1. Introduction
Surgical resection remains a central component of the multidisciplinary management of tumors of the central nervous system (CNS), particularly diffuse gliomas. Despite major advances in molecular classification, imaging, and adjuvant therapies, cytoreductive surgery continues to represent the first therapeutic intervention for most patients. In contemporary neuro-oncology, surgery is still primarily evaluated in terms of extent of resection (EOR) and its association with survival outcomes. Large modern series and meta-analyses have consistently confirmed that a greater EOR correlates with improved overall survival in glioblastoma and other diffuse gliomas, reinforcing the paradigm of “maximal safe resection” as a cornerstone of treatment [
1,
2]. However, accumulating clinical and biological evidence suggests that the relationship between surgical cytoreduction and outcome is more complex than a simple quantitative measure of tumor removal. Even in the context of gross total resection, the majority of malignant CNS tumors recur, often with aggressive behavior and limited responsiveness to subsequent therapies. Contemporary analyses have shown that survival curves frequently overlap across different degrees of resection, particularly when molecular features are taken into account, indicating that EOR alone does not fully capture post-surgical disease dynamics [
3,
4].
One major limitation of surgery lies in the biological diffusivity of CNS tumors. Modern imaging–pathology correlation studies have demonstrated that tumor cells with distinct molecular and transcriptional programs persist outside the resection cavity, even after apparently complete resections [
5]. These findings challenge the notion that anatomical completeness equates to biological eradication.
In parallel, increasing attention has been devoted to the tumor microenvironment as a critical determinant of tumor behavior, recurrence, and treatment resistance. The CNS tumor microenvironment is now recognized as an active and adaptive ecosystem, composed of tumor cells, resident microglia, infiltrating macrophages, endothelial cells, and extracellular matrix components. Recent comprehensive reviews have highlighted the role of neuroinflammation, immune suppression, and hypoxia in sustaining tumor growth and mediating resistance to radiotherapy and systemic treatments [
6,
7,
8,
9].
Importantly, surgical intervention itself represents a profound biological perturbation of this ecosystem. Tissue injury induced by resection triggers inflammatory cascades, alters vascular permeability, disrupts the blood–brain barrier, and reshapes oxygen and nutrient gradients in the peritumoral region. Experimental and clinical data indicate that these surgery-induced changes can promote tumor cell plasticity, invasion, and resistance mechanisms, potentially influencing early recurrence patterns [
10,
11]
.
From an evolutionary standpoint, cancer progression is increasingly understood as a dynamic process driven by selective pressures acting on heterogeneous tumor cell populations. Within this framework, therapeutic interventions, including surgery, may function as evolutionary bottlenecks, eliminating sensitive clones while allowing resistant subpopulations to expand. Longitudinal genomic studies have provided compelling evidence that gliomas undergo marked clonal evolution between diagnosis and recurrence. Using paired primary and recurrent tumor samples, Barthel et al. [
12] and Kim et al. [
13] demonstrated that recurrent tumors frequently harbor distinct genetic and transcriptional profiles, consistent with treatment-driven selection.
The clinical relevance of tumor evolution has been further underscored by the integration of molecular profiling into the World Health Organization (WHO) classification of CNS tumors. Large-scale studies have shown that molecular features such as IDH mutation status, TERT promoter alterations, EGFR amplification, and H3 histone mutations not only define prognosis but also influence patterns of invasion, recurrence, and therapeutic vulnerability [
14,
15]. Within this context, it remains uncertain whether surgical strategies grounded exclusively in anatomical criteria are sufficient to account for the biological heterogeneity and infiltrative behavior of CNS tumors.
Collectively, these observations support a shift in perspective: surgery should no longer be regarded merely as a technical step preceding adjuvant therapies, but rather as a biological driver that actively shapes tumor evolution and treatment response. Understanding how surgical trauma modulates the tumor microenvironment, exerts selective pressure on residual tumor cells, and interacts with radiotherapy and systemic treatments is essential for refining postoperative strategies. In this review, we explore the emerging concept of surgical biology in CNS tumors, examining its implications for recurrence patterns, molecular evolution, and the development of integrated, biology-driven approaches to precision neuro-oncology.
2. Surgical Trauma as a Biological Event
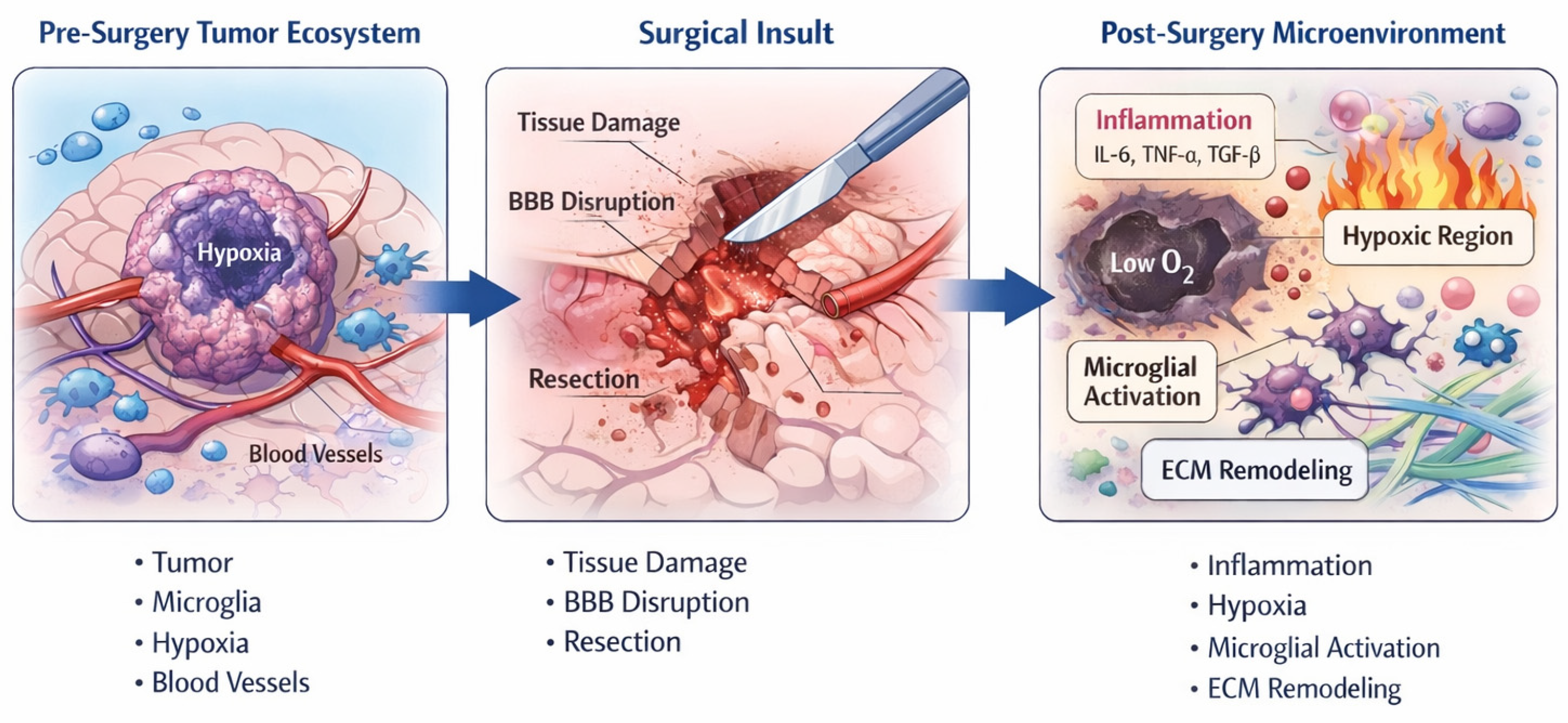
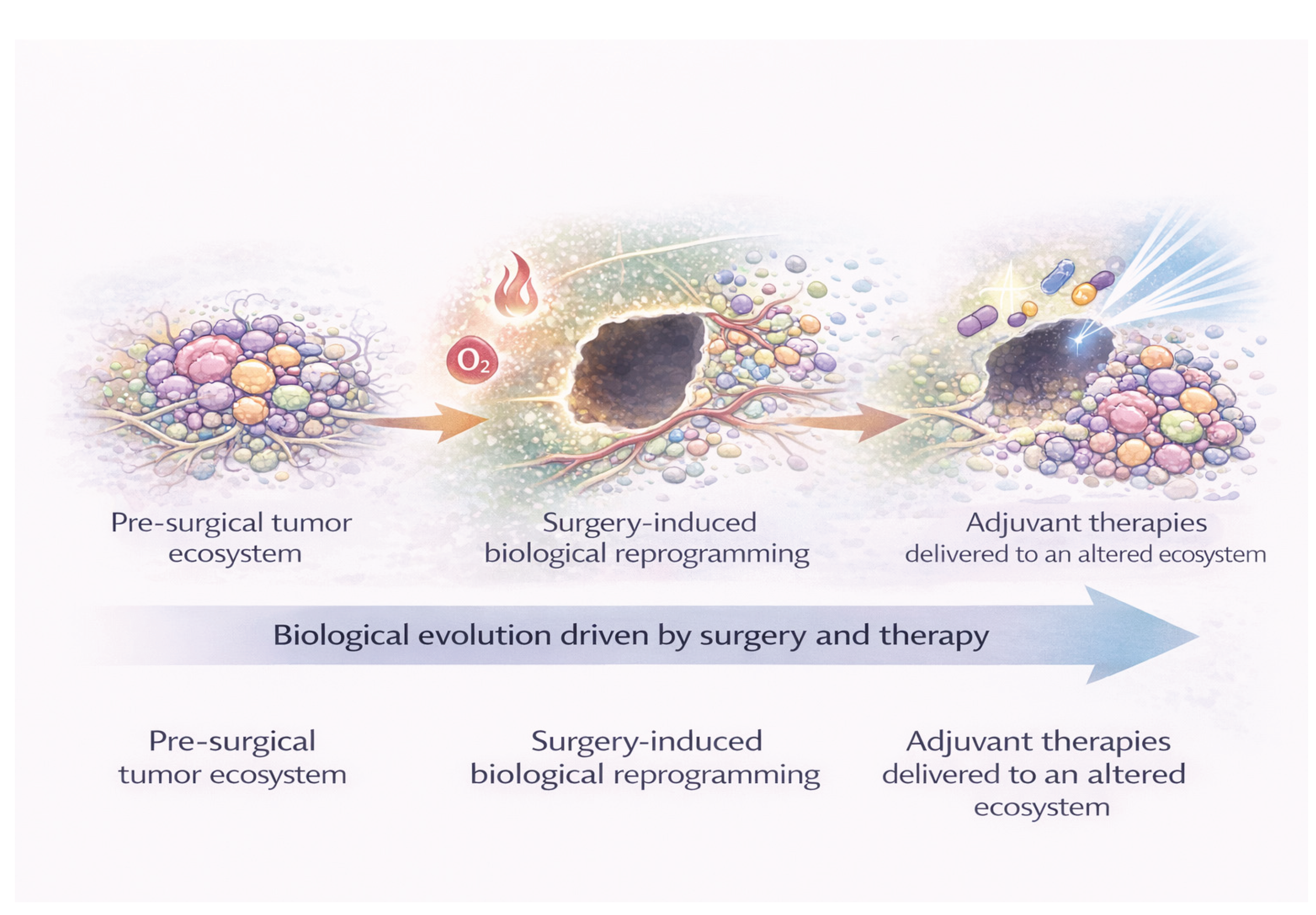
Surgical resection of CNS tumors has traditionally been conceptualized as a mechanical intervention aimed at cytoreduction and symptom relief. However, accumulating experimental and clinical evidence indicates that surgery constitutes a profound biological perturbation of the tumor ecosystem, with consequences that extend well beyond the immediate removal of tumor tissue. Rather than representing a neutral reset of the disease burden, surgical trauma initiates a cascade of inflammatory, vascular, and metabolic changes that reshape the residual tumor microenvironment and may influence subsequent tumor behavior (
Figure 1).
Tissue injury induced by resection triggers an acute inflammatory response characterized by the release of cytokines, chemokines, and growth factors. Recent studies have shown that surgical trauma promotes the activation of microglia and recruitment of peripheral macrophages, leading to a transient but biologically relevant pro-inflammatory milieu within the peritumoral brain. Buonfiglioli et al. [
16] reported that microglia–macrophage activation states are highly plastic and can shift toward tumor-supportive phenotypes in response to tissue damage. Similarly, Van der Sanden et al. reported that post-surgical inflammatory signaling may enhance tumor cell survival and invasiveness through cytokine-mediated pathways [
17].
In parallel, surgical disruption of the blood-brain barrier (BBB) represents a critical biological consequence of tumor resection. BBB breakdown alters vascular permeability, facilitates immune cell infiltration, and modifies the pharmacokinetics of adjuvant therapies. Advanced imaging and molecular studies have shown that BBB permeability remains altered for weeks following surgery, potentially creating spatially heterogeneous niches within the resection cavity and surrounding brain tissue. Brooks and Parrinello highlighted how vascular and endothelial responses to tissue injury can contribute to adaptive tumor responses, including angiogenesis and metabolic reprogramming [
18].
Another key consequence of surgical trauma is the generation of peri-cavitary hypoxia. Resection inevitably disrupts local blood supply, leading to transient ischemia and altered oxygen gradients in the surrounding brain parenchyma. Hypoxic conditions are well recognized as potent drivers of tumor aggressiveness, promoting stemness, invasion, and resistance to radiotherapy. Recent translational studies have demonstrated that hypoxia-inducible signaling pathways, including HIF-1α–mediated transcriptional programs, are upregulated in residual tumor cells located near the resection margin. Singh et al. and Bayona et al. provided evidence that hypoxic stress fosters adaptive phenotypes associated with treatment resistance and recurrence [
8,
19].
Beyond inflammatory and hypoxic effects, surgical trauma induces extensive remodeling of the extracellular matrix (ECM) and mechanical properties of the tumor niches. Changes in ECM composition and stiffness have been shown to modulate tumor cell migration and invasion. Suarez-Meade et al. emphasized that physical and biochemical alterations of the tumor microenvironment following therapeutic interventions can actively shape tumor evolution [
20]. In the post-surgical setting, ECM remodeling may facilitate the dispersal of invasive tumor cells into surrounding brain tissue, contributing to diffuse recurrence patterns.
Collectively, these processes support the notion that surgical resection initiates a biological reprogramming of the tumor microenvironment rather than simply debulking disease. As schematically illustrated in
Figure 1, the transition from the pre-surgical tumor ecosystem to the post-surgical microenvironment involves coordinated changes in inflammation, vascular integrity, oxygenation, and stromal interactions. These surgery-induced alterations may create selective conditions that favor the survival and expansion of tumor cell subpopulations with enhanced adaptive capacity, thereby influencing early recurrence dynamics and response to adjuvant therapies.
Recognizing surgical trauma as a biologically active event has important implications for postoperative management. It suggests that the timing, targeting, and intensity of adjuvant radiotherapy and systemic treatments should be considered in light of the evolving post-surgical tumor ecosystem. Rather than treating surgery and adjuvant therapies as sequential and independent steps, a biologically informed integration of surgical and postoperative strategies may be required to counteract surgery-induced adaptive responses.
3. Clonal Selection and Tumor Evolution After Surgical Resection
Diffuse CNS tumors, and gliomas in particular, are characterized by marked intratumoral heterogeneity at genetic, epigenetic, and transcriptional levels [
21]. Contemporary molecular profiling studies have demonstrated that even treatment-naïve tumors harbor multiple coexisting subclones with distinct biological properties, including differential proliferative capacity, invasive potential, and therapeutic sensitivity [
22]. This intrinsic heterogeneity provides the substrate upon which therapeutic interventions, including surgery, can act as selective pressures, shaping tumor evolution over time.
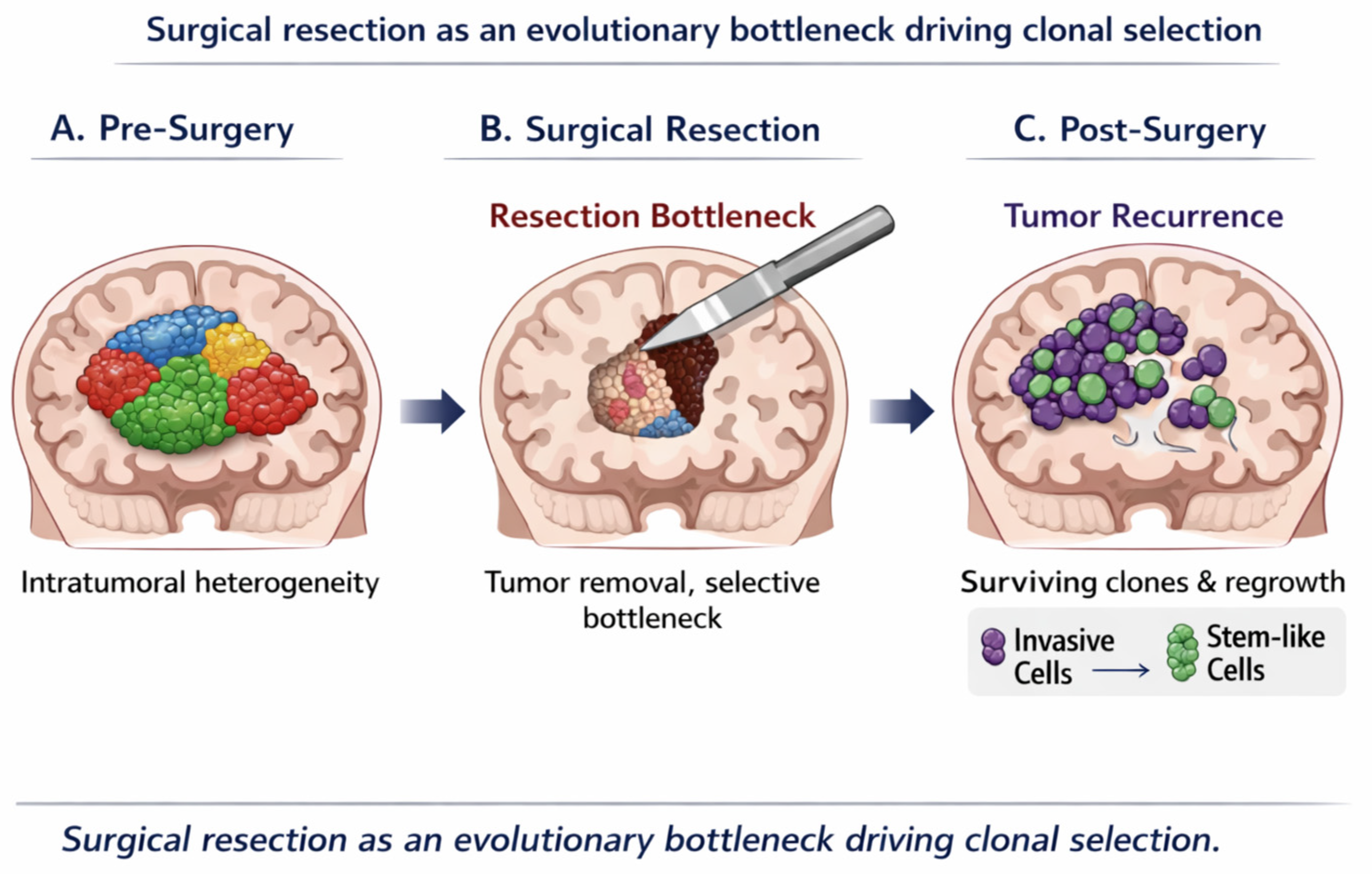
From an evolutionary perspective, surgical resection can be conceptualized as an acute selective bottleneck that dramatically reduces tumor burden while simultaneously altering the ecological context in which residual tumor cells persist. Rather than eliminating the disease uniformly, surgery preferentially removes spatially accessible and often more proliferative tumor populations, while leaving behind subclones adapted to hostile microenvironmental conditions such as hypoxia, nutrient deprivation, and immune surveillance. This concept aligns with modern evolutionary models of cancer progression, in which treatment-induced bottlenecks favor the expansion of resistant phenotypes rather than complete eradication [
23]
.
Compelling evidence for surgery-driven clonal selection has emerged from longitudinal genomic analyses of paired primary and recurrent CNS tumors. Using integrative genomic and transcriptomic profiling, Barthel et al. [
12] demonstrated that recurrent gliomas frequently diverge substantially from their primary counterparts, acquiring novel driver alterations and transcriptional programs consistent with adaptive evolution under therapeutic pressure. Importantly, these changes were not random but followed reproducible evolutionary trajectories, suggesting that treatment-related selective forces actively shape recurrence biology.
Spatially resolved analyses have further refined this concept by revealing that distinct tumor regions contribute differentially to recurrence [
24]. Jung et al. [
25] highlighted that subclones residing at the invasive margins of the tumor, often less affected by surgical resection, are disproportionately represented in recurrent disease. These marginal populations frequently exhibit gene expression signatures associated with migration, stemness, and therapy resistance, supporting the hypothesis that surgery selects for biologically aggressive phenotypes rather than merely reducing tumor volume [
26].
Tumor stem-like cells appear to play a central role in this process. Glioma stem cells (GSCs), characterized by enhanced self-renewal capacity and resistance to cytotoxic therapies, are preferentially enriched in hypoxic and peri-vascular niches that may be relatively preserved following resection [
27]. Recent studies have shown that surgical trauma and subsequent microenvironmental remodeling can further promote stem-like states through inflammatory and hypoxia-driven signaling pathways providing mechanistic evidence linking microenvironmental stress to stem cell enrichment and clonal dominance at recurrence [
28,
29]. Importantly, clonal selection after surgery is not limited to genetic alterations but also involves epigenetic and transcriptional reprogramming. Single-cell sequencing studies have revealed that residual tumor cells can undergo rapid phenotypic transitions in response to post-surgical cues, adopting mesenchymal or invasive transcriptional states associated with poor prognosis [
30]. Neftel et al. [
31] demonstrated that glioblastoma cells dynamically transition between cellular states rather than remaining fixed in discrete lineages, highlighting the plastic nature of tumor evolution. Surgical perturbation may accelerate these transitions by reshaping the surrounding niche.
Collectively these findings support a model in which surgical resection acts as an evolutionary filter, selectively favoring tumor cell populations with enhanced adaptive capacity (
Figure 2). The biological properties of the residual disease, rather than the absolute extent of resection, may therefore be critical determinants of recurrence pattern, aggressiveness, and response to adjuvant therapies. Recognizing surgery as a driver of clonal selection provides a conceptual framework for understanding why anatomically complete resections do not necessarily translate into durable disease control and underscores the need for postoperative strategies tailored to the evolving biology of residual tumor cells.
4. Surgical Margins Versus Molecular Margins
The concept of surgical margins has historically been grounded in anatomical and radiological criteria, with gross total resection is commonly defined by the absence of contrast-enhancing tumor on postoperative magnetic resonance imaging [
32]. While this approach remains clinically relevant, growing molecular and biological evidence indicates that anatomical margins do not coincide with the true biological extent of CNS tumors. In diffuse gliomas and other infiltrative brain tumors, residual disease often persists beyond visible surgical boundaries, challenging the adequacy of anatomy-based definitions of completeness.
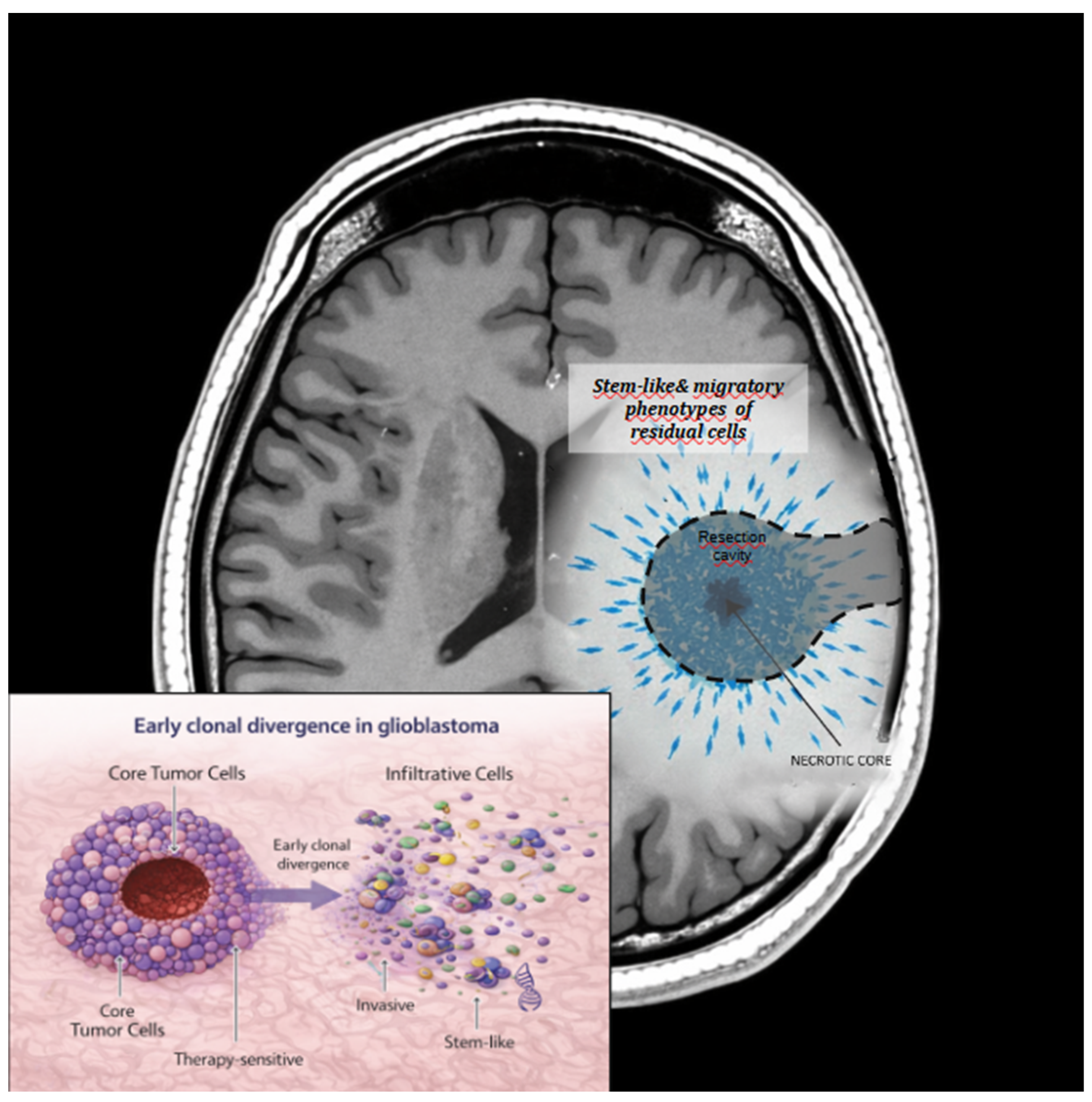
Recent molecular and imaging studies have demonstrated that tumor cell infiltration extends far beyond radiographically detectable lesions [
33]. Using advanced imaging-genomic correlations, Gill et al. [
34] showed that tumor-associated molecular alterations can be detected in non-enhancing brain regions adjacent to the surgical cavity. These findings support the notion that the apparent surgical margin frequently underestimates the spatial distribution of biologically active tumor cells.
At the molecular level, residual tumor cells located at the invasive front often harbor distinct genetic and transcriptional profiles compared with cells from the tumor core. Bhaduri et al. [
35] demonstrated that peripheral tumor populations exhibit stem-like and migratory phenotypes associated with treatment resistance. Similarly, Nomura et al. [
36] identified transcriptional programs at the tumor-brain interface that promote invasion and adaptation, underscoring the biological relevance of marginal disease.
The dissociation between surgical and molecular margins is further amplified by intratumoral heterogeneity and clonal evolution. As discussed in the previous section, surgical resection acts as an evolutionary bottleneck, preferentially removing central tumor populations while sparing infiltrative cells at the periphery. These marginal cells frequently exhibit molecular features associated with aggressive behavior, including mesenchymal transition, metabolic flexibility, and resistance to radiotherapy and chemotherapy [
31]. Suvà and Tirosh [
37] highlighted how spatially distinct tumor compartments contribute disproportionately to recurrence, emphasizing that the biological “edge” of the tumor may be more relevant than the radiological boundary.
Molecular classification has further refined this concept. In IDH-wildtype glioblastoma, alterations such as EGFR amplification, TERT promoter mutations, and chromosomal instability have been associated with highly infiltrative growth patterns that extend beyond surgical margins. Conversely, IDH-mutant gliomas often demonstrate diffuse infiltration along white matter tracts despite relatively indolent radiological appearances. Large integrative analyses by Louis et al. [
14] and Weller et al. [
38] underscore that molecular features, rather than anatomical extent alone, are key determinants of tumor behavior and recurrence risk.
These observations have profound implications for postoperative management, particularly for radiotherapy planning. Target volume delineation based solely on surgical cavities and contrast-enhancing disease may fail to encompass biologically relevant residual tumor regions [
39]. Emerging evidence suggests that incorporating molecular and functional imaging biomarkers, such as diffusion abnormalities, metabolic signatures, or radiomic features, may better reflect the true extent of disease. Kickingereder et al. [
40] demonstrated that radiomic features beyond contrast enhancement correlate with molecular aggressiveness and recurrence patterns, supporting a shift toward biologically informed target definition.
As illustrated in
Figure 3, the discrepancy between surgical and molecular margins represents a fundamental limitation of anatomy-based treatment paradigms. Recognizing that “complete resection” does not equate to biological eradication is essential for understanding recurrence dynamics and optimizing adjuvant therapies. A paradigm that integrates surgical anatomy with molecular and microenvironmental information may offer a more accurate framework for risk stratification and personalized postoperative treatment strategies.
5. Surgery, Recurrence Patterns, and Molecular Shift
Despite maximal surgical resection and contemporary multimodal treatment, recurrence remains an almost universal feature of malignant CNS tumors, particularly glioblastoma. Importantly, tumor recurrence is not a random event but follows reproducible spatial and biological patterns that reflect the interaction between surgical intervention and tumor biology. Increasing evidence suggests that surgery not only influences the location of recurrence but also shapes its molecular and phenotypic characteristics, contributing to a progressive shift toward more aggressive disease.
Several modern imaging-based studies have demonstrated that the majority of glioblastoma recurrences arise within or adjacent to the surgical cavity, yet a substantial proportion occur at the margins or at distant sites, particularly in tumors with highly infiltrative growth patterns. Using longitudinal imaging analyses, Chiranth et al. [
41] reported that distant progression is an aggressive progression pattern associated with poor prognosis. These findings indicate that recurrence patterns are not solely dictated by treatment fields but are deeply influenced by the biological properties of residual tumor cells.
From a molecular standpoint, recurrent tumors often differ significantly from their primary counterparts. Integrated genomic studies have shown that surgical resection and subsequent therapies select for tumor cell populations with distinct genetic and transcriptional profiles. Barthel et al. [
12] demonstrated that recurrent gliomas frequently acquire novel driver alterations and chromosomal aberrations that were absent or subclonal at diagnosis. This molecular divergence supports the concept that recurrence represents a selective evolved disease state rather than simple regrowth of the primary tumor.
Single-cell and bulk RNA sequencing studies have revealed that recurrent glioblastomas are enriched for mesenchymal-like and stem-like transcriptional states associated with invasiveness, immune evasion, and resistance to therapy. Neftel et al. [
31] showed that glioblastoma cells dynamically transition between cellular states, with therapy acting as a catalyst for phenotypic switching. Surgical trauma, adjuvant therapies and the associated microenvironmental changes described in
Section 2 may initiate and accelerate this plasticity, favoring the emergence of aggressive recurrent phenotypes.
Spatial analyses have further linked molecular evolution to recurrence patterns. Tumor cells located at the invasive front, often beyond the surgical margin, are more likely to seed marginal or distant recurrences and exhibit molecular features distinct from those of the tumor core. Bhaduri et al. [
35] identified peripheral tumor populations with enhanced migratory capacity and resistance to cytotoxic stress, underscoring the biological relevance of marginal disease. These observations align with the concept of a molecular margin extending beyond the anatomical resection boundary, as discussed in
Section 4.
The molecular shift observed at recurrence has important clinical implications. Recurrent tumors frequently display reduced sensitivity to standard therapies, including radiotherapy and temozolomide, and may exhibit altered immune landscapes that limit the efficacy of immunotherapeutic approaches. Wang et al. [
42] reported significant changes in immune cell composition and checkpoint expression between primary and recurrent tumors, highlighting the adaptive nature of the disease. These changes may, in part, reflect surgery-induced spatial and biological selection pressures acting on heterogeneous tumor populations.
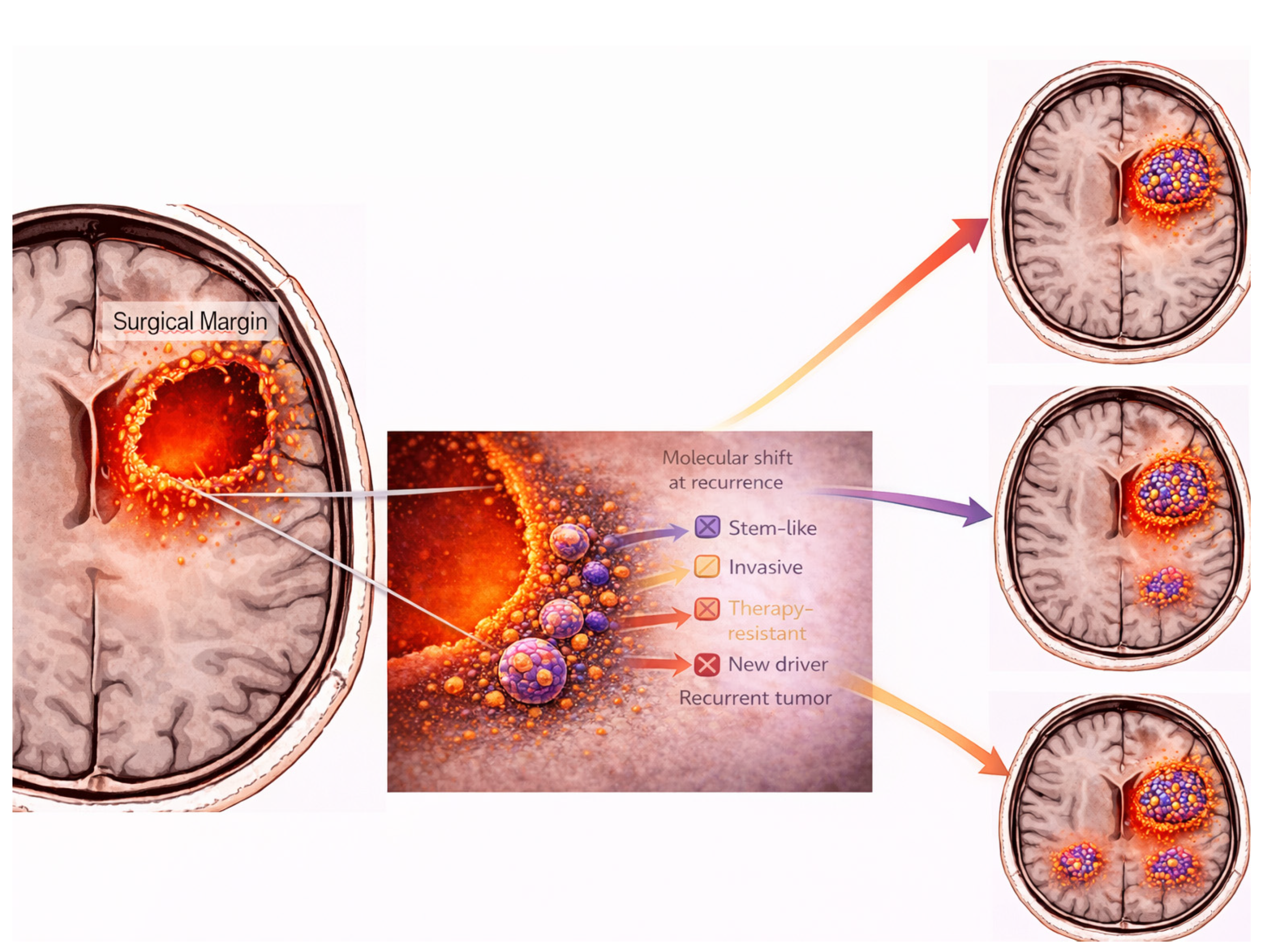
Collectively, these findings support a model in which surgery influences not only when and where tumors recur, but also how they recur at the molecular level. Recurrence should therefore be viewed as the outcome of an evolutionary process shaped by surgical cytoreduction, microenvironmental remodeling, and subsequent adjuvant therapies. As schematically summarized in
Figure 4, the interplay between surgical margins, residual biologically active disease, and clonal evolution determines recurrence patterns and molecular progression.
Understanding the biological underpinnings of recurrence patterns may help refine postoperative risk stratification and guide more personalized treatment strategies. Rather than assuming biological continuity between primary and recurrent disease, clinicians and researchers should consider recurrence as a distinct, evolved entity that emerges from selective pressures imposed by surgery and multimodal therapy.
Beyond the surgical margin, residual molecularly active disease may persist and drive local, marginal, or distant recurrence. Selective pressures induced by surgery and multimodal therapy promote clonal evolution, resulting in molecular and phenotypic shifts at recurrence, including stem-like features, invasiveness, therapy resistance, or new oncogenic drivers.
6. Implications for Radiotherapy and Adjuvant Therapies
The recognition of surgery as a biologically active event, rather than a purely cytoreductive intervention, has profound implications for postoperative radiotherapy and systemic treatments in CNS tumors. Traditional adjuvant strategies have largely been designed under the assumption that surgery merely reduces tumor burden, often underestimating the extent to which surgical intervention may alter tumor biology both spatially and molecularly. However, the evidence discussed in the preceding sections indicates that surgical resection reshapes the tumor microenvironment, drives clonal selection, and contributes to molecular evolution, thereby influencing treatment response and patterns of failure as schematically illustrated in
Figure 5. Radiotherapy remains a cornerstone of postoperative management in high-grade gliomas, yet its efficacy is tightly linked to the biological characteristics of residual disease. Surgery-induced hypoxia, inflammation, and vascular remodeling can directly modulate radiosensitivity. Hypoxic tumor cells are well known to exhibit reduced responsiveness to ionizing radiation due to impaired oxygen-mediated fixation of DNA damage. Recent translational studies have demonstrated that peri-cavitary hypoxia following surgical resection may persist during the early postoperative period, potentially reducing the effectiveness of adjuvant radiotherapy when delivered to biologically unfavorable niches. Horsman and Overgaard [
43] emphasized that spatial and temporal hypoxia remains a critical determinant of radiation response in brain tumors.
Target volume delineation represents another major challenge. Contemporary radiotherapy planning is primarily guided by anatomical imaging, incorporating the surgical cavity and surrounding margins based on contrast enhancement and edema. However, as illustrated above, the molecular margin of disease often extends beyond these radiological boundaries. Failure to account for biologically active infiltrative tumor cells may contribute to high rate of recurrences. Advanced imaging and radiomic approaches have shown promise in identifying regions of aggressive biology beyond contrast-enhancing disease. Kim et al. [
44] demonstrated that radiomic features derived from non-enhancing regions correlate with molecular aggressiveness and recurrence risk, supporting a shift toward biologically informed target definition.
Surgical biology also intersects with systemic therapy responsiveness. Temozolomide efficacy, for instance, may be influenced by the selective survival of resistant subclones enriched at the tumor margins following resection. Molecular evolution at recurrence frequently includes alterations associated with DNA repair, cell cycle regulation, and metabolic adaptation, which may reduce sensitivity to alkylating agents. Barthel et al. [
12] and Kim et al. [
13] provided evidence that recurrent tumors represent biologically distinct entities with altered therapeutic vulnerabilities.
Immunotherapeutic strategies are similarly affected by surgery-induced changes in the tumor microenvironment. Surgical trauma triggers inflammatory signaling and immune cell recruitment, yet this response may paradoxically promote immunosuppressive phenotypes within the tumor niche. Recent studies have highlighted dynamic shifts in immune cell composition and checkpoint expression following standard therapy, including surgery. Pombo Antunes et al. [
45] showed that macrophage polarization and T-cell dysfunction evolve over time, potentially limiting the efficacy of immune-based treatments if not appropriately timed or combined with other modalities.
These observations collectively argue for a more integrated approach to postoperative therapy that accounts for the evolving biological landscape shaped by surgery. Rather than viewing surgery, radiotherapy, and systemic treatments as sequential and independent steps, a surgery-informed, biology-driven therapeutic framework may be required. This could include adaptive radiotherapy strategies guided by molecular imaging, optimized timing of adjuvant treatments to counteract surgery-induced hypoxia or inflammation, and tailored systemic therapies targeting biologically selected residual disease.
In this context, surgery should be considered an integral component of precision neuro-oncology, not only as a means of tissue removal but also as a source of biological information and a determinant of subsequent treatment response. Incorporating surgical biology into postoperative decision-making may help refine risk stratification, improve local control, and ultimately mitigate the aggressive evolutionary trajectories that characterize recurrent CNS tumors.
7. Future Directions: Toward Surgery-Integrated Precision Neuro-Oncology
The cumulative evidence reviewed in the preceding sections supports a paradigm shift in the conceptualization of surgery within neuro-oncology. Rather than representing a purely cytoreductive step preceding adjuvant therapies, surgical resection emerges as a central biological determinant of tumor evolution, recurrence patterns, and treatment response. This recognition opens new avenues for integrating surgical information into precision neuro-oncology frameworks.
One major future direction lies in the biological exploitation of surgical intervention. Surgical specimens already serve as the primary source for histological and molecular diagnosis; however, their potential to inform postoperative risk stratification remains underutilized. Advances in spatial transcriptomics and single-cell sequencing now allow high-resolution mapping of tumor heterogeneity and microenvironmental interactions across different tumor regions, including the invasive margin. Ravi et al. [
46]
demonstrated that molecular programs associated with invasion and therapy resistance are spatially enriched at tumor–brain interfaces, suggesting that surgical margin-specific profiling could provide actionable biological insights.
Beyond tissue-based analyses, surgery may also act as a temporal anchor for dynamic disease monitoring. Liquid biopsy approaches, including circulating tumor DNA and extracellular vesicles, have shown promise in capturing tumor evolution longitudinally. Miller et al. [
47] and Sabedot et al. [
48] highlighted how postoperative molecular dynamics may reflect residual disease burden and emerging resistance mechanisms. Integrating surgical timing with liquid biopsy could enable early identification of high-risk evolutionary trajectories before radiological recurrence becomes evident.
Artificial intelligence (AI) and advanced imaging analytics represent another critical frontier. Radiomic and deep learning approaches applied to perioperative imaging have demonstrated the ability to infer molecular features, invasive behavior, and recurrence risk from standard MRI sequences. Kickingereder et al. [
49] and Chang et al. [
50] suggested that AI-derived imaging biomarkers could bridge the gap between anatomical margins and molecular disease, enabling surgery-informed radiotherapy planning and adaptive treatment strategies.
These technological advances also raise the possibility of adaptive, biology-guided adjuvant therapy. Rather than applying uniform postoperative treatment protocols, future strategies may tailor radiotherapy volumes, dose distributions, and systemic therapy choices based on surgery-induced biological states. Early-phase studies exploring adaptive radiotherapy and response-guided treatment intensification provide proof-of-concept that dynamic treatment modification is feasible. Tseng et al. [
51] emphasized that integrating biological information into adaptive workflows may improve local control while limiting unnecessary toxicity.
Crucially, realizing a surgery-integrated precision oncology model will require a shift from discipline-specific workflows to truly multidisciplinary integration. Surgeons, radiation oncologists, medical oncologists, molecular pathologists, and data scientists must collaborate around a shared biological framework, in which surgical decisions are informed by anticipated downstream effects on tumor evolution and therapeutic response. Such integration aligns with emerging systems-level approaches to cancer care, as advocated by Prados et al. [
52].
In this context, surgery should be repositioned not as the beginning of a linear treatment sequence, but as a biological inflection point that shapes the trajectory of disease. Embracing this perspective may facilitate the development of postoperative strategies that anticipate, rather than react to, tumor evolution. Ultimately, surgery-integrated precision neuro-oncology has the potential to transform postoperative management from a standardized protocol into a biologically informed, adaptive continuum of care.
8. Discussion: Surgery as a Biological Driver of Tumor Evolution in CNS Tumors
This review advances a conceptual reinterpretation of surgery in CNS tumors, proposing that surgical resection should be understood not merely as a cytoreductive intervention, but as a biologically active event that shapes tumor evolution, recurrence patterns, and treatment response. By integrating evidence from tumor biology, clonal evolution, imaging, and clinical outcomes, we argue that surgery represents a critical inflection point in the disease trajectory rather than a neutral prelude to adjuvant therapy. Importantly, recognizing surgery as a biologically transformative event opens the possibility of reconsidering how subsequent therapies are sequenced and integrated, with the goal of adapting treatment strategies to the biological state imposed by surgical intervention itself.
A central message emerging from this review is that anatomical completeness does not equate to biological completeness. While extent of resection remains an important prognostic factor, its clinical value cannot be fully understood without considering the biological properties of residual disease. The dissociation between surgical margins and molecular margins highlights a fundamental limitation of anatomy-based paradigms and provides a biological explanation for the high rate of recurrence observed even after radiologically complete resections. This perspective reconciles seemingly paradoxical observations in the literature, such as improved survival with greater resection without corresponding durable disease control. More importantly, it suggests that postoperative strategies should not simply assume a continuation of preoperative tumor biology, but rather be recalibrated in light of the new biological equilibrium established after surgery.
By framing surgery as an evolutionary bottleneck, this review situates surgical intervention within contemporary models of cancer evolution. Surgical resection imposes a strong selective pressure on heterogeneous tumor populations, preferentially eliminating dominant but therapy-sensitive clones while allowing adaptive, invasive, or stem-like populations to persist. The recurrent tumor that emerges is therefore not simply a regrowth of the primary lesion, but an evolved biological entity, shaped by the combined pressures of surgery and adjuvant therapies. This evolutionary framing provides a unifying explanation for molecular divergence at recurrence and for the frequent emergence of treatment resistance. It also implies that postoperative treatments might achieve greater efficacy if they are timed and selected to specifically target the biological characteristics of the residual, surgery-selected populations, rather than following rigid treatment sequences.
Importantly, this review emphasizes that the biological consequences of surgery extend beyond tumor cell selection to include microenvironmental reprogramming. Surgical trauma induces inflammation, hypoxia, vascular remodeling, and immune modulation, all of which influence tumor cell behavior and therapeutic sensitivity. These changes challenge the implicit assumption that postoperative radiotherapy and systemic treatments act on a biological substrate comparable to the pre-surgical tumor. Instead, adjuvant therapies are delivered into a dynamically altered ecosystem, with important implications for treatment timing, target definition, and therapeutic efficacy. A deeper understanding of these processes may allow clinicians to integrate treatments that counteract or exploit surgery-induced biological changes, potentially improving local control and delaying resistance.
Another key contribution of this review lies in its multidisciplinary integration. By bridging neurosurgery, radiation oncology, medical oncology, molecular pathology, and imaging sciences, we propose a framework in which surgical decisions are informed not only by immediate anatomical considerations but also by anticipated downstream biological effects. Such a framework naturally leads to reconsideration of conventional treatment sequencing, encouraging closer coordination among specialties to design treatment pathways that dynamically respond to biology modified by prior interventions. This integrative perspective aligns with emerging precision oncology paradigms and underscores the need for closer collaboration across disciplines to address the complex biology of CNS tumors.
At the same time, several limitations should be acknowledged. Much of the evidence supporting surgery-induced biological effects is derived from retrospective analyses, preclinical models, or indirect clinical observations. Prospective studies specifically designed to capture perioperative biological dynamics remain scarce. Furthermore, translating biological insights into actionable changes in clinical practice, such as adaptive radiotherapy, surgery-informed systemic therapy, or modified treatment sequencing, will require careful validation to avoid overtreatment or unnecessary toxicity. Nonetheless, the convergence of evidence across multiple domains supports the validity and clinical relevance of the proposed conceptual framework.
In summary, the message of this review is not that surgery is detrimental, but that surgery matters biologically. Recognizing surgery as a driver of tumor evolution rather than a neutral intervention offers a more coherent understanding of recurrence, resistance, and treatment failure in CNS tumors. At the same time, acknowledging the biological impact of surgical resection invites a shift from rigid, linear treatment sequences toward adaptive therapeutic strategies that integrate and respond to treatment-induced biological changes. Ultimately, this perspective supports the development of a dynamic, biology-driven continuum of care, in which surgical intervention becomes a fully integrated component of precision neuro-oncology aimed at improving long-term patient outcomes.
9. Conclusions
Surgery should be viewed as a biological “inflection point” capable of shaping residual disease behavior and subsequent treatment response. Because anatomical completeness does not necessarily equate to biological completeness, the concept of a molecular margin provides a plausible framework to explain marginal and distant recurrences. Importantly, postoperative radiotherapy and systemic therapies are delivered into a dynamically altered ecosystem, reinforcing the need for biology-informed timing and targeting. Moving forward, perioperative spatial profiling, integrated imaging–liquid biopsy monitoring, and truly multidisciplinary workflows will be essential to translate surgical biology into actionable postoperative strategies.
Author Contributions
Conceptualization, P.T. and S.C.; methodology, P.T., F.D., G.R., and G.B.; validation, P.T., G.R. and F.D.; formal analysis, P.T. and F.D.; investigation, P.T., F.D., G.R., G.B., P.P., M.V. and T.C.; resources, S.C., G.M., G.T. and L.C.; data curation, P.T. and F.D.; writing-original draft preparation, P.T. and F.D.; writing-review and editing, P.T., G.R., G.B., P.P., M.V., T.C., G.T., L.C., G.M. and S.C.; visualization, P.T., F.D.; supervision, G.M and S.C.; project administration, P.T. and S.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding. The work was carried out within the authors’ institutional academic activities at their respective affiliated institutions.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Brown, T.J.; Brennan, M.C.; Li, M.; Church, E.W.; Brandmeir, N.J.; Rakszawski, K.L.; Patel, A.S.; Rizk, E.B.; Suki, D.; Sawaya, R.; et al. Association of the Extent of Resection With Survival in Glioblastoma: A Systematic Review and Meta-analysis. JAMA Oncol. 2016, 2, 1460–1469. [Google Scholar] [CrossRef]
- Geens, W.; Rizani, G.; Del Gaudio, N.; Buyck, F.; Van Gestel, F.; Bruneau, M.; Neyns, B.; Duerinck, J. Extent of resection and its association with overall survival in newly diagnosed IDH wildtype glioblastoma treated with concomitant radiochemotherapy: A systematic review and meta-analysis. Brain Spine 2025, 5, 105867. [Google Scholar] [CrossRef]
- Molinaro, A.M.; Hervey-Jumper, S.; Morshed, R.A.; Young, J.; Han, S.J.; Chunduru, P.; Zhang, Y.; Phillips, J.J.; Shai, A.; Lafontaine, M.; et al. Association of Maximal Extent of Resection of Contrast-Enhanced and Non-Contrast-Enhanced Tumor With Survival Within Molecular Subgroups of Patients With Newly Diagnosed Glioblastoma. JAMA Oncol. 2020, 6, 495–503. [Google Scholar] [CrossRef]
- Cahill, D.P. Extent of Resection of Glioblastoma: A Critical Evaluation in the Molecular Era. Neurosurg. Clin. N. Am. 2021, 32, 23–29. [Google Scholar] [CrossRef]
- Quail, D.F.; Joyce, J.A. The Microenvironmental Landscape of Brain Tumors. Cancer Cell 2017, 31, 326–341. [Google Scholar] [CrossRef]
- Kalluri, A.L.; Shah, P.P.; Lim, M. The Tumor Immune Microenvironment in Primary CNS Neoplasms: A Review of Current Knowledge and Therapeutic Approaches. Int. J. Mol. Sci. 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Feldman, L. Hypoxia within the glioblastoma tumor microenvironment: A master saboteur of novel treatments. Front. Immunol. 2024, 15, 1384249. [Google Scholar] [CrossRef]
- Bayona, C.; Ranđelović, T.; Ochoa, I. Tumor Microenvironment in Glioblastoma: The Central Role of the Hypoxic-Necrotic Core. Cancer Lett. 2026, 639, 218216. [Google Scholar] [CrossRef] [PubMed]
- Donnini, F.; Minniti, G.; Rubino, G.; Battaglia, G.; Pastina, P.; Carfagno, T.; Vannini, M.; Mazzei, M.A.; Tini, P. Glia Between Resistance and Radiotoxicity in Glioblastoma: Mechanisms and Translational Perspectives - A Narrative Review. Neuroglia 2025, 6, 44. [Google Scholar] [CrossRef]
- Gu, D.; Hu, L.; Yang, K.; Yuan, W.; Shan, D.; Gao, J.; Li, J.; Gimple, R.C.; Dixit, D.; Zhu, Z.; et al. Stress-induced pro-inflammatory glioblastoma stem cells secrete TNFAIP6 to enhance tumor growth and induce suppressive macrophages. Dev. Cell 2025, 60, 2558–2575.e6. [Google Scholar] [CrossRef]
- Knudsen, A.M.; Halle, B.; Cédile, O.; Burton, M.; Baun, C.; Thisgaard, H.; Anand, A.; Hubert, C.; Thomassen, M.; Michaelsen, S.R.; et al. Surgical resection of glioblastomas induces pleiotrophin-mediated self-renewal of glioblastoma stem cells in recurrent tumors. Neuro-Oncology 2022, 24, 1074–1087. [Google Scholar] [CrossRef]
- Barthel, F.P.; Johnson, K.C.; Varn, F.S.; Moskalik, A.D.; Tanner, G.; Kocakavuk, E.; Anderson, K.J.; Abiola, O.; Aldape, K.; Alfaro, K.D.; et al. Longitudinal molecular trajectories of diffuse glioma in adults. Nature 2019, 576, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, I.H.; Cho, H.J.; Park, C.K.; Jung, Y.S.; Kim, Y.; Nam, S.H.; Kim, B.S.; Johnson, M.D.; Kong, D.S.; et al. Spatiotemporal Evolution of the Primary Glioblastoma Genome. Cancer Cell 2015, 28, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro-Oncology 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Kalidindi, N.; Or, R.; Babak, S.; Mason, W. Molecular Classification of Diffuse Gliomas. Can. J. Neurol. Sci. 2020, 47, 464–473. [Google Scholar] [CrossRef]
- Buonfiglioli, A.; Hambardzumyan, D. Macrophages and microglia: the cerberus of glioblastoma. Acta Neuropathol. Commun. 2021, 9, 54. [Google Scholar] [CrossRef]
- van der Sanden, B.; Ratel, D.; Berger, F.; Wion, D. Glioma Recurrence Following Surgery: Peritumoral or Perilesional? Front. Neurol. 2016, 7, 52. [Google Scholar] [CrossRef]
- Brooks, L.J.; Parrinello, S. Injury programs shape glioblastoma. Trends Neurosci. 2022, 45, 865–876. [Google Scholar] [CrossRef]
- Singh, S.; Dey, D.; Barik, D.; Mohapatra, I.; Kim, S.; Sharma, M.; Prasad, S.; Wang, P.; Singh, A.; Singh, G. Glioblastoma at the crossroads: current understanding and future therapeutic horizons. Signal Transduct. Target. Ther. 2025, 10, 213. [Google Scholar] [CrossRef]
- Suarez-Meade, P.; Whitehead, R.; Rosenfeld, S.; Schiapparelli, P.; Konstantopoulos, K.; Quinones-Hinojosa, A. Extracellular Matrix Stiffness Conditions Glioblastoma Cells for Long-term Migration: Mechanical Memory as a Driver of Invasion and Recurrence in Glioblastoma. Neuro-Oncology 2025. [Google Scholar] [CrossRef]
- Parker, N.R.; Hudson, A.L.; Khong, P.; Parkinson, J.F.; Dwight, T.; Ikin, R.J.; Zhu, Y.; Cheng, Z.J.; Vafaee, F.; Chen, J.; et al. Intratumoral heterogeneity identified at the epigenetic, genetic and transcriptional level in glioblastoma. Sci. Rep. 2016, 6, 22477. [Google Scholar] [CrossRef]
- Ratliff, M.; Karimian-Jazi, K.; Hoffmann, D.C.; Rauschenbach, L.; Simon, M.; Hai, L.; Mandelbaum, H.; Schubert, M.C.; Kessler, T.; Uhlig, S.; et al. Individual glioblastoma cells harbor both proliferative and invasive capabilities during tumor progression. Neuro-Oncology 2023, 25, 2150–2162. [Google Scholar] [CrossRef] [PubMed]
- Gatenby, R.A.; Brown, J.S. Integrating evolutionary dynamics into cancer therapy. Nat. Rev. Clin. Oncol. 2020, 17, 675–686. [Google Scholar] [CrossRef]
- Blobner, J.; Tonn, J.C. Resection of glioma—Feeding the beast? Neuro-Oncology 2022, 24, 1088–1089. [Google Scholar] [CrossRef]
- Jung, E.; Osswald, M.; Ratliff, M.; et al. Tumor cell plasticity, heterogeneity, and resistance in crucial microenvironmental niches in glioma. Nat. Commun. 2021, 12, 1014. [Google Scholar] [CrossRef]
- Putavet, D.A.; de Keizer, P.L.J. Residual Disease in Glioma Recurrence: A Dangerous Liaison with Senescence. Cancers 2021, 13, 1560. [Google Scholar] [CrossRef] [PubMed]
- Taga, T.; Tabu, K. Glioma progression and recurrence involving maintenance and expansion strategies of glioma stem cells by organizing self-advantageous niche microenvironments. Inflamm. Regen. 2020, 40, 33. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Yan, X.; Hu, S. Glioma stem cells: drivers of tumor progression and recurrence. Stem Cell Res. Ther. 2025, 16, 293. [Google Scholar] [CrossRef]
- Bastola, S.; Pavlyukov, M.S.; Yamashita, D.; et al. Glioma-initiating cells at tumor edge gain signals from tumor core cells to promote their malignancy. Nat. Commun. 2020, 11, 4660. [Google Scholar] [CrossRef]
- Doroszko, M.; Stockgard, R.; Uppman, I.; et al. The invasion phenotypes of glioblastoma depend on plastic and reprogrammable cell states. Nat. Commun. 2025, 16, 6662. [Google Scholar] [CrossRef]
- Neftel, C.; Laffy, J.; Filbin, M.G.; et al. An Integrative Model of Cellular States, Plasticity, and Genetics for Glioblastoma. Cell 2019, 178, 835–849.e21. [Google Scholar] [CrossRef]
- Lacroix, M.; Abi-Said, D.; Fourney, D.R.; et al. A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J. Neurosurg. 2001, 95, 190–198. [Google Scholar] [CrossRef]
- Brenner, S.; Hartzendorf, S.; Vogt, P.; et al. Progression patterns in non-contrast-enhancing gliomas support brain tumor responsiveness to surgical lesions. Pathol. Oncol. Res. 2022, 28, 1610268. [Google Scholar] [CrossRef]
- Gill, B.J.; et al. MRI-localized biopsies reveal subtype-specific differences in molecular and cellular composition at the margins of glioblastoma. Proc. Natl. Acad. Sci. USA 2014, 111, 12550–12555. [Google Scholar] [CrossRef]
- Bhaduri, A.; Di Lullo, E.; Jung, D.; et al. Outer Radial Glia-like Cancer Stem Cells Contribute to Heterogeneity of Glioblastoma. Cell Stem Cell 2020, 26, 48–63.e6. [Google Scholar] [CrossRef] [PubMed]
- Nomura, M.; Spitzer, A.; Johnson, K.C.; et al. The multilayered transcriptional architecture of glioblastoma ecosystems. Nat. Genet. 2025, 57, 1155–1167. [Google Scholar] [CrossRef]
- Suvà, M.L.; Tirosh, I. Single-Cell RNA Sequencing in Cancer: Lessons Learned and Emerging Challenges. Mol. Cell 2019, 75, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Weller, M.; van den Bent, M.; Preusser, M.; et al. EANO guideline on the diagnosis and treatment of diffuse gliomas of adulthood. Nat. Rev. Clin. Oncol. 2021, 18, 170–186. [Google Scholar] [CrossRef] [PubMed]
- Minniti, G.; Amelio, D.; Amichetti, M.; et al. Patterns of failure and comparison of different target volume delineations in patients with glioblastoma treated with conformal radiotherapy plus concomitant and adjuvant temozolomide. Radiother. Oncol. 2010, 97, 377–381. [Google Scholar] [CrossRef]
- Kickingereder, P.; Burth, S.; Wick, A.; et al. Radiomic Profiling of Glioblastoma: Identifying an Imaging Predictor of Patient Survival with Improved Performance over Established Clinical and Radiologic Risk Models. Radiology 2016, 280, 880–889. [Google Scholar] [CrossRef]
- Chiranth, S.; Fougner, V.; Christensen, I.J.; Trip, A.K.; Christiansen, T.; Nørøxe, D.S.; Yde, C.W.; Berthelsen, A.K.; Hasselbalch, B.; Lassen, U.; Poulsen, H.S.; Urup, T. Identifying Targetable Alterations Predictive of Distant Progression in Glioblastoma Patients Undergoing Standard Therapy. Neuro-Oncology Advances 2025, 7, vdaf092. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, B.; Hu, X.; et al. Tumor Evolution of Glioma-Intrinsic Gene Expression Subtypes Associates with Immunological Changes in the Microenvironment. Cancer Cell 2017, 32, 42–56.e6. [Google Scholar] [CrossRef]
- Horsman, M.R.; Overgaard, J. The impact of hypoxia and its modification of the outcome of radiotherapy. J. Radiat. Res. 2016, 57 (Suppl 1), i90–i98. [Google Scholar] [CrossRef]
- Kim, J.Y.; Yoon, M.J.; Park, J.E.; et al. Radiomics in peritumoral non-enhancing regions: fractional anisotropy and cerebral blood volume improve prediction of local progression and overall survival in patients with glioblastoma. Neuroradiology 2019, 61, 1261–1272. [Google Scholar] [CrossRef] [PubMed]
- Pombo Antunes, A.R.; Scheyltjens, I.; Duerinck, J.; Neyns, B.; Movahedi, K. Understanding the glioblastoma immune microenvironment as basis for the development of new immunotherapeutic strategies. eLife 2020, 9, e52176. [Google Scholar] [CrossRef]
- Ravi, V.M.; Will, P.; Kueckelhaus, J.; et al. Spatially resolved multi-omics deciphers bidirectional tumor-host interdependence in glioblastoma. Cancer Cell 2022, 40, 639–655.e13. [Google Scholar] [CrossRef]
- Miller, A.M.; Shah, R.H.; Pentsova, E.I.; et al. Tracking Tumor Evolution in Glioma through Liquid Biopsies of Cerebrospinal Fluid. Nature 2019, 565, 654–658. [Google Scholar] [CrossRef] [PubMed]
- Sabedot, T.S.; Malta, T.M.; Snyder, J.; et al. A serum-based DNA methylation assay provides accurate detection of glioma. Neuro-Oncology 2021, 23, 1494–1508. [Google Scholar] [CrossRef]
- Kickingereder, P.; Bonekamp, D.; Nowosielski, M.; et al. Radiogenomics of Glioblastoma: Machine Learning–based Classification of Molecular Characteristics by Using Multiparametric and Multiregional MR Imaging Features. Radiology 2016, 281, 907–918. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Bai, H.X.; Zhou, H.; et al. Residual Convolutional Neural Network for the Determination of IDH Status in Low- and High-Grade Gliomas from MR Imaging. Clin. Cancer Res. 2018, 24, 1073–1081. [Google Scholar] [CrossRef]
- Tseng, C.-L.; Zeng, K.L.; Mellon, E.A.; et al. Evolving concepts in margin strategies and adaptive radiotherapy for glioblastoma: A new future is on the horizon. Neuro-Oncology 2024, 26 (Suppl 1), S3–S16. [Google Scholar] [CrossRef] [PubMed]
- Prados, M.D.; Byron, S.A.; Tran, N.L.; et al. Toward precision medicine in glioblastoma: the promise and the challenges. Neuro-Oncology 2015, 17, 1051–1063. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).