1. Introduction
The most common oral health disorder is dental caries which results in hard tissue loss[
1]. Nowadays, resin-based composites (RBCs) have replaced amalgam as the gold standard of dental restorative materials, especially after the Minamata Convention on Mercury where it has been advised to drift away from amalgam due to its environmental concerns[
2,
3,
4,
5]. Further advantages of RBCs are the adhesive character to the tooth which allows for minimal-invasive procedures and excellent aesthetics through tooth-coloured restorations. Dentin adhesives function as intermediary connections between dentin and RBCs. They are a solvated monomeric blend of lipophilic and hydrophilic components[
6]. Through research, several approaches of dental adhesive have been established such as etch-and-rinse and self-etch systems. Self-etch dentin adhesives reduce the number of application steps by leaving out the separate process of conditioning the tooth structure and thus saving time in the treatment[
7,
8,
9,
10]. However, the containing monomers in adhesive systems have also been subject of research regarding possible cytotoxic and genotoxic effects[
4,
11,
12,
13].
It is important to investigate the cytotoxicity of dentin adhesives according to ISO standards, as they come into contact with intraoral tissues such as gingival or pulp cells during intraoral application[
11,
14]. It has already been demonstrated in other studies that dentin adhesives have a potential to cause cell damage [
11,
15,
16,
17,
18].
In our first study, the eluates of six different dentin adhesives were tested for their cytotoxicity
in vitro on cell cultures with primary fibroblasts after an observation period of 24 hours[
19]. This screening study showed an initial ranking of cytotoxicity based on qualitative assessments of dentin adhesives[
19,
20,
21]. In order to lean more towards
in vivo situations where residues of adhesives may have prolonged cell contact, a longer period should be tested according to ISO 0993-5[
20]. This study functions as an extension of our first study, in which the same dentin adhesives were investigated but now for a period of 48 hours.
The unique feature of our study remains the triangular evaluation of cytotoxicity[
19]. However, it is precisely the combination of qualitative and quantitative assessment which is crucial for more meaningful conclusions. As in our first study, the comparability with existing studies is difficult because our methods and parameters are unique[
12,
18,
22,
23,
24,
25,
26,
27,
28,
29,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39]. In several studies, cytotoxicity is detected only after 24 hours[
18,
26,
27,
29,
33,
35,
36,
37,
38]. These studies show the short-term effect of cytotoxicity of dentin adhesives. However, only a few studies give results after the combination of different observation periods [
23,
25,
31,
34,
39,
40,
41,
42].
In this study, the comparison of the cytotoxicity of self-etch and etch-and-rinse dentin adhesives after 48 hours was evaluated and allows a comparison to the first observation period of 24 hours.
The study hypothesis of our screening study are as following:
- -
Self-etch adhesives show no different cytotoxicity in relation to etch-and-rinse adhesives regarding the triangulated evaluation.
- -
Sequentially applied substances have different cytotoxic effects other than single applied substances regarding the triangulated evaluation.
4. Discussion
Materials that come into contact with living cells should be biocompatible, i.e. they should not cause cytotoxic, pro-inflammatory, mutagenic or adverse immune reactions. Thus, cytotoxicity tests are crucial for evaluating the biocompatibility of materials. Other studies similar to ours have researched a time-dependent release of monomers due to incomplete polymerization[
4,
40]. The study design was adopted from our first study and is an extension from 24 to 48 hours, thus enabling a direct comparison between the results [
19].
The most important conclusion of the second part of our study consists in no statistically significant differences regarding the cytotoxicity between self-etch and etch-and-rinse dentin adhesives after 48 hours which is consistent with the first part of our study in terms of qualitative evaluation and reactivity index and is in accordance with other studies[
19,
22,
23,
24,
25,
26,
27]. Contradictory, other studies found that self-etch respectively etch-and-rinse dentin adhesives showed more cytotoxicity in the comparison[
18,
22,
28,
31,
33,
34,
35,
43]. However, in all of the comparative studies from other researchers, different methods and materials were used. It should be noted that in the clinical application of etch-and-rinse adhesives conditioning with 37% phosphoric acid is necessary. This step has not been tested as in our first investigation because there are already numerous studies addressing this topic[
19,
44,
45].
First of all, it should be highlighted that the cytotoxicity of the individual dentin adhesives is linked to the cytotoxicity of the individual composition of the substances. Significantly involved in this are the concentrations of the different monomers[
46,
47,
48]. Adhesive systems usually contain a mixture of those monomers which have already been proven to have cytotoxic and cell-modulating properties[
18,
49,
50,
51]. The typical components are HEMA, bis-GMA, UDMA and TEGDMA which have shown to exhibit cytotoxic effects in a time- and concentration dependent manner[
40,
52,
53]. The widely accepted ranking of cytotoxicity of monomers from the highest to the lowest: Bis-GMA, UDMA, TEGDMA, and HEMA [
46,
50,
54]. Bis-GMA can impair protein synthesis and induce reactive oxidative stress resulting in cell death and has been shown to be toxic even in small amounts compared to other monomers[
13]. HEMA, while accepted as the least toxic, is able to delay cell cycle progression in fibroblast by increased formation of reactive oxygen species (ROS)[
55]. Furthermore, due to the hydrophilic character of small monomers like HEMA and TEGDMA they can more easily penetrate through cell membranes[
56,
57,
58]. The induced oxidative stress leads to mitochondrial dysfunctions which may lead to cellular damage, inflammatory response and caspase-mediated cell death (apoptosis)[
13,
59,
60,
61]. Apart from the monomers, free-radical based photo-initiators are also expected to induce ROS formation[
4]. The ranking of cytotoxicity of monomers could not be confirmed in our study regarding the evaluation with significant differences. However, it must be noted that the number of monomers in the eluates of adhesives do not exactly represent the real amount since more hydrophilic monomers as TEGDMA are more likely to be eluted than bis-GMA[
62].
Only AdheSE, Clearfil SE Bond, One-up Bond F Plus and Optibond Solo Plus showed a statistically significant difference to the cell control after 48 hours. However, cultures with Optibond Solo Plus did not present this effect after 24 hours. One possible reason for this could be that the complete extent of the cytotoxicity of Optibond Solo Plus only becomes apparent after 48 hours or that cell changes take time before showing an effect. Similar observations were made in a study where the cytotoxicity universal adhesives were tested after 24 , 48 and 72 hours. While cytotoxic effects have been shown after 48h and 72h, no such effects arose after 24hours [
41]. A possible explanation might be that residual unpolymerized monomers are released over time. However, other studies found that adhesives like Clearfil SE Bond showed highest toxicity in the first 24 hours[
63]. In accordance with most authors, various factors determine the extent of toxic effects such as chemical composition of the material, time of exposure and the specific cell type[
4,
41,
64,
65].
As in the first part of the study, it must be stated that the solvents of the dentin adhesive also have an influence on their cytotoxicity[
24,
48]. The test materials are based on acetone, ethanol and water. The water-based AdheSE, Clearfil SE Bond, One-up Bond F Plus and the ethanol-based Optibond Solo Plus showed statistically less viable cells compared to the cell control. For the acetone-based dentin adhesives this effect could not be observed. As already explained, this might lead to the conclusion that the cytotoxicity is hardly influenced by the individual composition but rather by the summation of their ingredients[
24,
39,
48]. However, the effect of the mentioned water-based adhesives, which were all self-etch adhesives in our study, might rely on the fact that they contain acidic functional monomers, such as 10-Methacryloyloxydecyl dihydrogen phosphate (10-MDP; Hybrid Bond Base), 11-methacryloyloxyundecan-1,1-decarboxylic acid (MAC-10; One-up Bond F Plus Agent A) or 4-Methacryloxyethyl-trimellitic-anhydride (4-META; Clearfil SE Bond Primer). Those might contribute to the cytotoxicity due to their acidic character. It has been shown that 10-MDP promotes inflammatory responses and may suppress cell differentiation[
66]. On the other hand, 4-META has been shown to be more biocompatible which might support the less toxic effect observed in our study of the acetone-based adhesive Hybrid Bond[
67]. Optibond Solo Plus is the only adhesive that we tested which is ethanol-based. Thus, it is hard to draw conclusions on its cytotoxic effect in our study based on its solvent. It would be necessary to have more adhesives with the same solvent in order to determine a link to a possible cytotoxicity based on the solvent. Furthermore, as part of our triangulated evaluation, the number of viable cells is not sufficient to draw possible conclusions about possible harmful cellular responses as not all may end in cell death[
62].
Contrary to the quantitative assessment, every adhesive showed a statistically significant difference to the cell control regarding the qualitative evaluation including the reactivity index. Thus, all the adhesives showed a cytotoxic effect, which is in accordance with previous studies[
23,
24,
25,
26,
28,
34,
36,
37]. Syntac, which is also acetone-based, presented one of the highest reactivity-indexes even after 48 hours with no change compared to 24hours. This is also in accordance with other investigations, which classify Syntac as highly cytotoxic[
32,
35,
39]. The investigation of the working group of Sigusch exhibited also a high cytotoxicity of Syntac in the initial phase of observation with no statistically significant differences between 24 and 48 hours. They argue that Syntac contains a larger proportion of non-linked substances after polymerization, which can then be released to the culture medium over a prolonged period[
39]. Contrary, one study also found a lower cytotoxicity of the dentin adhesive Syntac[
68]. They could not show the known increasing effect of cytotoxicity by mixing primer and bonding adhesive parts. As mentioned in the first part of the study, the proportion of glutaraldehyde could play a role in the high cytotoxicity. Free glutaraldehyde has a high potency of cytotoxicity[
32,
43]. Most of the glutaraldehyde is bound irreversibly in the bonding process of the dentin adhesives. It stabilizes the collagen fiber network, which is exposed after etching. However, it has not yet been possible to prove how many molecules are free in the clinical application process[
39].
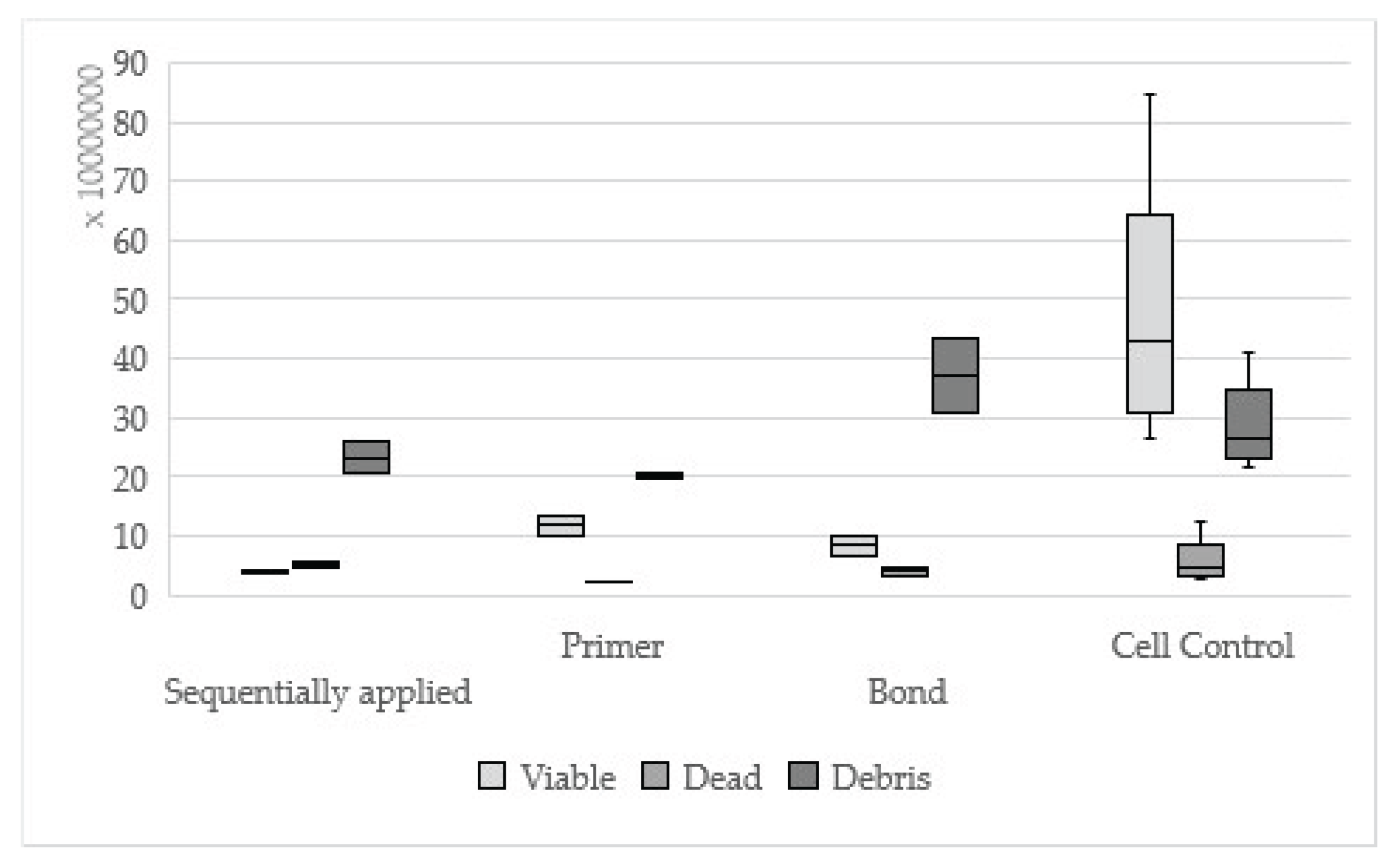
It was reported that Clearfil SE Bond showed a reactivity index of mild to moderate, which has also been confirmed by our results of the second part of this study[
24]. The question arises if an observed high reactivity index that does not cause cell death, might promote cancerous formations. One study has found increased numbers of micro-nucleated cells due to methacrylates which indicates mutagenicity[
4]. Another observed an increase in hypodiploid cell numbers linked to DNA damage in gingival fibroblasts[
69]. As mentioned, glutaraldehyde might be a potential reason for the toxic effect of Syntac and research has already suggested that glutaraldehyde might have carcinogenic potential[
70]. Furthermore, it might be plausible to assume that any toxic effect on a cell might contribute to a malignant transformation as any cell damage and stress response can contribute to DNA damage[
71]. This might be enforced by the toxicity of the other monomers and/or the altered pH-level by the maleic acid in Syntac.
Aside from the composition of adhesives, pH levels also affect cytotoxicity. Other studies have shown that lower pH levels may increase the toxicity of adhesives[
40,
72,
73].
In the second part of the study after 48 hours, Optibond Solo Plus remains the dentin adhesive with the highest reactivity index. When looking at the components, Optibond Solo contains silica in different forms. They have been the subject of studies regarding possible cytotoxic effects. As studies have shown silica might be responsible for cellular stress responses which might cause cell death or promote transformation[
74]. Those properties combined with the cytotoxic effects of the other components of Optibond Solo Plus like bis-GMA, might be a possible explanation for the few viable cells and the high reactivity index observed in this study[
75].
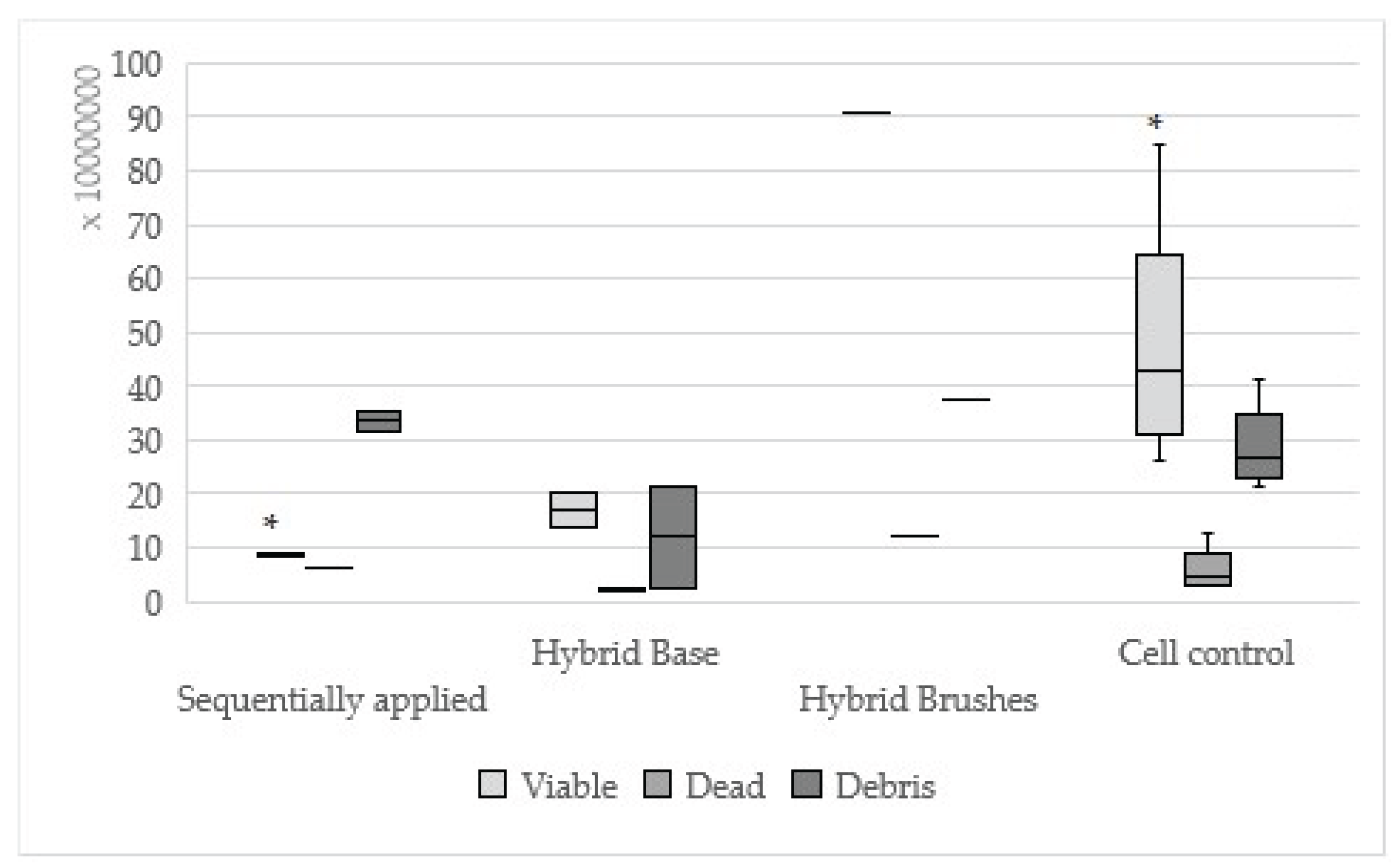
In terms of quantitative evaluation only Hybrid Bond sequentially applied showed statistically significant less viable cells in comparison to Hybrid Brushes single applied. The Hybrid Brushes contain no monomers which might explain the low cytotoxicity. This effect was also mentioned in the first part of our investigation after 24 hours[
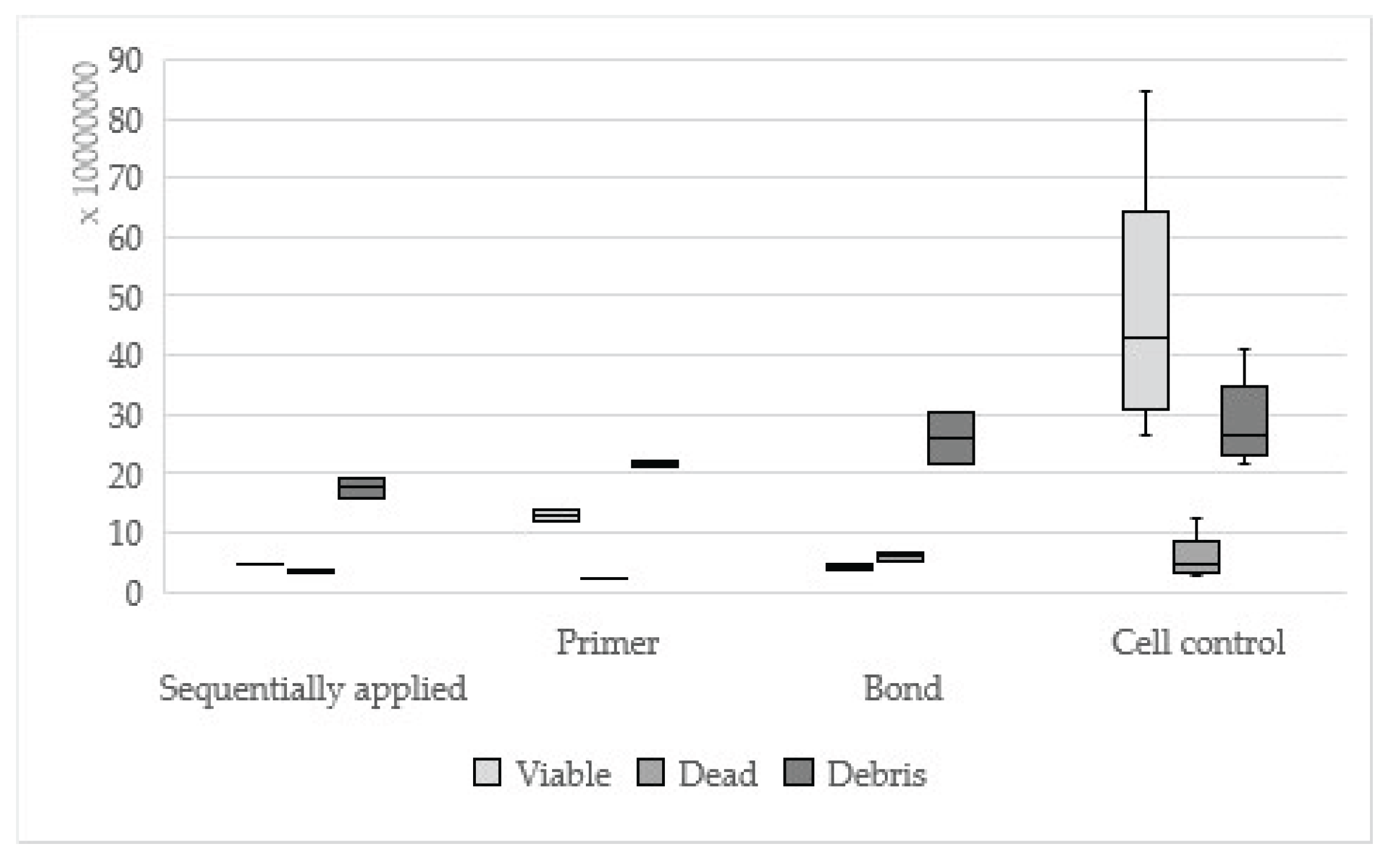
19]. The dentin adhesives AdheSE, Hybrid Bond, One-up Bond F Plus and Syntac showed statistically significant differences between sequentially and single applied adhesive parts, which is in accordance with our first investigation[
19]. Contrary, Clearfil SE Bond showed no statistically significant difference between sequentially and single applied adhesive part in comparison to the observation term of 24 hours. Possibly, the cell regeneration that has already occurred could be a reason for this observation. Recent studies have assumed the same where toxicity of adhesives appeared to decrease after 72 hours[
41].
It should be emphasized again that the experimental conditions of the quantitative method can best be transferred to further investigations and thus the best possible comparison of different examinations is possible. We consider this to be the most reliable method for evaluation as qualitative assessments may vary from person to person.
In future research, the evaluation for these adhesives should be extended up to a long-term trial over 30 days as described in the 10993-5[
20]. In order to further assimilate the cytotoxicity of the individual dentin adhesives, further investigation methods such as MTT or WST-1 assay are planned after the screening. This investigation can also be supplemented by further screening studies with other adhesives to confirm the result of the comparison of the cytotoxicity of self-etch and etch-and-rinse adhesives. Nevertheless, our study does not exactly mimic the environment in the oral cavity which is a major limitation.
In vivo studies consider reactions of the surrounding tissue and cells, as well as the composition and temperature of human saliva which is highly complicated to adapt in
in vitro studies. However, cell culture models have gained value as an alternative to animal experiments which are ethically controversial, costly and time-consuming. Those
in vitro models are already widely accepted in restorative dentistry for testing the biocompatibility of materials and therefore further replacing the need for animal experiments[
76,
77]. It can definitely be stated that far more detailed studies regarding the mechanisms of toxicity, e.g. with immunohistological staining or flow cytometry, are necessary in order to develop a comprehensive understanding and be able to develop more biocompatible materials in the future[
4,
62].
Figure 1.
Results of Hybrid Bond sequentially applied, single applied (Hybrid Base, Hybrid Brushes) and cell control; (Sequentially applied vs. Hybrid Brushes *viable: p=0.01).
Figure 1.
Results of Hybrid Bond sequentially applied, single applied (Hybrid Base, Hybrid Brushes) and cell control; (Sequentially applied vs. Hybrid Brushes *viable: p=0.01).
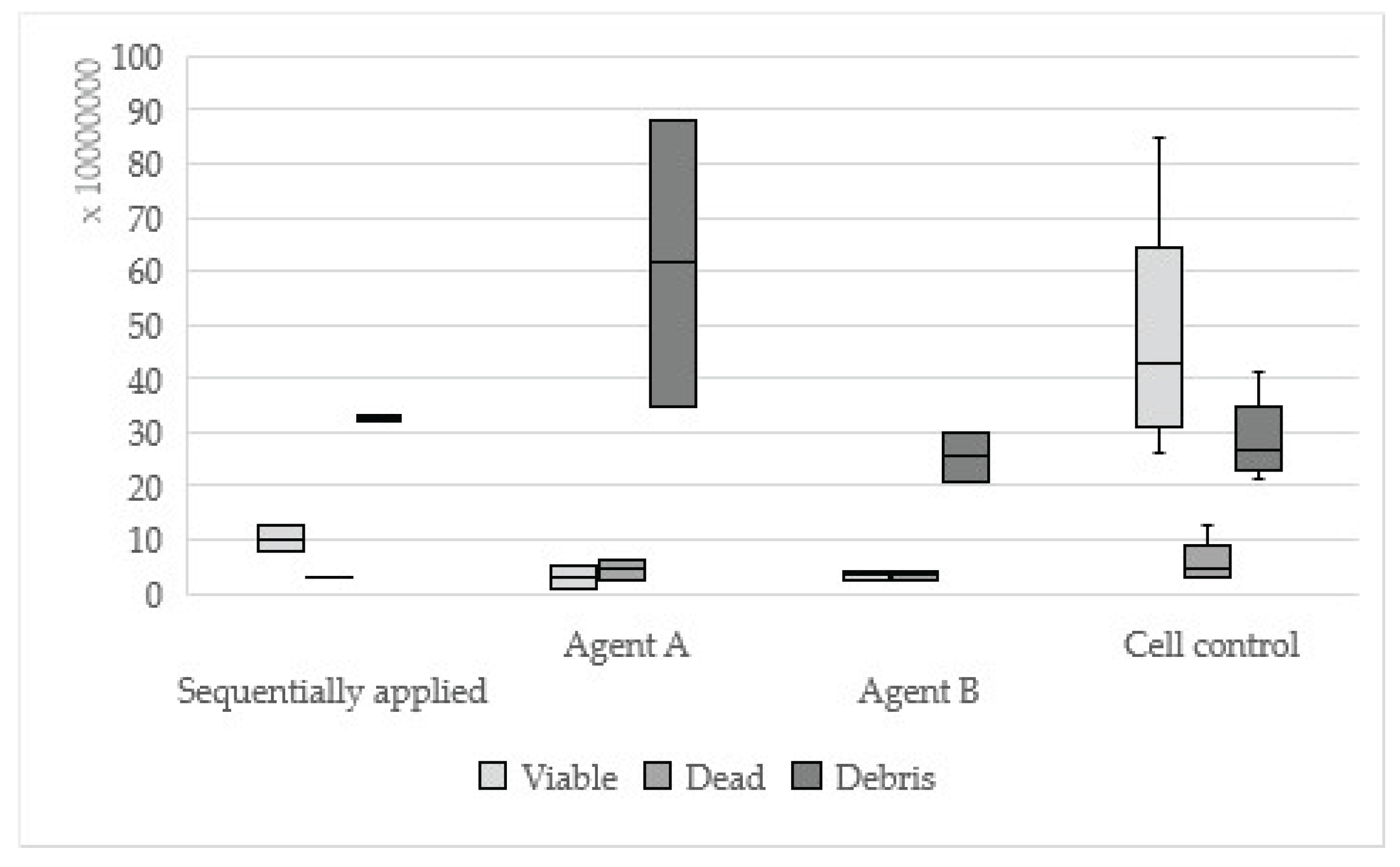
Figure 2.
Results of One-up Bond F Plus sequentially applied, single applied (Agent A, Agent B) and cell control.
Figure 2.
Results of One-up Bond F Plus sequentially applied, single applied (Agent A, Agent B) and cell control.
Figure 3.
Results of AdheSE sequentially applied, single applied (Primer, Bond) and cell control.
Figure 3.
Results of AdheSE sequentially applied, single applied (Primer, Bond) and cell control.
Figure 4.
Results of Clearfil SE Bond sequentially applied, single applied (Primer, Bond) and cell control
Figure 4.
Results of Clearfil SE Bond sequentially applied, single applied (Primer, Bond) and cell control
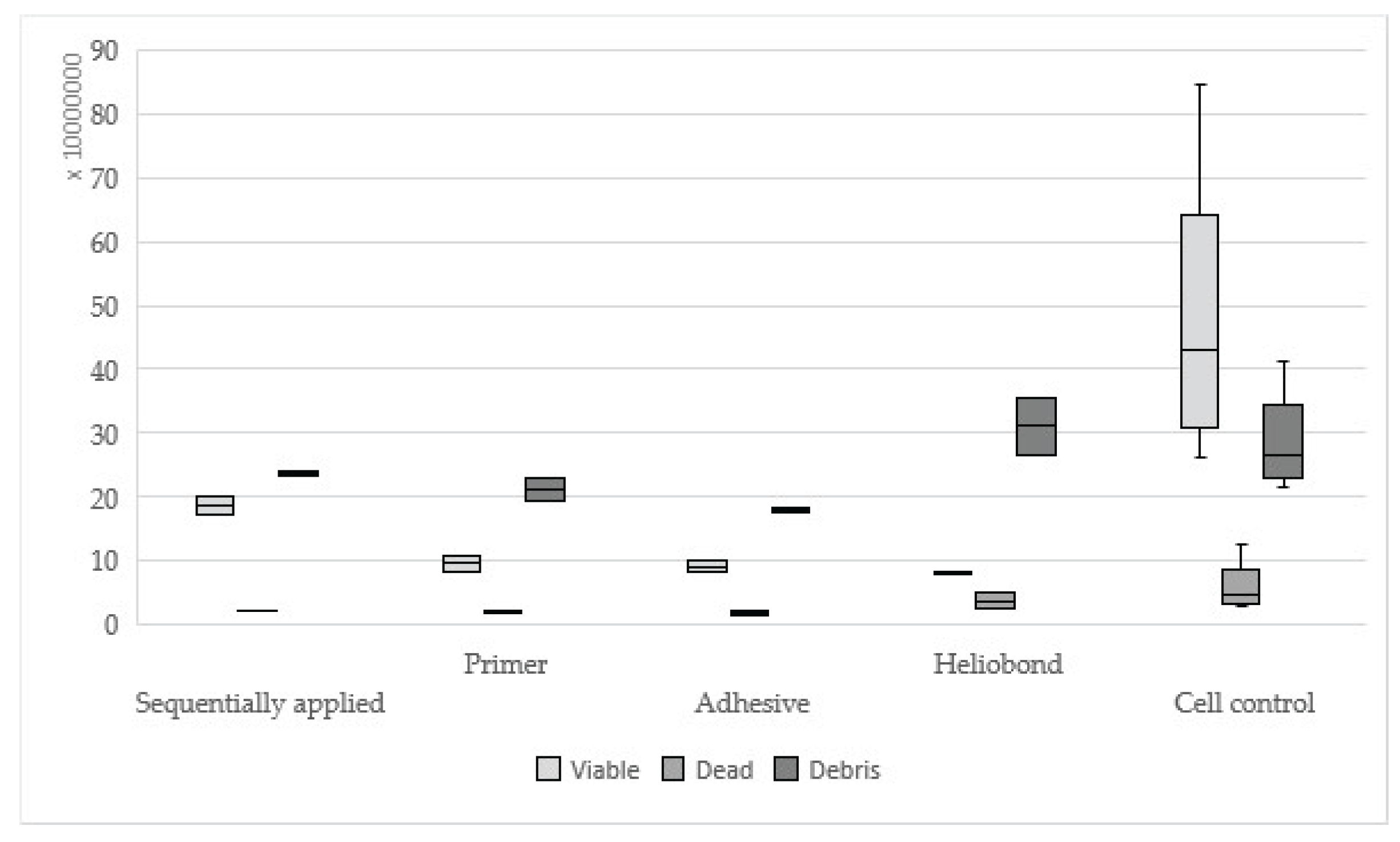
Figure 5.
Results of Syntac sequentially applied, single applied (Primer, Adhesives, Heliobond) and cell control
Figure 5.
Results of Syntac sequentially applied, single applied (Primer, Adhesives, Heliobond) and cell control
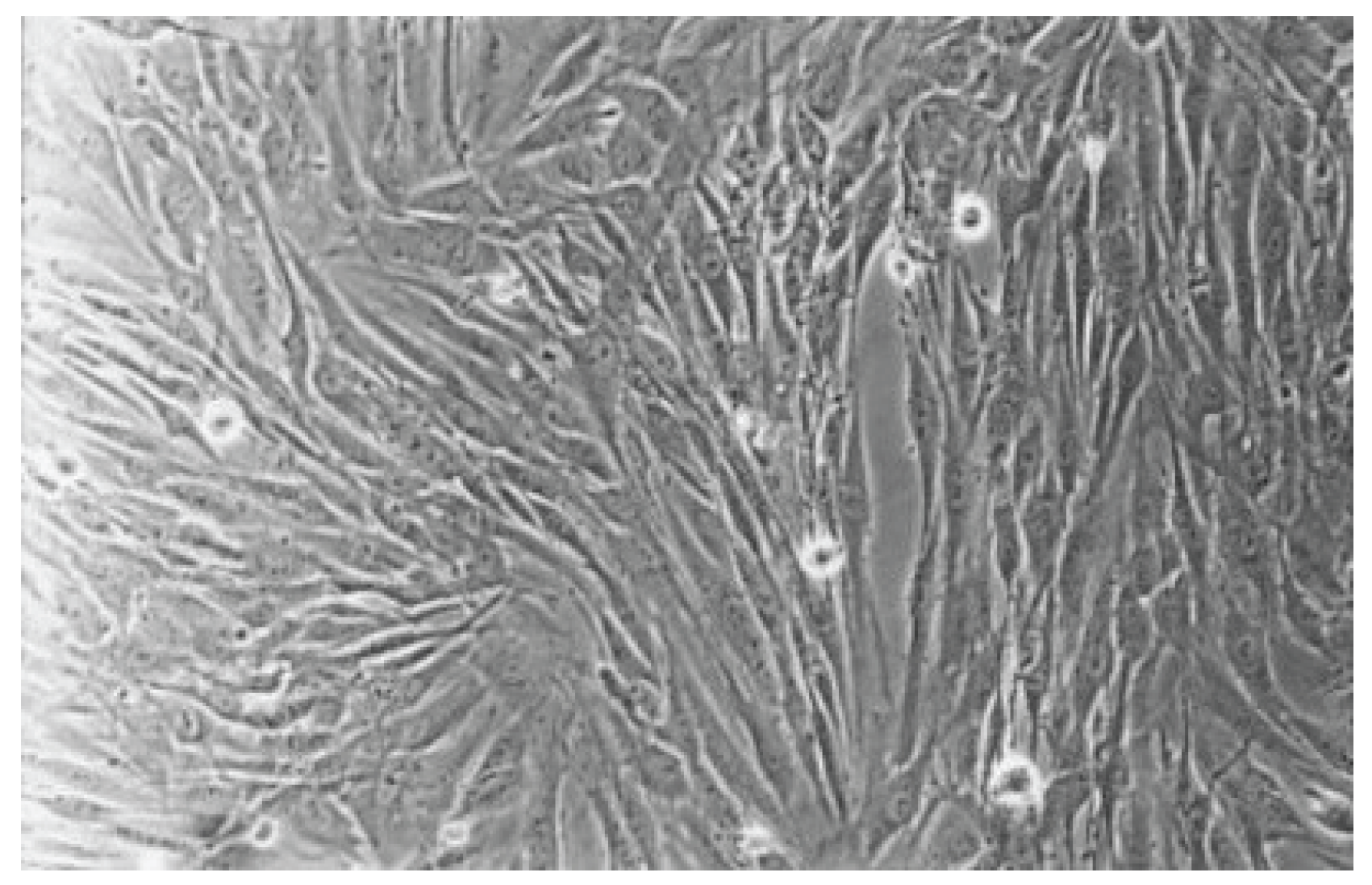
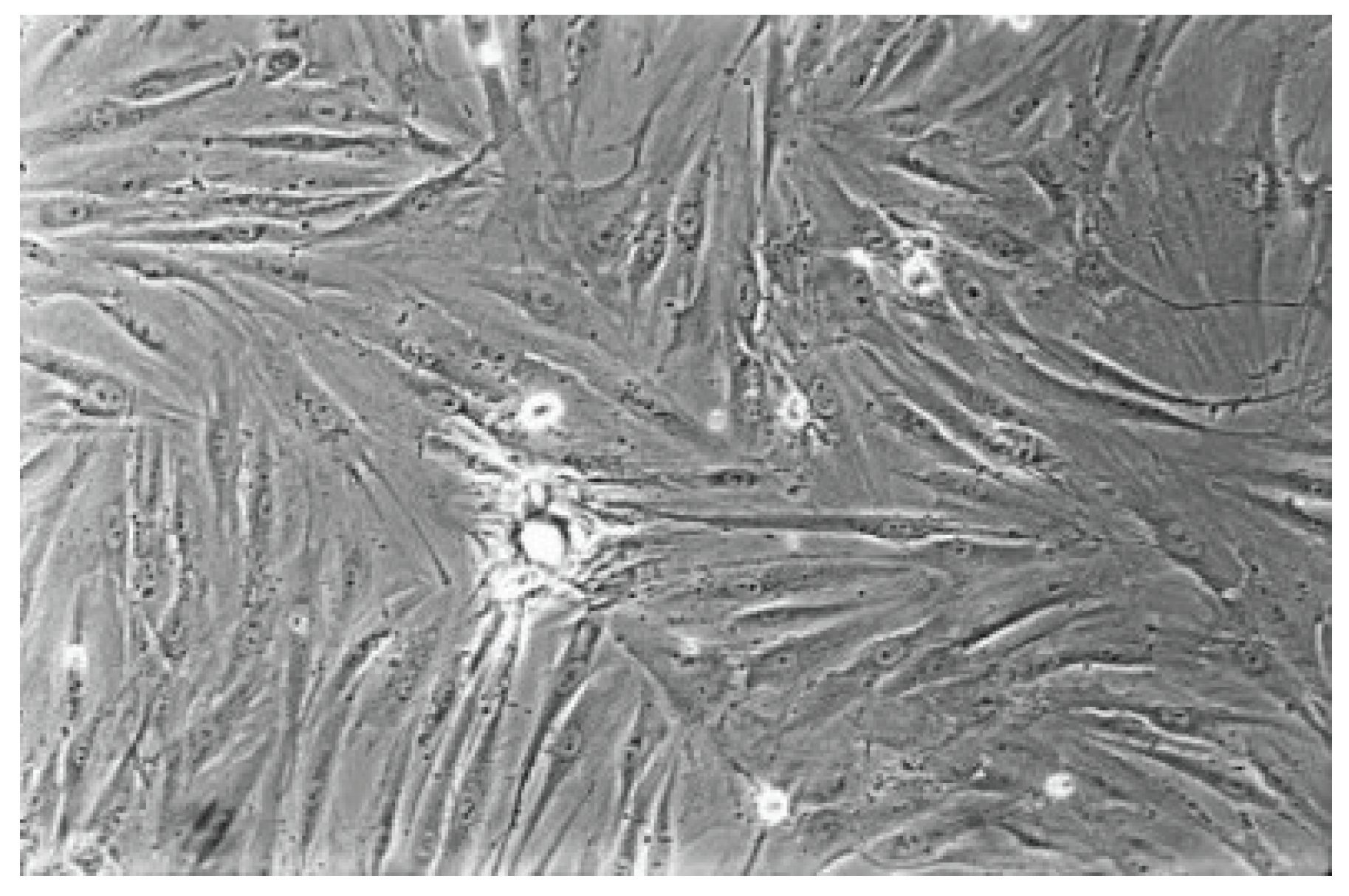
Figure 6.
The cell control (No. 7) presents a regular dense lawn of fibroblasts with characteristic long cells after 48 hours (100-fold magnification).
Figure 6.
The cell control (No. 7) presents a regular dense lawn of fibroblasts with characteristic long cells after 48 hours (100-fold magnification).
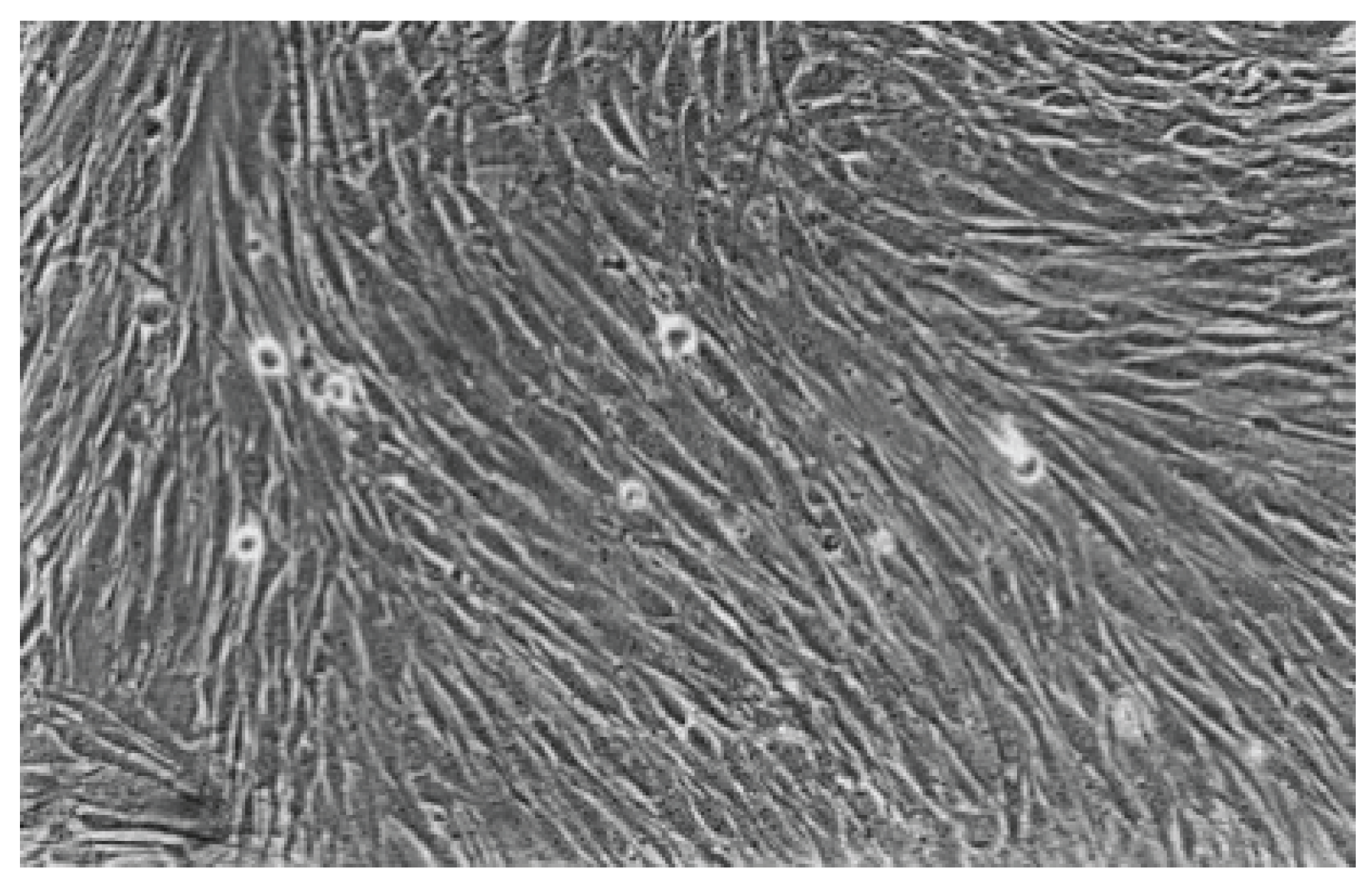
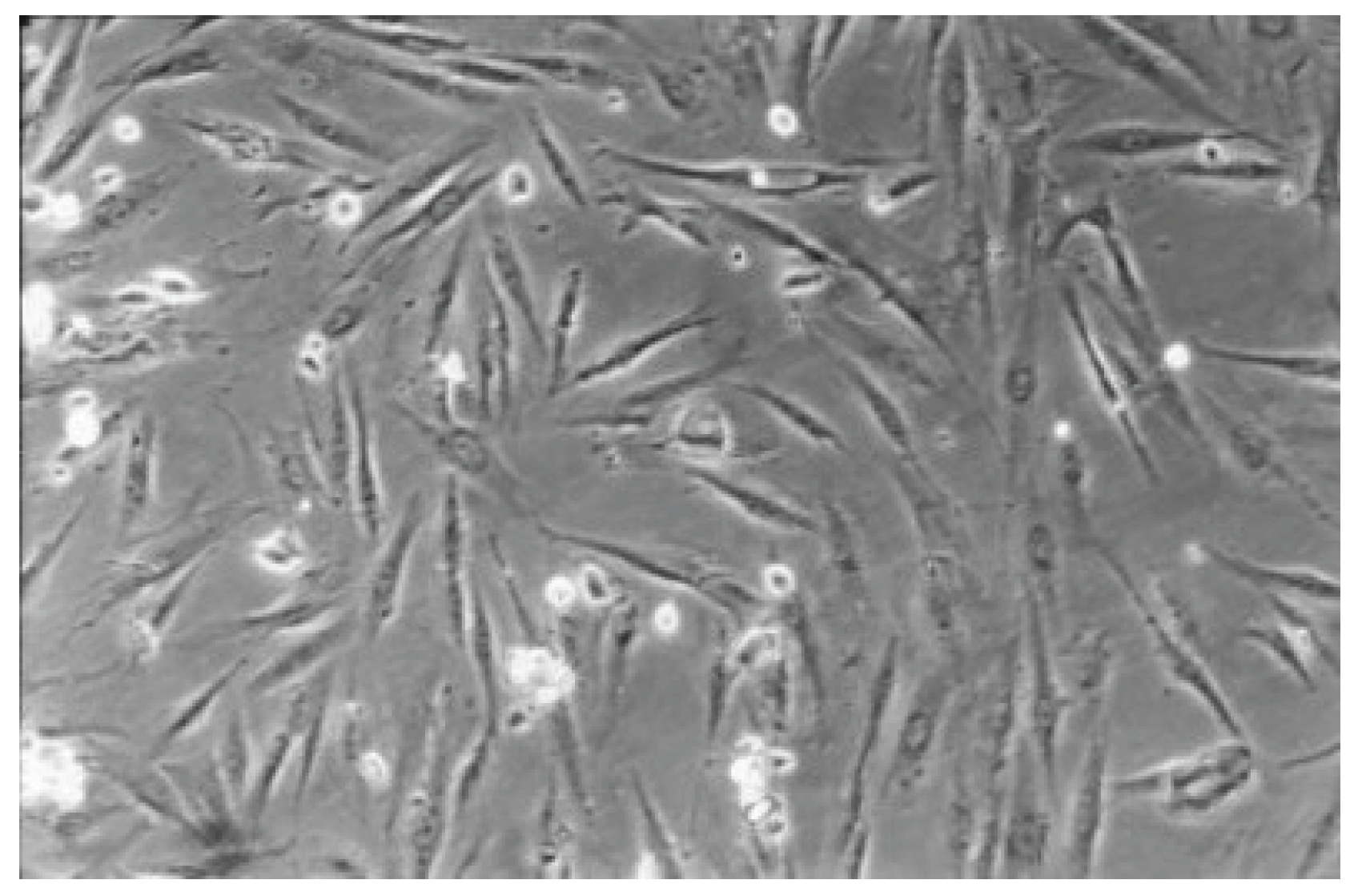
Figure 7.
With Hybrid Brushes (No. 1), the fibroblast lawn appears very dense, similar to the cell control after 48 hours. Normal cells are predominating (100-fold magnification).
Figure 7.
With Hybrid Brushes (No. 1), the fibroblast lawn appears very dense, similar to the cell control after 48 hours. Normal cells are predominating (100-fold magnification).
Figure 8.
Cell culture with One-up Bond F Plus (No. 2) after 48 hours, shows many viable and some rounded cells. The fibroblast lawn is less compared to the cell control (100-fold magnification).
Figure 8.
Cell culture with One-up Bond F Plus (No. 2) after 48 hours, shows many viable and some rounded cells. The fibroblast lawn is less compared to the cell control (100-fold magnification).
Figure 9.
With AdheSE (No. 3), the cell culture appears less dense than the cell control. Some vital cells are recognizable (100-fold magnification).
Figure 9.
With AdheSE (No. 3), the cell culture appears less dense than the cell control. Some vital cells are recognizable (100-fold magnification).
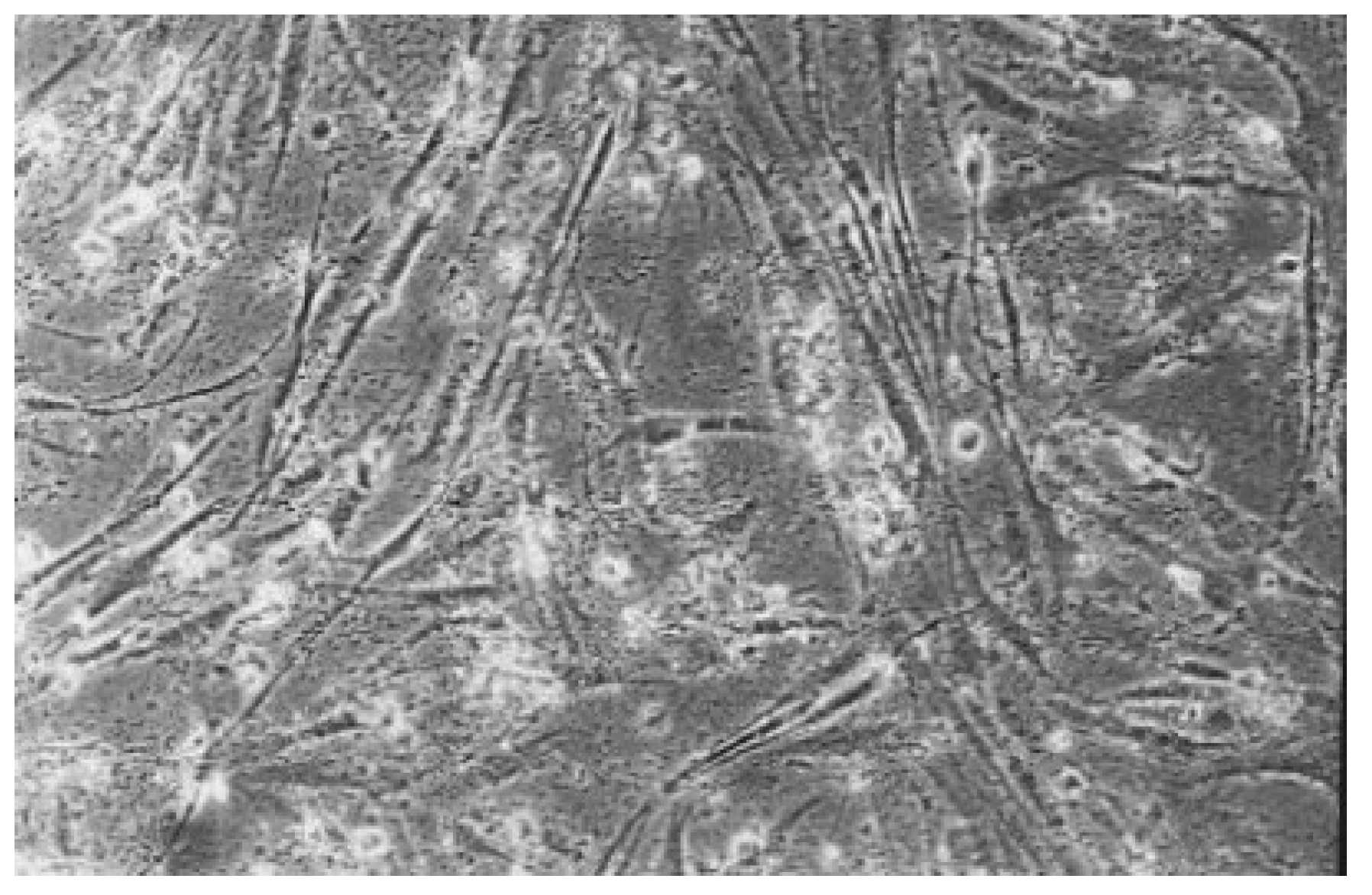
Figure 10.
The cell culture exposed to dentin adhesive Clearfil SE Bond (No. 4) is less dense than the cell control with rounded and dead cells (100-fold magnification).
Figure 10.
The cell culture exposed to dentin adhesive Clearfil SE Bond (No. 4) is less dense than the cell control with rounded and dead cells (100-fold magnification).
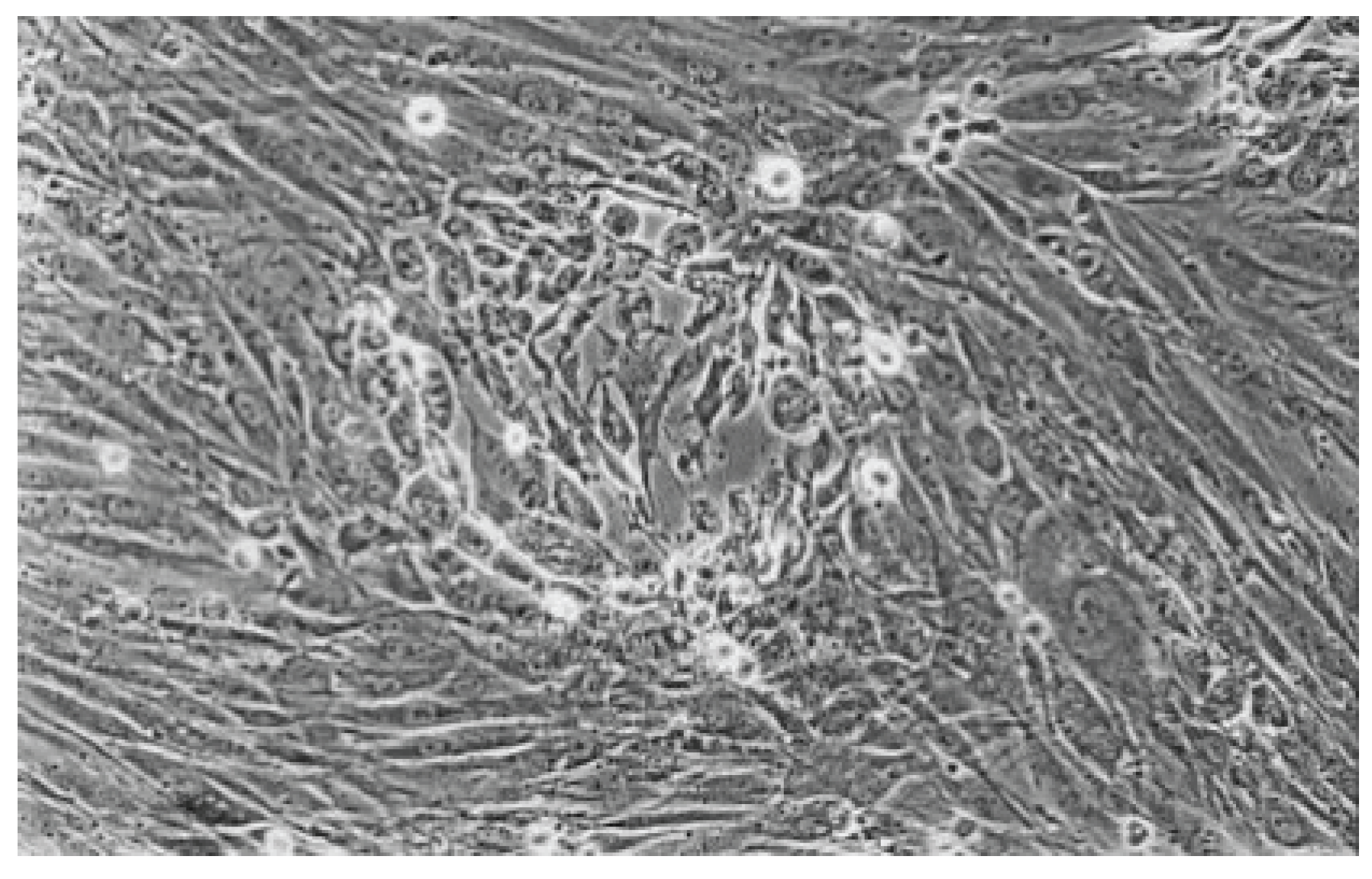
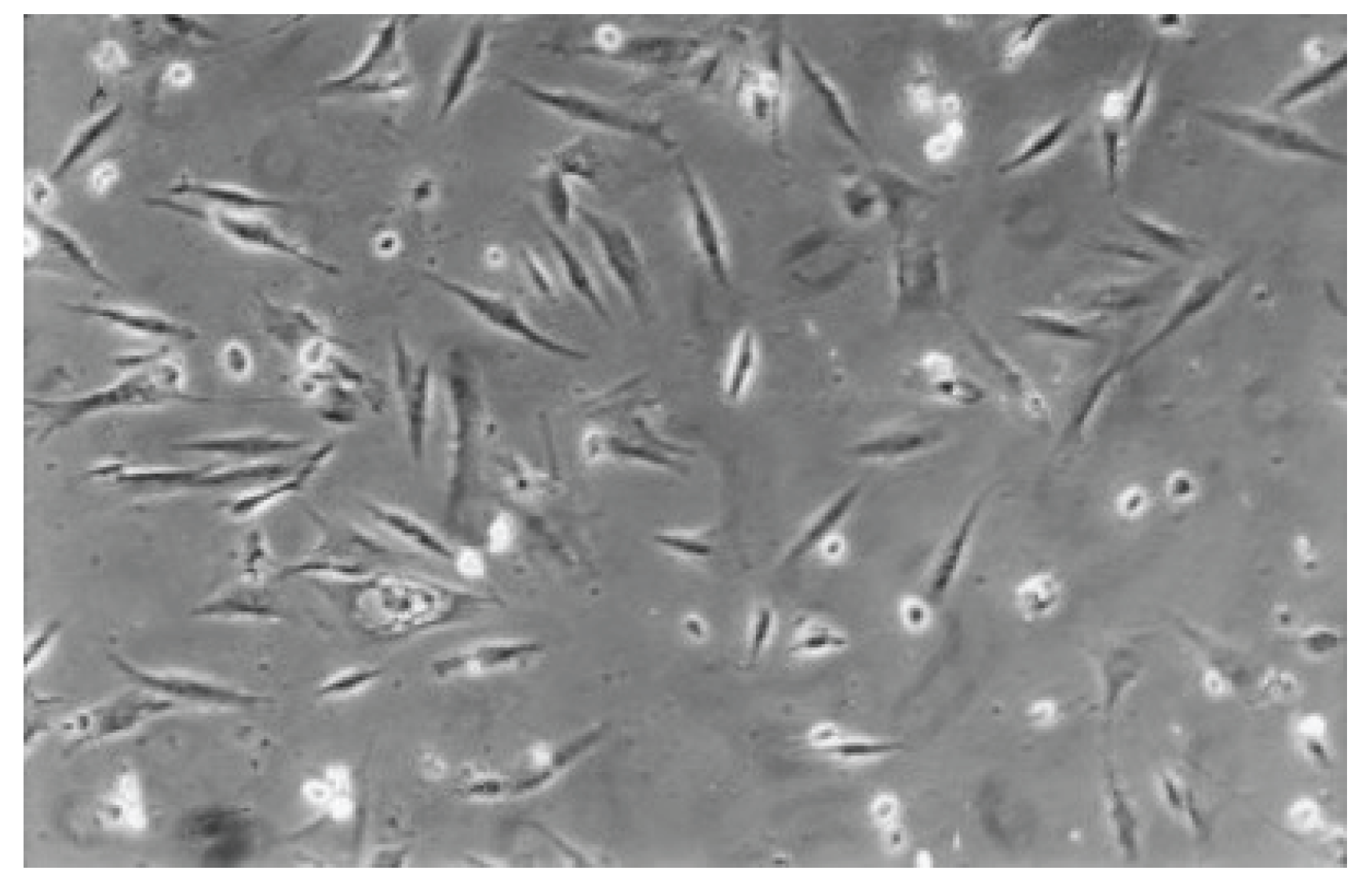
Figure 11.
Rounded and viable fibroblasts can be found in the cell culture of Syntac (No. 5). The fibroblast lawn is much less dense than the cell control (100-fold magnification).
Figure 11.
Rounded and viable fibroblasts can be found in the cell culture of Syntac (No. 5). The fibroblast lawn is much less dense than the cell control (100-fold magnification).
Figure 12.
Few fibroblasts are observed under the influence of Optibond Solo Plus (No. 6) and seem to be in the process of dying with some viable cells. The cell culture is much less dense than the cell control (100-fold magnification).
Figure 12.
Few fibroblasts are observed under the influence of Optibond Solo Plus (No. 6) and seem to be in the process of dying with some viable cells. The cell culture is much less dense than the cell control (100-fold magnification).
Table 1.
Mean, Standard deviation (sd), Minimum (Min.), Maximum (Max.) and Median Values of the six dentin adhesives after 48 hours (Viable cells).
Table 1.
Mean, Standard deviation (sd), Minimum (Min.), Maximum (Max.) and Median Values of the six dentin adhesives after 48 hours (Viable cells).
| No. |
Dental adhesive (1-6) |
Mean |
sd |
Min. |
Max. |
Median |
Significance in Rel. To No.* |
| 1 |
Hybrid Bond |
284.154.000,00 |
350.824.759,65 |
86.440.000,00 |
906.100.000,00 |
136.100.000,00 |
- |
| 2 |
One-up Bon F Plus |
55.790.000,00 |
41.645.983,24 |
10.480.000,00 |
126.800.000,00 |
46.205.000,00 |
7 |
| 3 |
AdheSE |
71.540.000,00 |
43.545.782,34 |
34.640.000,00 |
136.900.000,00 |
47.375.000,00 |
7 |
| 4 |
Clearfil SE Bond |
79.585.000,00 |
38.042.208,53 |
38.280.000,00 |
134.200.000,00 |
82.885.000,00 |
7 |
| 5 |
Syntac |
113.225.000,00 |
46.508.508,59 |
77.690.000,00 |
200.200.000,00 |
91.895.000,00 |
- |
| 6 |
Optibond Solo Plus |
65.305.000,00 |
14.941.166,29 |
54.740.000,00 |
75.870.000,00 |
65.305.000,00 |
7 |
| 7 |
Cell Control |
467.340.000,00 |
223.378.877,69 |
263.300.000,00 |
846.300.000,00 |
430.600.000,00 |
2,3,4,6 |
Table 2.
Qualitative evaluation Hybrid Bond (No. 1).
Table 2.
Qualitative evaluation Hybrid Bond (No. 1).
| Concentration |
Components Sequentially applied |
Components Single applied |
| |
|
Hybrid Base |
Hybrid Brushes |
| I |
0,1-2,0 µl: few viable fibroblasts, no mitosis, material strongly distributed on petri dish bottom, up to 100% cell death |
0,04-2,0 µl: rounded fibroblasts, mitosis, material strongly distributed on petri dish bottom, less dense fibroblast lawn, up to 100% cell death |
0,1-1,0 µl: like the cell control |
| II |
2,5-5,0 µl: material spreads very strongly on Petri dish bottom, 100% cell death |
3,0-5,0 µl: material spreads very strongly on Petri dish bottom, 100% cell death |
1,1-3,4 µl: like concentration I |
Table 3.
Qualitative evaluation One up Bond F Plus (No. 2)
Table 3.
Qualitative evaluation One up Bond F Plus (No. 2)
| Concentration |
Components Sequentially applied |
Components Single applied |
| |
|
Agent A |
Agent B |
| I |
1,0-6,0 µl: few rounded fibroblasts, few mitosis, material strongly distributed on petri dish bottom, dense fibroblast lawn |
1,0-4,0 µl: rounded fibroblasts, up to 100% cell dead |
1,0-5,0 µl: rounded fibroblasts, partially still viable appearing cells, fibroblasts growing on material, not so dense fibroblast lawn |
| II |
7,0-14,0 µl: rounded fibroblasts, fibroblast lawn less dense than the cell control |
5,0-8,0 µl: rounded fibroblasts, less mitosis, few viable cells, up to 100% cell dead |
6,0-10,0 µl: few fibroblasts appearing viable, fibroblasts growing on material, material strongly distributed on petri dish bottom |
Table 4.
Qualitative evaluation AdheSE (No. 3)
Table 4.
Qualitative evaluation AdheSE (No. 3)
| Concentration |
Components Sequentially applied |
Components Single applied |
| |
|
AdheSE Primer |
AdheSE Bond |
| I |
2,5-6,0 µl: hardly any mitosis, vital fibroblasts, slightly less dense fibroblast lawn than cell control |
5,0-9,0 µl: vital fibroblasts, mitosis, slightly less dense fibroblast lawn than cell control |
3,0-4,0 µl: many rounded cells, viable fibroblasts at the border of the petri dish, not so dense fibroblast lawn |
| II |
7,0-12,0 µl: like concentration I |
10,0-14,0 µl: like concentration I |
5,0-7,0 µl: 100% cell dead |
Table 5.
Qualitative evaluation Clearfil SE Bond (No. 4)
Table 5.
Qualitative evaluation Clearfil SE Bond (No. 4)
| Concentration |
Components Sequentially applied |
Components Single applied |
| |
Clearfil SE Bond |
Clearfil SE Bond Primer |
Clearfil SE Bond Bond |
| I |
2,0-3,0 µl: many dead fibroblasts, some viable appearing atypical cells, 75-95% cell dead |
4,0-5,0 µl: few viable fibroblasts, material spreads strongly on Petri dish bottom, up to 100% cell dead |
3,0-4,0 µl: vital and many dead cells, fibroblast lawn less dense than the cell control |
| II |
4,0-5,0 µl: 90-100% cell dead |
6,0-8,0 µl: 100% cell dead |
5,0-6,0 µl: 100% cell dead |
Table 6.
Qualitative evaluation Syntac (No. 5)
Table 6.
Qualitative evaluation Syntac (No. 5)
| Concentration |
Components Sequentially applied |
Components Single applied |
| |
|
Syntac Primer |
Syntac Adhesive |
Syntac Heliobond |
| I |
0,1-1,0 µl: few viable fibroblasts, rounded cells, material spreads very strongly on Petri dish bottom, 99-100% cell dead |
1,0-6,0 µl: many viable cells, dead fibroblasts, not so dense fibroblast lawn |
0,2-2,0 µl: few viable cells, rounded fibroblasts, mitoses, not so dense fibroblast lawn |
1.0-5.0 µl: rounded and viable cells, many dead cells, not so dense fibroblast lawn |
| II |
2,0-2,5 µl: some small viable fibroblasts, most cells dead, material spreads very strongly on Petri dish bottom, 90-100% cell dead |
7,0-12,0 µl: many rounded cells, less dense fibroblast lawn than the cell control |
3,0-5,0 µl: 100% cell dead |
6.0-10.0 µl: many rounded cells, up to 100% cell dead |
Table 7.
Qualitative evaluation Optibond Solo Plus (No. 6)
Table 7.
Qualitative evaluation Optibond Solo Plus (No. 6)
| Concentration |
Optibond Solo Plus |
| I |
1,0-4,0 µl: many viable cells, rounded fibroblasts, vacuolated cells, material spreads strongly on petri dish bottom, first fibroblasts grow on material, less dense fibroblast lawn than cell control |
| II |
5,0-8,0 µl: material spreads strongly on petri dish bottom, 100% cell dead |