | Ca(OH)2, waste brick powder | − | 25−33 | [86] |
| India | Slag | SS, NaOH, | Dholpur Stone | Sand | 15 | [87] |
| Jaiselmer Stone | 24 | |||||
| Kota Stone | 25 | |||||
| Makarana Stone | 25 | |||||
| Turkey | Slag | SS, NaOH | Slaked lime Brick powder |
River Sand | 18.1−30.6 | [88] |
| NaOH | 11.5−26.2 | |||||
| KOH | 11.1−27.1 |
3.5. Pyroclastic Material-Based Geopolymers
| Country | Precursors In Molar Ratio: VA: Si/Al=2.5 VS: Si/Al=2.2 PM:Si/Al=3.1 |
Activators | Admixture | Aggregate | 28-day compressive strength (MPa) | Ref. |
|---|---|---|---|---|---|---|
| Italy | Volcanic ash | SS, NaOH | Metakaolin |
18–50 (21d) |
[89] | |
| Volcanic soil | 17–51 (21 d) | |||||
| Volcanic ash | KOH, KS | 29–71 (21 d) | ||||
| Volcanic soil | 30–89 (21 d) | |||||
| Italy | Volcanic ash | SS, NaOH | Metakaolin | Volcanic rock | 39 | [90] |
| None | 45 | |||||
| Volcanic soil | Volcanic soil | 21 | ||||
| None | 25 | |||||
| Italy | Volcanic ash, | SS, NaOH | Metakaolin, Ca(OH)2 | None | 34 | [91] |
| Metakaolin | 25 | |||||
| Italy | Volcanic ash | SS, NaOH | Metakaolin | None | 45 | [92] |
| Volcanic rock | 39 | |||||
| Volcanic soil | None | 25 | ||||
| Volcanic soil | 21 | |||||
| Italy | Volcanic ash, | SS, NaOH | Metakaolin, Aluminum Powder |
– | – | [93] |
| Volcanic soil, | – | |||||
| Pumice | – | |||||
| Italy | Pumice | SS, NaOH | Metakaolin, | 7.2–12.7 | [94] |
3.6. Ceramic Waste-Based Geopolymers
| Country | Precursors SiO2=57.3, Al2O3=32.7, Fe2O3=3.5, K2O=1.4, Na2O=0.78, CaO=0.83, with quartz, mica, feldspar, and hematite |
Activators | Admixture | Aggregate | 28-day compressive strength (MPa) | Ref. |
|---|---|---|---|---|---|---|
| Italy | Porcelain and stoneware waste, | SS, NaOH | Metakaolin | – | 30 | [95] |
| Raw pressed ceramic waste, | 25 | |||||
| Calcined Raw pressed ceramic, | 41 | |||||
| Gypsum | 4 | |||||
| Mixed | 38 | |||||
| Italy | Ceramic tile waste | SS, NaOH | Metakaolin, lime | 3.5–14.3 | [96] | |
| Italy | Tile waste or Brick Wate |
SS, NaOH | Metakaolin, lime | Carbonate sand, | – | [97] |
| Volcanic aggregate, | – | |||||
| marble powder | – | |||||
| Italy | Ceramic tile waste | SS, NaOH | Metakaolin | None | 14–27 | [98] |
| Carbonate sand | 18–32 | |||||
| Silica sand | 6–7 | |||||
| Tile waste | 6–16 | |||||
| Italy | Brick waste | SS, NaOH | Fly Ash | Brick waste | 13.4 | [99] |
| None | 5.3 |
3.7. Geopolymers Formulated from Other Materials
| Country | Precursors | Activators | Admixture | Aggregate | 28-day compressive strength (MPa) | Ref. |
|---|---|---|---|---|---|---|
| Italy | Tuff Waste (SiO2=58.8%, Al2O3=31.82% Fe2O3=2.99% CaO=3.10% Na2O=3.44% MgO=1.11% K2O=9.34% TiO2=0.53%) |
SS, NaOH | Fly ash | Tuff waste | 1.3−1.5 | [99] |
| None | 0.8 | |||||
| Portugal | Silica fume waste (SiO2=97% Al2O3=0.15% Fe2O3=0.03% CaO=0.2% Na2O=0.05% MgO=0.30% K2O=0.80%) |
SS, NaOH | Polycarboxylate-based superplasticizer | − | 10−12 | [100] |
| None | 14−15 | |||||
| Portugal | Tungsten mine mud waste (SiO2=46.7% Al2O3=17.0% Fe2O3=15.7% CaO=0.69% Na2O=0.85% MgO=4.83%) |
SS, NaOH | Portland cement, glass, metakaolin |
Expanded Cork | 10−19.5 | [101] |
| Italy | Santena Clay (Smectite- Montmorillonite, Kaolinite, Illite, quartz, feldspars) |
KOH, KS | − | − | Not conducted | [102] |
4. Compatibility Criteria in Using Geopolymers for Architectural Heritage Structure Conservation
4.1. Aesthetic Compatibility
4.1.1. Color
| Binder Composition | Alkali Activator Ratio (NS/ NaOH) |
Aggregates | Reference Color (Lref) | Color Difference (ΔE) | Change in Color Coordinate | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| ΔL | Δa | Δb | ||||||
| MK | In molar ratio | Dolostone | Dolostone | 4.7 |
-1.1 | 1.1 | 4.4 | [69] |
| Sandstone | Sandstone | 10.9 | -10.8 | -0.52 | 1.6 | |||
| Tile+10 wt% MK | 1 | SS | Messina Bricks (Ancient Brick) |
19 | n.v. | [97] |
||
| CS | 9 | |||||||
| CER | 6 | |||||||
| Tile+20 wt% MK | 1 | SS | 21 | |||||
| CS | 12 | |||||||
| CER | 14 | |||||||
| Tile+20 wt% MK | 2.3 | SS | n.v. | |||||
| CS | 12 | |||||||
| CER | 18 | |||||||
| Tile+10 wt% MK | 1 | SS | Gela Bricks (Modern Brick) |
6.5 | ||||
| CS | 12 | |||||||
| CER | 13 | |||||||
| Tile+20 wt% MK | 1 | SS | 8.5 | |||||
| CS | 8.3 | |||||||
| CER | 10 | |||||||
| Tile+ 20 wt% MK | 2.3 | SS | n.v. | |||||
| CS | 7 | |||||||
| CER | 8 | |||||||
| Tile+10 wt% MK | 1 | CS | Odéon Bricks (Ancient Brick) | Recognizable | n.v. | [98] | ||
| Brick+10 wt% MK | Very Similar | |||||||
| Pumice+20 wt% MK | 1.33 | None | Raw Pumice | 1.3 | 0.5 | 0.25 | 1.16 | [94] |
| Pumice+30 wt% MK | 1.19 | 3.2 | -2.9 | 0.24 | 1.2 | |||
| Santena Clay | 1 (KS/ KOH) | None | Raw Clay | 11.09 |
-9.49 | -2.16 | –5.32 | [102] |
| Tile+10 wt% MK | 1 | SS | Raw Ceramic Tile |
12 | n.v | [98] | ||
| CS | 4 | |||||||
| CER | 6 | |||||||
| Tile+20 wt% MK | 1 | SS | 3.2 | n.v. | ||||
| CS | 9.2 | |||||||
| CER | 9.5 | |||||||
| Tile+20 wt% MK | 2.3 | SS | n.v. | |||||
| CS | 5 | |||||||
| CER | 1.5 | |||||||
| VA+ 20 wt% MK | n.v. | None | Prior to Weathering |
1.87 | n.v. | [94] | ||
| VA | 2.49 | |||||||
| VS+20 wt% MK | None | 7.10 | ||||||
| VS | 6.19 | |||||||
| Santena Clay | 1 | None | Prior to Weathering | 2.09 | 2.05 | 0.36 | 0.23 | [102] |
4.1.2. Presence of Efflorescence
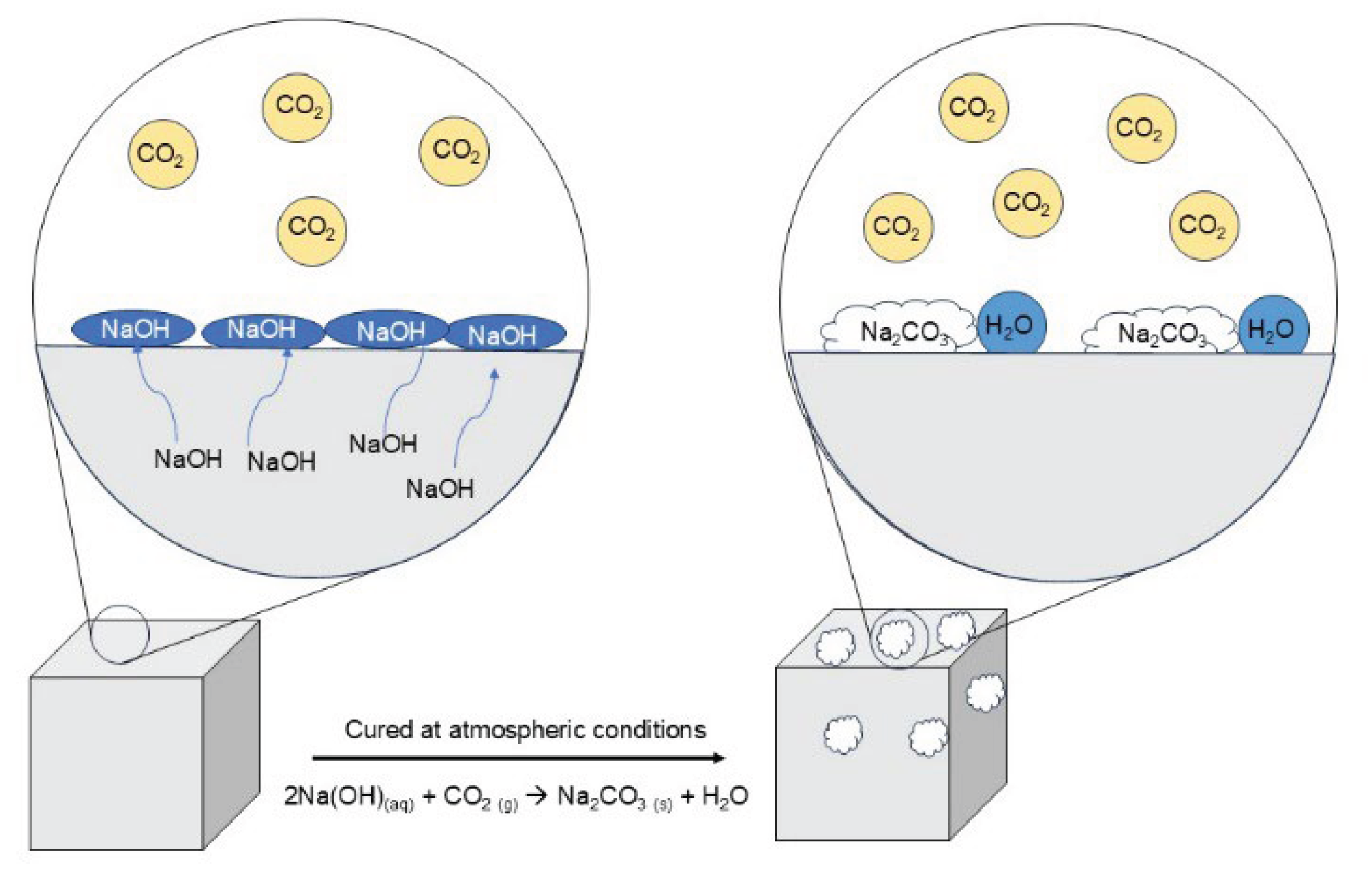
4.1.3. Texture
4.2. Chemical Compatibility
4.2.1. Microstructure
4.2.2. Ionic Release
4.3. Physical Compatibility
| Property | Geopolymer type | Geopolymer value | Reference material | Reference material value | Percent change (%) | Similarity | Ref. |
|---|---|---|---|---|---|---|---|
| Open Porosity (%) | Pumice+MK | 36.5 | Sicilian Limestone | − | − | − | [94] |
| MK+dolostone | 17.8 | Dolostone | − | − | Similar (from author) | [69] | |
| MK+sandstone | 14.1 | Sandstone | 2−6 | 57−85 | High | ||
| FA | 20-40 | Natural Hydraulic Lime | 50 | 20−60 | Very High to High | [100] | |
| Tuff | 39.4-42.3 |
Aerial Lime Mortar | 25.9 | 52−63 | High | [99] | |
| NHL | 23.5 | 67−80 | High | ||||
| OPC | 18.8 | 113−128 | Medium | ||||
| Repair Mortars | 26.2 | 50−61 | High | ||||
| Historical Magnesian Mortar | 30−40 | 6−31 | Very High | ||||
| Natural Limestone | 24−30 | 39−40 | Very High | ||||
| Brick | 26.2-34.2 | Aerial Lime Mortar | 25.9 | 1.1−23 | Very High | ||
| NHL | 23.5 | 11.5−45.5 | Very High | ||||
| OPC | 18.8 | 39.4−81 | Very High | ||||
| Repair Mortars | 26.2 | 0−31 | Very High | ||||
| Historical Magnesian Mortar | 30−40 | 12.7−15 | Very High | ||||
| Natural Limestone | 24−30 | 8.3−13 | Vert High | ||||
| Water Absorption (%) | Tuff | 26.7-30.97 |
n.d. | − | − | − | [99] |
| Brick | 15.71-19.75 | n.d. | − | − | − | ||
| Capillarity Absorption (mg/cm2 s-1/2) | Tuff | 20.9-33.4 | Aerial Lime Mortar | 13.9 | 50−140 | Medium to High | [99] |
| NHL | 9.40 | 122−225 | Medium | ||||
| OPC | 3.4 | 514−882 | Very Low | ||||
| Repair Mortars | 24.2 | 13.6−38.01 | Very High | ||||
| Historical Magnesian Mortar | 28.5 | 17−27 | Very High | ||||
| Natural Limestone | 31.3 | 6.7−33 | Very high | ||||
| Brick Based | 19.5-26.6 | Aerial Lime Mortar | 13.9 | 40−91 | High | ||
| NHL | 9.40 | 107−183 | Medium | ||||
| OPC | 3.4 | 473−682.3 | Low | ||||
| Repair Mortars | 24.2 | 10−19 | Very High | ||||
| Historical Magnesian Mortar | 28.5 | 6.6−31 | Very High | ||||
| Natural Limestone | 31.3 | 15−37 | Very High | ||||
| Bulk Density (g/cm3) | MK+dolostone | 2.8 | Dolostone | 2.7 | 3.7 | Very High | [69] |
| MK+sandstone | 2.9 | Sandstone | 2.9 | 0 | Very High | ||
| Pumice+MK | 1.43-1.46 | Silician Limestone | − | − | [94] | ||
| Silica Fume | 1.0-1.2 | NHL | 1.35 | 11−25 | Very High | [100] | |
| Tuff | 1.35-1.4 | - | − | − | [99] | ||
| Brick | 1.73-1.76 | - | − | − |
4.4. Mechanical Compatibility
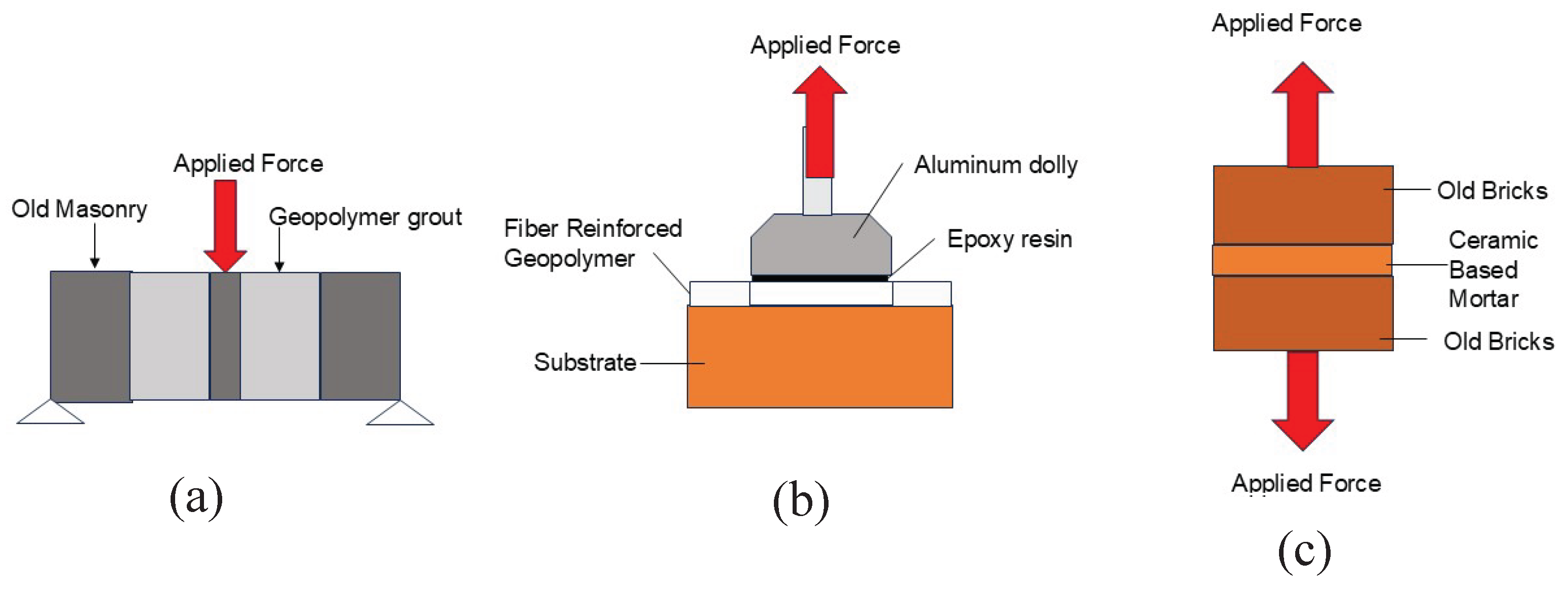
4.4.1. Adhesion
4.4.2. Surface Hardness
4.4.3. Compactness
4.4.4. Compressive Strength

4.4.5. Tensile Strength
4.4.6. Flexural Strength
| Property | Geopolymer type | Geopolymer | Reference | Ref. material value | Percent Change% | Similarity | Ref. |
|---|---|---|---|---|---|---|---|
| Adhesion (MPa) | Silica fume | 0.6−0.9 | NHL | 0.1 | 500−800 | Very Low | [100] |
| Slag | 0.89−1.65 | SM Bricks (substrate 1) | − | − | − | [85] | |
| Slag | 1.21−2.81 | EM Bricks (substrate 2) | − | − | − | ||
| Surface Hardness | Slag | 31−39 | Historical Mortars | 10−20 | 95−210 | Medium | [86] |
| Tuff | 26.5−53 | In-situ Mortar | − | − | − | [99] | |
| Brick | 31.2−75.8 | In-situ Mortar | − | − | − | ||
| Compactness (m/s) | Slag | 3000−4000 | In-situ Mortar | 2000-3000 m/s | 50 | Very High | [86] |
| Compressive (MPa) | Pumice | 3−6 MPa | Type A WSL | 8−20 MPa | 62−70 | High | [94] |
| Type B1 WSL | 3−10 MPa | 0−40 | Very High | ||||
| Type B2 WSL | 1−3 MPa | 0−200 | Very High to Medium | ||||
| Type C WSL | 0.5−1.5 MPa | 100−1100 | Medium to Very Low | ||||
| Silica Fume | 10−16 | NHL | 9 MPa | 11−77 | High to Very High | [100] | |
| Tuff | 0.81-1.48 | Aerial Lime Mortar | 0.5 | 62−196 | Medium to High | [99] | |
| NHL | 3.7 | 78−60 | High | ||||
| OPC | 24.8 | 94−96 | High | ||||
| Repair Mortars | 3.5 | 57−77 | High | ||||
| Historical Magnesian Mortar | − | − | − | ||||
| Natural Limestone | 10.1 | 85−92 | High | ||||
| Brick | 5.3−13.4 | Aerial Lime Mortar | 0.5 | 960−2580 | Very Low | ||
| NHL | 3.7 | 43−262 | Medium to High | ||||
| OPC | 24.8 | 46−75 | High to Very High | ||||
| Repair Mortars | 3.5 | 51−282 | Medium to High | ||||
| Historical Magnesian Mortar | − | − | − | ||||
| Natural Limestone | 10.1 | 47.5−32.7 | Very High | ||||
| MK | 18 | Dolostone | − | − | [69] | ||
| MK | 21 | Sandstone | − | − | |||
| FA | 1.8−12.1 | Plain Mortar | 12.5 | 0−86 | High to Very High | [82] | |
| Tensile (MPa) | FA | 0.6−2.2 | Plain Mortar | 1.6 | 37.5−62.5 | High to Very High | [85] |
| Slag | 4.8 | None | n.d. | − | − | ||
| Flexural (MPa) | Silica Fume | 0.2 | NHL | 1.0−1.5 | 80−86 | High | [100] |
| Tuff | 0.4−1.23 | Aerial Lime Mortar | 0.24 | 66−412 | Medium to High | [99] | |
| NHL | 1.00 | 23−60 | Very High | ||||
| OPC | 4.70 | 74−91 | High | ||||
| Repair Mortars | − | − | |||||
| Historical Magnesian Mortar | − | − | |||||
| Natural Limestone | − | − | |||||
| Brick | 2.85−4.58 | Aerial Lime Mortar | 0.24 | 1087−1808 | Very Low | ||
| NHL | 1.00 | 185−358 | Low to Medium | ||||
| OPC | 4.70 | 2−39 | Very High | ||||
| Repair Mortars | − | − | − | ||||
| Historical Magnesian Mortar | − | − | − | ||||
| Natural Limestone | − | − | − | ||||
| MK | 3.2 | Dolostone | − | − | − | [69] | |
| MK | 3.6 | Sandstone | − | − | − |
5. Recent Applications of Geopolymers in Architectural Heritage Structure Conservation
5.1. Repair Mortars
5.2. Consolidants
5.3. Masonry Strengthening
5.4. Others
| Country | Geopolymer | Purpose/Application | Original Materials | Architectural Heritage Structure | Ref |
|---|---|---|---|---|---|
| Morrocco | Metakaolin | Repair mortar | Lime-based mortar | Moroccan Historical Monument | [68] |
| Italy | Metakaolin | Repair mortar | Sandstone and dolostone | Not specified | [69] |
| Turkey | Slag | Repair mortars | Low strength mortars | Turkish Historical Masonry | [86] |
| Italy | Volcanic ash | Repair mortars/binders | Not specified | Catania Architectures | [89] |
| Volcanic soil | |||||
| Italy | Volcanic ash | Repair mortars/binders | Not specified | Catania Architectures | [90] |
| Volcanic soil | |||||
| Italy | Volcanic ash | Repair mortars/binders | Not specified | Catania Architectures | [92] |
| Volcanic soil | |||||
| Italy | Volcanic ash, | Repair mortars/binders | Not specified | Not specified | [93] |
| Volcanic soil, | |||||
| Pumice | |||||
| Italy | Volcanic ash, | Repair mortars | Mosaics | Cathedral of Monreale, Sicily | [91] |
| Italy | Pumice | Repair mortars | Sicilian limestones | Sicilian Baroque Cities | [94] |
| Italy | Tile waste or Brick waste |
Repair mortars | Ancient bricks | Cathedral of Messina, Sicily, and Archeological Ruins in Gela, Caltanissetta |
[97] |
| Italy | Ceramic tile waste | Repair mortar | Ancient bricks | Roman Odéon of Catania | [98] |
| Italy | Brick waste | Repair mortar | Limestone, lime mortars | Not specified | [99] |
| Italy | Tuff waste | Repair mortar | Limestone, lime-based mortars | Not specified | [99] |
| Portugal | Tungsten mine waste | Repair mortar | None | Not specified | [101] |
| Italy | Metakaolin | Consolidant | Tuff, concrete, ceramic | None | [70] |
| Portugal | Metakaolin | Consolidant | Ceramic tiles | None | [71] |
| Portugal | Metakaolin | Consolidant | Ceramic tiles | None | [72] |
| Italy | Metakaolin | Consolidant | Ceramic artworks | None | [75] |
| Czech Republic | Metakaolin (LO5) | Consolidant | Ceramic tiles | None | [77] |
| Italy | Porcelain stoneware Waste | Consolidant | Ceramic artworks | None | [95] |
| Raw pressed ceramic | |||||
| Calcined raw pressed ceramic | |||||
| Gypsum | |||||
| Mixed | |||||
| Portugal | Silica fume | Consolidant/Grout | Hydraulic lime | Non specified | [100] |
| Italy | Local clay | Consolidant/Plaster | Local clay | Earthen Architectures | [102] |
| Turkey | Slag | Strengthening historical masonry | Ceramic brick masonry | Turkish Historical Architectures | [90] |
| Italy | Fly ash | Strengthening historical masonry | Tuff and brick masonry | Not specified | [83] |
| Philippines | Fly Ash | Strengthening historical masonry | Unreinforced stone masonry | Not specified | [82] |
| Italy | Slag | Strengthening historical masonry | Ancient and modern brick masonry | Not specified | [85] |
| Czech Republic | Metakaolin | Valorization | None | None | [73] |
| Czech Republic | Metakaolin (LO5) | Valorization | None | None | [74] |
| Italy | Ceramic tile waste | Valorization | None | None | [96] |
| Italy | Fly ash | Valorization | None | None | [81] |
| Italy | Metakaolin | Colored geopolymer | None | None | [76] |
| India | Slag | Colored geopolymer | None | None | [87] |
| Czech Republic | Metakaolin (LO5) | Investigation of maturation environment | None | None | [78] |
| China | Metakaolin, | Investigation of modified activators (Na2SiO3 and K2SiO3) | None | None | [79] |
| Italy | Fly ash | Investigation of lightweight Aggregates | None | None | [106] |
6. Case Study of Compatible Geopolymers for Heritage Structures in the Philippines
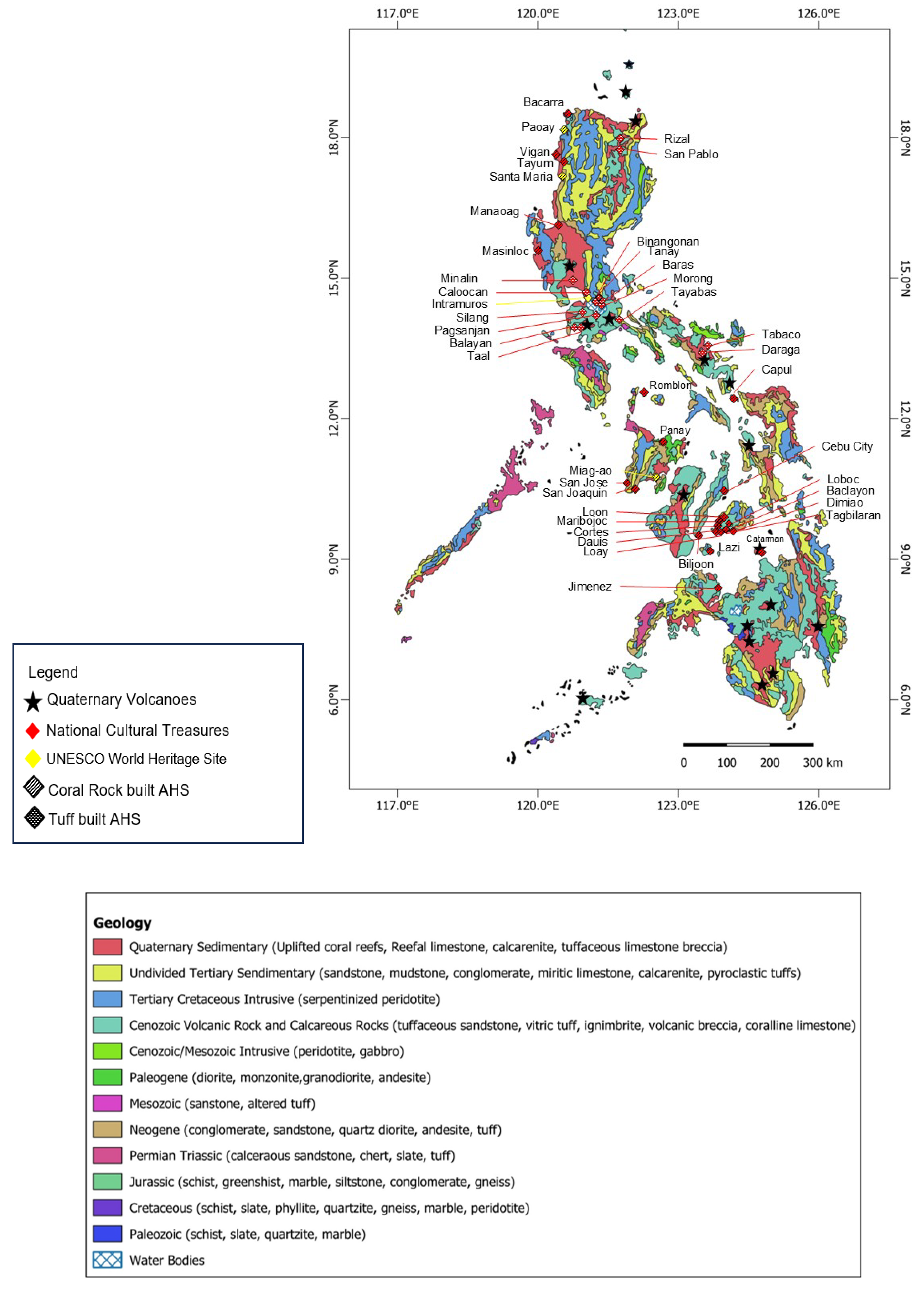
| Type | Inherent color | Chromatic similarity to Philippine tuff | Chromatic similarity to coral rock | Degree of valorization | Abundancy in Philippines |
|---|---|---|---|---|---|
| Metakaolin | Creamy white | Yes | Yes | Low | High |
| Fly Ash | Grey | Yes | No | High | High |
| GGBS | White | Variable | Yes | High | High |
| Volcanic ash | Light grey | Yes | No | Medium | High |
| Volcanic soil (Ghaira) | Red | No | No | Medium | High |
| Pumice | Grey to whitish | Yes | No | Low | Low |
| Ceramic | Red | No | No | High | Variable |
| Tuff waste | Grey | Yes | No | High | Low |
| Silica fume | Grey | Yes | No | High | High |
| Mine waste | − | − | − | High | High |
7. Summary and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AHS | Architectural heritage structures |
| ASM | Artificial stone mortars |
| EC | Electrical conductivity |
| FA | Fly Ash |
| FRCM | Fiber reinforced cementitious materials |
| ICOMOS | International council on monument and sites |
| MK | Metakaolin |
| NDT | Non-destructive techniques |
| NHL | Natural hydraulic lime |
| NMF | Non-metallic fraction |
| OPC | Ordinary Portland cement |
| PCB | Printed circuit board |
| PM | Pumice |
| SEM | Scanning electron microscope |
| SF | Silica fume |
| TEOS | Tetraethyl orthosilicate |
| UCS | Unconfined compressive strength |
| UNESCO | United Nations Educational, Scientific, and Cultural Organization |
| UPV | Ultrasonic pulse velocity |
| URM | Unreinforced masonry |
| VA | Volcanic ash |
| VS | Volcanic soil |
| XRD | X-ray diffraction |
References
- Siegesmund, S.; Török, Á. Building Stones. In: Stones in Architecture: Properties and Durability, 5th ed.; Siegesmund, S., Snethlage, R., Eds; Springer: Heidelberg, Berlin, Germany, 2014, 11-97. [CrossRef]
- Pötzl, C.; Siegesmund, S.; López-Doncel, R.; Dohrmann, R. Key parameters of volcanic tuffs used as building stone: A statistical approach. Environ. Earth Sci. 2022, 81(10). [CrossRef]
- Martínez-Martínez, J.; Pola, A.; García-Sánchez, L.; Reyes Agustin, G.; Osorio Ocampo, L. S.; Macías Vázquez, J. L.; Robles-Camacho, J. Building stones used in the architectural heritage of Morelia (México): Quarries location, rock durability and stone compatibility in the monument. Environ. Earth Sci. 2018, 77(5), 167. [CrossRef]
- Di Benedetto C.; Cappelletti, P.; Favaro, M.; Graziano, S.F.; Langella, A.; Calcaterra, D.; Colella, A. Porosity as key factor in the durability of two historical building stones: Neapolitan Yellow Tuff and Vicenza Stone, Eng. Geol. 2015, 193, 310–319. [CrossRef]
- Germinario, L.; Török, Á. Variability of technical properties and durability in volcanic tuffs from the same quarry region – examples from Northern Hungary. Eng. Geol. 2019, 262, 105319. [CrossRef]
- Lubelli, B.; Nijland, T. G.; Tolboom, H.J. Moisture induced weathering of volcanic tuffstone. Constr. Build. Mater. 2018, 187, 1134–1146. [CrossRef]
- Nijland, T. G.; Van Hees, R. P. J. The volcanic foundation of Dutch architecture: Use of Rhenish tuff and trass in the Netherlands in the past two millennia. Heron 2016, 61(2), 69–98. https://www.researchgate.net/publication/310614592_The_volcanic_foundation_of_Dutch_architecture_Use_of_Rhenish_tuff_and_trass_in_the_Netherlands_in_the_past_two_millennia.
- Siedel, H.; Rust, M.; Goth, K.; Krüger, A.; Heidenfelder, W. Rochlitz porphyry tuff (Rochlitzer Porphyrtuff): A candidate for ‘Global Heritage Stone Resource’ designation from Germany. Episodes 2019, 42(2), 81–91. [CrossRef]
- Korat, L.; Mirtič, B.; Mladenovič, A.; Pranjić, A. M.; Kramar, S. Formulation and microstructural evaluation of tuff repair mortar. J. Cult Herit 2015, 16(5), 705–711. [CrossRef]
- Cárdenes, V.; Cabrera-Guillén, D.; López-Piñeiro, S.; de Argandoña, V. G. R.; Rubio-Ordóñez, A. The Historical Significance of the Welded Tuffs from Arucas, Canary Islands. Geoheritage 2022, 14(46). [CrossRef]
- Valido, J. A.; Cáceres, J. M.; Sousa, L. M. O. Physical and mechanical properties of Ignimbrite from Arucas, Canary Islands. Environ Earth Sci. 2023, 82(13), 342. [CrossRef]
- Siegesmund, S.; Pötzl, C.; López-Doncel, R.; Gross, C. J.; Dohrmann, R.; Ufer, K. Overview and quality assessment of volcanic tuffs in the Mexican building heritage. Environ. Earth Sci. 2022, 81(17). [CrossRef]
- López-Doncel, R.; Wedekind, W.; Aguillón-Robles, A.; Dohrmann, R.; Molina-Maldonado, S.; Leiser, T.; Wittenborn, A.; Siegesmund, S. Thermal expansion on volcanic tuff rocks used as building stones: Examples from Mexico. Environ. Earth Sci. 2018., 77(9). Scopus. [CrossRef]
- Ruiz-Ruiz, R.; Alonso-Guzman, E. M.; Martinez-Molina, W.; Chavez-Garcia, H. L.; Arreola-Sanchez, M.; Borrego-Perez, J. A.; Navarrete-Seras, M. A.; Velazquez-Perez, J. A.; Morales-Rosales, L. A. Environmental Decay of Ignimbrite Patrimonial Monuments in the Dry, Urban, and Non-Industrial Atmosphere of Morelia (México). Heritage 2023, 6(3), 3137–3158. [CrossRef]
- Padilla-Ceniceros, R.; Pacheco-Martínez, J.; López-Doncel, R.A.; Orenday-Tapia, E.E. Rock deterioration in the masonry walls of the Cathedral Basilica of Aguascalientes. Mexico. Rev. Mex Cienc. Geol 2017. 34, 138–139. [CrossRef]
- López-Doncel, R.; Wedekind, W.; Dohrmann, R.; Siegesmund, S. Moisture expansion associated to secondary porosity: An example of the Loseros Tuff of Guanajuato, Mexico. Environ. Earth Sci. 2013, 69(4), 1189–1201. [CrossRef]
- González-León, L. I.; Canet, C.; Lozada-Amador, E.; Alvarado-Sizzo, I.; Lagarda-García, F. O.; Cruz-Pérez, M. Á.; García-Alonso, E.; Mora-Chaparro, J. C.; Torres, P. S. U.; Salgado-Martínez, E. Tezoantla Tuff («Cantera de Tezoantla», Hidalgo state): The first Mexican “Heritage Stone.” Episodes 2024, 47(1), 109–119. [CrossRef]
- Elert, K.; Ruiz-Agudo, E.; Jroundi, F.; Gonzalez-Muñoz, M. T.; Fash, B. W.; Fash, W. L.; Valentin, N.; de Tagle, A.; Rodriguez-Navarro, C. Degradation of ancient Maya carved tuff stone at Copan and its bacterial bioconservation. Npj Mater. Degrad. 2021, 5(1). [CrossRef]
- Jroundi, F.; Elert, K.; Ruiz-Agudo, E.; Gonzalez-Muñoz, M. T.; Rodriguez-Navarro, C. Bacterial Diversity Evolution in Maya Plaster and Stone Following a Bio-Conservation Treatment. Front. Microbiol. 2020, 11, 599144. [CrossRef]
- Deniz, B. E.; Topal, T. A new durability assessment method of the tuffs used in some historical buildings of Cappadocia (Turkey). Environ. Earth Sci. 2021, 80(7). [CrossRef]
- Çelik, M. Y.; Güven, Ö. An assessment of the durability of untreated and water repellent-treated cultural heritage stone (Döğer tuff-Turkey) by salt mist and salt crystallization tests. Bull. Eng. Geol. Environ. 2024, 83(5). [CrossRef]
- Çelik, M. Y.; Güven, Ö. Evaluation of the Durability of Untreated and Water-Repellent-Treated Cultural Heritage Stone (Döğer Tuff-Turkey) through Capillary Water Absorption and Freeze-Thaw Tests. Geoheritage 2024, 16(4). [CrossRef]
- Çelik, M. Y.; Sert, M. The role of different salt solutions and their concentration ratios in salt crystallization test on the durability of the Döğer tuff (Afyonkarahisar, Turkey) used as building stones of cultural heritages. Bull. Eng. Geol. Environ. 2020, 79(10), 5553–5568. Scopus. [CrossRef]
- Çelik, M. Y.; Sert, M. An assessment of capillary water absorption changes related to the different salt solutions and their concentrations ratios in the Döğer tuff (Afyonkarahisar-Turkey) used as building stone of cultural heritages. J. Build. Eng. 2021, 35. Scopus. [CrossRef]
- Çelik, M. Y.; Sert, M. An investigation of the pore size distribution variations with salt crystallization tests of Dӧǧer tuff (Afyonkarahisar-Turkey). Bull. Eng. Geol. Environ. 2022, 81(1). [CrossRef]
- Maras, M. M.; Kose, M. M.; Rızaoglu, T. Microstructural characterization and mechanical properties of volcanic tuff (Malatya, turkey) used as building stone for the restoring cultural heritage. Period. Polytechn. Civ. Eng. 2020, 65(1), 309–319. [CrossRef]
- Pötzl, C.; Siegesmund, S.; Dohrmann, R.; Koning, J. M.; Wedekind, W. Deterioration of volcanic tuff rocks from Armenia: Constraints on salt crystallization and hydric expansion. Environ. Earth Sci. 2018, 77(19). [CrossRef]
- Menningen, J.; Klein, C.; Pötzl, C.; Gross, C. J.; Siegesmund, S. Development of restoration mortars and artificial stones for use in restoring cultural heritage sites made from volcanic tuffs. Environ. Earth Sci. 2022, 81(20). [CrossRef]
- Guo, X.; Zhang, Y.; Xu, X.; Dai, S. Petrographic and geochemical analyses to characterise the source of built historical natural stones—A case study of the volcanic stones from historical quarries and Baoguosi Temple in the city of Ningbo, China. Built herit. 2023, 7(1). [CrossRef]
- Wu, Y.; Shen, J.; Zhang, J.; Zhang, B. Environmental Factor Accelerate the Deterioration of Tuff Stone Heritage: A Case Study of a Stone House in Southeast China. Buildings 2022, 12(2). Scopus. [CrossRef]
- Jo, Y. H.; Lee, C. H. Weathering features of a five-story stone pagoda compared to its quarrying site in Geumgolsan Mountain, Korea. Environ Earth Sci. 2022, 81(6). [CrossRef]
- Germinario, L.; Oguchi, C. T. Gypsum, mirabilite, and thenardite efflorescences of tuff stone in the underground environment. Environ. Earth Sci. 2022, 81(242). [CrossRef]
- Germinario, L.; Oguchi, C. T. Underground salt weathering of heritage stone: Lithological and environmental constraints on the formation of sulfate efflorescences and crusts. J. Cult. Herit. 2021, 49, 85–93. Scopus. [CrossRef]
- Manohar, S., Bala, K., Santhanam, M., Menon, A. Characteristics and deterioration mechanisms in coral stones used in a historical monument in a saline environment. Constr. Build. Mater. 2020, 241. [CrossRef]
- Meng, Q.; Dong, Y.; Li, H.; Cui, L. Static and dynamic compressive performance of coral reef limestone: Interpretations of rate effect from laboratory tests. Bull. Eng. Geol. Environ. 2024, 83(5), 194. [CrossRef]
- Xu, J., Huang, X., Zhang, Z.; Jin, G. Pore structure characteristics of coral reef limestone: A combined polarizing microscope and CT scanning study. In Proceedings of Geo Shanghai International Conference 2024 - Volume 3: Transportation Geotechnics, Shanghai, China, 26 May. [CrossRef]
- Wei, X.; Luo, Y.; Tao, Y.; Li, X.; Meng, F. Experimental Research into the Uniaxial Compressive Strength of Low-Density Reef Limestone Based on Image Recognition. Materials 2023, 16(15), 5465. [CrossRef]
- Siegesmund, S.; Gross, C. J.; Dohrmann, R.; Marler, B.; Ufer, K.; Koch, T. Moisture expansion of tuff stones and sandstones. Environ. Earth Sci. 2023, 82(6). [CrossRef]
- Urbina Leonor, L. M.; Sosa Echeverría, R.; Alarcón Jiménez, A. L.; Solano Murillo, M.; Pérez, N. A.; Sánchez Alvarez, P.; Hernández Tellez, J.; Vega, E.; Kahl, J. D. W. Wet Atmospheric Deposition Damage on Coral Limestone: An Assessment for Conservation Purposes. Geoheritage 2025, 17(2), 79. [CrossRef]
- Blows, J. F.; Carey, P. J.; Poole, A. B. Preliminary investigations into Caen Stone in the UK; its use, weathering and comparison with repair stone. Build. Environ. 2003, 38(9-10), 1143-1149. [CrossRef]
- Přikryl, R.; Török, Á. Natural stones for monuments: Their availability for restoration and evaluation. Geol. Soc. Spec. Publ. 2010, 333, 1 – 9. [CrossRef]
- Rozenbaum, O.; Barbanson, L.; Muller, F.; Bruand, A. Significance of a combined approach for replacement stones in the heritage buildings’ conservation frame. C. R. Geosci. 2008, 340(6), 345-355. [CrossRef]
- Török, Á.; Vogt, T.; Löbens, S.; Forgó, L. Z.; Siegesmund, S.; Weiss, T. Weathering forms of rhyolite tuffs. Zeitschrift der Deutschen Gesellschaft für Geowissenschaften 2005, 177-187. [CrossRef]
- Stefanidou, M.; Pachta, V.; Papayianni, I. Design and testing of artificial stone for the restoration of stone elements in monuments and historic buildings. Constr. Build. Mater. 2015, 93, 957-965. [CrossRef]
- Giacobello, F.; Ielo, I.; Belhamdi, H.; Plutino, M. R. Geopolymers and Functionalization Strategies for the Development of Sustainable Materials in Construction Industry and Cultural Heritage Applications: A Review. Materials 2022, 15(5). [CrossRef]
- Abulencia, A. B.; Villoria, M. B. D.; Libre, R. G. D.; Jr., Quiatchon, P. R. J.; Dollente, I. J. R.; Guades, E. J.; Promentilla, M. A. B., Garciano, L. E. O.; Ongpeng, J. M. C. Geopolymers as Sustainable Material for Strengthening and Restoring Unreinforced Masonry Structures: A Review. Buildings 2021, 11(11), 532. [CrossRef]
- Cong, P.; Cheng, Y. Advances in geopolymer materials: A comprehensive review. J. Traffic Transp. Eng. 2021, 8(3), 283-314. [CrossRef]
- Matsimbe, J.; Dinka, M.; Olukanni, D.; Musonda, I. Geopolymer: A Systematic Review of Methodologies. Materials 2022, 15(19):6852. [CrossRef]
- Singh, N.B.; Middendorf, B. Geopolymers as an alternative to Portland cement: An overview. Constr. Build Mater. 2020. 237. [CrossRef]
- Amar, M.; Ladduri, B.; Alloul, A.; Benzerzour, M.; Abriak, N. Microstructure and mechanical properties of geopolymers utilizing excavated soils, metakaolin and slags. J. Build. Eng 2024, 86. [CrossRef]
- Elert, K.; Rodriguez-Navarro, C. Degradation and conservation of clay-containing stone: A review. Construction and Building Materials 2022, 330. [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [CrossRef]
- Andrews, R. The place of systematic reviews in education research. Br. J. Educ. Stud. 2005, 53, 399–416. [CrossRef]
- Jindal-Snape, D.; Hannah, E.F.S.; Cantali, D.; Barlow, W.; MacGillivray, S. Systematic literature review of primary–secondary transitions: International research. Rev. Educ. 2020, 8, 526–566. [CrossRef]
- Pradines, S.; Balestra, F. Excavations on the Coral Mosques of the Maldives. J. Mater. Cult. Muslim World 2021, 2(1–2), 200–226. [CrossRef]
- Cayme, J.M.C. Chemistry of Lime Mortared Rubble Masonry in Bohol, Philippines. Int. J. Conserv. Sci. 2021, 12 (3), 997-986. https://ijcs.ro/public/IJCS-21-72_Cayme.pdf.
- Abdel–Aty, Y. Y.; Mahmoud, H. S.; Al-Zahrani, A. A. Experimental Evaluation of Consolidation Techniques of Fossiliferous Limestone in Masonry Walls of Heritage Buildings at Historic Jeddah, Kingdom of Saudi Arabia. Adv. Res. Conserv. Sci. 2020, 1(1), 16–33. [CrossRef]
- Cassar, J.; Cefai, S.; Grima, R.; Stroud, K. Sheltering archaeological sites in Malta: Lessons learnt. Herit. Sci. 2018, 6(1), 36. [CrossRef]
- Moradi, Z. The role of coral in art and architecture: An overview. Int. J. Aquat. Biol. (2016)., 4(2), 125–142. [CrossRef]
- Pradines, S. The ‘Trees from the Sea’ Coral Architectures and Indian Ocean Maritime Resources. J. Res. Archit. Plan. 2024, 34(1), 01–17. [CrossRef]
- Ali, B. S.; Castro, J. J.; Omi, S.; Nazimi, K. Preserving the Past: Investigating Zanzibar’s Ancient Construction Materials for Sustainable Heritage Conservation. Buildings 2024, 14(7), 2129. [CrossRef]
- Xiaoli Ji; Xiangbo Huang; Shiyun Zhong; Junli Zhou. Advancing toward a low-carbon infrastructure: Emission reduction potential of geopolymer road maintenance. Mater. Today Sustain 2025, 30, 101121. [CrossRef]
- Wong, L.S. Durability Performance of Geopolymer Concrete: A Review, Polymers 2022, 14 (5), 868. [CrossRef]
- Opiso, E.M.; Tabelin, C.B.; Maestre, C.V.; Aseniero, J.P.J.; Arima, T.; Villacorte-Tabelin, M. Utilization of Palm Oil Fuel Ash (POFA) as an Admixture for the Synthesis of a Gold Mine Tailings-Based Geopolymer Composite. Minerals 2023, 13(2), 232. [CrossRef]
- Opiso, E.M.; Tabelin, C.B., Maestre, C.V.; Aseniero, J.P.J.; Park, I.; Villacorte-Tabelin, M. Synthesis and characterization of coal fly ash and palm oil fuel ash modified artisanal and small-scale gold mine (ASGM) tailings based geopolymer using sugar mill lime sludge as Ca-based activator. Heliyon 2021, 7(4). [CrossRef]
- Aseniero, J.P.J.; Opiso, E.M.; Banda M.H.T.; Tabelin, C.B. Potential utilization of artisanal gold-mine tailings as geopolymeric source material: Preliminary investigation. SN Appl. Sci. 2019, 1(35). [CrossRef]
- Mabroum, S; Moukannaa, S.; El Machi, A.; Taha, Y.; Benzaazoua, M.; Hakkou, R. Mine wastes based geopolymers: A critical review. Clean. Eng. Technol 2020, 1. [CrossRef]
- Allali, F.; Joussein, E.; Kandri, N. I.; Rossignol, S. The influence of calcium content on the performance of metakaolin-based geomaterials applied in mortars restoration. Mater. Des. 2016., 103, 1–9. [CrossRef]
- Clausi, M.; Tarantino, S. C.; Magnani, L. L.; Riccardi, M. P.; Tedeschi, C.; Zema, M. Metakaolin as a precursor of materials for applications in Cultural Heritage: Geopolymer-based mortars with ornamental stone aggregates. Appl. Clay Sci. 2016, 132–133, 589–599. Scopus. [CrossRef]
- Ricciotti, L.; Molino, A. J.; Roviello, V.; Chianese, E.; Cennamo, P.; Roviello, G. Geopolymer composites for potential applications in cultural heritage. Environments 2017, 4(4), 1–15. [CrossRef]
- Moutinho, S.; Costa, C.; Cerqueira, Â.; Rocha, F.; Velosa, A. Geopolymers and polymers in the conservation of tile facades. Constr. Build. Mater. 2019, 197, 175–184. [CrossRef]
- Moutinho, S.; Costa, C.; Andrejkovičová, S.; Mariz, L.; Sequeira, C.; Terroso, D.; Rocha, F.; Velosa, A. Assessment of properties of metakaolin-based geopolymers applied in the conservation of tile facades. Constr. Build. Mater. 2020, 259. [CrossRef]
- Mácová, P.; Sotiriadis, K.; Slížková, Z.; Šašek, P.; Řehoř, M.; Závada, J. Evaluation of Physical Properties of a Metakaolin-Based Alkali-Activated Binder Containing Waste Foam Glass. Materials 2020, 13(23), 5458. [CrossRef]
- Perná, I.; Novotná, M.; Řimnáčová, D.; Šupová, M. New Metakaolin-Based Geopolymers with the Addition of Different Types of Waste Stone Powder. Crystals 2021, 11(8), 983. [CrossRef]
- Ricciotti, L.; Occhicone, A.; Manzi, S.; Saccani, A.; Ferone, C.; Tarallo, O.; Roviello, G. Sustainable Materials Based on Geopolymer–Polyvinyl Acetate Composites for Art and Design Applications. Polymers 2022, 14(24). [CrossRef]
- Angelo, A. D.; Arconati, V.; Vertuccio, L.; Viola, V.; Catauro, M. Use of Organic Dyes to Color the Metakaolin-Based Geopolymer: A Physico-Chemical Characterization. Macromol. Symp. 2023, 411(1). [CrossRef]
- Perná, I.; Novotná, M.; Hanzlíček, T.; Rafl, J.; Nováková, M. The application of geopolymers in the renovation of the historical ceramic tiles in the pilgrimage church of St. John of Nepomuk (Czech Republic). Case Stud. Constr. Mater. 2025, 22. [CrossRef]
- Perná, I.; Novotná, M.; Hanzlíček, T.; Šupová, M.; Řimnáčová, D. Metakaolin-based geopolymer formation and properties: The influence of the maturation period and environment (air, demineralized and sea water). J. Ind. Eng. Chem. 2024, 134, 415–424. [CrossRef]
- Jin, G.; Wang, X.; Mao, H.; Ji, S.; Shi, Q. Preparation and properties of metakaolin-fumed-silica geopolymer modified with sodium silicate and potassium silicate activators. Chin. J. Anal. Chem. 2024, 52(1), 100352. [CrossRef]
- Singh, N. B. Fly Ash-Based Geopolymer Binder: A Future Construction Material. Minerals 2018, 8(7), 299. [CrossRef]
- Kakria, K.; Thirumalini, S.; Secco, M.; Shanmuga Priya, T. A novel approach for the development of sustainable hybridized geopolymer mortar from waste printed circuit boards. Resour. Conserv. Recycl. 2020, 163, 105066. [CrossRef]
- Dollente, I. J. R.; Valerio, D. N. R.; Quiatchon, P. R. J.; Abulencia, A. B.; Villoria, M. B. D.; Garciano, L. E. O.; Promentilla, M. A. B.; Guades, E. J.; Ongpeng, J. M. C. Enhancing the Mechanical Properties of Historical Masonry Using Fiber-Reinforced Geopolymers. Polymers 2023, 15(4). [CrossRef]
- Longo, F.; Cascardi, A.; Lassandro, P.; Aiello, M. A. A new Fabric Reinforced Geopolymer Mortar (FRGM) with mechanical and energy benefits. Fibers 2020, 8(8). [CrossRef]
- Vasanelli, E.; Calò, S.; Cascardi, A.; Aiello, M. A. The Use of Lightweight Aggregates in Geopolymeric Mortars: The Effect of Liquid Absorption on the Physical/Mechanical Properties of the Mortar. Materials 2024, 17(8), 1798. [CrossRef]
- Tamburini, S.; Natali, M.; Garbin, E.; Panizza, M.; Favaro, M.; Valluzzi, M. R. Geopolymer matrix for fibre reinforced composites aimed at strengthening masonry structures. Constr. Build. Mater. 2017, 141, 542–552. [CrossRef]
- Maras, M. M. Characterization of performable geopolymer mortars for use as repair material. Struct. Concr. 2021, 22(5), 3173–3188. Scopus. [CrossRef]
- Gupta, V.; Pathak, D. K.; Kumar, R.; Miglani, A.; Siddique, S.; Chaudhary, S. Production of colored bi-layered bricks from stone processing wastes: Structural and spectroscopic characterization. Constr. Build. Mater. 2021, 278, 122339. [CrossRef]
- Kutlusoy, E.; Maras, M. M.; Ekinci, E.; Rihawi, B. Production parameters of novel geopolymer masonry mortar in heritage buildings: Application in masonry building elements. J. Build. Eng. 2023, 76. [CrossRef]
- Finocchiaro, C.; Barone, G.; Mazzoleni, P.; Leonelli, C.; Gharzouni, A.; Rossignol, S. FT-IR study of early stages of alkali activated materials based on pyroclastic deposits (Mt. Etna, Sicily, Italy) using two different alkaline solutions. Constr. Build. Mater. 2020, 262, 120095. [CrossRef]
- Occhipinti, R.; Caggiani, M. C.; Andriulo, F.; Barone, G.; de Ferri, L., Mazzoleni, P. Effect of atmospheric exposure on alkali activated binders and mortars from Mt. Etna volcanic precursors. Mater. Lett. 2022, 315. [CrossRef]
- Finocchiaro, C.; Occhipinti, R.; Barone, G.; Mazzoleni, P.; Andreola, F.; Romagnoli, M.; Leonelli, C. Effects of the addition of slaked lime to alkali-activated pastes based on volcanic ashes from Mt. Etna volcano (Italy). Ceram. Int. 2024., 50(13), 24479–24486. [CrossRef]
- Occhipinti, R.; Caggiani, M. C.; De Ferri, L.; Xu, Z.; Steindal, C. C.; Razavi, N.; Andriulo, F.; Mazzoleni, P.; Barone, G. Structural properties of volcanic precursors-based geopolymers before and after natural weathering. Ceram. Int. 2023, 49(13), 21892–21902. [CrossRef]
- Occhipinti, R.; Lanzafame, G.; Lluveras Tenorio, A.; Finocchiaro, C.; Gigli, L.; Tinè, M. R.; Mazzoleni, P.; Barone, G. Design of alkali activated foamy binders from Sicilian volcanic precursors. Ceram. Int. 2023, 49(23), 38835–38846. [CrossRef]
- Occhipinti, R.; Stroscio, A.; Finocchiaro, C.; Fugazzotto, M.; Leonelli, C.; José Lo Faro, M.; Megna, B.; Barone, G.; Mazzoleni, P. Alkali activated materials using pumice from the Aeolian Islands (Sicily, Italy) and their potentiality for cultural heritage applications: Preliminary study. Constr. Build. Mater. 2020, 259. [CrossRef]
- Ricciotti, L.; Occhicone, A.; Ferone, C.; Cioffi, R.; Tarallo, O.; Roviello, G. Development of Geopolymer-Based Materials with Ceramic Waste for Artistic and Restoration Applications. Materials 2022, 15(23). [CrossRef]
- Fugazzotto, M.; Mazzoleni, P.; Lancellotti, I.; Camerini, R.; Ferrari, P.; Tiné, M.; Centauro, I.; Salvatici, T.; Barone, G. Industrial Ceramics: From Waste to New Resources for Eco-Sustainable Building Materials. Minerals 2023, 13(6), 815. [CrossRef]
- Fugazzotto, M.; Barone, G.; Mazzoleni, P. Mortars Produced by Alkaline Activation of Construction Ceramic Waste: Suitability Assessment within Conservation Issue. Int. J. Archit. Herit 2024. [CrossRef]
- Fugazzotto, M.; Mazzoleni, P.; Stroscio, A.; Barone, G. Creating Mortars through the Alkaline Activation of Ceramic Waste from Construction: Case Studies on Their Applicability and Versatility in Conservation. Sustainability 2024, 16(3). [CrossRef]
- Capasso, I.; D’Angelo, G.; Fumo, M.; del Rio Merino, M.; Caputo, D.; Liguori, B. Valorisation of Tuff and Brick Wastes by Alkali Activation for Historical Building Remediation. Materials 2023, 16(20). [CrossRef]
- Baltazar, L. G. Performance of Silica Fume-Based Geopolymer Grouts for Heritage Masonry Consolidation. Crystals 2022, 12(2). [CrossRef]
- Beghoura, I.; Castro-Gomes, J.; Ihsan, H.; Estrada, N. Feasibility of alkali-activated mining waste foamed materials incorporating expanded granulated cork. Min. Sci. 2017. [CrossRef]
- Rescic, S.; Mattone, M.; Fratini, F.; Luvidi, L. Earthen plasters stabilized through sustainable additives: An experimental campaign. Sustainability 2021, 13(3), 1–31. [CrossRef]
- Geraldes, C. F. M.; Lima, A. M.; Delgado-Rodrigues, J.; Mimoso, J. M.; Pereira, S. R. M. Geopolymers as potential repair material in tiles conservation. Appl. Phys A. 2016, 122(3), 197. [CrossRef]
- Pappalardo, G.; Mineo, S.; Caliò, D.; Bognandi, A. Evaluation of Natural Stone Weathering in Heritage Building by Infrared Thermography. Heritage 2022, 5(3), 2594–2614. [CrossRef]
- Salami, B.; Oyehan, T., Gambo, Y.; Badmus, S.; Tanimu, G.; Adamu, S.; Lateef, S.; Saleh, T. Technological trends in nanosilica synthesis and utilization in advanced treatment of water and wastewater. Environ. Sci. Pollut. Res. 2022, 29. [CrossRef]
- Vasanelli, E.; Calò, S.; Cascardi, A.; Aiello, M. A. The Use of Lightweight Aggregates in Geopolymeric Mortars: The Effect of Liquid Absorption on the Physical/Mechanical Properties of the Mortar. Materials 2024., 17(8), 1798. [CrossRef]
- The Geology of Guadalupe Tuff. UP Geological Society E-Bedrock, UP Geological Society 2021. Available Online: https://medium.com/up-geological-society-e-bedrock/the-geology-of-the-guadalupe-tuff-2ecdb187f55a (Accessed on Nov 2025).
- Korat, L.; Mirtič, B.; Mladenovič, A.; Pranjić, A. M.; Kramar, S. Formulation and microstructural evaluation of tuff repair mortar. J. Cult. Herit 2015, 16(5), 705–711. [CrossRef]
- Heap, M. J.; Farquharson, J. I.; Kushnir, A. R. L.; Lavallée, Y.; Baud, P.; Gilg, H. A.; Reuschlé, T. The influence of water on the strength of Neapolitan Yellow Tuff, the most widely used building stone in Naples (Italy). Bull. Volcanol. 2018, 80(6), 51. [CrossRef]
- Azevedo, A.R.G.; Vieira, C.M.F.; Ferreira, W.M.; Faria, K.C.P.; Pedroti, L.G.; Mendes, B.C. Potential use of ceramic waste as precursor in the geopolymerization reaction for the production of ceramic roof tiles. J. Build. Eng. 2020. [CrossRef]
- Ministero per i beni e le attività culturali Roma, Giugno 2004. Code of Cultural and Landscape Heritage. Available Online: https://www.eui.eu/Projects/InternationalArtHeritageLaw/Documents/NationalLegislation/Italy/itcultlandscapeheritge2004engtof.pdf (Accessed on Nov. 2025).
- Fourteenth (14th) Congress. Republic of the Philippines. Republic Act No. 10066. An act providing for the protection and conservation of the national cultural heritage, strengthening the national commission for culture and the arts (NCCA) and its affiliated cultural agencies, and for other purposes. Availabl Online: https://lawphil.net/statutes/repacts/ra2010/ra_10066_2010.html (Accessed on Nov. 2025).
- Jo, Y. H.; Lee, C. H. Weathering features of a five-story stone pagoda compared to its quarrying site in Geumgolsan Mountain, Korea. Environ. Earth Sci. 2022, 81(6). [CrossRef]
- Liu, J.; Xie, G.; Wang, Z.; Li, Z.; Fan, X.; Jin, H.; Zhange, W.; Xing, F.; Tang, L. Synthesis of geopolymer using municipal solid waste incineration fly ash and steel slag: Hydration properties and immobilization of heavy metals. J. Environ. Manag. 2023, 341, 118053. [CrossRef]
- Lei, Z.; Pavia, S. Geopolymer based on biomass ash from agricultural residues. Constr. Build Mater. 2024., 441, 137471. [CrossRef]
- Lucero, R., Jr.; Salisid, K. B.; Oros, R.; Bongabong, A.; Alguno, A.; Villacorte-Tabelin, M.; Silwamba, M.; Phengsaart, T.; Tabelin, C. B. Nanosilica-Based Hybrid Hydrophobic Coatings for Stone Heritage Conservation: An Overview. Minerals 2025, 15(11), 1134. [CrossRef]
- Eang, K.E.; Igarashi, T.; Fujinaga, R.; Kondo, M.; Tabelin, C.B. Groundwater monitoring of an open-pit limestone quarry: Water-rock interaction and mixing estimation within the rock layers by geochemical and statistical analysis. Int. J. Min. Sc. Technol. 2018, 28, 849-857. [CrossRef]
- Dahan, A.M.E.; Alorro, R.D.; Pacaña, M.L.C.; Baute, R.M.; Silva, L.C.; Tabelin, C.B.; Resabal, V.J.T. Hydrochloric Acid Leaching of Philippine Coal Fly Ash: Investigation and Optimisation of Leaching Parameters by Response Surface Methodology (RSM). Sustain. Chem. 2022, 3(1), 76-91. [CrossRef]
- Pacaña, M.L.C.; Ranay, K.A.; Tabelin, C.B.; Alorro, R.D.; Resabal, V.J.T. Optimization of Mixed Mineral–Organic Acid Leaching for Critical Rare Earth Element Extraction from Philippine Coal Fly Ash. Sustainability 2025, 17(24), 11076. [CrossRef]
- Tabelin, C.B.; Igarashi, T.; Arima, T.; Sato, D.; Tatsuhara, T.; Tamoto, S. Characterization and evaluation of arsenic and boron adsorption onto natural geologic materials, and their application in the disposal of excavated altered rock. Geoderma 2014, 213, 163–172. [CrossRef]
- Tabelin, C.B.; Igarashi, T.; Yoneda, T.; Tamamura, S. Utilization of natural and artificial adsorbents in the mitigation of arsenic leached from hydrothermally altered rock. Eng. Geol. 2013, 156, 58–67. [CrossRef]
- The Philippine Registry of Heritage (PRH), Talampamana ng Pilipinas. Available Online: https://talapamana.ncca.gov.ph (Accessed on Jan 2026).
- National Museum of The Philippines (NMP). Annual Report 2021. Available online: https://weblinks.nationalmuseum.gov.ph/wp-content/uploads/2023/01/30111112/NMP-FY-2021-Annual-Report_compressed.pdf (Accessed on Jan 2026).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).