4. Dissipation of Heat Through a Gravitational System
4.1. Gravitational Dissipative Structures (GDS) Model of the Tropospheric Water Cycle (TWC)
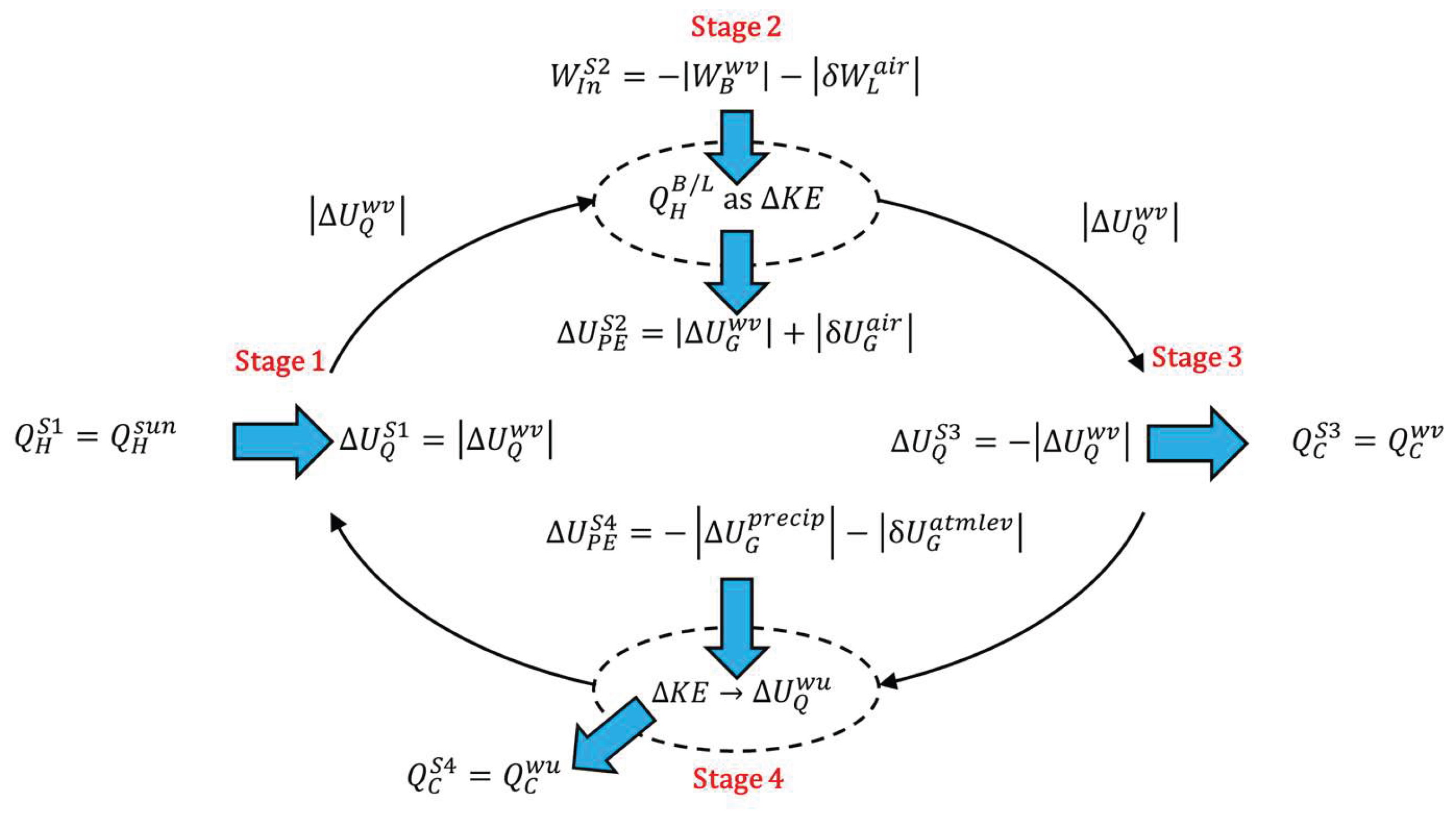
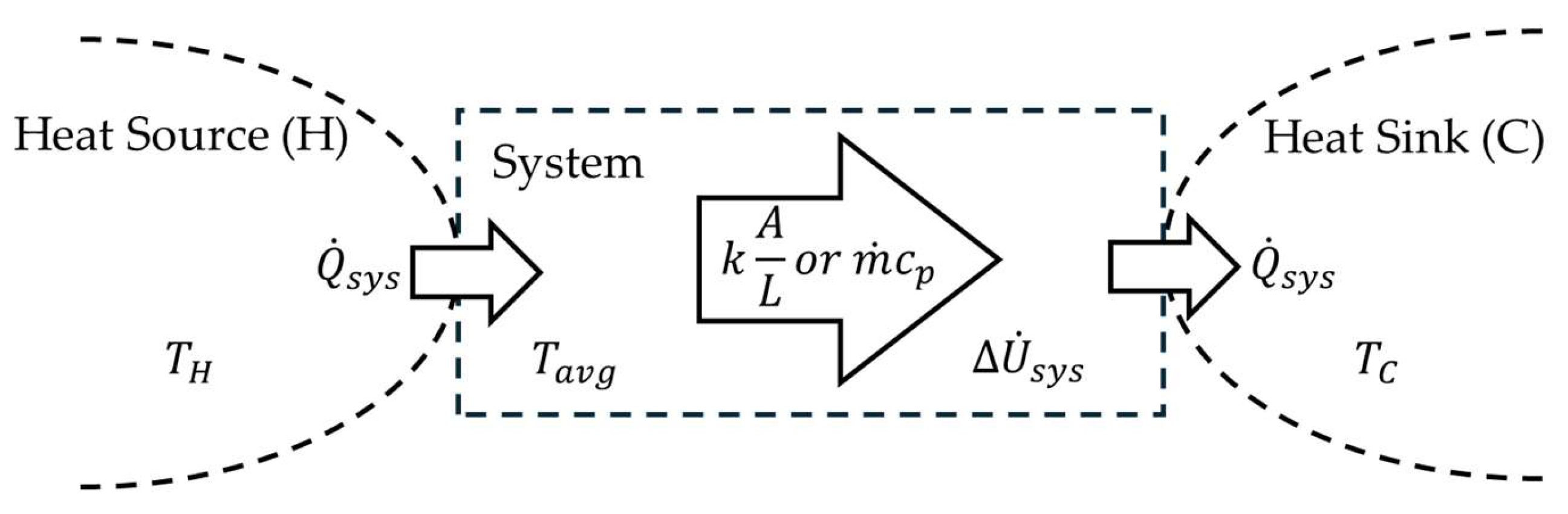
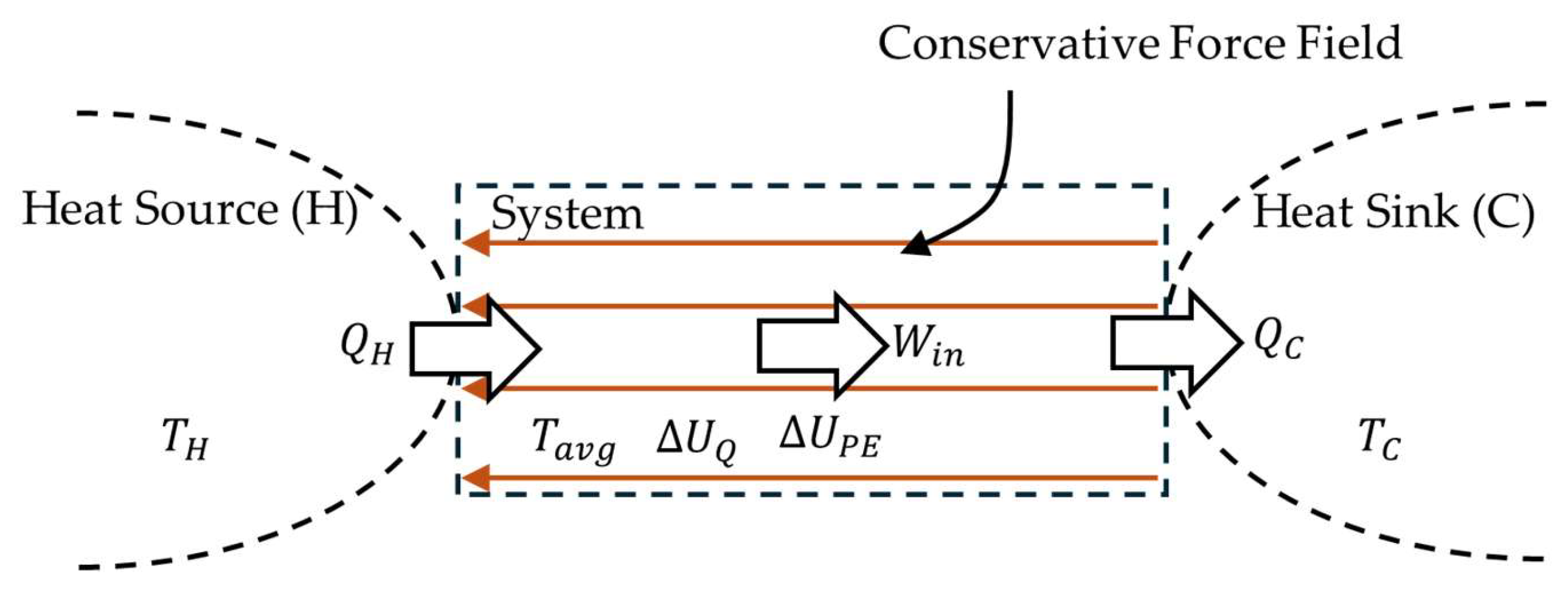
When the force field in
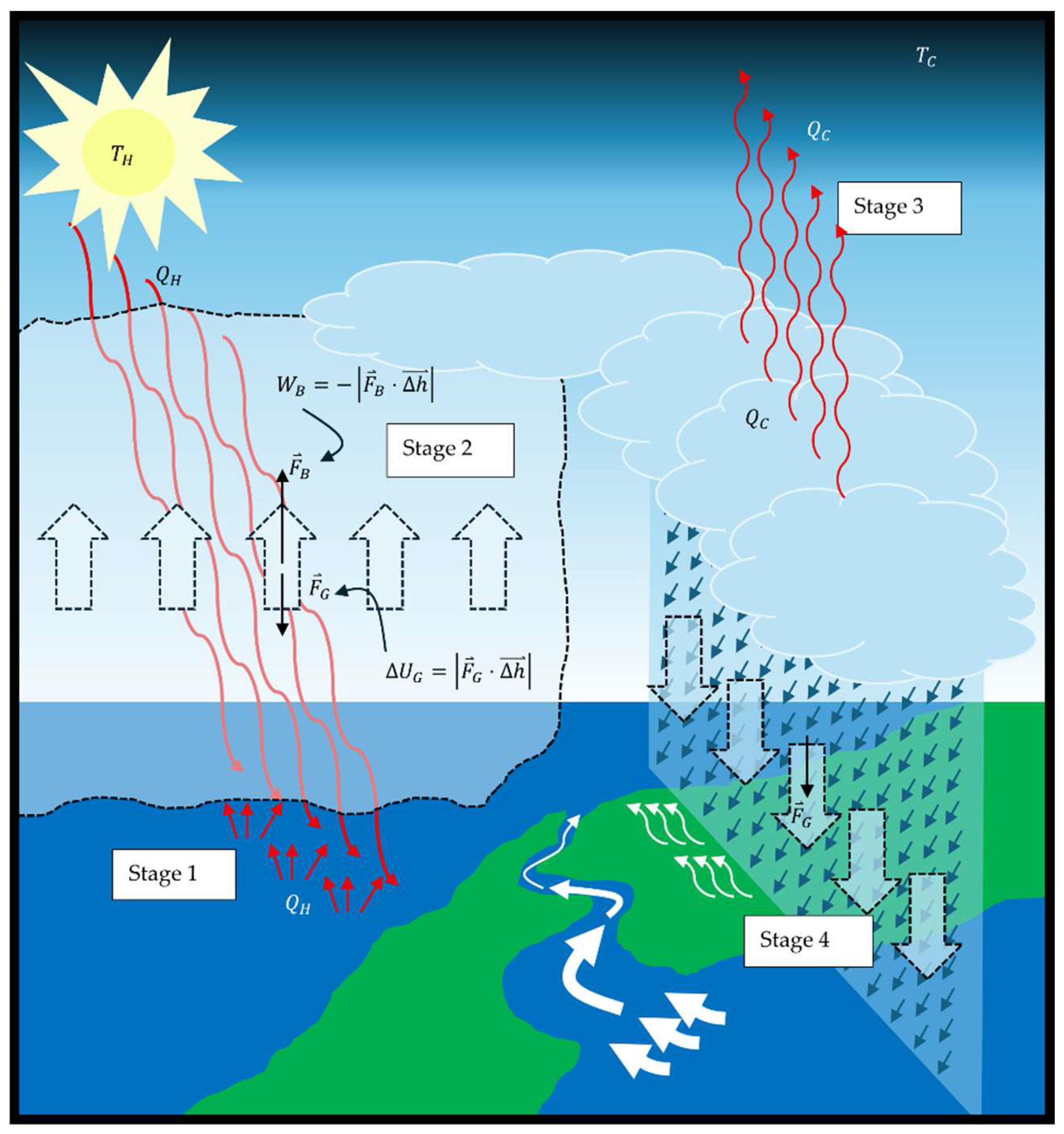
Figure 2 is replaced by gravity, it is a model of how the tropospheric water cycle (TWC) on Earth enables heat to escape the gravity well of Earth. However, the model does not include the entire TWC shown in
Figure 4. The TWC includes the return of the water to ground level. The gravitational dissipative structure (GDS) model of the TWC is analyzed in
Appendix A. Stage 1 (superscript
S1), stage 2 (superscript
S2), stage 3 (superscript
S3), and stage 4 (superscript
S4) of
Figure 4 align with the same stages in
Figure A1. Application of the first law of thermodynamics and the work energy theorem to the model results in the energy balance (also Equation A5),
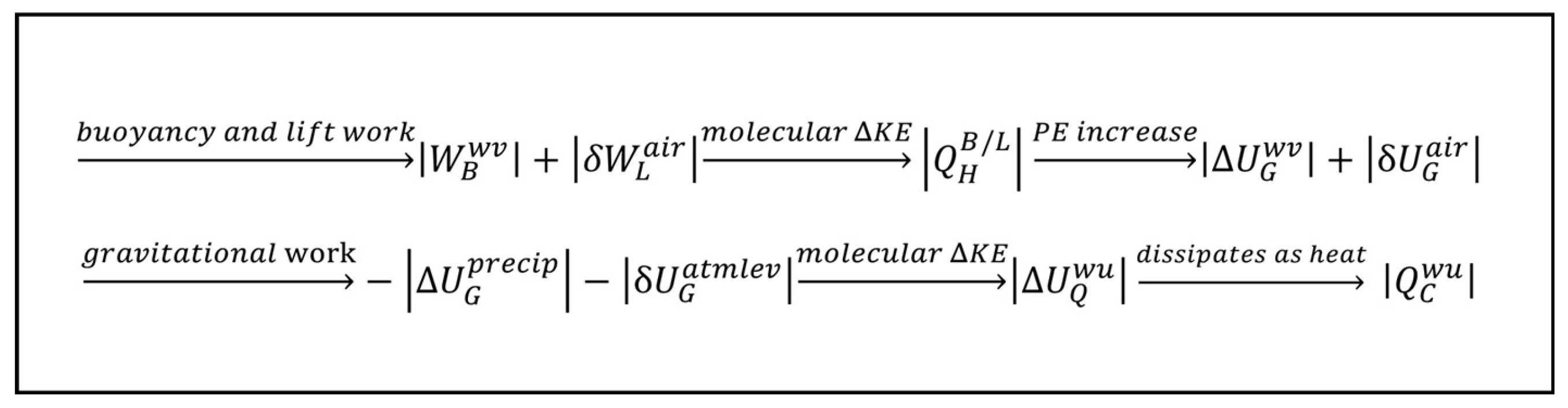
The Sun is a heat source reservoir for Earth, because heat in the form of radiation is emitted by the nuclear fusion of hydrogen into helium in the Sun and travels at the speed of light as photons (electromagnetic packets of energy) to Earth with an energy equivalent to the temperature of the Sun at which it was radiated, A photon from the Sun comes into contact with the electric field of an atom on Earth and is thereby absorbed by the atom. The absorption of the energy of the photon by the atom causes the atom to increase the average speed of its vibration. An influx of such photons against a large mass of material, such as the surface of a lake, results in the temperature of the mass of material rising as its internal energy increases based on its heat capacity. This is stage 1 of the heat engine model of the TWC: heat input. The heat from the Sun, , enters water on Earth’s surface at stage 1 of the heat engine, increasing the internal energy of some of the water by an amount required to raise it to the temperature of vaporization and then transition its phase to water vapor (superscript wv), .
The warm water vapor that is formed is less dense than the cooler air around it and above it. At stage 2, this difference in density creates a buoyant force upward on the water vapor in opposition to the gravitational force on the water vapor. The expansion of the water vapor also lifts the atmosphere above it by the displacement of the cooler air, just as bath water rises when you sit in it. The buoyancy and lift forces (subscripts
B and
L) together result in work input being done on the atmospheric column made up of water vapor and air (subscript
air) (from Equation A8),
The buoyancy work pushes evaporated water skyward with the internal energy it holds in its molecular-kinetic molecules,
The work of the buoyancy and lift forces accelerates the molecules in the water vapor and air column, increasing the molecular kinetic energy of the water and air molecules, resulting in convective heat,
The convective heat of the acceleration is simultaneously converted into the potential energy of gravity (subscript
G) in an equal amount to the work of the buoyancy and lift (from Equation A13 and Equation A16),
As it is lifted skyward, the water vapor continues to take in more heat from the solar radiation, even as it dissipates some heat to cooler air molecules around it. This maintains the water vapor at the lower density needed to produce buoyancy work that is converted into gravitational potential energy as it continues to rise. At various altitudes, dependent upon environmental conditions of the atmosphere, stage 3 occurs with the heat dissipation from the water vapor becoming greater than the heat transfer into the water vapor from solar radiation. Water vapor releases the latent heat of vaporization,
, condensing and releasing the heat that ultimately dissipates to space,
.
4
During stage 4, condensed water falls from the sky by the force of gravity. The total volume of water vapor decreases by the loss of vapor to condensation. This results in the lifted air falling as the total vapor bubble reduces in size. This is a gravitational leveling/rounding effect of the troposphere by the force of gravity (
Figure 5), described by Irons and Irons [
5] (Presentation 1). The energy balance for stage 4 just involves gravitational potential energy and heat (also Equation A23),
The decrease of the potential energy of the rain as it precipitates (superscript
precip) and the lifted air as it atmospherically levels (superscript
atmlev) at stage 4 is equal and opposite to the increase in potential energy of the buoyant water vapor and lifted air at stage 2,
The potential energy is converted to the kinetic energy of bulk water, molecular water, and air, a continuation of the convective cycle started by the rising water vapor and air column in stage 2. The water hits the ground and sinks into the ground or flows over the surface to collect back into streams, rivers, ponds, lakes, and oceans from which the water evaporated, with all kinetic energy gained from the gravitational potential energy being converted into the internal energy of the water and air warming up (superscript
wu),
both from friction during the precipitation and leveling and from the impact of water hitting the ground, at which point its remaining kinetic energy is converted to internal energy The increase in internal energy then dissipates into the environment as heat. The energy balance gives the result that the heat dissipated into the environment at stage 4 due to the warmup is equal to the original increase in potential energy of the water vapor and air during stage 2,
4.2. Characteristics of DSs
Three interesting characteristics of a DS are revealed by this model. The first characteristic is that the restoration of internal energy from stored energy that does not deplete another energy source or reduce the quality of the restored energy makes stage 4 of the GDS act as an ideal pump, with conservative forces performing all work in the thermodynamic cycle. This means that the thermodynamic cycle of the DS is semi-reversible [
45].
Sands [
46] demonstrates by computational analysis that the canonical theory of Clausius that states that a Carnot cycle requires quasi-static conditions to achieve reversibility is incorrect and that a Carnot cycle is theoretically possible for any heat engine quasi-statically near to or dynamically far from thermodynamic equilibrium. It does not require a violation of the second law of thermodynamics, considering the universal entropy associated with such a dynamic Carnot cycle is zero, whether it is run forward or run in reverse. As demonstrated by Sands, a Carnot cycle still requires a heat engine that uses an ideal gas (no internal dissipation) and that has no heat losses. The Sands dynamic Carnot cycle must also strictly control when each stage of the heat engine ends and the next stage begins. It is notable in the literature that such controls theoretically and empirically (thus far) generate their own irreversible universal entropy by the process of deleting their information [
47]. This suggests that the Sands dynamic Carnot cycle does have an information loss universal entropy.
The DS model presented here in heat transformation theory highlights a way by which the Sands unstated assumption of a zero-entropy control system can be achieved. The heat to work (stage 2) must physically capture the heat and hold it with no losses and the work to heat (stage 4) must return that exact same amount of heat with no change in quantity or quality (i.e., no temperature drop). For the GDS presented in
Section 4.1 and
Appendix A, stage 4 is a reversal of stage 2 with no universal entropy generation resulting from the action of the conservative force field at each stage. No loss in quantity and no loss of quality (heat is released without the need for a differential temperature to cause the release) means that specific timing of stage 2 and stage 4 of a GDS is not required, therefore the cycle need not follow a Carnot cycle and no control system is needed. True, the energy with no loss of quantity, quality, or information that is added back to the heat engine at stage 4 is eventually exhausted to the universal heat sink, but that is after the potential energy is converted back into internal energy of the same quantity and quality as it had when the potential energy was generated. The force acting at stage 2 and stage 4 is completely conservative and is independent of the boundary of the open system where internal energy is dissipated as heat to the heat sink as governed by the second law of thermodynamics. The TWC GDS does exactly this. This is a characteristic of all DSs. This is what makes them semi-reversible. Stages 1 and 3 of the DS cannot be reversible, because they are not controlled by conservative forces and they do not follow a Carnot cycle.
In contrast, stages 2 and 4 of an engineered forced-convection heat engine are performed by non-conservative forces in constrained but non-isolated energy conversion systems. Heat dissipation and loss still occurs through scattering of work output at stage 2 into degrees of freedom of heat dissipation that do not perform work and through imperfectly insulated barriers as a result of dampening. If the work output of stage 2 of an engineered forced-convection heat engine were to be utilized to perform the work input at stage 4 to pump the heat transfer medium back to stage 1, the work input power available at stage 4 would be less than the work output power at stage 2 due to such heat dissipation and loss at both the stage 2 work output and stage 4 work input machinery, both losing power and generating universal entropy.
When considering the TWC GDS and the portion of heat input from the Sun that is non-reflective (i.e., only considering heat that must convectively escape Earth’s gravity well), stage 2 and stage 4 of the GDSs occurring in the atmosphere have no such constrained, non-isolated energy conversion systems because the atmosphere acts as a whole, effectively isolating it from heat losses through any surface other than the stage 3 surface at the top of the troposphere. The buoyancy effect caused by density differences works because of pressure differences caused by the depth of a fluid in a gravitational field. The pressure differences drive motion by particle interactions of non-ideal fluids that can result in dissipation of the upward momentum of water molecules into transverse degrees of freedom, resulting in dissipation of the heat. However, greatly isolatable, single-dimensional motion of the water vapor results from barrier channeling. The vaporous ejections of high temperature water molecules from liquid water that occurs during evaporation are channeled in a common direction of the pressure gradient by the more compact and almost twice-as-massive air molecules made up mostly of diatomic oxygen and diatomic nitrogen that are also moving in the direction of the pressure gradient caused by a tropospheric air cycle (TAC) driven by the solar-radiative heating of the air. The compact, massive, moving air molecules act like walls against the spread out, less-massive water molecules, resulting in the water molecules giving up very little of their momentum to dispersion to degrees of freedom that are perpendicular to the pressure gradient. Thus, the water molecules mostly maintain their momentum along the pressure gradient that generally progresses vertically even as horizontal flow of air occurs barometrically. This upward movement of water vapor can be physically experienced by the bump of an airplane flying over a river on a hot day.
As a result of the dense air around the sparse water, superheating of the water vapor by solar radiation tends to be dissipated into the air, and the molecules of water that manage to continue to rise gradually vertically along the pressure gradient have sufficient internal energy to maintain their latent heat of vaporization. The dissipation of water vapor superheating being captured in the air of the atmosphere effectively still requires the dispersed heat captured by air molecules to transfer along the pressure gradient to the top of the troposphere. For the proportion of solar heating that is absorbed by water and air, the only way to escape efficiently from Earth’s gravity well is by convection of both water and air to the top of the troposphere where it is output at stage 3. Earth’s gravity well has no heat losses of absorbed solar radiation that depletes the natural convection of combined water vapor and air because it is a sphere with a radial conservative force field. Thus, though the combination of the TWC and TAC do not meet the theoretical requirements of a zero-entropy cycle at stage 1 and stage 3, they do at stage 2 and stage 4. Thus, the postulate of the DS being a semi-reversible heat engine cycle as proposed by Irons and Irons is supported theoretically.
The second interesting characteristic of a DS revealed by the TWC GDS is that the conversion of buoyancy to heat and then to potential energy,
Figure 6, without depleting an energy source or reducing the ongoing quality of action of the gravitational field reveals that buoyancy is an extension of the conservative nature of the gravitational force field. Buoyancy is a force caused by a permanent differential pressure that does not dissipate, making it conservative. This conservative nature of the buoyant force is a result of being coupled to the force of gravity by the variation in depth pressure of matter in the presence of gravity. Unlike a temperature difference that results in a pressure gradient to drive heat convection until the temperature and pressure equalize as a result of heat dissipation, depth pressure never equalizes, due to the constant presence of gravity. Effectively, the gravidynamic energy of buoyancy and lift are added to the environment as new heat energy without depleting gravity or buoyancy. The moment the fallen water is evaporated again, the buoyant force returns, fully conserved.
5
The third interesting characteristic of a DS is that it operates as an open system on a closed state cycle. This means that the properties of matter making up the DS return to the same values the matter has at the start of stage 1 when the matter reaches the end of stage 4. The net change in temperature, net change in pressure, net change in volume, net change in internal energy, net change in stored potential energy, and net change in non-equilibrium entropy of matter cycling back to the beginning of stage 1 are zero. Matter of the DS that is in process at any point between the beginning of stage 1 and the end of stage 4 are at non-zero net change values. The overall result is that the DS achieves average values of positive net changes in internal energy, stored potential energy, and non-equilibrium entropy when the DS achieves far-from-equilibrium steady state.
At steady state, the heat transfer rate out of the DS equals the heat transfer rate into the DS. It is at this steady state level where the DS achieves maximum universal entropy production equivalent to simple heat transfer. This is a principle of operation of DSs known as the
maximum entropy production principle [
48,
49,
50,
51,
52]. The model of the GDS demonstrates that this principle is a result of stage 4 decay occurring at the same rate as stage 2 growth upon reaching steady state, thus resulting in total heat output equaling total heat input. However, on the growth path to steady state, the GDS must output less heat than it takes in to achieve growth of stored potential energy. It is noteworthy that this means that the average net change in non-equilibrium entropy of the DS at steady state is positive and the net change in non-equilibrium entropy of the local heat source and heat sink appears to be negative (see
Section 5.1). To understand the function of a GDS further, it is necessary for heat transformation theory to define mathematical equations for state properties. These equations assist in analyzing the functions of a GDS.
4.3. Auto-Powering Capacity and Auto-Restoring Order of a DS
The DS has six thermodynamic properties defined by heat transformation theory. Four of these properties are proposed at a high level in Irons and Irons [
45] (pp. 2-6) as self-restoring order, capacity, entropy, and exergy. The following presents a refinement of terminologies, definitions, and mathematical developments of these properties.
The first two properties refined by heat transformation theory are associated with the extensive macroscopic properties of process of the DS.
Auto-powering capacity (APC), referred to as capacity in [
45], is defined as the flow within the DS that is generated by heat transfer per unit area of a DS.
Auto-restoring order (ARO), referred to as self-restoring order in [
45], is the flow within the DS that is generated by the release of the potential energy gained by work input at stage 2 and released at stage 4 to return the DS to its starting conditions. Flow is a network ecology term that can be expressed many ways, such as in properties of mass, energy, or population (number of “individuals”) and in quantities of sums, rates, or fluxes [
53] (pp. 213-260). Flow (
is usually associated with a transfer between two nodes (subscripts
and
) of a network. A DS is the node of a network that receives
flows from
local heat sources and outputs stage 3
flows to
local heat sinks. The APC and ARO are flows within the node that sum, re-divide, and route the
heat inputs to
stage 3 heat outputs. Ecosystem networks on Earth today are made up of both physical (inanimate) and biological (animate) DS nodes. For a pre-biotic Earth, such a network is only comprised of physical components. Developing heat transformation theory in terms of network ecology gives advantages of leveraging the science of ecological thermodynamics to explain the growth phenomena covered by the theory and suits the context of pre-biotic evolution of inanimate material.
Equations for APC and ARO can be mathematically modeled by starting with the heat engine model of the DS in question. Specific DSs have unique heat engine models, the model developed in
Appendix A for the TWC being an example. Using the heat engine model, thermodynamic equations specific to the model are developed for APC and ARO based upon heat inputs and work inputs seen in nature for the given DS.
6 The equations and empirical values are used to calculate values.
4.3.1. The Tropospheric Water Cycle (TWC)
For the TWC model of
Appendix A, APC is driven by the heat input of stages 1 through 3 (also Equation A17),
It is desired to put APC in terms of power fluxes, i.e., energy flow per unit time per unit area, considering these are common units used in the study of network ecology. Using Equation 20, APC (superscript
APC) for the TWC (subscript
twc) takes the form,
Calculation S1 in the
Supplementary Materials provides the analysis, empirical research on Earth’s energy budget [
54,
55,
56], and calculations for these values. The result from Equation S32 in the
Supplementary Materials calculates the APC of the TWC of Earth to be,
For the TWC model of
Appendix A, ARO is driven by the heat output of stage 4, Equation A32. Putting ARO (superscript
ARO) in terms of power fluxes takes the form,
Research on Earth’s energy budget [
54,
55,
56] indicates the Earth receives an average flow of radiative-heat-transfer-rate flux from the node of the Sun (subscript
sun) to the node of the DS of the TWC of
. The result is an equivalent average convective-internal-energy-transfer-rate flux of rising water vapor through the atmosphere. This results in an average of
of kinetic-mass power flux due to the work input of buoyancy and lift that goes into gravitational potential energy, which kinetic-mass power flux is associated with the average convective-heat power flux of evaporated water.
4.3.2. The Tropospheric Air Cycle (TAC)
Any heat dissipated away from causing superheating can also be utilized by other DSs in the environment, such as what happens when the superheating of water vapor is dissipated into the surrounding air. This dissipation of superheating away from water vapor becomes part of the
of average convective-heat power flux due to solar heating of the air in the atmosphere [
54,
55,
56].
The TAC is a result of the interaction of two collections of air molecules, one being a collection of warmer, low-altitude air and the other being cooler, high-altitude air. The interaction results in an upward natural convection due to the pressure difference. This convective gradient is a result of a differential pressure between the hotter, higher-pressure end of the system where the heat is input to internal energy of the air and the cooler end of the system at the top of the atmosphere where the pressure is lower and heat output occurs. The heat is carried by the mass convection of warmed air into the cooler, higher altitudes, carrying an internal energy of the air based on its specific heat capacity.
The work of natural convection that drives the air is driven by gravity and therefore conservative, and the resulting TAC is semi-reversible in the same way as explained for the TWC. Calculation S2 in the
Supplementary Materials provides analysis, empirical research on Earth’s energy budget [
54,
55,
56], and calculations for these values. The reader is left to prepare their own TAC GDS heat engine model similar to that of the TWC in
Appendix A. The result of Equation S51 in the
Supplementary Materials calculates the APC for the TAC (subscript
tac) of Earth to be,
with the ARO of the TAC (Equation S53 in the
Supplementary Materials) only including a gravitational-potential-energy-release-rate flux equal to the storage-rate flux,
All power fluxes used in APC and ARO are proportional to the kinematic-mass-flow-rate flux of the natural convection (subscript
NC) of air,
(Calculation S2, Equation S43 in the
Supplementary Materials), considering it only involves specific heat capacity with no phase change. The result is
of average kinetic-mass power flux that is driven by natural convection into a gravitational-potential-energy-storage-rate flux of air displacement that is almost triple the associated
of average convective-internal-energy transfer rate flux of heat-capacity-carrying air.
The specific heat capacity of air results in much less internal energy carrying capacity than that of water vapor with its latent heat of vaporization. Air is also denser than water vapor. These two factors result in the kinetic-mass power flux of air needing to be greater than that of water vapor by two orders of magnitude. This high kinetic-mass power flux of air compared to water vapor is what mitigates the superheating of water vapor with the two DSs working together to mix the atmosphere.
4.3.3. Kinematic-Mass-Flow-Rate Flux Proportionality and the Combined Effect of TWC and TAC
The example of the TWC and TAC reveal an interesting aspect of GDSs. The kinetic-mass-flow-rate flux of the heat-carrying medium of a DS is proportional to both the APC and the ARO. For the TWC of Calculation S1 in the
Supplementary Materials, the contribution of the kinematic-mass-power flux of air to APC and ARO is two orders of magnitude smaller than the total APC and one order of magnitude smaller than the total ARO. The lesser contribution of kinematic-mass-power flux of air to APC and ARO allows an approximation of proportionality. Therefore, all the power fluxes used in APC and ARO are treated as proportional to the kinematic-mass-flow-rate flux of water vapor as shown in Calculation S1 in the
Supplementary Materials. The example of the TWC reveals that DSs in which the heat-carrying medium undergoes a phase transition require an estimation of proportionality. However, the TAC analysis in Calculation S2 in the
Supplementary Materials shows that the APC and ARO are exactly proportional to the kinetic-mass-flow-rate flux, thus needing no approximation. GDSs that do not undergo a phase transition have this proportionality as an exact property.
For this approximation to be acceptable for the TWC in which the heat-carrying medium undergoes a phase transition, an assumption built into the calculations of Calculation S1 in the
Supplementary Materials is that there is no superheating of the water medium of the DS (i.e., raising the temperature of the water above the vaporization temperature at atmospheric pressure). All the heat input from the heat source goes into the latent heat of vaporization of water with the temperature of the water vapor remaining at
, the temperature of phase transition of liquid water into gaseous water at standard temperature and pressure.
This assumption requires questioning, considering kinematic-mass-flow-rate flux is governed not only by the quantity of mass involved but also by the velocity of flow. Superheating allows for the carrying of more heat by the same amount of mass. However, superheating would also result in an increase in velocity of flow due to a decrease in the density of the water vapor and an increase in the buoyancy force. These are competing changes that make it difficult to determine what is exactly happening in the environment. To address this unknown in the analysis, the heat transformation theory defines superheating as the carrying of a heat load above and beyond the mechanism of the DS to transform the extra heat load to potential energy. Considering the purpose of the heat transformation theory is to understand the mechanism of the DS that transforms heat into potential energy, the element of superheating is not a function of the DS and, therefore, is assumed to not be included in the transfer of heat through the DS. This is a valid assumption in that the open-system nature of DSs, even those without phase transitions, results in any superheating above and beyond what the DS can use for energy storage dissipating out of the DS and into the surrounding environment to either be used by another DS or to engage more mass in the given DS.
Based on this, the heat transformation theory excludes superheating from the APC calculations of the DS considering we are defining it as a capacity. In other words, it is a maximum engagement of available mass in the environment assuming no superheating. As a result, the kinematic-mass-flow-rate flux of a DS adjusts proportionally to changes in convective-internal-energy-transfer-rate flux by engaging a greater amount of mass from the environment in the DS or by passing the heat to other surrounding DSs for their use, both of which eliminate superheating from the function of the DS, making these fluxes proportional.
For the TWC and TAC that operate in the same space, any potential superheating that water vapor might tend toward as a result of solar radiation continuing to heat the water vapor as it rises is transferred to the surrounding air and becomes part of the contribution of
of solar radiation to heating the air. The APC flows of the heat engines that drive the TWC and the TAC power the cycles from stage 1 to stage 3 where the portion of the flow associated with the direct solar heating is dissipated to the tropopause (subscript
tropau),
and eventually to the stratosphere and upper layers of atmosphere, and to space. The ARO flows in the troposphere (subscript
trosph),
release the potential energies and restore the internal energies of the DSs at stage 4, returning the DS to the state properties it has at the start of stage 1, thus maintaining Earth in average thermal steady state. The heat associated with ARO becomes part of the background environmental internal energy that supports average Earth temperatures. Combined, the APC and ARO of the TWC and TAC in the troposphere are substantially larger than the input flow from the radiative heat rate flux of the Sun to the troposphere. This makes up part of the total power flux that drives Earth weather.
7
4.4. Specific Universal Entropies and Complexity Yield of a GDS
The last four properties of a DS as refined by heat transformation theory are intensive and associated with the macroscopic properties of state of the DS. Intensive properties are being used to reveal how the differences in matter and states of matter affect the dissipation of heat through a DS. The first is specific universal entropy (), defined as the amount of universal entropy per unit mass of a DS that is generated by the DS. This could also be called the change in universal entropy per unit mass. Considering it is a specific quantity, meaning “per unit mass,” it is already a differential. Calling it change of specific universal entropy would be redundant.
This causal property of the DS system is defined by the effect of the DS on universal entropy, and not by the non-equilibrium entropy of the DS itself. This follows heat engine theory in which the entropy of the heat engine is calculated based upon the output of heat from the heat source and the input of heat to the heat sink. However, unlike heat engine theory that uses local heat source and local heat sink temperatures, equilibrium heat source and equilibrium heat sink temperatures of the universe are used. By defining this property, the heat transformation theory uses the state property of the entropy of the universe in thermal equilibrium to define a process property of a far-from-equilibrium system. This avoids using the non-equilibrium entropy of the DS itself (i.e., taking the difference of the increase in non-equilibrium entropy of the DS of the input of heat from the local heat source and the decrease in non-equilibrium entropy of the DS of the output of heat to the local heat sink). Such non-equilibrium entropy cannot be compared to universal entropy, as discussed in
Section 2. Referring to it loosely as “the entropy of the DS” can suggest it is the specific entropy of the heat-carrying medium of the DS that is being calculated. This is an incorrect interpretation. Non-equilibirum entropy of the system of a DS is discussed in
Section 4.2.There are three quantities of specific universal entropy that assist in the analysis of the DS.
The first quantity of specific universal entropy to discuss is
specific maximum universal entropy (
), defined as the maximum (subscript
max) change of universal entropy per unit mass of the medium (subscript
med) of the DS (superscript
DS) involved in the heat transfer. It is the result of a total heat output from the universal heat source (subscript
uni-H) that goes through a DS and is subsequently input to the universal heat sink (subscript
uni-C),
As discussed in
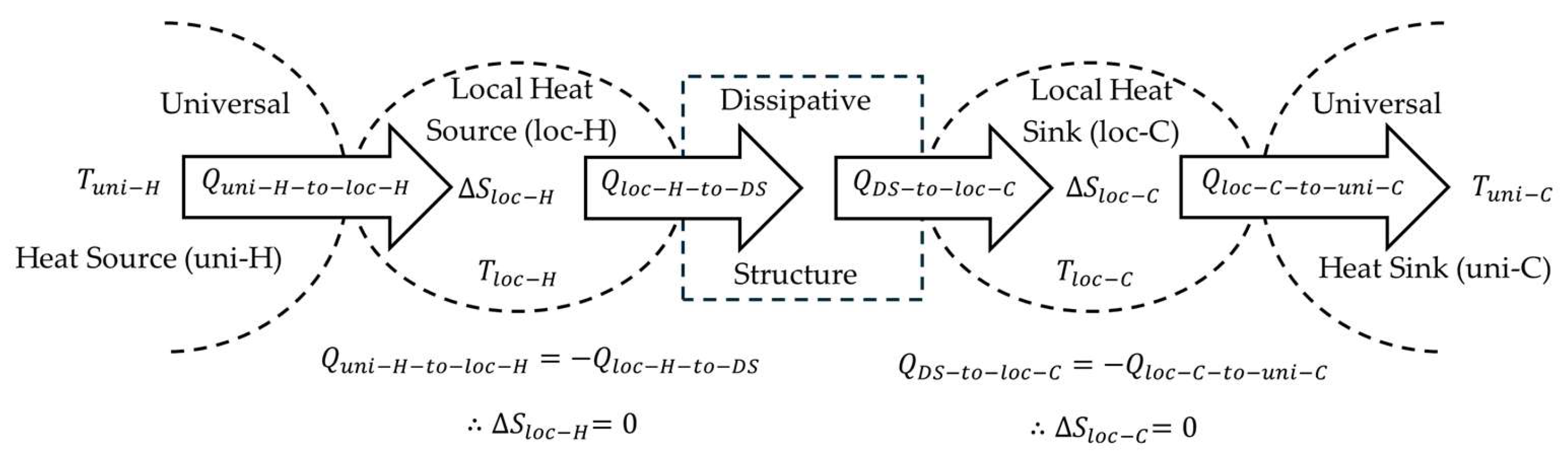
Section 4.3, a DS can be a node in a network. The quantity of heat that is passed to the DS from its local heat source originated from the Sun. It is heat that is output from a universal equilibrium heat source and input to the GDS that is used to calculate universal entropy. Subscripting it as a local heat source (subscript
loc-H) even though it originates from a universal heat source ensures inputs and outputs are properly tracked when the DS is analyzed in the context of a network. Notice, however, that the universal heat source temperature and universal heat sink temperature are used. These temperatures are used because the property is the amount of specific entropy that the DS is affecting on the universe.
In any given timeframe of operation of a DS that has achieved steady state, the same quantity of heat that is input is also output, such that the total heat input is equal to the total heat output. The equation could use either total heat input or total heat output, considering they are the same magnitude. For convention, the total heat input is used. To obtain the specific maximum universal entropy, the equation divides by the total mass of the heat-carrying medium of the DS (). The property is in units of kilojoules per kilogram-kelvin.
The objective of the specific maximum universal entropy is to include the total change of universal entropy per unit mass of the DSs that results from the complete dissipation to the universal heat sink of the heat from the heat source that passes through the DS. The maximum universal entropy is exactly equal to that generated for simple heat dissipation from the heat source reservoir to the heat sink reservoir without passing through a DS, mentioned previously as the maximum entropy production principle [
48,
49], from which the heat transformation theory derives the name specific maximum universal entropy.
For the specific maximum universal entropy of the TWC,
is the total sum of the heat inputs from stages 1 and 2 that originate from the universal heat source and that are passed through the network and input to the GDS from the local heat source that feeds it. Even though the heat input from the buoyancy and lift does not originate from the Sun, the mass that makes up Earth to produce the gravity with a corresponding buoyancy and lift does originate from stars that preceded the Sun, the elements making up Earth being the result of the high temperature fusion and supernova of stars operating at similar temperatures as the Sun. Therefore, the same universal heat source temperature is used for the heat input from buoyancy and lift. See
Section 5.2 for further discussion on this topic.
To calculate specific maximum universal entropy for the TWC DS, it is convenient to put Equation 30 in terms of the empirical and calculated values of Calculation S1 in the
Supplementary Materials,
As discussed in
Section 4.3.3 and in Calculation S1 in the
Supplementary Materials, the APC and kinematic-mass-flow-rate flux of water vapor are approximately proportional as calculated empirically and based on the modeled assumption of no superheating of water vapor. The ratio is approximately the same as the ratio of Equation 30 of the total heat input of a fixed period of time and the mass of the heat transfer medium used in that time period. Note that the
term is the temperature of the sun where fusion occurs,
the universal heat source of radiative heat for the TWC, and the
term is the temperature of space as indicated by the cosmic microwave background radiation (CMBR), 2.7 K. The specific maximum universal entropy for the TAC DS similarly follows using the empirical and calculated values of Calculation S2 in the
Supplementary Materials,
Total changes in maximum universal entropy over a given time-period are determined by the total amount of mass engaged in the medium of the DS in that time period. Considering the TAC has a kinematic-mass-flow-rate flux that is two orders of magnitude greater than that of the water vapor and air captured in the TWC, the reader can satisfy themselves that the maximum universal entropy generation rate of the TAC is the same order of magnitude as the TWC, even though the specific maximum universal entropy of the TAC is two orders of magnitude lesser than that of the TWC.
The specific maximum universal entropy is governed by the temperature of the heat source and heat sink, not by the strength or type of conservative force field and not by the quantity of mass of the DS. A separate intensive property is needed to quantify the conservative force field element of a DS. The fourth new property of the theory is used to do this and differentiate the transformation of heat.
Complexity yield,
related to
exergy in [
45], is a quantization of the ability of the DS to generate
complexity (subscript
Cx), i.e., conservatively store and release energy per mole of the heat carrying medium as a result of heat transfer through the DS,
This equation is a general form that can be applied to any DS with
being the energy that is stored during the endothermic stage of a DS (always stage 2 for GDSs) and
is the number of moles of heat carrying medium used in the storage of that energy. The reason for dividing by the number of moles rather than by mass is because Clausius’s temperature-based entropy and Boltzmann’s statistical-microstate-based entropy [
57] are both based on particle dynamics and particle quantity, both based on molecular constitution. Temperature is a macroscopic intensive property that is based on kinetic-molecular energy according to Maxwell [
35]. Boltzmann statistical entropy is based on the number of accessible microstates that are determined by molecular aggregation and granularity, both properties of the numbers and varieties of molecules. This is discussed further in part II of this paper series [
58].
Complexity yield is a measure of the DS’s ability to produce a change in stored energy. The complexity yield is a result of the energy that is stored or released in the interaction between the matter that is the heat transfer medium of the DS and the conservative force field of the DS as a result of a property of the matter that couples the interaction (e.g., the mass property of matter couples with the gravitational force). A DS transforms heat into stored energy. Stored energy,
can include a change in what is traditionally called internal energy in the context of chemistry that is held by the matter of the medium in addition to a change in what is traditionally called potential energy in the context of gravity. Internal energy is considered in
Section 5. The work input of a DS is a result of universal heat source input to the DS. Therefore, conservation of energy using the first law of thermodynamics combined with the work-energy theorem requires that the change in stored energy is equal to the total heat input at stages 1 and 2 minus the heat output at stage 3 at steady state.
For the complexity yield of the TWC,
is the total sum of the solar (superscript sun) and buoyancy and life (superscript B/L) heat inputs from stages 1 and 2 that originate from the universal heat source and that are passed through the network and input to the GDS from the local heat source that feeds it and is the heat output at stage 3.
To calculate complexity yield for the TWC, it is again convenient to put Equation 34 in terms of the empirical and calculated values of Calculation S1 in the
Supplementary Materials,
As discussed above and in Calculation S1 in the
Supplementary Materials, the total gravitational-potential-energy-storage-rate flux and kinematic-mass-flow-rate flux of water vapor are approximately proportional as calculated empirically and based on the assumption of no superheating of water vapor. The ratio is approximately the same as the ratio of Equation 34 of the total heat input of a fixed period of time and the number of moles of the heat transfer medium used in that time period. This makes complexity yield a constant property of the DS. Thus, the differences in the heat source and heat sink that affect specific maximum universal entropy do not affect how the DS converts heat to potential energy at a constant per-unit-of-matter ratio. Complexity yield is not defined by the heat source and heat sink of the DS. Complexity yield is an intensive state property of the DS that is a result of the properties of matter and matter’s interaction with the conservative force field of the DS. However, the actual potential energy storage rate is extensive and is a portion of the APC. The complexity yield for the TAC DS similarly follows using the empirical and calculated values of Calculation S2 in the
Supplementary Materials,
Both TWC and TAC calculations of complexity yield multiply by the molar mass of the heat transfer medium to convert the denominator to moles per unit time per unit area.
This leads to a second form of specific universal entropy. The
specific conditional universal entropy (
) is defined as the change in universal entropy per unit mass of the heat transfer medium of the DS, which change is associated with direct pass-through heat that is not transformed to potential energy,
It is a general form that can be applied to any DS in a network. As with specific maximum universal entropy, specific conditional universal entropy is determined by local heat source inputs and local heat sink outputs that are ultimately from and to the universal heat source and universal heat sink and by the universal heat source and heat sink temperatures. The term conditional entropy is specifically used to relate to the conditional entropy of information theory, which relationship is discussed in Section 6. Heat transformation theory defines specific conditional universal entropy specifically to address the heat leaving the system at stage 3. The specific conditional universal entropy is a quantity that applies in exactly the same way to forced convection heat engines as it does to DSs. The specific conditional universal entropy results from the exhausting of the remaining heat from the heat source that is carried as internal energy in the heat engine medium following the work performance of the heat engine.
For the specific conditional universal entropy of the TWC,
the values of Calculation S1 in the
Supplementary Materials are applied using similar ratios of power flux to mass flow rate flux as used for specific maximum universal entropy,
The specific conditional universal entropy of the TAC is calculated using values in Calculation S2 in the
Supplementary Materials,
For these cycles, even though the heat of warmup as condensed water falls back to Earth and cooled air levels eventually ends up in space along with the heat from the Sun, only the heat carried by the medium from the Sun is considered in conditional entropy. This is because the warmup heat comes from the ARO of the heat engine, which ARO is a result of decay of the stored potential energy at stage 4. That warmup heat is not generated at the same time and place in stage 4 as the heat exhausted at stage 3. This difference in time and place of heat generation is consequential in heat transformation theory, especially in the case of GDSs that extend over great distances.
To understand the importance, the warmup heat associated with the decay of stored potential energy must be tracked separately by a third form of specific universal entropy of a DS. The
specific mutual universal entropy (
) is defined as the difference between specific maximum universal entropy and specific conditional universal entropy and is the change in universal entropy per unit mass of the heat transfer medium of the DS, which change is mutual to the complexity yield or mutual to the heat that is transformed by the complexity yield,
This equation is a general form that can be applied to any DS in a network. It reveals that specific mutual universal entropy is fixed to the constant universal heat sink temperature. It is the quantity of specific maximum universal entropy that remains to be generated following the generation of specific conditional universal entropy at stage 3. It is specific universal entropy resulting from the decay of complexity by ARO at stage 4. The word mutual is specifically used to relate to the average mutual information of information theory and is discussed below in Section 6.
In the case of the TWC and TAC, there is no stage 4 heat input. There is only a stage 4 output that is equal to the heat originally converted to stored energy in stage 2. For the specific mutual universal entropy of the TWC,
the values of Calculation S1 in the
Supplementary Materials are applied,
The specific mutual universal entropy of the TAC is calculated using values in Calculation S2 in the
Supplementary Materials,
The reader can verify that these values are exactly equal to the specific maximum universal entropy minus the specific conditional universal entropy.
The three types of specific universal entropy of a DS presented above follow the convention of heat engine theory and the standard definition of universal entropy as a property of heat sources and heat sinks that are near or at thermal equilibrium. Entropy is discussed and calculated in heat engine theory. Non-conservative forced-convection heat engines are far-from-equilibrium systems. The way that entropy is calculated for such heat engines in theory is treated as if it is a universal entropy, being the calculated change in entropy of the universe with the universe defined as the local heat source connected to the engine (typically of a burning fuel) and the local heat sink of the engine cooling system connected to the heat engine (typically circulating air, water, or chemical coolant). The heat engine with local heat source and local heat sink are typically in the Earth environment (laying aside for now any discussion of heat engines on spaceships). Thus, the heat captured by the cooling system is carried away by design from the heat engine and exposed to the surrounding Earth environment (i.e., troposphere) to which the heat further dissipates. An example is the steam coming from the cooling tower of a power plant. That heat carried in the troposphere is transferred by the TWC and TAC to the tropopause, to the stratosphere and upper layers of atmosphere, and eventually to space. Thus, for complete and honest disclosure of universal entropy generated by such a heat engine, it should use the temperature of the Sun as the universal heat source and the temperature of space as the universal heat sink to calculate its universal entropy and its best case Carnot efficiency, because that is where true equilibrium in the universe is achieved. In such a case, the real efficiency of the heat engine in comparison to the Carnot efficiency is much less optimistic. This truly reveals the waste of heat engines that don’t capture and use heat exhausted to perform other functions. This is the big difference between DSs in nature and most non-conservative forced-convection heat engines designed and built by humans. DSs network together, with the heat exhaust from upstream DSs being used as heat sources for downstream DSs.
How do DSs compare to non-conservative forced-convection heat engines? The comparison can be made by considering local heat source temperatures and heat sink temperatures for DSs, just as is done in heat engine theory for non-conservative forced-convection heat engines. The questions to be answered generally by heat transformation theory for
basal [
45] (for usage of basal) DSs on Earth and specifically for the TWC and TAC that we have already discussed are (1) what quality of the exhausted heat of a DS is now available in the environment for use by other DSs and (2) what are these other DSs. Considering the heat quantities exhausted at stage 3 and stage 4 of a DS are known and do not immediately go to the universal heat sink, the calculated specific conditional universal entropy and specific mutual universal entropy suggest a quality of that heat in the local environment. Heat transformation theory needs far-from-equilibrium properties to represent the heat quality that remains in the environment prior to heat meeting its end fate of maximum universal entropy generation in the heat sink of space.
4.5. Specific Heat Qualities, Heat Transformation Effectivity, and Efficiency of a GDS
To address this need, the heat transformation theory proposes a fifth property called specific heat quality. Heat quality is another term that is occasionally used in the literature, typically to refer to the potential of heat to produce work based on the temperature of the local source of the heat. As such, it is related to entropy in that heat quality is considered to be lost as heat moves from a higher temperature environment into a lower temperature environment. Heat transformation theory defines and quantifies the specific heat quality available for use by a DS as being the amount of heat available per unit mass per unit temperature. It has the same units as specific universal entropy and is determined in a way that considers the specific universal entropy that can be produced from the point where the heat is made available to the DS by a heat source — following having been received by a local heat source from a universal heat source, perhaps through a network of DS — to poetically and literally the ends of the universe. When considered this way, specific heat quality is a measure of the potential specific universal entropy that can be generated as the available heat quality is used to perform work in the open system of Earth as heat dissipates on a path through Earth from the universal heat source to the universal heat sink.
In the equations of specific universal entropies, the heat source temperature of the Sun and the heat sink temperature of space are used, and . However, in calculating the specific heat quality depleted by a DS on Earth, the temperature of the heat source and heat sink of the local environment need to be used, and . The intent is to determine the amount of specific heat quality depleted by the DS and the amount remaining when the heat exhausts from the DS. Remembering that the heat quantities exhausted at stage 3 and stage 4 of a DS are different amounts and can be at different locations with different local temperatures, the specific heat qualities at stage 3 and stage 4 are considered separately.
The
specific conditional heat quality output,
is the quantity of heat per unit mass per unit temperature of the medium of a far-from-equilibrium DS receiving heat directly from the universal heat source and that is output at stage 3 and made available to the environment,
It is related to the APC of the DS. It has the same units as specific universal entropy, suggesting that it is a potential entropy. The second term in the expression is what the specific conditional universal entropy of the DS would be if the universal heat sink were the local environment rather than space. Considering that local heat sources and heat sinks are simple heat transferring open systems that are also far-from-equilibrium systems, this term cannot be called a specific universal entropy that is an equilibrium entropy. The first term of the equation is the specific conditional universal entropy. The second term of the equation is the specific total heat quality drop from the universal heat source to the local heat sink of the DS. It is the amount of heat quality that is depleted after leaving the universal heat source and entering the local heat sink of the DS.
The form of this second term of Equation 45 is used to define a
specific conditional heat quality drop,
of the DS,
This equation represents the amount of specific heat quality depleted by the DS because of the down step from the local heat source temperature to the local heat sink temperature of the environment at the location where the heat is released from the DS at stage 3 and, therefore, an amount of specific heat quality that is no longer available to be used by other DSs. For a DS that is using a local heat source rather than heat directly from a universal heat source, it is important to recognize the network effect of subdividing the original heat from the heat source to go down multiple branching and paralleling paths that break up the heat into multiple flows. This network effect results in each subsequent DS having specific heat quality drops and specific heat quality outputs based on local heat sources. Therefore, Equation 46 is defined by starting with the second term of Equation 45 and replacing with .
For the cases of the TWC and TAC dissipating heat at stage 3, the local environment is the tropopause. The average temperature of the environment at the tropopause of 222.15 K (–51 °C). To calculate specific conditional heat quality output for the TWC, put Equation 46 in terms of the empirical and calculated values of Calculation S1 in the
Supplementary Materials,
The specific conditional heat quality output of the TAC is calculated using values in Calculation S2 in the
Supplementary Materials,
Comparing these values to the specific conditional universal entropy values, it is interesting to note that the TWC has a specific conditional heat quality drop of 20 kJ/kg-K and the TAC has a specific conditional heat quality drop of 0.2 kJ/kg-K. What is exhausted to the environment as specific conditional heat quality output in both cases is 98.8% of the initial specific heat quality from the Sun. They each use 1.2% of the specific heat quality.
There are DSs in the layers of the atmosphere above the troposphere that can utilize the 98.8% of the remaining specific heat quality from stage 3 of the TWC and TAC. Between the troposphere and the stratosphere is another GDS that circulates the thinner, dryer air. This circulation is horizontal due to both the rotation of Earth and a temperature inversion in the stratosphere that prevents vertical convection.
8 The result is the phenomena of jet streams. The jet stream GDSs utilize heat given off by the TWC and TAC GDSs of the troposphere, as well as the drag of the spinning Earth on the atmosphere and direct heating by solar radiation. For the purpose of calculating a maximum possible effect, assume the heat exhausted at stage 3 of the TWC and TAC of the troposphere goes mostly to these jet streams due to the temperature inversion of the stratosphere. The contribution of these heat inputs to drive the jet streams are estimated in Calculation S3 in the
Supplementary Materials.
It is noteworthy in the calculations of Calculation S3 in the
Supplementary Materials that a heat-quality-transfer-rate flux delivered from the TWC and the TAC to the jet streams is determined by multiplying the specific conditional heat quality outputs of the TWC and TAC by their kinematic-mass-flow-rate fluxes. How the jet streams receive this amount of power in the form of heat at this level of quality (i.e., per unit temperature) depends upon the quantity of air mass available as a heat transfer medium to the jet streams and the specific heat capacity of the air. A similar consideration applies to all transfers of specific heat quality outputs from upstream DSs as inputs to downstream DSs. This exhibits the use of the specific heat quality output property in enabling the tracking of heat quality drops through an ecosystem network.
The
specific mutual heat quality output,
is the quantity of heat per unit mass per unit temperature of the medium of a far-from-equilibrium DS that is output at stage 4 and made available to the environment,
This equation developed similarly to the specific conditional heat quality output of Equation 45 and is related to the ARO of the DS. It has the same units as specific universal entropy, suggesting that it also is a potential entropy, as the specific conditional heat quality output. The
specific mutual heat quality drop,
of the DS similarly follows,
based upon a local heat source temperature rather than the universal heat source temperature due to network effects. This equation represents the amount of heat quality that is depleted by the DS because of the release of stored energy as heat to the local heat sink temperature of the environment at the location of stage 4 and, therefore, an amount of specific heat quality that is no longer available to be used by other DSs.
For the cases of the TWC and TAC, again there is no stage 4 heat input. There is only a stage 4 output that is equal to the heat originally converted to stored energy in stage 2. The stage 4 heat output is to the local environment that is lower in the troposphere. The average temperature is 288.15 K (15 °C). To calculate specific mutual heat quality output for the TWC, put Equation 50 in terms of the empirical and calculated values of Calculation S1 in the
Supplementary Materials,
The specific mutual heat quality output of the TAC is calculated using values in Calculation S2 in the
Supplementary Materials,
Comparing these values to the specific mutual universal entropy values, it is interesting to note that the TWC specific mutual heat quality drop is 0.4 kJ/kg-K and that of the TAC is 0.3 kJ/kg-K. What is exhausted to the environment in both cases is 99.1% of the initial specific heat quality from the Sun. They each use 0.9% of the specific heat quality that initially goes into complexity yield and is exhausted following decay at stage 4.
A specific maximum heat quality output and a specific maximum heat quality drop are not defined, considering specific heat quality is a property that can affect separate local heat sinks at different heat sink temperatures, at different times, and with different quantities of heat. In some cases, it is possible for the local heat sinks at stage 3 and stage 4 to be co-located. However, the heat outputs at stage 3 and stage 4 are different amounts and occur at different times, which can result in the specific heat quality outputs being utilized by different downstream DSs. For this reason, there is no explanatory value in calculating a specific maximum heat quality output and specific maximum heat quality drop.
The property of specific heat quality suggests an efficacy of heat in being transformable to stored energy and the associated complexity yield, as discussed above. Therefore, heat transformation theory defines
heat transformation effectivity,
as a sixth property of a DS and defined as the effectiveness of the DS at transforming heat into stored energy based on the specific heat quality,
The factor of one over the molar mass of the medium of the DS is used to put the numerator in terms of per unit mass just as the denominator. Heat transformation effectivity is based on the heat storage occurring during stage 2. This definitional equation provides the first theorem of heat transformation theory when Equation 33 and Equation 50 are substituted into it, the
heat transformation effectivity theorem,
Theorem 1. Heat transformation effectivity theorem: A greater local heat sink temperature results in a DS yielding the same complexity yield at a greater specific heat quality output.
It reveals that a DS with a greater local heat sink temperature has a greater heat transformation effectivity. Though the simplified units are in temperature, it is helpful to think of heat transformation effectivity in terms of units of complexity yield per unit of specific heat quality.
Proof of Theorem 1. To understand the heat transformation effectivity theorem, it is helpful to do an analysis of how it changes with environmental conditions of the DS. For any DS in steady state, a greater local heat sink temperature also results in an equivalently greater average temperature of the DS and an equivalently greater local heat source temperature to accomplish an unchanged local heat input and APC of the DS. This is because the internal energy of the local heat source that is available to be moved as heat is independent of the DS. The local temperatures are driven to increase or decrease for the heat source to reach a steady state in which its internal energy remains constant as it transfers heat into the DS. This results in the heat transfer from the local heat source to the local heat sink through the DS remaining constant with local changes in temperatures. Recall that the heat source and heat sink do not affect how the DS uses properties of matter to store energy, resulting in the complexity yield of a DS remaining constant even if the local heat sink temperature and associated heat transformation effectivity increase. A DS that involves the same conservative force field but has a greater heat transformation effectivity produces the same complexity yield with a lesser specific mutual heat quality drop. Also note that considering local heat input, , does not change and the energy storage of the constant complexity yield is only dependent on the properties of mass, this means that mass engagement is constant and local heat output at stage 3, , also does not change. Thus, a greater local heat sink temperature and associated heat transformation effectivity with the same local heat output at stage 3 results in a greater specific conditional heat quality output. In summary, a greater local heat sink temperature results in a greater heat transformation effectivity, which means that the complexity yield of the DS does not change but the specific heat quality output increases.
Heat transformation effectivity is not the same as real heat engine efficiency. The heat transformation effectivity is a conversion ratio. Real heat engine efficiency,
, is work output divided by heat input,
The Carnot efficiency of an ideal heat engine,
is the best possible efficiency that the DS could theoretically achieve if it were a dynamic zero-non-equilibrium-entropy cycle. As noted at the end of
Section 4.4, the Carnot efficiency of a given heat engine is historically based on the local heat source and heat sink temperatures.
Putting heat transformation effectivity in terms of these efficiencies results in
for which the specific maximum heat quality drop is
Equation 57 provides a second theorem that is a corollary of the heat transformation effectivity theorem of Equation 54.
Theorem 2. Corollary to the heat transformation effectivity theorem: Heat transformation effectivity is greater and the real heat engine efficiency is constant for a DS with a greater local heat sink temperature.
Proof of Theorem 2. Relating Equation 53 and Equation 57 results in,
Equation 59 reveals how heat transformation effectivity and real heat engine efficiency vary in relation to each other. As discussed previously, if the local heat sink temperature is greater for the same local heat input, then the steady state heat transformation effectivity is greater with complexity yield being a constant of the DS. Unlike specific universal entropy that is calculated based upon the universal heat source and heat sink temperatures, specific heat quality drop is affected by the conditions of the local temperatures. An analysis specific mutual heat quality drop of Equation 50 and the of Equation 58 reveals the effects. Recall that regardless of how heat input might change, the ratio of any energy flow through a DS to the mass flow through the DS is a constant. Equation 53 shows an inverse relationship of a greater local heat sink temperature to a lesser specific mutual heat quality drop with a constant complexity yield.
Considering the local heat source temperature must be greater by an equivalent amount, this results in a lesser difference of the inverse of the local heat sink temperature and the inverse of the local heat source temperature, , and, thus, a lesser as shown by Equation 58. Considering the specific mutual heat quality drop goes as the inverse of a single changing temperature multiplied by the difference in total heat input and heat output at stage 3 whereas goes as the difference of the inverses of two different temperatures that are changing by the same amount multiplied by the total heat input, therefore is lesser by a greater factor than specific mutual heat quality drop. The result is that specific conditional heat quality drop, Equation 46, is lesser by the greatest factor, it being the difference between and specific mutual heat quality drop. Using this analysis, the ratio on the left side of Equation 59 is greater with a greater heat sink temperature.
On the right-hand side of Equation 59, the Carnot efficiency of Equation 56 is lesser for equivalently greater local heat source and local heat sink temperatures. The real heat engine efficiency, Equation 55, does not change considering the local heat input and stage 3 heat output do not change. For heat engines that drive work output using non-conservative forces, a lesser real heat engine efficiency results from a greater local heat sink temperature. However, because of the fixed nature of the conservative work output per unit mass of a DS, local heat sink temperature does not affect the energy balance provided by the first law of thermodynamics, resulting in a constant real heat engine efficiency. This results in a greater ratio on the right side of Equation 59 equivalent to the greater ratio on the left side of the equation. DSs are just as efficient at any temperature and are more effective at great temperatures.
The two theorems of Equation 53 and Equation 57 are important to understand the function of DSs. DSs that operate in environments of greater local temperatures have less specific heat quality drop for the same complexity yield. This also means that specific heat quality outputs are greater even as local heat input and the local heat outputs at stage 3 and stage 4 do not change. Constant complexity yields and local heat flows with greater specific heat quality outputs at greater local heat sink temperatures means that the heat can support more DSs with their complexity yields in downstream in a network of DSs before the specific heat quality is routed into the universal heat sink of space or “burns out.” This is discussed further in Section 6.6.
9
Though higher local heat sink temperatures are better for DSs operating in an ecosystem, this is not the case for
augmentational [
45] (for usage of augmentational) non-conservative forced-convection heat engines. The efficiency of the non-conservative forced convection heat engine decreases for an increase in heat sink temperature. This means less work output is generated for the same heat due to the non-conservative nature of the involved forces. Together, these mean that the power of the heat engine drops even as the engine requires more fuel to be burned for the dropping power. Augmentational non-conservative forced convection heat engines have greater power and greater efficiency with lesser heat sink temperatures, together resulting in less exhaust heat that is just waste, though at a lower specific heat quality output. The heat sink is usually the environment, and the environment is not always as cold as is needed for a heat engine to work at the power desired and without overheating and failing as a result. Contrary to the non-conservative forced convection heat engine, the DS has the same efficiency with a greater specific heat quality output with greater local heat sink temperatures resulting in greater complexity production on Earth due to a greater number of DSs with lesser specific heat quality drops.
4.6. Steady-State Seeking and Transient Growth of GDSs
To make it easier to develop the models and the properties associated with single DSs and networks of DS, the analyses used to initially develop the models are based on steady-state conditions. However, the models and associated equations are based on the laws of thermodynamics that apply to all conditions, including transient conditions. Therefore the models and equations of the properties also apply to transient conditions. Analyses of these models indicate that when transient conditions occur in the environment of a DS, the DS seeks to return to steady state, just as any heat engine, as governed by the laws of thermodynamics. This steady-state-seeking function shifts the properties of the DS to drive toward total heat output coming into equality with total heat input as the DS asymptotically approaches a maximum APC based upon the available heat and available materials of the DS’s heat transfer medium. This characteristic prevents a cascading temperature effect and maximizes universal entropy production to an amount equivalent to simple heat transfer. The difference between the DS and simple heat transfer is that DSs have complexity yield.
It is noteworthy that DSs are a result of two things, the first being collections of matter with properties that produce conservative force fields and the second being a universal heat source and universal heat sink that provide the ultimate temperature difference that drives heat through the collections of material. Without either, the DS does not exist. If both are initially present, and then either the source of matter or the source of heat are lost, the DS grows smaller and finally disperses. The matter with its conservative force fields can hypothetically be placed anywhere in the universe with any local solar or galactic heat source and the heat sink of space and still function to seek steady state heat dissipation while producing complexity yield. The DS is as an emergent property of the unique combination of heat source, heat sink, and matter with its conservative force fields.
Imagine an Earth-like planet in orbit around a star that provides a greater or lesser heat rate flux or has a greater or lesser temperature. The gravitational field, structure, and material makeup of the Earth-like planet is the same, but the solar heat source is different due to the size of the star in combination with the distance of the plant from the star, reducing the solar flux as the inverse of square of the distance from the star. The DS of the planet will adjust its steady state average temperature to bring the total heat output from the planet equal to the total heat input to the planet by the entire network of DSs that form on the planet coming into quasi-steady state with each other. This means that an Earth-like planet around some other star is driven by the same ecological thermodynamics. This science is not unique to Earth. It also means that over the course of the astro geological history of our solar system, as the heat generation rate and temperature of the Sun have varied, so has the average temperature of Earth to adjust up and down to maintain a steady state.
The steady-state-seeking characteristic of a DS results in a transient process associated with APC, ARO, complexity yield, specific heat quality drop, and specific heat quality output. The DS grows as both additional mass and additional heat are available and shrinks as either mass or heat or both become unavailable. This happens in a process in which either additional mass or additional heat is fed into a DS that has existing excess heat or excess mass respectively available. This puts a DS in a growth transient that increases APC. While in this transient, the DS is using its complexity yield, a property of the heat transfer material medium and the conservative force field, to increase APC, resulting in an increase in total stored energy to a new, greater level of total average stored energy. Occurring with this is a transient increase in heat input at stages 1 and 2 with a lagging increase in heat output at stage 3 such that heat input at stage 1 continues to increase its difference from the heat output at stage 3. This generally will not affect local heat sink temperature at the stage 3 location of the cycle considering that excess heat dissipation through the local ecosystem is typically greater than the amount of heat being utilized by the DS due to
massive mixing, discussed below in the
Section 4.7. Specific conditional heat quality drop and specific conditional heat quality output are generally not affected if local heat source and heat sink temperatures do not change. They are specific properties, so the increased heat input and APC are directly related to an increase in mass engaged by the DS. As this occurs, ARO and heat output at stage 4 increase in a lagging response to the greater APC as the decay portion of the cycle increases its processing of energy-storing mass through stage 4 in response to the increase in mass being engaged in the storage of energy at stage 2. As with stage 3, this generally does not affect local heat sink temperature, specific mutual heat quality drop, or specific mutual heat quality output at the stage 4 location of the cycle due to massive mixing.
The specific mutual universal entropy of a DS is the measure of the amount of universal entropy that is the result of the ARO of the DS as the stored energy built up by the DS is released as heat, resulting in a specific mutual heat quality drop. Because the heat that is captured as stored energy during stage 2 is not released right away as heat output as stage 3, the heat is not part of the specific conditional universal entropy. This means that the release of the heat at stage 4 and the result of the specific mutual universal entropy that it generates in the universal heat sink are delayed. What this looks like in an early Earth filled with DSs that are in a geological time period of transient growth is that the total heat output rate flux from Earth is less than the total heat input rate flux to Earth. During such a time period, growth of stored energy and decrease in Earth-wide average temperature can occur as heat is stored in mass interactions with conservative force fields resulting from heat-dissipating work. With a lower rate of heat output needed to keep up with a quasi-steady heat input rate because some of the heat input is going to stored energy simultaneously all over Earth, the local heat sink temperatures can drop, allowing the average temperature of the DS to decrease to maintain the heat output rate. When a growing DS finally achieves a far-from-equilibrium condition that is near steady state, this means that the stored energy decay rate of ARO has caught up with the energy storage rate associated with APC, a result of the total local heat output rate at stages 3 and 4 being equal to the total local heat input rate at stages 1 and 2. Temperatures also return to their steady-state levels.
4.7. Networking of GDSs Results in Massive Mixing, Heat Delivery, and Complexity
Before heat released from a GDS can pass all the way to the universal heat sink, it must pass through a network of GDSs that eventually allow the heat to radiate to space. The TWC, TAC, and jet stream cycles are not the only GDSs on Earth. There are natural convective heat engine cycles in the oceans of water (i.e., the hydrosphere) that are driven by direct solar heating and by inputs from other GDSs, resulting in ocean current cycles that move massive amounts of water to change longitude, latitude, and depth. The other GDSs that can have inputs to the ocean current cycles include lithospheric GDSs and the DS that is the rotation of Earth under the pulls of gravity from the Sun and the Moon.
There are also lithospheric GDSs in the core, mantel, and crust of Earth. They include lava cycles that surface at volcanoes and the mineral laden water of a lithospheric water cycle that carry heat generated in the lithosphere to the surface, such as by way of hot springs. They also include cycles that drive plate tectonics that produce new crust at mid-ocean ridges and drive old crust back underground at subduction zones. The GDSs of the lithosphere have various heat sources, one being the internal energy left in the core from the original collapse of matter into the formation of the Earth, a second being the friction of the liquid swirling pool of the core and the friction of earth being pulled and squeezed by the gravity of the Sun the Moon, both of these a result of the Earth rotation cycle DS. A third source of lithospheric heat is nuclear in nature and is a result of the radioactive decay of elements in the mantel and crust of Earth that have their origin in the fusion and eventual collapse and supernova of stars.
All heat that transfers to the surface of the lithosphere and hydrosphere escapes into the atmosphere by way of transferring their heat into the TWC and the TAC. The TWC and the TAC are a combination of direct solar heating and the specific heat quality outputs of all of the other GDSs of the Earth. The primary means of heat escaping from Earth is by gravity and buoyancy driving natural convection through the atmosphere, and not by black-body radiation directly from the lithosphere and hydrosphere.
All these GDSs of the atmosphere, the hydrosphere, and the lithosphere produce what heat transformation theory calls
massive mixing. The massive mixing is a phenomenon of the emergent property of
complexity that is driven by the complexity yields of the GDSs. The mathematical basis of complexity is developed in part II of this paper series [
58]. For the conceptual basis of complexity, consider the example of water entering the TWC from a lake high in a mountain range that is fed by a hot spring at the bottom of the lake. A separate lithospheric water cycle GDS that is transferring heat from the mantel of Earth to the surface of Earth uses the water to lift the heat to the altitude of the lake, storing an increase in gravitational potential energy per mole of the water based on the change in distance from the center of Earth from the depth below the ground where the water received the heat from the mantel to the altitude of the mountain lake. That starting potential energy per mole of the water in the lake is transferred to the TWC when the water in the lake evaporates because of solar power and enters the TWC. That transfer of potential energy is what heat transformation theory defines as complexity. However, the potential energy per mole brought from the lithospheric water cycle with the water into the TWC is not included in the complexity yield of the TWC. The complexity yield of the TWC is the amount of potential energy per mole that the TWC imparts to the water as it takes the water to its new level of potential energy at the top of the troposphere. The starting potential energy that the water had in the mountain lake is still with that water. Thus, the networking of the TWC with the lithospheric water cycle increases complexity. If the water goes on to feed into another DS from the top of the troposphere, the complexity is a combination of the potential energy per unit mole imparted by the complexity yield of the lithospheric water cycle plus the potential energy per unit mole imparted by the complexity yield of the TWC.
On the decay portion of the TWC when the water precipitates, if it falls into a lake at the bottom of the mountain range and then flows downriver to an ocean, the amount of released potential energy per mole includes some of the potential energy per mole received from the lithospheric water cycle DS that lifted the water to the mountain lake. The remaining amount of stored gravitational potential energy per unit mole remaining in the water once its reaches the ocean is summative to the complexity of the network if it is taken up by another DS. The lesser amount of gravitational energy per unit mole that remains is transferred on the next DS as yet another positive, though lesser contribution to total network complexity. As the water continues to be passed from GDS to GDS, the total network grows in complexity as potential energy per mole stored in the water is passed along, taking heat with it. The same argument applies to lithospheric earth cycles (i.e., as in elements making up the mantel and crust) and atmospheric air cycles, as well as the cross connection from earth to water (e.g., magma to hot spring), water to air (e.g., TWC to jet stream), and water and air to earth (e.g., water and air penetrating and mixing with soil to start the carbon, nitrogen, and sulfur cycles).
The universal solvency and volatility of water is what ties this together. The TWC with the help of other DSs in the atmosphere move water all over the surface of Earth, picking it up from large bodies of water and then moving it with atmospheric air currents over land where it rains. The water is then driven by gravity downhill by soaking into the ground and down into subterranean lithospheric streams, or by running over the surface and collecting in streams and rivers where it eventually flows into ponds, lakes, seas, and oceans. As the water travels, it physically wears away at earth materials on and below the surface, resulting in it carrying silt and dissolved elements and minerals with it as it travels upward in lithospheric water cycles of hot springs on the surface or hydrothermal vents, smokers, and magma seafloor spreading along the Mid-Atlantic Ridge at the bottom of oceans, or downward on the decay side of the atmospheric, hydrospheric, and lithospheric water cycles. Some of these materials settle out in subterranean mineral deposits or surface level mineral deposits or sediments along the way, while others make it all the way into a larger body of water, where they mix with other materials that have collected there over geological periods of time. Once in a body of water that is large enough to have its own large-scale GDSs, the material gets moved around more in sea and ocean currents that spread the material out over great distances. This massive mixing is a result of the complexity of potential energy per mole that continues to drive water, air, and earth to the lowest gravitational point as heat is given off, only for water, air and earth to pick up heat again to be driven by buoyancy and natural convection away from the center of Earth to provide more complexity yield. The more water, air, and earth that becomes engaged in variations of these DSs, the greater the lengthening and spreading of these variations, the greater the complexity of the network as calculated by the stoichiometric summing of the hand-offs of potential energy per mole.
All of this massive mixing with water as the main solvent results in growing concentrations in the waters of Earth of simple chemicals that form when elements prevalent in the earth and in the air react with water: dissolved salts, acids, and bases (as the first-formed simplest chemicals) made from elements from the first three lines of the periodic table and particulates of minerals that include heavier elements. These concentrations are a direct result of the APCs, AROs, and complexities of the GDSs driven by solar heating, buoyancy, and the local heat sources in the Earth. The concentrations of simple chemicals grow over geological time, going through various surges resulting from large-scale events such as volcanoes, meteor strikes, and hurricanes. The more consistent growth toward high concentrations is a result of the regular, ongoing GDSs of the atmosphere, hydrosphere, and lithosphere driving either steadily higher or asymptotically toward a steady state of saturated concentrations that level out due to the plating out of minerals and the precipitation of salts.
The greater the complexity, the greater the massive mixing, the greater the material and heat carrying capacity as functions of the emergent property. The complexity of Earth resulting in massive mixing prevents superheating and mass disengagement, making DSs optimal in efficiency by reducing local heat sink temperatures, equalizing local heat source and heat sink temperatures to minimize specific heat quality drops, and maximizing use of available heat with available DS medium material. With the massive mixing of concentrations of ions, simple chemicals, and particulate minerals along with the heat being carried by the ions, molecules, and particulates themselves, comes the opportunity for more complex chemical reactions.