1. Introduction
Nutrition plays a fundamental role in sports performance, as it influences energy production, muscle recovery and injury prevention (1). Within the essential micronutrients for athletes, vitamin B12 (VB12) has gained special interest due to its role in DNA synthesis, red blood cell formation and neuromuscular function (2). Deficiency of this vitamin can compromise oxygen transport, increase fatigue, and reduce recovery capacity, factors that directly affect athletic performance (3).
VB12, also known as cobalamin, is a water-soluble vitamin found in foods of animal origin, such as meats, eggs, dairy products and seafood (4). Its absorption in the body depends on the presence of intrinsic factor, a glycoprotein produced in the stomach, which means that its bioavailability can be affected by various medical conditions or dietary habits (5), although it can also be absorbed via passive diffusion across the gastrointestinal mucosa, including the oral cavity. In this context, vegetarian and vegan athletes are at an increased risk of deficiency, which could have a negative impact on their performance (6). In fact, studies have shown that up to 62% of vegans and up to 86% of vegetarians may have suboptimal VB12 levels, depending on supplementation practices (7). In addition, individuals with VB12 deficiency increase with age, gastrointestinal disorders or different medical treatments, like metformin, H2 antagonists or chronic intake of proton pump inhibitors (8-10). Older adults and individuals with malabsorption syndromes may require higher doses or alternative routes of administration to maintain adequate levels (5).
Vitamin B12 exists in several forms, including cyanocobalamin, methylcobalamin (MeB12), hydroxocobalamin, and adenosylcobalamin. (11). Cyanocobalamin is a synthetic form of VB12, commonly used in supplements and fortified foods due to its stability and lower cost. Among the natural forms, adenosylcobalamin and MeB12 are biologically active, while hydroxocobalamin is a natural form that can be converted into either active form. Adenosylcobalamin functions as a cofactor of methylmalonyl Co-A mutase in mitochondria, whereas MeB12 serves as a cofactor for methionine synthase in the cytosol. Although cyanocobalamin and hydroxocobalamin are not active coenzymes themselves, they can be converted into the biologically active forms within the body. The human body, however, does not directly use VB12; instead, it has to be converted into its active coenzyme forms, MeB12 or adenosylcobalamin (11). Compared to cyanocobalamin, MeB12 has a methyl group in place of the cyanide ligand (12). It is a cofactor necessary for the methylation cycle, which methylates proteins or DNA, to produce methionine from homocysteine (13, 14). This biochemical pathway is also linked to the regulation of neurotransmitter synthesis and mood, which may be relevant for psychological resilience during competition (15). MeB12 has been reported to exhibit the best absorption rate by neuronal subcellular organelles when compared to other forms. Studies using radiolabeled MeB12 in both rats and humans demonstrated that its absorption is comparable to that of cyanocobalamin; however, urinary excretion is about one-third that of cyanocobalamin, and liver uptake was greater with MeB12 (16). Consequently, MeB12 may offer superior therapies for neurological conditions by means of efficient systemic or local administration (11, 17).
The effects of VB12 on athletic performance have been extensively studied. It has been found that its deficit can lead to megaloblastic anemia, fatigue, muscle weakness, and impaired cognitive function, which together may decrease endurance and responsiveness to physical exertion (18). Furthermore, inadequate VB12 levels may compromise mitochondrial energy production, a critical factor for both aerobic and anaerobic performance (19). Moreover, subclinical VB12 deficiency in certain population groups may go unrecognized and generate negative effects in the long term (2).
Despite the importance of VB12 in sports physiology, currently, the research conducted to evaluate its influence on the sports performance of athletes is very limited and with little scientific evidence (20-22). Furthermore, these studies only evaluated the effect of one form of VB12 such as hydroxocobalamin, with limited data evaluating the active form MeB12 on exercise performance. Therefore, as there is limited evidence on the efficacy of VB12 supplementation on physical exercise performance, it is necessary to continue investigating this relationship. The main objective of this exploratory study was to investigate the effect of short-term (3 days) MeB12 intake on anaerobic performance and cognitive variables in amateur cyclists. Our hypothesis is that acute MeB12 intake may improve markers of physical and cognitive performance.
2. Methodology
2.1. Study Design
A randomized, triple-blind, placebo-controlled crossover and clinical trial was conducted. Participants ingested MeB12 (1 mg/day) or placebo formulation for 3 consecutive days: days −2 and −1, and a final dose ~5 h before testing on day 0. A washout period of 7 days was carried out between treatments. The product administered, whether active or placebo, was a proprietary formulation prepared at Farmacia Daya Nueva C.B. (Alicante, Spain), being matched in appearance and composition. The active ingredient methylcobalamin (MecobalActive®) was manufactured by HTBA (Murcia, Spain). The study was submitted for evaluation and approved by the Ethics Committee of the Catholic University of Murcia (UCAM) on May 31 of 2024 (CE052416). The trial was conducted in accordance with the Declaration of Helsinki for human research (23). All eligible participants provided informed consent before participating in the study. This study was registered in ClinicalTrials.gov with ID: NCT06639789.
2.2. Participants
The sample consisted of a total of 18 well-trained amateur cyclists (
Table 1), whose enrolment criteria were: age between 18 and 45 years, training 4-6 days per week, no pathology incompatible with the evaluations to be performed, not presenting any condition that contraindicates vitamin B12 supplementation, not competing at a professional level and not taking any supplementation during the study period.
Table 1.
General Characteristics of participants.
Table 1.
General Characteristics of participants.
| Age |
Height |
BM |
% FM |
FM |
% MM |
MM |
32.2
(9.49) |
167.1
(5.37) |
67.3
(6.73) |
11.0
(2.78) |
8.1
(2.75) |
47.7
(1.21) |
34.2
(3.01) |
| SD = Standard deviation; BM = body mass, FM = fat mass, MM = muscle mass. |
1.3. Procedures
The participants visited the laboratory 3 times (1 time per week). Visit 1: the informed consent to participate in the study was signed, a blood sample for a general health analysis, anthropometry, diet questionnaire and familiarization with cognitive tests were completed. Visits 2 & 3: In both visits the study protocol was executed in the randomly assigned condition order (product-placebo / placebo-product). The assessment consisted of blood analysis, cognitive test, fatigue protocol (5 x Wingate 30"/3' of rest period) and repetition of cognitive test and blood analysis. All subjects who participated in our study performed tests at the same time to avoid interference of circadian rhythms in the results.
1.4. Assessments
1.4.1. Blood Analysis
A total of 5 blood samples were taken for subsequent analysis, being a general health analysis (general biochemistry and hemogram) at visit 1 and four VB12 blood analyses at pre- and post-fatigue times during visits 2 and 3 (2 samples per day).
A duly accredited nursing professional performed the venous blood draw, collecting two tubes: a 3 mL tube with EDTA anticoagulant for the hemogram, and a 3.5 mL tube made of polyethylene terephthalate (PET) for general health studies. Red blood cell analysis was performed using a Cell-Dyn 3700 automated analyzer (Abbott Diagnostics, Chicago, IL, USA), under a quality control system that included internal controls (Cell-Dyn 22) and external controls provided by the Program of Excellence for Medical Laboratories (PEML). This instrument estimated the levels of erythrocytes, hemoglobin, hematocrit and hematometric parameters.
1.4.2. Anthropometry
Anthropometric assessments were carried out by a researcher certified at ISAK Level 1. For measuring height and body weight, a clinical-grade digital scale with an integrated stadiometer (SECA 780; Vogel & Halke GmbH & Co., Hamburg, Germany) was used. Skinfold measurements were obtained using Holtain Skinfold Calipers (Holtain, Ltd., Crymych, Pembrokeshire, UK), following the standardized procedures outlined by the International Society for the Advancement of Kinanthropometry (24). Body fat percentage was estimated using the Faulkner equation (25), while muscle mass percentage was derived from a modified version of the Matiegka equation (26). Additionally, the sum of eight specific skinfold sites (triceps, subscapular, biceps, iliac crest, supraspinale, abdominal, thigh, and calf) was calculated.
1.4.3. Cognitive Response Test
The cognitive response test was carried out with a reaction light system (Lummic®, Pitk Pelotas SL, Navarra, Spain), which has 6 devices placed in series at a distance of 15 cm (from center to center of each device). The participant was seated on a chair 30 cm from the table with the palms of the hands resting on the thighs and he had to react to the lights to turn them off with the dominant hand, returning to the starting position (palm on the thigh) before switching off the next light. The test consisted of completing 3 sets of 40 lights with 1 minute of recovery between sets. The variables recorded were mean reaction time and the total time of each series. During the familiarization session (visit 1), a complete series was performed to familiarize the participant with the operation, protocol and position. Data were automatically recorded by the system application, which communicated to the photocell via Bluetooth with a smartphone.
1.4.4. Repeated Wingate Test
The fatigue protocol consisted of 5 sets of Wingate test (30 sec all out) separated by 3 minutes of recovery. The load was estimated based on the body weight of each participant following the standardized formula for this test (load = 0.075 · body weight). The test was performed seated, and instructions were given to perform the test at maximum from the first second. A warm-up of 5 minutes at 50 W at 75-85 rpm had been performed previously. During each series of the test, the maximum power, maximum relative power, mean power, mean relative power and fatigue index were recorded. The fatigue index was calculated as the percentage decrease in power from the maximum peak to the minimum recorded during each set. During recovery, active pedaling without load was performed for 2 minutes, followed by 30 seconds of stop and 30 seconds of pre-test reactivation (50 W at 75-85 rpm).
1.4.5. Diet Questionnaires
Participants were instructed to consume a standardized breakfast two hours prior to testing, which included 0.41 grams of protein, 1.35 grams of carbohydrates, and 0.53 grams of fat per kilogram of body mass. Diet questionnaires were carried out by means of a personal interview, in which all the dietary intake in 72h was collected. This survey should always include 2 weekdays (M-F) and 1 weekend or holiday. Once the information was collected, it was analyzed with analysis software (Dietowin®) to obtain macronutrients (fats, carbohydrates and proteins) and caloric intake. The data were analyzed to corroborate that there were no substantial modifications in the diet during the study.
1.4.6. Vitamin B12
Serum vitamin B12 concentration was determined by electrochemiluminescence immunoassay (ECLIA), using the Cobas-e automated system (Roche Diagnostics; Sandhofer Strasse 116, D-68305 Mannheim, Germany), specifically the Elecsys Vitamin B12 II test. This assay, based on a competitive principle, employs intrinsic factor as the specific binding protein for vitamin B12. The serum sample (9 μL) was subjected to a first incubation with pretreatment reagents that release bound vitamin B12, followed by a second incubation with ruthenium-labeled intrinsic factor, which forms a complex whose amount is inversely proportional to the concentration of B12 in the sample. Subsequently, in a third incubation, streptavidin-coated microparticles and biotin-labeled vitamin B12 were added, forming a stable complex that was fixed to a solid phase by biotin-streptavidin interaction. Finally, the mixture was transferred to the measuring cell where, under electric current, a chemiluminescent signal proportional to the amount of analyte was produced, detected by a photomultiplier. The results were calculated with a specific calibration curve generated by a two-point calibration and a master curve supplied by the Cobas link system.
1.4.7. Statistical Analysis
The JAMOVI software (Jamovi v 2.6. Jamovi.org, Sydney, Australia) was used for statistical analysis. The homogeneity and normality of the data were tested with the Levene and Shapiro-Wilk tests, respectively. Given the crossover design, comparisons between MeB12 and placebo conditions were performed using paired T-test for the variables of the Wingate test, mental agility and biochemical markers. The effect size (ES) was calculated for the comparison of the variables analyzed. The following thresholds were used for ES: ≤0.20, no effect; 0.21-0.49, small effect; 0.50-0.79, moderate effect; ≥0.80, large effect. The significance level was set at p < 0.05.
3. Results
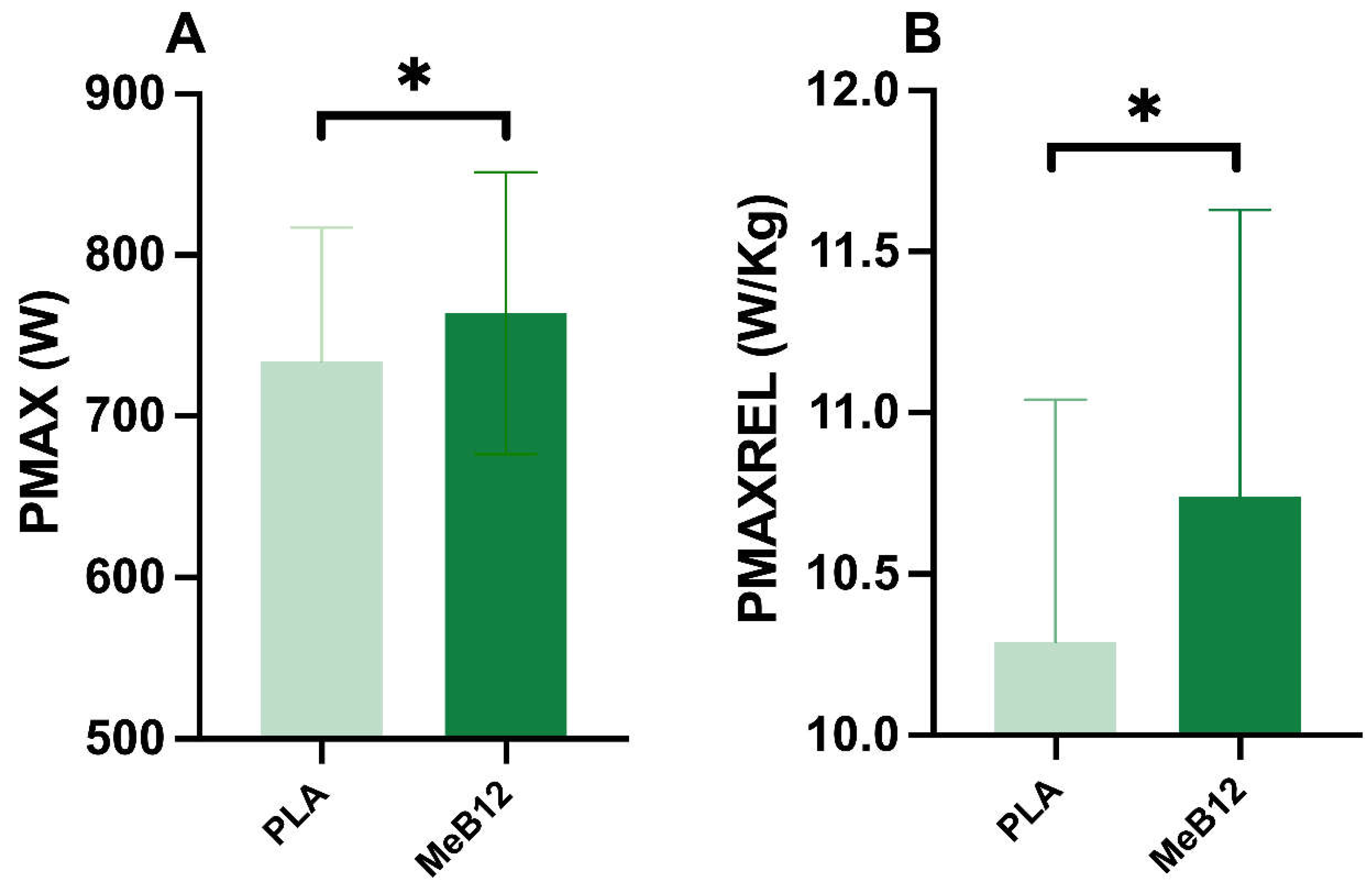
Compared with placebo, the MeB12 group showed greater maximum peak power (PMAX) (764.1±87.38W) than the placebo (PLA) (734.2±83.12W) (p = 0.016; ES = 0.628) after performing the 5 sprints of the Wingate test (
Figure 1A). Furthermore, in maximum relative power (PMAXREL) we found that MeB12 recorded higher PMAXREL values (10.74±0.89W/kg) than PLA (10.29±0.75W/kg) (p = 0.013; ES = 0.655) following the same protocol (
Figure 1B).
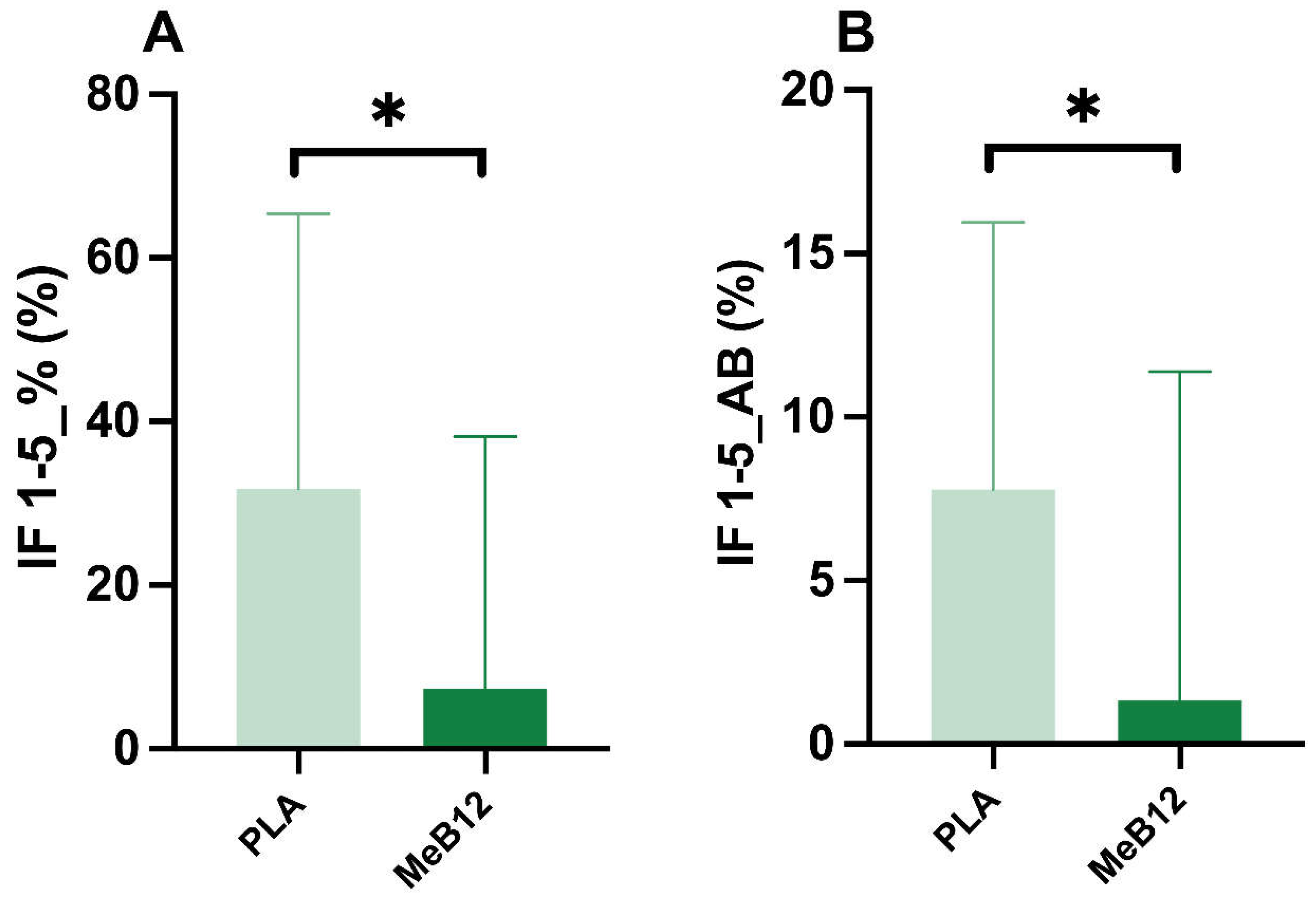
We also analyzed the difference between the 1
st and 5
th sprint (1-5) of the Wingate test for the percentage change and for the absolute value (AB) of the fatigue index (IF%). Results indicated that MeB12 intake decreased in percentage IF% between the 1
st and 5
th effort of the Wingate test %IF 1-5_B12 (7.39±30.76 %) compared to PLA %IF 1-5_PLA (31.80±33.60%) (p = 0.012; ES = 0.664) (
Figure 2A), and similarly in absolute values in %IF 1-5 AB_B12 (1.33±10.05%) and %IF 1-5 AB_PLA (7.78±8.16%) compared to PLA (p = 0.022; ES = 0.594) (
Figure 2B).
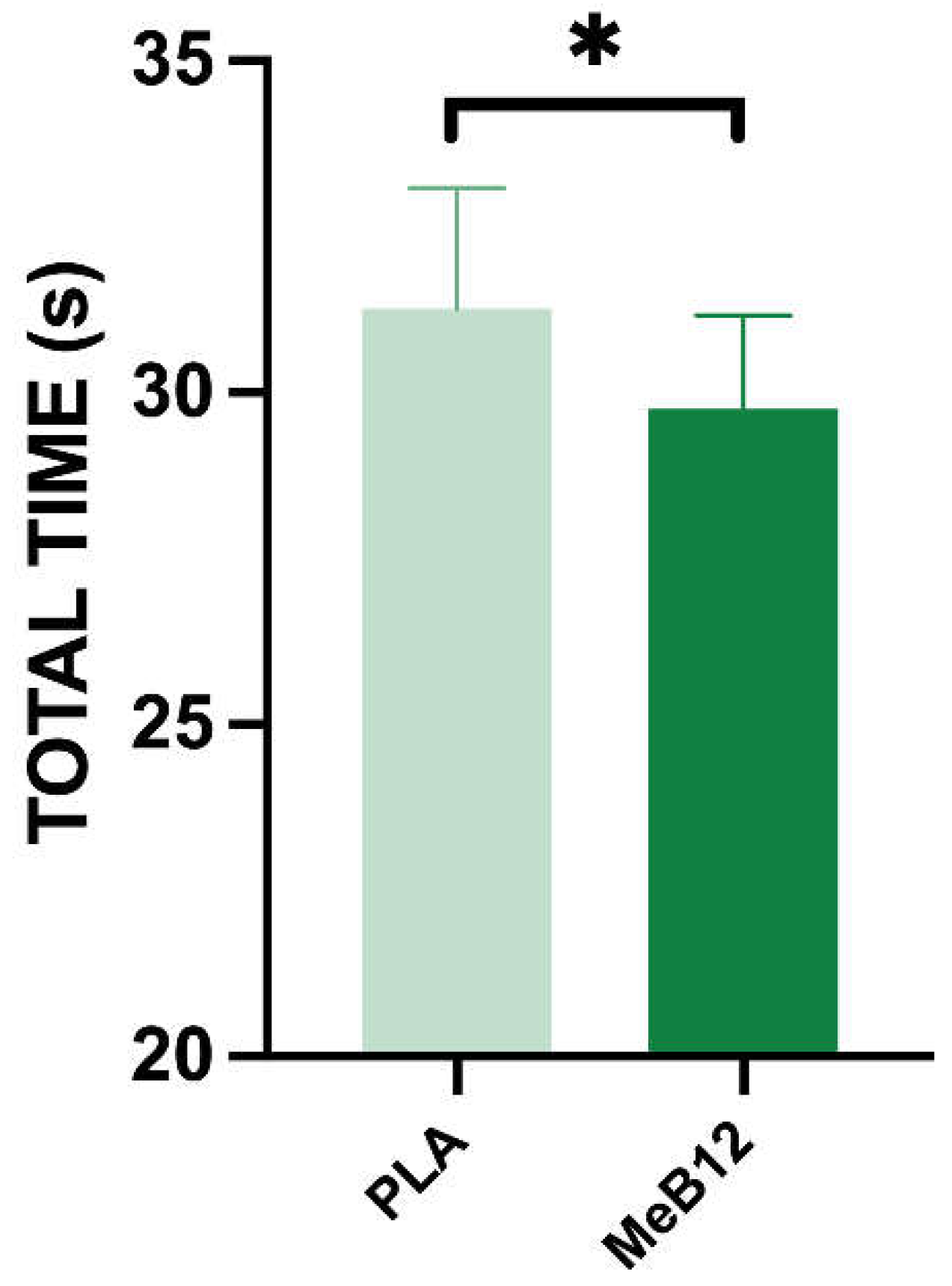
Statistical analysis of the mental agility test data revealed that the total time recorded before the Wingate test was significantly lower in MeB12 (29.75 ± 1.42 s) compared to the PLA (31.27 ± 1.82 s) (p < 0.001; ES = 0.459) (
Figure 3). These findings suggest that a 3-day supplementation with MeB12 can improve cognitive performance, as reflected by faster completion times in the mental agility test under resting conditions, compared to placebo.
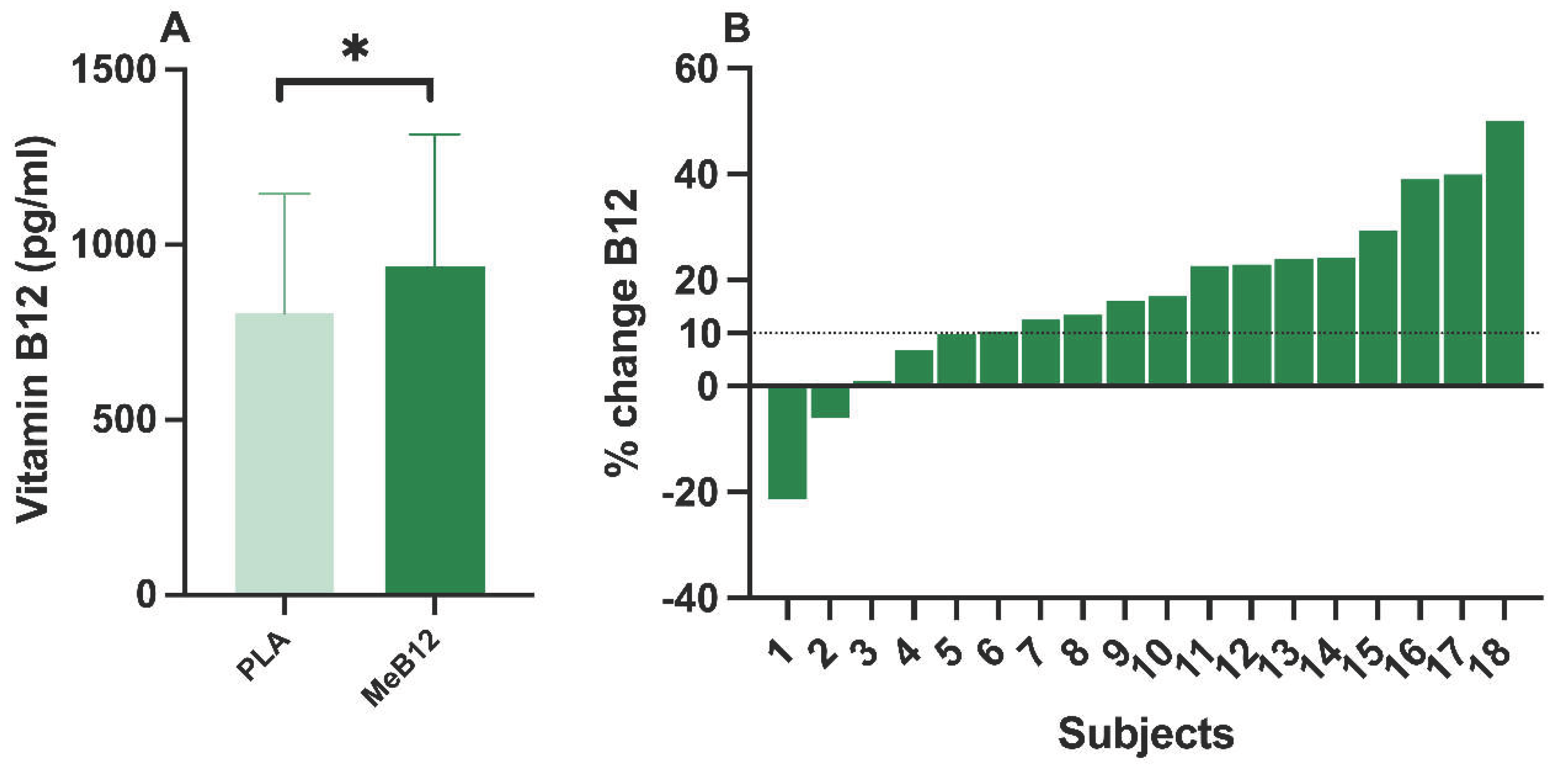
The analysis of VB12 values before the Wingate test showed that the group supplemented with MeB12 increased plasma VB12 levels (924±378 pg/ml) compared to PLA (804±343 pg/ml) (p = 0.008; ES = 0.706). After 3 days of MeB12 supplementation, it increased by 16.8% from its basal levels (PLA), considering the values before performing the Wingate test (
Figure 4A). 13 of the 18 subjects increased plasma VB12 concentrations by more than 10% after 3 days of supplementation compared to their baseline values (PLA), taking into account the values before performing the Wingate test (resting conditions) (
Figure 4B).
4. Discussion
The main findings after completion of this intervention were that acute intake (3 days) of an active form of VB12 (MeB12) resulted in higher PMAX and PMAXREL, and lower %IF (between 1st and 5th sprint) compared to PLA during the Wingate test in amateur cyclists. In addition, we observed a cognitive improvement with MeB12 supplementation, as the total time to complete a mental agility task was decreased compared to PLA. Finally, acute supplementation with this active VB12 form increased plasma VB12 concentrations by 16.8%.
After supplementation with MeB12 amateur cyclists had higher peak power output (PMAX: +30W, +4.1%) and relative peak power (PMAXREL: +0.45W/kg, +4.4%) during the Wingate test compared to PLA. In contrast, Mya-Tu et al. (22) evaluated the effects of administering 1 mg cyanocobalamin injections 3 times per week over 6 weeks in healthy male students on aerobic capacity performance (incremental test to exhaustion). They found that cyanocobalamin did not improve aerobic performance, since no improvements in maximal oxygen consumption (VO2MAX) were found. Differences between cyanocobalamin and MeB12 activity and bioavailability may explain their different effects. Conversely, Lee et al. (27) found an improvement in endurance performance (25% increase in time to exhaustion) and a decrease in fatigue-related biomarkers (decrease in lactate and ammonia) derived from exercise, after 28 days supplementation with a B-vitamin complex (36 mg B1; 10 mg B2; 50 mg B6 and 750 µg B12) in non-athletes. Notably, there are methodological differences between their study and ours, since they used an exercise protocol with a high aerobic component and we used one with a high anaerobic component. In addition, they did not use an isolated molecule, and the intake was long term.
Potential mechanisms by which MeB12 have contributed to improve performance in a Wingate test may be derived from a cognitive or central nervous system improvement. It has been shown that MeB12 supplementation can reduce diabetic peripheral neuropathic pain and associated leg symptomatology such as burning paresthesia and spontaneous pain (28, 29) and improving the distance of neurogenic claudication (30). On the other hand, it has been shown that the administration of MeB12 can improve nerve conduction velocity mediated by the recovery of myelin that promotes nerve regeneration, suppresses spontaneous ectopic discharges of peripheral primary sensory neurons in neuropathic pain conditions and restores neuromuscular functioning in peripheral hyperalgesia and allodynia (11). Therefore, the intake of MeB12 could improve performance in the Wingate test through a reduced perception of effort. Since, as it is characteristic in this type of test, the effort is maximal, and the perception of effort is a determining factor in maintaining a high-power output. These neuromuscular effects may partly explain the observed improvements in repeated sprint performance.
Furthermore, in vitro models have reported that VB12 promotes indirect antioxidant activity, as it can increase the expression of superoxide dismutase and catalase enzymes responsible for degrading reactive oxygen species (31). As is well known, reactive species can be a factor in anticipating fatigue (32). Additionally, it has been demonstrated that administering VB12 (more especially, MeB12) to melanocytes can enhance nuclear factor erythroid 2-related factor 2 (NRF2) activation, which in turn stimulates the transcription of the target genes catalase and superoxide dismutase and reduces the oxidative stress caused by hydrogen peroxide (33). Likewise improved Wingate test results may be related to a potential enhancement of the antioxidant system following supplementation.
In addition, acute supplementation with MeB12 decreased the total time (–4.9%) to complete a mental agility test before performing the Wingate test compared to placebo. In line with our results, Zhou et al. (34) after administering VB12 (cobamamide 0.25 mg/d and methylcobalamin 0.50 mg/d) for six months in middle-aged and elderly patients with cognitive impairment, found that they improved attention, calculation and visual- constructive ability compared to PLA. In addition, van Asselt et al. (35) evaluated intramuscular administration of hydroxocobalamin for five months (1000 g weekly for 4 weeks and 1000 g monthly for 4 months) on cognitive performance and brain function in elderly subjects with low cobalamin concentrations but no cognitive dysfunction. This author found that hydroxocobalamin administration decreased homocysteine and improved performance on the verbal word learning test and verbal fluency, and the quantitative electroencephalograph showed more fast activity and less slow activity. In addition, increased fast activity and decreased slow activity was associated with improved performance on word learning and verbal fluency tasks (35). Therefore, supplementation with MeB12 may decrease the time in performing the mental agility test due to an improvement in attentional or psychomotor components of cognitive function. To determine the cause of the performance and cognitive enhancement triggered by MeB12 supplementation, more clinical research is required.
The findings in this study are consistent, as the improvements in both performance and cognitive level occurred in amateur cyclists where their VB12 levels prior to MeB12 supplementation were in the normal range (mean±sd: 804±333 pg/ml). Despite this, the three-day supplementation with MeB12 formulation increases plasma VB12 levels by 16.8%, and in 72% of the subjects, it raises VB12 concentrations by more than 10%. This indicates a high efficacy in increasing VB12 levels in a short period of time of intake. In some studies, the efficacy of VB12 has been demonstrated in relation to cognitive function, but with subjects where there was a previous deficiency of VB12 (34, 35). This adds power to our results, since previously the positive effects after VB12 intake or administration had occurred in subjects with VB12 deficiency and never in subjects with normal levels.
This study has several limitations that should be considered when interpreting the results. Firstly, although the statistical power was sufficient to detect within-subject differences, future studies with larger samples are needed to confirm these results. Secondly, the study design focused on short-term supplementation (3 days), providing valuable insight into immediate effects, though it limits the possibility of assessing long-term outcomes or sustainability of the observed improvements. Finally, potential confounding variables such as sleep quality, stress or micronutritional status, which could modulate performance and cognitive outcomes, were not assessed.
One of the main strengths of this study is the use of a randomized, placebo-controlled, crossover design, and triple-blind, which minimizes interindividual variability and reinforces the reliability of the observed effects. The inclusion of multiple outcome measures, such as anaerobic power output (PMAX, PMAXREL), fatigue index (%IF), cognitive performance (mental agility test), and plasma VB12 levels, allows for a comprehensive assessment of both physical and neurocognitive responses to supplementation. In addition, the use of MeB12 a biologically active form of vitamin B12 with high neuronal affinity-provides mechanistic relevance to the findings and distinguishes this study from others using less bioavailable VB12 forms such as cyanocobalamin.
Given the limited sample size and short intervention period, further well-powered studies are required to confirm these observations, explore underlying mechanisms, and determine their relevance across different populations and exercise modalities.
5. Conclusions
This study suggests that short-term (3 days) supplementation with formulation containing methylcobalamin (MecobalActive®, HTBA) is associated with changes in selected markers of anaerobic performance and cognitive response in amateur cyclists. Compared with placebo, supplementation was associated with higher absolute and relative peak power during a repeated Wingate protocol, a smaller decline in fatigue index across sprints, and shorter completion times in a reaction-based cognitive task. Short-term intake was also associated with increased plasma vitamin B12 concentrations, including in participants with baseline values within the normal range. Taken together, these findings suggest that short-term methylcobalamin supplementation may be associated with changes in selected performance and cognitive measures under high-intensity exercise conditions in recreationally trained individuals. Given the exploratory nature, small sample size, and short intervention period, these results warrant confirmation in larger and longer-term studies.
Author Contributions
Conceptualization, F.J.M.N., P.E.A. and C.M.P.; methodology, F.J.M.N., P.E.A. and C.M.P.; software, F.J.M.N. and C.M.P.; validation, F.J.M.N., P.E.A. and C.M.P.; formal analysis, F.J.M.N. and C.M.P.; investigation, F.J.M.N., F.J.G.B., T.G.H. and C.M.P.; resources, F.J.M.N., T.G.H. and C.M.P.; data curation, F.J.M.N., F.J.G.B., T.G.H. and C.M.P.; writing—original draft preparation, F.J.M.N.; writing—review and editing, F.J.M.N., P.E.A., F.J.G.B., T.G.H. and C.M.P.; visualization, F.J.M.N. and C.M.P.; supervision, F.J.M.N. and C.M.P.; project administration, F.J.M.N. and C.M.P.; funding acquisition, P.E.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by HealthTech BioActives S.L.U. (Murcia, Spain). The funder had no role in the study design; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Acknowledgments
This study was supported by Research Center in High Performance Sport of the Catholic University of Murcia and HealthTech BioActives S.L.U. (Murcia, Spain). We thank Javier Zaragozá Santamaría (Farmacia Daya Nueva C.B.) for his contribution to the product formulation.
References
- Rodriguez, NR; Di Marco, NM; Langley, S. American College of Sports Medicine position stand. Nutrition and athletic performance. Med Sci Sports Exerc. 2009, 41(3), 709–31. [Google Scholar]
- Thomas, DT; Erdman, KA; Burke, LM. Position of the Academy of Nutrition and Dietetics, Dietitians of Canada, and the American College of Sports Medicine: nutrition and athletic performance. Journal of the Academy of Nutrition and Dietetics 2016, 116(3), 501–28. [Google Scholar] [CrossRef] [PubMed]
- Woolf, K; Manore, MM. B-vitamins and exercise: does exercise alter requirements? Int J Sport Nutr Exerc Metab. 2006, 16(5), 453–84. [Google Scholar] [CrossRef]
- Institute of Medicine Standing Committee on the Scientific Evaluation of Dietary Reference I, its Panel on Folate OBV, Choline. The National Academies Collection: Reports funded by National Institutes of Health. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B(6), Folate, Vitamin B(12), Pantothenic Acid, Biotin, and Choline; National Academies Press (US) Copyright © 1998, National Academy of Sciences.: Washington (DC), 1998. [Google Scholar]
- O'Leary, F; Samman, S. Vitamin B12 in health and disease. Nutrients 2010, 2(3), 299–316. [Google Scholar] [CrossRef] [PubMed]
- Craig, WJ. Health effects of vegan diets. Am J Clin Nutr. 2009, 89(5), 1627s–33s. [Google Scholar] [CrossRef]
- Pawlak, R; Lester, SE; Babatunde, T. The prevalence of cobalamin deficiency among vegetarians assessed by serum vitamin B12: a review of literature. Eur J Clin Nutr. 2016, 70(7), 866. [Google Scholar] [CrossRef]
- Palacios, G; Sola, R; Barrios, L; Pietrzik, K; Castillo, MJ; González-Gross, M. Algorithm for the early diagnosis of vitamin B12 deficiency in elderly people. Nutricion Hospitalaria 2013, 28(5), 1447–52. [Google Scholar] [PubMed]
- Battat, R; Kopylov, U; Szilagyi, A; Saxena, A; Rosenblatt, DS; Warner, M; et al. Vitamin B12 deficiency in inflammatory bowel disease: prevalence, risk factors, evaluation, and management. Inflammatory bowel diseases 2014, 20(6), 1120–8. [Google Scholar] [CrossRef] [PubMed]
- Salinas, M; Flores, E; López-Garrigós, M; Leiva-Salinas, C. Vitamin B12 deficiency and clinical laboratory: Lessons revisited and clarified in seven questions. International journal of laboratory hematology 2018, 40, 83–8. [Google Scholar] [CrossRef]
- Zhang, M; Han, W; Hu, S; Xu, H. Methylcobalamin: a potential vitamin of pain killer. Neural Plast. 2013, 2013, 424651. [Google Scholar] [CrossRef]
- McDowell, LR. Vitamins in animal and human nutrition; John Wiley & Sons, 2000. [Google Scholar]
- Pfohl-Leszkowicz, A; Keith, G; Dirheimer, G. Effect of cobalamin derivatives on in vitro enzymatic DNA methylation: methylcobalamin can act as a methyl donor. Biochemistry 1991, 30(32), 8045–51. [Google Scholar] [CrossRef] [PubMed]
- Toohey, JI. Vitamin B12 and methionine synthesis: a critical review. Is nature's most beautiful cofactor misunderstood? Biofactors 2006, 26(1), 45–57. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, DO. B Vitamins and the Brain: Mechanisms, Dose and Efficacy--A Review. Nutrients 2016, 8(2), 68. [Google Scholar] [CrossRef]
- Okuda, K; Yashima, K; Takara, I; Kitasaki, T; Kurashige, M; Takamatsu, M. ABSORPTION OF METHYLCOBALAMIN IN COMPARISON WITH CYANOCOBALAMIN. VITAMINS 1969, 40(4), 224–31. [Google Scholar]
- Paul, C; Brady, DM. Comparative bioavailability and utilization of particular forms of B12 supplements with potential to mitigate B12-related genetic polymorphisms. Integrative Medicine: A Clinician's Journal 2017, 16(1), 42. [Google Scholar]
- Maughan, RJ; Burke, LM; Dvorak, J; Larson-Meyer, DE; Peeling, P; Phillips, SM; et al. IOC consensus statement: dietary supplements and the high-performance athlete. Br J Sports Med. 2018, 52(7), 439–55. [Google Scholar] [CrossRef]
- Janssen, JJE; Grefte, S; Keijer, J; de Boer, VCJ. Mito-Nuclear Communication by Mitochondrial Metabolites and Its Regulation by B-Vitamins. Front Physiol. 2019, 10, 78. [Google Scholar] [CrossRef]
- Lukaski, HC. Vitamin and mineral status: effects on physical performance. Nutrition 2004, 20(7-8), 632–44. [Google Scholar] [CrossRef]
- Read, MH; McGuffin, S. The effect of B-complex supplementation on endurance performance. Journal of sports medicine and physical fitness 1983, 23(2), 178–84. [Google Scholar]
- Mya-Tu, M. The effect of vitamin B12 on physical performance capacity. British Journal of Nutrition 1978, 40(2), 269–73. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. Jama 2013, 310(20), 2191–4. [CrossRef]
- Stewart, A; Marfell-Jones, M; Olds, T; De Ridder, H. International Society for advancement of Kinanthropometry. In International standards for anthropometric assessment; 2011; pp. 50–3. [Google Scholar]
- Faulkner, JA. Physiology of Swimming. Research Quarterly American Association for Health, Physical Education and Recreation 1966, 37(1), 41–54. [Google Scholar] [CrossRef]
- Norton, KI. Standards for anthropometry assessment; Routledge: Kinanthropometry and exercise physiology, 2018; pp. 68–137. [Google Scholar]
- Lee, MC; Hsu, YJ; Shen, SY; Ho, CS; Huang, CC. A functional evaluation of anti- fatigue and exercise performance improvement following vitamin B complex supplementation in healthy humans, a randomized double-blind trial. Int J Med Sci. 2023, 20(10), 1272–81. [Google Scholar] [CrossRef]
- Li, G. Effect of mecobalamin on diabetic neuropathies. Beijing Methycobal Clinical Trial Collaborative Group. Zhonghua Nei Ke Za Zhi 1999, 38(1), 14–7. [Google Scholar]
- Ide, H; Fujiya, S; Asanuma, Y; Tsuji, M; Sakai, H; Agishi, Y. Clinical usefulness of intrathecal injection of methylcobalamin in patients with diabetic neuropathy. Clin Ther. 1987, 9(2), 183–92. [Google Scholar] [PubMed]
- Waikakul, W; Waikakul, S. Methylcobalamin as an adjuvant medication in conservative treatment of lumbar spinal stenosis. J Med Assoc Thai 2000, 83(8), 825–31. [Google Scholar] [PubMed]
- Theiss, EL; Griebsch, LV; Lauer, AA; Janitschke, D; Erhardt, VKJ; Haas, EC; et al. Vitamin B12 Attenuates Changes in Phospholipid Levels Related to Oxidative Stress in SH-SY5Y Cells. Cells 2022, 11(16). [Google Scholar] [CrossRef]
- Cheng, AJ; Yamada, T; Rassier, DE; Andersson, DC; Westerblad, H; Lanner, JT. Reactive oxygen/nitrogen species and contractile function in skeletal muscle during fatigue and recovery. J Physiol. 2016, 594(18), 5149–60. [Google Scholar] [CrossRef] [PubMed]
- An, R; Li, D; Dong, Y; She, Q; Zhou, T; Nie, X; et al. Methylcobalamin Protects Melanocytes from H(2)O(2)-Induced Oxidative Stress by Activating the Nrf2/HO-1 Pathway. Drug Des Devel Ther. 2021, 15, 4837–48. [Google Scholar] [CrossRef]
- Zhou, L; Bai, X; Huang, J; Tan, Y; Yang, Q. Vitamin B12 supplementation improves cognitive function in middle aged and elderly patients with cognitive impairment. Nutr Hosp. 2023, 40(4), 724–31. [Google Scholar]
- van Asselt, DZ; Pasman, JW; van Lier, HJ; Vingerhoets, DM; Poels, PJ; Kuin, Y; et al. Cobalamin supplementation improves cognitive and cerebral function in older, cobalamin-deficient persons. J Gerontol A Biol Sci Med Sci. 2001, 56(12), M775–9. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).