2.1. Effect of DMAM and DMAPS on the Salt Resistance of Polymers
Polyacrylamide (PAM) is a thickening agent widely used in oilfield operations, including drilling, profile control, water blocking, oil displacement, and fracturing [
16]. However, PAM’s resistance to high temperature and salinity was insufficient. N, N-Dimethylacrylamide contains rigid dimethyl groups, providing better temperature resistance than AM. The steric hindrance from these dimethyl groups increases intermolecular repulsion, preventing polymer chains from curling and from hydrolysis in high-temperature, high-salinity environments. DMAA is more suitable than AM as a backbone for high-temperature and high-salinity-resistant polymers [
17,
18,
19]. Zwitterionic polymers are polyelectrolytes that contain both cationic and anionic groups. In electrolyte solutions, electrostatic repulsion between these groups is shielded, allowing polymer chains to fully extend. This results in increased drilling fluid viscosity and shear strength, thereby enhancing rheological properties [
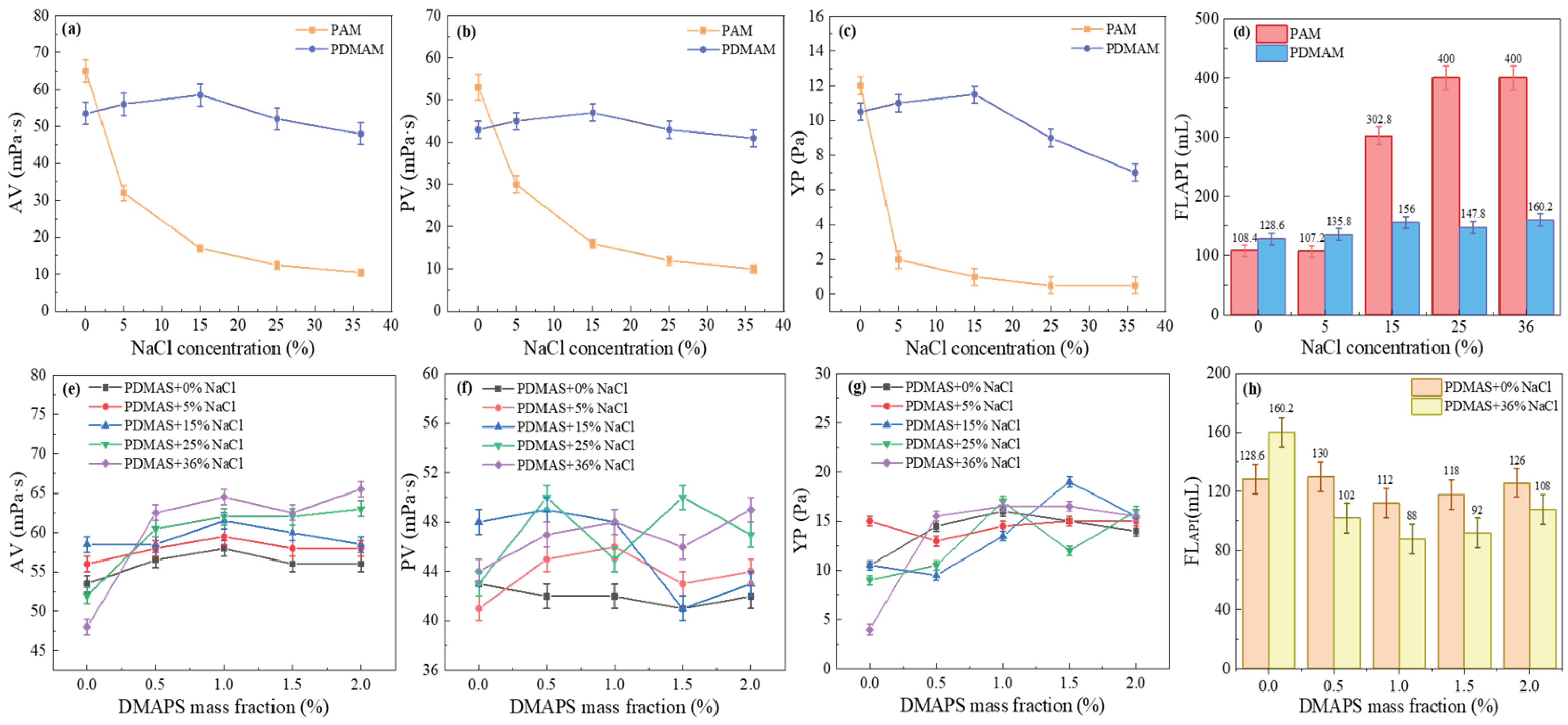
20]. The effects of PAM, PDMAA, and PDMASs on solution properties were evaluated at different NaCl concentrations, with a polymer concentration of 3.0 wt%.
As shown in
Figure 1(a)-(d), the AV, PV, and YP of the PAM/CFWBDFs reduced significantly with increasing NaCl concentration from 0% to 36%. As shown in
Figure 2(d), the FL
API of the PAM solution increased with increasing NaCl concentration, with all filtration losses observed at 25% NaCl. Under positive pressure, the polymers could form filter cakes that seal micropores and microcracks. A higher polymer molecular weight or larger hydrodynamic volume enhanced the sealing effect on micro/nanopores within the filter cake, resulting in a dense cake and less filtration loss [
21]. Greater molecular weight also strengthened the binding effect of highly hydrating groups (such as amide and sulfonate groups) on free water, further reducing the filtration loss. PAM has good viscosity-increasing properties in freshwater. However, a higher concentration of Na⁺ caused PAM chains to curl, weakening the viscosity enhancement and the filtration loss reduction properties. Surprisingly, there were no significant declines in AV, PV, or YP of PDMAA/CFWBDFs with increasing NaCl concentration, and higher NaCl concentrations did not increase FL
API. It was indicated that PDMAA exhibited better salt tolerance than PAM. From
Figure 1(e)-(h), it was clear that the AV of PDMAS/CFWBDFs increased as the NaCl concentration rose after adding the DMAPS to the PDMAA molecular chains. The PV stayed within a narrow range of 40.0 to 48.0 mPa·s, and the YP ranged from 9.5 to 16.5 Pa. When the mass fraction of DMAPS was 1.0 wt%, there was the best performance of PDMAS compared to PDMAA.
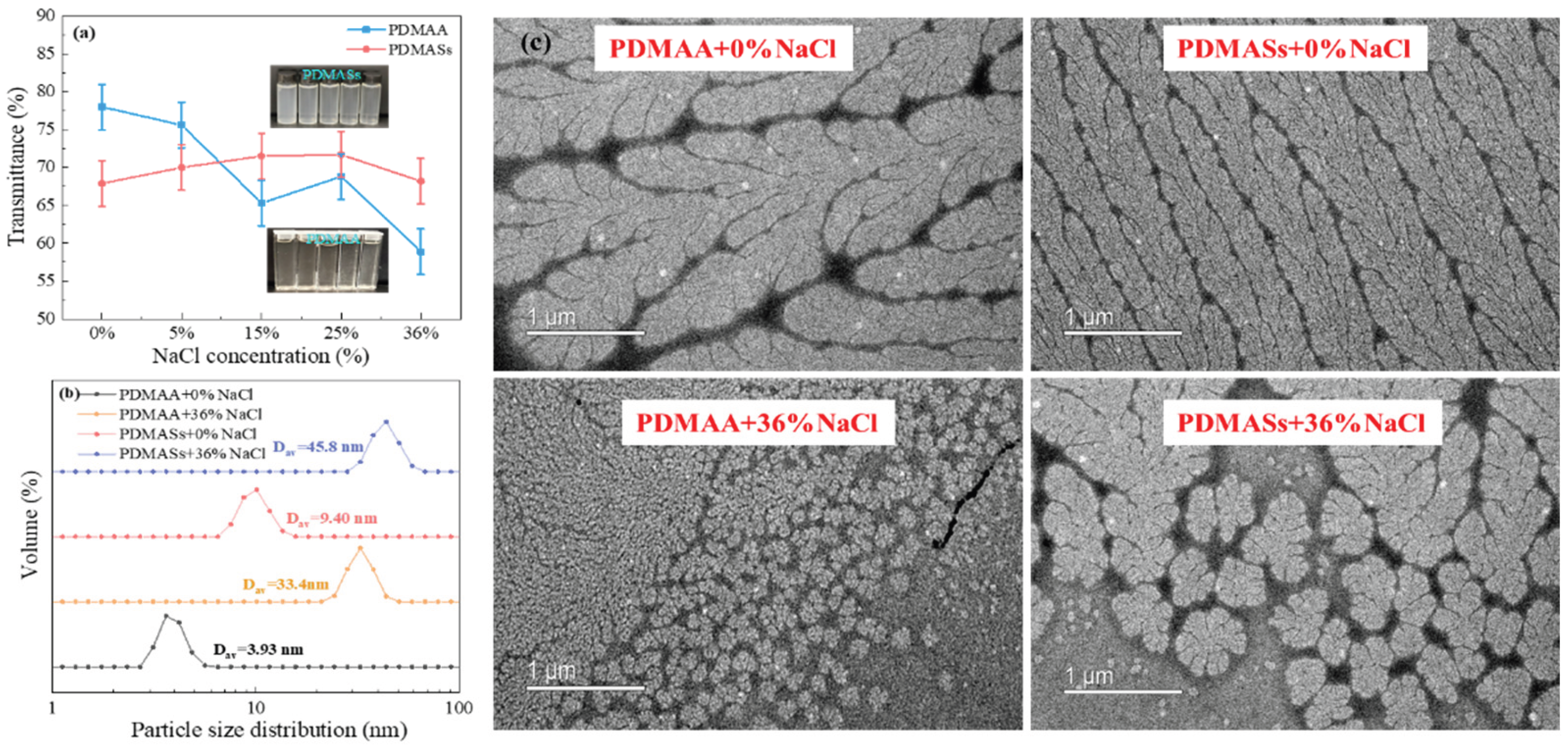
As shown in
Figure 2(a), the transmittance of the PDMAA/CFWBDFs decreased steadily with increasing NaCl concentration. The transmittance of PDMAS/CFWBDFs remained mainly stable at around 70%. The PDMAM/CFWBDFs became slightly turbid as NaCl concentration increased, while the PDMAS/CFWBDFs shifted from clear to slightly turbid. This phenomenon may be due to the formation of hydrophobic regions by PDMAM and PDMAS within the solutions, leading to decreased light transmittance [
22].
From
Figure 2(b), it was shown that at 0% NaCl, the average particle sizes (D
av) of PDMAA and PDMAS were 3.93 nm and 9.40 nm, respectively. At 36% NaCl, D
av increased to 33.4 nm for PDMAA and 45.8 nm for PDMAS. It suggested that NaCl promoted polymer extension, allowing polymers to adsorb more water, and the D
av of polymers was enlarged.
As shown in
Figure 2(c), both PDMAA and PDMAS had slender, willow-like shapes at 0% NaCl. More side chains were stretching out; the polymers were fully extended in a high-salinity environment. After the polymer chains extended, they enhanced the interpolymer interactions, thereby forming a three-dimensional associative network. The network likely resulted from weak hydrophobic interactions of dimethyl groups in DMAA. A higher concentration of Na+ increased the polarity of the solutions and promoted hydrophobic association among a small number of dimethyl groups, resulting in the formation of a three-dimensional associative network [
23]. Undoubtedly, PDMAA/CFWBDFs exhibited higher AV, PV, and YP and lower FL
API than PAM/CFWBDFs under high-salinity conditions. In addition, PDMASs possessed betaine side chains, which extended and resulted in a larger hydrodynamic radius in a high-salinity environment [
24], thereby enhancing AV, PV, and YP, and reducing filtration loss.
2.2. Effect of AM Mass fraction on the Properties of the Crosslinked System
Polyethyleneimine was introduced to crosslink polymers via transamidation reactions at high temperatures, preventing polymer degradation and performance failure due to high-temperature hydrolysis. Polyethyleneimine (PEI) is an aziridine polymer containing abundant imine groups. These imine groups are highly reactive and easily crosslink with functional groups in polymers, such as amide, carboxyl, or hydroxyl groups. PEI is often used as the crosslinker in oilfield operations, including profile adjustment, water blocking, and fracturing [
25]. The number of amide groups, which serve as crosslinking sites, will influence the crosslinking efficiency between the polymer and PEI. Under conditions of 180 °C and 36% NaCl, the crosslinked product between synthesized polymers (PDMASA-0, PDMASA-1, PDMASA-2, PDMASA-3, and PDMASA-4) and 0.25wt%(w/v) LPEI
1800 after aging was observed. The rheological and filtrate loss properties of each sample were evaluated.
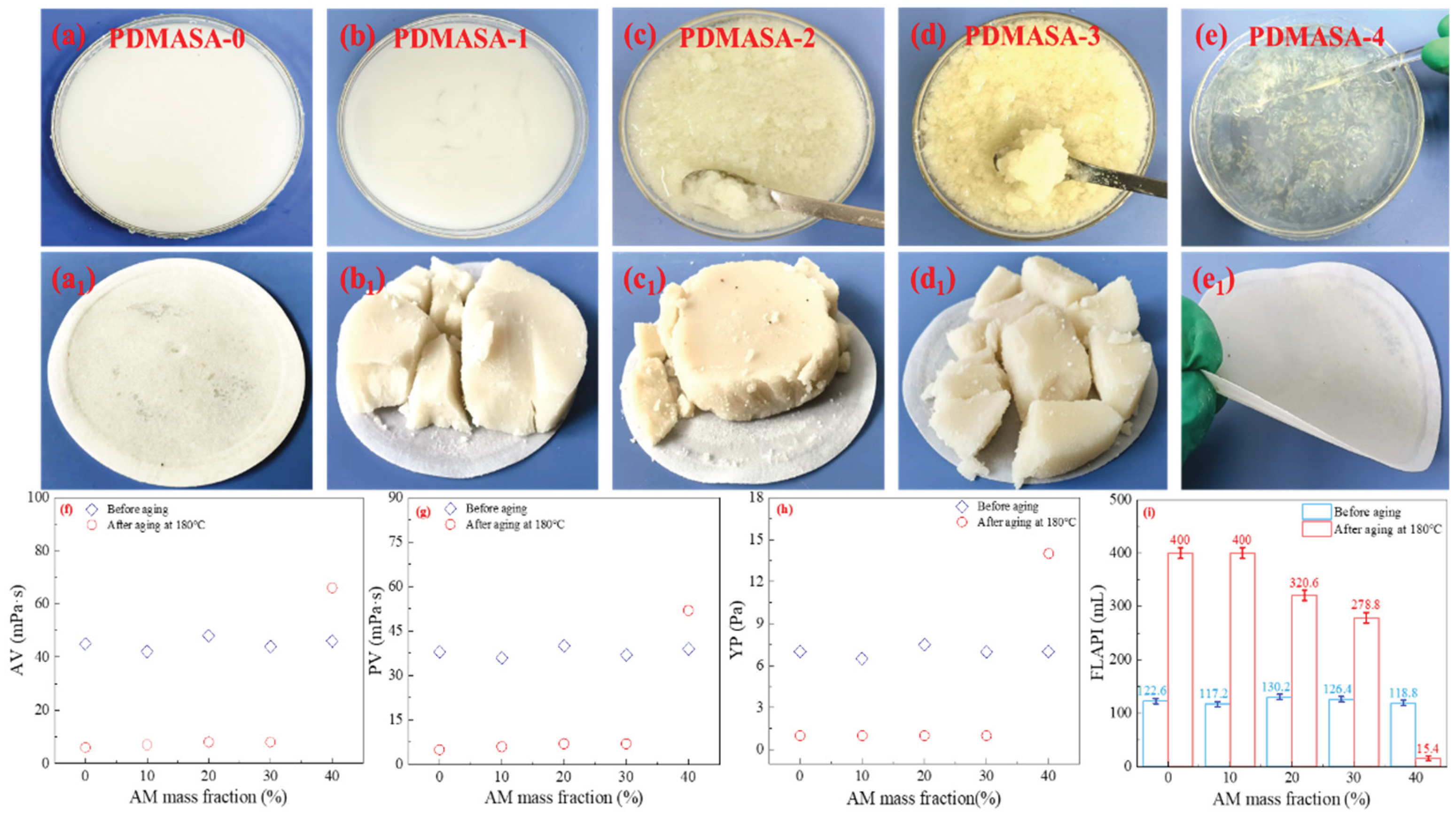
As shown in
Figure 3. From
Figure 3(a)-(e), under 36% NaCl conditions, PDMASA-0(LPEI
1800)/CFWBDFs and PDMASA-1(LPEI
1800)/CFWBDFs appeared milky white and highly diluted after aging at 180 °C. In contrast, many macroscopic gel particles existed in the PDMASA-2(LPEI
1800)/CFWBDFs and PDMASA-3(LPEI
1800)/CFWBDFs. Surprisingly, the PDMASA-4(LPEI
1800)/CFWBDFs formed a gel that did not flow after aging [
26]. As demonstrated in
Figure 3(f)-(h), under 180 °C and 36% NaCl conditions, the combining systems of PDMASA-0, PDMASA-1, PDMASA-2, and PDMASA-3 with 0.25wt%LPEI
1800 exhibited the AV of 6.0 to 8.0 mPa·s, the PV of only 5.0 to 7.0 mPa·s, and the YP of only 1.0 Pa. The weakly gel formed by PDMASA-4 and LPEI
1800 became a flowable viscous solution after shearing, with the AV of 66.0 mPa·s, the PV of 52.0 mPa·s, and the YP of 14.0 Pa. The results suggested excessive crosslinking between PDMASA-4 and LPEI
1800.
From
Figure 3(i), it was clear that the FL
API of systems from
Figure 3(a) to (d) are 350.0, 350.0, 320.6, and 278.8 mL, respectively. However, the FL
API of the system shown in
Figure 2(e) was only 15.4 mL.
The filter cakes from each system are shown in
Figure 3 (a
1). The PDMASA-0(LPEI
1800)/CFWBDFs had a thinner filter cake but experienced significant filtration loss. It was suggested that PDMASA-0, lacking amide groups, could not crosslink with LPEI
1800 at high temperatures. The filter cakes of the CFWBDFs shown in
Figure 3(b
1)-(d
1) were thicker, with a large amount of filtration loss, indicating that the water-insoluble gel particles were formed by PDMASA-1, PDMASA-2, and PDMASA-3 crosslinking with LPEI
1800.It confirmed that PDMASA-1, PDMASA-2, and PDMASA-3 could crosslink with LPEI
1800. However, the gel particles could not effectively improve the rheological properties or reduce the filtration loss.
After PDMASA-4 crosslinking with LPEI
1800, the viscous solution could be obtained. It showed increased AV, PV, and YP and a decreased FL
API. As shown in
Figure 3(e
1), the filter cake was thin, with no visible gel particles on its surface [
27]. The results indicated that adding 0.25% LPEI
1800 (m/v) with an AM mass fraction of 40 wt% to PDMASA polymerization enabled PDMASA and LPEI
1800 to crosslink [
28]. Even with excessive crosslinking, it still aligned with our predicted model. Therefore, the optimal mass fraction for AM polymerization was 40 wt%.
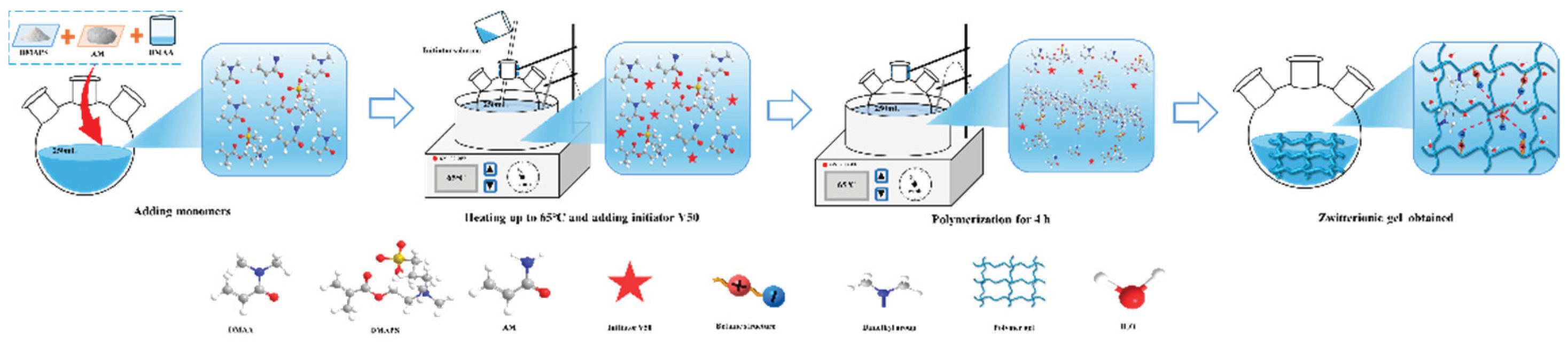
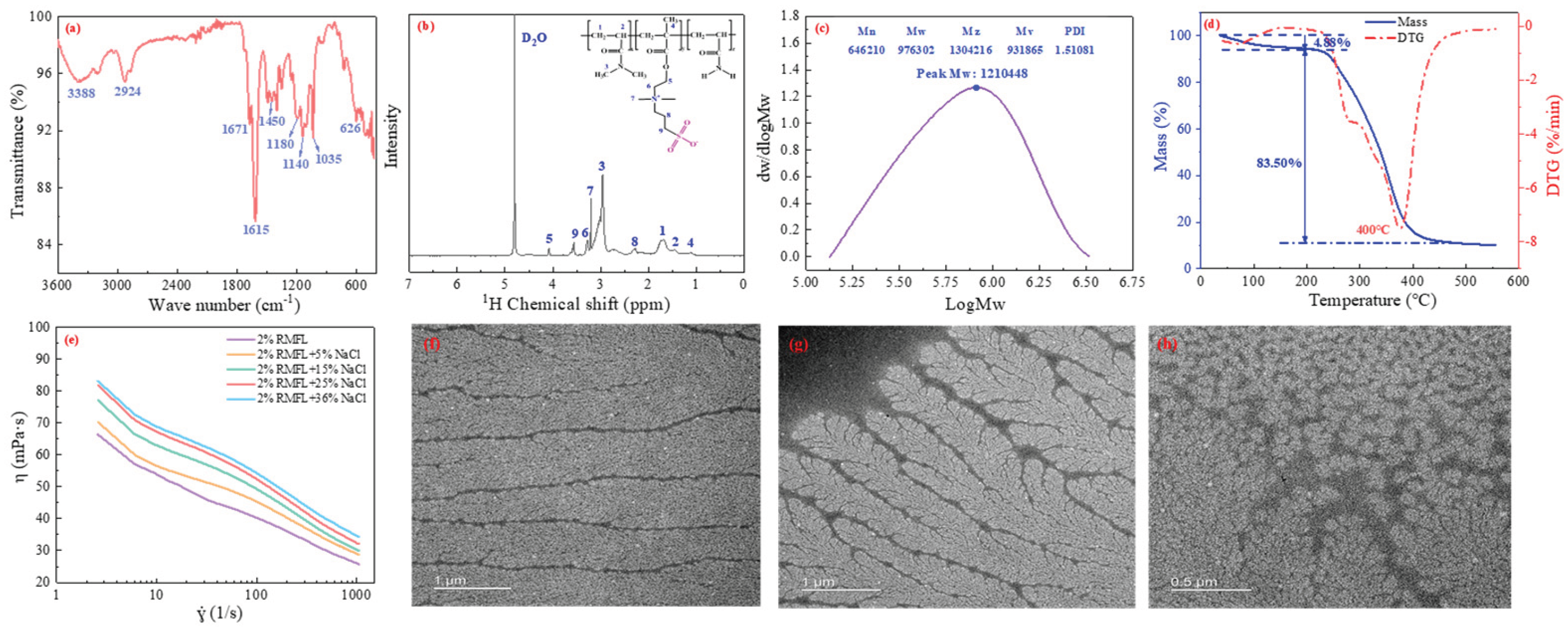
2.3. Preparation and Characterization of RMFL
The preparation method is shown in
Figure 4. The synthesis route was illustrated in the above section. The characterization of RMFL is shown in
Figure 5. The infrared spectrum is shown in
Figure 5(a). The characteristic peak at 3388 cm⁻¹ was from N-H stretching in the amide groups; the peaks at 2924 and 2870 cm⁻¹ from C-H stretching in methyl and methylene groups [
29]; the peaks at 1671 cm⁻¹ were from the C=O stretching in the amide groups; the peak at 1615 cm⁻¹ was from N-H bending vibration; the peak at 1450 cm⁻¹ was from the N⁺-C stretch in the quaternary ammonium cation; the peaks at 1180 cm⁻¹ and 1037 cm⁻¹ were from S=O and S-O stretching in the sulfonic acid group; the peak at 628 cm⁻¹ was from C-S stretching [
30]. Infrared spectroscopy analysis indicated that the product contained amide, methyl, carbonyl, quaternary ammonium, and sulfonic acid groups.
The
1H NMR spectrum is shown in
Figure 5(b), the peak at 1.11 ppm was attributed to the chemical shift of H on the methyl groups attached to the polymer main chains; the peak at 1.53 ppm was from the chemical shift of H in the methylene groups of the main chains; multiple peaks between 1.60 and 1.80 ppm originated from the chemical shift of H on the methylene groups within the main chain[
31]; the peak at 2.27 ppm came from the chemical shift of H on the intermediate methylene group of N+-CH
2-CH
2-CH
2-SO
3[
32]; The peaks from 2.80 to 3.20 ppm correspond to the chemical shift of H on -CON(CH₃)₂; the peak at 3.20 ppm was related with the chemical shift of H in N+(CH₃)₂; and the peak at 3.28 ppm corresponded to the chemical shift of H in N+-CH₂; the peak at 3.56 ppm resulted from the chemical shift of H in the methylene bridge connected to the sulfonate group within N+-CH₂-CH₂-CH₂-SO₃. The peak at 4.08 ppm could correspond to the H chemical shift of -COO-CH₂- [
33];
1H NMR and FTIR indicated that the product contained functional groups of the monomers. The molecular structure aligned with the design, confirming the successful polymerization of RMFL.
As shown in
Figure 5(c), the number-average molecular weight (M
n) of RMFL was 897940 Da, and the weight-average molecular weight (M
w) was 1176302 Da. The polydispersity index (PDI) was 1.31081, indicating a narrow molecular weight distribution and good polymerization efficiency, which helped to minimize experimental errors. The thermogravimetric (TGA) curve of RMFL is shown in
Figure 5(d). As the temperature increased from 50 °C to 550 °C, the weight loss of RMFL was mainly divided into three stages. The first stage was from 50 to 197 °C, resulting in approximately 4.88% mass reduction. The mass loss was primarily due to the evaporation of bound water adsorbed on amide and sulfonic acid groups, as well as free water between the polymer chains. The second stage was from 197 to 475 °C; approximately 83.50% mass reduction was observed, primarily due to the thermal degradation of amide, sulfonic acid, the quaternary ammonium cation, and methyl groups. The third stage was from 475 °C to 550 °C; RMFL exhibited less mass loss, with a decrease of 1.87%. During this stage, the backbone of RMFL underwent scission until the polymer was completely carbonized.
The microtopography of RMFL is shown in
Figure 5(f)-(h). From
Figure 5(f), RMFL displayed a fine and willow-branch-like structure under 0% NaCl conditions, which can be attributed to the electrostatic attraction between the anionic and cationic groups on the polymer side chains. The electrostatic attraction led to the contraction of the side chains. In
Figure 5(g), the side chains of RMFL become extended under 36% NaCl conditions. Because the electrostatic attraction between quaternary ammonium cations and sulfonate groups was shielded by a large amount of Na⁺. Furthermore, under saturated salt conditions, the network structure formed by RMFL was observed in
Figure 5(h), which may result from intermolecular electrostatic and hydrophobic interactions among RMFL molecules, with electrostatic interactions being dominant. Such a network structure contributes to the regulation of the rheological properties of clay-free water-based drilling fluids.
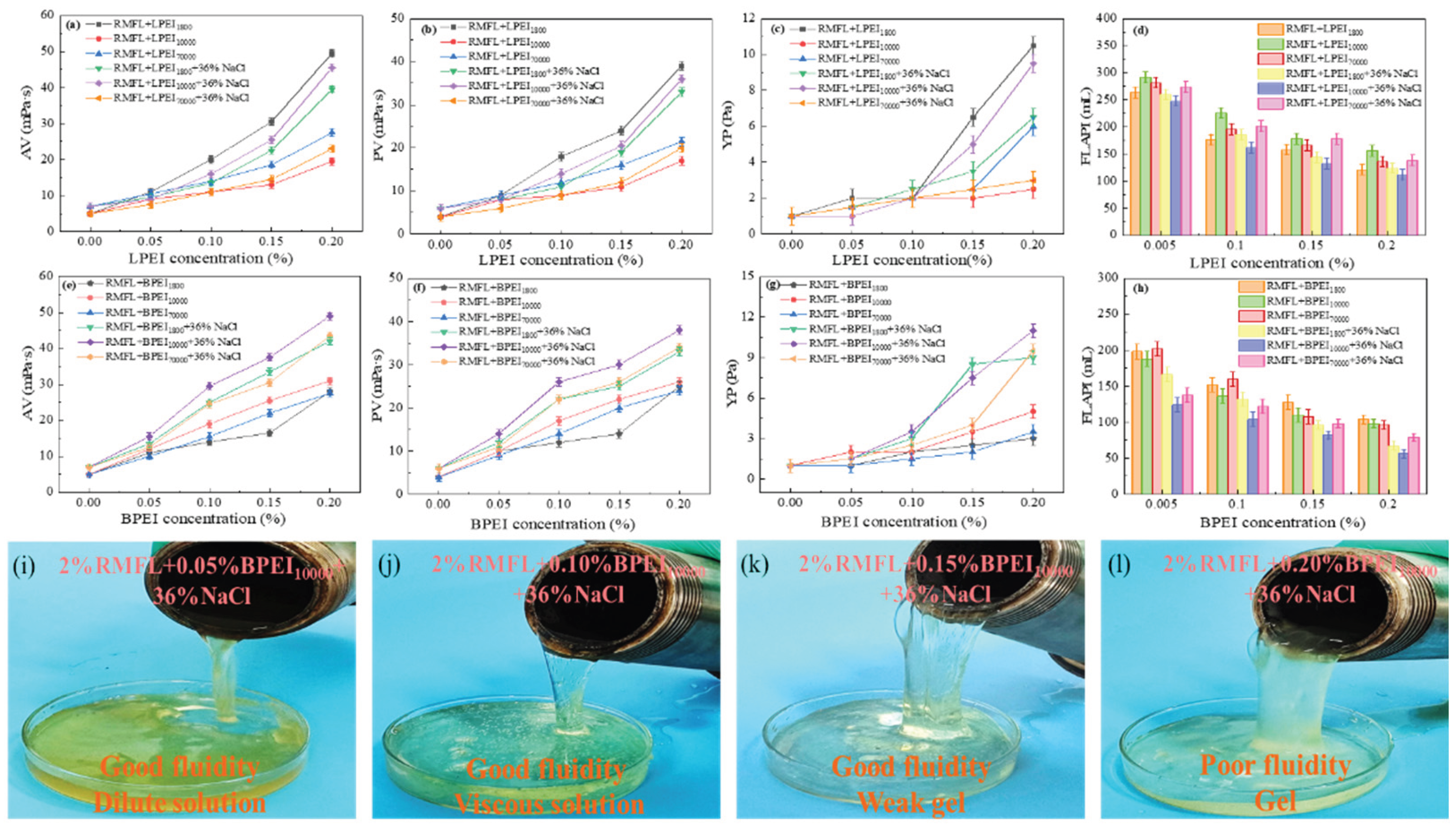
2.4. Effect of Polyethyleneimine on the Performance of RMFL
The performance of the RMFL crosslinked with PEI of different molecular weights, configurations, and concentrations (w/v) is illustrated in
Figure 6. As shown in
Figure 6(a)-(d), increasing LPEI
x concentration gradually raised the AV, PV, and YP of the RMFL(LPEI
x)/CFWBDFs after aging at 180 °C for 16h. The FLAPI gradually decreased. Higher LPEI
x concentrations led to increased crosslinking between RMFL and LPEI
x, enhancing the rheological properties of the RMFL(LPEI
x)/CFWBDFs and reducing filtration loss.
LPEIx with different molecular weights exhibited different crosslinking effects at the same concentration. At 0% NaCl, LPEI1800 demonstrated optimal crosslinking with RMFL, as the RMFL(LPEI1800)/CFWBDFs achieved the highest AV, PV, and YP after aging at 180 °C, along with the lowest FLAPI. This difference arises because the three LPEIx have different molarity at the same concentration. The molarity of LPEI1800 was 2.22×10-3 mol/L. And the molarity of LPEI10000 was 0.4×10−3 mol/L, whereas the molarity of LPEI70000 was only 0.057×10-3 mol/L. Although LPEI1800 has a lower molecular weight, its molar concentration is more than five times that of LPEI10000 and thirty-nine times that of LPEI70000. This indicated that the molarity of the crosslinker is the primary factor influencing the crosslinking efficiency in freshwater environments, and it has a greater impact than molecular weight.
But LPEI10000 was the most effective in crosslinking with RMFL under 36% NaCl conditions. This is because the longer polymer chains of LPEI10000 can simultaneously interact with multiple RMFL chains, facilitating binding and the formation of a more extensive three-dimensional network. In high salinity environments, electrostatic interactions are strongly screened, so the crosslinking efficiency is mainly determined by the chain length and spatial bridging ability. Therefore, although LPEI1800 has a higher molar concentration, its shorter chain length limits the number of effective crosslinking sites.
As shown in
Figure 6(e)-(h), with increasing BPEI
y concentration, the AV, PV, and YP of the RMFL(BPEI
y)/CFWBDFs gradually increased after aging at 180 °C. The RMFL(BPEI
y)/CFWBDFs exhibited higher AV, PV, and YP, with the lowest FLAPI. BPEI
y demonstrated a superior crosslinking effect compared to LPEI
x. The results were attributed to the branched structure of BPEI
y. BPEI
y exposed a greater number of highly reactive imine groups at the terminal ends of side chains, thereby increasing the probability of reaction with RMFL. Therefore, BPEI
y was more effective than LPEI
x in crosslinking with RMFL [
34], significantly improving the rheological properties of RMFL(BPEI
y)/CFWBDFs under high-temperature and high-salinity conditions.
Comparing the effects of BPEIy with different molecular weights on the RMFL(BPEIy)/CFWBDFs, BPEI10000 exhibited the most effective crosslinking with RMFL under both 0% NaCl and 36% NaCl conditions. The RMFL(BPEI10000)/CFWBDFs showed the highest AV, PV, and YP, and the lowest FLAPI after aging. This is the combined effect of molecular configuration, molecular weight, and molarity. The crosslinking efficiency of BPEI70000 was lower because of its lower molarity at the same concentration. When the molar concentrations were comparable, such as between BPEI1800 and BPEI10000, a higher molecular weight may lead to improved crosslinking performance. Under saturated NaCl conditions, BPEI1800 and BPEI10000 were subject to strong charge screening. However, due to its higher molecular weight and larger hydrodynamic volume, BPEI10000 exhibited stronger bridging ability and could simultaneously interact with multiple RMFL chains. In contrast, the smaller hydrodynamic volume of BPEI1800 limited it to localized, single-point interactions.
As shown in
Figure 6(i)-(l). When the BPEI
10000 concentration was 0.05%, the RMFL(BPEI
10000)/CFWBDFs formed a dilute solution with good fluidity after aging at 180 °C. At a BPEI
10000 concentration of 0.10%, the RMFL(BPEI
10000)/CFWBDFs became thicker after aging, exhibiting wall-clinging, easy-flow and possessed weak gel-like properties.
At a BPEI
10000 concentration of 0.15%, RMFL and BPEI
10000 created a gel with a stronger internal network. A “spitting tongue” phenomenon demonstrated significant flow resistance during pouring [
35]. At a BPEI
10000 concentration of 0.20%, the internal network of the crosslinked polymer became even stronger, forming a cohesive gel. The results indicated that at BPEI
10000 concentrations of 0.10% or higher, the crosslinked polymer network was sufficiently robust to form a solid gel. It was always better to ensure adequate fluid flow during drilling rather than allow solid gel formation. Solid gel formation can cause serious problems, such as drill bit embedding, difficulty pumping fluids, and inefficient rock penetration. In summary, the optimal BPEI
10000 concentration was 0.10%. When 0.10%(m/V) BPEI
10000 was crosslinked with 2% RMFL, it did not form a soft gel. Still, it effectively increased the AV, PV, and YP of CFWBDFs under 180 °C and 36% NaCl conditions.
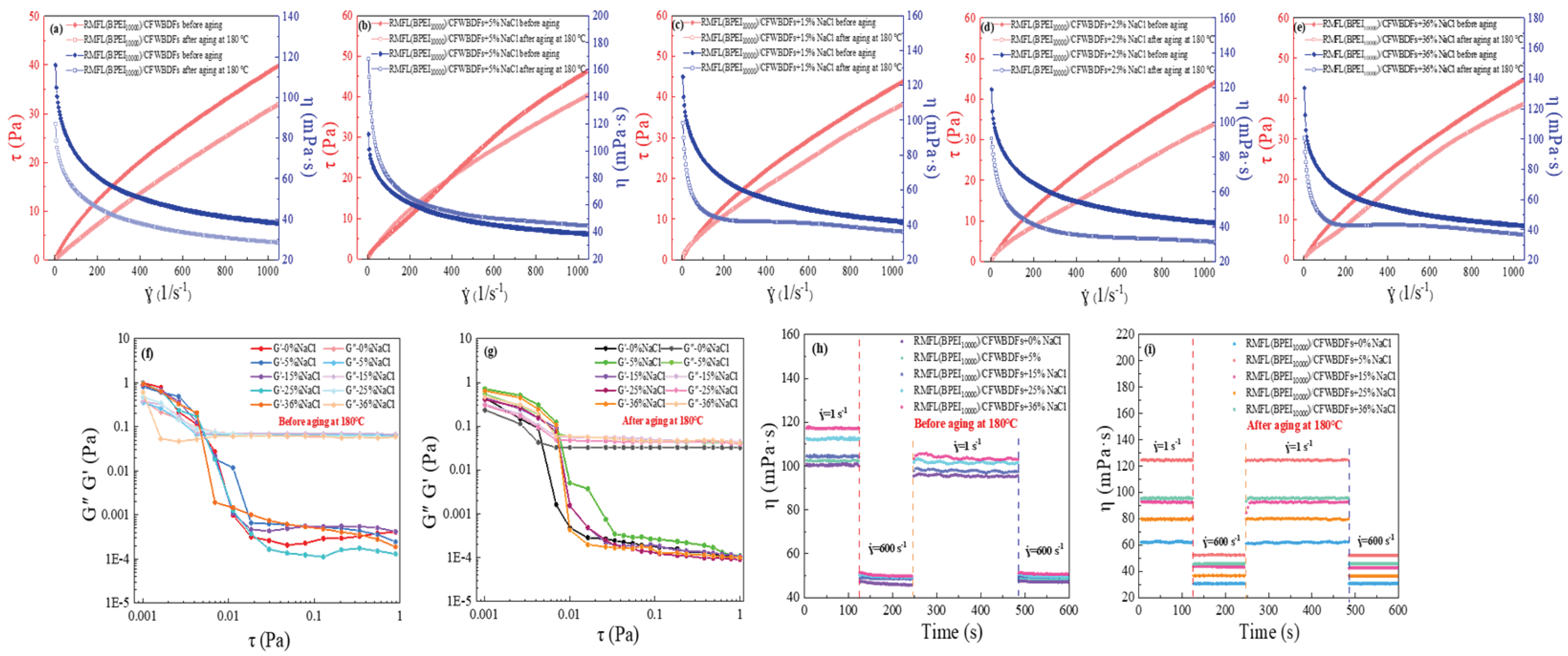
2.5. Rheological Properties Evaluation of RMFL(BPEI10000)/CFWBDFs
The rheological curves of the RMFL(BPEI
10000)/CFWBDFs before and after aging at 180 °C under different NaCl conditions are shown in
Figure 6(a)-(e). With increasing NaCl concentration, the viscosity of the RMFL(BPEI
10000)/CFWBDFs gradually increased before aging, confirming the anti-polyelectrolyte effect of RMFL. After aging at 180 °C, the viscosity of the RMFL(BPEI
10000)/CFWBDFs showed an initial increase, followed by a decrease, then a renewed increase. Under 5% NaCl conditions, the viscosity of the RMFL(BPEI
10000)/CFWBDFs reached its highest within the shear rate range of 0–1050 s⁻¹. As the NaCl concentration increased to 36%, the viscosity of the RMFL(BPEI
10000)/CFWBDFs did not decrease significantly after aging at 180 °C. The results indicated that the RMFL(BPEI
10000)/CFWBDFs exhibited temperature resistance and salt tolerance.
The viscoelastic properties of the RMFL(BPEI
10000)/CFWBDFs are shown in
Figure 6(f)-(g). Within the shear stress range of 0.001 to 0.006 Pa, G’ consistently exceeded G’’, indicating the RMFL(BPEI
10000)/CFWBDFs mainly exhibited elastic properties. When shear stress exceeded 0.07 Pa, G’’ surpasses G’, revealing primarily viscous properties. The results indicated that the RMFL(BPEI
10000)/CFWBDFs was a kind of viscoelastic fluid. Intermolecular forces, such as electrostatic forces and hydrogen bonds, created a network structure between polymer chains. At low shear stress, the network could recover from disturbances, demonstrating elasticity and effectively suspending rock cuttings when static. When the shear stress increased to a certain point, the three-dimensional network experienced irreversible damage. As a result, the RMFL(BPEI
10000)/CFWBDFs transferred from static to flowing, facilitating bottomhole cleaning and enhancing rock fracturing efficiency [
36].
The thixotropy of the RMFL(BPEI
10000)/CFWBDFs was shown in
Figure 6(h)-(i). At a NaCl concentration of 36%, the viscosity of the RMFL(BPEI
10000)/CFWBDFs was 117.07 mPa·s. At the shearing rate of 600 s⁻¹, the viscosity decreased to 51.48 mPa·s. When the shear rate was restored to 1 s⁻¹, the viscosity was 103.33 mPa·s, with a viscosity recovery rate of 88.26%. After aging, the RMFL(BPEI
10000)/CFWBDFs with 36% NaCl exhibited a viscosity of 95.22 mPa·s at 1 s⁻¹. At the shear rate of 600 s⁻¹, the weak gel structure in the solution was disrupted, and the viscosity was reduced to 45.41 mPa·s. After the shearing rate was restored to 1 s⁻¹, the viscosity recovered to 94.34 mPa·s, representing a recovery rate of 99.07%. The results demonstrated that RMFL(BPEI
10000)/CFWBDFs exhibited thixotropy under high-temperature and high-salinity conditions. The internal network was broken at high shearing rates, causing viscosity reduction and enabling drilling fluids to be ejected from the drill bit to break rocks and perform wellbore cleaning. The internal network structure rapidly recovered at low shearing rates, allowing drilling fluids to suspend and transport bit cuttings effectively [
37].
The results demonstrated that RMFL synergized with BPEI10000 to effectively enhance the rheological properties of RMFL(BPEI10000)/CFWBDFs. The RMFL(BPEI10000)/CFWBDFs possessed suitable viscoelasticity, thixotropy, shear-thinning behaviour, and appropriate AV, PV, and YP for RMFL(BPEI10000)/CFWBDFs, ensuring safe and efficient deep well drilling operations.
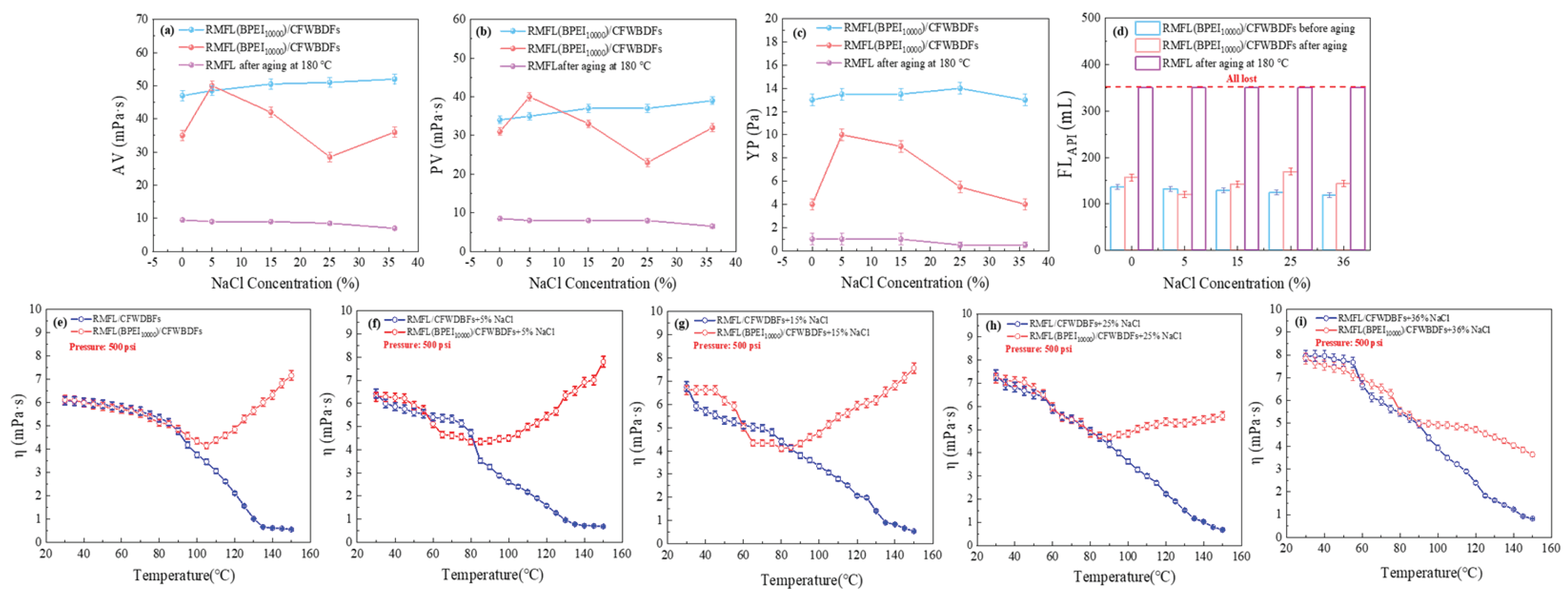
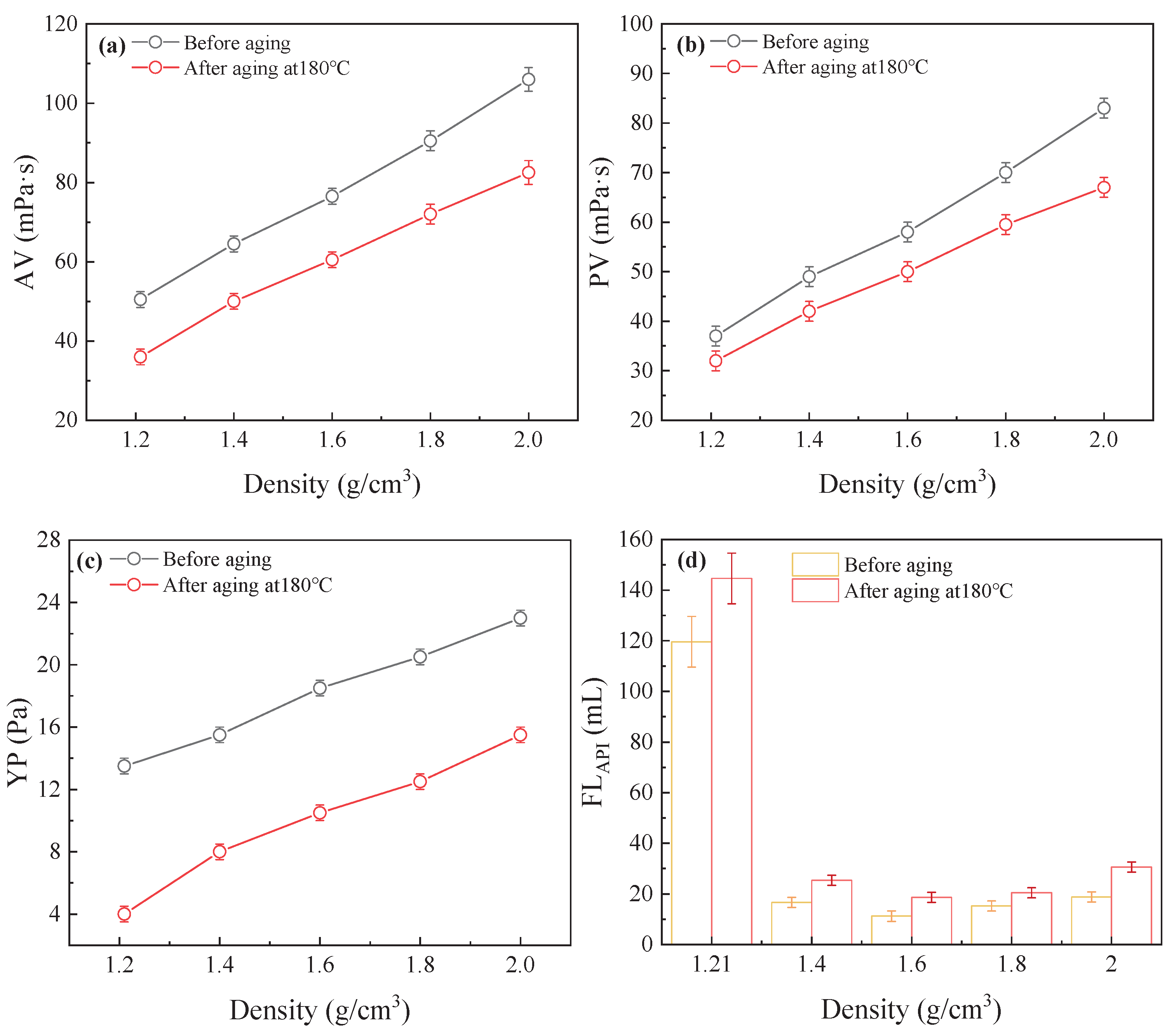
Figure 7(a)-(d) illustrates the rheological properties and filtration loss of the RMFL(BPEI
10000)/CFWBDFs under different NaCl concentrations. As the NaCl concentration increased from 0% to 36%, the PV of the RMFL(BPEI
10000)/CFWBDFs was stable at 37.0 mPa·s before aging. And the PV was 31.0, 40.0, 33.0, 23.0, and 32.0 mPa·s after aging at 180 °C, with PV retention rates exceeding 60%. In contrast, the RMFL/CFWBDFs without BPEI
10000 exhibited PV less than 10.0 mPa·s. The YP of the RMFL(BPEI
10000)/CFWBDFs was approximately 13.5 Pa before aging. The YP was higher than 4.0 Pa after aging. Without BPEI
10000, the YP of the RMFL/CFWBDFs was only 1.0 Pa. In terms of filtration performance, when the NaCl concentration was 5%, 15%, 25%, and 36%, the FL
API of RMFL (BPEI
10000)/CFWBDFs after aging were 156.6, 122.2, 143.0, 170.8 and 144.6 mL. In contrast, in the absence of BPEI
10000, the RMFL/CFWBDFs exhibited complete fluid loss during the API filtration test. The results indicated that BPEI
10000 and RMFL effectively modified the rheological behaviour and filtration performance of RMFL (BPEI
10000)/CFWBDFs.
The viscosity-temperature curves of RMFL(BPEI
10000)/CFWBDFs are shown in
Figure 7(e)-(i). The viscosity of the RMFL/CFWBDFs decreased significantly with temperature increasing from 30 °C to 150 °C. RMFL degraded severely and could not maintain the viscosity of CFWBDFs in the absence of BPEI
10000. After adding BPEI
10000, the viscosity of the RMFL(BPEI
10000)/CFWBDFs first decreased, then increased within the NaCl concentration range of 0% to 25%, with transition temperature points at 115, 118, 128, and 132 °C, respectively. Under 36% conditions, no transition temperature was observed. The viscosity of the RMFL(BPEI
10000)/CFWBDFs gradually decreased. However, due to the crosslinking interaction between BPEI
10000 and RMFL, the viscosity of the RMFL(BPEI
10000)/CFWBDFs remained stable at 3.63 mPa·s at 150 °C and 500 psi, which was increased compared to the RMFL/CFWBDFs without BPEI
10000. As NaCl concentration increased, the crosslinking temperature of BPEI
10000 with RMFL shifted towards higher temperatures. It could result in the charge-shielding effect of Na⁺ on both BPEI
10000 and RMFL, inhibiting the crosslinking reaction between BPEI
10000 and RMFL. However, the fully extended RMFL retained effective crosslinking capability with BPEI
10000, thereby improving the rheological properties of the RMFL(BPEI
10000)/CFWBDFs under high-temperature, high-salinity conditions.
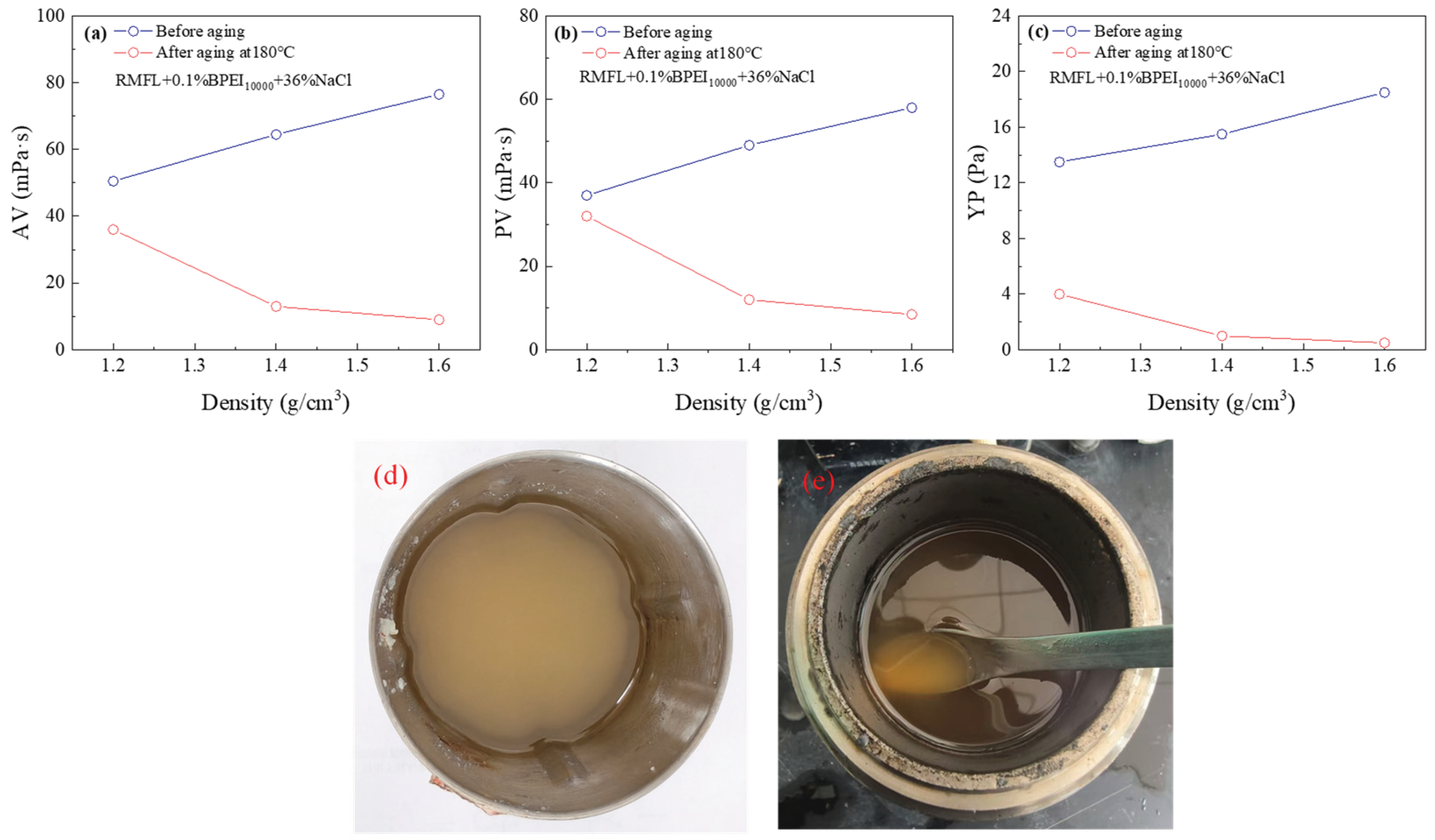
2.6. Evaluation of RMFL(BPEI10000)/CFWBDFs Under High-Density Conditions
The results are shown in
Figure 8. As illustrated in
Figure 8(a)-(c), the AV, PV, and YP of the drilling fluid gradually decreased with increasing density. When the drilling fluid density was 1.40 g/cm³, the PV dropped sharply to 13.0 mPa·s after aging, and the YP was only 1.0 Pa. At a density of 1.60 g/cm³, the PV further decreased to 9.0 mPa·s after aging, with the YP reduced to only 0.5 Pa. It was suggested that the crosslinking efficiency between RMFL and BPEI
10000 was weakened with increasing drilling fluid density, thereby diminishing the rheological enhancement effect of the crosslinking polymer. The phenomenon was mainly due to barite, a large inert solid particle, which created pronounced steric hindrance within the drilling fluids, interfering with effective crosslinking between RMFL and BPEI
10000.
As discussed previously, increasing the molar concentration of BPEI
10000 can enhance the crosslinking efficiency between RMFL and BPEI
10000. Therefore, the RMFL/BPEI
10000 ratio was optimised for high-density drilling fluids, and an appropriate ratio suitable for high-density barite-weighted systems was identified. For RMFL(BPEI
10000)/CFWBDFs with a density of 1.40 g/cm
3, the recommended mass ratio of RMFL to BPEI
10000 was 1:0.10. When the density is increased to 1.60 g/cm³, the recommended mass ratio of RMFL to BPEI
10000 was 1:0.15. For RMFL (BPEI
10000)/CFWBDFs with densities of 1.80 and 2.00 g/cm3, the recommended mass ratios of RMFL to BPEI
10000 were 1:0.25 and 1:0.50. After optimization of the RMFL/BPEI
10000 ratio, the basic properties of RMFL (BPEI
10000)/CFWBDFs under saturated salt conditions before and after ageing at 180 °C are shown in
Figure 9. It could be observed that RMFL (BPEI
10000)/CFWBDFs maintained good rheological properties after aging. In addition, the presence of barite significantly reduced the filtration loss.
Further investigations were conducted to evaluate the performance of RMFL and BPEI
10000 in Ca²-based CFWBDFs. The concentrations of RMFL and BPEI
10000 were fixed at 2.0%, 0.1%, as shown in
Figure 9. From
Figure 9(a), it was shown that the appearance of the CaCl
2 drilling fluid with a density of 1.26 g/cm
3 after aging at 180 °C. Bulk insoluble aggregates were observed. Large insoluble precipitates were observed in all CaCl
2 drilling fluids with densities of 1.35, 1.41, and 1.43 g/cm
3, as shown in
Figure 9(b), (c) and (d). As shown in
Figure 9(e)-(h), the same phenomenon was also observed in CaBr₂ drilling fluids of different densities. With increasing amounts of CaCl
2 and CaBr
2, more precipitates formed, and the viscosity of CFBWDFs decreased to a nearly negligible level. This was because BPEI
10000 not only formed a crosslinked polymer network with RMFL but also chelated Ca
2+. The crosslinked polymer could adsorb a significant amount of Ca
2+. The abundant Ca
2+ could strongly chelate with other crosslinked polymers, leading to extensive polymer adsorption. A large, insoluble, complex precipitate eventually formed.
The unsuitability of the RMFL/BPEI10000 system in Ca2+-based CFWBDFs indicated that the crosslinker must possess specific crosslinking functionality. It should selectively target and react with the intended polymer to form a crosslinked network, rather than reacting with other components in the system. Otherwise, the interactions with other components will undermine the effectiveness of both the polymer and the crosslinker, leading to failure of rheological regulation.
Figure 10.
Compatibility of the RMFL(BPEI10000)/CFWBDFs with Ca2+ and Mg2+. (a)-(d) Compatibility of BPEI10000 with different concentrations of calcium chloride; (e)-(h) Compatibility of branched polyethyleneimine with different concentrations of calcium bromide.
Figure 10.
Compatibility of the RMFL(BPEI10000)/CFWBDFs with Ca2+ and Mg2+. (a)-(d) Compatibility of BPEI10000 with different concentrations of calcium chloride; (e)-(h) Compatibility of branched polyethyleneimine with different concentrations of calcium bromide.
2.7. Mechanism Exploration
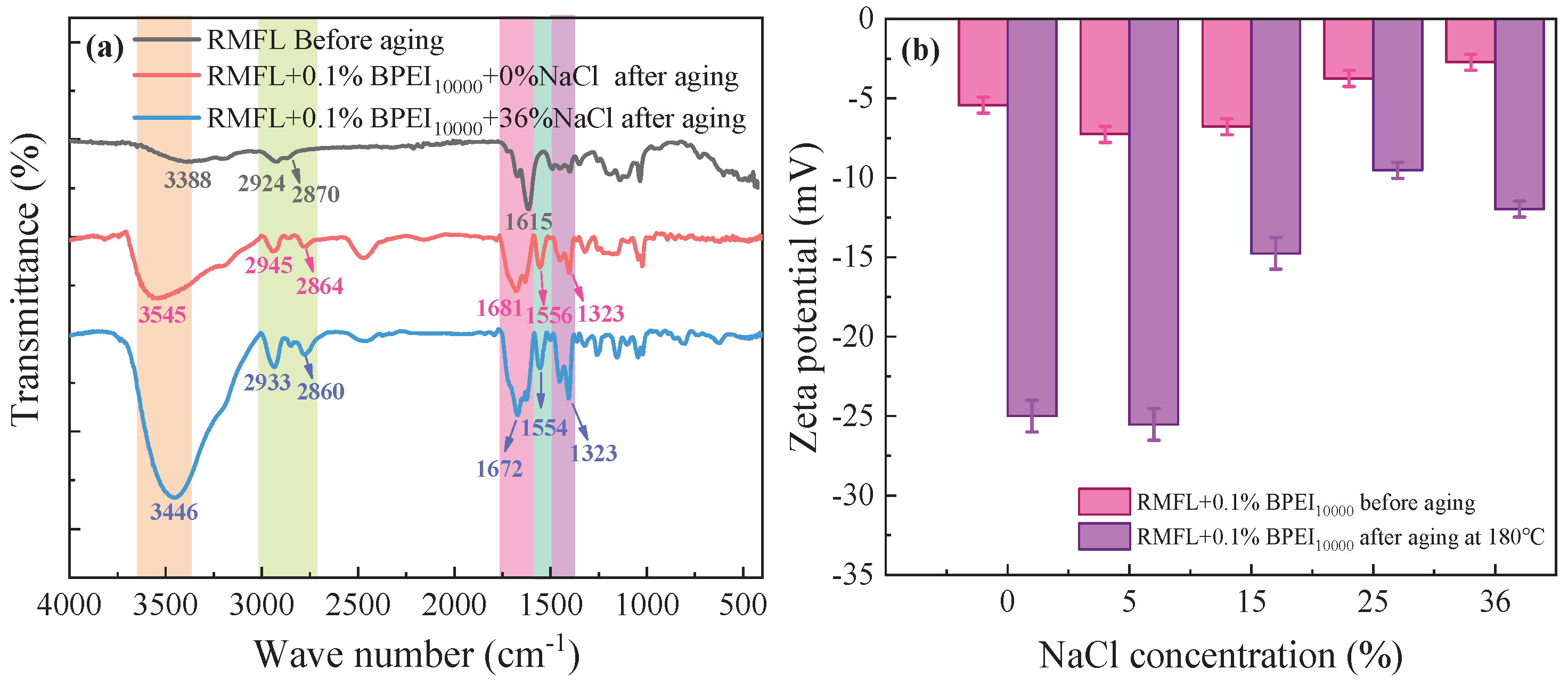
The infrared spectrum of the crosslinked polymer is shown in
Figure 11(a). The broad characteristic peaks at 3545 and 3446 cm⁻¹ should originate from the stretching vibrations of hydroxyl, amine, and amide groups. After RMFL crosslinking with BPEI
10000, the crosslinked polymer contained abundant amine groups. Hydrogen bonding interactions between amine groups, carboxyl, and amide groups gave rise to the broad characteristic peaks observed [
38]. The characteristic peaks at 2924, 2945, and 2933 cm⁻¹ were related to the asymmetric stretching vibrations of the C-H bonds in the methylene groups. The characteristic peaks at 2870, 2864, and 2860 cm⁻¹ came from the symmetric stretching vibrations of the C-H bonds in the methylene groups. The characteristic peak intensity and peak area associated with methylene stretching vibrations in the cross-linked product of RMFL and BPEI
10000 were greater, indicating an increased number of methylene groups. The characteristic peak at 1681 cm⁻¹ corresponded with the stretching vibration of the C=O bond in the amide group, while the peaks at 1723 cm⁻¹ and 1702 cm⁻¹ likely stem from the stretching vibration of the C=O bond in the carboxyl group. The amide groups might have undergone prolonged high-temperature hydrolysis, forming carboxyl groups [
39]. The peaks at 1554 cm⁻¹ and 1323 cm⁻¹ resulted from coupled vibrations of the N-H and C-N bonds in the amine groups. After interaction between RMFL and BPEI
10000, the intensities of these peaks for the N-H and C-N bonds significantly increased. The peak at 1040 cm⁻¹ originated from the stretching vibration of the C-N bond. Its intensity in the cross-linked product was notably enhanced. The crosslinked polymer exhibited detectable C=O, -OH, C-H, C-N, and N-H bonds, suggesting a marked increase in carboxyl, imine, amide, and methylene groups. The results confirmed that BPEI
10000 and RMFL underwent crosslinking under 0% and 36% NaCl conditions [
40]. The zeta potential of the crosslinked polymer was measured to analyse the charge properties of the RMFL/BPEI
10000 crosslinked polymer. As shown in
Figure 11(b), RMFL contained betaine-type zwitterionic structures, with sulfonate groups hydrating to carry a negative charge [
41]. Due to the compressive effect of Na⁺ on the double electric layer of polymers, the Zeta potential of RMFL gradually decreased. At different NaCl concentrations, the RMFL molecular chains carried negative charges before and after aging at 180 °C. It suggested that the crosslinked polymer carried a negative charge, mainly due to carboxyl groups produced by the high-temperature hydrolysis of amide groups. The decrease in zeta potential indicated an increase in negative charge on the crosslinked polymer, which improved the dispersion stability of polymers, nanoparticles, and clay during drilling operations, thereby enhancing the colloidal stability of drilling fluids [
42].
The microstructure of the polymers was shown in
Figure 12(a) and (d). Under 0% NaCl conditions, RMFL exhibited a willow-like morphology. Under 36% NaCl conditions, RMFL exhibited a densely crosslinked three-dimensional network structure. A three-dimensional network structure was observed in the crosslinked weak gel. As shown in
Figure 12(e) and (f), without BPEI
10000 addition, RMFL underwent severe thermal degradation, with polymer chains visibly shortening and diminishing in size. The results demonstrated that RMFL was crosslinked with BPEI
10000 via a transamidation reaction under high-temperature stimulation, forming an extended crosslinking network weak gel. The crosslinking polymer could effectively enhance viscosity, increase shear strength, improve rheological properties, and reduce filtration loss in RMFL(BPEI
10000)/CFWBDFs under high-temperature, high-salinity conditions.
The microstructure of the filter cakes is shown in
Figure 12(g)-(l). Under 0% NaCl conditions, RMFL chains were tightly packed, forming a dense filter cake with fewer apparent pores or cracks. After adding BPEI
10000, the filter cake surface remained flat, smooth, and dense after aging at 180 °C.Under 36% NaCl conditions, the RMFL(BPEI
10000)/CFWBDFs formed compact filter cakes that were smooth and devoid of discernible dehydration pores or fissures before and after aging at 180 °C. In contrast, RMFL experienced severe thermal hydrolysis in the absence of BPEI
10000, resulting in filter cakes with larger micropores and higher filtration losses. The results indicated that RMFL could form a crosslinked weak gel with a three-dimensional network structure synergistic with BPEI
10000. The crosslinking polymer helped form a dense mud cake and effectively reduced filtration loss.