1. Introduction
Splenic marginal zone lymphoma (SMZL) is a rare, indolent B-cell non-Hodgkin lymphoma characterized by primary involvement of the spleen, bone marrow, and peripheral blood, typically presenting in older adults with splenomegaly and cytopenias. [
1,
2,
3] Accounting for less than 2% of all lymphoid malignancies, SMZL exhibits a highly heterogeneous clinical course, ranging from prolonged survival with minimal intervention to progressive disease requiring systemic therapy [
4,
5]. Despite its generally favorable prognosis compared with aggressive lymphomas, long-term outcomes are influenced by a complex interplay of patient characteristics, disease biology, and treatment selection [
4,
5,
6,
7].
Management of SMZL has evolved substantially over the past two decades. Historically, splenectomy played a central role in both diagnosis and symptom control, often producing durable hematologic responses [
4,
5]. More recently, the introduction of immunochemotherapy and anti-CD20–based regimens has shifted practice patterns toward non-surgical management, particularly for patients with advanced age, comorbidities, or disseminated disease [
5,
8]. However, owing to the rarity of SMZL, prospective randomized trials are limited, and clinical decision-making frequently relies on retrospective studies, small series, and expert consensus rather than high-level evidence [
4,
5,
8].
In addition to disease- and treatment-related factors, sociodemographic characteristics such as sex, race, ethnicity, marital status, and socioeconomic context have emerged as important determinants of survival across multiple hematologic malignancies [
9]. These factors may reflect differences in baseline health, comorbidity burden, access to care, treatment tolerance, social support, and competing risks of mortality, particularly in indolent lymphomas affecting older populations [
9,
10,
11,
12]. However, the extent to which sociodemographic factors independently influence survival in SMZL remains poorly characterized, with prior studies limited by small sample sizes, short follow-up, or incomplete adjustment for competing causes of death [
7,
13,
14].
To address these gaps by leveraging large-scale analyses of rare malignancies with extended follow-up, we performed this study using the Surveillance, Epidemiology, and End Results (SEER) dataset. This dataset captures detailed demographic, diagnostic, treatment, and survival information across diverse U.S. populations, making it a valuable resource for evaluating real-world outcomes in SMZL [
15]. Importantly, in indolent lymphomas such as SMZL, where non-lymphoma mortality is substantial and cause-of-death attribution may be unreliable, overall survival represents a particularly robust and clinically meaningful endpoint for registry-based research [
16].
In this study, we used SEER data spanning 2000 to 2022 to evaluate sociodemographic factors and treatment patterns associated with overall survival among patients with primary splenic marginal zone lymphoma. By leveraging a large, contemporary cohort with long-term follow-up, we aimed to clarify the relative contributions of patient characteristics and treatment exposures to survival outcomes and to provide population-level evidence to inform risk stratification and survivorship-focused care in this rare lymphoma subtype.
2. Materials and Methods
2.1. Study Design and Data Source:
This retrospective cohort study used data from the National Cancer Institute’s SEER program to evaluate sociodemographic factors and treatment patterns associated with overall survival among patients with SMZL. [
15] The study period spanned January 1, 2000, through December 31, 2022. SEER is a population-based registry that captures incident cancers, selected initial treatment indicators, vital status, and cause of death for defined geographic regions in the United States. The dataset includes patient-level demographic characteristics, tumor and staging variables as available, treatment recodes, follow-up time, and vital status. Because this study relied on deidentified registry data, it qualifies as non–human participants research and hence did not require IRB review.
2.2. Study Population
Patients were eligible if they had (1) histologically confirmed splenic marginal zone lymphoma defined using ICD-O-3 histology code 9689 and (2) primary site of the spleen (ICD-O-3 site code C42.2). Patients were excluded if required variables for time-to-event analyses were missing or invalid (eg, missing survival time or event indicator).
2.3. Study Variables
Sociodemographic characteristics included sex, race, Hispanic ethnicity, marital status at diagnosis, county-level rurality, and county-level median household income. Sex was analyzed as female versus male; race was grouped as White, Black, Asian/Pacific Islander, American Indian/Alaska Native, or unknown; ethnicity was classified as Hispanic or non-Hispanic; and marital status was categorized as married, single, divorced, separated, widowed, unmarried/domestic partner, or unknown. Rurality was measured using a seven-level county classification, and median household income, adjusted to 2023 dollars, was presented as increasing income categories. Clinical factors included age and year at diagnosis, both treated as continuous variables, and Ann Arbor stage, categorized as stages I–IV, unknown, or missing. Treatment variables included receipt of chemotherapy, receipt of radiation therapy, primary site surgery, and regional lymph node surgery, with treatments analyzed according to whether they were received and, for surgical variables, by the extent or type of procedure, with infrequent categories retained and unknown or not applicable categories used as reference where appropriate.
2.4. Outcomes
The primary endpoint was overall survival (OS), defined as the time from diagnosis to death from any cause; individuals alive at last follow-up were censored. Survival time was measured in months using the SEER survival time variable.
Disease-specific survival (DSS) was not selected as the primary endpoint because SEER cause-of-death attribution is derived from death certificate coding and may be misclassified, particularly for indolent lymphomas where death may occur from treatment toxicity, infections, secondary malignancies, or comorbid disease rather than direct lymphoma progression. This concern is amplified in an older cohort with substantial competing risks. In this study, the distribution of causes of death demonstrated prominent non-lymphoma mortality (eg, cardiovascular, pulmonary, infectious, and other causes), which limits interpretability of DSS and increases susceptibility to differential misclassification across sociodemographic strata. OS was therefore chosen as the most clinically meaningful and methodologically robust endpoint for registry-based comparisons. (Supplemental
Table 1)
2.5. Statistical Analysis
Descriptive statistics were used to summarize baseline characteristics and treatment patterns. Continuous variables were reported as mean (SD) and categorical variables as frequency (percentage).
Time-to-event analyses were conducted using Kaplan–Meier methods to estimate survival distributions by selected categorical predictors. Differences in survival curves were evaluated using log-rank tests (pooled over strata). Median survival times and corresponding standard errors and confidence intervals were reported when estimable; results for categories with very small sample sizes were interpreted cautiously given instability of estimates.
Associations between covariates and OS were evaluated using Cox proportional hazards regression. Univariate Cox models were fit for each predictor separately. For categorical variables, indicator (dummy) coding was used as appropriate. Continuous predictors (age, year of diagnosis, and median household income ordinal category) were modeled per 1-unit increase as presented in the output.
A multivariable Cox model was then fit including covariates meeting the prespecified inclusion threshold from univariate analyses (p<0.10) and those carried forward in the reported multivariable output with indicator parameter coding. The final adjusted model included sex, year of diagnosis, race, ethnicity, marital status, regional lymph node surgery, and chemotherapy. Adjusted hazard ratios (HRs) with 95% CIs were reported. Two-sided p values <0.05 were considered statistically significant.
Missing data were handled using complete-case analysis within each model. The rural–urban continuum indicator-coded model demonstrated non-convergence/singularity due to sparse categories, and estimates from that model were considered unstable and were not emphasized in inference. All analyses were performed using IBM SPSS Statistics (v31).
3. Results
3.1. Cohort characteristics
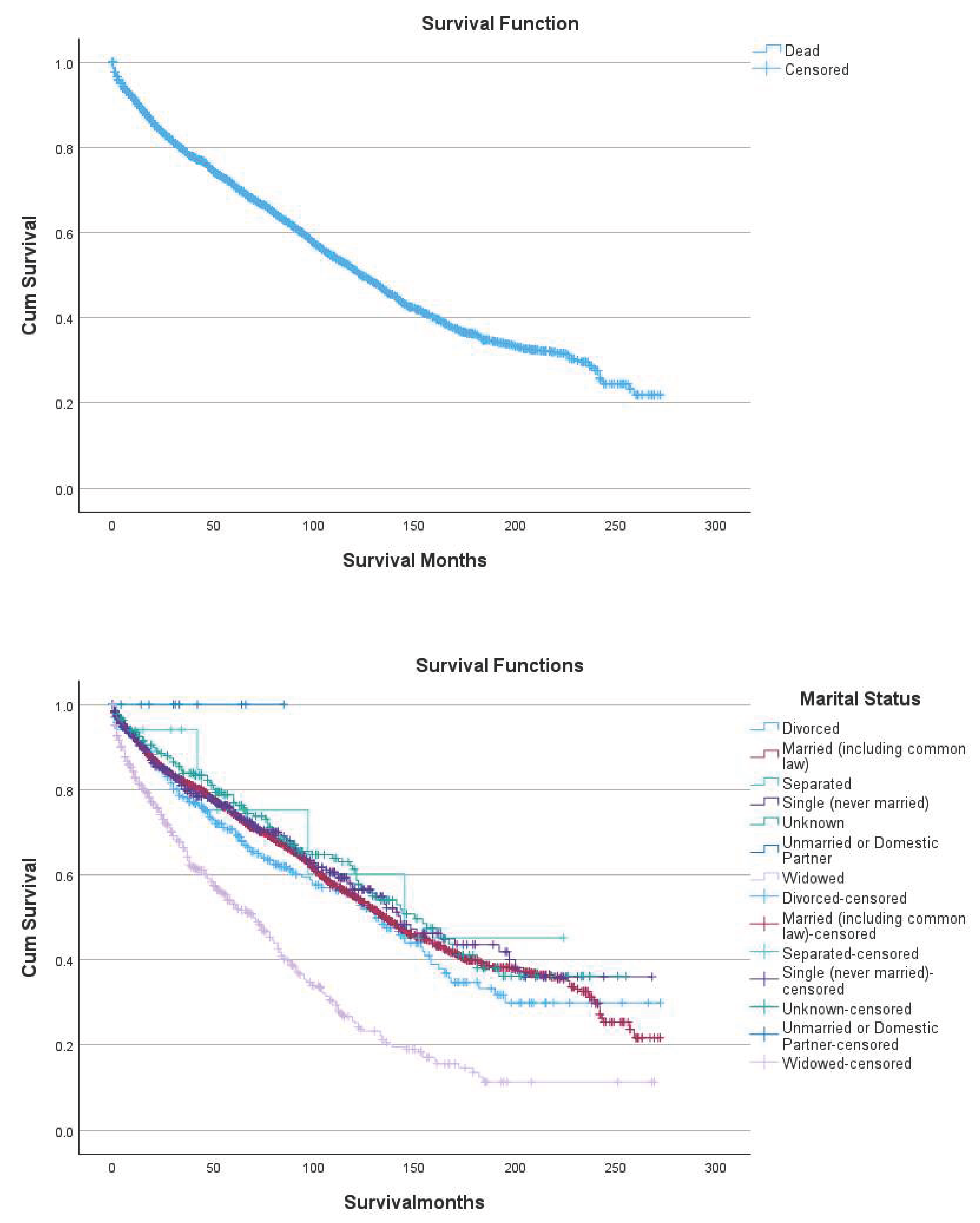
A total of 3,548 patients with histologically confirmed primary splenic marginal zone lymphoma were identified in SEER (2000–2022). The mean age at diagnosis was 68.2 years (SD 11.6), and 53.6% were female. Most patients were White (89.8%) and non-Hispanic (92.1%). Marital status at diagnosis was most commonly married (57.9%), followed by widowed (14.6%), single (11.0%), divorced (9.4%), and unknown (6.3%); separated (0.5%) and unmarried/domestic partner (0.3%) were uncommon. Most patients resided in metropolitan counties (≥1 million population, 61.3%). Ann Arbor stage was recorded as stage IV in 38.9% and stage I in 13.2%; however, staging was missing in 39.4% of cases. (
Table 1)
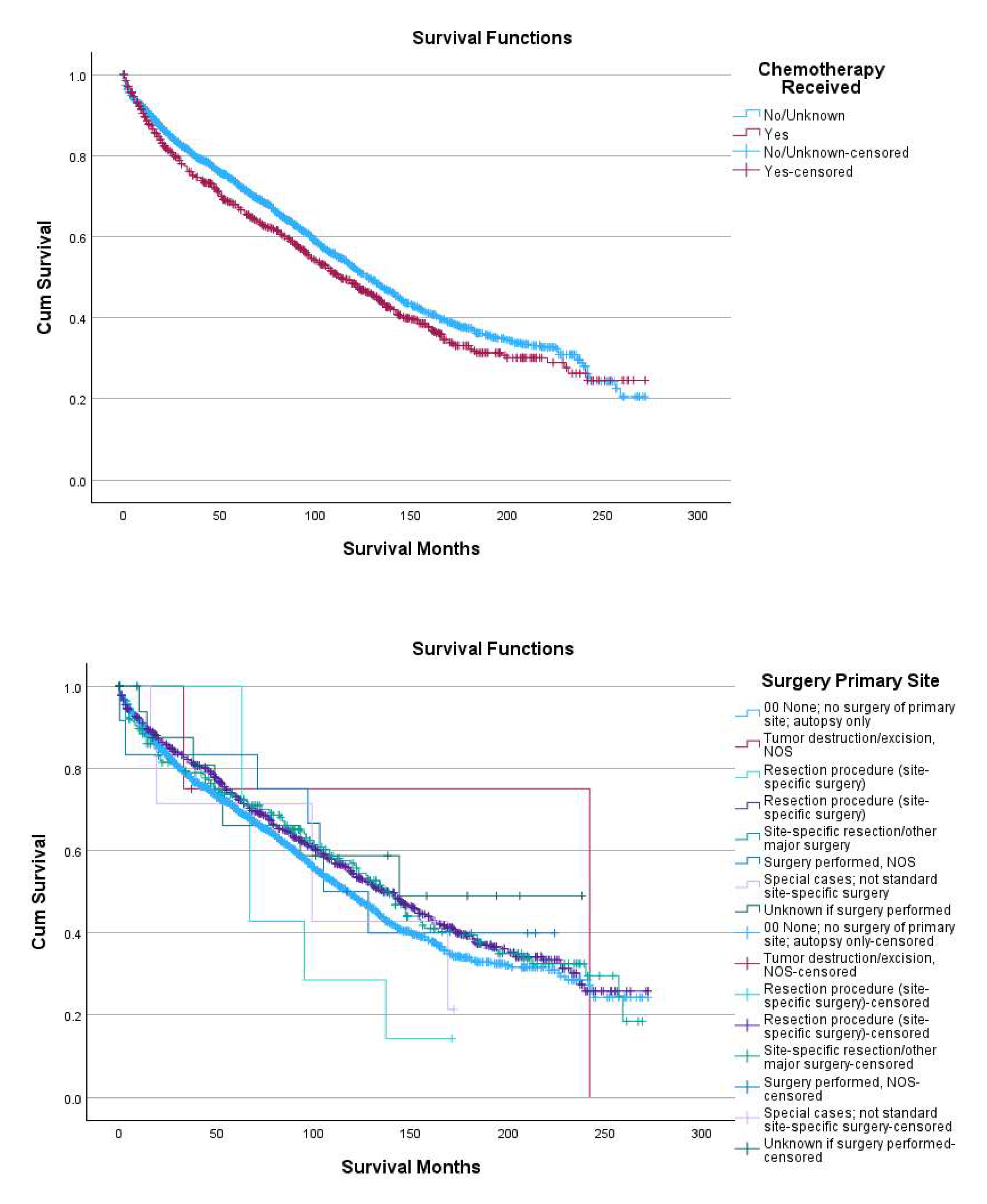
With respect to initial treatment, 26.4% received chemotherapy, 0.7% received beam radiation, and 21.4% underwent primary site surgery. Regional lymph node surgery was uncommon: 49.9% had no lymph node surgery, 1.5% had 1–3 nodes removed, and 0.7% had ≥4 nodes removed; lymph node surgery was unknown/not applicable in 40.4%. (
Table 1)
3.2. Cause of Deaths:
56.8% were alive at last follow-up. Among decedents, the most frequent recorded cause of death was non-Hodgkin lymphoma (15.8% of the entire cohort), followed by major competing causes including diseases of heart (6.1%) and a heterogeneous category of other causes of death (5.2%). Additional non-lymphoma causes each contributed smaller proportions, most commonly chronic respiratory disease (1.7%), cerebrovascular disease (1.3%), lung cancer (1.2%), and infection-related mortality (e.g., pneumonia/influenza 0.8%, septicemia 0.4%). Collectively, this pattern indicates that while lymphoma-related mortality constitutes a substantial share of deaths, competing cardiovascular, pulmonary, infectious, and secondary malignancy causes are prominent, consistent with the older age distribution of SMZL and underscoring the importance of survivorship-focused management alongside lymphoma-directed care. (Supplementary
Table 1)
3.3. Survival Analyses
Kaplan-Meier analyses showed no statistically significant differences in overall survival by extent of primary site surgery (log-rank p=0.376), despite numerical variation in median survival across surgical categories. Patients who underwent site-specific resection or other major surgery exhibited median survival estimates comparable to those managed without primary site surgery, while categories with very small sample sizes showed unstable estimates with wide confidence intervals, limiting interpretability. In contrast, overall survival differed markedly by marital status (log-rank p<0.001), with widowed patients experiencing substantially shorter median survival (70 months) compared with married, divorced, single, or unknown-status patients, consistent with the strong prognostic effect observed in multivariable Cox modeling. Chemotherapy exposure was also associated with statistically significant differences in survival distributions (log-rank p=0.013), with patients receiving no or unknown chemotherapy status demonstrating longer median survival (128 months) compared with those who received chemotherapy (113 months). (
Figure 1)
3.4. Univariate predictors of overall survival
In univariate Cox analysis, female sex was associated with lower mortality than male sex (HR 0.86, 95% CI 0.78 to 0.95; p = 0.003). Increasing age was strongly associated with death (HR 1.07 per year, 95% CI 1.06 to 1.08; p < 0.001). Non-Hispanic ethnicity was associated with improved survival compared with Hispanic ethnicity (HR 0.82, 95% CI 0.69 to 0.98; p = 0.030). Marital status showed a strong association with OS, with married patients demonstrating lower mortality than widowed patients (HR 0.47, 95% CI 0.41 to 0.53; p < 0.001). Chemotherapy receipt was associated with higher mortality, as patients with no or unknown chemotherapy had lower risk of death (HR 0.87, 95% CI 0.78 to 0.97; p = 0.013). (
Table )
3.5. Multivariable predictors of overall survival
After adjustment for covariates meeting prespecified inclusion criteria, increasing age strongly predicted worse OS (HR 1.07 per year, 95% CI 1.07 to 1.08; p < 0.001). Male sex was independently associated with higher mortality (HR 1.34, 95% CI 1.20 to 1.49; p < 0.001). Hispanic ethnicity remained associated with increased risk of death (HR 1.43, 95% CI 1.19 to 1.70; p < 0.001). Chemotherapy exposure was also associated with higher mortality (HR 1.25, 95% CI 1.12 to 1.39; p < 0.001). (
Table 3)
4. Discussion
To our knowledge, this study represents the largest population based cohort of primary SMZL reported to date. In multivariable analysis, age, sex, Hispanic ethnicity, marital status, year of diagnosis, race, and chemotherapy exposure were independently associated with OS. Increasing age was the strongest continuous predictor of mortality. Male sex remained associated with higher risk of death. Hispanic ethnicity was associated with inferior survival compared with non Hispanic ethnicity. Race showed an overall association with OS, with Asian or Pacific Islander patients demonstrating higher mortality relative to White patients, while other racial comparisons were not statistically significant. These findings are consistent with prior reports in indolent B cell lymphomas that document survival differences across demographic groups [
10,
13,
17,
18].
Marital status retained a significant association with OS after adjustment. Compared with divorced patients, married patients and those with unknown marital status had lower mortality, while widowed and single patients did not differ significantly in the adjusted model. Prior oncology literature has reported survival differences by marital status [
9,
10,
11,
12,
13,
14,
15,
16,
17,
18,
19]. Our analysis confirms that marital status remains associated with OS in SMZL within a population-based cohort.
In the context of SMZL, an indolent disease predominantly affecting older adults, the magnitude and persistence of the marital-status association suggest that social determinants and supportive care factors play a central role in shaping long-term outcomes. This interpretation is reinforced by the observed distribution of causes of death, which showed a substantial contribution from non-lymphoma causes, including cardiovascular, pulmonary, infectious, and secondary malignancy-related mortality [
13,
20].
Primary site surgery and radiation therapy were not associated with differences in OS in univariate analyses. Chemotherapy exposure was associated with higher mortality in the adjusted model. These findings should be interpreted within the context of registry based data. SEER does not capture treatment intent, specific regimens, dose intensity, performance status, or detailed measures of disease burden. As a result, the observed association between chemotherapy and OS most likely reflects underlying disease severity and treatment selection rather than a direct adverse treatment effect [
5,
13,
22]. These results provide valuable population level insight into real world practice patterns while underscoring the need for prospective clinical data to define treatment efficacy.
Ann Arbor stage was not independently associated with OS in univariate analysis. Although stage was not recorded in 39.4 percent of cases, the available data reflect real world registry reporting patterns in SMZL and allow evaluation of survival within a large, contemporary population based cohort. The absence of a strong stage signal is consistent with prior observations that traditional anatomic staging has limited prognostic discrimination in SMZL [
13,
18].
This study has several notable strengths, including a large population-based cohort, extended follow up, and broad geographic representation. These features provide robust and generalizable estimates of survival in a rare lymphoma subtype that is not feasible to study in randomized trials. Although registry data do not capture detailed clinical variables or treatment intent, they offer a valuable real-world perspective on outcomes across diverse patient populations. Despite inherent limitations such as stage missingness and limited treatment granularity, the consistency and magnitude of the observed associations support the validity of the findings. Within this framework, our results demonstrate that demographic and social variables remain independently associated with OS in SMZL. These data provide clinically relevant insight and reinforce the importance of comprehensive assessment, survivorship planning, and comorbidity management in this predominantly older patient population.
5. Conclusions
In this large population-based analysis of SMZL, overall survival was independently associated with age, sex, ethnicity, marital status, and chemotherapy exposure. Male sex, Hispanic ethnicity, advancing age, and receipt of chemotherapy were associated with higher mortality, while married status was associated with improved survival. Local treatment approaches, including splenectomy and radiation, were not associated with superior survival. The substantial contribution of non-lymphoma causes of death highlights the importance of competing health risks in this older population. These findings provide population level evidence to inform risk assessment and long-term management in SMZL.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Table S1: Cause of death distribution (COD to site recode) in the SMZL cohort (SEER 2000–2022; N=3,548).
Author Contributions (CRediT)
Conceptualization: Manas Pustake, Atulya Aman Khosla, Jesus Gomez; Methodology: Manas Pustake, Atulya Aman Khosla, Nishant Tiwari; Formal Analysis: Manas Pustake; Data Curation: Manas Pustake Investigation: Manas Pustake, Oboseh Ogedegbe, Sakditad Saowapa, Mohammad Arfat Ganiyani; Validation: Atulya Aman Khosla, Jesus Gomez; Writing – Original Draft Preparation: Manas Pustake; Writing – Review and Editing: Oboseh Ogedegbe, Atulya Aman Khosla, Sakditad Saowapa, Mohammad Arfat Ganiyani, Avi Harisingani, Nishant Tiwari, Stevenson Ongsyping, Jesus Gomez; Supervision: Jesus Gomez; Project Administration: Manas Pustake; All authors reviewed the manuscript critically for important intellectual content and approved the final version for publication.
Funding
This research received no external funding.
Institutional Review Board Statement
This study used deidentified, publicly available SEER registry data and qualifies as non human subjects research. Institutional Review Board review and approval were not required.
Informed Consent Statement
Not applicable. The study used deidentified registry data and did not involve direct patient contact.
Data Availability Statement (required):
The data used in this study are publicly available through the Surveillance, Epidemiology, and End Results Program (SEER), National Cancer Institute. Access to SEER data requires completion of a data use agreement and can be obtained at
https://seer.cancer.gov.
Acknowledgments
This study was presented as a poster at the American Society of Hematology Annual Meeting 2025 in Orlando, FL and Author Manas Pustake has received Abstract Achievement Award for this abstract and poster presentation. This study used data from the Surveillance, Epidemiology, and End Results Program of the National Cancer Institute. The interpretation and reporting of these data are the sole responsibility of the authors. Grammarly AI was used for language editing and grammar refinement. The authors take full responsibility for the content and integrity of the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Arcaini, L; Rossi, D; Paulli, M. Splenic marginal zone lymphoma: from genetics to management. Blood, The Journal of the American Society of Hematology 2016, 127(17), 2072–81. [Google Scholar] [CrossRef]
- Franco, V; Florena, AM; Iannitto, E. Splenic marginal zone lymphoma. Blood, The Journal of the American Society of Hematology 2003, 101(7), 2464–72. [Google Scholar] [CrossRef] [PubMed]
- Thieblemont, C; Felman, P; Callet-Bauchu, E; Traverse-Glehen, A; Salles, G; Coiffier, B; Berger, F. Splenic marginal-zone lymphoma: a distinct clinical and pathological entity. The lancet oncology 2003, 4(2), 95–103. [Google Scholar] [CrossRef]
- Cheah, CY; Zucca, E; Rossi, D; Habermann, TM. Marginal zone lymphoma: present status and future perspectives. Haematologica 2022, 107(1), 35. [Google Scholar] [CrossRef]
- Santos, TS; Tavares, RS; Farias, DL. Splenic marginal zone lymphoma: a literature review of diagnostic and therapeutic challenges. Revista brasileira de hematologia e hemoterapia 2017, 39, 146–54. [Google Scholar] [CrossRef]
- Perrone, S; D'Elia, GM; Annechini, G; Ferretti, A; Tosti, ME; Foà, R; Pulsoni, A. Splenic marginal zone lymphoma: Prognostic factors, role of watch and wait policy, and other therapeutic approaches in the rituximab era. Leuk Res 2016, 44, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Alderuccio, JP; Lossos, IS. Prognostic factors and risk of transformation in marginal zone lymphoma. Annals of Lymphoma 2020, 4. [Google Scholar] [CrossRef]
- Flinn, IW; Van Der Jagt, R; Kahl, BS; Wood, P; Hawkins, TE; MacDonald, D; Hertzberg, M; Kwan, YL; Simpson, D; Craig, M; Kolibaba, K. Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood, The Journal of the American Society of Hematology 2014, 123(19), 2944–52. [Google Scholar] [CrossRef]
- Fei-Zhang, DJ; Wu, E; Stanisic, AV; Hou, L; Platanias, LC; Ansell, SM; Lewis-Thames, MW; Badawy, SM; Paludo, J. Socioeconomic, racial-ethnic, household, and infrastructural disparities of hematologic cancer outcomes in the United States. Blood Adv. 2025, 9(6), 1463–1471. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Habib, A; Huang, Y; Paskett, ED; Devarakonda, S; Fisher, JL; Kittai, AS. Effect of racial disparities on cancer survival in hematologic malignancies: a population-based analysis of SEER data (2009–2019). Preventive Oncology & Epidemiology 2025, 3(1), 2483704. [Google Scholar] [CrossRef]
- Nielsen, LH; Kristensen, DT; Jakobsen, LH; Bøgsted, M; Gregersen, H; Madsen, J; Severinsen, MT; Brøndum, RF. Socioeconomic status and overall survival among patients with hematological malignant neoplasms. JAMA network open 2024, 7(3), e241112-. [Google Scholar] [CrossRef]
- Miranda-Galvis, M; Tjioe, KC; Balas, EA; Agrawal, G; Cortes, JE. Disparities in survival of hematologic malignancies in the context of social determinants of health: a systematic review. Blood advances 2023, 7(21), 6466–91. [Google Scholar] [CrossRef]
- Florindez, JA; Alderuccio, JP; Reis, IM; Lossos, IS. Splenic marginal zone lymphoma: A US population-based survival analysis (1999-2016). Cancer 2020, 126(21), 4706–4716. [Google Scholar] [CrossRef] [PubMed]
- Kalashnikov, I; Tanskanen, T; Viisanen, L; Malila, N; Jyrkkiö, S; Leppä, S. Transformation and survival in marginal zone lymphoma: a Finnish nationwide population-based study. Blood cancer journal 2023, 13(1), 62. [Google Scholar] [CrossRef] [PubMed]
- Surveillance, Epidemiology, and End Results (SEER) Program. (2025). SEER*Stat Database: Incidence –
SEER Research Data, 18 Registries, Nov 2024 Sub (2000–2022) – Linked To County Attributes – Total U.S.,
1969–2023 Counties [Data set]. National Cancer Institute, Division of Cancer Control and Population
Sciences, Surveillance Research Program. https://seer.cancer.gov.
- Al-Mashhadi, AL; Simonsen, MR; Cheah, CY; Amini, RM; Arboe, B; Cerhan, JR; Chanchiri, I; Clausen, MR; Day, J; Eyre, TA; Gang, AO. Favorable Outcomes of Splenic Marginal Zone Lymphoma in an International Study of 934 Patients with Long Follow-up. Blood 2023, 142, 4396. [Google Scholar] [CrossRef]
- Munir, M; Vaughn, JL; Rimmalapudi, S; Epperla, N. Site-specific prognosis and temporal trends in extranodal marginal zone lymphoma patients in the United States. Blood Cancer Journal 2025, 15(1), 201. [Google Scholar] [CrossRef] [PubMed]
- Khor, S; Haupt, EC; Hahn, EE; Lyons, LJ; Shankaran, V; Bansal, A. Racial and ethnic bias in risk prediction models for colorectal cancer recurrence when race and ethnicity are omitted as predictors. JAMA Network Open 2023, 6(6), e2318495-. [Google Scholar] [CrossRef]
- Jain, P; Ganatra, N; Patel, S; Abdullah, J; Anwar, J. Racial and socioeconomic disparities in incidence and survival of splenic marginal zone lymphoma: United States national database analysis. Blood 2025, 146, 7095. [Google Scholar] [CrossRef]
- Vaughn, JL; Pinheiro, LC; Olszewski, AJ; Epperla, N. Decreased Lymphoma-Specific Mortality in Patients with Splenic Marginal Zone Lymphoma in the United States. Blood 2020, 136:9. [Google Scholar] [CrossRef]
- Ogbue, O; Zhou, J; Adeoye, O; Falade, A; Hwang, S; Wang, Y; Godby, R. Real-world treatment patterns and survival in splenic marginal zone lymphoma: A national analysis in the rituximab era. Blood 2025, 146, 1813. [Google Scholar] [CrossRef]
- Perrone, S; D'Elia, GM; Annechini, G; Ferretti, A; Tosti, ME; Foà, R; Pulsoni, A. Splenic marginal zone lymphoma: Prognostic factors, role of watch and wait policy, and other therapeutic approaches in the rituximab era. Leuk Res 2016, 44, 53–60. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |