Submitted:
19 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Harvesting method significantly influences both product quality and operational efficiency in medicinal and aromatic plant production. This study comparatively evaluated manual and machine harvesting in Salvia fruticosa (Anatolian sage) cultivated under semi-arid continental conditions in Central Anatolia (Karaman, Türkiye). Plants were harvested at full flowering stage, and assessments included biomass yield, essential oil (EO) content and yield, EO composition, elemental composition, antioxidant activity, harvest losses, and field performance. Manual harvesting resulted in higher fresh and dry biomass yields (6670 and 2440 kg ha⁻¹) compared to machine harvesting (5800 and 2120 kg ha⁻¹). Essential oil content was significantly greater under manual harvesting (2.03%) than machine harvesting (1.57%) Manually harvested samples also showed higher concentrations of Ca, Zn, Cu, and B, whereas antioxidant activity did not differ significantly between treatments. Machine harvesting achieved substantially greater field capacity (0.315 ha h⁻¹) than manual harvesting (0.0114–0.0138 ha h⁻¹) but was associated with higher harvest losses (12.04% vs. 8.40%). Overall, results indicate a trade-off between operational efficiency and quality preservation, highlighting the need to optimize machine harvesting parameters for sustainable large-scale production.
Keywords:
1. Introduction
2. Results and Discussion
2.1. Some Pre- and Post-Harvest Physical Characteristics
2.2. Essential Oil Content(%) and Yield (kg/da)
2.3. Essential Oil Composition (%)
| RT | RI | Compound | Amount% |
|---|---|---|---|
| 8.700 | 972 | α-pinene | 2.77 |
| 10.483 | 1027 | Camphene | 2.57 |
| 12.411 | 1075 | β-pinene | 4.03 |
| 14.939 | 1131 | Myrcene | 2.13 |
| 16.834 | 1170 | Limonene | 1.64 |
| 17.746 | 1188 | Eucalyptol | 43.07 |
| 18.815 | 1206 | γ-terpinene | 0.57 |
| 19.862 | 1218 | o-cymene | 0.29 |
| 20.431 | 1225 | α-terpinolene | 0.48 |
| 26.208 | 1294 | Thujone | 2.83 |
| 26.934 | 1303 | 4(10)-Thujen-3-ol | 3.70 |
| 27.695 | 1429 | trans-β-Terpineol | 0.42 |
| 29.861 | 1490 | Camphor | 20.12 |
| 30.329 | 1504 | Linalool | 0.55 |
| 30.614 | 1514 | Linalyl acetate | 0.88 |
| 31.656 | 1547 | Bornyl acetate | 1.68 |
| 32.247 | 1565 | Caryophyllene | 2.54 |
| 32.299 | 1567 | Terpinen-4-ol | 0.54 |
| 34.425 | 1634 | Humulene | 1.16 |
| 35.124 | 1656 | (+)-4-Carene | 1.25 |
| 35.368 | 1664 | Endo-Borneol | 0.99 |
| 36.657 | 1704 | Geranyl acetate | 0.14 |
| 39.212 | 1785 | Geraniol | 0.11 |
| 43.266 | 1914 | Caryophyllene oxide | 0.4 |
| 45.613 | 1987 | Viridiflorol | 0.85 |
| 47.128 | 2036 | Thymol | 0.03 |
| 47.341 | 1501 | Eugenol | 0.02 |
| TOTAL | 95.76 | ||
| RT | RI | Compound | Amount% |
|---|---|---|---|
| 8.680 | 972 | α-pinene | 2.50 |
| 8.764 | 976 | α-thujene | 0.45 |
| 10.461 | 1026 | Camphene | 2.50 |
| 12.354 | 1073 | β-pinene | 2.69 |
| 14.896 | 1078 | β-myrcene | 1.35 |
| 15.678 | 1146 | α-terpinolene | 0.43 |
| 16.693 | 1166 | D-Limonene | 2.20 |
| 17.548 | 1184 | Eucalyptol | 21.77 |
| 18.778 | 1205 | γ-terpinene | 0.85 |
| 19.876 | 1218 | p-cymene | 1.75 |
| 26.422 | 1296 | Thujone | 19.94 |
| 26.966 | 1391 | cis-sabinol | 3.94 |
| 29.82 | 1489 | Camphor | 16.12 |
| 30.308 | 1504 | Linalool | 0.33 |
| 30.614 | 1513 | Linalyl acetate | 0.88 |
| 31.629 | 1545 | Bornyl acetate | 1.3 |
| 32.27 | 1566 | Caryophyllene | 5.22 |
| 32.492 | 1573 | Aromadendrene | 0.12 |
| 34.436 | 1634 | Humulene | 1.36 |
| 35.124 | 1656 | (+)-4-Carene | 0.29 |
| 35.358 | 1663 | Endo-Borneol | 0.96 |
| 36.648 | 1704 | Geranyl acetate | 0.05 |
| 39.206 | 1785 | Geraniol | 0.06 |
| 43.299 | 1914 | Caryophyllene oxide | 1.5 |
| 45.699 | 1990 | Viridiflorol | 5.84 |
| 47.128 | 2035 | Thymol | 0.13 |
| 47.344 | 2042 | Eugenol | 0.04 |
| TOTAL | 94.57 | ||
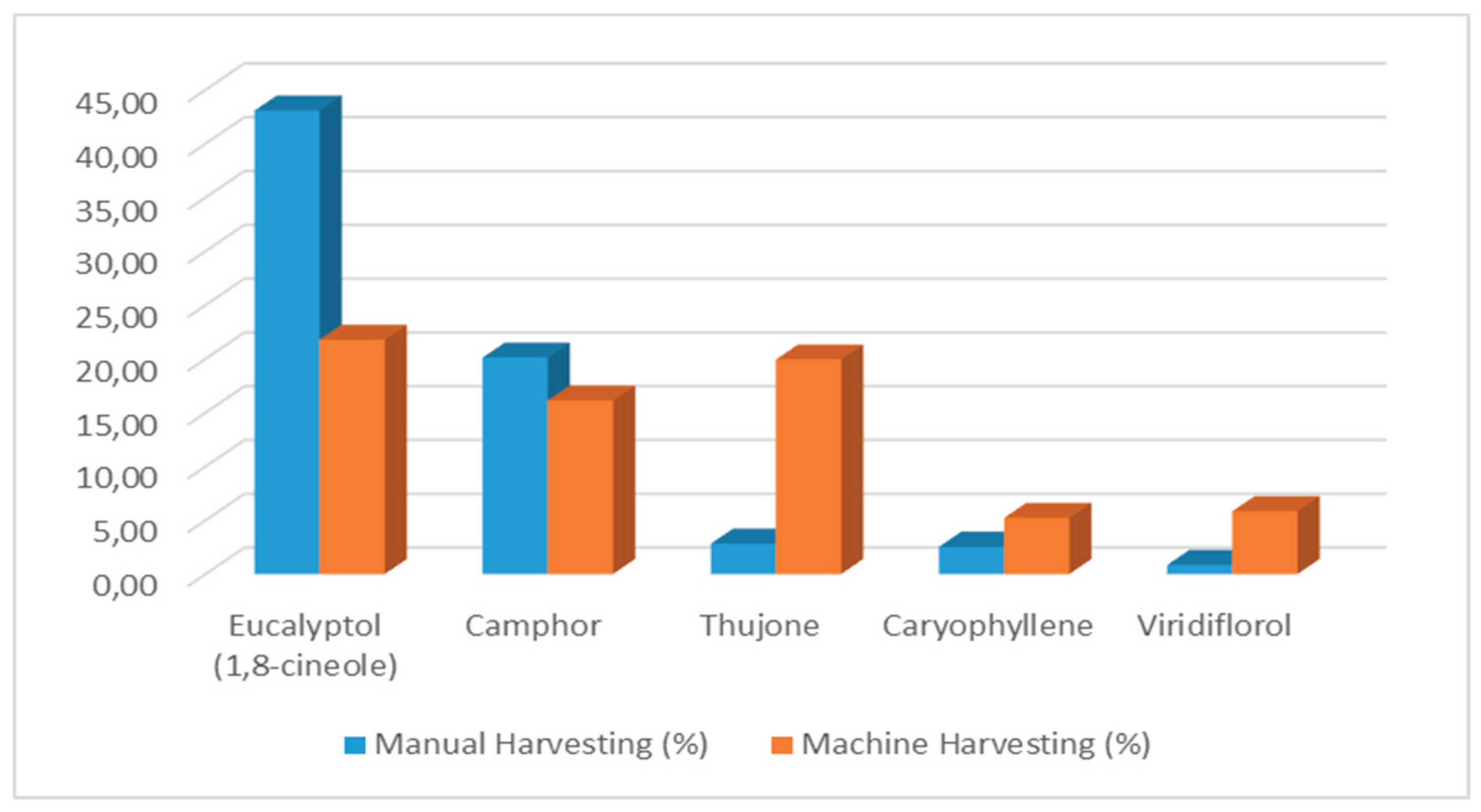
2.4. Macro and Micro Elements (ppm)
| Harvesting Methods | Macro Elements | ||||
|---|---|---|---|---|---|
| P (ppm) | K (ppm) | Ca (ppm) | Mg (ppm) | S (ppm) | |
| Manual Harvesting | 1683.13±2.45 | 11350.17±5.73 | 32564.92±57.72 | 4960.92±4.26 | 2838.67±11.80 |
| Machine Harvesting | 1560.66±1.33 | 10640.23±28.93 | 12573.18±32.27 | 5894.43±11.11 | 1845.32±2.27 |
| Harvesting Methods | Micro Elements | ||||
|---|---|---|---|---|---|
| Fe (ppm) | Zn (ppm) | Cu (ppm) | Mn (ppm) | B (ppm) | |
| Manual Harvesting | 840.28±2.06 | 15.12±0.54 | 6.69±0.11 | 30.98±0.10 | 40.58±0.09 |
| Machine Harvesting | 805.52±3.02 | 9.53±0.09 | 4.72±0.09 | 31.71±0.36 | 33.20±0.63 |
2.5. Antioxidant Activity
| Harvesting Methods | Antioxidant Activity (mg TE mL⁻¹) |
|---|---|
| Manual Harvesting | 0,093±0,001 |
| Machine Harvesting | 0,096±0,001 |
2.6. Some Operational Characteristics of Machine and Manual Harvesting
3. Materials and Methods
3.1. Trial Area and Plant Material
3.2. Harvesting Methods
- Machine harvesting
- Manual harvesting
3.3. Determination of Some Pre- and Post-Harvest Physical Characteristics
3.4. Determination of Essential Oil Content and Yield
3.5. Essential Oil Composition
3.6. Determination of Macro and Micro Elements
3.7. Determination of Antioxidant Activity
- Preparation of Plant Extracts
- Antioxidant Activity
3.8. Determination of Some Operational Characteristics of Machine and Manual Harvesting
- Sa= 0,1.B.V. K
- Sa: Machine field capacity (ha h⁻¹)
- B: Working width (m)
- V: Forward speed of the machine (km h⁻¹)
- K: Field efficiency coefficient (%)
- Labor (worker) field capacity: During manual harvesting, the time required for a single worker to harvest one plant and a defined group of plants within a specified distance was recorded. Harvest duration was determined accordingly, and the harvested area within that time was converted to a unit area basis (ha).
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EO | Essential Oil |
| GC–MS | Gas Chromatography–Mass Spectrometry |
| ICP–MS | Inductively Coupled Plasma–Mass Spectrometry |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| TE | Trolox Equivalent |
| Ha | hectare |
References
- Başer, K.H.C.; Buchbauer, G. Handbook of Essential Oils: Science, Technology, and Applications; CRC Press: London, UK, 2015.
- Perfumi, M.; Arnold, N.; Tacconi, R. Hypoglycemic activity of Salvia fruticosa Mill. from Cyprus. J. Ethnopharmacol. 1991, 34, 135–140. [CrossRef]
- Badalamenti, N.; Salbitani, G.; Cianciullo, P.; Bossa, R.; De Ruberto, F.; Greco, V.; Basile, A.; Maresca, V.; Bruno, M.; Carfagna, S. Chemical composition of Salvia fruticosa Mill. essential oil and its protective effects against heavy metal-induced oxidative stress. Antioxidants 2023, 12, 1990. [CrossRef]
- Davis, P.H. Flora of Turkey and the East Aegean Islands, Vol. 7; Edinburgh University Press: Edinburgh, UK, 1982; pp. 400–439.
- Vokou, D.; Kokkini, S.; Bessiere, J.-M. Geographic variation of Greek oregano essential oils. Biochem. Syst. Ecol. 1993, 21, 287–295. [CrossRef]
- Öztekin, S.; Martinov, M. Medicinal and Aromatic Crops: Harvesting, Drying and Processing; Haworth Press: New York, NY, USA, 2007.
- Comparetti, A.; Vallone, M.; Catania, P.; Schillaci, G.; Rizzo, G.; Tornese, F. Comparison of mechanical, assisted and manual harvesting of Origanum vulgare L. Sustainability 2022, 14, 2562. [CrossRef]
- Malik, S.; Sharma, K.; Kanaujia, A. Harvest and post-harvest management for ensuring quality of medicinal plants. Int. J. Adv. Res. 2021, 9, 602–606. [CrossRef]
- Zuazo, V.H.D.; Cárceles, B.; Gálvez Ruiz, B.; Cermeño Sacristán, P. Response of essential oil yield of aromatic and medicinal plants to harvesting strategies. Comunicata Sci. 2020, 10, 429–437. [CrossRef]
- Vinogradova, N.; Vinogradova, E.; Chaplygin, V.; Mandzhieva, S.; Kumar, P.; Rajput, V.D.; Minkina, T.; Seth, C.S.; Burachevskaya, M.; Lysenko, D.; Singh, R.K. Phenolic compounds of medicinal plants in anthropogenically transformed environments. Plants 2023, 12, 3133.
- Meier, U.; Bleiholder, H.; Buhr, L.; Feller, C.; Hack, H.; Lancashire, P.D.; Schnock, U.; Stauß, R.; van den Boom, T.; Weber, E.; Ebbinghaus, R.; Zwerger, P. The BBCH system to coding the phenological growth stages of plants. J. Kulturpflanzen 2009, 61, 41–52.
- Al-Sammarraie, M.A.J. Diskli tip silaj makinelerinde bıçak–karşı bıçak açıklığının makine performansına etkisi. Master’s Thesis, Selçuk University, Konya, Türkiye, 2019.
- UPOV. Guidelines for the Conduct of Tests for Distinctness, Uniformity and Stability; UPOV: Geneva, Switzerland, 2005.
- AOAC. Official Methods of Analysis, 21st ed.; AOAC International: Gaithersburg, MD, USA, 2019.
- European Directorate for the Quality of Medicines & HealthCare. European Pharmacopoeia, 10th ed.; Council of Europe: Strasbourg, France, 2019.
- NMKL. Determination of trace elements in food by ICP-MS after pressure digestion. NMKL Method No. 186; Nordic Committee on Food Analysis: Oslo, Norway, 2007.
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [CrossRef]
- Vokou, D., Kokkini, S., & Bessiere, J.-M. (1993). Geographic variation of Greek oregano (Origanum vulgare ssp. hirtum) essential oils. Biochemical Systematics and Ecology, 21(2), 287–295. [CrossRef]
- Leontaritou, P.; Lamari, F.N.; Papasotiropoulos, V.; Iatrou, G. Morphological, genetic and essential oil variation of Greek sage populations. Ind. Crops Prod. 2020, 150, 112346. [CrossRef]
- Skoula, M.; Abbes, J.E.; Johnson, C.B. Genetic variation of volatiles and rosmarinic acid in Salvia fruticosa. Biochem. Syst. Ecol. 2000, 28, 551–561. [CrossRef]
- Karioti, A.; Vokou, D.; Skoula, M.; Demetzos, C. Composition of essential oils of Salvia fruticosa. J. Agric. Food Chem. 2003, 51, 6505–6510.
- Türkmen, M. Chemical composition of essential oils of Salvia spp. leaves. Bangladesh J. Bot. 2021, 50, 1069–1076. [CrossRef]
- Wasternack, C.; Hause, B. Jasmonates in plant stress responses. Ann. Bot. 2013, 111, 1021–1058.
- Jacobo-Velázquez, D.A.; Cisneros-Zevallos, L. Stressed plants as biofactories of phenolic compounds. Agriculture 2012, 2, 259–271.
- Ghorbanpour, M.; Varma, A., Eds. Medicinal Plants and Environmental Challenges; Springer: Cham, Switzerland, 2017.
- Blande, J.D.; Holopainen, J.K.; Niinemets, Ü. Plant volatiles in polluted atmospheres. Plant Cell Environ. 2014, 37, 1892–1904. [CrossRef]
- Loreto, F.; Schnitzler, J.-P. Abiotic stresses and induced BVOCs. Trends Plant Sci. 2010, 15, 154–166. [CrossRef]
- Marschner, P. Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Academic Press: London, UK, 2011.
- Broadley, M.; Brown, P.; Cakmak, I.; Ma, J.F.; Rengel, Z.; Zhao, F. Beneficial elements. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Academic Press: London, UK, 2011; pp. 249–269.
- White, P.J.; Brown, P.H. Plant nutrition for sustainable development. Ann. Bot. 2010, 105, 1073–1080. [CrossRef]
- Maathuis, F.J.M. Physiological functions of mineral macronutrients. Curr. Opin. Plant Biol. 2009, 12, 250–258. [CrossRef]
- Hänsch, R.; Mendel, R.R. Physiological functions of mineral micronutrients. Curr. Opin. Plant Biol. 2009, 12, 259–266. [CrossRef]
- Dinçer, C.; Topuz, A.; Özdemir, K.S.; Şahin-Nadeem, H.; Çam, İ.B.; Tontul, İ.; Göktürk, R.S.; Tuğrul Ay, S. Phenolic composition, antioxidant activity and essential oil content of wild and cultivated sage. Ind. Crops Prod. 2012. [CrossRef]
- Ververis, A.; Kyriakou, S.; Ioannou, K.; Chatzopoulou, P.S.; Panayiotidis, M.I.; Plioukas, M.; Christodoulou, K. Chemical profiling and antioxidant capacities of Salvia fruticosa extracts. Plants 2023, 12, 3191.
- Jacobo-Velázquez, D. A., & Cisneros-Zevallos, L. (2012). An alternative use of horticultural crops: Stressed plants as biofactories of bioactive phenolic compounds. Agriculture, 2(3), 259–271. [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410.
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [CrossRef]
- Skotti, E.; Anastasaki, E.; Kanellou, G.; Polissiou, M.; Tarantilis, P.A. Total phenolic content and antioxidant activity of Greek MAPs. Ind. Crops Prod. 2014, 53, 46–54.
- Lamaison, J.L.; Petitjean-Freytet, C.; Carnat, A. Medicinal Lamiaceae with antioxidant properties. Pharm. Acta Helv. 1991, 66, 185–188.
- ASABE. ASAE D497.7: Agricultural Machinery Management Data; American Society of Agricultural and Biological Engineers: St. Joseph, MI, USA, 2011.
- Pingali, P.L. Agricultural mechanization: Adoption patterns and economic impact. In Handbook of Agricultural Economics; Elsevier: Amsterdam, The Netherlands, 2007; Vol. 3, pp. 2779–2805. [CrossRef]

| Harvesting Methods | Essential Oil Content (%) | Essential Oil Yield (L ha⁻¹) |
|---|---|---|
| Manual Harvesting | 2.03±0.15 | 49.5±0.37 |
| Machine Harvesting | 1.57±0.22 | 33.3±0.56 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




