1. Introduction
Unconventional radiotherapy approaches (including high and ultra-high dose rate or spatially varying dose delivery) are currently under investigation [
1], mainly preclinically, with particular attention to producing improved tolerance of healthy tissues or to determine an improved tumour control. Radiation sources include high-energy (MeV) protons, electrons, or photons, as well as low-energy (keV) photons produced by orthovoltage X-ray tubes. A class of promising strategies, which potentially permits the implementation of this approach, is spatially fractionated radiation therapy (SFRT), in which the radiation dose is delivered purposely in a nonuniform spatial pattern, creating alternating regions of high and low dose values. Various SFRT techniques have been proposed and are well investigated in the literature, including GRID therapy, lattice therapy, microbeam radiation therapy (MRT), and minibeam radiation therapy (MBRT) [
2,
3,
4]. Current evidence suggests that MBRT can achieve effective tumour control while significantly reducing normal tissue radiotoxicity [
5]. Indeed, over the past decades, extensive preclinical research in animal models has demonstrated that MBRT with kilovoltage photons may provide superior normal tissue sparing and tumour control, compared with conventional homogeneous irradiation fields with broad beams [
5]. Spatial dose modulation is typically achieved through incident beam attenuation using a multi-slit collimator positioned in front of the radiation source, which, in a common approach, determines miniplanar beams with beamlets spaced in a one-dimensional array. In typical implementations, MBRT is characterized by arrays of submillimetric beams, typically 0.1–2 mm wide, separated by a fraction of a millimeter to several millimeters [
6]; single or few-view irradiations of the target volume are typically utilized in preclinical radiotherapy treatments.
Moreover, beyond the practical advantage of using hospital-based commercial X-ray irradiators rather than large synchrotron radiation facilities as in MRT research, the relatively larger beamlet widths of MBRT treatment (with respect to 0.05-0.10 mm beamlets) also provide increased tolerance to organ and tumour motion during treatment. This feature expands the range of feasible preclinical studies at the hospital level and may ultimately facilitate the clinical translation of the technique.
Previous investigations with kilovoltage X-rays have sought to optimize photon-minibeam energy [
7], generally showing that orthovoltage X-rays are preferable to gamma rays due to reduced lateral scattering, which preserves a higher peak-to-valley dose ratio (PVDR). Studies have also emphasized the biological relevance of the valley doses [
8,
9] mainly produced by scattered photons within the target volume. Conversely, other reports have shown that higher PVDR values correlate with improved healthy tissue sparing [
10,
11]. Preclinical MBRT studies with kilovoltage X-rays (x-MBRT) have been performed using various small-animal irradiation platforms, each employing different beam delivery systems and collimator geometries. In 2024, the first human patients were treated with x-MBRT [
12] using an orthovoltage X-ray source, marking a milestone in the translation of this technique to the clinic.
Despite extensive
in silico collimator design studies [
13,
14], there is still no consensus on the “optimal” collimator geometry for achieving the best therapeutic window in x-MBRT. Indeed, the identification of a universally optimal geometry – if such configuration exists – depends on a complex combination of geometrical factors (related to both irradiator and collimator), dosimetric parameters, X-ray beam quality, available power source and dose rate, collimator material, beam divergence, beam field size, distance of the beam from the collimator, as well as the size, shape and position of the target within the animal body. In this context, Monte Carlo (MC) simulations play a significant role in optimizing the dose delivery pattern via virtual dosimetry studies. This approach enables the systematic and independent variation of a reduced set of key irradiation parameters in controlled virtual experiments, through the use of a digital twin of the irradiation source, irradiation setup, and target object (including small-animal computational models).
Following our previous study on a virtual dosimetry platform for preclinical x-MBRT [
15], the present study focuses on optimizing the geometric design of x-MBRT collimators for preclinical irradiations with a commercial system. MC simulations are employed to investigate the relationship between geometrical parameters of both parallel and diverging collimators and key dose parameters in the irradiation session.
A specific investigative goal is to show, computationally, the difference of three-dimensional (3D) dose distributions within the target, using an x-MBRT collimator, compared with an open beam irradiation, having the same field size. This strategy isolates the dosimetric effect of the MBRT collimator itself, independently of the irradiation geometry (i.e., distances and field size), which remains common to a corresponding conventional broad-beam irradiation, while systematically varying the collimator design parameters.
Such an approach closely reflects typical preclinical MBRT studies, where treatment outcomes in tumor-bearing small animals irradiated with minibeams are compared with cohorts of animals receiving either equivalent open-beam irradiation (without an MBRT collimator) or no irradiation.
In this framework, a direct one-to-one comparison of 3D dose distributions (MBRT vs. open beam) via virtual dosimetry would be beneficial for analyzing treatment outcomes and estimating the x-MBRT dose needed to deliver the same target dose as in the corresponding open-beam treatment. A further advantage of this approach is the possibility of incorporating into the MC-based dosimetry plan a digital twin of the tumor-bearing animal, derived from a computed tomography scan of the animal, performed pre-treatment on the same irradiation platform if equipped with a built-in imaging unit. Finally, this study represents a basic step in our efforts for devising a preclinical x-MBRT treatment planning system based on MC simulations, for which we are also implementing GPU-based MC simulation tools (called VIT-MBRT platform) for fast and validated virtual imaging and dosimetry studies with kilovoltage X-rays [
15,
16].
4. Discussion
The present work provides a systematic Monte Carlo–based analysis of how individual geometric parameters of multi-slit collimators influence the fundamental dosimetric descriptors of X-ray minibeam radiation therapy. While several studies have explored collimator fabrication strategies or reported empirical performance metrics [
13,
14,
21], a quantitative mapping between geometric design variables and beam-quality indicators such as
PVDR,
valley dose, and
mean dose has remained largely unexplored. The results presented here fill this gap by establishing explicit functional dependencies that can guide rational collimator optimization for preclinical x-MBRT.
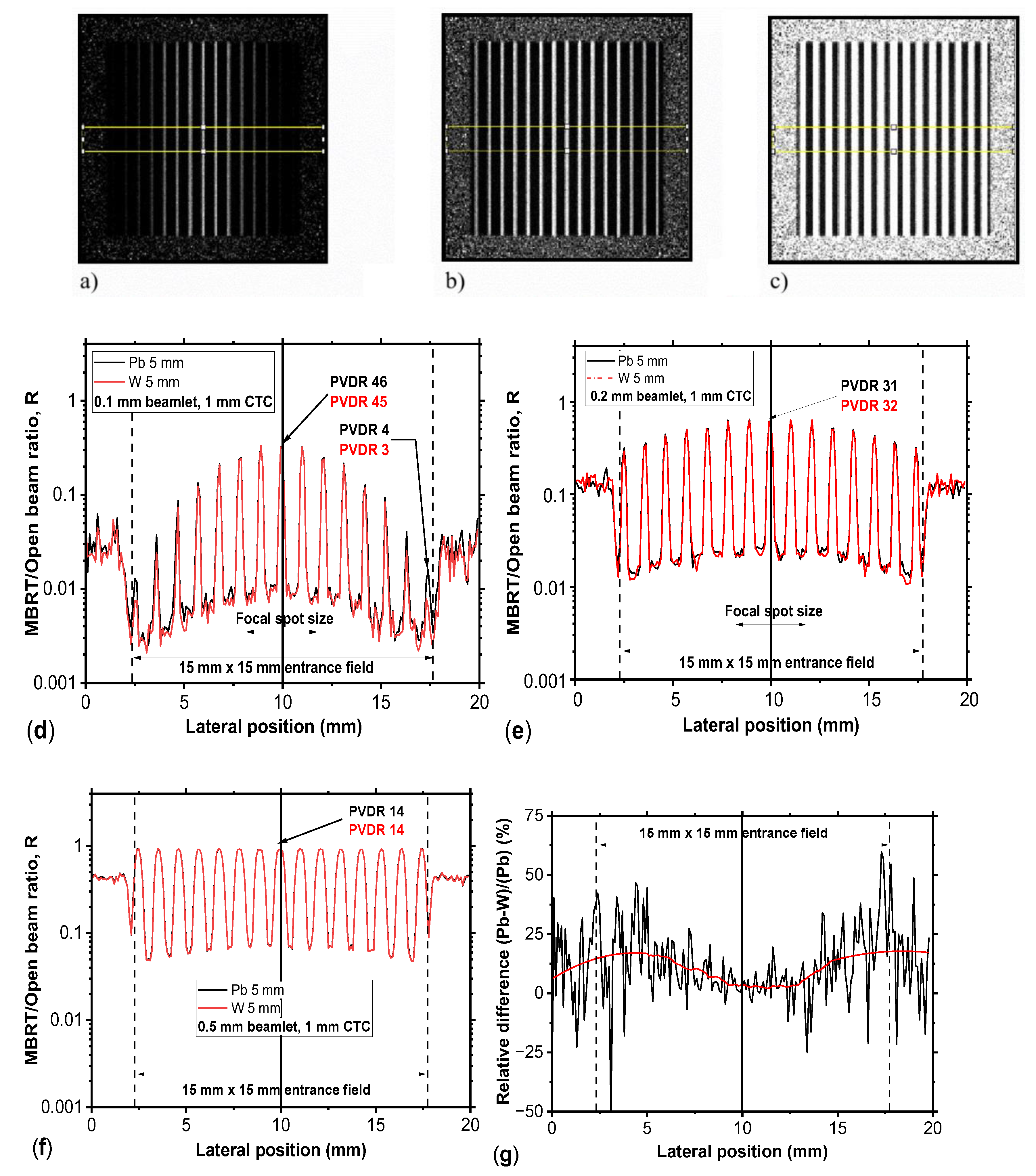
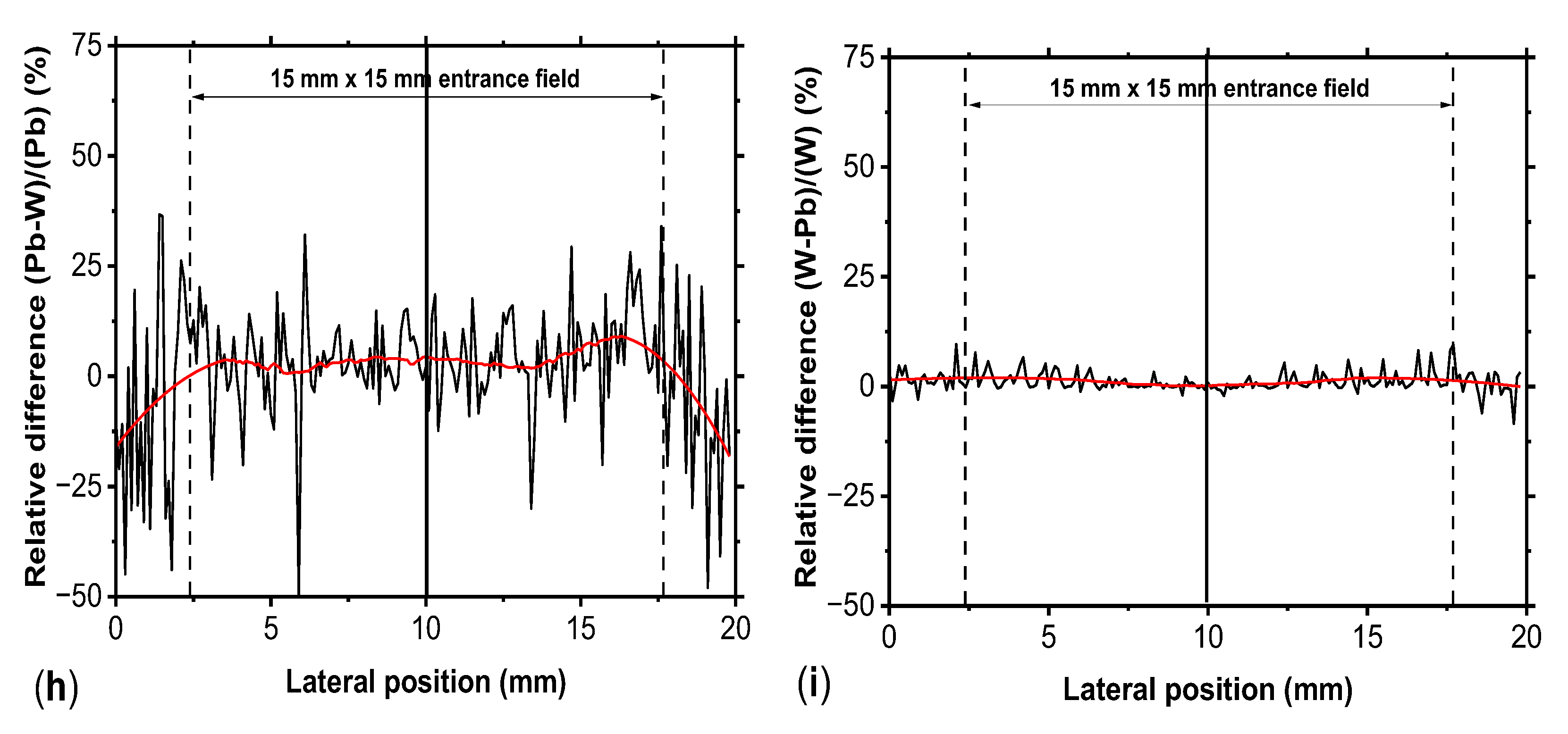
The comparison between lead and tungsten collimators (
Figure 5) demonstrates that, for thicknesses on the order of 5 mm, both materials yield nearly indistinguishable central-axis PVDR values. This is consistent with the high attenuation coefficients of both materials in the orthovoltage range, where photoelectric absorption dominates. The different profile shapes (
Figure 5d–f) due to the parallel collimator geometry and not to collimator material, show a lateral reduction of the peak intensity from 25% to 2% for the smallest (
w/CTC = 10%,
Figure 5g) and greater (
w/CTC = 50%,
Figure 5i) apertures, respectively. This arises from the differential attenuation of the oblique photons’ beam, which becomes increasingly relevant at off-axis positions. These results suggest that the choice of collimator material, in the case of a 5-mm-thick MBRT collimator is related only on practical considerations, such as cost, machinability, achievable tolerances, and mechanical stability.
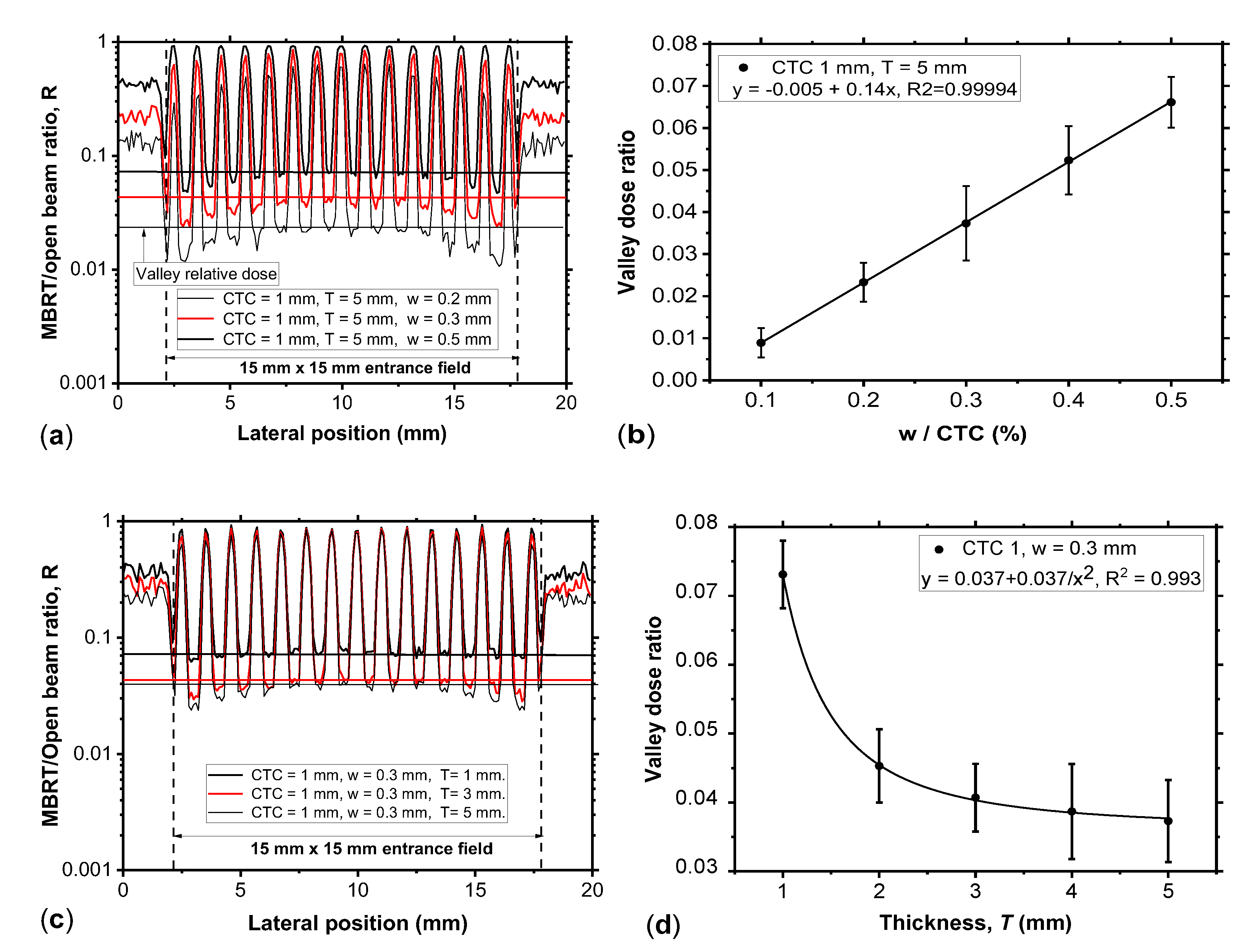
A central outcome of this study is the demonstration that the valley dose exhibits a linear dependence on the
w/CTC ratio (
Figure 6b), confirming that the geometric filling factor governs the fraction of scattered photons reaching the inter-beam regions. This observation is consistent with theoretical models of spatially fractionated beams, where valley dose is dominated by low-energy scattered photons whose fluence scales with the open fraction of the collimator [
6]. Conversely, the inverse-square dependence of
valley dose on collimator thickness (
D ∝ 1/
T²) (
Figure 6d) reflects the suppression of lateral photon transport within the slit channel, in agreement with previous Geant4-based analyses of microbeam and minibeam collimators [
14,
17]. These relationships provide a compact parametric framework for predicting valley-dose behaviour without requiring full MC simulations for each geometry.
Thicker collimators more effectively suppress laterally scattered photons within the slits, thereby increasing PVDR. However, beyond approximately 4 mm (for tungsten), additional thickness yields diminishing returns while unnecessarily reducing beam transmission and increasing manufacturing complexity and costs—particularly for tungsten.
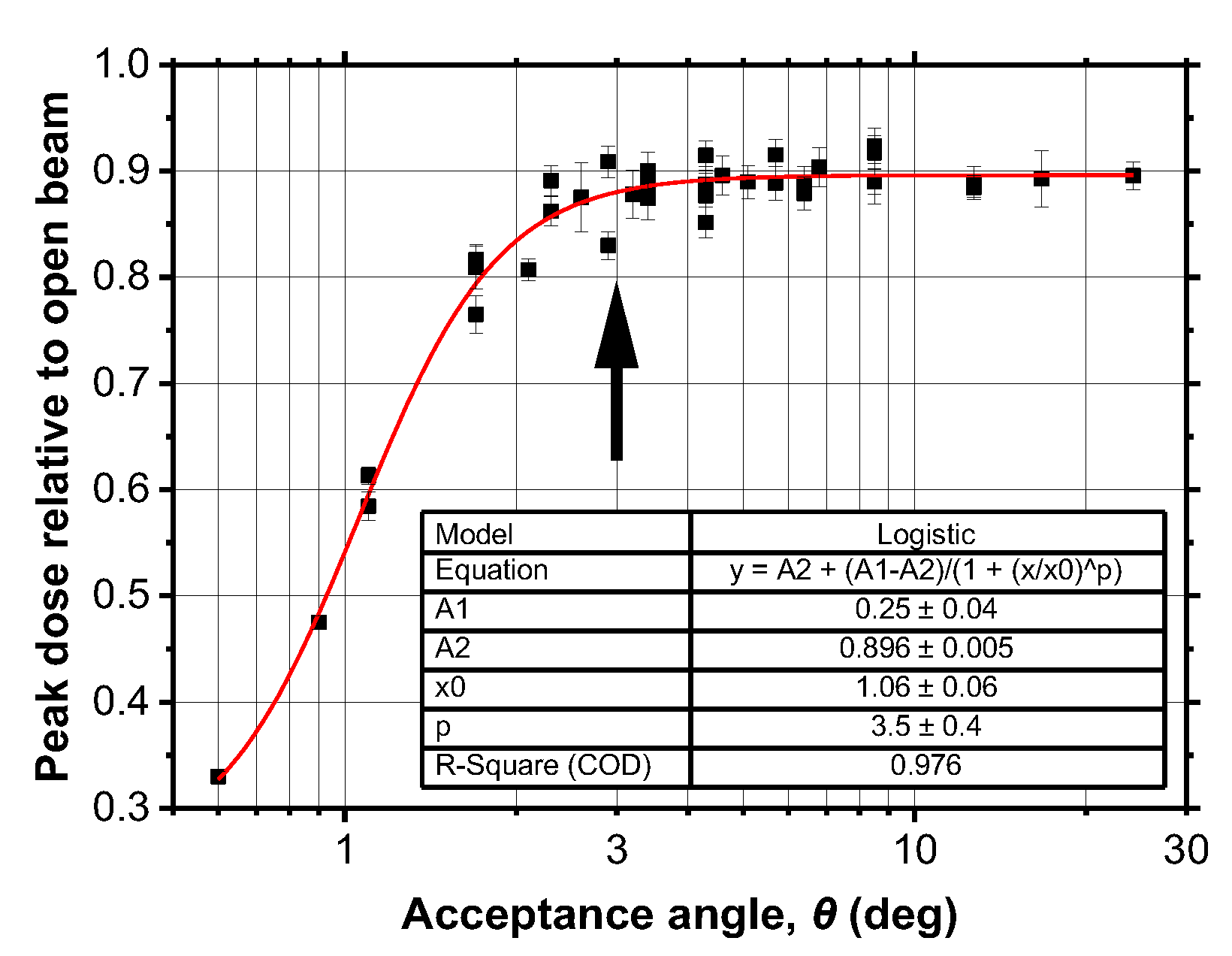
The acceptance angle
θ emerges as another critical determinant of peak-dose transmission. As shown in
Figure 9, the peak dose increases with
θ until reaching a saturation plateau at approximately 0.9 (normalized to open beam) for
θ ≳ 3°. This behaviour reflects the interplay between the intrinsic beam divergence of the X-RAD225Cx source and the angular acceptance of the slit channel. When
θ is smaller than the beam divergence, a portion of the primary fluence is intercepted by the collimator walls, reducing the peak intensity. Once
θ matches or exceeds the beam divergence, further increases do not enhance transmission. Similar saturation effects have been reported in synchrotron-based MRT studies, where matching the collimator acceptance to the source divergence is essential to preserve peak integrity [
5,
6].
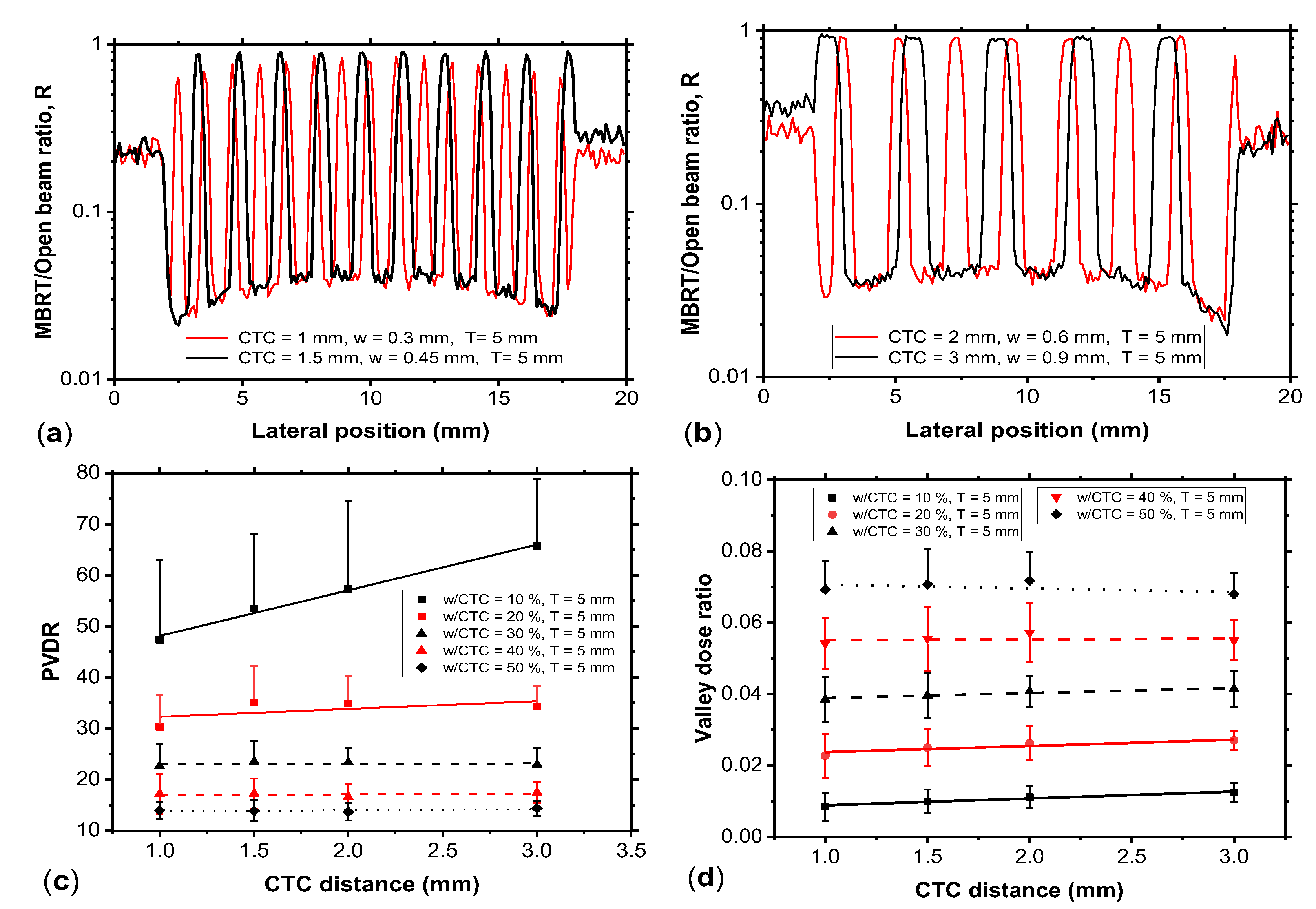
The dependence of
PVDR on
CTC spacing (
Figure 7 and
Figure 8) further clarifies the geometric trade-offs inherent to MBRT design. For fixed
w/CTC ratios, increasing
CTC enhances
PVDR primarily through an increase in
peak dose, while
valley dose remains approximately constant. This is consistent with the reduced overlap of lateral scatter tails from adjacent beamlets at larger separations, a phenomenon also observed in proton and electron minibeam configurations [
2,
3,
4]. However, excessively large
CTC values reduce the spatial frequency of the minibeam pattern, potentially diminishing the biological benefits associated with spatial fractionation [
5,
10]. Thus, the optimal CTC must balance dosimetric modulation with biological efficacy.
From these findings, empirical relationships can be derived to guide collimator’s design optimization:
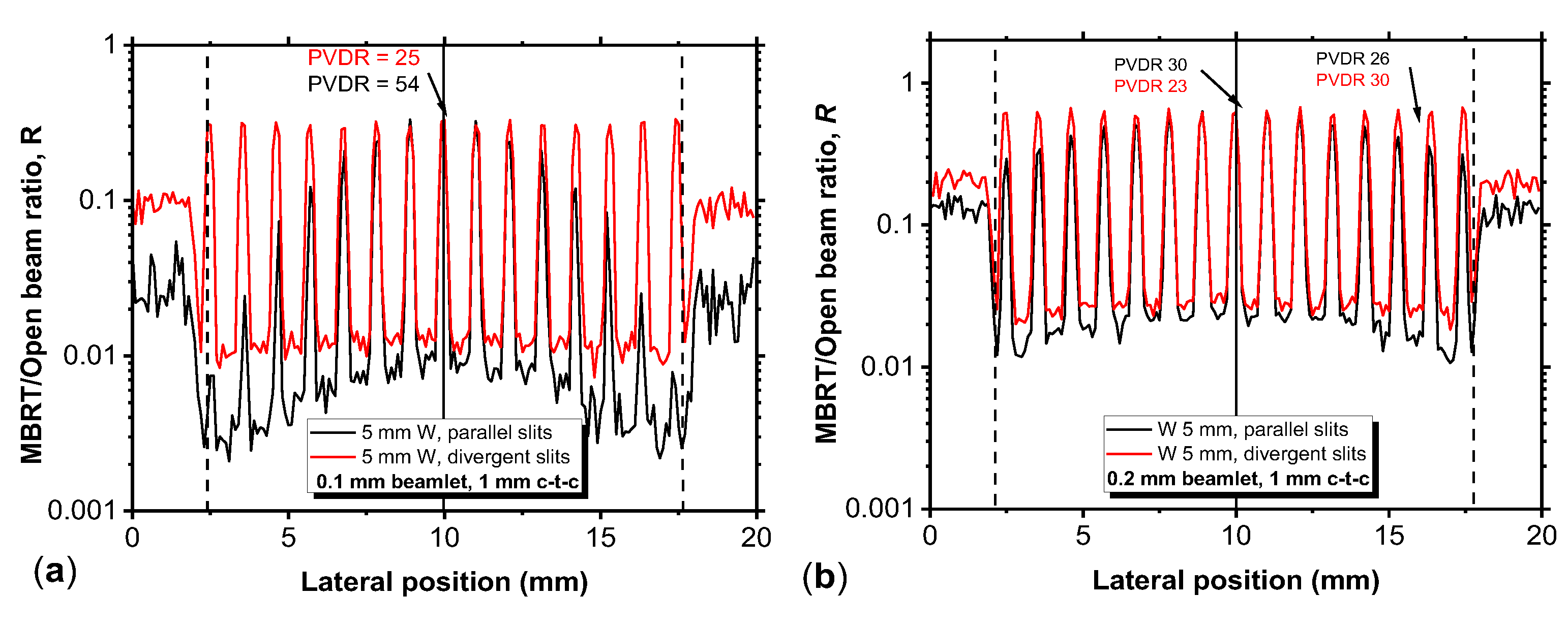
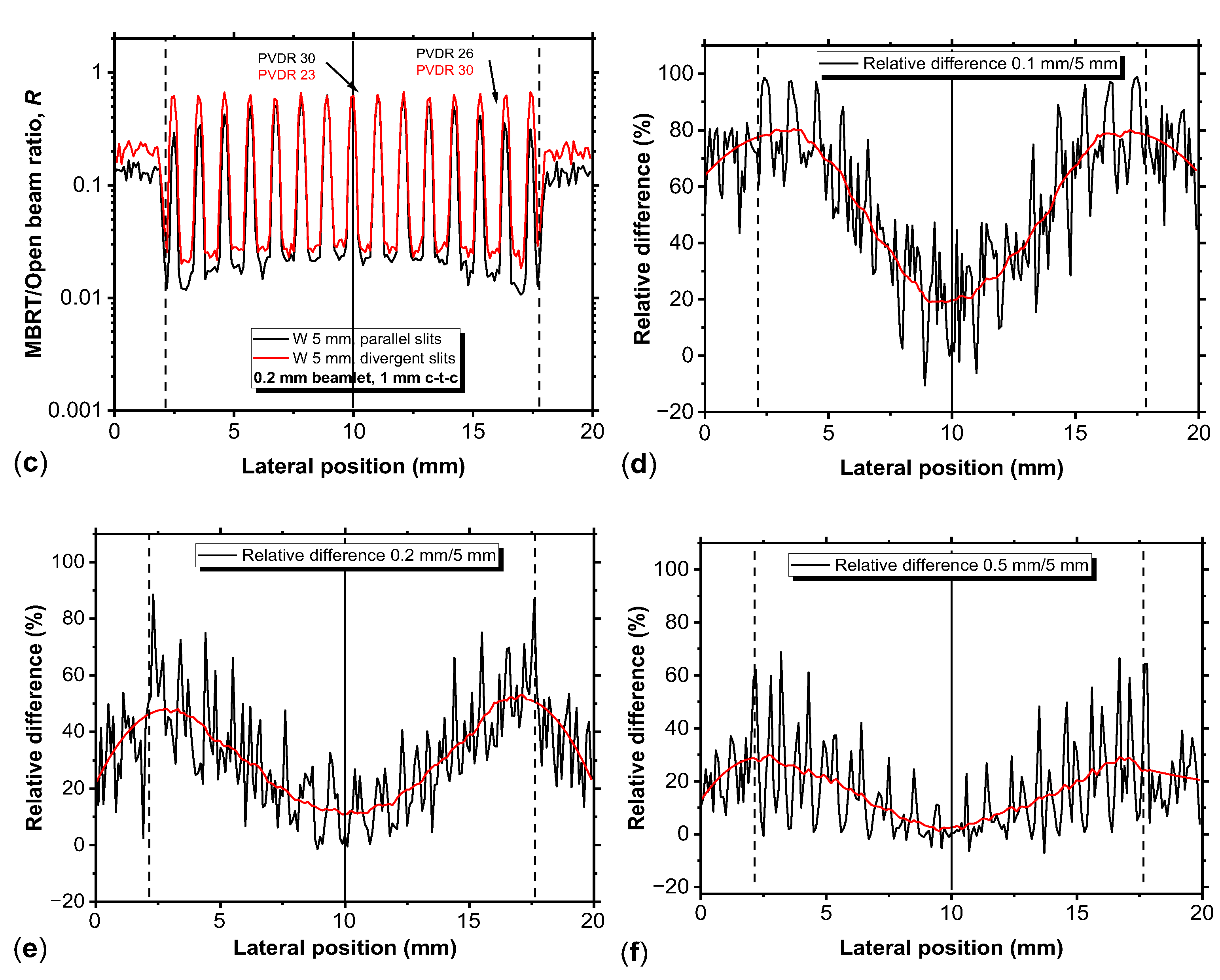
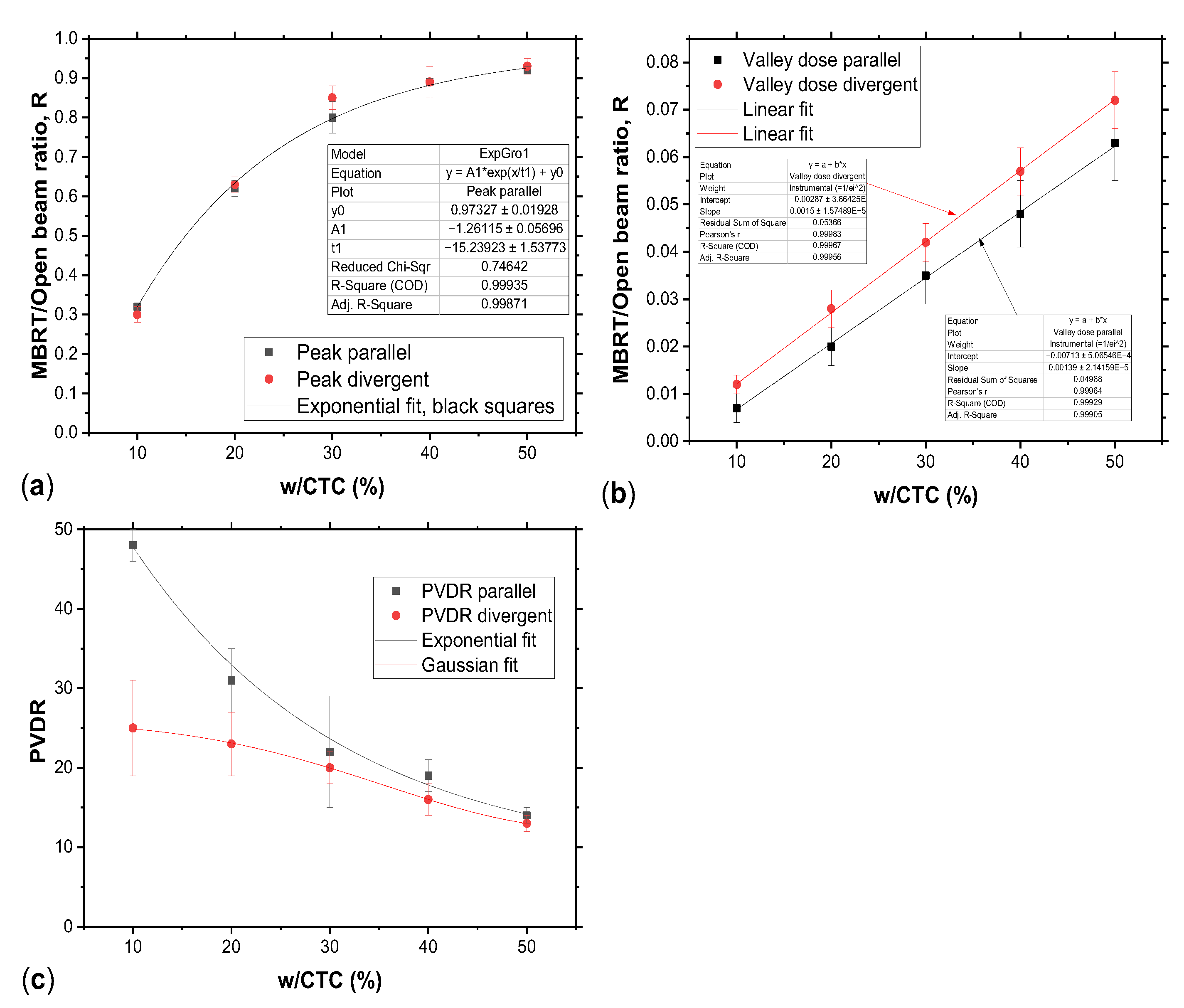
The comparison between parallel and divergent collimators (
Figure 10 and
Figure 11) further clarifies the trade-offs between geometric configurations. When slit width, thickness, and spacing are matched, both geometries yield similar central peak doses (
Figure 11a). However, divergent collimators produce flatter lateral profiles (
Figure 10a–c), improving field uniformity but increasing
valley dose due to enhanced transmission of scattered photons. These findings are consistent with prior MRT studies showing that divergent geometries increase the contribution of scattered photons to the valley region due to improved alignment between the slit channel and the beam divergence [
6]. As a result,
PVDR is consistently higher for parallel collimators (
Figure 11c), making them more suitable for applications where strong spatial modulation and normal-tissue sparing are prioritized. Nevertheless, beams exhibiting strong lateral inhomogeneities could be advantageous during the planning phase, similarly to the use of flattening filter–free (FFF) beams, which have been shown to improve dose delivery efficiency and treatment conformity [
20]. Divergent geometries may instead be advantageous for larger irradiation fields where uniformity is desired, although they inherently constrain the focus-to-target distance, reducing flexibility in experimental setups.
Overall, the present results suggest that intermediate geometries (i.e., collimator thicknesses of 2–4 mm and
w/CTC ratios of 20–40%) offer a favourable compromise between
PVDR, transmission efficiency, and manufacturability. These findings are consistent with the parameter ranges adopted in successful preclinical x-MBRT studies [
5,
7,
12]. Importantly, the relationships derived here provide a basis for future treatment-planning strategies, including integration into GPU-accelerated MC platforms such as VIT-MBRT [
15,
16], enabling rapid optimization of collimator designs tailored to specific biological endpoints, beam qualities, and anatomical targets.
Future work will include experimental validation of the MC estimated dose maps using radiochromic film dosimetry and micro-ionization chambers, as well as preclinical
in vivo studies to assess the predictive accuracy of the simulation-derived
PVDR and
valley-dose metrics. Given the increasing interest in clinical translation of x-MBRT [
12], establishing robust, physics-based design criteria for collimators is essential for ensuring reproducible and biologically effective dose delivery.
5. Conclusions
This study presented a systematic Monte Carlo–based analysis of mini-beam collimator geometries for an MBRT investigation in small animals using a preclinical X-ray irradiator. The simulations of various metal collimators investigated the influence of collimator material, slit width, CTC spacing, thickness, and geometry on the dose to water distribution, in a water phantom representative of small-animal experiments. Expressing the dose maps in relative terms with respect to open beam irradiation permits linking MBRT absorbed dose fields to open field beams, whose dosimetry is already available to the experimenter by the irradiator’s preclinical treatment planning system. Results showed that the valley-dose was found to increase linearly with the width-to-spacing ratio (w/CTC) and to decrease approximately with the inverse square of the collimator thickness, establishing the role of these two parameters as geometric determinants of spatial modulation in x-MBRT. The peak dose saturated for acceptance angles around 3°, confirming that collimator divergence should be matched to the beam geometry to minimize unnecessary attenuation or valley overexposure. Parallel geometries preserved higher PVDR and stronger spatial modulation, whereas divergent geometries offered smoother lateral profiles, making them more suitable for large irradiation fields. Overall, intermediate configurations, with thickness between 2–4 mm and w/CTC ratio of 20–40%, emerged as the most balanced design window, combining efficient dose modulation, acceptable transmission, and practical manufacturability. However, the optimal geometry remains application-specific, determined by the intended biological endpoint and specific experimental setup.
Author Contributions
Conceptualization, P.R. and G.M.; methodology, P.R.; software, U.C. and N.L.; validation, U.C., N.L., L.A.C., G.M. and P.R.; formal analysis, U.C.; writing—original draft preparation, U.C., PR.; writing—review and editing, U.C., G.M., N.L., L.A.C., R.P. and P.R.; supervision, G.M., P.R.; project administration, G.M., R.P. All authors have read and agreed to the published version of the manuscript.
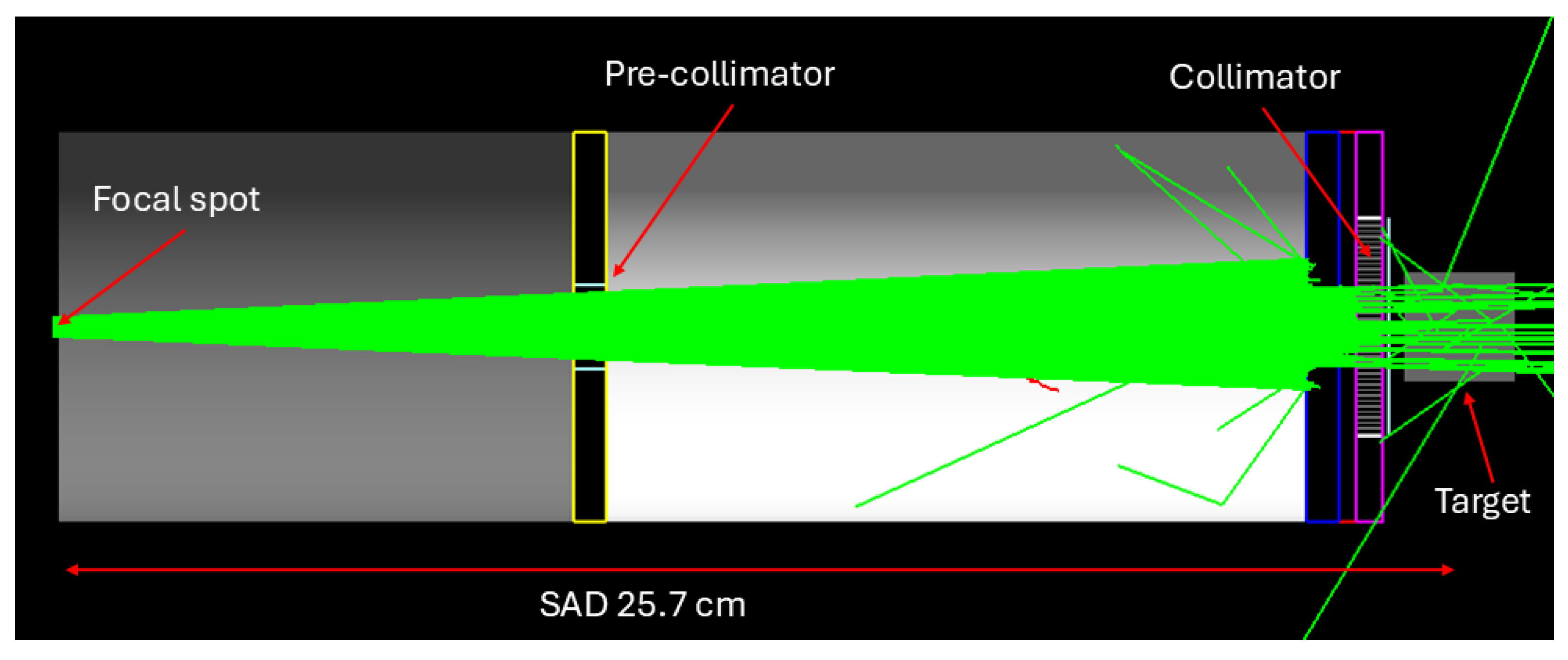
Figure 1.
The Monte Carlo simulations were performed in TOPAS, with the geometry shown, using custom-designed MBRT collimators that fit within the 15×15 mm² field collimator of the X-ray irradiator. Dose distribution was scored in a digital 2×2×2 cm³ water phantom with 0.1×0.1×0.1 mm3 voxels; the source-to-object distance was 25.7 cm.
Figure 1.
The Monte Carlo simulations were performed in TOPAS, with the geometry shown, using custom-designed MBRT collimators that fit within the 15×15 mm² field collimator of the X-ray irradiator. Dose distribution was scored in a digital 2×2×2 cm³ water phantom with 0.1×0.1×0.1 mm3 voxels; the source-to-object distance was 25.7 cm.
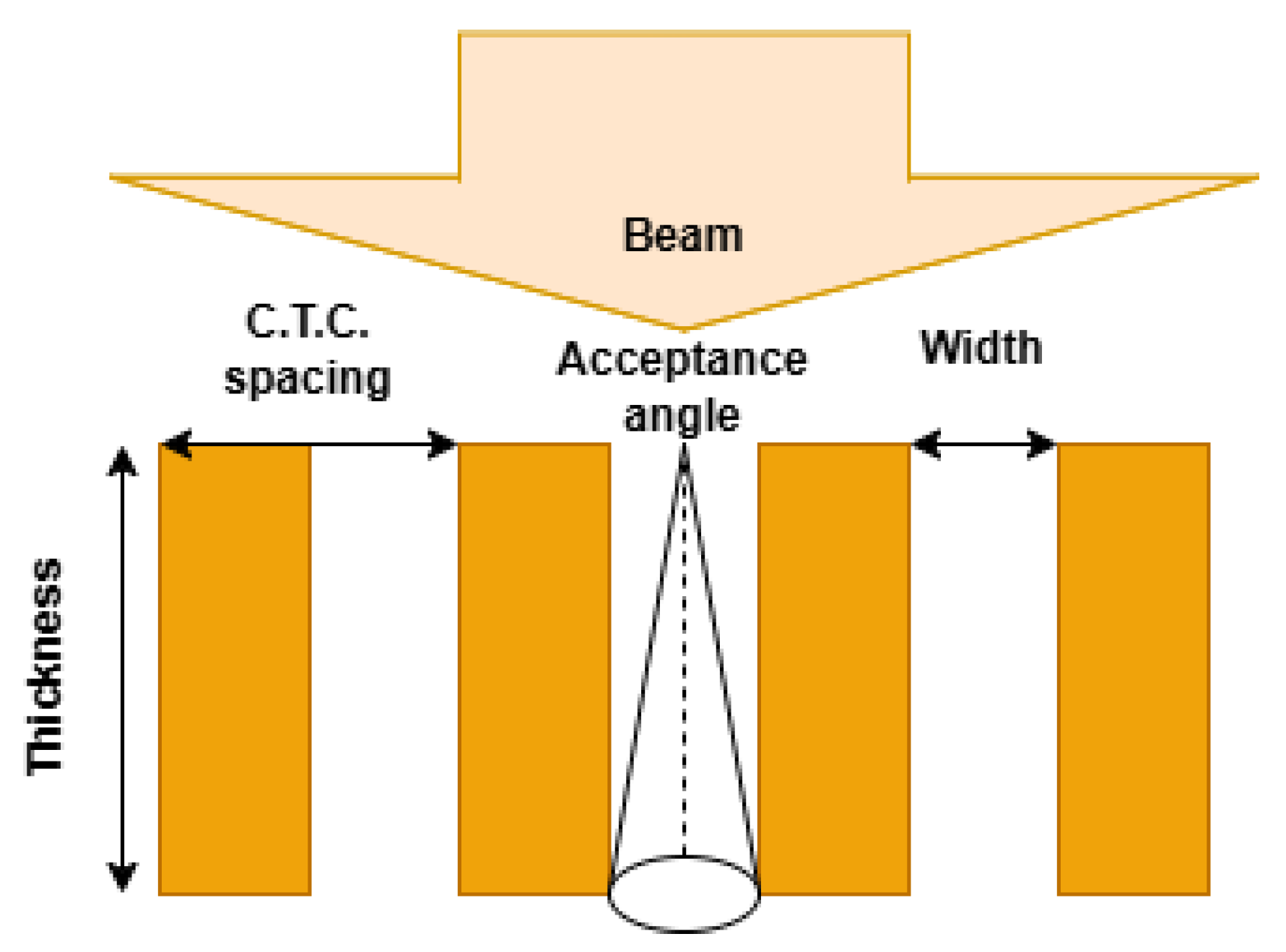
Figure 2.
Schematic representation of the minibeam geometry in x-MBRT. The primary beam is fractionated into narrow sub-beams by a multi-slit collimator characterized by width, center-to-center (CTC) distance and thickness (T).
Figure 2.
Schematic representation of the minibeam geometry in x-MBRT. The primary beam is fractionated into narrow sub-beams by a multi-slit collimator characterized by width, center-to-center (CTC) distance and thickness (T).
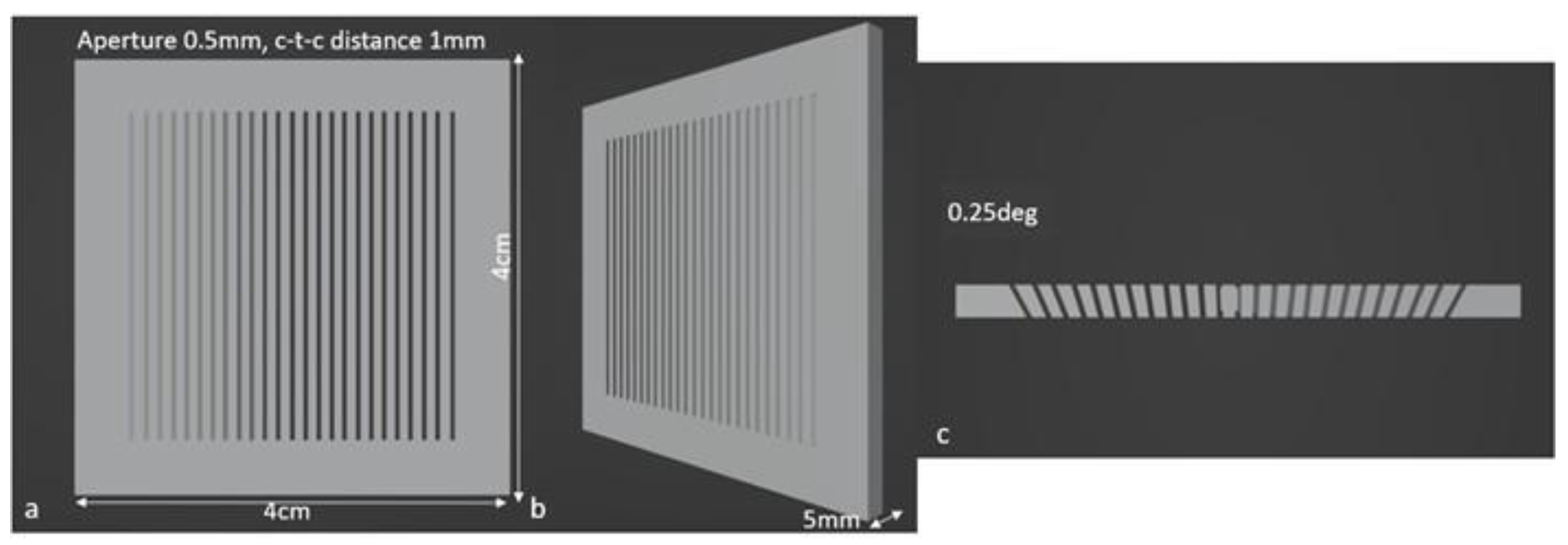
Figure 3.
Geometry of a parallel collimator: (a) front view, (b) 45° view, and (c) transversal section.
Figure 3.
Geometry of a parallel collimator: (a) front view, (b) 45° view, and (c) transversal section.
Figure 4.
Geometry of a divergent collimator: (a) front view, (b) 45° view, and (c) transversal section.
Figure 4.
Geometry of a divergent collimator: (a) front view, (b) 45° view, and (c) transversal section.
Figure 5.
Example of the R(x,y,z) distribution (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) produced by a tungsten parallel MBRT collimators, 5-mm-thick, 1 mm CTC. Beamlet width: (a) 0.1 mm, (b) 0.2 mm, (c) 0.5 mm. The profiles obtained with similar MBRT collimators, but different materials (tungsten, red line, and lead, black line) are shown in panels (d-f). In (g-i) are reported the relative difference between the profile in (d-f).
Figure 5.
Example of the R(x,y,z) distribution (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) produced by a tungsten parallel MBRT collimators, 5-mm-thick, 1 mm CTC. Beamlet width: (a) 0.1 mm, (b) 0.2 mm, (c) 0.5 mm. The profiles obtained with similar MBRT collimators, but different materials (tungsten, red line, and lead, black line) are shown in panels (d-f). In (g-i) are reported the relative difference between the profile in (d-f).
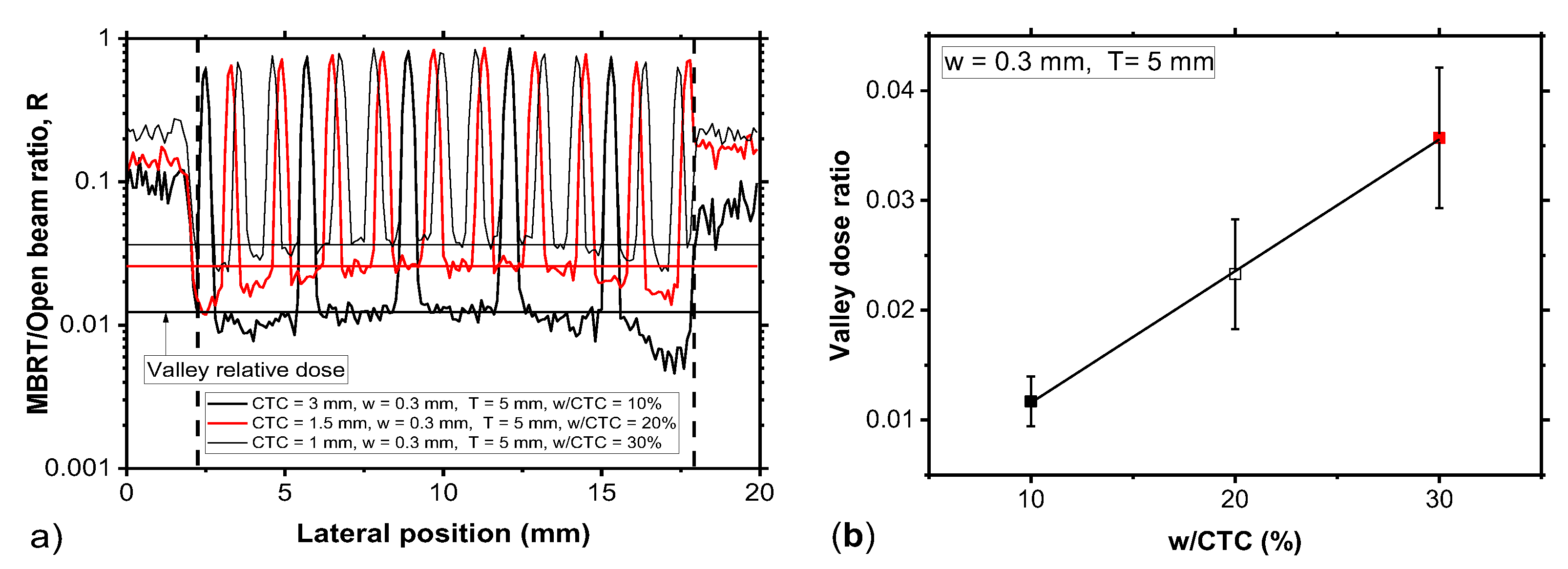
Figure 6.
(a) Comparison of profiles of the R(x,y,z) distribution (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) produced by a tungsten MBRT collimators with collimator thickness of 5 mm, CTC of 1 mm and w of 0.2 mm, 0.3 mm and 0.5 mm. (b) Valley dose ratio as a function of the w/CTC ratio. (c) Comparison of profiles of the R(x,y,z) distribution (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) produced by a tungsten MBRT collimators with collimator thickness of 1 mm, 3 mm and 5 mm, w = 0.3 mm and the CTC distance of 1 mm. (d) Valley dose ratio as a function of thickness T. An inverse-square law trend was fitted to the data points.
Figure 6.
(a) Comparison of profiles of the R(x,y,z) distribution (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) produced by a tungsten MBRT collimators with collimator thickness of 5 mm, CTC of 1 mm and w of 0.2 mm, 0.3 mm and 0.5 mm. (b) Valley dose ratio as a function of the w/CTC ratio. (c) Comparison of profiles of the R(x,y,z) distribution (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) produced by a tungsten MBRT collimators with collimator thickness of 1 mm, 3 mm and 5 mm, w = 0.3 mm and the CTC distance of 1 mm. (d) Valley dose ratio as a function of thickness T. An inverse-square law trend was fitted to the data points.
Figure 10.
Profiles of the MBRT/Open beam ratio produced by parallel (black line) and divergent (red line) tungsten MBRT collimators (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) for slit widths of 0.1 mm (a), 0.2 mm (b) and 0.5 mm (c). In all collimators the collimator thickness is fixed at 5 mm and the distance CTC at 1 mm. Relative differences between the parallel and divergent MBRT collimators profile are reported in d-f.
Figure 10.
Profiles of the MBRT/Open beam ratio produced by parallel (black line) and divergent (red line) tungsten MBRT collimators (at 1 cm depth in water, 15×15 mm2 field size at the entrance surface of the water phantom) for slit widths of 0.1 mm (a), 0.2 mm (b) and 0.5 mm (c). In all collimators the collimator thickness is fixed at 5 mm and the distance CTC at 1 mm. Relative differences between the parallel and divergent MBRT collimators profile are reported in d-f.
Figure 11.
(a) Peak dose as a function of the w/CTC ratio for parallel and diverging collimators. (b) Valley dose curve for parallel and diverging collimators as a function of the w/CTC ratio. Note that the values for the diverging collimator are always higher than those for the parallel counterpart. (c) Central PVDR curve for parallel and diverging collimators as a function of the w/CTC ratio. Fits have been shown to follow the data trends.
Figure 11.
(a) Peak dose as a function of the w/CTC ratio for parallel and diverging collimators. (b) Valley dose curve for parallel and diverging collimators as a function of the w/CTC ratio. Note that the values for the diverging collimator are always higher than those for the parallel counterpart. (c) Central PVDR curve for parallel and diverging collimators as a function of the w/CTC ratio. Fits have been shown to follow the data trends.