Submitted:
26 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Clarification of Research Questions and Study Objectives
- –
- What is the role of cytokine and interleukin networks in the pathophysiology AUD?
- –
- Specifically, how do alcohol consumption and abstinence modulate pro-inflammatory and anti-inflammatory signalling?
- –
- What are the mechanistic pathways linking immune dysregulation to AUD-related neurobiology and clinical phenotypes?
- –
- What are the sources of heterogeneity in immune responses among individuals with AUD?
- –
- What are the translational implications of immune dysregulation in AUD?
- –
- Can cytokines and interleukins serve as biomarkers for disease progression, treatment response, or risk stratification in AUD?
- To synthesize and critically appraise current evidence on the role of cytokines and interleukins in AUD.
- To identify dynamic immune signatures that vary across different stages of AUD, including active drinking, withdrawal, and abstinence.
- To highlight potential biomarkers and immunomodulatory therapeutic strategies that could improve clinical outcomes in AUD.
- To identify research gaps and propose future priorities for advancing the understanding of immune dysregulation in AUD.
2. Materials and Methods
3. Results
3.1. Cytokine and Interleukin Networks in Individuals with Alcohol Use Disorder (AUD)
3.2. Changes in Cytokine Levels Across Different Stages of Alcohol Use Disorder (AUD)
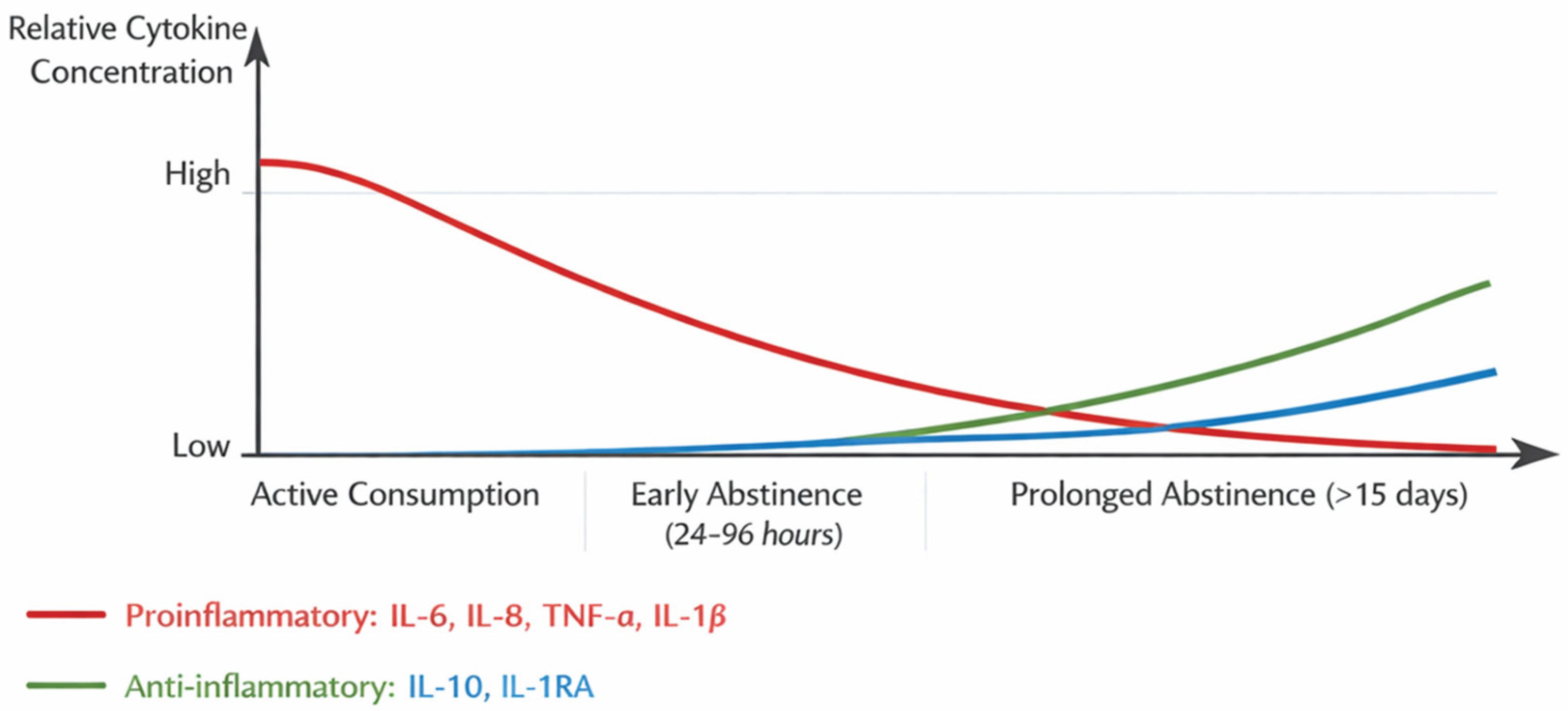
3.3. Associations of Clinical Severity and Affective Symptoms with Inflammatory and Neurotrophic Markers in Individuals with Alcohol Use Disorder (AUD)
3.4. Main Limitations and Future Lines of Research
4. Discussion
4.1. Clinical Implications
4.2. Future Lines of Research
- –
- Longitudinal study designs: Conduct extended follow-up studies to monitor the temporal evolution of cytokine and interleukin levels during different stages of AUD, including active drinking, withdrawal, and sustained abstinence. These studies should incorporate repeated biomarker assessments at multiple time points to capture dynamic changes in immune profiles.
- –
- Advanced methodologies: Utilize cutting-edge techniques such as single-cell RNA sequencing and proteomics to investigate cytokine interactions and immune cell-specific responses. These methods can provide deeper insights into the molecular mechanisms driving immune dysregulation in AUD.
- –
- Interdisciplinary collaborations: Establish partnerships between psychologist, psychiatrists, addiction specialists, immunologists, neuroscientists, and bioinformaticians to develop integrated models of AUD pathophysiology. Collaborative efforts can facilitate the synthesis of data across immune, neurobiological, and behavioural domains, enabling a more holistic understanding of the disorder.
- –
- Clinical trials for biomarker validation: Design randomized controlled trials to evaluate the utility of cytokines and interleukins as biomarkers for AUD diagnosis, treatment response, and relapse prediction. These trials should include diverse patient populations, such as individuals with varying levels of alcohol consumption and comorbid conditions.
- –
- Development of targeted interventions: Investigate the efficacy of immunomodulatory therapies, such as cytokine inhibitors or anti-inflammatory agents, in mitigating the pro-inflammatory state associated with AUD. Additionally, explore the potential of lifestyle interventions, including dietary modifications and exercise, to improve immune function and clinical outcomes.
- –
- Global and population-based studies: Expand research to include diverse populations across different geographic regions and cultural contexts. This approach will help identify population-specific immune signatures and inform tailored interventions.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Global status report on alcohol and health and treatment of substance use disorders; World Health Organization: Geneva, Switzerland, 2024; Available online: https://www.who.int (accessed on 21 February 2026).
- World Health Organization. Alcohol (Fact sheet); World Health Organization: Geneva, Switzerland, 2024; Available online: https://www.who.int (accessed on 21 February 2026).
- Karoly, H.C.; Kirk-Provencher, K.T.; Schacht, J.P.; Gowin, J.L. Alcohol and brain structure across the lifespan: A systematic review of large-scale neuroimaging studies. Addict. Biol. 2024, 29, e13439. [Google Scholar] [CrossRef]
- Quelch, D.; Roderique-Davies, G.; John, B. Alcohol-related brain damage: An umbrella (term) for the approaching post-COVID monsoon. Future Healthc. J. 2023, 10, 313–320. [Google Scholar] [CrossRef]
- Clergue-Duval, V.; Barré, T.; Cognat, E.; et al. Patients with severe alcohol-related cognitive impairment improve in flexibility when abstinence is maintained: A comparative study with Alzheimer’s disease. Front. Psychol. 2022, 13, 936639. [Google Scholar] [CrossRef] [PubMed]
- Rutt, L.N.; Liu, M.; Melamed, E.; et al. Emerging concepts in alcohol, infection & immunity: A summary of the 2023 Alcohol and Immunology Research Interest Group (AIRIG) meeting. Alcohol 2024, 11, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Tyler, R.E.; Vizioli, C.; Barb, J.J.; Farokhnia, M.; Leggio, L. Circulating immune and endocrine markers in currently drinking and abstinent individuals with alcohol use disorder and controls. Addict. Biol. 2025, 30, e70039. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. IARC Handbooks of Cancer Prevention Volume 20A: Reduction or cessation of alcohol consumption (FAQ). International Agency for Research on Cancer: Lyon, France, 2024; Available online: https://www.iarc.who.int (accessed on 21 February 2026).
- Melkumyan, M.; Randall, P.A.; Silberman, Y. Central amygdala neuroimmune signaling in alcohol use disorder. Addict. Neurosci. 2025, 14, 100194. [Google Scholar] [CrossRef]
- Ye, J.-H.; Zuo, W.; Chaudhry, F.; Chinn, L. Neuroimmune mechanisms in alcohol use disorder: Microglial modulation and therapeutic horizons. Psychoactives 2025, 4, 33. [Google Scholar] [CrossRef]
- Rasool, A.E.; Furlong, T.; Prasad, A.A. Microglia activity in the human basal ganglia is altered in alcohol use disorder and reversed with remission from alcohol. Addict. Biol. 2024, 29, e13374. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.H.; Pillai, S. Cellular and Molecular Immunology, 10th ed.; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Achur, R.N.; Freeman, W.M.; Vrana, K.E. Circulating cytokines as biomarkers of alcohol abuse and alcoholism. J. Neuroimmune Pharmacol. 2010, 5, 83–91. [Google Scholar] [CrossRef]
- Chatzipanagiotou, S.; Kalykaki, M.; Tzavellas, E.; Karaiskos, D.; Paparrigopoulos, T.; Liappas, A.; et al. Alteration of biological markers in alcohol-dependent individuals without liver disease during the detoxification therapy. In Vivo 2010, 24, 325–328. [Google Scholar]
- Moura, H.F.; Hansen, F.; Galland, F.; Silvelo, D.; Rebelatto, F.P.; Ornell, F.; et al. Inflammatory cytokines and alcohol use disorder: Systematic review and meta-analysis. Braz. J. Psychiatry 2022, 44(5), 548–556. [Google Scholar] [CrossRef]
- Crotty, K.; Anton, P.; Coleman, L.G.; Morris, N.L.; Lewis, S.A.; Samuelson, D.R.; McMahan, R.H.; Hartmann, P.; Kim, A.; Ratna, A.; Mandrekar, P.; Wyatt, T.A.; Choudhry, M.A.; Kovacs, E.J.; McCullough, R.; Yeligar, S.M. A critical review of recent knowledge of alcohol’s effects on the immunological response in different tissues. Alcohol. Clin. Exp. Res. 2023, 47, 36–44. [Google Scholar] [CrossRef]
- Tharmalingam, J.; Gangadaran, P.; Rajendran, R.L.; Ahn, B.-C. Impact of alcohol on inflammation, immunity, infections, and extracellular vesicles in pathogenesis. Cureus 2024, 16, e56923. [Google Scholar] [CrossRef]
- Crews, F.T.; Macht, V.A.; Vetreno, R.P. Epigenetic regulation of microglia and neurons by proinflammatory signaling following adolescent intermittent ethanol (AIE) exposure and in human AUD. Adv. Drug Alcohol Res. 2024, 4, 12094. [Google Scholar] [CrossRef]
- Ramos, A.; Joshi, R.S.; Szabo, G. Innate immune activation: Parallels in alcohol use disorder and Alzheimer’s disease. Front. Mol. Neurosci. 2022, 15, 910298. [Google Scholar] [CrossRef]
- Chen, G.; Shi, F.; Yin, W.; Guo, Y.; Liu, A.; Shuai, J.; Sun, J. Gut microbiota dysbiosis: The potential mechanisms by which alcohol disrupts gut and brain functions. Front. Microbiol. 2022, 13, 916765. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Jee, Y.-M.; Yang, K.; Ryu, T. Alcohol-induced oxidative stress and gut–liver–brain crosstalk: Expanding the paradigm from ALD to MetALD. Antioxidants 2025, 14, 1196. [Google Scholar] [CrossRef] [PubMed]
- Janicova, A.; Haag, F.; Xu, B.; Garza, A.P.; Dunay, I.R.; Neunaber, C.; Nowak, A.J.; Cavalli, P.; Marzi, I.; Sturm, R.; Relja, B. Acute alcohol intoxication modulates monocyte subsets and their functions in a time-dependent manner in healthy volunteers. Front. Immunol. 2021, 12, 652488. [Google Scholar] [CrossRef] [PubMed]
- García-Marchena, N.; Araos, P.F.; Barrios, V.; et al. Plasma chemokines in patients with alcohol use disorders: Association of CCL11 (Eotaxin-1) with psychiatric comorbidity. Front. Psychiatry 2017, 7, 214. [Google Scholar] [CrossRef] [PubMed]
- Grodin, E.N.; Baskerville, W.A.; McManus, K.R.; Irwin, M.R.; Ray, L.A. Elevations in interleukin-8 levels in individuals with alcohol use disorder and clinical insomnia symptoms. Alcohol. Clin. Exp. Res. 2024, 48, 2079–2088. [Google Scholar] [CrossRef]
- Bramness, J.G.; Pandey, S.; Moe, J.S.; Toft, H.; Lien, L.; Walby, F.A.; Myhre, M.Ø.; Bolstad, I. Levels of IL-6 are associated with lifetime attempted suicide in alcohol use disorder patients. Neuropsychiatr. Dis. Treat. 2023, 19, 2141–2148. [Google Scholar] [CrossRef]
- Balan, I.; Lopez, A.G.; Gilmore, T.; Bremmer, M.; O’Buckley, T.K.; Xia, K.; Hendershot, C.S.; Morrow, A.L. Identification of interleukin-1β in whole blood as a candidate biomarker for alcohol use disorder risk based on AUDIT scores. Addict. Biol. 2025, 30, e70088. [Google Scholar] [CrossRef]
- García-Calvo, X.; et al. Significance of markers of monocyte activation (CD163 and sCD14) and inflammation (IL-6) in patients admitted for alcohol use disorder treatment. Alcohol. Clin. Exp. Res. 2020, 44, 152–158. [Google Scholar] [CrossRef]
- Tyler, R.E.; Kim, S.W.; Guo, M.; Jang, Y.J.; Damadzic, R.; Stodden, T.; Vendruscolo, L.F.; Koob, G.F.; Wang, G.-J.; Wiers, C.E.; Volkow, N.D. Detecting neuroinflammation in the brain following chronic alcohol exposure in rats: A comparison between in vivo and in vitro TSPO radioligand binding. Eur. J. Neurosci. 2019, 50, 1831–1842. [Google Scholar] [CrossRef]
- Nikou, T.; Ioannidis, A.; Zoga, M.; Tzavellas, E.; Paparrigopoulos, T.; Magana, M.; Pliatsika, P.; Nikolaou, C.; Chatzipanagiotou, S. Alteration in the concentrations of interleukin-7 (IL-7), interleukin-10 (IL-10) and granulocyte colony stimulating factor (G-CSF) in alcohol-dependent individuals without liver disease, during detoxification therapy. Drug Alcohol Depend. 2016, 163, 77–83. [Google Scholar] [CrossRef]
- Papantoniou, M.; Chatzipanagiotou, S.; Kokotis, P.; Nikolaou, C.; Gargalionis, A.; Tzavellas, E.; Paparrigopoulos, T.; Rentzos, M. Interleukin-1 beta and Interleukin-6 serum concentrations correlate with neuropathy and liver enzyme levels in patients diagnosed with alcohol use disorder. J. Neuroimmunol. 2025, 378, 578599. [Google Scholar] [CrossRef]
- Coller, J.K.; Hutchinson, M.R. Implications of central immune signaling caused by drugs of abuse: Mechanisms, mediators and new therapeutic approaches for prediction and treatment of drug dependence. Pharmacol. Ther. 2012, 134, 219–245. [Google Scholar] [CrossRef]
- Gan, X.; Zhang, L.; Newton, T.; Chang, S.L.; Ling, W.; Kermani, V.; Berger, O.; Graves, M.C.; Fiala, M. Cocaine infusion increases interferon-gamma and decreases interleukin-10 in cocaine-dependent subjects. Clin. Immunol. Immunopathol. 1998, 89, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Historical insights into cytokines. Eur. J. Immunol. 2007, 37, S34–S45. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A. The blood-brain barrier in neuroimmunology: Tales of separation and assimilation. Brain Behav. Immun. 2015, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Pascual, M.; Baliño, P.; Aragón, C.M.G.; Guerri, C. Cytokines and chemokines as biomarkers of ethanol-induced neuroinflammation and anxiety-related behavior: Role of TLR4 and TLR2. Neuropharmacology 2015, 89, 352–359. [Google Scholar] [CrossRef]
- Crews, F.T.; Vetreno, R.P. Neuroimmune basis of alcoholic brain damage. Int. Rev. Neurobiol. 2014, 118, 315–357. [Google Scholar] [CrossRef]
- Crews, F.T.; Lawrimore, C.J.; Walter, T.J.; Coleman, L.G., Jr. The role of neuroimmune signaling in alcoholism. Neuropharmacology 2017, 122, 56–73. [Google Scholar] [CrossRef] [PubMed]
- Hillmer, A.T.; Sandiego, C.M.; Hannestad, J.; Angarita, G.A.; Kumar, A.; McGovern, E.M.; Huang, Y.; O’Connor, K.C.; Carson, R.E.; O’Malley, S.S.; Cosgrove, K.P. In vivo imaging of translocator protein, a marker of activated microglia, in alcohol dependence. Mol. Psychiatry 2017, 22, 1759–1766. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Wiers, C.E.; Tyler, R.; Shokri-Kojori, E.; Jang, Y.J.; Zehra, A.; Freeman, C.; Ramirez, V.; Lindgren, E.; Miller, G.; Cabrera, E.A.; Stodden, T.; Guo, M.; Demiral, Ş.B.; Diazgranados, N.; Park, L.; Liow, J.-S.; Pike, V.; Morse, C.; Vendruscolo, L.F.; Innis, R.B.; Koob, G.F.; Tomasi, D.; Wang, G.-J.; Volkow, N.D. Influence of alcoholism and cholesterol on TSPO binding in brain: PET [11C]PBR28 studies in humans and rodents. Neuropsychopharmacology 2018, 43, 1832–1839. [Google Scholar] [CrossRef]
- Yen, C.-H.; Ho, P.-S.; Yeh, Y.-W.; Liang, C.-S.; Kuo, S.-C.; Huang, C.-C.; Chen, C.-Y.; Shih, M.-C.; Ma, K.-H.; Huang, S.-Y. Differential cytokine levels between early withdrawal and remission states in patients with alcohol dependence. Psychoneuroendocrinology 2017, 76, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Adams, C.; Conigrave, J.H.; Lewohl, J.; Haber, P.; Morley, K.C. Alcohol use disorder and circulating cytokines: A systematic review and meta-analysis. Brain Behav. Immun. 2020, 89, 501–512. [Google Scholar] [CrossRef]
- Neupane, S.P. Neuroimmune interface in the comorbidity between alcohol use disorder and major depression. Front. Immunol. 2016, 7, 655. [Google Scholar] [CrossRef]
- González-Quintela, A.; Vidal, C.; Lojo, S.; Pérez, L.F.; Otero-Antón, E.; Gude, F.; Barrio, E. Serum cytokines and increased total serum IgE in alcoholics. Ann. Allergy Asthma Immunol. 1999, 83, 61–67. [Google Scholar] [CrossRef]
- Crews, F.T.; Sarkar, D.K.; Qin, L.; Zou, J.; Boyadjieva, N.; Vetreno, R.P. Neuroimmune function and the consequences of alcohol exposure. Alcohol Res. Curr. Rev. 2015, 37, 331–341. [Google Scholar] [CrossRef]
- Leclercq, S.; de Timary, P.; Delzenne, N.M.; Stärkel, P. The link between inflammation, bugs, the intestine and the brain in alcohol dependence. Transl. Psychiatry 2017, 7, e1048. [Google Scholar] [CrossRef]
- Heberlein, A.; Käser, M.; Lichtinghagen, R.; Rhein, M.; Lenz, B.; Kornhuber, J.; Bleich, S. TNF-α and IL-6 serum levels: Neurobiological markers of alcohol consumption in alcohol-dependent patients? Alcohol 2014, 48, 671–676. [Google Scholar] [CrossRef]
- Leclercq, S.; Cani, P.D.; Neyrinck, A.M.; et al. Role of intestinal permeability and inflammation in the biological and behavioral control of alcohol-dependent subjects. Brain Behav. Immun. 2012, 26, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Costello, E.J.; Copeland, W.E.; Shanahan, L.; Worthman, C.M.; Angold, A. C-reactive protein and substance use disorders in adolescence and early adulthood: A prospective analysis. Drug Alcohol Depend. 2013, 133, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Manzardo, A.M.; Poje, A.B.; Penick, E.C.; Butler, M.G. Multiplex immunoassay of plasma cytokine levels in men with alcoholism and the relationship to psychiatric assessments. Int. J. Mol. Sci. 2016, 17, 472. [Google Scholar] [CrossRef]
- Agarwal, K.; Manza, P.; Chapman, M.; Nawal, N.; Biesecker, E.; McPherson, K.; Dennis, E.; Johnson, A.; Volkow, N.D.; Joseph, P.V. Inflammatory Markers in Substance Use and Mood Disorders: A Neuroimaging Perspective. Front. Psychiatry 2022, 13, 863734. [Google Scholar] [CrossRef] [PubMed]
- Decker Ramirez, E.B.; Schank, J.R. Sex Differences in Inflammatory Mechanisms Related to Alcohol Use Disorder and Depression. Brain Behav. Immun. 2026, 131, 106172. [Google Scholar] [CrossRef]
- Kazmi, N.; Wallen, G.R.; Yang, L.; Alkhatib, J.; Schwandt, M.L.; Feng, D.; Gao, B.; Diazgranados, N.; Ramchandani, V.A.; Barb, J.J. An Exploratory Study of Pro-Inflammatory Cytokines in Individuals with Alcohol Use Disorder: MCP-1 and IL-8 Associated with Alcohol Consumption, Sleep Quality, Anxiety, Depression, and Liver Biomarkers. Front. Psychiatry 2022, 13, 931280. [Google Scholar] [CrossRef]
- Shafiee, A.; Jafarabady, K.; Rafiei, M.A.; et al. Effect of Alcohol on Brain-Derived Neurotrophic Factor (BDNF) Blood Levels: A Systematic Review and Meta-Analysis. Sci. Rep. 2023, 13, 17554. [Google Scholar] [CrossRef]
- Gellé, T.; Vinais, T.; Lacroix, A.; Plansont, B.; Nubukpo, P.; Girard, M. Serum BDNF and pro-BDNF Levels in Alcohol Use Disorders According to Depression Status: An Exploratory Study of Their Evolution Two Months After Withdrawal. Heliyon 2024, 10, e38940. [Google Scholar] [CrossRef]
- Goldsmith, D.R.; Rapaport, M.H.; Miller, B.J. A meta-analysis of blood cytokine network alterations in psychiatric patients: Comparisons between schizophrenia, bipolar disorder and depression. Mol. Psychiatry 2016, 21, 1696–1709. [Google Scholar] [CrossRef]
- Quagliato, L.A.; Nardi, A.E. Cytokine alterations in panic disorder: A systematic review. J. Affect. Disord. 2018, 228, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Martinez, P.; Lien, L.; Zemore, S.; et al. Circulating cytokine levels are associated with symptoms of depression and anxiety among people with alcohol and drug use disorders. J. Neuroimmunol. 2018, 318, 80–86. [Google Scholar] [CrossRef] [PubMed]
- García-Marchena, N.; et al. Abstinent patients with alcohol use disorders show an altered plasma cytokine profile: Identification of both interleukin 6 and interleukin 17A as potential biomarkers of consumption and comorbid liver and pancreatic diseases. J. Psychopharmacol. 2020, 34, 1250–1260. [Google Scholar] [CrossRef] [PubMed]
- Sturm, R.; Haag, F.; Janicova, A.; et al. Acute alcohol consumption increases systemic endotoxin bioactivity for days in healthy volunteers—With reduced intestinal barrier loss in female. Eur. J. Trauma Emerg. Surg. 2022, 48, 1569–1577. [Google Scholar] [CrossRef]
- Sokolova, S.; Skryabin, V.; Pozdniakov, S.; Ivanchenko, V.; Vinokurova, N.; Ardamatskaya, L.; Masyakin, A. Phase-Dependent Cytokine Signatures in Alcohol Use Disorder: A Systematic Review of Psychoneuroimmune Links to Withdrawal Severity and Return to Drinking. Psychoneuroendocrinology 2026, 186, 107771. [Google Scholar] [CrossRef]
- Donnadieu-Rigole, H.; Pansu, N.; Mura, T.; Pelletier, S.; Alarcon, R.; Gamon, L.; Perney, P.; Apparailly, F.; Lavigne, J.-P.; Dunyach-Remy, C. Beneficial Effect of Alcohol Withdrawal on Gut Permeability and Microbial Translocation in Patients with Alcohol Use Disorder. Alcohol. Clin. Exp. Res. 2018, 42, 32–40. [Google Scholar] [CrossRef]
| Category | Marker | Description |
|---|---|---|
| Pro-inflammatory cytokines. | IL-6. | Associated with pro- and anti-inflammatory activities; linked to the pathology of alcohol-induced liver disease. |
| Pro-inflammatory cytokines. | IL-7. | Contributes to the inflammatory response. |
| Pro-inflammatory cytokines. | IL-8. | Related to inflammatory processes and tissue injury. |
| Pro-inflammatory cytokines. | TNF-α. | Implicated in inflammation and liver damage in AUD. |
| Anti-inflammatory cytokines. | IL-10. | Has anti-inflammatory properties; concentrations may fluctuate depending on the stage of AUD. |
| Other markers. | IL-1β. | Related to inflammation and neuronal injury. |
| Other markers. | IL-12. | Associated with immune responses in patients with AUD. |
| Other markers. | TGF-β1. | Linked to inflammatory processes and immunological alterations. |
| Study | Main limitations |
|---|---|
| Nikou et al. [29] |
|
| García-Marchena et al. [23] |
|
| García-Calvo et al. [27] |
|
| Tyler et al. [28] |
|
| Balan et al. [26] |
|
| Bramness et al. [25] |
|
| Grodin et al. [24] |
|
| Papantoniou et al. [30] |
|
| Clinical domain | Biomarker signal(s) | Practical application in clinical practice | Example implementation | Clinical value / expected benefit |
|---|---|---|---|---|
| Biomarker-guided diagnosis and risk stratification. | Elevated IL-6, IL-8, TNF-α, IL-1β. | Support identification of individuals at risk for AUD or in early-stage AUD as an adjunct to standard clinical assessment. | Add cytokine panel to routine laboratory work-up in patients with suspected AUD (alongside clinical interview and screening tools). | Earlier detection, improved phenotyping, and more objective risk stratification. |
| Treatment monitoring across detoxification and abstinence. | Longitudinal trajectories of IL-6, IL-10; persistent elevation of IL-8. | Track biological response to treatment and flag persistent immune activation during abstinence. | Serial measurements during detoxification and follow-up visits; persistent IL-8 elevation prompts closer monitoring and targeted supportive care. | More individualized follow-up intensity; potential early warning for ongoing inflammation and vulnerability. |
| Personalized pharmacological adjuncts. | High pro-inflammatory profile (e.g., TNF-α, IL-18; IL-1β). | Identify candidates for immunomodulatory strategies as adjuncts to standard AUD care (investigational/off-label depending on context). | Consider evaluation in clinical trials of TNF-α inhibitors or IL-1 receptor antagonists in severe AUD with marked inflammation. | Potential reduction of systemic inflammation and improvement in symptom burden or medical comorbidity (requires rigorous validation). |
| Personalized lifestyle and behavioral interventions. | Elevated inflammatory markers; gut-related inflammatory signatures. | Implement anti-inflammatory lifestyle interventions to complement standard AUD treatment. | Nutritional counseling emphasizing omega-3 fatty acids, polyphenols, and high-fiber diets; structured physical activity plans. | Reduced inflammatory tone, improved metabolic and gut health, and broader supportive effects on recovery. |
| Mental health integration within AUD care. | Associations of IL-18 and TNF-α with anxiety/depression symptoms. | Integrate psychiatric evaluation and treatment in patients with inflammatory activation and affective symptoms. | Combined care pathways: evidence-based psychotherapy (e.g., CBT, mindfulness-based interventions) plus optimized AUD treatment; consider inflammation-informed monitoring. | Improved management of comorbidity, potentially better adherence and outcomes via integrated biopsychosocial care. |
| Relapse prevention and stepped-care intensity. | Persistent inflammatory activation; reduced neuroplasticity-related markers (e.g., BDNF) | Identify patients at elevated relapse risk to escalate relapse-prevention intensity. | High-risk profiles prompt stepped-up care: residential programs, pharmacotherapy, and structured continuing care. | More targeted allocation of intensive resources; potentially improved relapse prevention. |
| Early preventive intervention before severe AUD. | Elevated IL-1β in individuals with high AUDIT scores. | Offer preventive support to high-risk individuals prior to severe AUD onset. | Use biomarker + AUDIT-informed triage to deliver motivational interviewing, brief interventions, and peer-support referral. | Earlier intervention window; potential reduction in progression to severe AUD. |
| Multidisciplinary care model development. | Multi-marker inflammatory profiles (cytokines/interleukins; neuroplasticity markers). | Coordinate integrated care addressing inflammation, neurobiology, and mental health. | Team-based management involving addiction specialists, psychologists, psychiatrists, immunology-informed input, nutrition, and physical therapy. | Holistic care, improved coordination, and more comprehensive management of comorbidities. |
| Patient education and engagement. | Biomarker-informed explanation of immune effects of alcohol. | Use biomarker results to enhance patient understanding and adherence. | Clinician-led education linking alcohol use to immune dysregulation and mental health; shared decision-making around treatment plan. | Greater engagement, adherence, and self-efficacy through personalized feedback. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




