Submitted:
15 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. VAERS Retrospective Analysis
2.2. Adverse Events Following Immunization
2.3. Manufacturing Contamination Signature Identification
3. Results
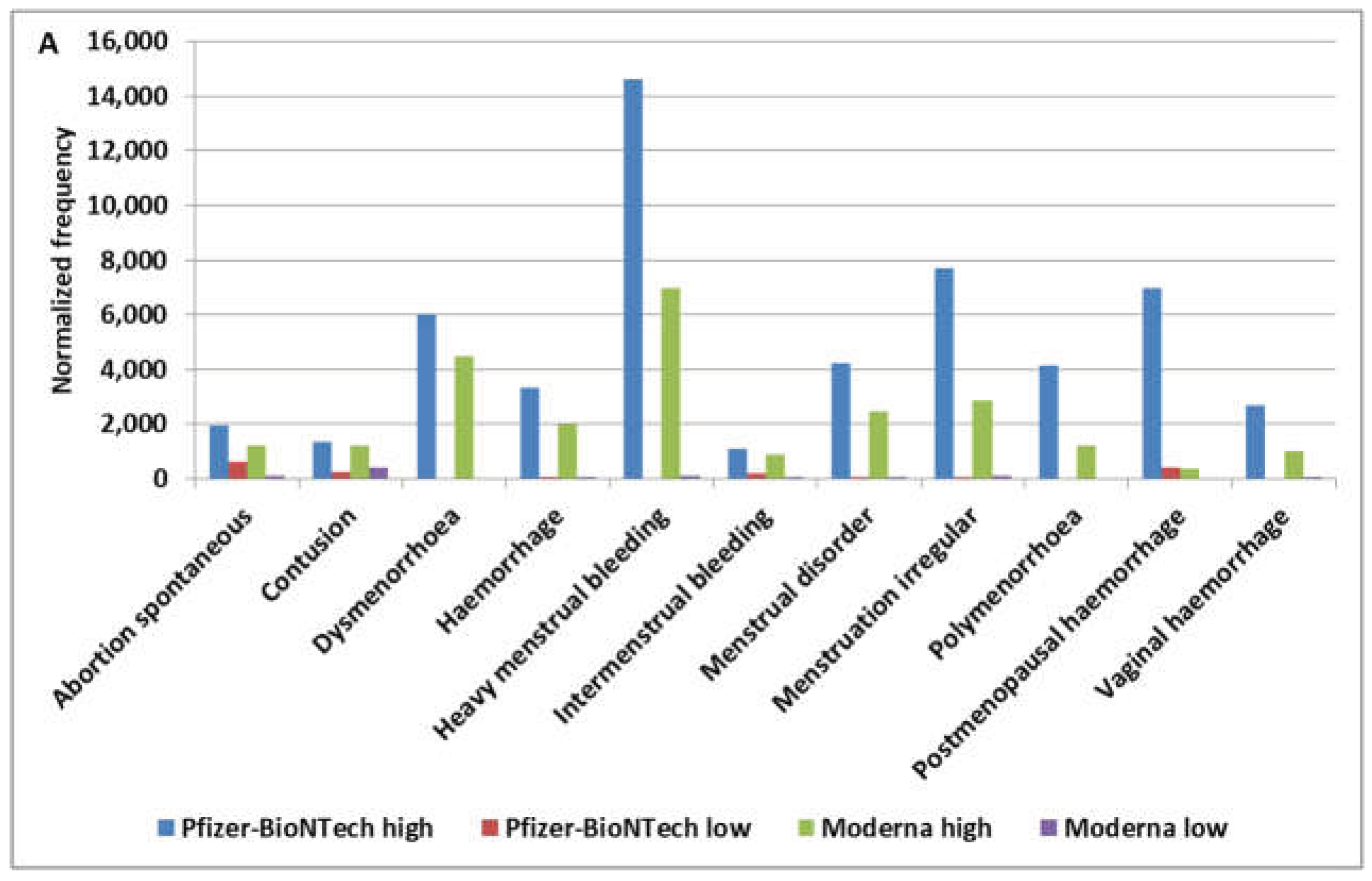
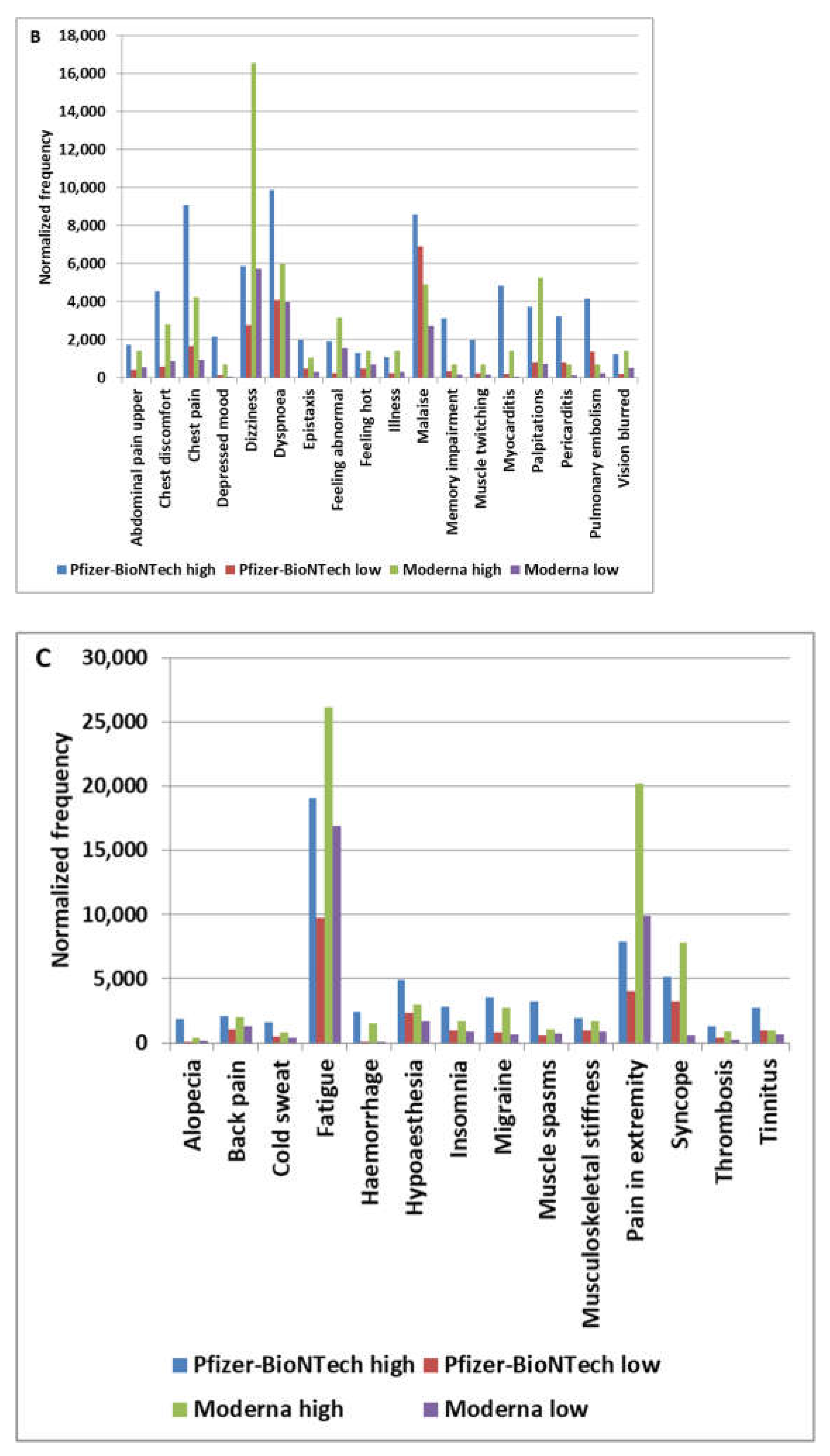
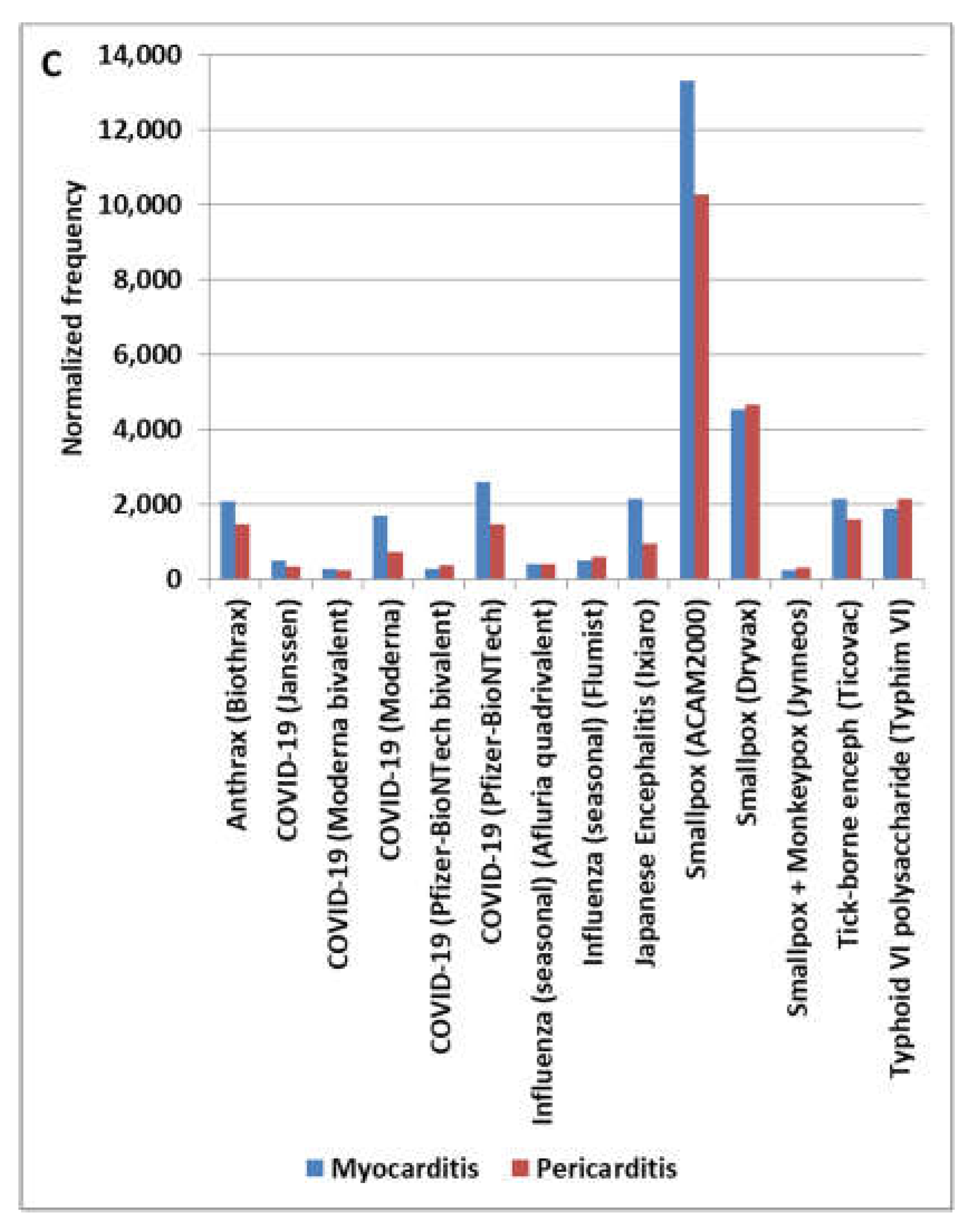
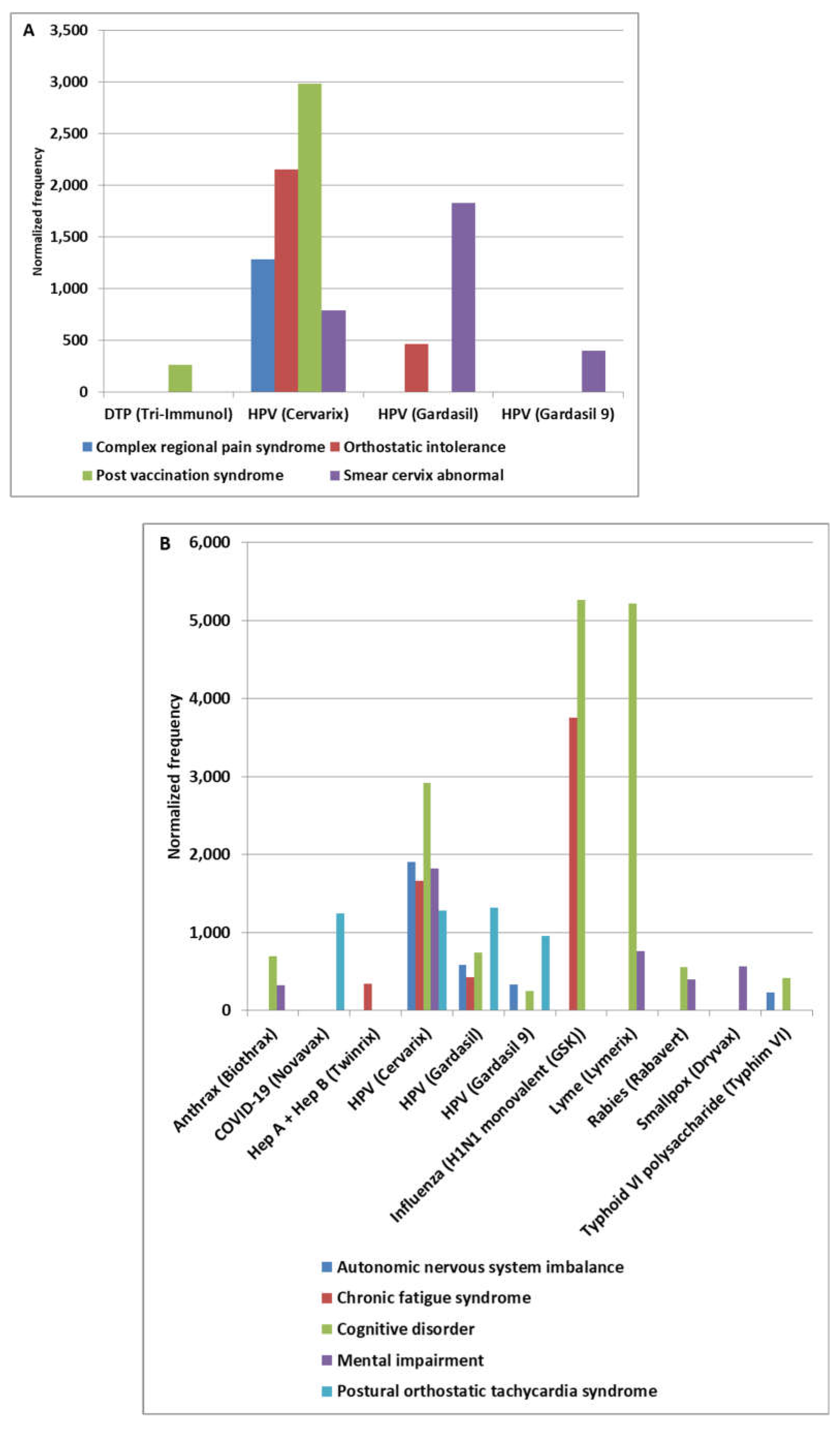
3.1. COVID-19 Vaccine Candidate Manufacturing Contamination Patterns
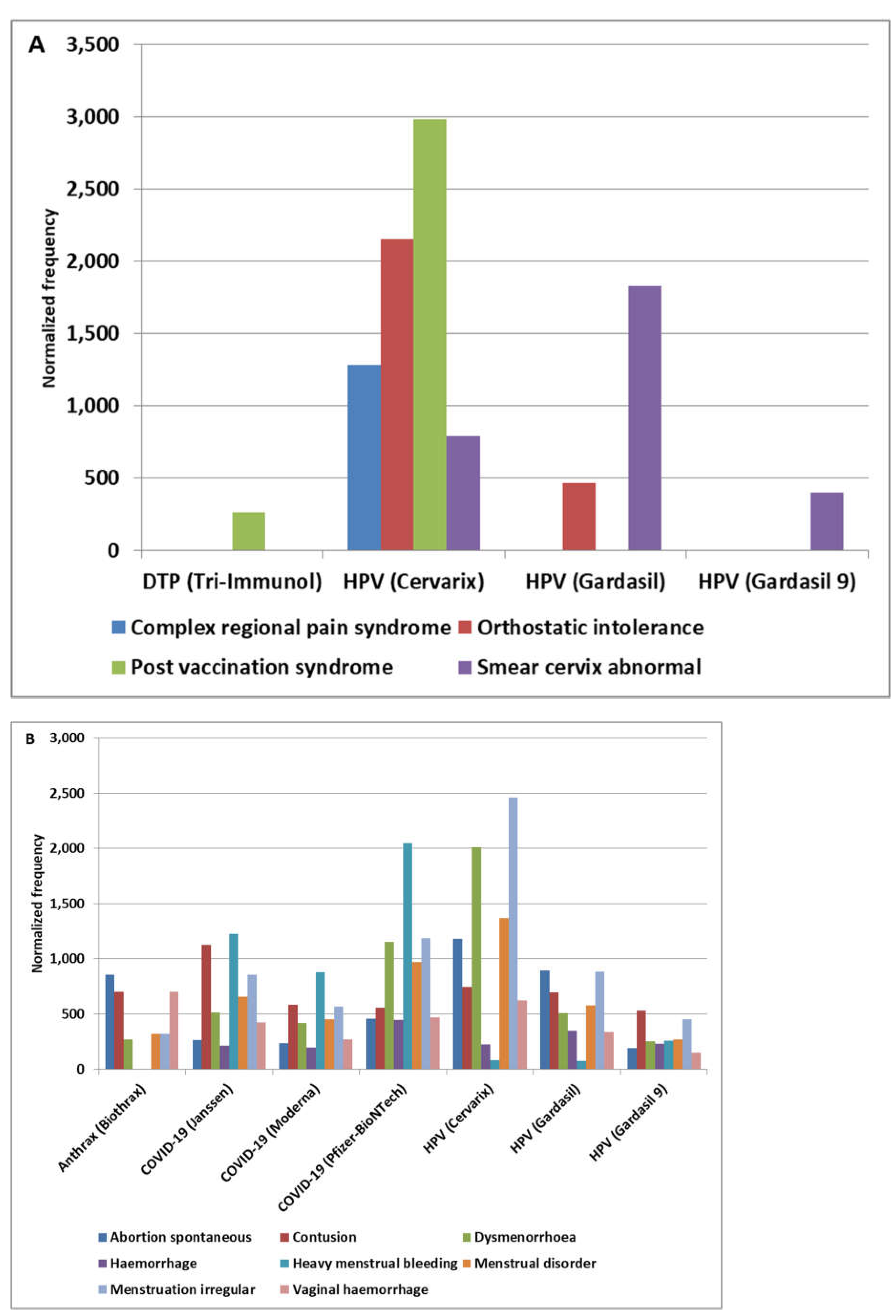
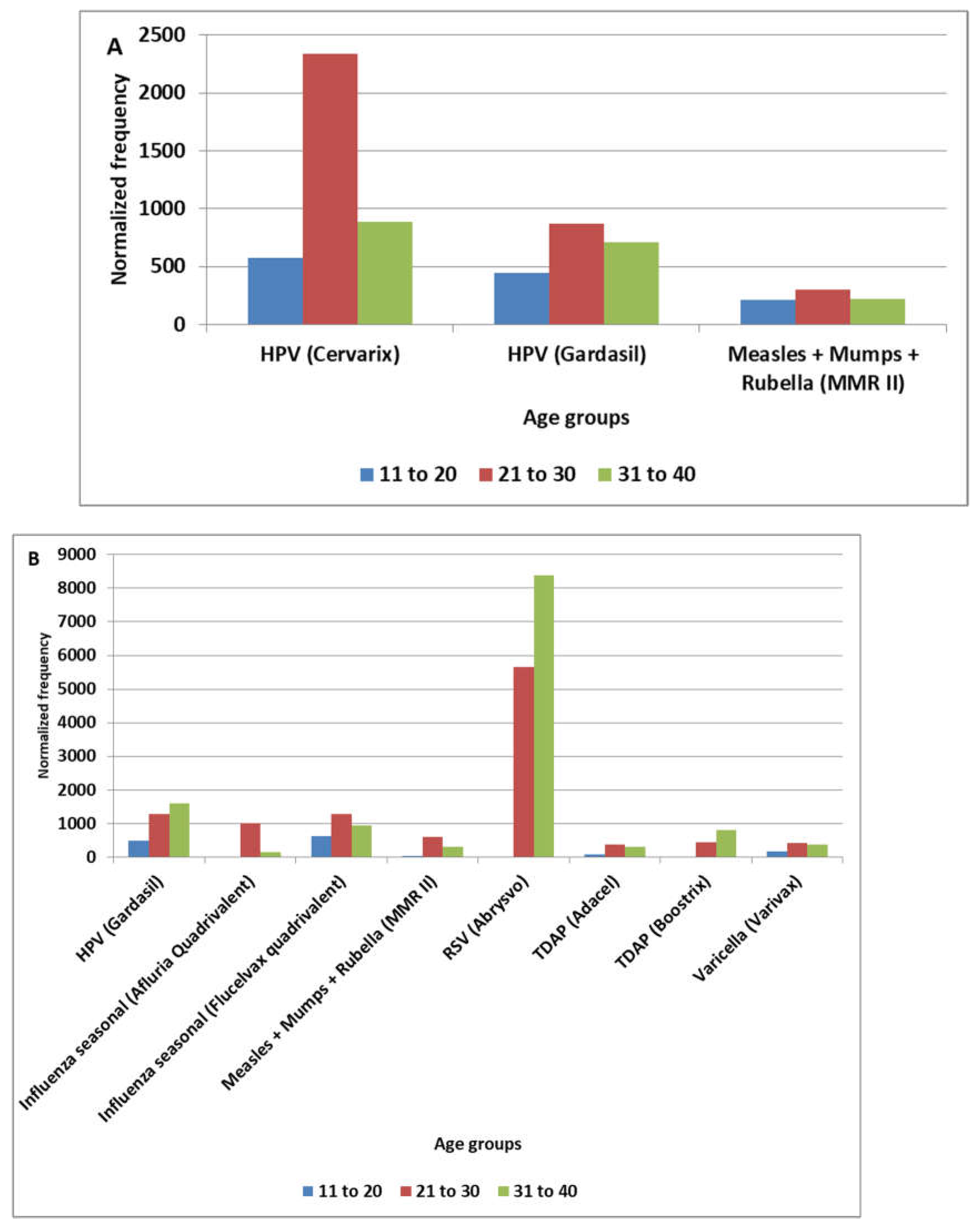
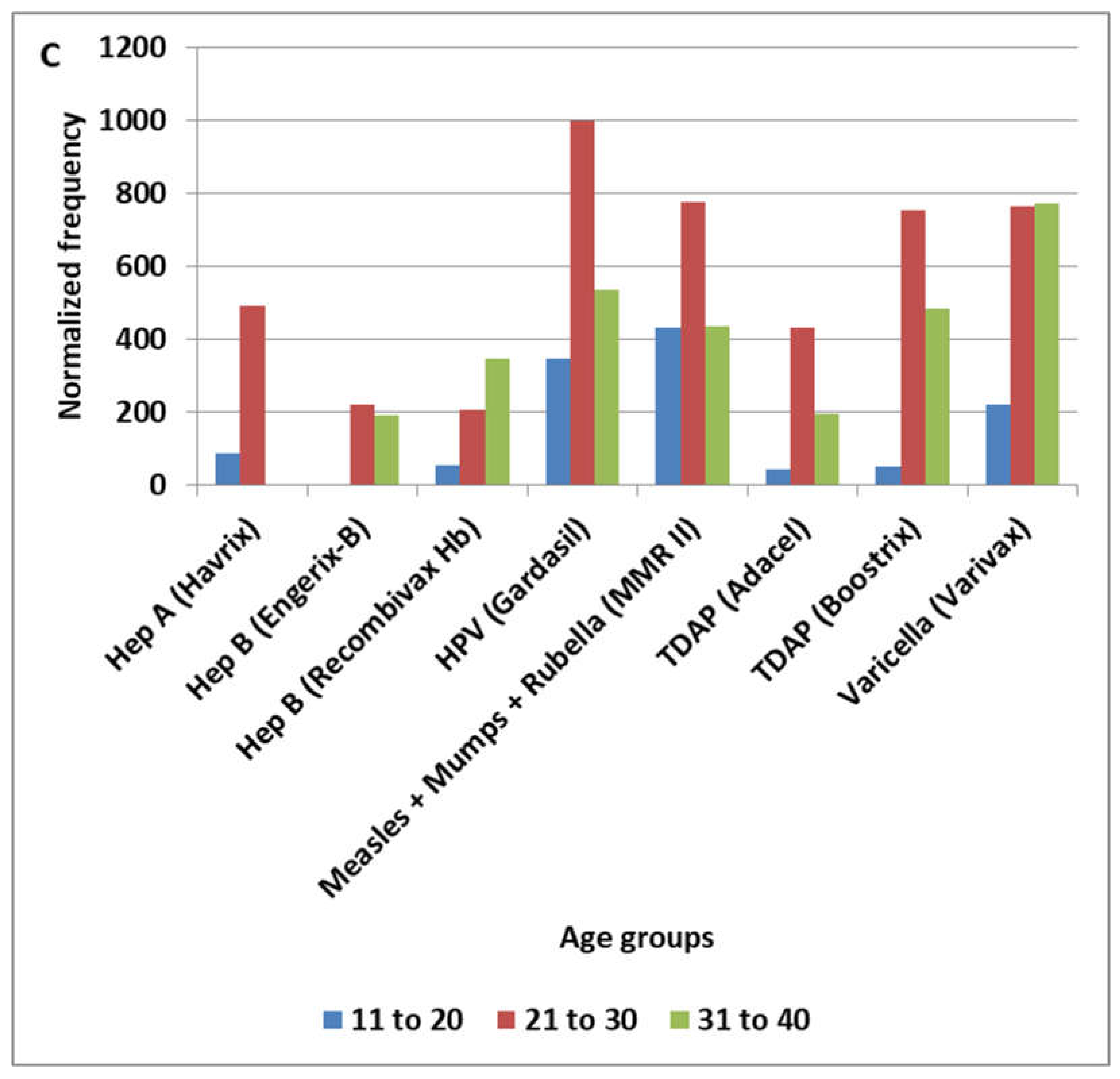
3.2. HPV Vaccine Candidate Manufacturing Contamination Patterns
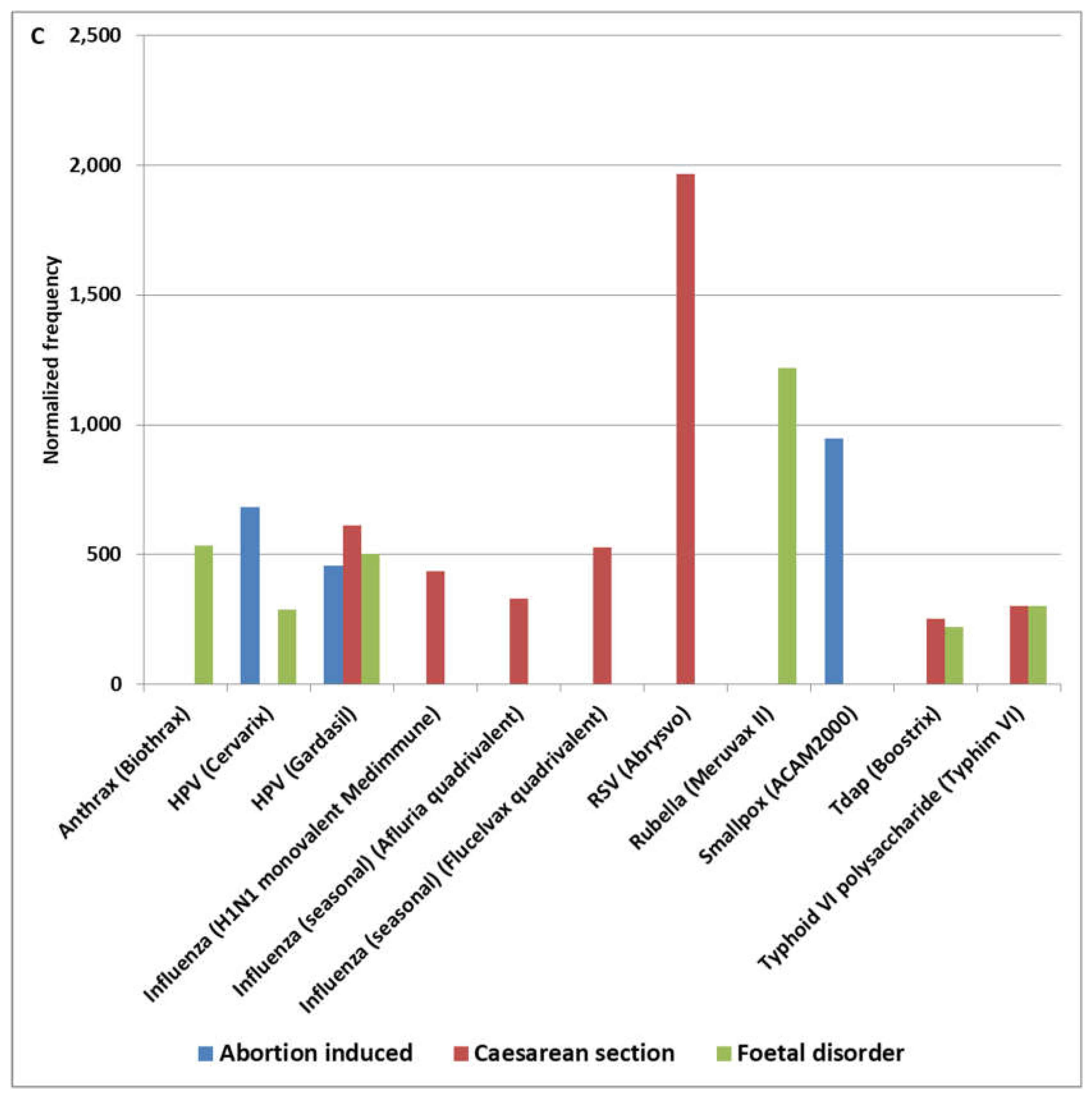
3.3. Identified Fetal Adverse Events
4. Discussion
4.1. Candidate Manufacturing Contamination Patterns
4.2. MChemorrhage Pattern
4.3. MCgynecologic Pattern
4.4. MCmyopericarditis Pattern
4.5. MCHPV Pattern
4.6. MCneuropathy Pattern
4.7. Abortion Spontaneous AEs and MCfetus Pattern
4.8. Endotoxins – Lipopolysaccharides - Pyrogens
4.9. Study Limitations
5. Conclusions
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
References
- Matar SG, Nourelden AZ, Assar A, et al. Effect of COVID-19 vaccine on menstrual experience among females in six Arab countries: A cross sectional study. Influenza Other Respir Viruses. 2023;17(1):e13088. [CrossRef]
- Baena-García L, Aparicio VA, Molina-López A, Aranda P, Cámara-Roca L, Ocón-Hernández O. Premenstrual and menstrual changes reported after COVID-19 vaccination: The EVA project. Womens Health. 2022;18:17455057221112237. [CrossRef]
- Rastegar T, Feryduni L, Fakhraei M. COVID-19 vaccine side effects on menstrual disturbances among Iranian women. New Microbes New Infect. 2023;53:101114. [CrossRef]
- Dabbousi AA, El Masri J, El Ayoubi LM, Ismail O, Zreika B, Salameh P. Menstrual abnormalities post-COVID vaccination: a cross-sectional study on adult Lebanese women. Ir J Med Sci 1971 -. 2023;192(3):1163-1170. [CrossRef]
- Lee KM, Junkins EJ, Luo C, Fatima UA, Cox ML, Clancy KB. Investigating trends in those who experience menstrual bleeding changes after SARS-CoV-2 vaccination. medRxiv. 2022. [CrossRef]
- Trogstad L, Laake I, Robertson AH, et al. Heavy bleeding and other menstrual disturbances in young women after COVID-19 vaccination. Vaccine. 2023;41(36):5271-5282. [CrossRef]
- Almomani EY, Hajjo R, Qablan A, Sabbah DA, Al-Momany A. A cross-sectional study confirms temporary post-COVID-19 vaccine menstrual irregularity and the associated physiological changes among vaccinated women in Jordan. Front Med. 2023;10. [CrossRef]
- Duijster JW, Schoep ME, Nieboer TE, Jajou R, Kant A, van Hunsel F. Menstrual abnormalities after COVID-19 vaccination in the Netherlands: A description of spontaneous and longitudinal patient-reported data. Br J Clin Pharmacol. 2023;89(10):3126-3138. [CrossRef]
- Laganà AS, Veronesi G, Ghezzi F, et al. Evaluation of menstrual irregularities after COVID-19 vaccination: Results of the MECOVAC survey. Open Med. 2022;17(1):475-484. [CrossRef]
- Muhaidat N, Alshrouf MA, Azzam MI, Karam AM, Al-Nazer MW, Al-Ani A. Menstrual Symptoms After COVID-19 Vaccine: A Cross-Sectional Investigation in the MENA Region. Int J Womens Health. 2022;14:395-404. [CrossRef]
- Al-Furaydi A, Alrobaish SA, Al-Sowayan N. The COVID-19 vaccines and menstrual disorders. Eur Rev Med Pharmacol Sci. 2023;27(3):1185-11911. [CrossRef]
- Khan GA, Althubaiti A, Alshrif A, Alsayed Z, Jifree H. Dysmenorrhea, intermenstrual bleeding, menstrual flow volume changes, and irregularities following COVID-19 vaccination and the association with vaccine skepticism: A retrospective observational study. Womens Health. 2023;19:17455057231210094. [CrossRef]
- Li K, Chen G, Hou H, et al. Analysis of sex hormones and menstruation in COVID-19 women of child-bearing age. Reprod Biomed Online. 2021;42(1):260-267. [CrossRef]
- Blix K, Laake I, Juvet L, et al. Unexpected vaginal bleeding and COVID-19 vaccination in nonmenstruating women. Sci Adv. 9(38):eadg1391. [CrossRef]
- Schmeling M, Manniche V, Hansen PR. Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine. Eur J Clin Invest. 2023;53(8):e13998. [CrossRef]
- Manniche V, Schmeling M, Gilthorpe JD, Hansen PR. Reports of Batch-Dependent Suspected Adverse Events of the BNT162b2 mRNA COVID-19 Vaccine: Comparison of Results from Denmark and Sweden. Medicina (Mex). 2024;60(8). [CrossRef]
- Ricke DO. Menstrual adverse events post-COVID-19 and human papillomavirus immunization. Microbes Immun. 2025. [CrossRef]
- Wastila L, Fu YH, Tung CC, Qato DM. Association Between Vaccination for Human Papillomavirus (HPV) and Autonomic Dysfunction and Menstrual Irregularities: A Self-Controlled Case Series Analysis. Drugs Real World Outcomes. 2025;12(3):467-477. [CrossRef]
- Little DT, Ward HRG. Adolescent Premature Ovarian Insufficiency Following Human Papillomavirus Vaccination: A Case Series Seen in General Practice. J Investig Med High Impact Case Rep. 2014;2(4):2324709614556129. [CrossRef]
- Gong L, Ji H huan, Tang X wen, Pan L yun, Chen X, Jia Y tao. Human papillomavirus vaccine-associated premature ovarian insufficiency and related adverse events: data mining of Vaccine Adverse Event Reporting System. Sci Rep. 2020;10(1):10762. [CrossRef]
- VAERS. Vaccine Adverse Event Reporting System. U.S. Department of Health & Human Services; 2025. https://vaers.hhs.gov/data/datasets.html. Accessed June 20, 2025.
- Ricke DO. VAERS-Tools. 2022. https://github.com/doricke/VAERS-Tools. Accessed November 15, 2025.
- Ricke DO. Vaccine-associated Kawasaki disease in children. Microbes Immun. 2025. [CrossRef]
- Ochiai M, Kataoka M, Toyoizumi H, et al. Endotoxin Content in Haemophilus influenzae Type b Vaccine. Jpn J Infect Dis. 2004;57(2):58-59. [CrossRef]
- Wilting FNH, Kotsopoulos AMM, Platteel ACM, van Oers JAH. Intracerebral Hemorrhage and Thrombocytopenia After AstraZeneca COVID-19 Vaccine: Clinical and Diagnostic Challenges of Vaccine-Induced Thrombotic Thrombocytopenia. Cureus. 2021;13(9):e17637. [CrossRef]
- See I, Su JR, Lale A, et al. US Case Reports of Cerebral Venous Sinus Thrombosis With Thrombocytopenia After Ad26.COV2.S Vaccination, March 2 to April 21, 2021. JAMA. 2021;325(24):2448-2456. [CrossRef]
- Greinacher Andreas, Thiele Thomas, Warkentin Theodore E., Weisser Karin, Kyrle Paul A., Eichinger Sabine. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N Engl J Med. 2021;384(22):2092-2101. [CrossRef]
- Muir Kate-Lynn, Kallam Avyakta, Koepsell Scott A., Gundabolu Krishna. Thrombotic Thrombocytopenia after Ad26.COV2.S Vaccination. N Engl J Med. 2021;384(20):1964-1965. [CrossRef]
- Sharma A, Upadhyay B, Banjade R, Poudel B, Luitel P, Kharel B. A Case of Diffuse Alveolar Hemorrhage With COVID-19 Vaccination. Cureus. 2022;14(1):e21665. [CrossRef]
- Syed K, Chaudhary H, Donato A. Central Venous Sinus Thrombosis with Subarachnoid Hemorrhage Following an mRNA COVID-19 Vaccination: Are These Reports Merely Co-Incidental? Am J Case Rep. 2021. [CrossRef]
- Chen QT, Liu Y, Chen YC, et al. Case report: Vaccine-induced immune thrombotic thrombocytopenia complicated by acute cerebral venous thrombosis and hemorrhage after AstraZeneca vaccines followed by Moderna COVID-19 vaccine booster and surgery. Front Neurol. 2022;13. [CrossRef]
- Sugasawa S, Kimura T, Bae Y, Kumasaka T, Ichi S. Two Cases of Rare Intratumoral Hemorrhage Following COVID-19 Vaccination. Cureus. 2022;14(12):e32400. [CrossRef]
- Takeyama R, Fukuda K, Kouzaki Y, et al. Intracerebral hemorrhage due to vasculitis following COVID-19 vaccination: a case report. Acta Neurochir (Wien). 2022;164(2):543-547. [CrossRef]
- Hulscher N, McCullough PA. Delayed Fatal Pulmonary Hemorrhage Following COVID-19 Vaccination: Case Report, Batch Analysis, And Proposed Autopsy Checklist. Preprints. 2024. [CrossRef]
- Yangi K, Demir DD, Uzunkol A. Intracranial Hemorrhage After Pfizer-BioNTech (BNT162b2) mRNA COVID-19 Vaccination: A Case Report. Cureus. 2023;15(4):e37747. [CrossRef]
- Bhurayanontachai R. Possible life-threatening adverse reaction to monovalent H1N1 vaccine. Crit Care. 2010;14(3):422. [CrossRef]
- Zhang J, Lian Z, Xue X, et al. Does HPV vaccination during periconceptional or gestational period increase the risk of adverse pregnancy outcomes?—An updated systematic review and meta-analysis based on timing of vaccination. Acta Obstet Gynecol Scand. 2024;103(10):1943-1954. [CrossRef]
- Lipkind HS, Vazquez-Benitez G, Nordin JD, et al. Maternal and Infant Outcomes After Human Papillomavirus Vaccination in the Periconceptional Period or During Pregnancy. Obstet Gynecol. 2017;130(3). [CrossRef]
- Kharbanda EO, Vazquez-Benitez G, Lipkind HS, et al. Risk of Spontaneous Abortion After Inadvertent Human Papillomavirus Vaccination in Pregnancy. Obstet Gynecol. 2018;132(1). [CrossRef]
- Kharbanda EO, Vazquez-Benitez G, DeSilva MB, et al. Association of Inadvertent 9-Valent Human Papillomavirus Vaccine in Pregnancy With Spontaneous Abortion and Adverse Birth Outcomes. JAMA Netw Open. 2021;4(4):e214340-e214340. [CrossRef]
- Ricke DO. Cardiac adverse events post-vaccination. Brain Heart. 2025;3(2):1-15. [CrossRef]
- Montgomery J, Ryan M, Engler R, et al. Myocarditis Following Immunization With mRNA COVID-19 Vaccines in Members of the US Military. JAMA Cardiol. 2021;6(10):1202. [CrossRef]
- Kim HW, Jenista ER, Wendell DC, et al. Patients With Acute Myocarditis Following mRNA COVID-19 Vaccination. JAMA Cardiol. 2021;6(10):1196. [CrossRef]
- Marshall M, Ferguson ID, Lewis P, et al. Symptomatic Acute Myocarditis in 7 Adolescents After Pfizer-BioNTech COVID-19 Vaccination. Pediatrics. 2021;148(3):e2021052478. [CrossRef]
- Larson KF, Ammirati E, Adler ED, et al. Myocarditis After BNT162b2 and mRNA-1273 Vaccination. Circulation. 2021;144(6):506-508. [CrossRef]
- Rosner CM, Genovese L, Tehrani BN, et al. Myocarditis Temporally Associated With COVID-19 Vaccination. Circulation. 2021;144(6):502-505. [CrossRef]
- Ramírez-García A, Lozano Jiménez S, Darnaude Ximénez I, Gil Cacho A, Aguado-Noya R, Segovia Cubero J. Pericarditis after administration of the BNT162b2 mRNA COVID-19 vaccine. Rev Espanola Cardiol Engl Ed. 2021;74(12):1120-1122. [CrossRef]
- Das BB, Moskowitz WB, Taylor MB, Palmer A. Myocarditis and Pericarditis Following mRNA COVID-19 Vaccination: What Do We Know So Far? Children. 2021;8(7):607. [CrossRef]
- Pepe S, Gregory AT, Denniss AR. Myocarditis, Pericarditis and Cardiomyopathy After COVID-19 Vaccination. Heart Lung Circ. 2021;30(10):1425-1429. [CrossRef]
- Hudson B, Mantooth R, DeLaney M. Myocarditis and pericarditis after vaccination for COVID-19. J Am Coll Emerg Physicians Open. 2021;2(4):e12498. [CrossRef]
- Diaz G, Parsons G, Gering S, Meier A, Hutchinson I, Robicsek A. Myocarditis and Pericarditis After Vaccination for COVID-19. JAMA. 2021;326(12):3.
- Lane S, Yeomans A, Shakir S. Reports of myocarditis and pericarditis following mRNA COVID-19 vaccination: a systematic review of spontaneously reported data from the UK, Europe and the USA and of the scientific literature. BMJ Open. 2022;12(5):e059223. [CrossRef]
- Poddighe D, Castelli L, Marseglia GL, Bruni P. A sudden onset of a pseudo-neurological syndrome after HPV-16/18 AS04-adjuvated vaccine: might it be an autoimmune/inflammatory syndrome induced by adjuvants (ASIA) presenting as a somatoform disorder? Immunol Res. 2014;60(2):236-246. [CrossRef]
- Ozawa K, Hineno A, Kinoshita T, Ishihara S, Ikeda S ichi. Suspected Adverse Effects After Human Papillomavirus Vaccination: A Temporal Relationship Between Vaccine Administration and the Appearance of Symptoms in Japan. Drug Saf. 2017;40(12):1219-1229. [CrossRef]
- Hineno A, Ikeda SI. A Long-Term Observation on the Possible Adverse Effects in Japanese Adolescent Girls after Human Papillomavirus Vaccination. Vaccines. 2021;9(8):856. [CrossRef]
- Kinoshita T, Abe R ta, Hineno A, Tsunekawa K, Nakane S, Ikeda S ichi. Peripheral Sympathetic Nerve Dysfunction in Adolescent Japanese Girls Following Immunization with the Human Papillomavirus Vaccine. Intern Med. 2014;53(19):2185-2200. [CrossRef]
- CDC. COVID-19 Vaccination for Women Who Are Pregnant or Breastfeeding. https://www.cdc.gov/covid/vaccines/pregnant-or-breastfeeding.html. Accessed August 1, 2025.
- Geier M R, Stanbro H, Merril C R. Endotoxins in commercial vaccines. Appl Environ Microbiol. 1978;36(3):445-449. [CrossRef]
- Brito LA, Singh M. COMMENTARY: Acceptable Levels of Endotoxin in Vaccine Formulations During Preclinical Research. J Pharm Sci. 2011;100(1):34-37. [CrossRef]
- Bidne KL, Dickson MJ, Ross JW, Baumgard LH, Keating AF. Disruption of female reproductive function by endotoxins. Reproduction. 2018;155(4):R169-R181. [CrossRef]
- Costa JP, Jesus S, Colaço M, Duarte A, Soares E, Borges O. Endotoxin contamination of nanoparticle formulations: A concern in vaccine adjuvant mechanistic studies. Vaccine. 2023;41(23):3481-3485. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




