Submitted:
24 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Subjects
| Scleral lens | Soft lens | |
|---|---|---|
| Contact lens brand | Comfort Optimum Extra | Acuvue Oasys 1 Day |
| Material | Roflufocon D | Senofilcon A |
| Refractive index | 1.43 | 1.40 |
| Dk value (Barrer) | 100 | 103 |
| Diameter (mm) | 16.5 | 14.3 |
2.2. Measurements and Data Collection Procedures
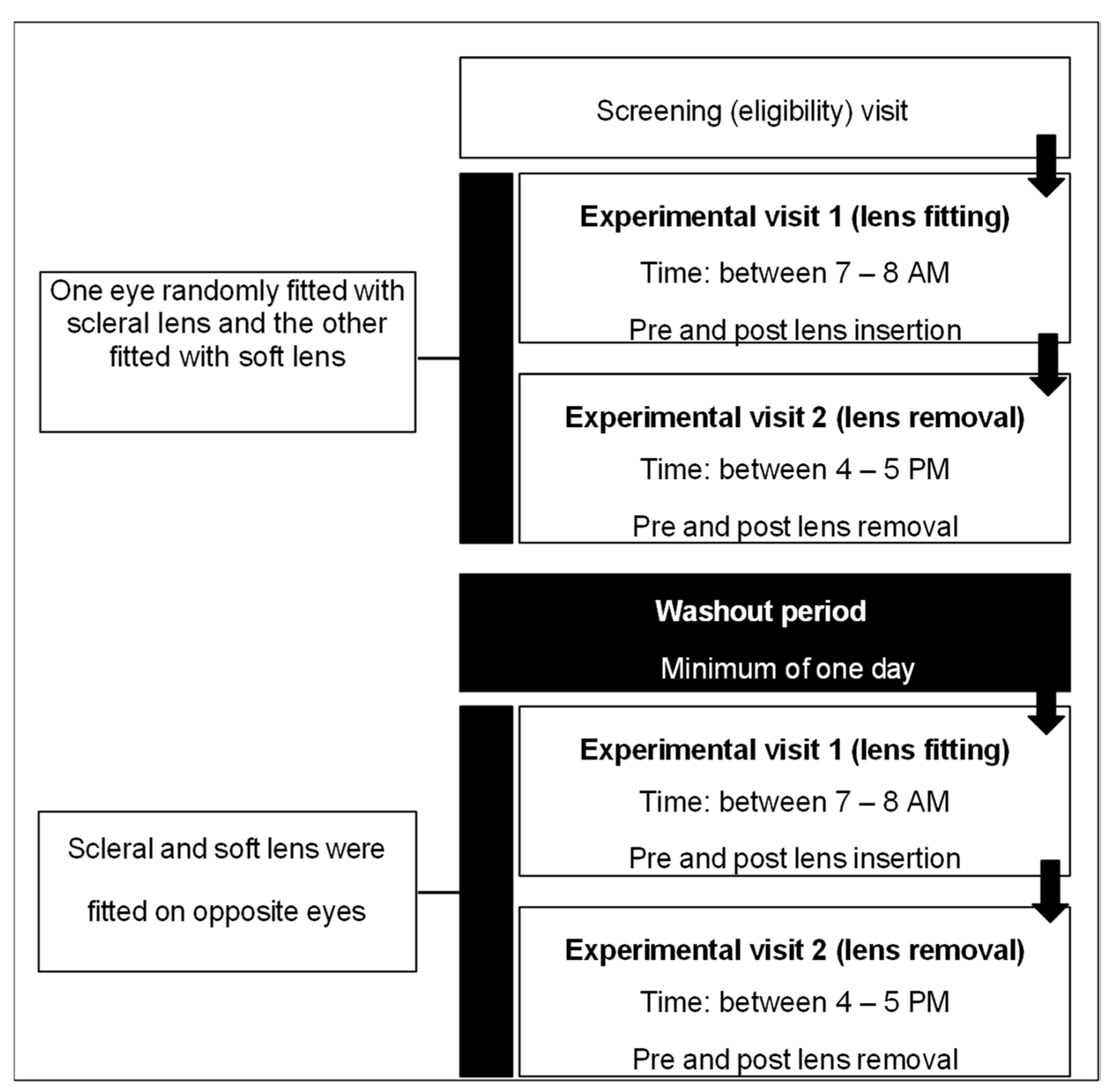
2.3. Randomization and Crossover Protocol
2.4. Statistical Analysis
3. Results
3.1. Demographics and Clinical Characteristics
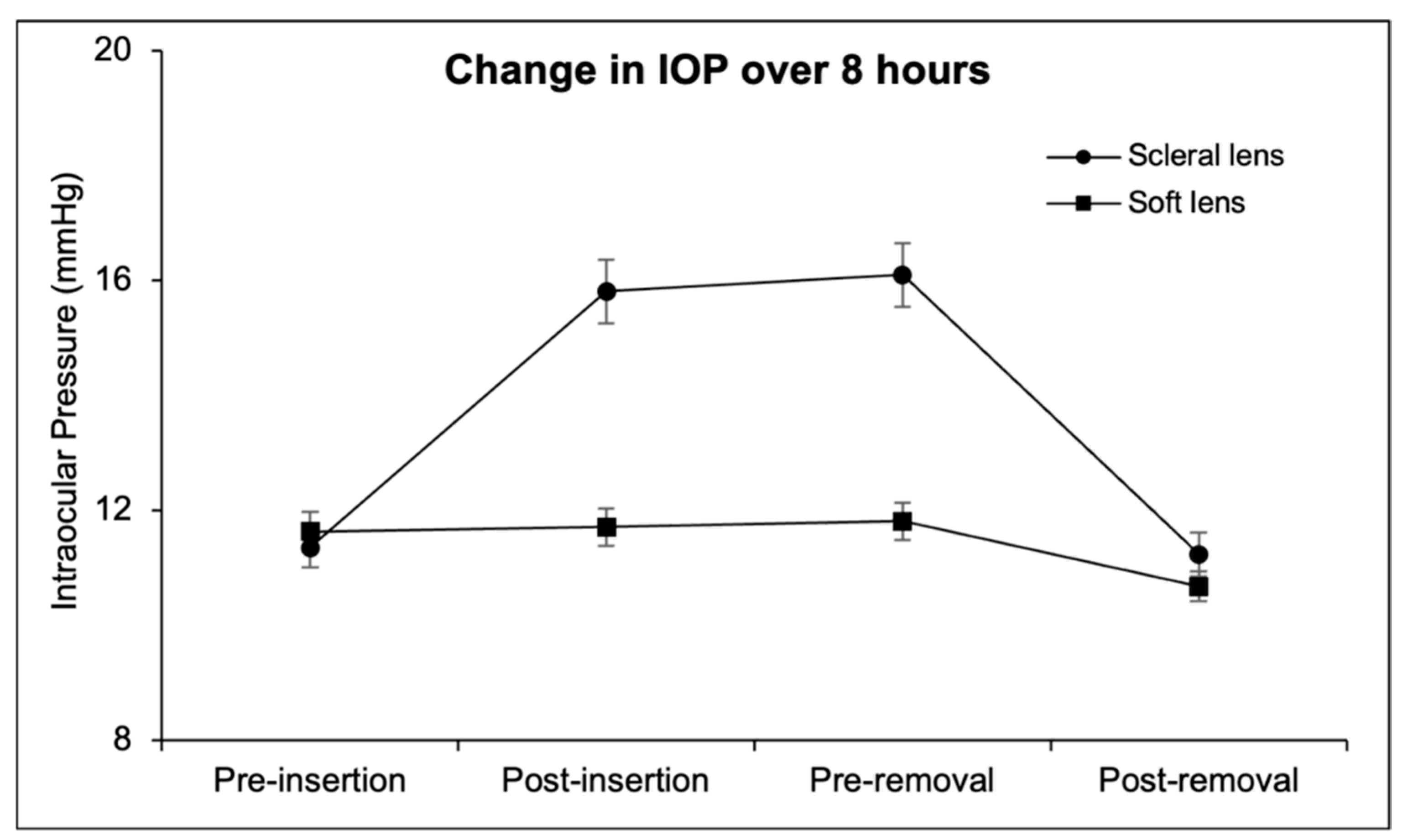
3.2. Intraocular Pressure (IOP)
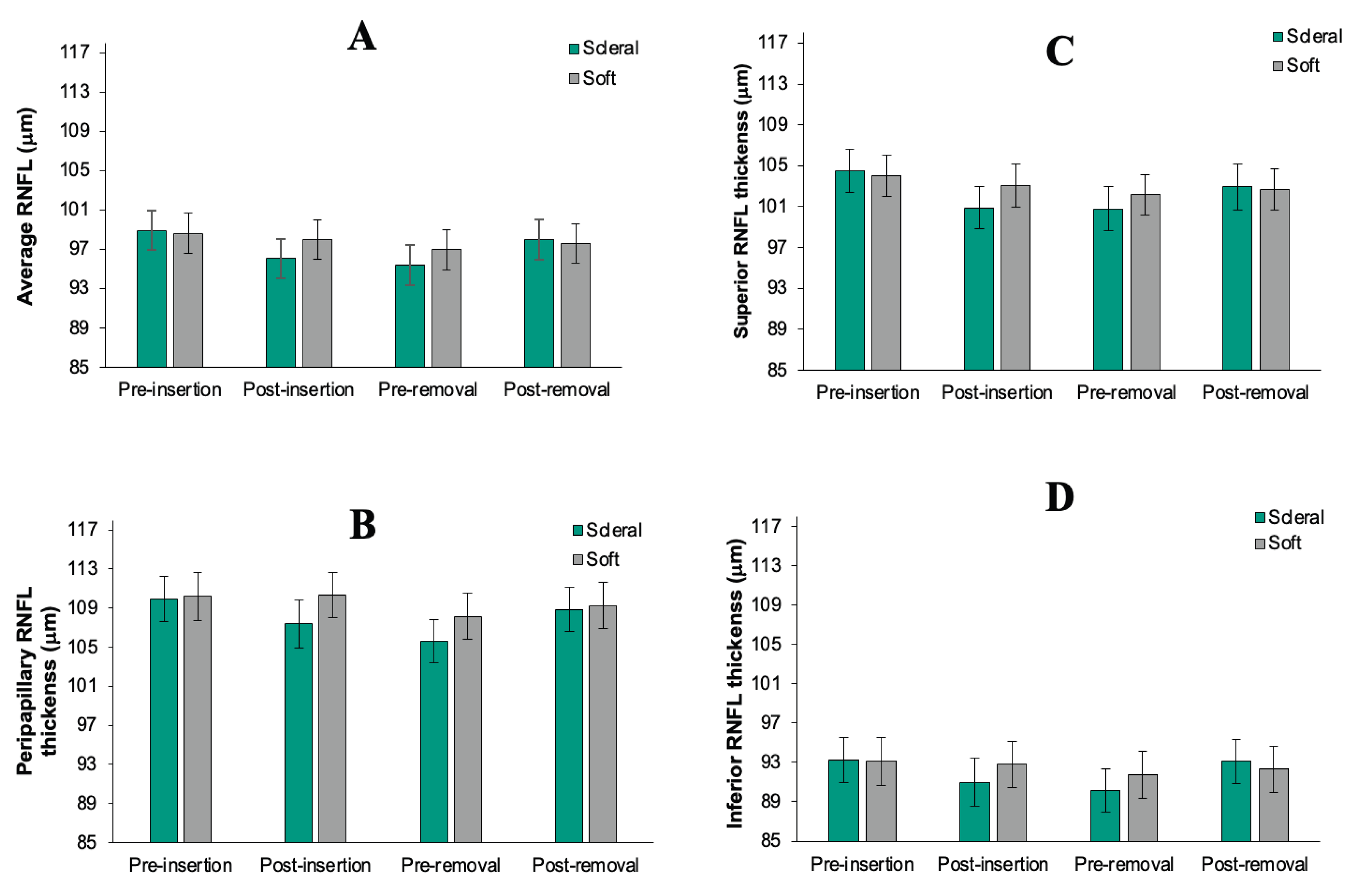
3.3. Retinal Nerve Fiber Layer (RNFL) Thickness
4. Discussion
5. Conclusions
6. Limitation
References
- Pearson, RM. Karl Otto Himmler, manufacturer of the first contact lens. Contact Lens and Anterior Eye 2007, 30(1), 11–16. [Google Scholar] [CrossRef]
- Pecego, M; Barnett, M; Mannis, MJ; Durbin-Johnson, B. Jupiter Scleral Lenses: The UC Davis Eye Center Experience. Eye & Contact Lens: Science & Clinical Practice 2012, 38(3), 179–182. [Google Scholar] [CrossRef]
- Harthan, JS; Shorter, E. Therapeutic uses of scleral contact lenses for ocular surface disease: patient selection and special considerations. OPTO 2018, Volume 10, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Pullum, KW; Whiting, MA; Buckley, RJ. Scleral Contact Lenses: The Expanding Role. Cornea 2005, 24(3), 269–277. [Google Scholar] [CrossRef]
- Visser, ES; Visser, R; van Lier, HJJ; Otten, HM. Modern Scleral Lenses Part II: Patient Satisfaction. Eye & Contact Lens: Science & Clinical Practice 2007, 33(1), 21–25. [Google Scholar] [CrossRef]
- Gumus, K; Gire, A; Pflugfelder, SC. The Impact of the Boston Ocular Surface Prosthesis on Wavefront Higher-Order Aberrations. American Journal of Ophthalmology 2011, 151(4), 682–690.e2. [Google Scholar] [CrossRef]
- Fadel, D. The influence of limbal and scleral shape on scleral lens design. Contact Lens and Anterior Eye 2018, 41(4), 321–328. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, P; Croteau, A. Fluid-Ventilated, Gas-Permeable Scleral Contact Lens Is an Effective Option for Managing Severe Ocular Surface Disease and Many Corneal Disorders That Would Otherwise Require Penetrating Keratoplasty. Eye & Contact Lens: Science & Clinical Practice 2005, 31(3), 130–134. [Google Scholar] [CrossRef]
- Van Der Worp, E; Bornman, D; Ferreira, DL; Faria-Ribeiro, M; Garcia-Porta, N; González-Meijome, JM. Modern scleral contact lenses: A review. Contact Lens and Anterior Eye 2014, 37(4), 240–250. [Google Scholar] [CrossRef]
- Michaud, L; Balourdet, S; Samaha, D. Variation of Bruch’s membrane opening in response to intraocular pressure change during scleral lens wear, in a population with keratoconus. Ophthalmic Physiologic Optic 2025, 45(2), 405–415. [Google Scholar] [CrossRef]
- Schornack, MM; Brown, WL; Siemsen, DW. The use of tinted contact lenses in the management of achromatopsia. Optometry - Journal of the American Optometric Association 2007, 78(1), 17–22. [Google Scholar] [CrossRef] [PubMed]
- Walker, MK; Bergmanson, JP; Miller, WL; Marsack, JD; Johnson, LA. Complications and fitting challenges associated with scleral contact lenses: A review. Contact Lens and Anterior Eye 2016, 39(2), 88–96. [Google Scholar] [CrossRef]
- Thoft, RA; Friend, J. Biochemical Aspects of Contact Lens Wear. American Journal of Ophthalmology 1975, 80(1), 139–145. [Google Scholar] [CrossRef]
- Kauffman, MJ; Gilmartin, CA; Bennett, ES; Bassi, CJ. A Comparison of the Short-Term Settling of Three Scleral Lens Designs. Optometry and Vision Science 2014, 91(12), 1462–1466. [Google Scholar] [CrossRef]
- Nau, CB; Schornack, MM; McLaren, JW; Sit, AJ. Intraocular Pressure After 2 Hours of Small-Diameter Scleral Lens Wear. Eye & Contact Lens: Science & Clinical Practice 2016, 42(6), 350–353. [Google Scholar] [CrossRef] [PubMed]
- Michaud, L; Samaha, D; Giasson, CJ. Intra-ocular pressure variation associated with the wear of scleral lenses of different diameters. Contact Lens and Anterior Eye 2019, 42(1), 104–110. [Google Scholar] [CrossRef]
- Nau, CB; Schornack, MM; McLaren, JW; Sit, AJ. Intraocular Pressure After 2 Hours of Small-Diameter Scleral Lens Wear. Eye & Contact Lens: Science & Clinical Practice 2016, 42(6), 350–353. [Google Scholar] [CrossRef] [PubMed]
- Walker, MK; Pardon, LP; Redfern, R; Patel, N. IOP and Optic Nerve Head Morphology during Scleral Lens Wear. Optom Vis Sci. 2020, 97(9), 661–668. [Google Scholar] [CrossRef]
- Vincent, SJ; Alonso-caneiro, D; Collins, MJ. Evidence on scleral contact lenses and intraocular pressure. Clinical and Experimental Optometry 2017, 100(1), 87–88. [Google Scholar] [CrossRef]
- Jung, EH; Lee, EJ; Kim, TW. Topographic Relationship with a Retinal Nerve Fiber Layer Defect Differs between β -Zone and γ -Zone Parapapillary Atrophy. Journal of Ophthalmology 2020, 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, K; Bowd, C; Weinreb, RN; Medeiros, FA; Sample, PA; Zangwill, LM. Retinal nerve fiber layer thickness measurements with scanning laser polarimetry predict glaucomatous visual field loss. American Journal of Ophthalmology 2004, 138(4), 592–601. [Google Scholar] [CrossRef]
- Diniz-Filho, A; Abe, RY; Zangwill, LM; et al. Association between Intraocular Pressure and Rates of Retinal Nerve Fiber Layer Loss Measured by Optical Coherence Tomography. Ophthalmology 2016, 123(10), 2058–2065. [Google Scholar] [CrossRef]
- Walker, MK; Pardon, LP; Redfern, R; Patel, N. IOP and Optic Nerve Head Morphology during Scleral Lens Wear. Optom Vis Sci. 2020, 97(9), 661–668. [Google Scholar] [CrossRef] [PubMed]
- Mcmonnies, CW. A hypothesis that scleral contact lenses could elevate intraocular pressure. Clinical and Experimental Optometry 2016, 99(6), 594–596. [Google Scholar] [CrossRef] [PubMed]
- Li, Y; Shi, J; Duan, X; Fan, F. Transpalpebral measurement of intraocular pressure using the Diaton tonometer versus standard Goldmann applanation tonometry. Graefe’s Archive for Clinical and Experimental Ophthalmology 2010, 248(12), 1765–1770. [Google Scholar] [CrossRef]
- Doherty, MD; Carrim, ZI; O’Neill, DP. Diaton tonometry: an assessment of validity and preference against Goldmann tonometry. Clinical Exper Ophthalmology 2012, 40(4). [Google Scholar] [CrossRef] [PubMed]
- Chauhan, BC; Danthurebandara, VM; Sharpe, GP; et al. Bruch’s Membrane Opening Minimum Rim Width and Retinal Nerve Fiber Layer Thickness in a Normal White Population: A Multicenter Study. Ophthalmology 2015, 122(9), 1786–1794. [Google Scholar] [CrossRef]
- The Trabecular Meshwork: A Basic Review of Form and Function. J Ocul Biol. 2014, 2(1). [CrossRef]
- Johnstone, M; Jamil, A; Martin, E. Aqueous Veins and Open Angle Glaucoma. In The Glaucoma Book; Schacknow, PN, Samples, JR, Eds.; Springer New York, 2010; pp. 65–78. [Google Scholar] [CrossRef]
- Acott, TS; Kelley, MJ; Keller, KE; et al. Intraocular Pressure Homeostasis: Maintaining Balance in a High-Pressure Environment. Journal of Ocular Pharmacology and Therapeutics 2014, 30(2-3), 94–101. [Google Scholar] [CrossRef]
- Johnstone, M; Jamil, A; Martin, E. Aqueous Veins and Open Angle Glaucoma. In The Glaucoma Book; Schacknow, PN, Samples, JR, Eds.; Springer New York, 2010; pp. 65–78. [Google Scholar] [CrossRef]
- Chuangsuwanich, T; Tun, TA; Braeu, FA; et al. How Myopia and Glaucoma Influence the Biomechanical Susceptibility of the Optic Nerve Head. Invest Ophthalmol Vis Sci. 2023, 64(11), 12. [Google Scholar] [CrossRef]
- Ohno-Matsui, K; Wu, PC; Yamashiro, K; et al. IMI Pathologic Myopia. Invest Ophthalmol Vis Sci. 2021, 62(5), 5. [Google Scholar] [CrossRef]
- Jia, X; Yu, J; Liao, SH; Duan, XC. Biomechanics of the sclera and effects on intraocular pressure. Int J Ophthalmol. 2016, 9(12), 1824–1831. [Google Scholar] [CrossRef]
- Boote, C; Sigal, IA; Grytz, R; Hua, Y; Nguyen, TD; Girard, MJA. Scleral structure and biomechanics. Prog Retin Eye Res. 2020, 74, 100773. [Google Scholar] [CrossRef]
- Jiang, J; Lin, T; Lin, F; et al. Effect of intraocular pressure reduction on progressive high myopia (PHM study): study protocol of a randomised controlled trial. BMJ Open. 2024, 14(6), e084068. [Google Scholar] [CrossRef] [PubMed]
| N (%) | Mean ± SD | Range | |
|---|---|---|---|
| Age(years) | 26 ± 3 | 20 - 31 | |
| Sex, male/female: n (%) | 20/11 (64.5%/35.5%) |
||
| Race: n (%) | |||
| · White | 18 (58.1%) | ||
| · Asian | 3 (9.7%) | ||
| · Asian/Indian | 3 (9.7%) | ||
| · Other | 7 (22.6%) | ||
| Refractive error (Spherical equivalent) (D) | -3.25 ± 3.25 | -9.25-+0.50 | |
| Average corneal curvature (D) | 43 ± 2.25 | 40.25 - 46.5 | |
| Baseline IOP (mmHg) | 14.3 ± 3 | 8 - 20 | |
| Pulse (BPM) | 72 ± 13 | 46 - 97 | |
| Systolic BP (mmHg) | 105 ± 13 | 84 - 140 | |
| Diastolic BP (mmHg) | 62 ± 8 | 43 - 86 | |
| OPP (mmHg) | 38 ± 7 | 18 - 63 |
| Source | Numerator df | Denominator df | F | Sig. |
|---|---|---|---|---|
| Intercept | 1 | 30.933 | 1826.294 | <.001 |
| Eye | 1 | 685.211 | 0.4 | 0.527 |
| LensType | 1 | 685.211 | 102.13 | <.001 |
| Conditions | 3 | 685.545 | 13.34 | <.001 |
| Eye * LensType | 1 | 685.211 | 0.06 | 0.806 |
| Eye * Conditions | 3 | 685.211 | 0.915 | 0.433 |
| LensType * Conditions a Dependent Variable: IOP. |
3 | 685.211 | 31.741 | <.001 |
| Source | Numerator df | Denominator df | F | Sig. |
|---|---|---|---|---|
| Intercept | 1 | 30.023 | 4891.669 | <.001 |
| Eye | 1 | 701 | 3.738 | 0.054 |
| LensType | 1 | 701 | 17.925 | <.001 |
| Conditions | 3 | 701 | 47.057 | <.001 |
| Eye * LensType | 1 | 701 | 0.172 | 0.678 |
| Eye * Conditions | 3 | 701 | 2.62 | 0.052 |
| LensType * Conditions | 3 | 701 | 13.552 | <.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





