Submitted:
21 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods and Evidence Acquisition
2.1. Defining the Border Zone Across Scales
2.2. Microstructure and Anisotropy in the Border Zone
2.3. Border-Zone Stress, Strain, and Shear After MI
2.4. Mechanotransduction Linking Surviving Myocytes to Fibrosis
2.5. Temporal Evolution from Edema and Inflammation to Granulation Tissue and Mature Scar
2.6. Electrical and Arrhythmogenic Implications of Border-Zone Remodelling
2.7. Implications for Therapy and Mechanomodulatory Interventions
3. Limitations and Controversies
Future Directions and Testable Hypotheses
4. Conclusions
References
- Pfeffer, M.A.; Braunwald, E. Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 1990, 81, 1161–1172. [Google Scholar] [CrossRef]
- Sutton, M.G.S.J.; Sharpe, N. Left ventricular remodeling after myocardial infarction: pathophysiology and therapy. Circulation 2000, 101, 2981–2988. [Google Scholar] [CrossRef] [PubMed]
- Frangogiannis, N.G. The inflammatory response in myocardial injury, repair, and remodelling. Nature Reviews Cardiology 2014, 11, 255–265. [Google Scholar] [CrossRef]
- Leancă, S.A.; Crișu, D.; Petriș, A.O.; Afrăsânie, I.; Genes, A.; Costache, A.D.; Tesloianu, D.N.; Costache, I.I. Left ventricular remodeling after myocardial infarction: from physiopathology to treatment. Life 2022, 12, 1111. [Google Scholar] [CrossRef] [PubMed]
- Weisman, H.F.; Healy, B. Myocardial infarct expansion, infarct extension, and reinfarction: pathophysiologic concepts. Progress in cardiovascular diseases 1987, 30, 73–110. [Google Scholar] [CrossRef] [PubMed]
- Travers, J.G.; Kamal, F.A.; Robbins, J.; Yutzey, K.E.; Blaxall, B.C. Cardiac fibrosis: the fibroblast awakens. Circulation research 2016, 118, 1021–1040. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, S.D.; Frangogiannis, N.G. The biological basis for cardiac repair after myocardial infarction: from inflammation to fibrosis. Circulation research 2016, 119, 91–112. [Google Scholar] [CrossRef]
- Guccione, J.M.; McCulloch, A.D.; Waldman, L. Passive material properties of intact ventricular myocardium determined from a cylindrical model; 1991. [Google Scholar]
- Fung, Y.C. What are the residual stresses doing in our blood vessels? Annals of biomedical engineering 1991, 19, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, J.D.; Rajagopal, K. A constrained mixture model for growth and remodeling of soft tissues. Mathematical models and methods in applied sciences 2002, 12, 407–430. [Google Scholar] [CrossRef]
- Leask, A. Getting to the heart of the matter: new insights into cardiac fibrosis. Circulation research 2015, 116, 1269–1276. [Google Scholar]
- Kidambi, A.; Mather, A.N.; Motwani, M.; Swoboda, P.; Uddin, A.; Greenwood, J.P.; Plein, S. The effect of microvascular obstruction and intramyocardial hemorrhage on contractile recovery in reperfused myocardial infarction: insights from cardiovascular magnetic resonance. Journal of Cardiovascular Magnetic Resonance 2013, 15, 58. [Google Scholar] [CrossRef]
- Holzapfel, G.A.; Ogden, R.W. Constitutive modelling of passive myocardium: a structurally based framework for material characterization. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 2009, 367, 3445–3475. [Google Scholar] [CrossRef] [PubMed]
- Fung, Y.; Liu, S. Determination of the mechanical properties of the different layers of blood vessels in vivo. Proceedings of the National Academy of Sciences 1995, 92, 2169–2173. [Google Scholar] [CrossRef] [PubMed]
- Bers, D.M. Cardiac excitation–contraction coupling. Nature 2002, 415, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Camelliti, P.; Borg, T.K.; Kohl, P. Structural and functional characterisation of cardiac fibroblasts. Cardiovascular research 2005, 65, 40–51. [Google Scholar] [CrossRef] [PubMed]
- De Bakker, J.; Van Capelle, F.; Janse, M.J.; Tasseron, S.; Vermeulen, J.T.; de Jonge, N.; Lahpor, J.R. Slow conduction in the infarcted human heart.'Zigzag'course of activation. Circulation 1993, 88, 915–926. [Google Scholar] [CrossRef] [PubMed]
- Spinale, F.G. Myocardial matrix remodeling and the matrix metalloproteinases: influence on cardiac form and function. Physiological reviews 2007, 87, 1285–1342. [Google Scholar] [CrossRef]
- Goldberger, J.J.; Basu, A.; Boineau, R.; Buxton, A.E.; Cain, M.E.; Canty, J.M., Jr.; Chen, P.-S.; Chugh, S.S.; Costantini, O.; Exner, D.V. Risk stratification for sudden cardiac death: a plan for the future. Circulation 2014, 129, 516–526. [Google Scholar] [CrossRef]
- Delmar, M.; Makita, N. Cardiac connexins, mutations and arrhythmias. Current opinion in cardiology 2012, 27, 236–241. [Google Scholar] [CrossRef]
- Verma, A.; Marrouche, N.F.; Schweikert, R.A.; Saliba, W.; Wazni, O.; Cummings, J.; ABDUL-KARIM, A.; Bhargava, M.; Burkhardt, J.D.; Kilicaslan, F. Relationship between successful ablation sites and the scar border zone defined by substrate mapping for ventricular tachycardia post-myocardial infarction. Journal of cardiovascular electrophysiology 2005, 16, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Berruezo, A.; Fernández-Armenta, J.; Andreu, D.; Penela, D.; Herczku, C.; Evertz, R.; Cipolletta, L.; Acosta, J.; Borràs, R.; Arbelo, E. Scar dechanneling: new method for scar-related left ventricular tachycardia substrate ablation. Circulation: Arrhythmia and Electrophysiology 2015, 8, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, A.F.; Bertelsen, L.; Jøns, C.; Jabbari, R.; Lønborg, J.; Kyhl, K.; Göransson, C.; Nepper-Christensen, L.; Atharovski, K.; Ekström, K. Scar border zone mass and presence of border zone channels assessed with cardiac magnetic resonance imaging are associated with ventricular arrhythmia in patients with ST-segment elevation myocardial infarction. Europace 2023, 25, 978–988. [Google Scholar] [CrossRef] [PubMed]
- D'hooge, J.; Heimdal, A.; Jamal, F.; Kukulski, T.; Bijnens, B.; Rademakers, F.; Hatle, L.; Suetens, P.; Sutherland, G.R. Regional strain and strain rate measurements by cardiac ultrasound: principles, implementation and limitations. European Journal of Echocardiography 2000, 1, 154–170. [Google Scholar]
- Nesbitt, G.C.; Mankad, S.; Oh, J.K. Strain imaging in echocardiography: methods and clinical applications. The international journal of cardiovascular imaging 2009, 25, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Mirea, O.; Pagourelias, E.D.; Duchenne, J.; Bogaert, J.; Thomas, J.D.; Badano, L.P.; Voigt, J.-U. Variability and reproducibility of segmental longitudinal strain measurement: a report from the EACVI-ASE strain standardization task force. JACC: Cardiovascular Imaging 2018, 11, 15–24. [Google Scholar] [PubMed]
- Gherbesi, E.; Gianstefani, S.; Angeli, F.; Ryabenko, K.; Bergamaschi, L.; Armillotta, M.; Guerra, E.; Tuttolomondo, D.; Gaibazzi, N.; Squeri, A. Myocardial strain of the left ventricle by speckle tracking echocardiography: From physics to clinical practice. Echocardiography 2024, 41, e15753. [Google Scholar] [CrossRef]
- Kim, R.J.; Wu, E.; Rafael, A.; Chen, E.-L.; Parker, M.A.; Simonetti, O.; Klocke, F.J.; Bonow, R.O.; Judd, R.M. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. New England Journal of Medicine 2000, 343, 1445–1453. [Google Scholar] [CrossRef]
- Rajiah, P.; Desai, M.Y.; Kwon, D.; Flamm, S.D. MR imaging of myocardial infarction. Radiographics 2013, 33, 1383–1412. [Google Scholar] [CrossRef]
- Kramer, C.M.; Barkhausen, J.; Bucciarelli-Ducci, C.; Flamm, S.D.; Kim, R.J.; Nagel, E. Standardized cardiovascular magnetic resonance imaging (CMR) protocols: 2020 update. Journal of Cardiovascular Magnetic Resonance 2020, 22, 17. [Google Scholar] [CrossRef]
- Segmentation, A.H.A.W.G.o.M.; Imaging, R.f.C.; Cerqueira, M.D.; Weissman, N.J.; Dilsizian, V.; Jacobs, A.K.; Kaul, S.; Laskey, W.K.; Pennell, D.J.; Rumberger, J.A.; et al. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 2002, 105, 539–542. [Google Scholar]
- Schuijf, J.D.; Kaandorp, T.A.; Lamb, H.J.; van der Geest, R.J.; Viergever, E.P.; van der Wall, E.E.; de Roos, A.; Bax, J.J. Quantification of myocardial infarct size and transmurality by contrast-enhanced magnetic resonance imaging in men. The American journal of cardiology 2004, 94, 284–288. [Google Scholar] [CrossRef]
- Messroghli, D.R.; Moon, J.C.; Ferreira, V.M.; Grosse-Wortmann, L.; He, T.; Kellman, P.; Mascherbauer, J.; Nezafat, R.; Salerno, M.; Schelbert, E.B. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: a consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI). Journal of Cardiovascular Magnetic Resonance 2016, 19, 75. [Google Scholar] [CrossRef]
- Moon, J.C.; Messroghli, D.R.; Kellman, P.; Piechnik, S.K.; Robson, M.D.; Ugander, M.; Gatehouse, P.D.; Arai, A.E.; Friedrich, M.G.; Neubauer, S. Myocardial T1 mapping and extracellular volume quantification: a Society for Cardiovascular Magnetic Resonance (SCMR) and CMR Working Group of the European Society of Cardiology consensus statement. Journal of Cardiovascular Magnetic Resonance 2013, 15, 92. [Google Scholar] [CrossRef]
- Kammerlander, A.A.; Marzluf, B.A.; Zotter-Tufaro, C.; Aschauer, S.; Duca, F.; Bachmann, A.; Knechtelsdorfer, K.; Wiesinger, M.; Pfaffenberger, S.; Greiser, A. T1 mapping by CMR imaging: from histological validation to clinical implication. JACC: Cardiovascular Imaging 2016, 9, 14–23. [Google Scholar]
- Han, D.; Lin, A.; Kuronuma, K.; Gransar, H.; Dey, D.; Friedman, J.D.; Berman, D.S.; Tamarappoo, B.K. Cardiac computed tomography for quantification of myocardial extracellular volume fraction: a systematic review and meta-analysis. Cardiovascular Imaging 2023, 16, 1306–1317. [Google Scholar]
- Turer, A.T.; Hill, J.A. Pathogenesis of myocardial ischemia-reperfusion injury and rationale for therapy. The American journal of cardiology 2010, 106, 360–368. [Google Scholar] [CrossRef]
- Nielles-Vallespin, S.; Khalique, Z.; Ferreira, P.F.; de Silva, R.; Scott, A.D.; Kilner, P.; McGill, L.-A.; Giannakidis, A.; Gatehouse, P.D.; Ennis, D. Assessment of myocardial microstructural dynamics by in vivo diffusion tensor cardiac magnetic resonance. Journal of the American College of Cardiology 2017, 69, 661–676. [Google Scholar] [CrossRef] [PubMed]
- Dall’Armellina, E.; Plein, S. Diffusion tensor imaging to assess myocardial microstructure after infarction by magnetic resonance; Oxford University Press: UK, 2025; Volume 46, pp. 470–472. [Google Scholar] [CrossRef] [PubMed]
- Chabiniok, R.; Wang, V.Y.; Hadjicharalambous, M.; Asner, L.; Lee, J.; Sermesant, M.; Kuhl, E.; Young, A.A.; Moireau, P.; Nash, M.P. Multiphysics and multiscale modelling, data–model fusion and integration of organ physiology in the clinic: ventricular cardiac mechanics. Interface focus 2016, 6, 20150083. [Google Scholar] [CrossRef] [PubMed]
- Niederer, S.A.; Lumens, J.; Trayanova, N.A. Computational models in cardiology. Nature reviews cardiology 2019, 16, 100–111. [Google Scholar] [CrossRef]
- Rodero, C.; Baptiste, T.M.; Barrows, R.K.; Lewalle, A.; Niederer, S.A.; Strocchi, M. Advancing clinical translation of cardiac biomechanics models: a comprehensive review, applications and future pathways. Frontiers in physics 2023, 11, 1306210. [Google Scholar] [CrossRef] [PubMed]
- Pathmanathan, P.; Gray, R.A. Verification of computational models of cardiac electro-physiology. International journal for numerical methods in biomedical engineering 2014, 30, 525–544. [Google Scholar] [CrossRef]
- Gray, R.A.; Pathmanathan, P. Patient-specific cardiovascular computational modeling: diversity of personalization and challenges. Journal of cardiovascular translational research 2018, 11, 80–88. [Google Scholar] [CrossRef]
- Avazmohammadi, R.; Li, D.S.; Leahy, T.; Shih, E.; Soares, J.S.; Gorman, J.H.; Gorman, R.C.; Sacks, M.S. An integrated inverse model-experimental approach to determine soft tissue three-dimensional constitutive parameters: application to post-infarcted myocardium. Biomechanics and modeling in mechanobiology 2018, 17, 31–53. [Google Scholar] [CrossRef]
- Ingber, D.E. Cellular mechanotransduction: putting all the pieces together again. The FASEB journal 2006, 20, 811–827. [Google Scholar] [CrossRef] [PubMed]
- Discher, D.E.; Janmey, P.; Wang, Y.-l. Tissue cells feel and respond to the stiffness of their substrate. Science 2005, 310, 1139–1143. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S. Role of YAP/TAZ in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Hinz, B. The myofibroblast: paradigm for a mechanically active cell. Journal of biomechanics 2010, 43, 146–155. [Google Scholar] [CrossRef]
- Maeda, T.; Sakabe, T.; Sunaga, A.; Sakai, K.; Rivera, A.L.; Keene, D.R.; Sasaki, T.; Stavnezer, E.; Iannotti, J.; Schweitzer, R. Conversion of mechanical force into TGF-β-mediated biochemical signals. Current Biology 2011, 21, 933–941. [Google Scholar] [CrossRef]
- Perreault, L.R.; Black, L.D., III. Mechanobiology of cardiac fibroblasts in cardiac remodeling. Cardiac Mechanobiology in Physiology and Disease 2023, 101–120. [Google Scholar]
- Fonseca, F.A.; Izar, M.C. Role of inflammation in cardiac remodeling after acute myocardial infarction. Frontiers in physiology 2022, 13, 927163. [Google Scholar] [CrossRef]
- Galis, Z.S.; Khatri, J.J. Matrix metalloproteinases in vascular remodeling and atherogenesis: the good, the bad, and the ugly. Circulation research 2002, 90, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Zardini, P.; Marino, P.; Golia, G.; Anselmi, M.; Castelli, M. Ventricular remodeling and infarct expansion. The American journal of cardiology 1993, 72, G98–G106. [Google Scholar] [CrossRef] [PubMed]
- Zerhouni, E.A.; Parish, D.M.; Rogers, W.J.; Yang, A.; Shapiro, E.P. Human heart: tagging with MR imaging--a method for noninvasive assessment of myocardial motion. Radiology 1988, 169, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Bujak, M.; Frangogiannis, N.G. The role of TGF-β signaling in myocardial infarction and cardiac remodeling. Cardiovascular research 2007, 74, 184–195. [Google Scholar] [CrossRef]
- Fomovsky, G.M.; Rouillard, A.D.; Holmes, J.W. Regional mechanics determine collagen fiber structure in healing myocardial infarcts. Journal of molecular and cellular cardiology 2012, 52, 1083–1090. [Google Scholar] [CrossRef]
- Li, S.; Yin, W.; Liu, Y.; Yang, C.; Zhai, Z.; Xie, M.; Ye, Z.; Song, X. Anisotropic conductive scaffolds for post-infarction cardiac repair. Biomaterials Science 2025, 13, 542–567. [Google Scholar] [CrossRef]
- Francisco, J.; Zhang, Y.; Im Jeong, J.; Mizushima, W.; Ikeda, S.; Ivessa, A.; Oka, S.; Zhai, P.; Tallquist, M.D.; Del Re, D.P. Blockade of fibroblast YAP attenuates cardiac fibrosis and dysfunction through MRTF-A inhibition. JACC: Basic to Translational Science 2020, 5, 931–945. [Google Scholar] [CrossRef] [PubMed]
- Del Re, D.P. Hippo-Yap signaling in cardiac and fibrotic remodeling. Current opinion in physiology 2022, 26, 100492. [Google Scholar] [CrossRef]
- Venugopal, H.; Hanna, A.; Humeres, C.; Frangogiannis, N.G. Properties and functions of fibroblasts and myofibroblasts in myocardial infarction. Cells 2022, 11, 1386. [Google Scholar] [CrossRef] [PubMed]
- Wells, R.G. Tissue mechanics and fibrosis. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 2013, 1832, 884–890. [Google Scholar] [CrossRef] [PubMed]
- Namashiri, P.; Allahverdizadeh, A.; Dadashzadeh, B.; Dokos, S. Electromechanical modeling of the left ventricle: considering hyperelastic and viscoelastic properties. Journal of the Brazilian Society of Mechanical Sciences and Engineering 2024, 46, 704. [Google Scholar] [CrossRef]
- Nishimura, R.A.; Carabello, B.A. Hemodynamics in the cardiac catheterization laboratory of the 21st century. Circulation 2012, 125, 2138–2150. [Google Scholar] [CrossRef]
- Choi, H.F.; Rademakers, F.E.; Claus, P. Left-ventricular shape determines intramyocardial mechanical heterogeneity. American Journal of Physiology-Heart and Circulatory Physiology 2011, 301, H2351–H2361. [Google Scholar] [CrossRef] [PubMed]
- Holzapfel, G.A.; Gasser, T.C.; Ogden, R.W. A new constitutive framework for arterial wall mechanics and a comparative study of material models. Journal of elasticity and the physical science of solids 2000, 61, 1–48. [Google Scholar]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Infarction, E.G. Fourth universal definition of myocardial infarction (2018). Journal of the American college of cardiology 2018, 72, 2231–2264. [Google Scholar]
- Humphrey, J.D. Mechanisms of arterial remodeling in hypertension: coupled roles of wall shear and intramural stress. Hypertension 2008, 52, 195–200. [Google Scholar] [CrossRef]
- Gibbs, T.; Villa, A.; Sammut, E.; Jeyabraba, S.; Carr-White, G.; Ismail, T.; Mullen, G.; Ganeshan, B.; Chiribiri, A. Quantitative assessment of myocardial scar heterogeneity using cardiovascular magnetic resonance texture analysis to risk stratify patients post-myocardial infarction. Clinical radiology 2018, 73, 1059. e1017–1059. e1026. [Google Scholar] [CrossRef]
- Moccia, S.; Banali, R.; Martini, C.; Muscogiuri, G.; Pontone, G.; Pepi, M.; Caiani, E.G. Development and testing of a deep learning-based strategy for scar segmentation on CMR-LGE images. Magnetic Resonance Materials in Physics, Biology and Medicine 2019, 32, 187–195. [Google Scholar] [CrossRef]
- Demer, L.L.; Yin, F. Passive biaxial mechanical properties of isolated canine myocardium. The Journal of physiology 1983, 339, 615–630. [Google Scholar] [PubMed]
- Sirry, M.S.; Butler, J.R.; Patnaik, S.S.; Brazile, B.; Bertucci, R.; Claude, A.; McLaughlin, R.; Davies, N.H.; Liao, J.; Franz, T. Characterisation of the mechanical properties of infarcted myocardium in the rat under biaxial tension and uniaxial compression. journal of the mechanical behavior of biomedical materials 2016, 63, 252–264. [Google Scholar] [CrossRef]
- Ngwangwa, H.; Nemavhola, F.; Pandelani, T.; Msibi, M.; Mabuda, I.; Davies, N.; Franz, T. Determination of cross-directional and cross-wall variations of passive biaxial mechanical properties of rat myocardia. Processes 2022, 10, 629. [Google Scholar] [CrossRef]
- Pandelani, T.; Semakane, L.; Msibi, M.; Kuchumov, A.G.; Nemavhola, F. Passive biaxial mechanical properties of sheep myocardium. Frontiers in Bioengineering and Biotechnology 2025, 13, 1549829. [Google Scholar] [CrossRef] [PubMed]
- Nemavhola, F. Study of biaxial mechanical properties of the passive pig heart: material characterisation and categorisation of regional differences. International Journal of Mechanical and Materials Engineering 2021, 16, 6. [Google Scholar] [CrossRef]
- Cerqueira, M.D. American Heart Association Writing Group on Myocardial Segmentation and Registration for Cardiac Imaging: standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 2002, 105, 539–542. [Google Scholar]
- Benz, D.C.; Kaufmann, P.A.; Dorbala, S. Transmural perfusion: A new direction for myocardial blood flow. Journal of Nuclear Cardiology 2022, 29, 1952–1955. [Google Scholar] [CrossRef] [PubMed]
- Richardson, W.J.; Clarke, S.A.; Quinn, T.A.; Holmes, J.W. Physiological implications of myocardial scar structure. Comprehensive Physiology 2015, 5, 1877–1909. [Google Scholar] [CrossRef]
- Bogaert, J.; Dymarkowski, S.; Taylor, A.M.; Muthurangu, V. Clinical cardiac MRI; Springer Science & Business Media, 2012. [Google Scholar]
- Unger, A.; Garot, J.; Toupin, S.; Duhamel, S.; Sanguineti, F.; Hovasse, T.; Champagne, S.; Unterseeh, T.; Chevalier, B.; Akodad, M. Prognostic value of cardiac MRI late gadolinium enhancement granularity in participants with ischemic cardiomyopathy. Radiology 2025, 314, e240806. [Google Scholar] [CrossRef]
- Eitel, I.; Stiermaier, T.; Lange, T.; Rommel, K.-P.; Koschalka, A.; Kowallick, J.T.; Lotz, J.; Kutty, S.; Gutberlet, M.; Hasenfuß, G. Cardiac magnetic resonance myocardial feature tracking for optimized prediction of cardiovascular events following myocardial infarction. JACC: Cardiovascular Imaging 2018, 11, 1433–1444. [Google Scholar] [CrossRef] [PubMed]
- Reindl, M.; Tiller, C.; Holzknecht, M.; Lechner, I.; Eisner, D.; Riepl, L.; Pamminger, M.; Henninger, B.; Mayr, A.; Schwaiger, J.P. Global longitudinal strain by feature tracking for optimized prediction of adverse remodeling after ST-elevation myocardial infarction. Clinical Research in Cardiology 2021, 110, 61–71. [Google Scholar] [CrossRef]
- Reindl, M.; Tiller, C.; Holzknecht, M.; Lechner, I.; Beck, A.; Plappert, D.; Gorzala, M.; Pamminger, M.; Mayr, A.; Klug, G. Prognostic implications of global longitudinal strain by feature-tracking cardiac magnetic resonance in ST-elevation myocardial infarction. Circulation: Cardiovascular Imaging 2019, 12, e009404. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yang, W.; Zhao, S.; Lu, M. State-of-the-art myocardial strain by CMR feature tracking: clinical applications and future perspectives. European radiology 2022, 32, 5424–5435. [Google Scholar] [CrossRef] [PubMed]
- Masithulela, F. Analysis of passive filling with fibrotic myocardial infarction. In ASME international mechanical engineering congress and exposition; American Society of Mechanical Engineers, 2015; p. V003T003A004. [Google Scholar]
- Masithulela, F. Bi-ventricular finite element model of right ventricle overload in the healthy rat heart. Bio-medical materials and engineering 2016, 27, 507–525. [Google Scholar] [CrossRef]
- Masithulela, F.J. Computational biomechanics in the remodelling rat heart post myocardial infarction; 2016. [Google Scholar]
- Masithulela, F. The effect of over-loaded right ventricle during passive filling in rat heart: A biventricular finite element model. In ASME international mechanical engineering congress and exposition; American Society of Mechanical Engineers, 2015; p. V003T003A005. [Google Scholar]
- Calvieri, C.; Riva, A.; Sturla, F.; Dominici, L.; Conia, L.; Gaudio, C.; Miraldi, F.; Secchi, F.; Galea, N. Left ventricular adverse remodeling in ischemic heart disease: emerging cardiac magnetic resonance imaging biomarkers. Journal of Clinical Medicine 2023, 12, 334. [Google Scholar] [CrossRef]
- Fung, Y.-c. Biomechanics: mechanical properties of living tissues; Springer Science & Business Media, 2013. [Google Scholar]
- Fung, Y.; Liu, S.; Zhou, J. Remodeling of the constitutive equation while a blood vessel remodels itself under stress. 1993. [Google Scholar] [CrossRef] [PubMed]
- Gasser, T.C.; Ogden, R.W.; Holzapfel, G.A. Hyperelastic modelling of arterial layers with distributed collagen fibre orientations. Journal of the royal society interface 2006, 3, 15–35. [Google Scholar] [CrossRef]
- Sermesant, M.; Delingette, H.; Ayache, N. An electromechanical model of the heart for image analysis and simulation. IEEE transactions on medical imaging 2006, 25, 612–625. [Google Scholar] [CrossRef]
- Holzapfel, G.A.; Ogden, R.W. Biomechanics of soft tissue in cardiovascular systems; Springer Science & Business Media, 2003; Volume 441. [Google Scholar]
- Camelliti, P.; Green, C.; Kohl, P. Structural and functional coupling of cardiac myocytes and fibroblasts. Advances in cardiology 2006, 42, 132. [Google Scholar]
- Takayama, Y.; Costa, K.D.; Covell, J.W. Contribution of laminar myofiber architecture to load-dependent changes in mechanics of LV myocardium. American Journal of Physiology-Heart and Circulatory Physiology 2002, 282, H1510–H1520. [Google Scholar] [CrossRef]
- Fomovsky, G.M.; Thomopoulos, S.; Holmes, J.W. Contribution of extracellular matrix to the mechanical properties of the heart. Journal of molecular and cellular cardiology 2010, 48, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Orr, A.W.; Helmke, B.P.; Blackman, B.R.; Schwartz, M.A. Mechanisms of mechanotransduction. Developmental cell 2006, 10, 11–20. [Google Scholar] [CrossRef]
- Nemavhola, F. Biaxial quantification of passive porcine myocardium elastic properties by region. Engineering Solid Mechanics 2017. [Google Scholar] [CrossRef]
- Nemavhola, F. Detailed structural assessment of healthy interventricular septum in the presence of remodeling infarct in the free wall–A finite element model. Heliyon 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Ngwangwa, H.M.; Nemavhola, F. Evaluating computational performances of hyperelastic models on supraspinatus tendon uniaxial tensile test data. Journal of Computational Applied Mechanics 2021, 52, 27–43. [Google Scholar]
- Nemavhola, F. Fibrotic infarction on the LV free wall may alter the mechanics of healthy septal wall during passive filling. Bio-medical materials and engineering 2017, 28, 579–599. [Google Scholar] [CrossRef]
- Nemavhola, F. Mechanics of the septal wall may be affected by the presence of fibrotic infarct in the free wall at end-systole. International Journal of Medical Engineering and Informatics 2019, 11, 205–225. [Google Scholar] [CrossRef]
- Asanuma, T.; Nakatani, S. Myocardial ischaemia and post-systolic shortening. Heart 2015, 101, 509–516. [Google Scholar] [CrossRef]
- Stewart, L.; Turner, N.A. Channelling the force to reprogram the matrix: Mechanosensitive ion channels in cardiac fibroblasts. Cells 2021, 10, 990. [Google Scholar] [CrossRef]
- Aimo, A.; Gaggin, H.K.; Barison, A.; Emdin, M.; Januzzi, J.L. Imaging, biomarker, and clinical predictors of cardiac remodeling in heart failure with reduced ejection fraction. JACC: Heart Failure 2019, 7, 782–794. [Google Scholar] [CrossRef]
- Sacks, M.S.; Enomoto, Y.; Graybill, J.R.; Merryman, W.D.; Zeeshan, A.; Yoganathan, A.P.; Levy, R.J.; Gorman, R.C.; Gorman, J.H., III. In-vivo dynamic deformation of the mitral valve anterior leaflet. The Annals of thoracic surgery 2006, 82, 1369–1377. [Google Scholar] [CrossRef]
- Hamill, O.P.; Martinac, B. Molecular basis of mechanotransduction in living cells. Physiological reviews 2001, 81, 685–740. [Google Scholar]
- Lyon, R.C.; Zanella, F.; Omens, J.H.; Sheikh, F. Mechanotransduction in cardiac hypertrophy and failure. Circulation research 2015, 116, 1462–1476. [Google Scholar] [CrossRef] [PubMed]
- Sugden, P.H.; Clerk, A. Cellular mechanisms of cardiac hypertrophy. Journal of molecular medicine 1998, 76, 725–746. [Google Scholar] [CrossRef]
- Ma, Y.; de Castro Brás, L.E.; Toba, H.; Iyer, R.P.; Hall, M.E.; Winniford, M.D.; Lange, R.A.; Tyagi, S.C.; Lindsey, M.L. Myofibroblasts and the extracellular matrix network in post-myocardial infarction cardiac remodeling. Pflügers Archiv-European Journal of Physiology 2014, 466, 1113–1127. [Google Scholar] [CrossRef]
- Burlew, B.S.; Weber, K.T. Cardiac fibrosis as a cause of diastolic dysfunction. Herz 2002, 27, 92–98. [Google Scholar] [CrossRef]
- Ravens, U. Mechano-electric feedback and arrhythmias. Progress in biophysics and molecular biology 2003, 82, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Zipes, D.P.; Jalife, J.; Stevenson, W.G. Cardiac electrophysiology: from cell to bedside E-book; Elsevier Health Sciences, 2017. [Google Scholar]
- Jain, N.; Moeller, J.; Vogel, V. Mechanobiology of macrophages: how physical factors coregulate macrophage plasticity and phagocytosis. Annual review of biomedical engineering 2019, 21, 267–297. [Google Scholar] [CrossRef] [PubMed]
- Kologrivova, I.; Shtatolkina, M.; Suslova, T.; Ryabov, V. Cells of the immune system in cardiac remodeling: main players in resolution of inflammation and repair after myocardial infarction. Frontiers in immunology 2021, 12, 664457. [Google Scholar] [CrossRef] [PubMed]
- Aloysius, A.; Saxena, S.; Seifert, A.W. Metabolic regulation of innate immune cell phenotypes during wound repair and regeneration. Current opinion in immunology 2021, 68, 72–82. [Google Scholar] [CrossRef]
- Phatharajaree, W.; Phrommintikul, A.; Chattipakorn, N. Matrix metalloproteinases and myocardial infarction. Canadian Journal of Cardiology 2007, 23, 727–733. [Google Scholar] [CrossRef]
- Chitiboi, T.; Axel, L. Magnetic resonance imaging of myocardial strain: a review of current approaches. Journal of Magnetic Resonance Imaging 2017, 46, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Imanli, H.; Ume, K.L.; Jeudy, J.; Bob-Manuel, T.; Smith, M.F.; Chen, W.; Abdulghani, M.; Ghzally, Y.; Mahat, J.B.; Itah, R. Ventricular tachycardia (VT) substrate characteristics: insights from multimodality structural and functional imaging of the VT substrate using cardiac MRI scar, 123I-metaiodobenzylguanidine SPECT innervation, and bipolar voltage. Journal of Nuclear Medicine 2019, 60, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Imbesi, A.; Greco, A.; Spagnolo, M.; Laudani, C.; Raffo, C.; Finocchiaro, S.; Mazzone, P.M.; Landolina, D.; Mauro, M.S.; Cutore, L. Targeting inflammation after acute myocardial infarction. Journal of the American College of Cardiology 2025, 86, 1146–1169. [Google Scholar] [CrossRef] [PubMed]
- Simon, M.A.; Kormos, R.L.; Murali, S.; Nair, P.; Heffernan, M.; Gorcsan, J.; Winowich, S.; McNamara, D.M. Myocardial recovery using ventricular assist devices: prevalence, clinical characteristics, and outcomes. Circulation 2005, 112, I-32-I-36. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.E.; Hsieh, P.C.; Grodzinsky, A.J.; Lee, R.T. Custom design of the cardiac microenvironment with biomaterials. Circulation research 2005, 97, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Vunjak-Novakovic, G.; Tandon, N.; Godier, A.; Maidhof, R.; Marsano, A.; Martens, T.P.; Radisic, M. Challenges in cardiac tissue engineering. Tissue Engineering Part B: Reviews 2010, 16, 169–187. [Google Scholar] [CrossRef] [PubMed]
- Tesch, G.H.; Young, M.J. Mineralocorticoid receptor signaling as a therapeutic target for renal and cardiac fibrosis. Frontiers in pharmacology 2017, 8, 313. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Zhou, B. Role of cardiac fibroblasts in cardiac injury and repair. Current Cardiology Reports 2022, 24, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Crampin, E.J.; Halstead, M.; Hunter, P.; Nielsen, P.; Noble, D.; Smith, N.; Tawhai, M. Computational physiology and the physiome project. Experimental Physiology 2004, 89, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.A.; DeSimone, D.W. Cell adhesion receptors in mechanotransduction. Current opinion in cell biology 2008, 20, 551–556. [Google Scholar] [CrossRef] [PubMed]
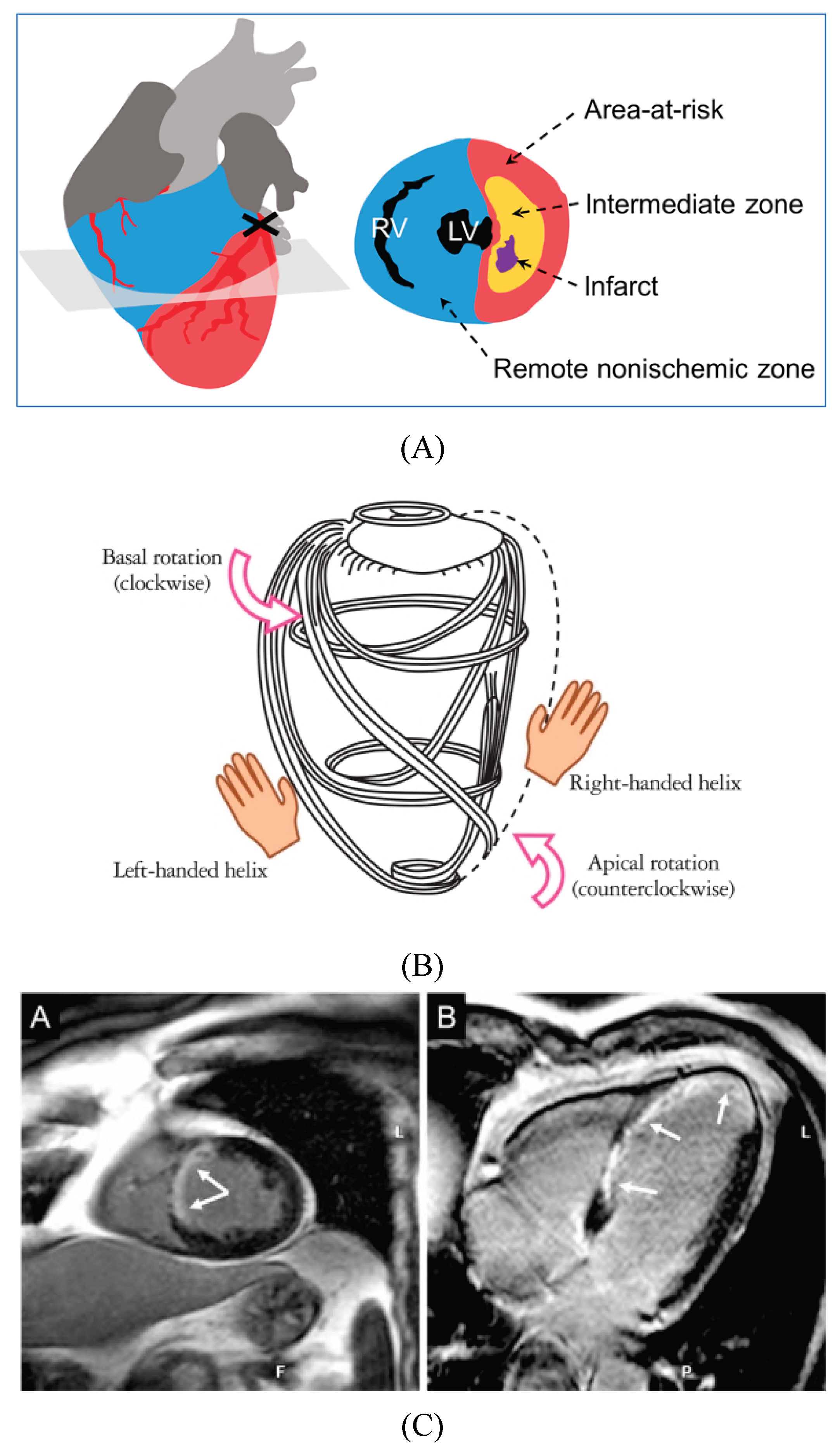
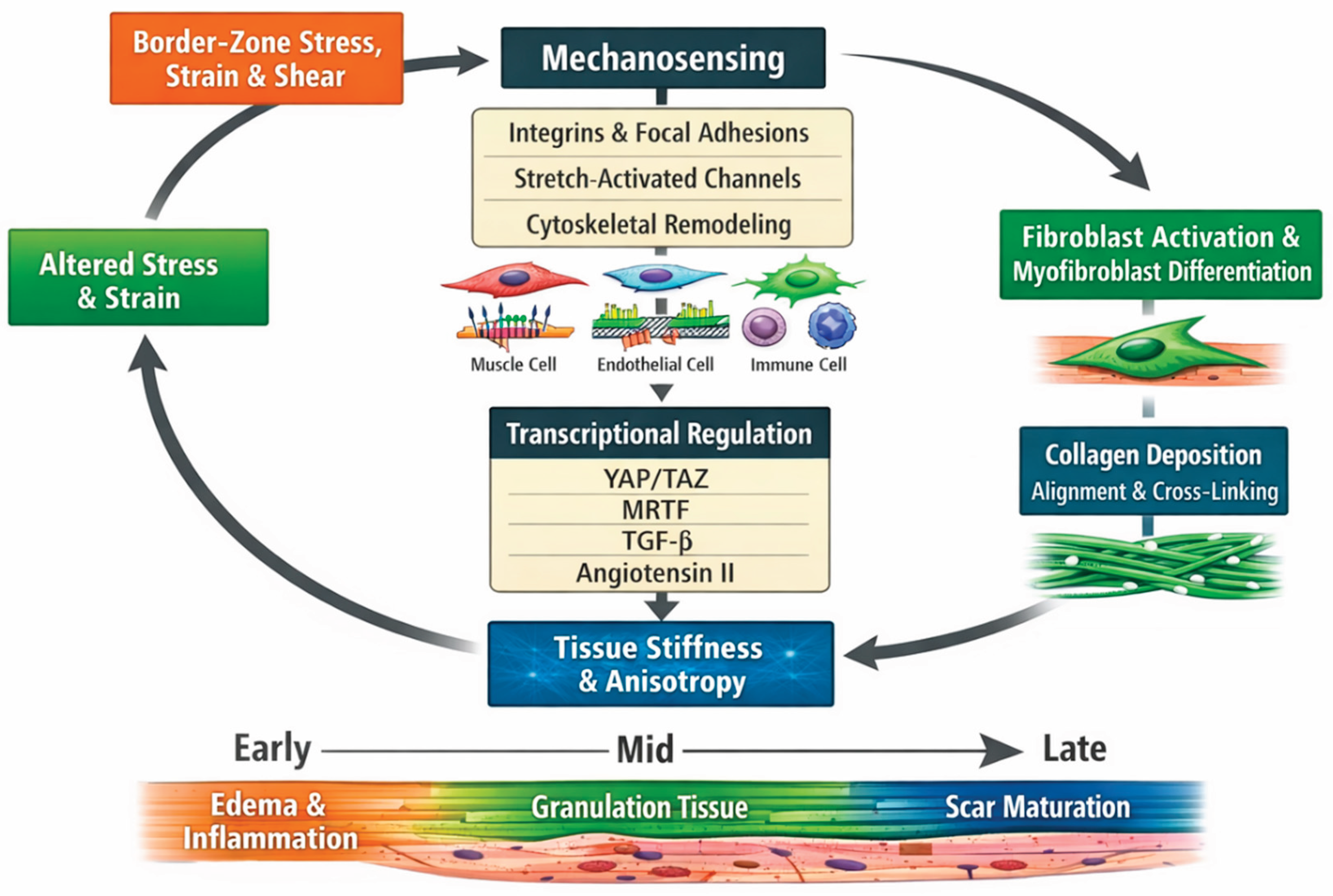
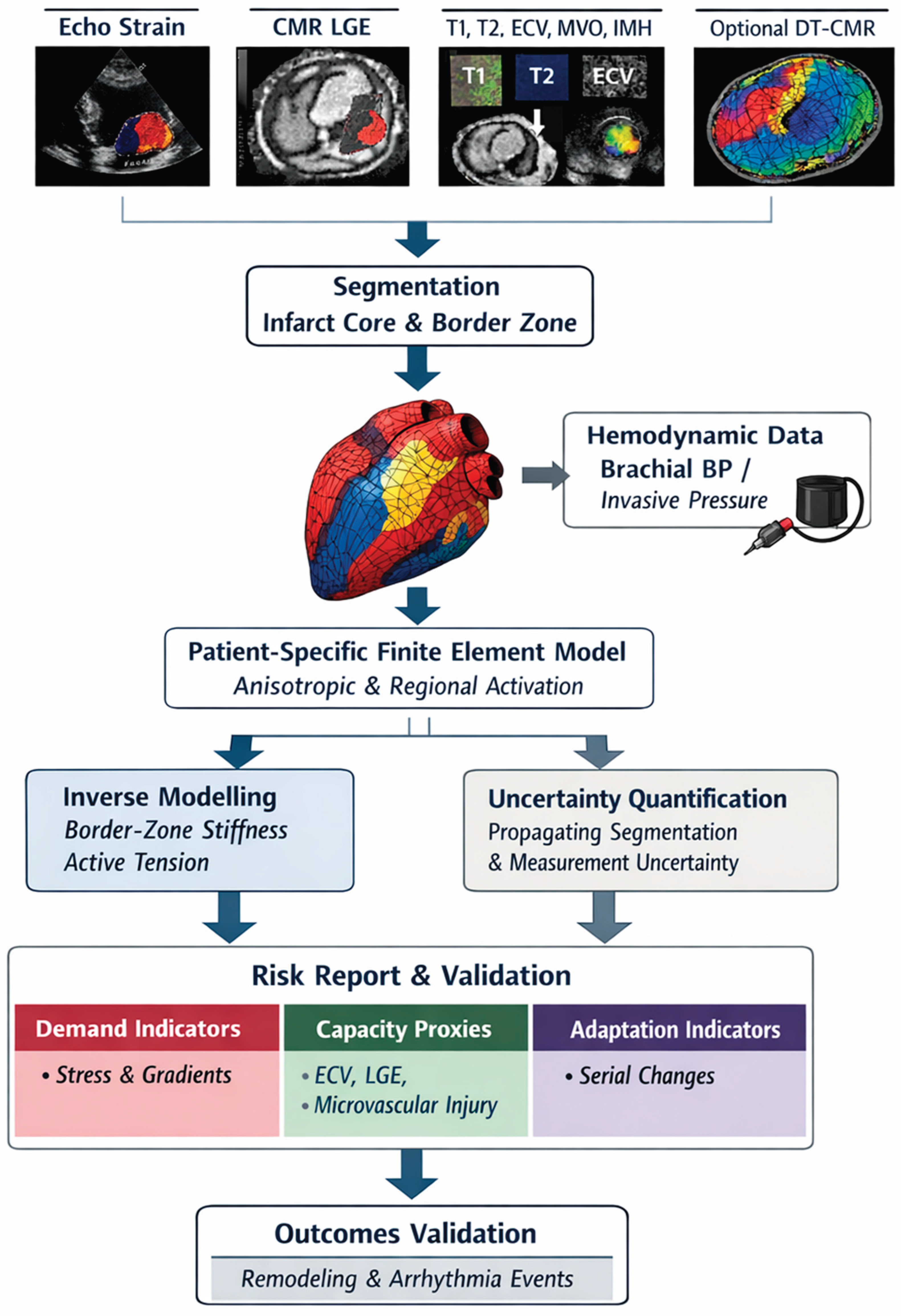
| Approach | Operational border-zone definition | What it measures most directly | What it does not measure directly | Key assumptions | Common confounders and misclassification |
|---|---|---|---|---|---|
| Histology and pathology | Peri-infarct region with mixed viability, inflammation, edema, and evolving fibrosis; often defined by staining transitions or myocyte survival islands | Cellular viability patterns, inflammation, microvascular injury, collagen content and organization at microscopic scale | In vivo stress, in vivo strain history, active tension; global load at sampling | Sampling represents in vivo state; fixation and slicing do not distort structure materially | Spatial undersampling; post-mortem changes; section orientation relative to fibers; difficulty mapping to imaging coordinates |
| Echo strain (speckle tracking) | Ring or band of reduced strain or delayed mechanical timing adjacent to infarcted segments | Deformation and timing of regional shortening under current loading conditions | Stress, stiffness, collagen alignment, and viability; separates contractility from loading imperfectly | Tracking accuracy and segmentation are adequate; frame rate captures timing; loading is similar across patients | Image quality and noise; heart rate and blood pressure; tethering and translational motion; vendor algorithm differences |
| CMR LGE | Intermediate signal intensity (“gray zone”) between high-intensity core and normal remote myocardium, using a chosen thresholding method | Relative gadolinium distribution reflecting extracellular space and kinetics at acquisition | Fiber architecture, stiffness, strength; definitive viability at cellular scale; mechanical demand | Thresholds produce consistent tissue classes; spatial resolution sufficient to resolve thin layers | Partial volume, motion, and inversion-time selection; threshold method variability; remodeling changes geometry over time |
| CMR T1/T2 mapping and ECV | Spatial transitions in T1, T2, or ECV around infarct or within peri-infarct region | Edema-related water content and diffuse fibrosis proxies, depending on sequence and time after MI | Direct collagen alignment and cross-linking; stress; active tension | Sequence calibration stable; hematocrit and field effects corrected; maps reflect tissue rather than artifacts | Heart rate dependence; field inhomogeneity; hematocrit uncertainty; overlap of edema and fibrosis signals early after MI |
| DT-CMR or microstructure imaging | Regions of altered fiber orientation, reduced fractional anisotropy, or increased dispersion near infarct edge | Microstructural orientation and disarray metrics at voxel scale | Stress and strength; functional loading response | Diffusion encoding captures myofiber orientation despite motion; post-processing robust | Limited availability; long acquisition; resolution and motion sensitivity; interpretation varies with model choice |
| Computational modeling (forward FE) | Property and activation gradient between infarct and remote myocardium, often informed by imaging intensity transitions | Consequences of assumed heterogeneity for stress, strain, and load transfer | True patient-specific properties unless calibrated; microvascular injury; molecular pathways | Constitutive law and boundary conditions are adequate; assigned gradients represent reality | Parameter sensitivity; non-uniqueness; segmentation uncertainty; neglect of residual stress or pericardial constraint |
| Inverse modeling and data assimilation | Border zone as region where inferred stiffness or active tension deviates from remote and changes smoothly toward infarct core | Estimated spatial variation in material parameters consistent with observed kinematics and pressures | Microscopic collagen features; unique identification without uncertainty; damage state | Measurements sufficiently informative; regularization does not dominate; priors appropriate | Identifiability limits; pressure estimation error; temporal mismatch; dependence on model form and smoothing |
| Determinant | Biomechanical mechanism in the border zone | Expected effect on stress and shear | Expected effect on fibrosis architecture | Key measurement proxies |
|---|---|---|---|---|
| Infarct thinning and early expansion | Reduces local thickness and increases local radius, amplifying pressure-driven demand and creating thickness gradients at infarct edges | Increases local stress and stress gradients; increases interface shear due to mismatch in deformation | Promotes aligned reinforcement along principal stretches; may increase replacement fibrosis at highly strained interfaces | CMR wall thickness, regional bulging; echo regional dyskinesia; serial geometry changes |
| Curvature changes and junction geometry | Creates notch-like regions at transitions between bulging scar and remote wall; concentrates stress at geometric discontinuities | Increases peak stress near junction; localizes shear to border-zone layers | Biases collagen alignment toward junction stress directions; may create patchy reinforcement and residual stress | CMR short-axis curvature indices; 3D shape models; strain gradients near scar edge |
| Stiffness gradient between infarct and remote tissue | Redistributes load depending on relative stiffness and geometry; can shift stress to stiffer regions and strain to softer regions | Can increase stress concentration at gradient; can increase shear if layers deform differentially | Shapes collagen organization via mechanoregulated deposition; may create anisotropic scar and heterogeneous interstitial fibrosis | Mapping-based ECV gradients; model-inferred stiffness; shear strain estimates from 3D strain imaging |
| Activation heterogeneity and depressed border-zone contractility | Remote contraction imposes traction on weakly contracting border zone; creates tethering and timing abnormalities | Increases cyclic shear and stress gradients; promotes post-systolic deformation | Promotes mechanosensitive signaling and myofibroblast activation; may reinforce regions with sustained stretch | Echo strain timing and dispersion; CMR feature tracking; electromechanical mapping where available |
| Microvascular obstruction and intramyocardial hemorrhage | Creates mechanically heterogeneous zones with edema and disrupted matrix; impairs perfusion and repair | Amplifies local heterogeneity and concentrates stress and shear at lesion boundaries | Associated with disorganized remodeling and persistent inflammation, potentially yielding patchy fibrosis and fragile interfaces | CMR MVO on early enhancement; IMH on T2*; T2/T1 mapping signatures |
| Myofiber and sheet disarray near infarct edge | Alters anisotropic load transmission and increases dispersion of principal strain directions | Increases local shear and nonuniform stress; reduces mechanical efficiency | Promotes heterogeneous collagen alignment and possible conduction heterogeneity via structural discontinuities | DT-CMR dispersion metrics; histology; strain directionality from 3D imaging |
| Residual stress and scar compaction | Changes unloaded configuration and alters stress distribution under physiological load | Can either homogenize or localize stress depending on distribution; affects border-zone stress gradients | Influences long-term collagen alignment and may stabilize or destabilize interfaces | Model calibration to measured deformation; serial geometry; indirect inference from remodeling patterns |
| Metric | Primary mechanical role | Plausible mechanistic link | Likely confounders | Clinical endpoints often studied | Validation design needed for prediction |
|---|---|---|---|---|---|
| Border-zone strain gradient (echo or CMR feature tracking) | Demand indicator | Higher gradients suggest stronger tethering and stress concentration; likely increases mechanosensing stimulus for fibrosis | Loading conditions, segmentation alignment to scar edge, tracking noise | Adverse remodeling, heart failure hospitalization | Prospective standardized imaging with controlled loading and scar-edge alignment; incremental prediction beyond infarct size |
| Mechanical dispersion (timing heterogeneity of peak strain) | Demand indicator with electromechanical coupling | Timing heterogeneity implies heterogeneous stress and shear and may correlate with arrhythmogenic substrate | Heart rate, conduction abnormalities, vendor algorithm differences | Arrhythmias, ICD therapies, remodeling | Prospective cohorts with electrical covariates; mechanistic linkage to shear or model-derived stress |
| LGE-defined gray zone extent | Capacity proxy and substrate marker | Intermediate intensity may reflect heterogeneous fibrosis and surviving myocytes; relates to anisotropic load transfer | Threshold method, partial volume, inversion time selection | Arrhythmias, mortality | Harmonized gray-zone definition and histologic or mapping validation; robust reproducibility |
| ECV gradient across scar edge (mapping) | Capacity proxy | ECV increases with matrix expansion and fibrosis; gradients may indicate evolving scar–border transition and stiffness gradient | Hematocrit, sequence variability, heart rate dependence | Remodeling, diastolic dysfunction | Standardized mapping protocols; serial studies to link ECV changes to mechanical changes and outcomes |
| T2 or edema mapping in peri-infarct region | Damage marker and early capacity modifier | Edema alters apparent stiffness and reflects inflammatory milieu that can weaken matrix continuity | Sequence differences, motion, timing after MI | Early remodeling, infarct expansion | Time-resolved studies that separate edema from fibrosis; integration with deformation measures |
| MVO and IMH presence and extent | Damage marker with capacity implications | Severe microvascular injury disrupts repair, creates heterogeneous mechanics, and predicts maladaptive remodeling | Timing of acquisition, contrast kinetics, T2* sensitivity | Adverse remodeling, heart failure, mortality | Prospective stratification and interaction analyses with mechanical metrics; mechanistic endpoints on healing trajectories |
| Model-inferred border-zone active tension fraction | Capacity proxy for contractile reserve | Lower active tension increases tethering and stress gradients; reflects surviving myocyte functional integrity | Pressure estimation error, identifiability, model form | Functional recovery, remodeling | Calibration to imaging and pressure with uncertainty bounds; independent validation against functional endpoints |
| Model-derived border-zone stress or shear index | Demand indicator | Estimated stress and shear integrate geometry, activation, and stiffness gradients; plausible driver of mechanotransduction | Model assumptions, boundary conditions, residual stress | Remodeling, arrhythmias | Benchmarking across modeling pipelines; prospective prediction with uncertainty-aware reporting |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





