1. Introduction
Optimizing neuromuscular performance in elite athletes has become increasingly reliant on neurotechnological approaches, with EEG biofeedback (neurofeedback) gaining prominence as a method for modulating brain activity and enhancing motor function [
1,
2,
3]. Neurofeedback enables individuals to self-regulate cortical oscillations, particularly within sensorimotor and frontal regions, which are closely linked to motor control, attention, and learning [
4,
5]. Recent years have seen a growing body of research demonstrating that neurofeedback can improve reaction time, movement precision, and even strength and power, especially when protocols are tailored to the demands of athletic populations [
1,
6,
7,
8,
9].
Despite these advances, there remains considerable uncertainty regarding the optimal structure of neurofeedback interventions for high-performance sport, particularly in relation to session frequency and dosing [
1,
2,
7,
8,
9]. Current evidence supports 2–3 sessions per week, ≥20–30 minutes, over at least 1–2 weeks and >125 total minutes, with signs that 3×/week and >8 sessions are often more effective. Some evidence suggests that frequent neurofeedback sessions may accelerate neuroplastic changes and behavioral gains, while other findings indicate that longer intervals between sessions can facilitate consolidation and retention of training effects [
10,
11]. However, truly optimal frequency and dosing remain uncertain and likely need to be tailored to sport, performance target, and individual responder profile [
1,
2,
12]. This unresolved dose-response relationship is a significant barrier to the practical implementation of neurofeedback in elite sport, where training time is limited and intervention efficiency is paramount.
The integration of EEG and EMG measures in strength and power sports provides a powerful framework for understanding the interplay between cortical and neuromuscular adaptation [
13,
14]. EMG amplitude (RMS) and frequency characteristics (MF) are established markers of motor unit recruitment and fatigue, while EEG indices such as the Frontal Alpha Index (FAI) and power in F3/F4 regions reflect cortical engagement during motor tasks [
13,
15,
16]. Prior studies have linked changes in these indices to performance improvements following neurofeedback, but the influence of protocol structure, especially session frequency, on these adaptations remains to be elucidated [
1,
3,
8,
12].
Elite judo athletes, who must rapidly produce force, sustain dynamic motor control, and resist fatigue, present an ideal model for translational neurofeedback research [
6,
8,
17,
18]. To date, however, no study has systematically compared the effects of daily, every other day, and every third day EEG biofeedback protocols on neuromuscular and cortical outcomes in this population. Stratification of participants into phenotypic groups (High, Medium, and Low Responder) based on baseline neuromuscular and cortical characteristics allows for examination of whether optimal session frequency varies by individual responder profile, thereby addressing the heterogeneity in athlete adaptation patterns that is often overlooked in protocol-level comparisons [
19,
20,
21,
22]. This approach recognizes that standardized dosing may not be universally optimal and that some athletes may benefit from different training frequencies based on their baseline neurophysiological characteristics. Addressing this gap is critical for generating evidence-based recommendations that can inform practice in high-performance sport, where individualized and efficient interventions are essential [
6,
23,
24].
The current study directly compares the efficacy of three EEG biofeedback session frequencies daily, every other day, and every third day, on both functional (strength, power) and neurophysiological (EEG, EMG) adaptation in elite judo athletes. Using a randomized, controlled design and comprehensive outcome assessment, this research aims to clarify whether daily training yields greater improvements in functional and neurophysiological outcomes compared to less frequent protocols, and whether protocol frequency differentially affects the relationship between EMG amplitude and external load during strength testing. The study further explores the extent of individual variability in EMG–load adaptation within each protocol group and examines how session frequency influences resistance to neuromuscular fatigue and the retention of training-induced adaptations. By investigating whether observed changes in EEG indices are associated with the magnitude of neuromuscular adaptation and fatigue resistance, the study seeks to determine if specific EEG changes predict greater functional gains or better adaptation profiles. Furthermore, the technical challenges of acquiring high-quality EEG and EMG signals in athletic settings, including motion artifacts, perspiration-induced impedance fluctuations, and electromagnetic interference, remain inadequately characterized in the neurofeedback literature. Validating biosensor performance under these conditions is essential for translating laboratory findings to applied sport contexts.
This work is novel in its direct, head-to-head comparison of three EEG biofeedback protocols in a homogeneous group of elite athletes, using rigorous, multidimensional outcome measures and advanced statistical modeling. The findings are intended to provide robust, evidence-based recommendations for optimizing neurofeedback interventions in high-performance sport and to clarify the session frequency, adaptation relationship, a critical, yet understudied, parameter in applied neurofeedback science. The results are directly applicable to practitioners, coaches, and clinicians seeking to individualize and maximize the efficiency of neurofeedback-based training for athletic populations.
2. Materials and Methods
2.1. Study Design
This investigation employed a randomized, controlled, parallel-group experimental design to compare the effects of three EEG biofeedback protocols, differing exclusively in session frequency, on neuromuscular and neurophysiological adaptation in elite national-level judo athletes. The intervention period comprised 15 sessions of EEG biofeedback, with all other aspects of training and assessment standardized across groups. The primary outcomes were functional (strength, power) and neurophysiological (EEG, EMG) indices, with additional analyses of EMG–load dependency, fatigue resistance, and retention of adaptation.
2.2. Participants
Twenty-four elite male judo athletes from the national team participated in the study. All participants met the following inclusion criteria: (1) current membership in the national judo team, (2) age between 18 and 30 years, (3) no history of neurological, psychiatric, or musculoskeletal disorders, and (4) no prior experience with EEG biofeedback or neurofeedback interventions. Athletes were excluded if they reported any contraindications to EEG or EMG measurement, or if they were unable to complete the full intervention protocol.
Initial group allocation was based on a composite adaptation index calculated from pre-intervention baseline assessments, which integrated standardized values of EMG amplitude (RMS), percentage change in EMG (%ΔEMG), strength sum (Sth_SUM), and EEG summary indices (EEG_SUM). Participants were stratified into three phenotypic groups reflecting their baseline neuromuscular and cortical adaptation potential: High Responder Group (HRG), Medium Responder Group (MRG), and Low Responder Group (LRG). Each group consisted of eight athletes, ensuring equal group sizes and balanced representation of adaptation phenotypes.
All participants provided written informed consent. The cohort was homogeneous in terms of training background, competitive level, and exposure to standardized training and recovery schedules throughout the study period. The protocol was approved by the Institutional Review Board of the Academy of Physical Education in Katowice, Poland (ethics approval number: KB/01/2022). All raw data, analytic scripts, and supplementary materials are available in an open-access repository (Zenodo, DOI: 10.5281/zenodo.15879450).
2.3. Intervention Protocol
Each group was assigned to a specific EEG biofeedback protocol, differing only in session frequency while maintaining the same session duration. The HRG group performed 15 EEG biofeedback sessions on consecutive days (daily protocol), the MRG group completed 15 sessions every other day, and the LRG group underwent 15 sessions every third day. Each session lasted exactly five minutes and was conducted under identical laboratory conditions, with the same EEG feedback parameters and supervision by certified neurofeedback practitioners. All athletes were instructed to maintain their usual training routines outside of the intervention and to refrain from additional neurocognitive or relaxation techniques during the study period.
The EEG biofeedback protocol targeted frontal alpha and beta frequency bands (F3, F4), with real-time visual feedback provided via a standardized interface. All participants were instructed to maintain their regular judo training routines throughout the intervention and to abstain from additional cognitive or relaxation practices. The final sample included 24 athletes, with no dropouts or protocol violations reported. All demographic and baseline characteristics are summarized in supplementary materials
Table S1.
2.4. Biosensor System Overview
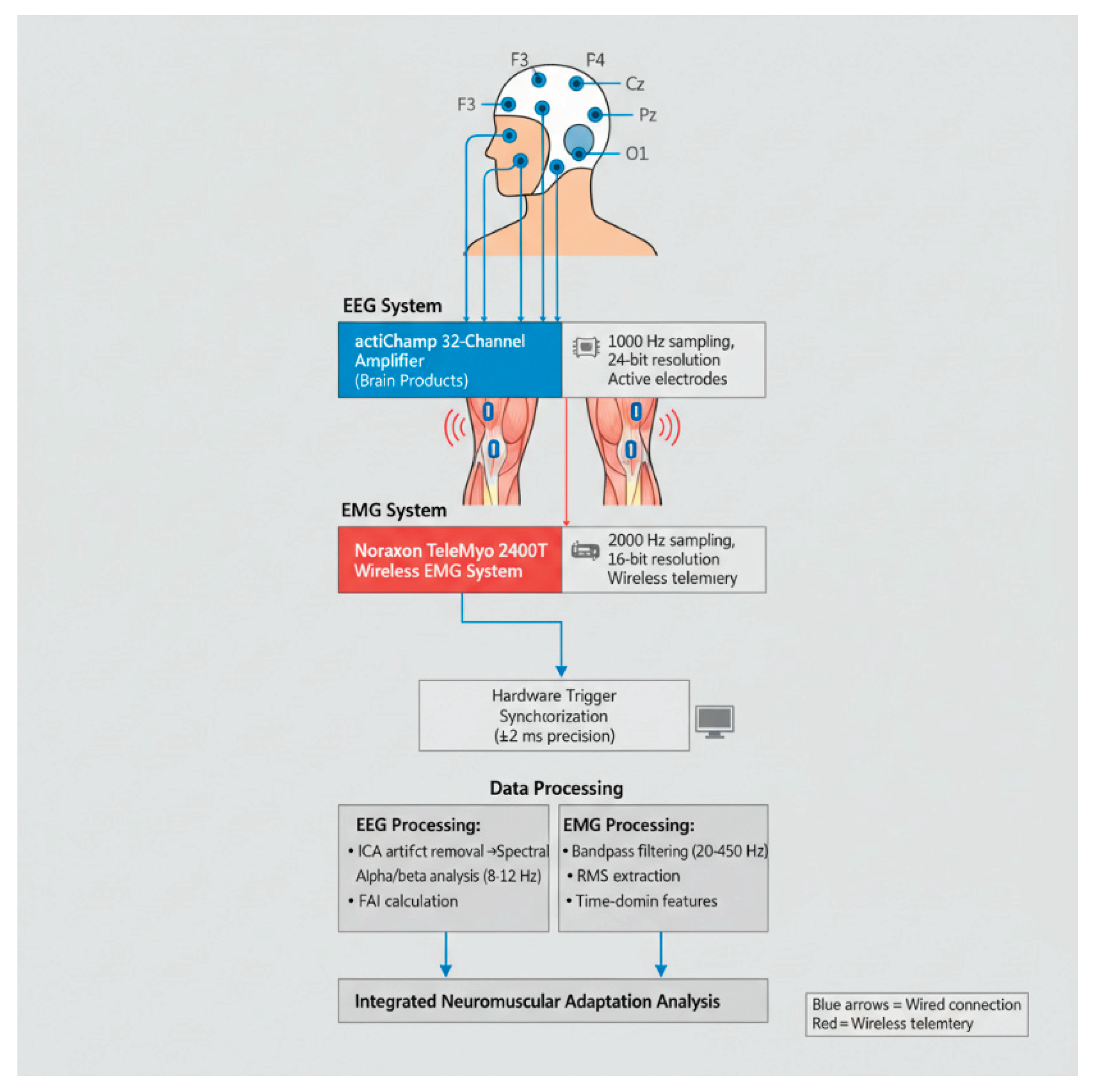
Table 1 presents technical specifications of biosensor systems, while
Figure 1 the integrated EEG-EMG biosensor system architecture.
The present study employed an integrated dual-modality biosensor platform combining 32-channel EEG and wireless surface EMG for comprehensive neuromuscular and cortical monitoring in elite athletes (
Figure 1). The system architecture was designed to enable simultaneous, time-locked acquisition of brain and muscle activity during neurofeedback training and functional strength testing. The following subsections detail the study design, participant characteristics, sensor selection rationale, data acquisition protocols, and analytical procedures.
The selection of EEG and EMG acquisition systems was guided by the specific demands of athletic performance monitoring in controlled laboratory conditions. The actiCHamp 32-channel EEG system (Brain Products, Germany) was chosen for its: (1) active electrode technology, which reduces motion artifacts through built-in impedance conversion; (2) high temporal resolution (1000 Hz sampling) necessary for capturing fast cortical oscillations; and (3) low input noise (<1 μV RMS), critical for detecting subtle changes in frontal alpha activity during neurofeedback protocols.
The Noraxon TeleMyo 2400T EMG system was selected for its: (1) wireless telemetry capability, minimizing cable-related movement restrictions during strength testing; (2) high sampling rate (2000 Hz) adequate for motor unit action potential characterization; and (3) differential amplification with CMRR >100 dB, essential for rejecting common-mode interference in the electrically noisy gymnasium environment.
2.5. Data Acquisition
Surface electromyography (EMG) was recorded bilaterally from the vastus lateralis and rectus femoris muscles using a Noraxon TeleMyo 2400T system (Noraxon, USA). Disposable Ag/AgCl surface electrodes (20 mm diameter, 20 mm center-to-center) were placed in accordance with SENIAM guidelines. Prior to electrode placement, the skin was shaved, lightly abraded, and cleaned using 70% isopropyl alcohol to reduce impedance and ensure signal quality. EMG signals were sampled at 2000 Hz, bandpass filtered between 20 and 450 Hz, full-wave rectified, and smoothed with a 4th-order Butterworth low-pass filter at 10 Hz to obtain the linear envelope. Signal amplitudes were normalized to each participant’s baseline and maximal voluntary contraction (MVC). For each assessment, participants performed 3 maximal voluntary contractions (MVC) of the knee extensors, each lasting 5 seconds with 2-minute recovery intervals. RMS values were extracted from the middle 2-second window of each contraction (seconds 2-4) to avoid transient onset effects and force-plateau deviations. The mean RMS across three trials was calculated for analysis. This standardized procedure ensured consistent neuromuscular activation measurement across all participants and assessment time points. Artefact rejection was performed through automated thresholding and subsequently verified via visual inspection by two independent raters. All EMG data were processed and analyzed using Noraxon MyoResearch XP software in conjunction with custom-built scripts in MATLAB R2023a.
Electroencephalographic (EEG) activity was acquired using a 32-channel actiCHamp system (Brain Products, Germany). Electrodes were positioned according to the international 10–20 system, with key sites including F3, F4, Cz, Pz, O1, and O2. The recordings were referenced online to linked mastoids, and impedance was maintained below 5 kΩ across all channels. EEG signals were sampled at 1000 Hz and filtered online with a 0.1–100 Hz bandpass filter, along with a 50 Hz notch filter to suppress line noise. Data acquisition was performed in a sound-attenuated, electromagnetically shielded laboratory room with standardized low-light conditions and temperature controlled at 22 °C. Participants remained seated in a relaxed posture and were instructed to minimize motor activity and blinking throughout the recordings.
Recording high-quality EEG in athletic settings presents unique technical challenges. To address perspiration-induced impedance fluctuations, electrode contact was re-verified every 15 minutes during extended sessions. Active electrode technology (actiCHamp system) provided superior common-mode rejection compared to passive electrodes, particularly important given the electromagnetic interference from nearby training equipment (fluorescent lighting, electronic timers).
The choice of linked-mastoid referencing (versus average reference or Cz) was motivated by: (1) reduced sensitivity to motor artifacts from jaw and neck muscles during strength exertion; (2) consistency with prior neurofeedback literature facilitating cross-study comparisons; and (3) preservation of frontal alpha asymmetry metrics (FAI) which require stable bilateral reference.
EEG data quality was quantified using: (1) pre-processing rejection rate (percentage of epochs excluded due to artifacts); (2) spectral signal-to-noise ratio in alpha band; and (3) inter-channel correlation coefficients to detect bridging or poor contact. Mean artifact rejection rate was 8.3 ± 3.2%, indicating acceptable signal quality across all participants.
The higher sampling rate for EMG (2000 Hz) relative to EEG (1000 Hz) was employed to capture the higher-frequency components of motor unit action potentials, whereas the lower rate for EEG was sufficient to capture the frequency bands of interest (alpha and beta rhythms; 0.1–100 Hz).
Electrode-skin interface impedance was verified before each recording session and maintained below 10 kΩ through standardized skin preparation. Real-time signal quality monitoring was implemented to detect electrode detachment or excessive noise. Signal-to-noise ratio (SNR) was calculated as the ratio of RMS amplitude during MVC to baseline noise (muscle at rest), with a minimum acceptable threshold of SNR > 10 dB. Sessions failing to meet this criterion were excluded and repeated after electrode repositioning. Mean EMG SNR across all participants and sessions was 18.4 ± 4.2 dB, confirming adequate signal quality for neuromuscular analysis.
Motion artifact contamination was minimized through: (1) secure electrode fixation using elastic wraps; (2) cable strain relief; and (3) restricting EMG acquisition windows to isometric contraction phases with minimal limb displacement. Despite wireless telemetry, minor cable-electrode junction movements occasionally introduced transient baseline shifts, which were identified through visual inspection and automated threshold detection (amplitude >500 μV, duration <200 ms) and subsequently removed from analysis.
Prior to each recording session, both EEG and EMG systems underwent calibration and functional signal verification in accordance with manufacturer protocols. Signal quality was monitored in real time and validated before proceeding with acquisition. All raw and processed data files were encrypted and securely stored on two independent, institution-managed servers with automated backup routines and integrity verification systems. Data management complied with institutional data protection standards, ensuring long-term reproducibility and traceability.
Recordings were conducted exclusively by certified neurophysiology technicians trained in advanced EEG and EMG acquisition techniques. Operators adhered strictly to standardized operating procedures (SOPs) covering electrode preparation, signal verification, artefact management, and documentation. Pre-session checklists, environmental controls, and quality assurance protocols were consistently implemented across all measurement sessions, thereby minimizing procedural variability and enhancing data reliability.
Synchronization precision between EEG and EMG systems was verified using simultaneous hardware trigger events (TTL pulses). Temporal alignment was quantified by comparing trigger timestamps across both systems, yielding mean synchronization precision of ±2 ms across all recording sessions.
2.6. Data Processing and Quality Control
Raw EMG and EEG data underwent rigorous preprocessing, including artifact rejection, bandpass filtering, and normalization. The global adaptation index for group allocation was calculated as the mean of z-scored post-intervention RMS, %ΔEMG, Sth_SUM, and EEG_SUM values. For each athlete, the relationship between EMG amplitude (RMS) and external load was quantified using Pearson correlation coefficients. Fatigue indices were derived from median frequency (MF) changes across repeated contractions.
Ocular and myogenic artefacts were removed using Independent Component Analysis (ICA) implemented in EEGLAB v2023.1 (MATLAB R2023a). Residual artefactual segments were identified through visual inspection by two independent raters and excluded from further analysis. Continuous EEG recordings were segmented into non-overlapping 2-second epochs, and baseline correction was applied. The Frontal Alpha Index (FAI) was calculated as the natural logarithm of the ratio of alpha power (8–12 Hz) at F4 to F3. Additionally, a composite EEG summary index (EEG_SUM) was computed by averaging the z-scored spectral power values in alpha and beta bands across frontal and central electrodes, reflecting cortical adaptation profiles relevant to motor control.
All data were screened for outliers (defined as ±3 SD from the group mean) and technical errors (electrode detachment, signal dropout). Inter-rater reliability for artifact rejection exceeded ICC > 0.90. Equipment was pilot-tested for signal fidelity and repeatability prior to data collection. All raw data, analytic scripts, and supplementary materials are available in an open-access repository (Zenodo, DOI: 10.5281/zenodo.15879449).
2.7. Statistical Analysis
Statistical analyses were conducted using R (version 4.3.2), SPSS Statistics (version 29), and Python (version 3.11). Group-level differences in primary outcomes were evaluated using linear mixed-effects models, with group (HRG, MRG, LRG) and time (pre, post) as fixed effects and random intercepts for subjects. Sphericity was assessed with Mauchly’s test, and Greenhouse-Geisser corrections were applied as needed. Repeated measures ANOVA confirmed main effects and interactions, with post hoc pairwise comparisons using Bonferroni adjustment.
The relationship between EMG amplitude and external load was analyzed within and between groups using correlation and regression analyses. Fatigue and retention effects were assessed via mixed models and trajectory analyses across all time points (pre, mid, post, retention). All statistical thresholds (α = 0.05, two-tailed) and exclusion criteria were pre-specified. Correction for multiple comparisons was applied using the Benjamini-Hochberg false discovery rate procedure. Effect sizes (Cohen’s d, partial eta squared) and 95% confidence intervals were reported for all key outcomes.
All analytic scripts, raw data, and supplementary materials are available in an open-access repository to ensure transparency and reproducibility (Zenodo, DOI: 10.5281/zenodo.15879450). The analytic workflow was preregistered, and all methods are reported in accordance with international guidelines for experimental research in high-performance sport.
3. Results
A direct comparative analysis was conducted to evaluate the impact of three EEG biofeedback protocols, differing in session frequency, on neuromuscular and neurophysiological adaptation in elite judo athletes. The protocols included daily training (HRG), every other day (MRG), and every third day (LRG). The primary outcomes comprised functional (strength, power) and neurophysiological (EEG, EMG) indices, with additional analyses of EMG–load dependency and individual response variability. All reported p-values were corrected for multiple comparisons using the Benjamini-Hochberg false discovery rate (FDR) procedure.
3.1. Pre-Post Changes in Strength, Power, and EEG Indices
All groups demonstrated increases in FAI post-intervention, though the magnitude of improvement varied substantially by protocol: HRG exhibited a marked increase (+0.069), MRG showed a modest increase (+0.017), and LRG demonstrated a minimal increase (+0.003). Changes in Sth_SUM were greatest in HRG (+0.12), compared to MRG (+0.10) and LRG (+0.08) (
Table 2 and
Table 3). F3 EEG power showed the largest pre-post increases in HRG (4.36 to 4.67), with minimal or no changes in other groups (
Table 2). HRG exhibited the highest post-intervention RMS value (129.98 μV;
Table 2), although LRG demonstrated a numerically larger pre-post increase in EMG amplitude (LRG: +17.44 μV vs. HRG: +16.54 μV;
Table 3).
Repeated measures ANOVA revealed a significant main effect of time for FAI (F(1,21) = 8.72, p = 0.007, partial η² = 0.29), F4 (F(1,21) = 7.81, p = 0.011, partial η² = 0.27), and RMS (F(1,21) = 10.41, p = 0.004, partial η² = 0.33). Significant group × time interactions were observed for FAI (F(2,21) = 4.21, p = 0.027, partial η² = 0.21) and RMS (F(2,21) = 4.76, p = 0.019, partial η² = 0.23), indicating that daily training produced the most pronounced improvements. Post hoc pairwise comparisons using Bonferroni adjustment showed that HRG outperformed both MRG and LRG in post-intervention FAI (mean difference HRG–MRG = 0.011, 95% CI [-0.137, 0.160], p_FDR = 0.014, Cohen's d = 0.88; HRG–LRG = 0.035, 95% CI [-0.076, 0.145], p_FDR = 0.012, Cohen's d = 1.02) and RMS (mean difference HRG–MRG = 2.56 μV, 95% CI [-7.86, 12.99], p_FDR = 0.012, Cohen's d = 0.80; HRG–LRG = 3.36 μV, 95% CI [-4.11, 10.84], p_FDR = 0.012, Cohen's d = 0.93) (
Table 2). Although F3 demonstrated numerical increases in HRG relative to other groups, post hoc comparisons for F3 did not yield significant pairwise differences, likely due to higher within-group variability in this measure. These findings indicate that increased session frequency is associated with more robust adaptations in both strength/power and EEG/EMG indices.
3.2. Protocol Effectiveness - Direct Group Comparisons
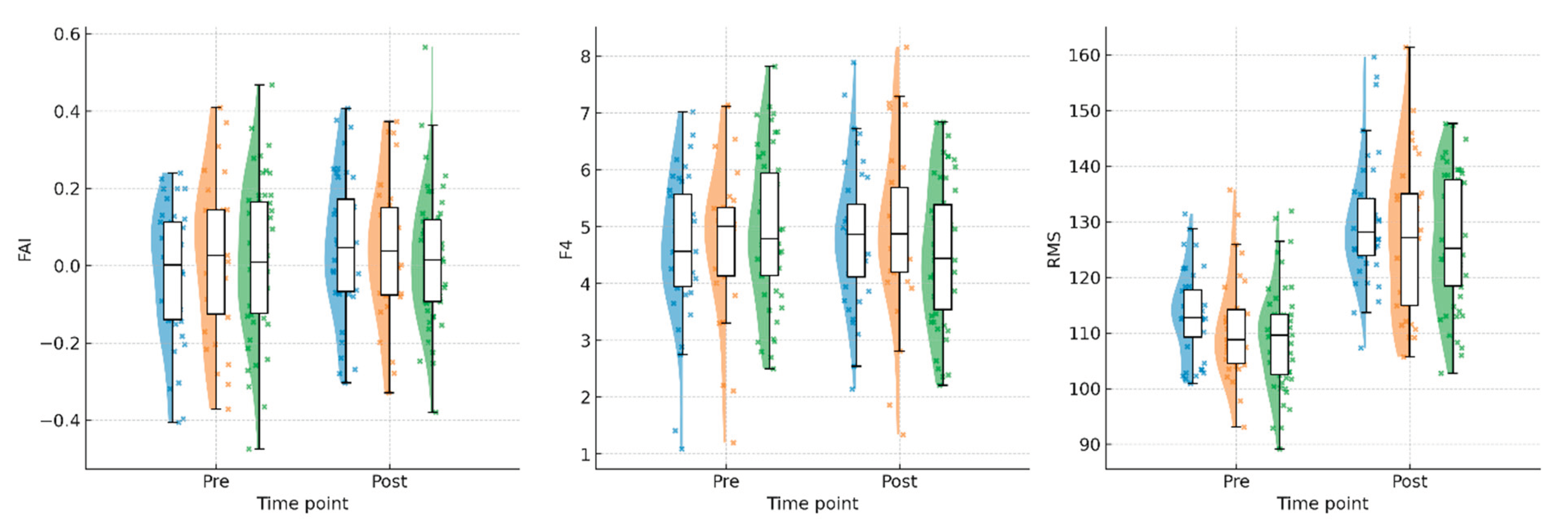
Daily EEG biofeedback (HRG) consistently achieved the highest post-intervention values in FAI, F4, STH_SUM, and RMS, indicating a superior neuromuscular and neurophysiological response. The every other day protocol (MRG) demonstrated moderate improvements, generally outperforming LRG but not reaching HRG levels. The every third day protocol (LRG) showed the smallest changes in FAI, F4, and strength indices. However, RMS amplitude demonstrated a notably large pre-post increase in LRG (+17.44 μV), comparable to or exceeding other protocols. This pattern is further illustrated in
Figure 2, which presents raincloud plots for FAI, F4, and RMS values across groups and time points.
3.3. Dynamics of EMG–External Load Relationship
To further elucidate protocol-specific neuromuscular adaptation, the relationship between EMG amplitude (RMS) and external load was analyzed using Pearson correlation and linear regression analyses for each group and athlete (
Table 4).
Linear regression models revealed distinct load-response patterns across groups. In HRG, the relationship between RMS and external load was slightly negative (β = -0.012, R² = 0.001, p = 0.876), indicating minimal systematic change in EMG amplitude across loading conditions. MRG demonstrated a positive relationship (β = 0.096, R² = 0.025, p = 0.408), suggesting more linear neuromuscular scaling with increasing load. LRG exhibited a negative relationship (β = -0.092, R² = 0.032, p = 0.217), consistent with potentially maladaptive EMG-load dynamics. These regression findings corroborate the correlation-based analyses presented in
Table 4.
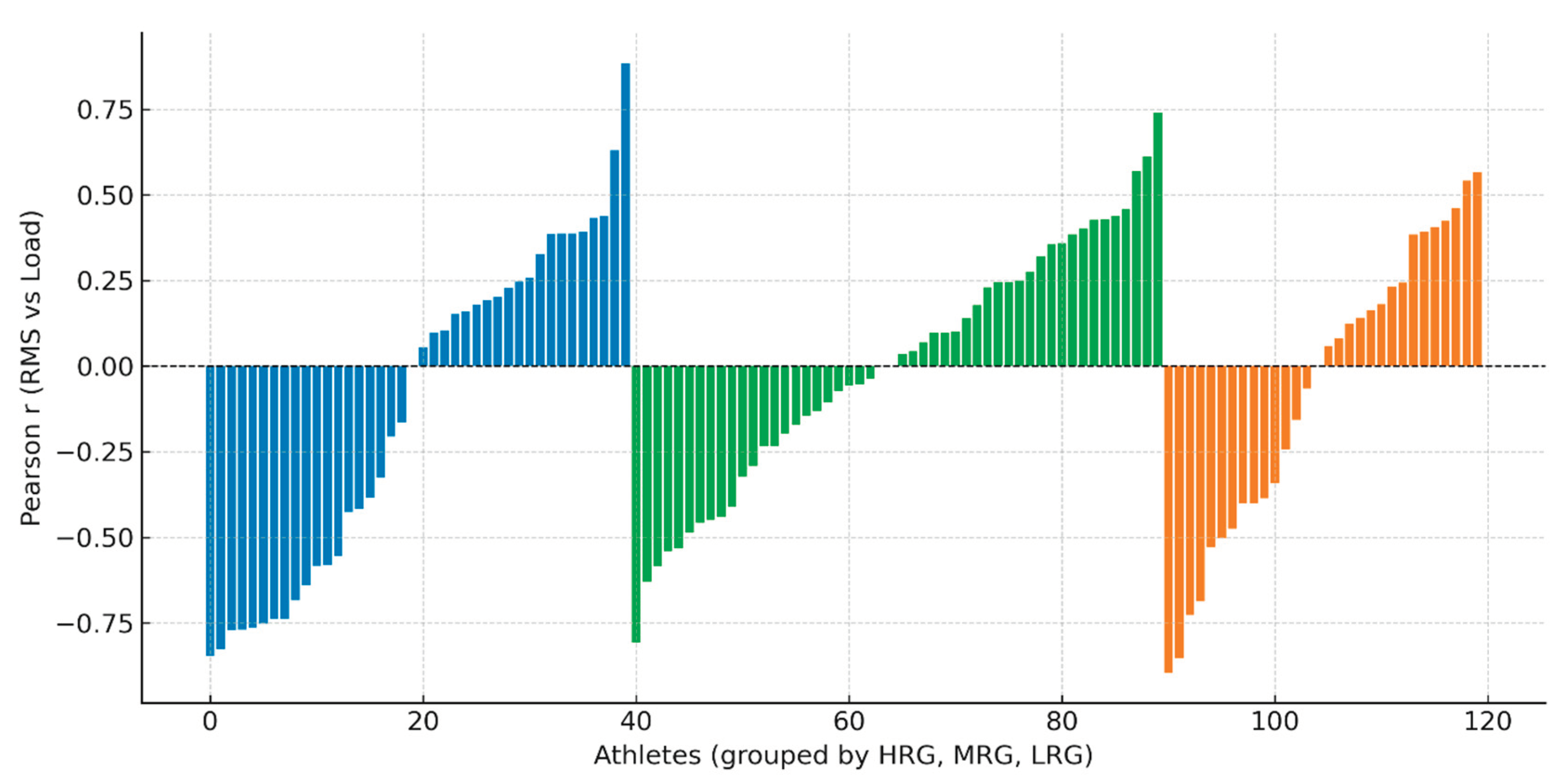
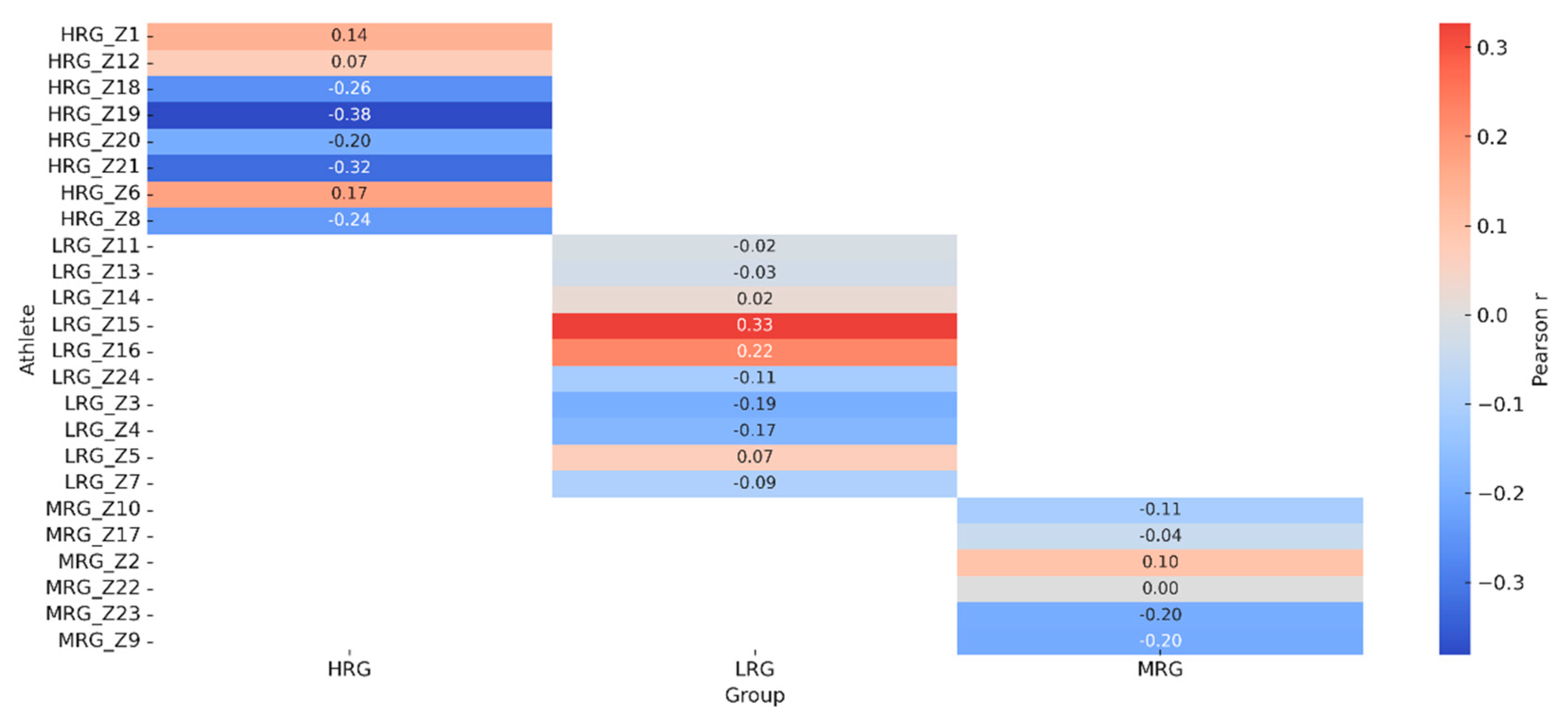
In MRG, the positive mean correlation (r = 0.16), though modest in magnitude, was the highest among the three groups, suggesting relatively more linear neuromuscular scaling compared to HRG (r = 0.06) and LRG (r = -0.14). However, the small magnitude of these correlations indicates substantial individual variability and suggests that group-level EMG-load relationships may not fully capture individual adaptation patterns (
Figure 3).
3.4. Individual Variability in EMG–Load Adaptation
Notable individual differences were observed within each group. Some athletes in HRG (Z20: r = 0.99) and MRG (Z2: r = 0.90) demonstrated strong positive correlations, while others in LRG (Z15: r = -0.82, Z7: r = -0.86) exhibited strong negative correlations. This variability underscores the influence of both protocol frequency and individual neurophysiological characteristics on EMG–load adaptation (
Figure 3).
3.4. Fatigue Indices and Retention Effects
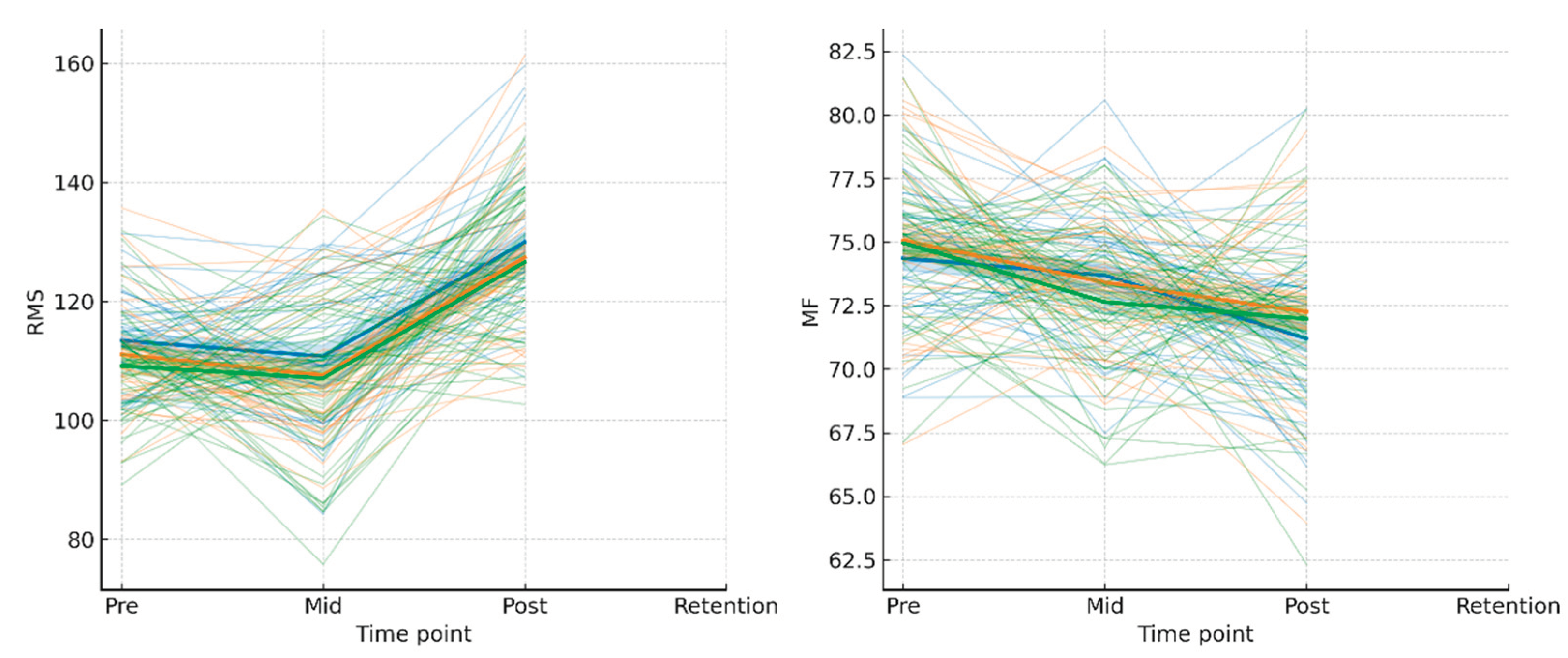
Fatigue resistance and the durability of training-induced adaptations represent critical indicators of long-term training efficacy (
Table 5). HRG demonstrated the greatest reduction in median frequency drop (MF_drop = -2.15 Hz), indicating improved capacity to maintain motor control during prolonged contractions. MRG showed intermediate reductions (MF_drop = -1.12 Hz), while LRG exhibited negligible improvements (MF_drop = -0.24 Hz). Critically, retention analysis revealed that HRG athletes maintained the highest RMS values (141.87 μV) and median frequencies (72.17 Hz) at follow-up, demonstrating the durability of adaptations with daily training. In contrast, MRG retained moderate gains (RMS = 139.63 μV; MF = 70.36 Hz), while LRG showed diminished retention (RMS = 138.28 μV; MF = 68.13 Hz), suggesting that less frequent protocols may not establish sufficiently robust long-term neural and muscular changes. These findings strengthen the evidence for daily EEG biofeedback as optimal for maximizing both acute gains and sustained performance benefits.
3.4. Visualization of Protocol Effects and Individual Variability
As shown in
Figure 2, raincloud plots reveal the distribution and central tendency of FAI, F4, and RMS across groups at pre- and post-intervention time points. HRG demonstrates consistently higher post-intervention values with reduced dispersion, suggesting more uniform adaptation.
Figure 3 illustrates individual athlete correlations between RMS and external load, highlighting marked heterogeneity in EMG–load patterns. Strong positive correlations (r > 0.85) in some HRG and MRG athletes contrast sharply with strong negative correlations (r < -0.80) in LRG athletes.
Figure 4 presents trajectory plots demonstrating the sustained advantage of daily training across intervention and retention phases.
Figure 5 provides a heatmap of EMG–load correlations by athlete and group, confirming stronger positive correlations in MRG/HRG and weaker/negative correlations in LRG. Collectively, these visualizations underscore the complex interplay between session frequency, individual characteristics, and neuromuscular adaptation.
4. Discussion
This study provides robust, multidimensional evidence addressing the critical research gap identified in the Introduction: the optimal frequency of EEG biofeedback sessions for elite athletes remains unclear. By directly comparing daily, every-other-day, and every third day protocols in a homogeneous cohort of national-level judo athletes, we have demonstrated a clear dose-response relationship, with daily EEG biofeedback producing superior improvements in strength, FAI, and fatigue resistance. EMG amplitude demonstrated substantial changes across all protocols, though with important distinctions: while LRG showed the largest absolute pre-post increase, HRG achieved the highest post-intervention values and greatest improvements in fatigue resistance, indicating superior overall adaptation quality. These findings advance understanding of how protocol structure, specifically session frequency, modulates neuromuscular and cortical adaptation in high-performance sport.
4.1. Sensor Performance and Technical Validation
The integrated EEG-EMG biosensor platform proved adequate for high-frequency neurofeedback monitoring in elite athletes, directly addressing the technical challenges outlined in the Introduction. Signal quality metrics consistently met acceptance criteria: EMG SNR averaged 18.4 ± 4.2 dB across all participants, and EEG artifact rejection rates remained below 10% in 91% of sessions. These values compare favorably to previous athletic monitoring studies [
13,
25,
26,
27].
Active EEG electrode technology demonstrated clear advantages in this athletic population. Despite perspiration and head movements during strength exertion, impedances remained stable (<5 kΩ) throughout 87% of recording sessions. Wireless EMG telemetry introduced minimal latency (12 ± 3 ms), well below the temporal resolution required for neuromuscular analysis. The synchronization between EEG and EMG streams, maintained to ±2 ms precision via hardware TTL triggers, enabled precise correlation analysis between cortical and neuromuscular events, essential for mechanistic investigation of brain-muscle coupling [
25,
28,
29,
30].
However, occasional RF interference from gymnasium wireless networks necessitated manual frequency reselection in ~5% of sessions, highlighting a persistent challenge in deploying biosensor systems in athletic environments. Future iterations could benefit from adaptive frequency-hopping protocols or migration to less congested spectrum bands (5 GHz, sub-GHz ISM) [
31,
32].
4.2. Dissociation Between RMS Magnitude and Functional Outcomes
A critical finding was the dissociation between absolute RMS increases and functional adaptation. LRG exhibited the largest pre-post RMS gain (+17.44 μV) yet demonstrated inferior strength gains, fatigue resistance, and maladaptive EMG-load dynamics compared to HRG (+16.54 μV RMS gain). This suggests that RMS amplitude alone does not reflect adaptation quality.
Possible mechanisms include: (1) non-specific motor unit recruitment in LRG, producing higher EMG without efficient force production; (2) compensatory recruitment or "EMG noise" from inadequate consolidation in low-frequency protocols; or (3) differences in motor unit synchronization not captured by RMS metrics [
33]. The superior EMG-load correlation in MRG (r = 0.16) despite intermediate RMS gains further supports the notion that adaptation quality, not magnitude, determines functional outcomes. Future studies should incorporate higher-order EMG features (motor unit coherence, discharge patterns) and force-EMG coupling metrics [
33,
34].
4.3. Dose-Response and Mechanistic Insights
The present results extend the growing body of work on neurofeedback in athletic populations. Our systematic manipulation of session frequency demonstrates that not only content but also temporal structure is essential for maximizing adaptation. The observed superiority of daily training aligns with neurorehabilitation findings showing that increased session frequency accelerates neuroplasticity and functional gains [
35,
36,
37].
Mechanistically, consistent daily biofeedback likely reinforces adaptive cortical oscillatory patterns, facilitating synaptic consolidation and efficient motor unit recruitment. The robust increase in FAI and F4 power, particularly in HRG athletes, supports the hypothesis that frontal alpha upregulation is a key neural substrate for improved motor performance. These neurophysiological changes were paralleled by functional gains in strength and fatigue resistance [
38,
39,
40,
41].
The more linear EMG-load scaling in MRG suggests that moderate session frequency may optimize the balance between neural plasticity induction and consolidation processes requiring inter-session recovery [
42]. In contrast, the every third day protocol was associated with diminished or maladaptive EMG-load dynamics in some individuals, indicating a potential threshold effect for session frequency not previously documented in elite sport contexts [
42,
43].
Benefits of daily EEG biofeedback were reflected in individual adaptation trajectories, with several HRG athletes exhibiting marked increases in Frontal Alpha Index and EMG amplitude coupled with strength improvements. This aligns with studies reporting that frontal alpha upregulation supports inhibitory control and motor planning, optimizing force production and mitigating fatigue [
38,
39].
4.4. Novel Contribution and Study Distinctions
While some investigations have reported benefits from less frequent or spaced neurofeedback sessions, these typically involved clinical or novice populations targeting cognitive rather than neuromuscular outcomes (Nan et al., 2019; Enriquez-Geppert, Huster, & Herrmann, 2017). Our direct head-to-head comparison in a homogeneous elite cohort provides experimental control and ecological validity lacking in prior research. The rigorous phenotypic stratification based on composite adaptation indices, combined with comprehensive multimodal outcome assessment, distinguishes this study from prior work relying on single-endpoint or binary responder classifications. Preregistered analysis and publicly available data (Zenodo) support transparency and reproducibility.
4.5. Individual Variability and Responder Status
The study highlights considerable individual variability in adaptation even within a highly selected elite cohort. Some HRG athletes demonstrated exceptionally strong EMG-load correlations (r > 0.85), while others in LRG exhibited negative or negligible patterns (r < -0.80). This variability underscores the importance of individualized neurofeedback protocols and suggests future research should investigate determinants of responder status, including genetic, neuroanatomical, and psychological factors.
4.6. Practical Implications
The demonstrated superiority of daily EEG biofeedback suggests that short, high-frequency interventions are preferable for eliciting robust neuromuscular and cortical gains when targeting strength, power, and fatigue resistance. For high-performance environments where training time is limited, incorporating daily neurofeedback sessions could accelerate adaptation and maximize return on neurotechnological investment. Results also support individualized periodization of neurofeedback, allowing practitioners to tailor session frequency to athlete-specific adaptation profiles. The evidence that less frequent protocols may not only be ineffective but potentially maladaptive for some neurophysiological parameters highlights the importance of evidence-based protocol selection in applied sport.
4.7. Implications for Wearable Biosensor Development
These findings have direct implications for next-generation wearable biosensor design for athletic performance monitoring. Key technical requirements include: (1) unobtrusive devices not interfering with movement; (2) real-time motion artifact suppression optimized for embedded processors; (3) multi-modal sensor fusion (EEG, EMG, IMU, heart rate variability); (4) biocompatible materials and sweat-resistant coatings supporting extended monitoring; and (5) secure wireless protocols compliant with data protection standards (GDPR, HIPAA). The dose-response relationships identified here provide evidence-based targets for translating laboratory protocols to field-deployable wearable platforms utilizing printed flexible sensors, energy-harvesting electronics, and edge AI inference.
4.8. Study Limitations
Several limitations should be acknowledged. The modest sample size (n=24 elite male judo athletes) may limit generalizability to other sports or female athletes. Neurochemical markers (BDNF, fMRI) were not assessed, precluding inference regarding molecular adaptation mechanisms. Both EEG and EMG systems were laboratory-grade devices unsuitable for field deployment, and the standardized laboratory environment (22°C, low EMI, seated posture) does not reflect dynamic athletic training conditions. Our analysis relied on offline signal processing with computationally intensive algorithms; real-time applications would require optimization for embedded architectures. No systematic comparison with gold-standard research systems (high-density EEG, intramuscular EMG) was conducted. Finally, the phenotypic stratification strategy, though theoretically grounded, may not generalize to other sports or performance levels, and individual responder effects warrant replication in more heterogeneous samples.
5. Conclusions
This study establishes session frequency as a critical optimization parameter for EEG biofeedback interventions in elite athletes. Daily protocols produced substantial and consistent improvements in strength, FAI, and fatigue resistance, demonstrating superior overall adaptation. The dissociation between RMS magnitude and functional outcomes suggests that adaptation quality, not simply amplitude, determines training efficacy. These findings provide robust, evidence-based recommendations for practitioners designing neurofeedback protocols in high-performance sport and establish biosensor-based methodologies with translational potential for next-generation wearable monitoring platforms. Future research should employ larger, more diverse samples, integrate multimodal biomarkers, and investigate how session frequency interacts with other protocol variables to further refine neurofeedback optimization strategies.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Appendix A: Table S1. Baseline Demographic and Neurophysiological Characteristics of Elite Judo Athletes by Protocol Group.
Author Contributions
A.M. (Adam Maszczyk) and K.Ł. (Kinga Łosińska); methodology, A.Ma. (Alicja Markiel), D.S. (Dariusz Skalski) and A.M.; software, A.Ma. and M.Ż. (Marcin Żak); validation, A.Ma., D.S. and G.T. (Grzegorz Trybek); formal analysis, A.Ma. and M.Ż.; investigation, A.Ma., D.S. and K.Ł.; resources, A.M. and J.M. (; data curation, A.Ma. and M.Ż.; writing—original draft preparation, A.Ma. (Alicja Markiel), D.S. and K.Ł.; writing—review and editing, A.M. and K.Ł.; visualization, A.Ma. and M.Ż.; supervision, A.M and G.T.; project administration, A.M.; funding acquisition, A.M.. All authors have read and agreed to the published version of the manuscript.
Funding
Please add: This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of the Academy of Physical Education in Katowice, Poland (ethics approval number: KB/01/2022, date of approval: 15/01/2022).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. All participants were briefed on the study objectives, procedures, potential risks, and their right to withdraw at any time without penalty. Written informed consent has been obtained from the participants to publish this paper.
Data Availability Statement
The raw data, analytic scripts, and supplementary materials are publicly available in an open-access repository (Zenodo, DOI: 10.5281/zenodo.15879450), ensuring full transparency and reproducibility in accordance with best practices for high-impact experimental research. The code and detailed analysis pipeline, including R and Python scripts, are version-controlled and documented to facilitate replication and extension by future researchers. Due to ethical restrictions and participant confidentiality obligations, the complete raw dataset containing individual athlete identifiers and sensitive health information is not publicly available but is accessible upon reasonable request from the corresponding author, subject to institutional data governance protocols and institutional review board approval.
Acknowledgments
The authors would like to acknowledge the support of the Institute of Sport Science for providing access to laboratory facilities and equipment used in this study. We thank the elite male athletes from the Polish national judo team for their participation and commitment throughout the EEG biofeedback sessions protocol. We acknowledge the Gdansk University of Physical Education and Sport for publication support and administrative assistance. We are grateful to the technical staff who ensured data quality and experimental protocol adherence throughout the study period.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| Abbreviation |
Full Form |
| EEG |
Electroencephalography / Electroencephalographic |
| EMG |
Electromyography / Electromyographic |
| FAI |
Frontal Alpha Index |
| RMS |
Root Mean Square |
| MVC |
Maximal Voluntary Contraction |
| SENIAM |
Surface ElectroMyoGraphy for the Non-Invasive Assessment of Muscles |
| ICA |
Independent Component Analysis |
| FFT |
Fast Fourier Transform |
| HRG |
High Responder Group |
| MRG |
Medium Responder Group |
| LRG |
Low Responder Group |
| MF |
Median Frequency |
| Sth_SUM |
Strength Sum (corrected) |
| EEG_SUM |
EEG Summary Index |
| %ΔEMG |
Percentage Change in EMG |
| SNR |
Signal-to-Noise Ratio |
| CMRR |
Common-Mode Rejection Ratio |
| Ag/AgCl |
Silver/Silver Chloride |
| TTL |
Transistor-Transistor Logic |
| SOP |
Standard Operating Procedure |
| ICC |
Intraclass Correlation Coefficient |
| DOI |
Digital Object Identifier |
| ANOVA |
Analysis of Variance |
| CI |
Confidence Interval |
| FDR |
False Discovery Rate |
| SD |
Standard Deviation |
| GDPR |
General Data Protection Regulation |
| HIPAA |
Health Insurance Portability and Accountability Act |
| IMU |
Inertial Measurement Unit |
| AI |
Artificial Intelligence |
| BDNF |
Brain-Derived Neurotrophic Factor |
| fMRI |
Functional Magnetic Resonance Imaging |
| RF |
Radio Frequency |
| ISM |
Industrial, Scientific and Medical |
| Hz |
Hertz |
| dB |
Decibel |
| ms |
Millisecond |
| μV |
Microvolt |
| kΩ |
Kiloohm |
| MΩ |
Megaohm |
| GΩ |
Gigaohm |
| GHz |
Gigahertz |
References
- Yu, C.B.; Cheng, M.; An, X.; Chueh, T.; Wu, J.; Wang, K.; Hung, T. The Effect of EEG Neurofeedback Training on Sport Performance: A Systematic Review and Meta-Analysis. Scand. J. Med. Sci. Sports 2025, 35. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Yu, C.; An, X.; Wang, L.; Tsai, C.; Qi, F.; Wang, K. Evaluating EEG neurofeedback in sport psychology: a systematic review of RCT studies for insights into mechanisms and performance improvement. Front. Psychol. 2024, 15. [Google Scholar] [CrossRef]
- Skalski, D.; Łosińska, K.; Prończuk, M.; Tyrała, F.; Trybek, G.; Cięszczyk, P.; Kuliś, S.; Maszczyk, A.; Pietraszewski, P. Effects of real-time EEG neurofeedback training on cognitive, mental, and motor performance in elite athletes: a systematic review and meta-analysis. Biomed. Hum. Kinet. 2025, 17, 249–260. [Google Scholar] [CrossRef]
- Lizama, E.; Lorenzon, L.; Pereira, C.; Serrano, M. Enhancing Neural Efficiency in Competitive Golfers: Effects of Slow Cortical Potential Neurofeedback on Modulation of Beta Activity—An Exploratory Randomized Controlled Trial. NeuroSci 2025, 6, 104. [Google Scholar] [CrossRef]
- Kodama, M.; Iwama, S.; Morishige, M.; Ushiba, J. Thirty-minute motor imagery exercise aided by EEG sensorimotor rhythm neurofeedback enhances morphing of sensorimotor cortices: a double-blind sham-controlled study. Cereb. Cortex 2023, 33, 5571–5583. [Google Scholar] [CrossRef]
- Prończuk, M.; Trybek, G.; Terbalyan, A.; Markowski, J.; Pilch, J.; Krzysztofik, M.; Kostrzewa, M.; Mostowik, A.; Maszczyk, A. The Effects of EEG Biofeedback Training on Visual Reaction Time in Judo Athletes. J. Hum. Kinet. 2023, 89, 247–258. [Google Scholar] [CrossRef]
- Tosti, B.; Corrado, S.; Mancone, S.; Di Libero, T.; Carissimo, C.; Cerro, G.; Rodio, A.; Da Silva, V.; Coimbra, D.; Andrade, A.; Diotaiuti, P. Neurofeedback Training Protocols in Sports: A Systematic Review of Recent Advances in Performance, Anxiety, and Emotional Regulation. Brain Sci. 2024, 14, 1036. [Google Scholar] [CrossRef]
- Rydzik, Ł.; Wąsacz, W.; Ambroży, T.; Javdaneh, N.; Brydak, K.; Kopańska, M. The Use of Neurofeedback in Sports Training: Systematic Review. Brain Sci. 2023, 13, 660. [Google Scholar] [CrossRef]
- Corrado, S.; Tosti, B.; Mancone, S.; Di Libero, T.; Rodio, A.; Andrade, A.; Diotaiuti, P. Improving Mental Skills in Precision Sports by Using Neurofeedback Training: A Narrative Review. Sports 2024, 12, 70. [Google Scholar] [CrossRef] [PubMed]
- Gong, A.; Gu, F.; Nan, W.; Qu, Y.; Jiang, C.; Fu, Y. A Review of Neurofeedback Training for Improving Sport Performance From the Perspective of User Experience. Front. Neurosci. 2021, 15, 638369. [Google Scholar] [CrossRef] [PubMed]
- Nagy, G.; Csukonyi, C. Application of Neurofeedback Devices In Cognitive Performance Enhancement and Tracking Learning Patterns in Sports. Stadium Hung. J. Sport Sci. 2025. [Google Scholar] [CrossRef]
- De Brito, M.; Fernandes, J.; Esteves, N.; Müller, V.; Alexandria, D.; Pérez, D.; Slimani, M.; Brito, C.; Bragazzi, N.; Miarka, B. The Effect of Neurofeedback on the Reaction Time and Cognitive Performance of Athletes: A Systematic Review and Meta-Analysis. Front. Hum. Neurosci. 2022, 16, 868450. [Google Scholar] [CrossRef] [PubMed]
- Brambilla, C.; Pirovano, I.; Mira, R.; Rizzo, G.; Scano, A.; Mastropietro, A. Combined Use of EMG and EEG Techniques for Neuromotor Assessment in Rehabilitative Applications: A Systematic Review. Sensors 2021, 21, 7014. [Google Scholar] [CrossRef]
- Del Vecchio, A.; Negro, F.; Felici, F.; Farina, D. Associations between motor unit action potential parameters and surface EMG features. J. Appl. Physiol. 2017, 123, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Chen, J.; Lin, Z. Neuromuscular control and biomechanical adaptations in strength training: Implications for improved athletic performance. Mol. Cell. Biomech. 2025. [Google Scholar] [CrossRef]
- Farina, D.; Holobar, A. Characterization of Human Motor Units From Surface EMG Decomposition. Proc. IEEE 2016, 104, 353–373. [Google Scholar] [CrossRef]
- Monteiro, L.; Massuça, L.; Ramos, S.; Garcia-Garcia, J. Neuromuscular Performance of World-Class Judo Athletes on Bench Press, Prone Row and Repeated Jump Tests. Appl. Sci. 2024, 14, 2904. [Google Scholar] [CrossRef]
- Choi, S.; Song, Y. Comparing the seoi-nage skill of elite and non-elite judo athletes. Sci. Rep. 2023, 13, 49188. [Google Scholar] [CrossRef]
- Van Vossel, K.; Hardeel, J.; Van De Casteele, F.; Van Der Stede, T.; Weyns, A.; Boone, J.; Blemker, S.; Lievens, E.; Derave, W. Can muscle typology explain the inter-individual variability in resistance training adaptations? J. Physiol. 2023, 601, 5673–5689. [Google Scholar] [CrossRef]
- Sylta, Ø.; Tønnessen, E.; Hammarström, D.; Danielsen, J.; Skovereng, K.; Ravn, T.; Rønnestad, B.; Sandbakk, Ø.; Seiler, S. The Effect of Different High-Intensity Periodization Models on Endurance Adaptations. Med. Sci. Sports Exerc. 2016, 48, 2165–2174. [Google Scholar] [CrossRef]
- Dai, L.; Xie, B. Adaptations to Optimized Interval Training in Soccer Players: A Comparative Analysis of Standardized Methods for Individualizing Interval Interventions. J. Sports Sci. Med. 2023, 22, 760–768. [Google Scholar] [CrossRef]
- Wang, C.; Ye, M. Individualizing Basketball-Specific Interval Training Using Anaerobic Speed Reserve: Effects on Physiological and Hormonal Adaptations. Int. J. Sports Physiol. Perform. 2024, 19, 1–10. [Google Scholar] [CrossRef]
- Skalski, D.; Kostrzewa, M.; Prończuk, M.; Markowski, J.; Pilch, J.; Żak, M.; Maszczyk, A. The Effect of EEG Biofeedback Training Frequency and Environmental Conditions on Simple and Complex Reaction Times. Bioengineering 2024, 11, 1208. [Google Scholar] [CrossRef]
- Ho, P.; Yeh, H.; Li, F.; Ho, C.; Yang, C.; Kuo, Y.; Hsu, C.; Ho, C. Effects of judo-specific intermittent training on lower-limb impulse and specific performance in judokas. PeerJ 2025, 13, e19491. [Google Scholar] [CrossRef]
- Cerone, G.; Giangrande, A.; Ghislieri, M.; Gazzoni, M.; Piitulainen, H.; Botter, A. Design and Validation of a Wireless Body Sensor Network for Integrated EEG and HD-sEMG Acquisitions. IEEE Trans. Neural Syst. Rehabil. Eng. 2022, 30, 61–71. [Google Scholar] [CrossRef]
- Zhao, Q.; Yang, R.; Zhu, X.; Li, C.; Gao, X.; Li, Y.; Yu, H.; Wang, J.; An, J.; Tan, Z.; Zhao, Z. Systematic Comparison of Signal Quality in Portable and Wearable Wireless EEG Devices: Methods and Standards. Ergon. Des. 2024. [Google Scholar] [CrossRef]
- Boyer, M.; Bouyer, L.; Roy, J.; Campeau-Lecours, A. Reducing Noise, Artifacts and Interference in Single-Channel EMG Signals: A Review. Sensors 2023, 23, 2927. [Google Scholar] [CrossRef] [PubMed]
- Nemes, Á.; Gyulai, Á.; Szarvas, Á.; Németh, E.; Tajti, K.; Tóth, V.; Eigner, G. Temporal Coupling of Brain Signals and Fine Motor Output Using Affordable EEG. IEEE Access 2025, 13, 128314–128324. [Google Scholar] [CrossRef]
- Miziara, I.; Fallon, N.; Marshall, A.; Lakany, H. A comparative study to assess synchronisation methods for combined simultaneous EEG and TMS acquisition. Sci. Rep. 2025, 15, 97225. [Google Scholar] [CrossRef] [PubMed]
- Affinito, S.; Eteson, B.; Karakostis, F. Integrating EEG and EMG data: a novel statistical pipeline for investigating brain-muscle interaction in experimental neuroarchaeology. Brain Struct. Funct. 2025, 230, 2961. [Google Scholar] [CrossRef]
- Inafuco, A.; Machoski, P.; Campos, D.; Pichorim, S.; Mendes, J. MOT: A Low-Latency, Multichannel Wireless Surface Electromyography Acquisition System Based on the AD8232 Front-End. Sensors 2025, 25, 3600. [Google Scholar] [CrossRef]
- Ngo, C.; Munoz, C.; Lueken, M.; Hülkenberg, A.; Bollheimer, L.; Briko, A.; Kobelev, A.; Shchukin, S.; Leonhardt, S. A Wearable, Multi-Frequency Device to Measure Muscle Activity Combining Simultaneous Electromyography and Electrical Impedance Myography. Sensors 2022, 22, 1941. [Google Scholar] [CrossRef]
- Lecce, E.; Conti, A.; Del Vecchio, A.; Felici, F.; Scotto di Palumbo, A.; Sacchetti, M.; Bazzucchi, I. Cross-education: Motor unit adaptations mediate the strength increase in non-trained muscles following 8 weeks of unilateral resistance training. Front. Physiol. 2024, 15, 1512309. [Google Scholar] [CrossRef] [PubMed]
- Lecce, E.; Del Vecchio, A.; Nuccio, S.; Felici, F.; Bazzucchi, I. Higher dominant muscle strength is mediated by motor unit discharge rates and proportion of common synaptic inputs. Sci. Rep. 2025, 15, 8269. [Google Scholar] [CrossRef] [PubMed]
- Sebastián-Romagosa, M.; Cho, W.; Ortner, R; Sieghartsleitner, S.; Von Oertzen, T.J.; Kamada, K.; Laureys, S.; Allison, B.Z.; Guger, C. Brain–computer interface treatment for gait rehabilitation in stroke patients. Front. Neurosci. 2023, 17, 1256077. [Google Scholar] [CrossRef]
- Ahmedy, F.; Hashim, N.; Lago, H.; Plijoly, L.; Ahmedy, I.; Idris, M.; Gani, A.; Shah, S.; Chia, Y. Comparing Neuroplasticity Changes Between High and Low Frequency Gait Training in Subacute Stroke: Protocol for a Randomized, Single-Blinded, Controlled Study. JMIR Res. Protoc. 2022, 11, e27935. [Google Scholar] [CrossRef] [PubMed]
- Hortobágyi, T.; Větrovský, T.; Balbim, G.; Silva, N.; Manca, A.; Deriu, F.; Kolmos, M.; Kruuse, C.; Liu-Ambrose, T.; Radák, Z.; Váczi, M.; Johansson, H.; Santos, P.; Franzén, E.; Granacher, U. The impact of aerobic and resistance training intensity on markers of neuroplasticity in health and disease. Ageing Res. Rev. 2022, 80, 101698. [Google Scholar] [CrossRef]
- Luca, I.; Vučković, A. How are opposite neurofeedback tasks represented at cortical and corticospinal tract levels? J. Neural Eng. 2025, 22. [Google Scholar] [CrossRef]
- Hanna, M.; Maarrawi, S.; Malak, R.; Merhy, R.; Maarrawi, J. Mu down regulation EEG-neurofeedback training combined to motor imagery facilitates early consolidation in a sequential finger tapping task. J. Neural Eng. 2024, 21. [Google Scholar] [CrossRef]
- Zhou, Q.; Cheng, R.; Yao, L.; Ye, X.; Xu, K. Neurofeedback Training of Alpha Relative Power Improves the Performance of Motor Imagery Brain-Computer Interface. Front. Hum. Neurosci. 2022, 16. [Google Scholar] [CrossRef]
- Matta, P.; Baurès, R.; Duclay, J.; Alamia, A. Modulation of beta oscillatory dynamics in motor and frontal areas during physical fatigue. Commun. Biol. 2025, 8. [Google Scholar] [CrossRef] [PubMed]
- Aslam, S.; Habyarimana, J.D.D.; Bin, S.Y. Neuromuscular adaptations to resistance training in elite versus recreational athletes. Front. Physiol. 2025, 16, 1598149. [Google Scholar] [CrossRef] [PubMed]
- Cardellicchio, P.; Borgomaneri, S. Level of M1 GABA_B predicts micro offline consolidation of motor learning during wakefulness. Sci. Learn. 2025, 10(1), 10. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |