Submitted:
23 February 2026
Posted:
26 February 2026
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Experimental Setup
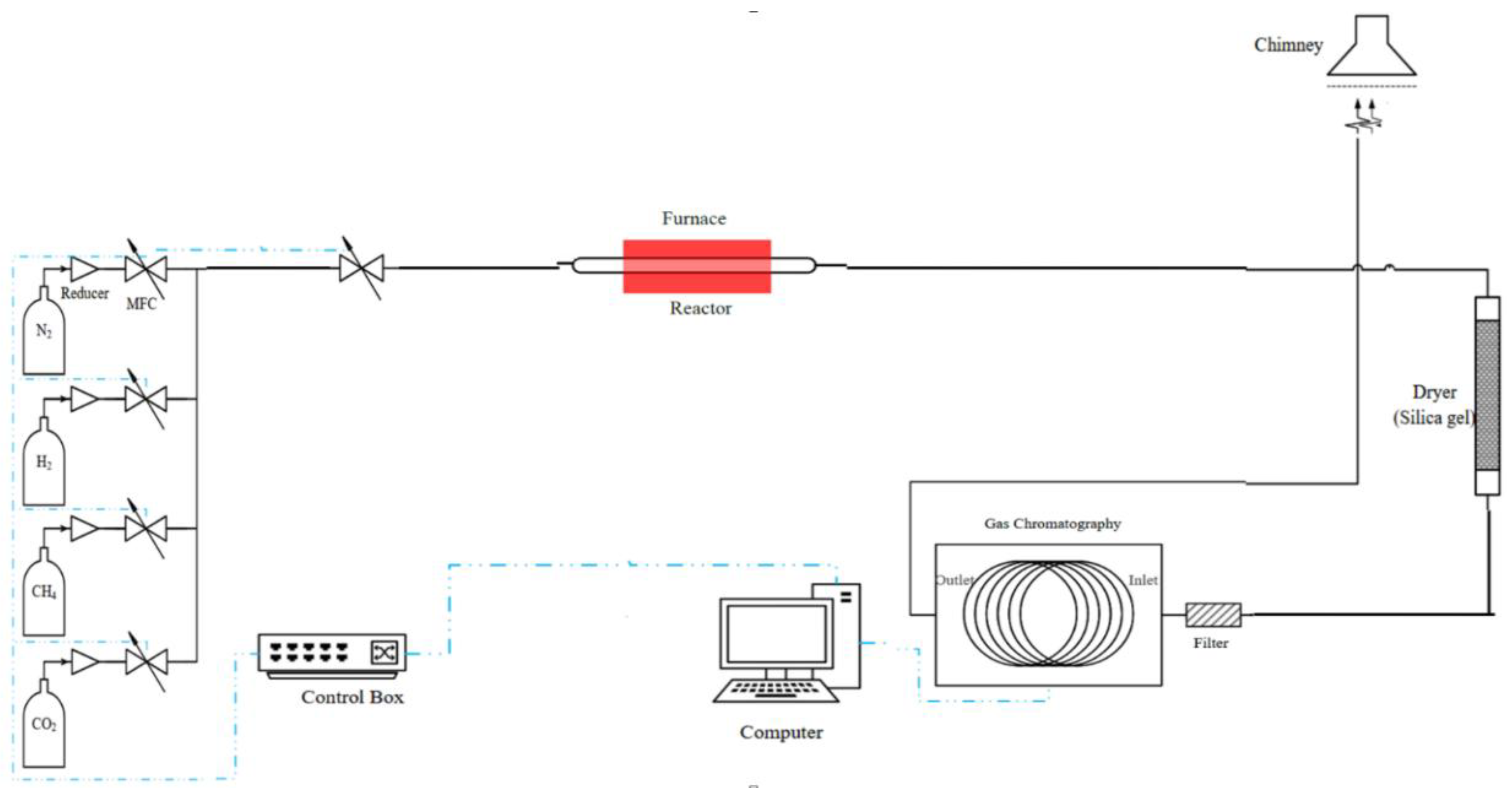
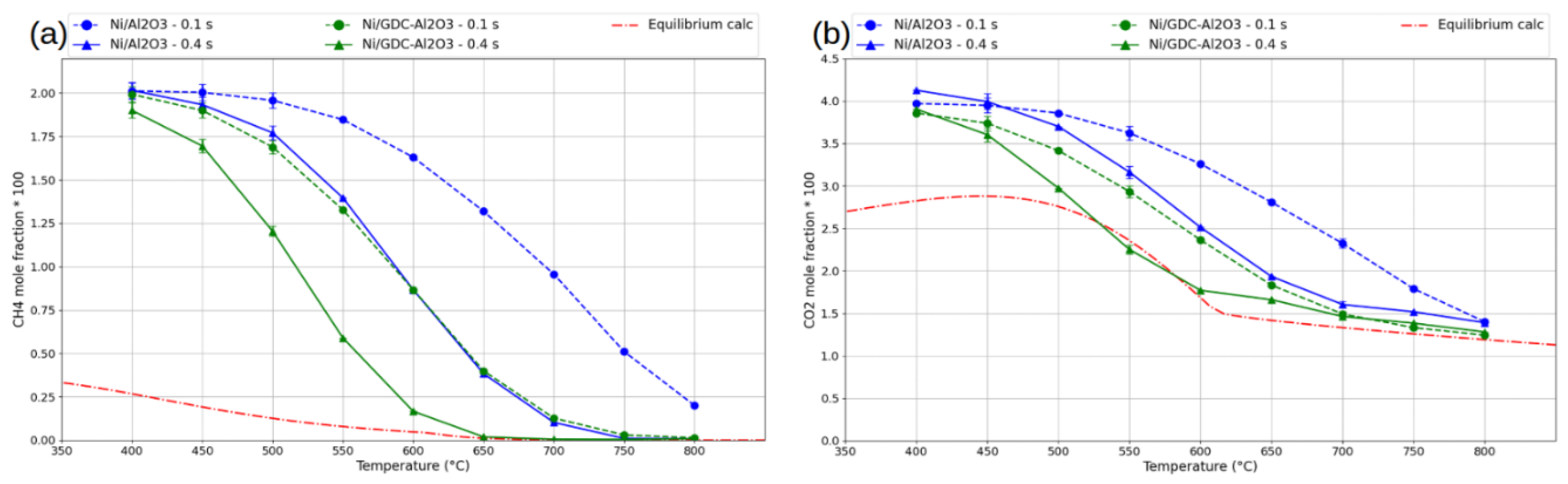
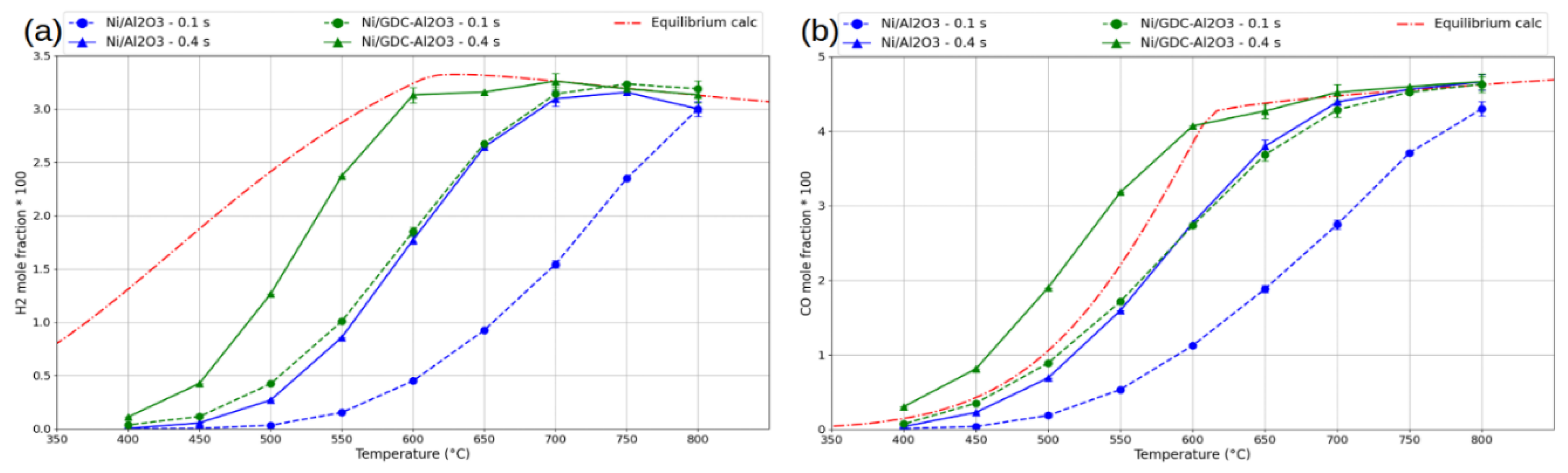
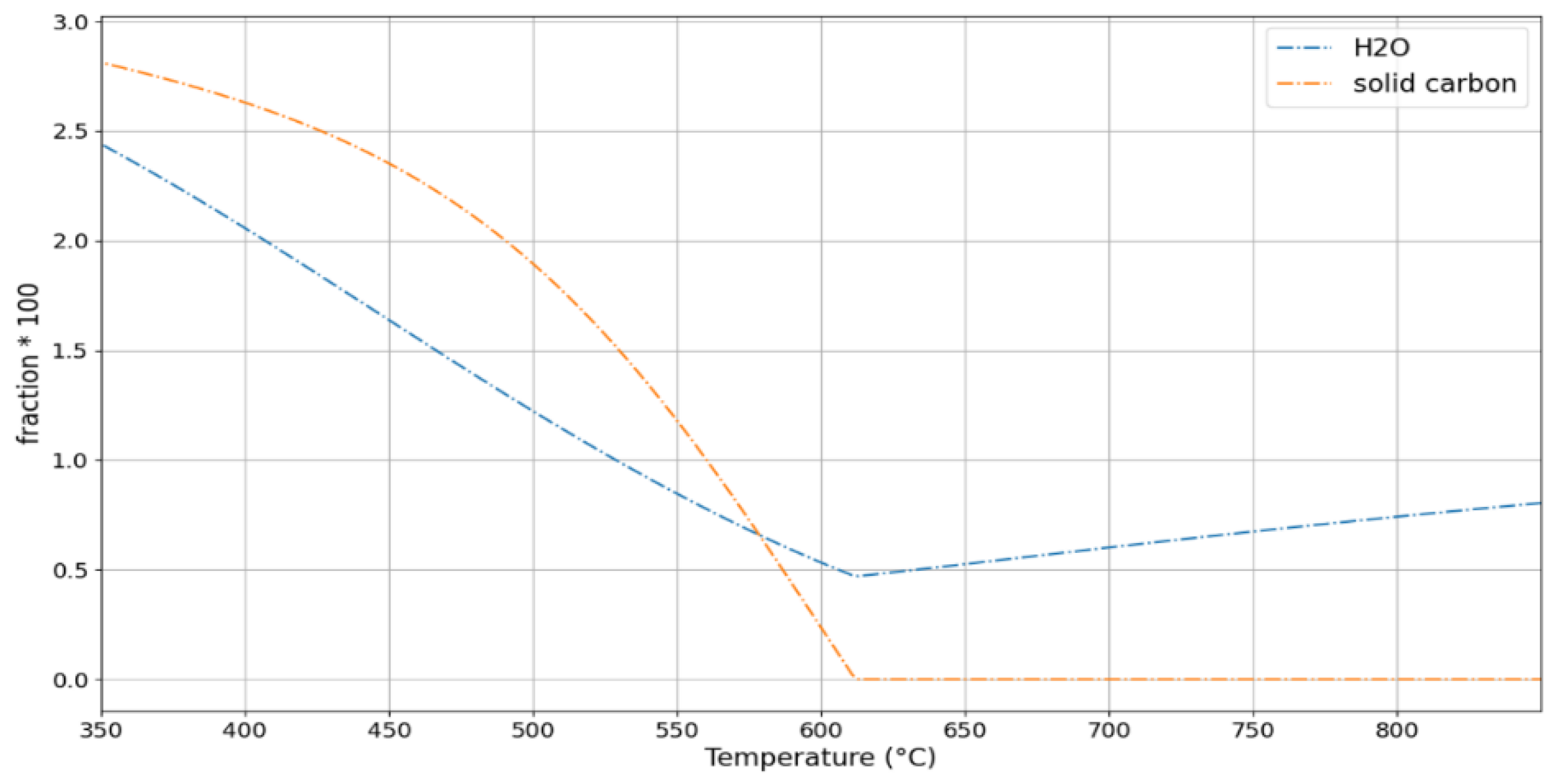
3. Results and Discussion
4. Conclusions
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, Y.; Keoleian, G. A.; Cooper, D. R. “The role of hydrogen in decarbonizing U.S. industry: A review”. Renewable and Sustainable Energy Reviews 2025, 214, 115392. [Google Scholar] [CrossRef]
- Green, D. W.; Southard, M. Z. “Perry’s Chemical Engineers’ Handbook”, 9th ed.; McGraw-Hill Education: New York, 2018. [Google Scholar]
- Pakhare, D.; Spivey, J. A review of dry (CO₂) reforming of methane over noble metal catalysts. Chem. Soc. Rev. 2014, 43, 7813–7837. [Google Scholar] [CrossRef]
- You, J.; Lai, L.; Chen, Y. “Recent Advances in Strong Metal-Support Interaction Engineering for Dry Reforming of Methane Catalysts”; Wiley, 2025. [Google Scholar]
- Seo, H. O. “Recent Scientific Progress on Developing Supported Ni Catalysts for Dry (CO₂) Reforming of Methane”. Catalysts 2018, 8, 110. [Google Scholar] [CrossRef]
- Meloni, E.; Martino, M.; Palma, V. “A Short Review on Ni Based Catalysts and Related Engineering Issues for Methane Steam Reforming”. Catalysts 2020, 10, 352. [Google Scholar] [CrossRef]
- Xu, Y.; Du, X.h.; Li, J.; Wang, P.; Zhu, J.; Ge, F.j.; Zhou, J.; Song, M.; Zhu, W.y. “A comparison of Al₂O₃ and SiO₂ supported Ni-based catalysts in their performance for the dry reforming of methane”. Fuel Process. Technol. 2019, 47, 199–208. [Google Scholar]
- Gao, X.; Ge, Z.; Zhu, G.; Wang, Z.; Ashok, J.; Kawi, S. “Anti-Coking and Anti-Sintering Ni/Al₂O₃ Catalysts in the Dry Reforming of Methane: Recent Progress and Prospects”. Catalysts 2021, 11, 1003. [Google Scholar] [CrossRef]
- Aghamohammadi, S.; Haghighi, M.; Maleki, M.; Rahemi, N. “Sequential impregnation vs. sol–gel synthesized Ni/Al₂O₃–CeO₂ nanocatalyst for dry reforming of methane: Effect of synthesis method and support promotion”. J. Mol. Catal. Chem. 2017, 431, 39–48. [Google Scholar]
- Han, J.; Zhan, Y.; Street, J.; To, F.; Yu, F. “Natural gas reforming of carbon dioxide for syngas over Ni–Ce–Al catalysts”. Int. J. Hydrogen Energy. 2017, 29, 18364–18374. [Google Scholar] [CrossRef]
- Laosiripojana, N.; Sutthisripok, W.; Assabumrungrat, S. “Synthesis gas production from dry reforming of methane over CeO2 doped Ni/Al2O3: Influence of the doping ceria on the resistance toward carbon formation”. Chem. Eng. J. 2005, 112, 13–22. [Google Scholar] [CrossRef]
- Babakouhi, R.; Alavi, S.M.; Rezaei, M.; Jokar, F.; Varbar, M.; Akbari, E. “Hydrogen production through combined dry reforming and partial oxidation of methane over the Ni/Al2O3−CeO2 catalysts”. Int. J. Hydrogen Energy. 2024, 60. [Google Scholar] [CrossRef]
- Marinho, A.L.A.; Toniolo, F.S.; Noronha, F.B.; Epron, F.; Duprez, D.; Bion, N. “Highly active and stable Ni dispersed on mesoporous CeO2−Al2O3 catalysts for production of syngas by dry reforming of methane”. Appl. Catal. B Environ. 2021, 281, 119459. [Google Scholar] [CrossRef]
- Ioannidou, E.; Neophytides, S. G.; Niakolas, D. K. “Au-Mo-Fe-Ni/CeO₂(Gd₂O₃) As Potential Fuel Electrodes For Internal CO₂ Reforming of CH₄ in Single SOFCs”. ECST 2023, 111, 2473–2485. [Google Scholar]
- Gurav, H.R.; Dama, S.; Samuel, V.; Chilukuri, S. “Influence of preparation method on activity and stability of Ni catalysts supported on Gd doped ceria in dry reforming of methane”. J. CO2 Util. 2017, 20, 357–367. [Google Scholar] [CrossRef]
- Unal; Meisuria, S.; Choolaei, M.; Reina, T.R.; Horri, B.A. “Synthesis and characteristics of nanocrystalline Ni1−xCoxO/GDC powder as a methane reforming catalyst for SOFCs”. Ceram. Int. 2018, 44, 6851–6860. [Google Scholar] [CrossRef]
- Wang, W.; Jiang, S.P.; Tok, A.L.Y.; Luo, L. “GDC-impregnated Ni anodes for direct utilization of methane in solid oxide fuel cells”. J. Power Sources 2006, 159, 68–72. [Google Scholar]
- Gurav, H.R.; Dama, S.; Samuel, V.; Chilukuri, S. “Influence of preparation method on activity and stability of Ni catalysts supported on Gd doped ceria in dry reforming of methane”. J. CO2 Util. 2017, 20, 357–367. [Google Scholar] [CrossRef]
- Marinho, A.L.A.; Rabelo-Neto, R.C.; Epron, F.; Toniolo, F.S.; Noronha, F.B.; Bion, N. “Effect of Metal Dopant on the Performance of Ni@CeMeO2 Embedded Catalysts (Me = Gd, Sm and Zr) for Dry Reforming of Methane”. Methane 2022, 1, 300–319. [Google Scholar] [CrossRef]
- AlFatesh, A.S. “Promotional effect of Gd over Ni/Y2O3 catalyst used in dry reforming of CH4 for H2 production”. Int. J. Hydrogen Energy 2017, 42, 18805–18816. [Google Scholar] [CrossRef]
- Zhang, G.X.; Wang, Y.; Li, X.K.; Bai, Y.K.; Zheng, L.; Wu, L.; Han, X.L. “Effect of Gd promoter on the structure and catalytic performance of mesoporous Ni/Al2O3-CeO2 in dry reforming of methane”. Ind. Eng. Chem. Res. 2018. [Google Scholar] [CrossRef]
- Khani, Y.; Bahadoran, F.; Shariatinia, Z.; Varmazyari, M.; Safari, N. “Synthesis of highly efficient and stable Ni/CexZr1−xGdxO4 and Ni/X−Al2O3 (X = Ce, Zr, Gd, Ce-Zr-Gd) nanocatalysts applied in methane reforming reactions”. Ceram. Int. 2020, 46, 25122–25135. [Google Scholar] [CrossRef]
- Fuel Cell Materials, Inc., USA. Available online: https://www.fuelcellmaterials.com.
- MaTecK GmbH, Germany. Available online: https://www.mateck.com.
- Hussien, G. S.; Polychronopoulou, K. “A Review on the Different Aspects and Challenges of the Dry Reforming of Methane (DRM) Reaction”. J. Nanomater. 2022, 12, 3400. [Google Scholar] [CrossRef] [PubMed]
- Kuo, K. K. Principles of Combustion; John Wiley and Sons, 2005. [Google Scholar]
- Smith, G. P.; Golden, D. M.; Frenklach, M.; Moriarty, N. W.; Eiteneer, B.; Goldenberg, M. GRI-Mech 3.0. 2000. Available online: http://combustion.berkeley.edu/gri-mech.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



