1. Introduction
Alcohol Use Disorder (AUD) represents a major global public health and social challenge, associated with substantial morbidity and mortality [
1,
2]. Chronic and excessive alcohol consumption can result in both reversible and irreversible cognitive deficits and is linked to structural alterations in the brain [
3,
4,
5]. Moreover, alcohol abuse may also affect immune function [
6,
7] and increases susceptibility to several malignancies (i.e., cancers of the colon, upper aerodigestive tract, liver, and breast, etc.) [
2,
8].
Recent work has increasingly focused on the neurobiological underpinnings of AUD, and converging evidence suggests that neuroimmune signalling—particularly microglial activation and cytokine-mediated pathways—may contribute to both the development and persistence of the disorder, including vulnerability to relapse [
9,
10,
11]. Cytokines comprise a heterogeneous group of proteins and glycoproteins produced by multiple cell types that function primarily as key regulators of immune and inflammatory responses. In addition, they act as growth and differentiation factors for a range of cells, particularly within the hematopoietic lineage. Their central role is to mediate intercellular communication, thereby modulating the activity of a broad spectrum of target cells. Cytokines can either induce or suppress the synthesis of other cytokines and their receptors, promoting synergistic interactions or, alternatively, exerting antagonistic effects. They are also characterized by pleiotropy, reflecting their capacity to act across diverse tissues and elicit multiple biological effects [
12]. In parallel, findings from immunopsychiatry in severe mental illnesses indicate that immune and inflammatory signalling differ from healthy controls and may vary by illness stage and symptom dimensions, highlighting substantial biological heterogeneity within diagnostic categories [
9,
10,
11].
In addition, several immunological parameters—including tumour necrosis factor-α (TNF-α) and interleukins (ILs)-1β, IL-8, IL-12, and IL-13—appear to be elevated in individuals with alcohol abuse or dependence. In contrast, IL-6 concentrations usually decrease during detoxification treatment in alcohol-dependent patients without liver disease [
12,
13,
14,
15].
Immune alterations associated with alcohol use are multifactorial and vary by exposure pattern, host comorbidities, and the specific immune pathways assessed [
16,
17]. Alcohol can activate innate immunity both centrally and peripherally. In the brain, ethanol-related neuroimmune signalling is closely linked to microglial activation and downstream proinflammatory cascades implicated in neurotoxicity and AUD pathophysiology [
18,
19]. Systemically, alcohol-induced dysbiosis and increased intestinal permeability facilitate translocation of microbial products such as lipopolysaccharide (LPS) into the portal circulation, amplifying hepatic inflammation and cytokine production that can propagate inflammatory signalling to the central nervous system via the bloodstream [
20,
21]. Moreover, acute and chronic alcohol exposure can exert divergent, time-dependent immunomodulatory effects. In fact, acute intoxication may show a biphasic response with early proinflammatory changes followed by later anti-inflammatory or immunosuppressive features, whereas sustained heavy use is more consistently associated with persistent immune dysregulation and proinflammatory tone [
16,
22]. Characterizing these dynamic, context-dependent immune signatures may help clarify mechanisms underlying AUD onset, maintenance, and progression, and inform targeted intervention strategies [
16,
17].
This narrative review provides a novel and integrative perspective on the role of cytokine and interleukin networks in AUD. While previous studies have mainly explored the relationship between alcohol consumption and immune dysregulation, they often focus on isolated biomarkers or specific stages of AUD, such as active drinking or withdrawal. This study advances the field by explicitly modelling the dynamic changes in inflammatory signatures across different phases of AUD, including intoxication, withdrawal, and early abstinence. By situating peripheral immune alterations within a mechanistic gut–liver–brain framework, this review offers a systems-level understanding of how AUD is initiated and maintained through innate immune pathways.
Unlike prior research that primarily relies on cross-sectional data, this study emphasizes the relevance of temporal variability in cytokine profiles, highlighting how inflammatory markers fluctuate based on drinking patterns, recency of alcohol exposure, and the progression of AUD. This approach is expected to strengthen causal plausibility by aligning immunological findings with clinically meaningful phases of the disorder, such as early recovery and detoxification. Furthermore, the review identifies dynamic biomarkers that may provide more accurate insights into disease progression and treatment response compared to static measurements, offering a unique contribution to the field.
Based on the growing body of scientific evidence linking immune dysregulation to the pathophysiology of AUD, this review aims to provide a comprehensive synthesis of current findings while addressing critical gaps in the literature. By examining the dynamic interplay between cytokine and interleukin networks, alcohol exposure, and different phases of AUD, the study aims to elucidate the mechanistic pathways underlying immune alterations and their impact on AUD-related neurobiology and clinical outcomes. Furthermore, this review emphasizes the relevance of identifying biomarkers and therapeutic targets that can inform personalized treatment strategies. The following section outlines the specific research questions and objectives that guide this investigation, providing a structured framework for exploring the complex relationship between immune signalling and AUD.
1.1. Clarification of Research Questions and Study Objectives
This narrative review aims to address the following research questions:
- –
What is the role of cytokine and interleukin networks in the pathophysiology AUD?
- –
Specifically, how do alcohol consumption and abstinence modulate pro-inflammatory and anti-inflammatory signalling?
- –
What are the mechanistic pathways linking immune dysregulation to AUD-related neurobiology and clinical phenotypes?
- –
What are the sources of heterogeneity in immune responses among individuals with AUD?
- –
What are the translational implications of immune dysregulation in AUD?
- –
Can cytokines and interleukins serve as biomarkers for disease progression, treatment response, or risk stratification in AUD?
The primary objectives of this review are as follows:
To synthesize and critically appraise current evidence on the role of cytokines and interleukins in AUD.
To identify dynamic immune signatures that vary across different stages of AUD, including active drinking, withdrawal, and abstinence.
To highlight potential biomarkers and immunomodulatory therapeutic strategies that could improve clinical outcomes in AUD.
To identify research gaps and propose future priorities for advancing the understanding of immune dysregulation in AUD.
3. Results
The results of this narrative review are organized into thematic domains to provide a structured and comprehensive synthesis of the evidence. First, the review examines the associations between cytokine and interleukin networks and AUD, highlighting key inflammatory markers and their roles in the pathophysiology of the disorder. Second, it explores the dynamic changes in cytokine levels across different stages of AUD, including withdrawal and abstinence, to elucidate the temporal variability of immune dysregulation. Third, the review investigates the correlations between clinical severity, affective symptoms, and inflammatory markers, emphasizing the interplay between immune activation and neuropsychological outcomes. Finally, the limitations of existing studies and future research priorities are discussed to address gaps in the literature and propose directions for advancing in this field.
3.1. Cytokine and Interleukin Networks in Individuals with Alcohol Use Disorder (AUD)
Several studies have documented associations between inflammatory dysregulation and AUD [
23,
24,
25,
26,
27,
28,
29,
30]. In this regard, a history of alcohol use was associated with significant differences across all assessed cytokines. Specifically, the alcohol group exhibited higher plasma concentrations of IL-1β, IL-6, and TNF-α compared with the control group. In contrast, plasma concentrations of IL-4, IL-17A, and IFN-γ were lower in the alcohol group than in controls [
23,
24,
25].
In the same line, Balan et al. [
26] reported that IL-1β levels are significantly elevated in high-risk individuals. Specifically, IL-1β—together with other immunological mediators such as IL-18, IL-7, and CCL11—was markedly increased among participants with Alcohol Use Disorders Identification Test (AUDIT) scores ≥ 6, consistent with potential immune dysregulation and a pro-inflammatory profile associated with alcohol consumption.
García-Calvo et al. [
27] also found that inflammatory cytokines, particularly interleukin-6 (IL-6), are associated with monocyte activation and systemic inflammation in patients with AUD. Elevated IL-6 levels correlated with clinical and laboratory characteristics, including older age and higher levels of AST, GGT, mean corpuscular volume (MCV), ferritin, an erythrocyte sedimentation rate (ESR) > 20 mm, and C-reactive protein (CRP) > 5 mg/L. In addition, IL-6 levels showed negative associations with alcohol consumption, total cholesterol, and albumin, which may indicate a state of systemic inflammation and early-stage liver injury.
Moreover, Tyler et al. [
28] analysed cytokines and interleukins in relation to immune dysregulation in AUD. Authors reported that alcohol exposure appears to perturb immune function and is associated with measurable shifts in circulating inflammatory mediators. Acute alcohol intake has been reported to decrease TNF-α while increasing interleukin-6 (IL-6). In contrast, chronic alcohol use is linked to more persistent immunological alterations, including elevated concentrations of proinflammatory markers such as TNF-α, IL-6, IL-8, and IL-18, alongside changes in anti-inflammatory mediators including interleukin-10 (IL-10) and interleukin-1 receptor antagonist (IL-1RA). In addition, correlation analyses further supported clinical relevance of inflammatory signalling. Proinflammatory markers, including IL-18 and TNF-α, were positively associated with AUD severity and with measures of anxiety and depressive symptoms, indicating that greater inflammatory activation may track with both disorder severity and comorbid affective burden. Conversely, IL-8 was negatively correlated with anxiety/depression measures within the abstinent AUD group, suggesting that the relationship between inflammation and psychiatric symptoms may vary by drinking status and phase of recovery.
Grodin et al. [
24] also highlighted the role of interleukins —including IL-6, IL-7, IL-8—and TNF-α, which are consistently elevated in individuals with AUD compared with controls. These proinflammatory cytokines are associated with alcohol consumption and AUD severity, suggesting that increased alcohol intake may modulate the immune system. Moreover, the study specifically identifies elevated IL-8 levels in individuals with AUD who exhibit clinically significant insomnia symptoms, a pattern not observed for other inflammatory markers such as IL-6, TNF-α, or CRP. IL-8, a proinflammatory chemokine, appears to be linked to sleep quality and insomnia symptoms in people with AUD. The authors also note that IL-8 production in peripheral blood mononuclear cells is associated with specific inflammatory signalling pathways, such as the activator protein 1 (AP-1) transcription factor pathway, which has been related to alcohol consumption and craving.
Nikou et al. [
29] reported that across the detoxification period, IL-7 concentrations were elevated in all patients. Levels remained consistently high during the first two weeks following initiation of detoxification, after which a decline was observed; however, IL-7 concentrations remained significantly higher than those of the control group at the end of treatment. IL-10 concentrations were significantly lower than in controls at admission and exhibited a linear increase throughout detoxification; nevertheless, values remained slightly lower than those of the control group at the end of therapy. In contrast, G-CSF concentrations were significantly elevated on admission and then decreased linearly over the course of detoxification, reaching near-normal values by the end of treatment. In this study the detoxification protocol comprised an initial 10 mg intramuscular injection of diazepam at admission, followed by oral diazepam 10 mg administered four times daily. From day 4 onward, the dose was progressively tapered to 5 mg orally, and diazepam was discontinued between days 12 and 14. Throughout hospitalization, patients received vitamins B1, B6, B12, A, E, and C, as well as folate [
29].
Table 1 provides a comprehensive overview of some of the most relevant inflammatory markers associated with AUD highlighting their roles in immune dysregulation and their potential implications for disease progression and treatment.
3.2. Changes in Cytokine Levels Across Different Stages of Alcohol Use Disorder (AUD)
Cytokine levels may vary across different stages of the clinical course of AUD [
23,
27,
28,
30]. In abstinent patients with AUD, different alterations in plasma cytokine concentrations were observed relative to control subjects. Specifically, interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and TNF-α were increased, whereas interleukin-4 (IL-4), interleukin-17A (IL-17A), and interferon gamma (IFN-γ) were reduced. These findings indicate that abstinent individuals with AUD exhibit a disrupted plasma cytokine profile that may be associated with inflammation related to chronic alcohol exposure. In addition, plasma IL-6 and IL-17A concentrations were associated with the presence of comorbid hepatic and pancreatic diseases [
23].
Similarly, Tyler et al. [
28] reported that IL-8 levels remained elevated in the abstinent AUD group (AB) compared to healthy controls and currently drinking individuals. This suggests a persistent proinflammatory state in individuals with severe AUD, even after 6+ weeks of abstinence.
In addition, the study of García-Calvo et al. [
27] pointed out that prior studies have reported reductions in biomarkers of inflammation and intestinal permeability (such as sCD14 and IL-6) after approximately 2–3 weeks of alcohol abstinence. This suggests that abstinence may beneficially attenuate systemic inflammation and monocyte activation in patients with AUD. However, in their research, biomarkers were assessed only once—two days after admission—so changes in these markers following a longer period of abstinence were not evaluated.
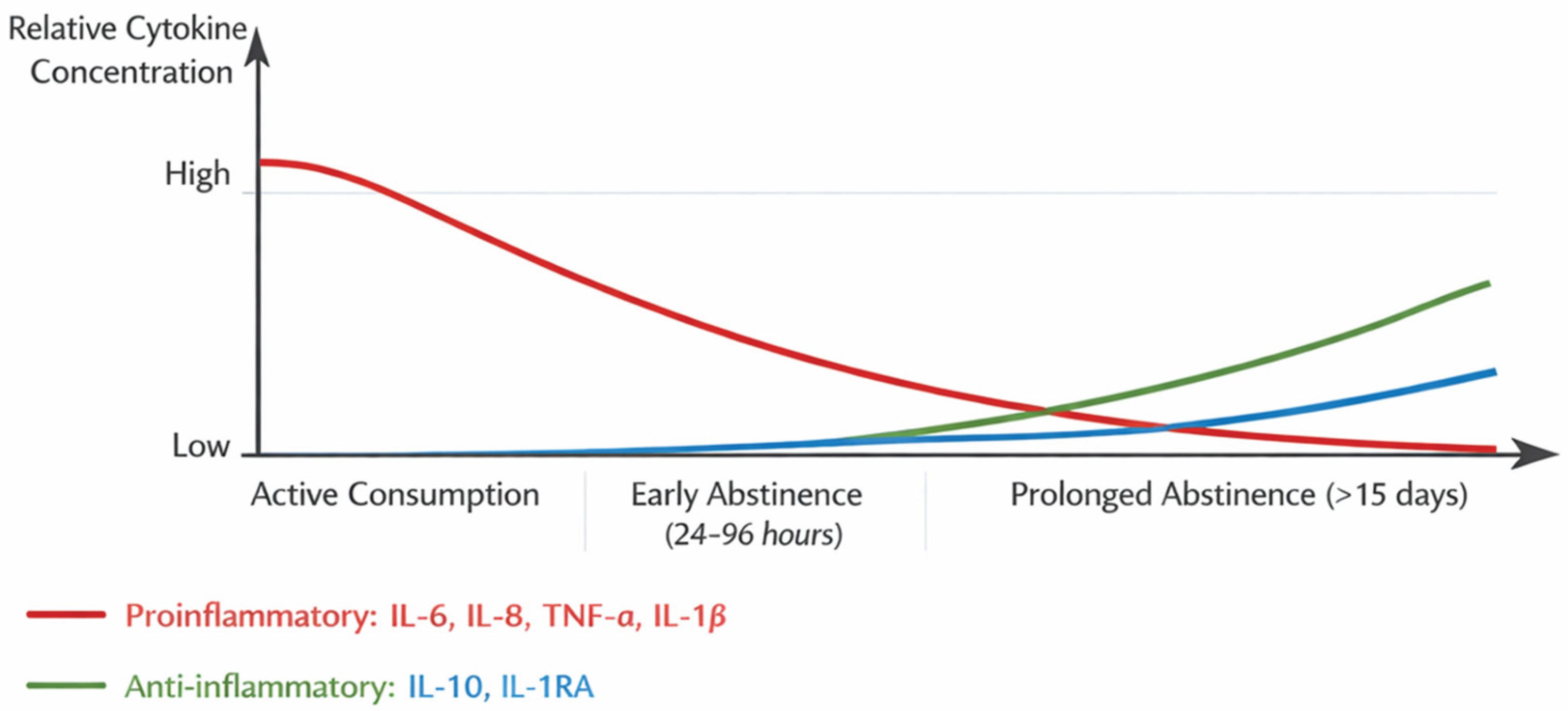
To visually represent the dynamic fluctuations in cytokine levels during the different stages of AUD, a diagram has been included below. This figure illustrates the relative concentrations of pro-inflammatory cytokines (IL-6, IL-8, TNF-α, IL-1β) and anti-inflammatory cytokines (IL-10, IL-1RA) across three key phases: active alcohol consumption, early abstinence (24–96 hours post-cessation), and prolonged abstinence (beyond 15 days). The diagram highlights the systemic inflammatory state observed during active drinking and early abstinence, characterized by elevated pro-inflammatory markers and suppressed anti-inflammatory mediators. It also demonstrates the gradual normalization of cytokine levels during prolonged abstinence, emphasizing the potential recovery of immune function. This visual aid complements the textual analysis and provides a clearer understanding of the immune dysregulation associated with AUD, offering insights into its clinical implications and potential therapeutic targets.
Figure 1.
Changes in Cytokine Levels Across the Stages of Alcohol Use Disorder (AUD). Note: Figure developed by authors from the data reported in the analysed studies. Systemic inflammation has been reported during active alcohol consumption and early abstinence. Prolonged abstinence appears to be associated with lower levels of pro-inflammatory cytokines and higher levels of anti-inflammatory cytokines. In severe AUD, IL-8 may remain elevated after weeks of abstinence.
Figure 1.
Changes in Cytokine Levels Across the Stages of Alcohol Use Disorder (AUD). Note: Figure developed by authors from the data reported in the analysed studies. Systemic inflammation has been reported during active alcohol consumption and early abstinence. Prolonged abstinence appears to be associated with lower levels of pro-inflammatory cytokines and higher levels of anti-inflammatory cytokines. In severe AUD, IL-8 may remain elevated after weeks of abstinence.
3.3. Associations of Clinical Severity and Affective Symptoms with Inflammatory and Neurotrophic Markers in Individuals with Alcohol Use Disorder (AUD)
It has been reported that cytokines are associated with mood states in patients with psychiatric disorders. For example, elevated levels of inflammatory cytokines such as interleukin-6 (IL-6) and TNF-α have been associated with depression. In addition, positive associations have been observed between anxiety (e.g., panic disorder) and the cytokines IL-6, IL-1β, and IL-5. It has also been described that, in patients with AUD, IL-6 is positively associated with depression and IL-10 is negatively associated with anxiety [
23].
Similarly, Balan et al. [
26] reported a significant association between alcohol craving in individuals with AUD and intestinal dysfunction, alongside elevations in pro-inflammatory biomarkers such as IL-1β and TNF-α. This finding reinforces the proposed relationship between immune activity and clinically relevant symptoms of AUD. Regarding neurotrophic markers, the study measured brain-derived neurotrophic factor (BDNF) but did not detect significant differences between high- and low-risk alcohol consumption groups. No meaningful associations were observed between BDNF levels and AUDIT or Alcohol Use Disorders Identification Test–Consumption (AUDIT-C) scores. Nevertheless, the authors suggest that future research incorporating anti-inflammatory mediators such as IL-1RA or IL-10 could provide a more balanced characterization of immune regulation in AUD.
García-Calvo et al. [
27] also indicated elevated levels of interleukin 6 (IL-6) were found in individuals with AUD compared to healthy controls. IL-6 has been associated with anxiety and depressive symptoms during alcohol withdrawal, although findings are inconsistent. IL-6 levels have also been linked to comorbid depressive disorders in individuals with AUD. Regarding other psychiatric disorders, higher blood IL-6 levels have been observed in other psychiatric conditions, such as post-traumatic stress disorder, major depression, and schizophrenia. However, only a few studies on AUD explicitly excluded individuals with other psychiatric disorders, which could act as confounding factors. Overall, while IL-6 appears to play a role in mood symptoms associated with AUD, further research is needed to clarify its relationship with anxiety and depression.
In the same line, Tyler et al. [
28] reported significant associations between clinical severity and biomarker profiles in AUD. Specifically, greater AUD severity and higher anxiety/depression scores were positively correlated with the proinflammatory cytokines IL-18 and TNF-α, whereas both clinical measures were negatively correlated with BDNF. IL-18 is described as an immune-activating cytokine implicated in chronic inflammation and immune dysregulation, and its positive association with AUD severity and anxiety/depression is presented as consistent with evidence of altered IL-18 signalling in stress- and addiction-relevant neurocircuitry in animal models, as well as increased IL-18 expression in peripheral immune cells in alcohol-related liver disease. Similarly, TNF-α—widely characterized as a key mediator of systemic inflammation—was positively correlated with AUD severity and with anxiety/depression, consistent with the view that AUD is associated with a heightened inflammatory tone and may be linked to neuroinflammation and mood symptoms. In contrast, BDNF, described as central to synaptic plasticity and overall brain health and noted to have anti-inflammatory effects, was inversely correlated with AUD severity and anxiety/depression, supporting the interpretation that more severe clinical presentation is accompanied by lower neurotrophic support. Overall, these correlation patterns are interpreted as reflecting a coordinated relationship between inflammatory activation (IL-18, TNF-α), reduced neuroplasticity-related signalling (BDNF), and the clinical expression of AUD and comorbid affective symptoms.
Bramness et al. [
25] also indicated that elevated levels of the cytokine IL-6 are associated with suicide attempts in patients with AUD. Although no statistically significant differences in IL-6 levels were observed between patients with and without suicide attempts in the bivariate analysis, the multivariable analysis—adjusted for factors such as sex, nicotine use, somatic comorbidities, and the use of anti-inflammatory medications—showed a significant association between higher IL-6 levels and suicide attempts (p = 0.033). These findings support prior evidence suggesting that IL-6 may represent a biological marker related to suicidal behaviour. However, the authors caution that the clinical applicability of IL-6 as an individual-level predictive biomarker remains limited.
Finally, the study by Grodin et al. [
24] found that individuals with AUD and clinically significant insomnia symptoms exhibited significantly higher levels of IL-8, a proinflammatory chemokine, than those without insomnia or with subclinical insomnia. No significant increases were observed in other inflammatory markers, including IL-6, TNF-α, or CRP. Regarding sex- and AUD severity–related interactions, men with AUD and clinical insomnia showed higher IL-8 levels than women and the other comparison groups. Likewise, individuals with severe AUD and clinical insomnia presented higher IL-8 concentrations than those with moderate AUD or without insomnia. Overall, the evidence suggests that both insomnia and AUD are associated with systemic inflammation, and that IL-8 may represent a potential biomarker linking these two disorders. Furthermore, interactions involving sex and AUD severity appear to shape IL-8 levels, indicating that men and individuals with severe AUD may be more vulnerable to the inflammatory consequences of insomnia and alcohol consumption. Further research is warranted to clarify these findings.
3.4. Main Limitations and Future Lines of Research
The reviewed studies on cytokine and interleukin networks in individuals with AUD present limitations that may undermine the robustness and generalizability of their findings. Sample sizes are frequently small, reducing statistical power, and many investigations rely on cross-sectional designs, precluding evaluation of within-subject changes in cytokine profiles across detoxification and sustained abstinence. Generalizability is further constrained by selective sampling strategies that exclude clinically relevant subgroups (e.g., currently drinking individuals; patients with severe liver or pancreatic disease; or those with psychiatric comorbidities). Across studies, inconsistent findings likely reflect substantial clinical heterogeneity—particularly differences in recency of alcohol intake, dependence severity, and comorbid conditions (including alcoholic liver disease, alcoholic hepatitis, and concurrent infections such as hepatitis B or C) that can amplify pro-inflammatory signalling.
Methodological variability also complicates comparisons, including differences in laboratory platforms, as well as variation in sample collection timing, storage, and processing, all of which may affect measurement accuracy and standardization. Finally, the limited use of standardized clinical instruments (e.g., the Clinical Institute Withdrawal Assessment for Alcohol) reduces the capacity to characterize withdrawal severity and objectively relate detoxification outcomes to inflammatory markers.
Future research should prioritize larger and more diverse cohorts, longitudinal designs, and standardized clinical and laboratory protocols to improve reliability and clinical interpretability.
Table 2 summarizes the main limitations identified in the reviewed studies, emphasizing methodological constraints and areas for future research to enhance the understanding of immune dysregulation in individuals with AUD.
Moreover, across the included studies, limitations and proposed future directions consistently pointed to the need for longitudinal designs with longer follow-up to track post-detoxification and abstinence-related changes in cytokines and related biomarkers (e.g., CD163, sCD14, IL-6), alongside objective clinical documentation of detoxification outcomes using standardized instruments such as CIWA-Ar. The literature also emphasized broader and more representative sampling—larger cohorts; greater inclusion of women with consideration of menopausal/hormonal factors; recruitment of individuals with active alcohol use, milder drinking phenotypes, and psychiatric patients without substance-use histories; and targeted cohorts with comorbid conditions, particularly established liver disease and severe hepatic/pancreatic involvement.
Methodologically, authors recommended expanding immune panels and evaluating candidate biomarkers (i.e., IL-7, IL-10, G-CSF, and IL-6) for diagnostic, staging, monitoring, and prognostic purposes, while advancing mechanistic work on cytokine interactions, immune-cell dysfunction, monocyte activation, metabolic disturbances, transcriptional regulation (e.g., STAT3), and alcohol-related autoimmune processes. Additional priorities included assessing the impact of detoxification medications (i.e., diazepam, vitamin supplementation), comparing detoxification protocols, examining how targeted interventions (e.g., HCV treatment, metabolic management, sustained abstinence) influence biomarker trajectories and outcomes, and exploring AUD–comorbidity interactions to inform preventive approaches and the clinical use of immunological parameters as diagnostic tools and biomarkers, including in psychiatric comorbidity.
In addition, the clinical implementation of biomarkers also entails several challenges. Ensuring validity and reproducibility is essential, such that biomarkers yield consistent and replicable results across different populations and clinical contexts. In this regard, variability in analytical methods, sample storage procedures, and study conditions can compromise measurement precision and, consequently, the reliability of findings.
A further barrier is the limited standardization of methods. The absence of standardized protocols for biomarker assessment can hinder clinical uptake, underscoring the need for uniform procedures for sample collection, processing, and analysis. In parallel, costs and accessibility remain important concerns, as some biomarkers require advanced and expensive technologies for detection, thereby restricting their use in resource-limited clinical settings.
Interpreting biomarker results can also be complex, particularly because biomarker levels may be influenced by multiple factors, including comorbidities, medications, age, sex, and lifestyle. This complexity is compounded by the relative scarcity of longitudinal studies, which are needed to clarify how biomarker levels change over time and how these trajectories relate to disease progression and treatment response.
Regulatory and approval requirements represent another substantive obstacle, as biomarkers must meet stringent regulatory standards before being authorized for clinical use, a process that can be lengthy and costly. Beyond regulation, clinical acceptance is critical: healthcare professionals require training in biomarker utilization and interpretation, and clinical utility must be clearly demonstrated for biomarkers to be adopted in routine practice. Finally, successful integration into healthcare systems depends on adequate infrastructure, including specialized laboratories and robust data-management systems, which may be challenging to establish in certain settings.
Addressing these challenges is essential to ensure that biomarkers can be used effectively and reliably in clinical practice, thereby improving the diagnosis, monitoring, and treatment of conditions such as AUD.
4. Discussion
The immune system has been reported to regulate behaviour via inflammatory signalling within the central nervous system (CNS), and multiple studies suggest that the pharmacodynamic effects of alcohol and other substances (e.g., cocaine) involve central modulation of immunological signalling pathways [
31,
32]. Within this signalling network, cytokines are pivotal mediators of cellular communication and activation. They act across tissues to regulate inflammatory processes, cell migration, and proliferation [
33]. Cytokines may access the brain via the blood–brain barrier or be produced locally by non-neuronal cells, including astrocytes and microglia [
34].
Current scientific evidence further indicates that alcohol directly modulates immune function and promotes neuroinflammation by altering the expression of inflammatory mediators at both central and peripheral levels [
35,
36]. Alcohol consumption has been proposed to disrupt microglia–neuron interactions mediated by cytokines [
37]; however, this interpretation has been challenged by in vivo human neuroimaging studies [
38,
39], and additional research is required to clarify these effects in adult patients with AUD [
28]. Given that inflammatory cytokine expression profiles change during the development of AUD and appear to normalize following cessation, it is plausible that such normalization contributes meaningfully to clinical improvement in individuals with AUD [
40].
In recent years, substantial attention has focused on the neurobiological basis of AUD, and neuroimmune signalling may play an important role in both the development and progression of the disorder [
41]. Chronic inflammation has been proposed as a mediator of AUD, as prolonged heavy alcohol consumption can alter the adaptive immune profile [
12]. Chronic heavy drinking activates toll-like receptor signalling, increasing intestinal permeability and the circulation of microbial products; this can stimulate peripheral immune cells to secrete pro-inflammatory cytokines, with subsequent priming of microglia and astrocytes to release cytokines within the central nervous system [
42]. Elevated cytokine levels have been associated with aberrant activation of other immune products, including IgE immunoglobulins [
43], and have also been implicated in alcohol-related organ dysfunction, with cytokines such as TNF-α and interleukin-6 (IL-6) contributing to the pathology of alcoholic hepatitis and alcoholic liver disease, respectively [
12]. This alcohol-induced chronic pro-inflammatory state is associated with oxidative stress and diminished neural function [
44] and may ultimately modulate neurotransmitter systems relevant to addiction [
45]. Consequently, immune dysregulation induced by chronic alcohol use may represent an important link to the neuropsychological changes observed in AUD [
41].
Several studies have reported associations between inflammatory alterations and AUD [
23,
24,
25,
26,
27,
28,
29,
30]. Peripheral cytokines, including IL-6 and TNF-α [
39,
46,
47], as well as IL-8, IL-10, and IL-12 [
24,
43], and inflammatory markers such as CRP [
47,
48], have been implicated in AUD. Increased peripheral cytokines have been associated with clinical (i.e., insomnia, intestinal problems) and psychiatric characteristics (i.e., anxiety, depression, suicide) [
23,
24,
25,
26,
27,
28,
49] and with drinking-related variables, including dependence severity, alcohol withdrawal score, and alcohol craving [
39,
46,
47].
In addition, anxiety and depressive symptoms in AUD were consistently associated with the biomarker profile previously analysed, aligning affective symptom burden with both heightened inflammatory signalling and reduced neurotrophic support. Specifically, anxiety/depression scores were positively correlated with the proinflammatory cytokines IL-18 and TNF-α and negatively correlated with BDNF. Taken together, these associations suggest that individuals presenting with greater anxiety and depressive symptomatology may also show a more pronounced proinflammatory milieu alongside lower levels of a factor linked to synaptic plasticity and overall brain health [
15,
28,
41,
50,
51,
52].
From a mechanistic standpoint, the observed positive relationships between anxiety/depression and IL-18 and TNF-α support an interpretation in which inflammatory activation is intertwined with the emotional and psychological manifestations of AUD. Within the framework, both IL-18 and TNF-α are positioned as key mediators of immune activation and systemic inflammation, and their correlations with anxiety/depression are therefore consistent with the notion that inflammatory processes may be relevant to mood-related symptom expression in this population. In this context, the correlations do not establish causality, but they reinforce the plausibility of an inflammation–affect link as part of the clinical phenotype of AUD [
15,
28,
51].
By contrast, the inverse association between anxiety/depression and BDNF complements the inflammatory findings by pointing to reduced neurotrophic signalling in individuals with higher affective symptom burden. Given BDNF’s described role in synaptic plasticity, neurogenesis, and brain health, lower BDNF in conjunction with higher anxiety/depression scores is compatible with the interpretation that affective symptoms in AUD co-occur with diminished neuroplasticity-related support. Importantly, this pattern mirrors the broader correlational structure observed in the study of Tyler et al [
28]—where greater clinical severity is associated with higher IL-18 and TNF-α and lower BDNF—suggesting that anxiety and depressive symptoms may cluster with the same biological correlates implicated in more severe AUD presentations [
15,
28,
50,
51,
52].
These association patterns underscore anxiety and depression as clinically relevant dimensions that co-vary with inflammatory and neurotrophic markers in individuals with AUD. Situating anxiety/depression within this biomarker context may refine the clinical characterization of AUD by emphasizing that affective symptoms are not simply secondary features but rather are embedded within the broader biological profile observed in the present study [
15,
28,
50,
51,
52,
53,
54].
The associations between AUD severity, anxiety/depression, and these biomarkers suggest a complex interplay between inflammation, neuroplasticity, and psychological health in AUD: IL-18 and TNF-α reflect the proinflammatory state associated with chronic alcohol use and its impact on mental health. BDNF deficits highlight the neuroplasticity impairments caused by AUD and their role in emotional dysregulation. These findings underscore the relevance of targeting inflammation and neuroplasticity in developing treatments for AUD and its associated comorbidities, such as anxiety and depression [
28]. Prior work suggests an association between cytokine alterations and mood states in psychiatric disorders, with higher IL-6 and TNF-α levels reported in relation to depression. While not implying causality, these observations support the consideration of cytokines as potential biomarkers for psychiatric conditions [
23,
55,
56,
57,
58].
In addition, cytokine levels may also fluctuate across the cycle of chronic alcohol exposure, including active drinking, withdrawal, and varying periods of abstinence. During active alcohol consumption and early abstinence, pro-inflammatory cytokines such as IL-6, IL-8, TNF-α, and IL-1β tend to be elevated relative to healthy controls. During prolonged abstinence, cytokine levels tend to decrease, suggesting a potential recovery of immune function. Mechanistically, alcohol-related increases in intestinal permeability permit bacterial endotoxins (e.g., lipopolysaccharide, LPS) to enter the circulation, activating toll-like receptors (TLR4) and the NF-κB pathway, thereby promoting inflammatory cytokine production; this process may contribute to systemic inflammation and organ damage, including hepatic and cerebral injury [
26,
42,
44]. A positive association has been reported between cytokine levels and daily alcohol consumption, whereas no significant correlation was observed between cytokine levels and alcohol craving [
26].
Detoxification studies suggest that cytokine concentrations can show directional changes during treatment. In alcohol-dependent individuals without liver disease, IL-7 remained steadily elevated from admission to two weeks later and then declined, while remaining significantly higher than levels in healthy controls at the end of detoxification; IL-10 was significantly low on admission, increased linearly during therapy, and remained non-significantly low at the end; and G-CSF was significantly elevated on admission and declined linearly to near-normal values by the end of detoxification therapy [
29]. These alterations—and their trend toward normalization during detoxification—have been interpreted as indicative of generalized immune system disturbance associated with alcohol abuse [
29,
61].
During abstinence, concentrations of certain cytokines may remain elevated, particularly IL-6, IL-8, TNF-α, and IL-10. In general, these levels appear higher in early abstinence (24–96 hours following cessation) and tend to normalize over time, with more sustained abstinence (e.g., beyond 15 days) associated with progressive normalization [
40,
46,
47]. Specifically, it has reported elevated levels of IL-8, during early withdrawal, reflecting acute inflammation [
28].
Regarding specific cytokines, IL-8 was higher in abstinent individuals with AUD compared with healthy controls and with individuals who were currently drinking. IL-8 is a proinflammatory chemokine central to neutrophil recruitment during inflammatory responses, and its elevation in abstinence may indicate ongoing inflammatory activation despite cessation of alcohol use. IL-18 also showed a tendency toward higher levels in abstinent participants. Given IL-18’s role in promoting immune activation and its involvement in chronic inflammatory states, this pattern may reflect sustained immune signalling after discontinuation of alcohol consumption. Anti-inflammatory pathways were also implicated: IL-10 has been linked to AUD through genetic variation, and IL-1RA has shown protective effects against alcohol-induced liver injury in preclinical models, suggesting compensatory anti-inflammatory activity in the context of alcohol-related immune stress [
28,
40,
59]. The findings emphasize the dynamic nature of cytokine regulation in AUD. Cytokine concentrations—particularly IL-8—may vary according to drinking pattern, alcohol dose, and the timing of biospecimen collection. For example, IL-8 may rise transiently following acute alcohol exposure and subsequently return toward baseline. Overall, these observations position cytokines and interleukins as key components of AUD-associated immune dysregulation and support their potential utility as biomarkers of disease state and as candidate therapeutic targets [
14,
15,
28,
41,
52].
While some studies suggest that proinflammatory markers decrease after a few weeks of abstinence, individuals with severe AUD may experience a prolonged proinflammatory state during abstinence [
45,
47]. It is hypostatized that elevated cytokine levels during abstinence may be linked to the chronicity and severity of AUD rather than abstinence itself. Severe AUD may lead to sustained inflammation that persists even after alcohol cessation [
14,
28]. This underscores the complex relationship between alcohol use, withdrawal, abstinence, and immune system dysregulation.
Alcohol abstinence may confer clinically meaningful immunological benefits in patients with AUD by attenuating systemic inflammation and monocyte activation. Consistent with this premise, previous studies have reported that biomarkers of inflammatory activity and gut-barrier dysfunction, including soluble CD14 (sCD14) and interleukin-6 (IL-6), decline after approximately two to three weeks of sustained abstinence. Mechanistically, chronic alcohol exposure promotes intestinal permeability and facilitates the translocation of bacterial products such as lipopolysaccharide (LPS) into the circulation, thereby driving monocyte activation and downstream inflammatory cascades. Discontinuation of alcohol use is therefore expected to reduce the burden of microbial-derived immune stimuli and, in turn, dampen systemic inflammatory signalling. In addition, abstinence may ameliorate co-occurring proinflammatory processes—such as oxidative stress, iron dysregulation, and subclinical hepatic injury—which together may improve clinical trajectories and decrease the likelihood of progression to advanced liver disease [
15,
23,
27,
28,
41].
The current narrative review also acknowledges several limitations inherent to the analysed studies, including small sample sizes, cross-sectional designs, lack of standardized biomarker measurement protocols, and limited representation of diverse populations. These limitations are critically appraised to provide a balanced interpretation of the findings and to inform future research priorities.
The limitations of the present research are primarily related to its methodological orientation as a narrative review. Although we consulted major bibliographic databases and applied a structured set of keywords to identify relevant studies, the search strategy was intended to support a conceptually driven synthesis rather than an exhaustive, protocol-based retrieval of all eligible records. In addition, we did not undertake a formal, standardized appraisal of study quality or risk of bias, which constrains our ability to evaluate findings quantitatively. These characteristics are typical of narrative syntheses and enable broader integration across a heterogeneous literature; however, they also warrant cautious interpretation of the conclusions and highlight the need for future systematic reviews and meta-analytic research to corroborate and refine the patterns described here.
Moreover, the integrative approach of this study represents a significant advancement in understanding the complex interplay between alcohol use, immune dysregulation, and neurobiology. While previous research has documented associations between AUD and elevated cytokine levels, this review uniquely synthesizes evidence to propose a comprehensive framework that links peripheral immune changes to central neuroimmune activation. This systems-level perspective not only enhances our understanding of AUD pathophysiology but also provides a foundation for developing targeted interventions that address the underlying immune mechanisms.
Furthermore, this study moves beyond the limitations of cross-sectional analyses by emphasizing the dynamic nature of cytokine regulation in AUD. By analysing on how inflammatory signatures vary across different stages of the disorder—active drinking, withdrawal, and abstinence—it offers a more nuanced understanding of the temporal evolution of immune dysregulation. This novel focus on time-sensitive immune changes is critical for identifying biomarkers that can better capture disease progression and inform personalized treatment strategies.
Another unique contribution of this review is its emphasis on the translational implications of immune dysregulation in AUD. By identifying specific cytokines and interleukins (e.g., IL-6, IL-8, TNF-α, IL-1β) as potential biomarkers, the study provides actionable insights for clinical practice, including the development of biomarker-guided diagnostic tools, personalized treatment approaches, and relapse prevention strategies. This focus on clinical applicability sets the study apart from previous research, which has often been limited to theoretical or preclinical findings.
In summary, this review’s novelty lies in its integrative and dynamic characterization of alcohol-related immune dysregulation, its explicit alignment with clinically relevant phases of AUD, and its emphasis on translational applicability. By identifying key research gaps and articulating future priorities, it makes a distinctive contribution to the field and provides a foundation for more effective and personalized approaches to AUD management.
Overall, the synthesized evidence reinforces the centrality of immune dysregulation in the pathophysiology and clinical expression of AUD. Through the integration of findings across methodologically diverse studies, the review highlights the potential utility of inflammatory and neurotrophic biomarkers for improving diagnostic precision, informing individualized treatment strategies, and supporting better long-term outcomes. Based on these insights, the following sections examine the clinical implications of the findings and outline future research directions to address outstanding gaps and advance the understanding and management of AUD.
4.1. Clinical Implications
Clinical implications emerging from this narrative synthesis support a more structured and multidimensional approach to the management of alcohol detoxification and its associated complications. In particular, clinical practice should incorporate an objective protocol for evaluating detoxification outcomes, given that a key limitation identified in the evidence base was the absence of objective clinical documentation of treatment response. Implementing standardized instruments such as the Clinical Institute Withdrawal Assessment for Alcohol Scale (CIWA-Ar) would strengthen the clinical characterization of withdrawal severity and detoxification progress and would provide a consistent framework for monitoring clinical response across settings. In addition, beyond the expected remission of alcohol-induced depressive symptoms, the evidence supports considering assessment of immune status in alcohol-dependent individuals as part of clinical care, with the explicit aim of preventing subsequent complications during and after detoxification.
The reported correlations between AUD severity, anxiety/depression, and biomarkers (IL-18, TNF-α, and BDNF) further argue for an integrated clinical perspective in which inflammatory activity, neuroplasticity-related signalling, and psychological health are considered jointly rather than in isolation. On purpose, Balan et al. [
26] highlights interleukin-1β (IL-1β) as a promising biomarker for assessing risk of AUD, as indicated by scores on the AUDIT and AUDIT-C screening instruments. Across predictive models, IL-1β emerged as the strongest and most consistent biomarker for AUDIT and AUDIT-C outcomes. In linear regression analyses, IL-1β accounted for up to 51% of the variance in AUDIT scores and 38% of the variance in AUDIT-C scores [
26].
From a diagnostic perspective, receiver operating characteristic (ROC) analyses indicated that IL-1β has good discriminative performance for identifying individuals with AUDIT scores ≥ 6 (AUC = 0.81) and excellent discrimination for AUDIT-C scores (AUC = 0.94), suggesting potential utility for detecting risky alcohol consumption patterns. Moderated multiple regression (MMR) analyses further supported the independent predictive value of IL-1β. IL-1β predicted AUDIT and AUDIT-C scores without significant interaction effects involving other immunological mediators, indicating that its association with alcohol-risk indices is not contingent on the levels of the other biomarkers examined [
26].
Clinically, these findings suggest that IL-1β may function as an early marker of AUD risk, including among individuals who do not yet meet full diagnostic criteria for AUD. This could facilitate earlier identification of at-risk individuals and enable more tailored preventive or therapeutic interventions. Overall, the study of Balan et al. [
26] underscores the potential of IL-1β as a clinically relevant biomarker for assessing AUD risk and its link to inflammatory and immune dysfunction pathways. Nevertheless, the authors emphasize the need to confirm these results in larger samples and in clinically well-characterized cohorts [
26].
Within this framework, biomarker findings are clinically relevant because they point to a biological signature that covaries with clinical severity and affective symptom burden. This supports the clinical rationale for viewing biomarkers as potential tools to confirm diagnosis, define current stage, facilitate earlier identification, and complement standard clinical assessments in monitoring the course and outcome of detoxification therapy. Consistent with the longitudinal priorities emphasized in the literature, biomarker monitoring is also positioned as potentially useful for characterizing trajectories during abstinence and for identifying patients at early stages who may be at higher risk of progression or poorer prognosis.
From a management standpoint, the recommendations organized by clinical domains emphasize inflammation as a relevant treatment target in severe presentations and in the presence of persistent inflammatory activation. In this context, anti-inflammatory strategies may warrant investigation, including immunomodulatory medications—such as IL-1 pathway inhibitors or TNF-α antagonists—in individuals with severe AUD and evidence of sustained inflammation. In parallel, naturally derived compounds with reported anti-inflammatory properties (e.g., omega-3 fatty acids, curcumin, or polyphenols) could be explored as adjunctive approaches. These approaches can be complemented by lifestyle modifications, including promotion of a balanced diet rich in anti-inflammatory foods and encouragement of regular physical activity. Given the proposed contribution of gut dysbiosis to systemic inflammation, addressing gut health through probiotics or dietary interventions such as high-fiber diets is also included within the recommended clinical domain focused on inflammation.
The clinical implications also extend to interventions aimed at enhancing neuroplasticity, particularly in the context of lower BDNF levels and comorbid depression/anxiety. Pharmacological avenues proposed within the recommendations include investigating medications that may enhance BDNF, such as antidepressants (e.g., Selective Serotonin Reuptake Inhibitors: SSRIs) or ketamine in individuals with AUD and comorbid affective symptoms, as well as exploring GLP-1 receptor agonists. Non-pharmacological strategies are also emphasized, including mindfulness-based practices such as meditation and yoga and the use of cognitive-behavioural therapy (CBT) to address anxiety, depression, and alcohol-related behaviours. Exercise is highlighted as a central component of this neuroplasticity-oriented domain, with recommendations to incorporate aerobic activity and/or resistance training as part of treatment planning.
Considering the frequent co-occurrence of anxiety and depression in AUD and their associations with the biomarker profile described, mental health assessment and management should be positioned as an integral component of care, not an adjunct. The recommendations therefore emphasize integrated, dual-diagnosis treatment that targets both AUD and co-occurring mental health disorders, alongside the use of psychiatric medications such as SSRIs or SNRIs when indicated. Psychotherapeutic approaches are also foregrounded, including trauma-focused therapy in the presence of early-life stress or PTSD, and motivational interviewing to support engagement and address ambivalence regarding recovery. In addition, participation in peer support groups—such as Alcoholics Anonymous or SMART Recovery—is recommended to reduce isolation and provide ongoing emotional support.
In terms of implementation and follow-up, the recommendations underscore the potential role of systematic biomarker monitoring to support clinical decision-making. This includes regular testing of inflammatory markers (IL-18 and TNF-α) and neuroplasticity-related biomarkers (BDNF) during treatment, with the goal of evaluating progress over time. Within this approach, biomarker-guided therapy is proposed, whereby clinical emphasis is tailored according to biomarker levels—for example, prioritizing anti-inflammatory strategies when IL-18 or TNF-α are elevated, and emphasizing neuroplasticity-oriented strategies in the context of low BDNF and prominent affective symptoms.
Finally, sustained abstinence support is identified as a critical component of management, particularly for individuals with severe AUD who may require more intensive early-phase stabilization. The recommendations include consideration of residential or inpatient programs during early abstinence to provide structured care and therapy, as well as relapse prevention strategies that combine pharmacotherapy to reduce cravings with ongoing therapy and relapse prevention planning. Across all domains, a holistic, personalized, and multidisciplinary model of care is recommended, involving collaboration among addiction specialists, psychologist, psychiatrists, nutritionists, and physical therapists. Patient education is also highlighted as an essential component, with an emphasis on explaining the connections between alcohol use, inflammation, and mental health to support informed engagement with treatment and recovery planning.
Table 3 summarizes the clinical implications and potential applications of cytokine and interleukin biomarkers in AUD. This table provides a comprehensive overview of how specific biomarkers can be utilized to enhance diagnostic precision, monitor disease progression, and inform personalized treatment strategies. By highlighting the translational relevance of these biomarkers, the table serves as a practical guide for integrating immunological insights into clinical practice and developing targeted interventions for AUD management.
4.2. Future Lines of Research
Based on analysed studies, future research should adopt longer follow-up periods and longitudinal designs to clarify the long-term effects of alcohol detoxification on cytokine concentrations and the recovery of immune system function. In addition to examining post-detoxification change, longitudinal work should specifically investigate temporal trajectories in biomarker levels—such as CD163, sCD14, and IL-6—particularly following alcohol abstinence, to better characterize their evolution over time and their potential roles in the progression of alcohol-related conditions. These approaches would also help address a key limitation noted in the current review, refer to the absence of objective clinical evidence documenting detoxification treatment outcomes.
To strengthen clinical characterization, future studies should incorporate standardized clinical evaluations of detoxification outcome, for example through implementation of protocols such as the Clinical Institute Withdrawal Assessment for Alcohol Scale (CIWA-Ar), thereby enabling immune measures to be interpreted alongside objectively documented clinical course.
Improving the representativeness and clinical scope of study populations is also a priority. Future research should expand to include more diverse patient groups, including alcohol-dependent individuals with liver disease or other co-existing conditions, in order to capture the broader impact of alcohol abuse on immune function. Targeted analyses in specific cohorts with alcohol-induced severe hepatic and pancreatic disease are warranted to identify the stages at which cytokines such as IL-6 and IL-17A may function as disease-specific biomarkers. Complementarily, including individuals who are actively consuming alcohol would enable more precise characterization of the direct effects of alcohol exposure on cytokine concentrations and their relationship with comorbidities. Broader sampling across drinking phenotypes—including individuals with milder patterns of alcohol use or moderate drinking—would allow comparisons of biomarker profiles and help clarify their relevance across different levels of alcohol consumption. Future studies should also increase efforts to recruit women, explicitly considering factors such as the menopausal transition and hormonal levels (e.g., estrogen and luteinizing hormone) that may influence cytokine concentrations. Furthermore, larger sample sizes are required to improve statistical validity and enhance the generalizability of findings. In addition, assessing psychiatric patients without a history of substance use disorders would help distinguish cytokine alterations attributable to AUD from those associated with other psychiatric conditions.
Beyond population-level refinements, future work should broaden the immunological scope of measurement. Studies incorporating additional immune parameters, together with longer follow-up periods and varied alcohol-dependent patient groups (including those with established liver disease), are needed to further elucidate pathophysiology. Within this framework, research should evaluate the potential utility of IL-7, IL-10, and G-CSF as biomarkers for monitoring progress and outcomes during detoxification therapy. More generally, biomarkers may be useful for confirming diagnosis, defining the current stage of AUD, and supporting early identification of patients. IL-6 elevation in individuals with AUD is also proposed as a potential risk marker for adverse medium-term outcomes even in the absence of advanced liver disease, and future studies should determine whether biomarker panels can identify early-stage AUD patients at increased risk of mortality or progression to severe liver disease.
Mechanistic investigation remains essential to explain observed biomarker patterns. Future research should examine interactions among cytokines—such as IL-7, IL-10, G-CSF, and other mediators—to improve understanding of immune modulation in alcohol dependence. Future research should further explore the mechanisms through which alcohol abuse leads to immune system disorders, including cytokine pathway dysregulation and immune cell dysfunction. Given the complexity of inflammatory signalling, developing integrated models that synthesize information across multiple inflammatory signals may better capture cytokine interaction patterns in alcohol addiction and its comorbidities. Additional mechanistic priorities include elucidating the links between monocyte activation, systemic inflammation, and metabolic disturbances in AUD even in the absence of advanced liver disease. At the genomic and molecular level, studies should investigate mechanisms underlying cytokine alterations during alcohol detoxification, including the role of transcription factors such as STAT3. The relationship between alcohol-induced autoimmune reactions and cytokine production—particularly IL-7—also warrants further study to clarify its proposed relevance for liver function and immune system regulation.
Interventional and comparative research directions are likewise indicated. Future studies should examine the specific effects of pharmaceutical treatments used during detoxification, including diazepam and vitamin supplementation, on cytokine levels and immune recovery. Comparative studies evaluating cytokine alterations across different detoxification protocols are needed to identify the most effective treatment approaches. In addition, research should assess how targeted interventions—such as treatment of hepatitis C virus infection, management of metabolic abnormalities, and sustained abstinence—affect biomarker levels and clinical outcomes. Given the clinical complexity of AUD, further work should explore interactions between AUD and comorbidities such as HCV infection, metabolic syndrome, and cardiovascular disease, with the aim of informing integrated therapeutic strategies.
Furthermore, future research should delve deeper into the relationship between biomarkers and comorbid conditions, such as hepatic and pancreatic diseases, which are frequently associated with AUD. While current studies have identified associations between elevated cytokine levels—such as IL-6 and IL-17A—and liver and pancreatic dysfunction, further investigation is needed to elucidate the underlying mechanisms driving these interactions. Understanding how specific biomarkers contribute to the progression of these comorbidities could provide a foundation for the development of targeted prevention and treatment strategies. For instance, identifying early-stage biomarkers of liver or pancreatic disease in AUD patients may enable timely interventions to mitigate disease progression. Additionally, exploring the role of anti-inflammatory cytokines, such as IL-10, in protecting against organ damage could inform the design of novel therapeutic approaches aimed at restoring immune balance and reducing the burden of comorbid conditions in AUD populations.
Finally, future research should translate these immunological insights into preventive and clinically useful applications. Developing strategies to assess and improve the immune status of alcohol-dependent individuals may help prevent complications during and after detoxification. Within detoxification and follow-up care, immunological parameters may serve as biological markers and diagnostic tools to assess the course and outcome of therapy. More broadly, the framing of cytokines as potential biomarkers of psychiatric disorders provides an additional rationale for integrating immune profiling into research that addresses AUD and its comorbidities.
In sum, to advance the understanding of immune dysregulation in AUD, future research should adopt more targeted and interdisciplinary approaches. Specifically, the following strategies are proposed:
- –
Longitudinal study designs: Conduct extended follow-up studies to monitor the temporal evolution of cytokine and interleukin levels during different stages of AUD, including active drinking, withdrawal, and sustained abstinence. These studies should incorporate repeated biomarker assessments at multiple time points to capture dynamic changes in immune profiles.
- –
Advanced methodologies: Utilize cutting-edge techniques such as single-cell RNA sequencing and proteomics to investigate cytokine interactions and immune cell-specific responses. These methods can provide deeper insights into the molecular mechanisms driving immune dysregulation in AUD.
- –
Interdisciplinary collaborations: Establish partnerships between psychologist, psychiatrists, addiction specialists, immunologists, neuroscientists, and bioinformaticians to develop integrated models of AUD pathophysiology. Collaborative efforts can facilitate the synthesis of data across immune, neurobiological, and behavioural domains, enabling a more holistic understanding of the disorder.
- –
Clinical trials for biomarker validation: Design randomized controlled trials to evaluate the utility of cytokines and interleukins as biomarkers for AUD diagnosis, treatment response, and relapse prediction. These trials should include diverse patient populations, such as individuals with varying levels of alcohol consumption and comorbid conditions.
- –
Development of targeted interventions: Investigate the efficacy of immunomodulatory therapies, such as cytokine inhibitors or anti-inflammatory agents, in mitigating the pro-inflammatory state associated with AUD. Additionally, explore the potential of lifestyle interventions, including dietary modifications and exercise, to improve immune function and clinical outcomes.
- –
Global and population-based studies: Expand research to include diverse populations across different geographic regions and cultural contexts. This approach will help identify population-specific immune signatures and inform tailored interventions.
By implementing these strategies, future research can address existing gaps in the literature, enhance the clinical applicability of findings, and contribute to the development of innovative approaches for the prevention and treatment of AUD.