Submitted:
19 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Adult Mortality Assay (Flight Cage)
2.2. Prospective Field Efficacy Study: Setting and Case Selection
2.2.1. Data Collection and Quality Assurance
2.2.2. Preliminary Study on the Need of Mechanical Destruction
2.3. Independent Field Validation (Switzerland, 2025) and Insecticide Comparator
2.4. Sample Preparation for Microscopy (Mechanistic Assessment)
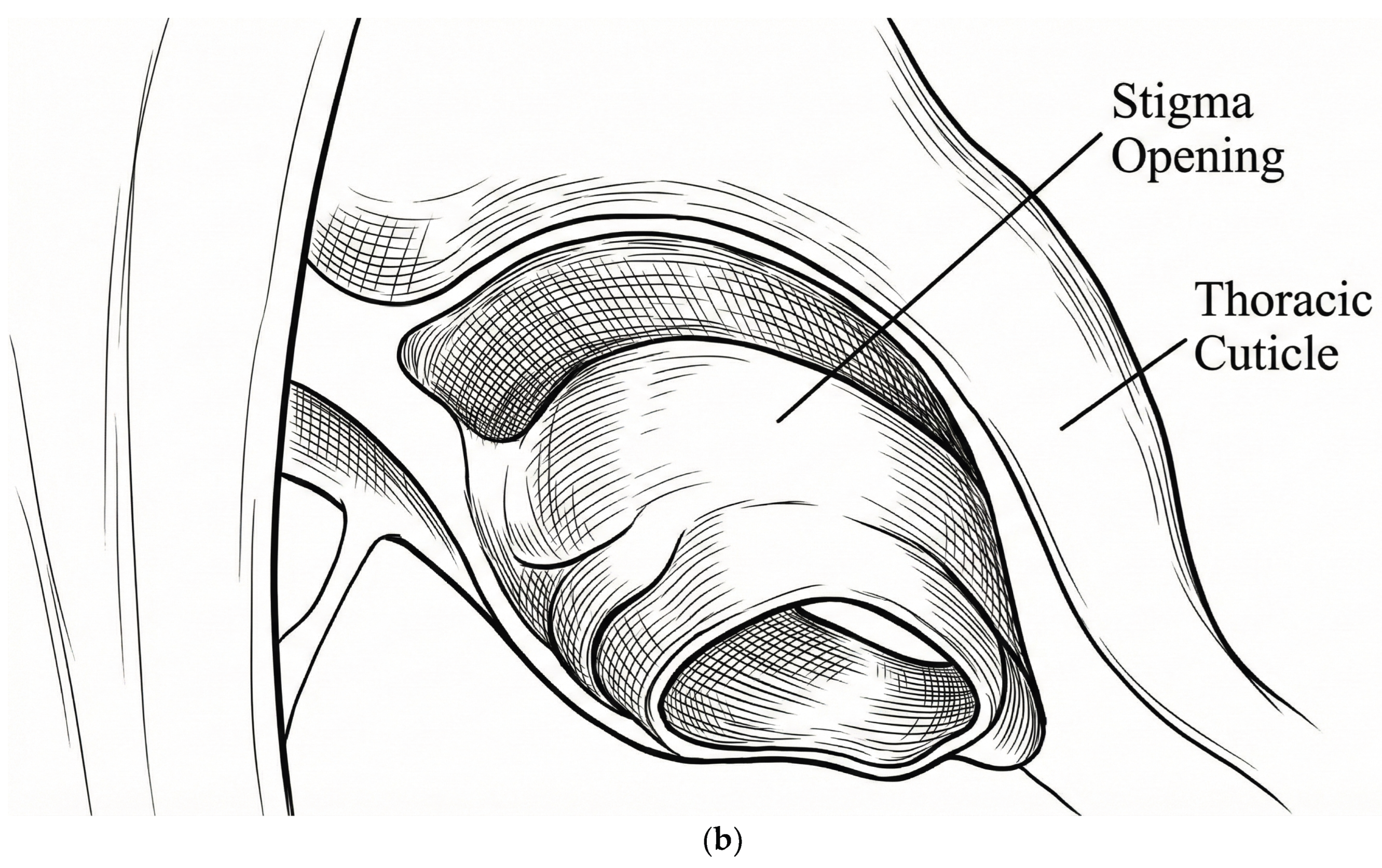
2.4.1. Spiracle Preparations
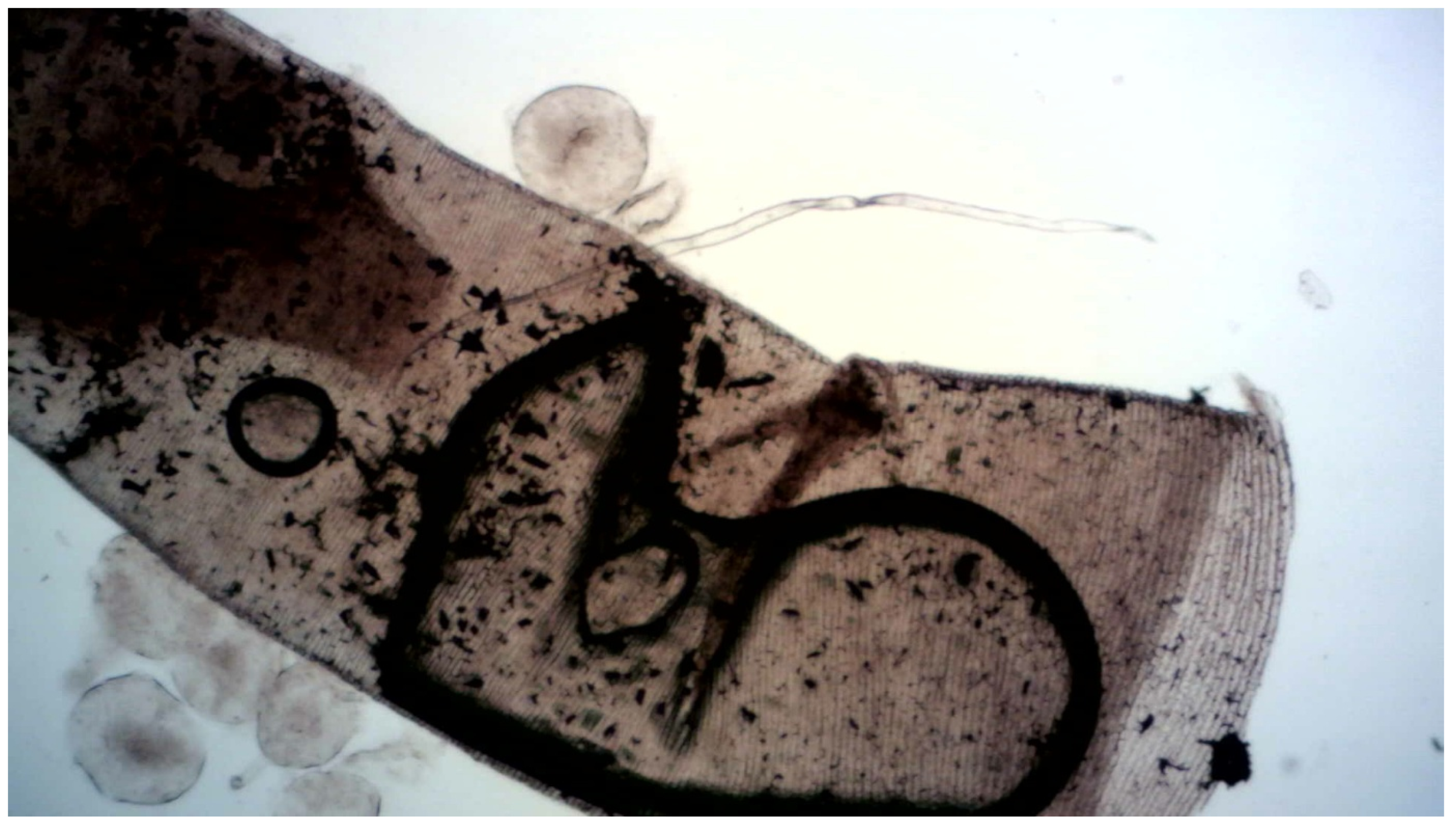
2.4.2. Tracheal Preparations
3. Results
3.1. Adult Mortality Assay (Flight Cage; ~400 Adults)
3.2. Prospective Field Series (Germany 2024; n = 15 Secondary Nests)
3.3. Cross-Site Operational Parameters (Germany 2024 vs Switzerland 2025; Subset Comparison)
3.4. Field Efficacy of Activated Charcoal and Comparison with a Biocide Dust (Day-7 Endpoint)
3.5. Microscopy
4. Discussion
4.1. Principal Findings
4.2. Mode of Action, Welfare Considerations, and Safety
4.3. Mechanistic Evidence from Microscopy
4.4. Operational Feasibility and Cost Considerations
4.5. Contextualization Relative to Existing Approaches
4.7. Implications and Future Directions
- Controlled comparative trials against alternative mineral/physical dusts (e.g., kaolin, zeolites) and, where legally permissible, benchmark insecticides, using standardized time-to-event endpoints.
- Optimization of dose and particle properties (size distribution, surface area, deposition).
- Stratified analyses across season and colony phase (nest stage, colony size, migration status).
- Targeted monitoring of non-target impacts and environmental pathways under operational conditions.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SD | Standard Deviation |
References
- Roy, H. E.; Pauchard, A.; Stoett, P.; Truong, T. Renard. “IPBES Invasive Alien Species Assessment: Full report”. [CrossRef]
- Lioy, S.; Bergamino, C.; Porporato, M. The invasive hornet Vespa velutina: distribution, impacts and management options. In CABI International; 01 Jan 2022. [Google Scholar] [CrossRef]
- Choi, M. B.; Martin, S. J.; Lee, J. W. Distribution, spread, and impact of the invasive hornet Vespa velutina in South Korea. J. Asia. Pac. Entomol. 2012, vol. 15(no. 3), 473–477. [Google Scholar] [CrossRef]
- Kim, J.; Choi, M.; Moon, T. Occurrence of Vespa velutina Lepeletier from Korea, and a revised key for Korean Vespa species (Hymenoptera: Vespidae). Entomol. Res. 2006, vol. 36(no. 2), 112–115. [Google Scholar] [CrossRef]
- Dillane, E.; Hayden, R.; O’Hanlon, A.; Butler, F.; Harrison, S. The first recorded occurrence of the Asian hornet (Vespa velutina) in Ireland, genetic evidence for a continued single invasion across Europe. J. Hymenopt. Res. 2022, vol. 93, 131–138. [Google Scholar] [CrossRef]
- Hoebeke, E. R.; Bartlett, L. J.; Evans, M.; Freeman, B. E.; Wares, J. P. First Records of Vespa velutina (Lepeletier) (Color form Nigrithorax) (Hymenoptera: Vespidae) in North America, an Invasive Pest of Domesticated Honeybees. Proc. Entomol. Soc. Wash. 2024, vol. 126(no. 2), 193–205. [Google Scholar] [CrossRef]
- Vespa velutina nigrithorax. von Kouba, DZ-AL, DZ am 17. August 2025 um 05:13 PM von Kevin Snair · iNaturalist. 21 Dec 2025. Available online: https://www.inaturalist.org/observations/307103550.
- Semmar, S. The First Record of Vespa velutina Lepeletier, 1836 in Algeria and Biocontrol Implications. Commagene J. Biol. 2025, vol. 9(no. 2), 243–244. [Google Scholar] [CrossRef]
- Public urged to look out for hornets in Auckland | NZ Government. 21 Dec 2025. Available online: https://www.mpi.govt.nz/news/media-releases/public-urged-to-look-out-for-hornets-in-auckland.
- Arias, A.-I.; Ferreira-Golpe, M.; Vázquez-González, I. V.; Nave, A.; Thiery, D.; Godinho, J. Economic costs and practices to control Vespa velutina nigrithorax in beekeeping: a survey in four regions in Europe 2023. [CrossRef]
- Requier, F.; Rome, Q.; Villemant, C.; Henry, M. A biodiversity-friendly method to mitigate the invasive Asian hornet’s impact on European honey bees. J. Pest Sci. (2004). 2020, vol. 93(no. 1), 1–9. [Google Scholar] [CrossRef]
- Lueje, Y. R.; Jácome, M. A.; Servia, M. J. New problems for old vineyards: Mitigating the impacts of yellow-legged hornets (Vespa velutina) in a historical wine-producing area. Agric. Ecosyst. Environ. 2024, vol. 367, 108969. [Google Scholar] [CrossRef]
- Rojas-Nossa, S. V.; Calviño-Cancela, M. The invasive hornet Vespa velutina affects pollination of a wild plant through changes in abundance and behaviour of floral visitors. Biol. Invasions 2020, vol. 22(no. 8), 2609–2618. [Google Scholar] [CrossRef]
- Barbet-Massin, M.; Salles, J.-M.; Courchamp, F. The economic cost of control of the invasive yellow-legged Asian hornet. NeoBiota 2020, vol. 55, 11–25. [Google Scholar] [CrossRef]
- Requier, F.; Fournier, A.; Pointeau, S.; Rome, Q.; Courchamp, F. Economic costs of the invasive Yellow-legged hornet on honey bees. Science of The Total Environment 2023, vol. 898, 165576. [Google Scholar] [CrossRef]
- Feás, X. Human Fatalities Caused by Hornet, Wasp and Bee Stings in Spain: Epidemiology at State and Sub-State Level from 1999 to 2018. Biology (Basel). 2021, vol. 10(no. 2), 73. [Google Scholar] [CrossRef]
- Pedersen, S. Broad ecological threats of an invasive hornet revealed through a deep sequencing approach. Science of The Total Environment vol. 970, 178978, 2025. [CrossRef]
- Haubrock, P. J. Economic costs of invasive alien species across europe. NeoBiota 2021, vol. 67, 153–190. [Google Scholar] [CrossRef]
- Lioy, S.; Laurino, D.; Capello, M.; Romano, A.; Manino, A.; Porporato, M. Effectiveness and selectiveness of traps and baits for catching the invasive hornet Vespa velutina. Insects 2020, vol. 11(no. 10), 1–13. [Google Scholar] [CrossRef]
- Teodoro Correia, A. Impact of Vespa velutina. monitoring baited traps on the entomofauna of three protected Portuguese areas. 09 Feb 2026. Available online: https://repositorio.ulisboa.pt/bitstream/10400.5/97363/1/TM_Alexandra_Correia.pdf.
- Pérez-Granados, B. Testing the selectiveness of electric harps: a mitigation method for reducing Asian hornet impact at beehives. J. Apic. Res. 2024, vol. 63(no. 2), 360–366. [Google Scholar] [CrossRef]
- Rojas-Nossa, S. V; Dasilva-Martins, D.; Mato, S.; Bartolomé, C.; Maside, X.; Garrido, J. Effectiveness of electric harps in reducing Vespa velutina predation pressure and consequences for honey bee colony development. Pest Manag. Sci. 2022, vol. 78(no. 12), 5142–5149. [Google Scholar] [CrossRef]
- Rein, Carolin. Bericht über die Versuche zu selektiven Fallen zum Fang der Asiatischen Hornisse (Vespa velutina nigrithorax. im Frühjahr 2025 in Baden-Württemberg, Hohenheim, Aug. 2025. 09 Feb 2026. Available online: https://bienenkunde.uni-hohenheim.de/fileadmin/einrichtungen/bienenkunde/PDF/Vespa_Velutina/Bericht_Selektive_Fallen_Fruehjahr_2025.pdf.
- Rojas-Nossa, S. V.; Mato, S.; Feijoo, P.; Lagoa, A.; Garrido, J. Comparison of Effectiveness and Selectiveness of Baited Traps for the Capture of the Invasive Hornet Vespa velutina. Animals 2024, vol. 14(no. 1). [Google Scholar] [CrossRef]
- Rojas-Nossa, S. V.; Novoa, N.; Serrano, A.; Calviño-Cancela, M. Performance of baited traps used as control tools for the invasive hornet Vespa velutina and their impact on non-target insects. Apidologie 2018, vol. 49(no. 6), 872–885. [Google Scholar] [CrossRef]
- Sánchez, O.; Arias, A. “All That Glitters Is Not Gold: The Other Insects That Fall into the Asian Yellow-Legged Hornet Vespa velutina ‘Specific’ Traps,”. Biology (Basel). 2021, vol. 10(no. 5), 448. [Google Scholar] [CrossRef]
- Monceau, K.; Thiéry, D. Vespa velutina nest distribution at a local scale: An 8-year survey of the invasive honeybee predator. Insect Sci. 2017, vol. 24(no. 4), 663–674. [Google Scholar] [CrossRef]
- Turchi, L.; Derijard, B. Options for the biological and physical control of Vespa velutina nigrithorax (Hym.: Vespidae) in Europe: A review. Journal of Applied Entomology 2018, vol. 142(no. 6), 553–562. [Google Scholar] [CrossRef]
- Commission. REGULATION (EU) No 1143/2014 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 22 October 2014 on the prevention and management of the introduction and spread of invasive alien species, Oct 2014.
- Guedes, R. N. C.; Berenbaum, M. R.; Biondi, A.; Desneux, N. The Side Effects of Pesticides on Nontarget Arthropods. Annu. Rev. Entomol. 2026, vol. 71(no. 1), 381–403. [Google Scholar] [CrossRef]
- Commission. REGULATION (EU) No 528/2012 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL, May 2022.
- Badre, N. H.; Martin, M. E.; Cooper, R. L. The physiological and behavioral effects of carbon dioxide on Drosophila melanogaster larvae. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2005, vol. 140(no. 3), 363–376. [Google Scholar] [CrossRef]
- Ruiz-Cristi, I.; Berville, L.; Darrouzet, E. Characterizing thermal tolerance in the invasive yellow-legged hornet (Vespa velutina nigrithorax): The first step toward a green control method. PLoS One 2020, vol. 15(no. 10), e0239742. [Google Scholar] [CrossRef]
- Gibbons, M. Measuring Pain in Insects. Animal Behaviour and Welfare Cases 2025. [Google Scholar] [CrossRef]
- Gibbons, M.; Crump, A.; Barrett, M.; Sarlak, S.; Birch, J.; Chittka, L. Can insects feel pain? A review of the neural and behavioural evidence. In Advances in Insect Physiology; Academic Press Inc., 2022; vol. 63, pp. 155–229. [Google Scholar] [CrossRef]
- Baua ECHA Hilfen. Available online: https://www.reach-clp-biozid-helpdesk.de/DE/Helpdesk.
- Commission. COMMISSION IMPLEMENTING REGULATION (EU) 2017/ 794 - of 10 May 2017 - approving silicon dioxide Kieselguhr as an existing active substance for use in biocidal products of product-type 18.
- Ogreten, A. Insecticidal efficacy of native raw and commercial diatomaceous earths against Tribolium confusum DuVal (Coleoptera: Tenebrionidae) under different environmental conditions. J. King Saud Univ. Sci. 2023, vol. 35(no. 7), 102827. [Google Scholar] [CrossRef]
- Van Den Noortgate, H.; Lagrain, B.; Pulinthanathu Sree, S.; Kerkhofs, S.; Wenseleers, T.; Martens, J. A. Material properties determining the insecticidal activity of highly divided porous materials on the pharaoh ant (Monomorium pharaonis). Pest Manag. Sci. 2018, vol. 74(no. 6), 1374–1385. [Google Scholar] [CrossRef]
- Alexander, P.; Kitchener, J.; Briscoe, H. Inert dust insecticides: Part I. Mechanism of action. Annals of Applied Biology 2008, vol. 31, 143–149. [Google Scholar] [CrossRef]
- BfR Übersicht zu Biozidprodukten. Available online: https://www.bfr.bund.de/chemikaliensicherheit/biozidprodukte-und-behandelte-waren/.
- Mewis, I.; Ulrichs, C. Action of amorphous diatomaceous earth against different stages of the stored product pests Tribolium confusum, Tenebrio molitor, Sitophilus granarius and Plodia interpunctella. J. Stored Prod. Res. 2001, vol. 37(no. 2), 153–164. [Google Scholar] [CrossRef]
- Schneiderman, H. A. Discontinuous Respiration in Insects: Role of the Spiracles. Biol. Bull. 1960, vol. 119(no. 3), 494–528. [Google Scholar] [CrossRef]
- Chiu, S. F. Toxicity Studies of So-Called ‘Inert’ Materials With the Bean Weevil, Acanthoscelides obtectus (Say). J. Econ. Entomol. 1939, vol. 32(no. 2), 240–248. [Google Scholar] [CrossRef]
- Webb, E. The Penetration of Derris through the Spiracles and Cuticle of Melophagus ovinus, L. Bull. Entomol. Res. 1946, vol. 36(no. 1), 15–22. [Google Scholar] [CrossRef]
- Hochenyos, G. L. Effect of Dusts on the Oriental Roach. J. Econ. Entomol. 1933, vol. 26(no. 4), 792–794. [Google Scholar] [CrossRef]
- Hochenyos, G. L. The Mechanism of Absorption of Sodium Fluoride by Roaches. J. Econ. Entomol. 1933, vol. 26(no. 6), 1162–1169. [Google Scholar] [CrossRef]
- Alexander, P.; Kitchener, J. A.; Briscoe, H. V. A. Inert dust insecticides: Part I. Mechanism of action. Annals of Applied Biology 1944, vol. 31(no. 2), 143–149. [Google Scholar] [CrossRef]
- Wang, Q. Short-term particulate matter contamination severely compromises insect antennal olfactory perception. Nat. Commun. 2023, vol. 14(no. 1), 4112. [Google Scholar] [CrossRef]
- Ebeling, W.; Wagner, R. E. Rapid Desiccation of Drywood Termites with Inert Sorptive Dusts and Other Substances. J. Econ. Entomol. 1959, vol. 52(no. 2), 190–207. [Google Scholar] [CrossRef]
- Van Den Noortgate, H. Material properties determining insecticidal activity of activated carbon on the pharaoh ant (Monomorium pharaonis). J. Pest Sci.. 2019, vol. 92(no. 2), 643–652. [Google Scholar] [CrossRef]
- Zalucki, M.; Furlong, M. Behavior as a mechanism of insecticide resistance: evaluation of the evidence. Curr. Opin. Insect Sci. 2017, vol. 21, 19–25. [Google Scholar] [CrossRef]
- Soderlund, D. M.; Bloomquist, J. R. Molecular Mechanisms of Insecticide Resistance. In in Pesticide Resistance in Arthropods; Springer US: Boston, MA, 1990; pp. 58–96. [Google Scholar] [CrossRef]
- Al Naggar, Y. Mechanisms and Genetic Drivers of Resistance of Insect Pests to Insecticides and Approaches to Its Control. Toxics 2025, vol. 13(no. 8), 681. [Google Scholar] [CrossRef]
- ffrench-Constant, R. H. The Molecular Genetics of Insecticide Resistance. Genetics 2013, vol. 194(no. 4), 807–815. [Google Scholar] [CrossRef]
- Liang, J. Insect Resistance to Insecticides: Causes, Mechanisms, and Exploring Potential Solutions. Arch. Insect Biochem. Physiol. 2025, vol. 118(no. 2). [Google Scholar] [CrossRef]

| Parameter (mean ± SD) | Germany 2024 (n = 15) | Switzerland 2025 (n = 25) |
|---|---|---|
| Nest height (m) | 9.2 ± 3.8 | 18± 8.2 |
| Estimated nest volume (L) | 46 ± 5.8 | 38 ± 7.8 |
| Activated charcoal (g) | 71 ± 12 | 96 ± 14 |
| Dose per volume (g/L) | 1.6 ± 0.3 | 2.6 ± 0.4 |
| Working time (min) | 54 ± 9.1 | 86 ± 48 |
| Treatment / group | Location (year) | Protocol status | n | Inactive at day 7, n (%) | Active/repair at day 7, n (%) | Notes on non-terminal outcomes |
|---|---|---|---|---|---|---|
| Activated charcoal (prospective series) | Germany (2024) | Protocol-adherent (incl. brood removal) | 15 | 15 (100%) | 0 (0.0%) | — |
| Activated charcoal (confirmation series) | Switzerland (2024/2025) | Protocol-adherent (incl. brood removal) | 25 | 25 (100%) | 0 (0.0%) | — |
| Activated charcoal (independent field test) | Switzerland (2025) | Protocol-adherent (incl. brood removal) | 105 | 102 (97.1%) | 3 (2.9%) | Small repair structures |
| Activated charcoal (pooled) | Germany + Switzerland (2024–2025) | Pooled protocol-adherent datasets | 145 | 142 (97.9%) | 3 (2.1%) | Repair and/or persistent activity |
| Activated charcoal (ancillary test) | Switzerland (2025) | Brood removal omitted (protocol deviation) | 3 | 0 (0.0%) | 3 (100%) | Flight activity and repair |
| Biocide dust (0.5% cypermethrin) | Germany (2024) | Operational comparator | 136 | 134 (98.5%) | 2 (1.5%) | Repair and/or emergency nest formation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




