Submitted:
23 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Clinical and Functional Assessment
2.3. Definition of Clinical Outcomes
2.4. Statistical Analysis
3. Results
3.1. Baseline Patient Features
3.2. Clinical Evolution
3.3. Safety
3.4. Dual Remission and Complete Recovery
4. Discussion
5. Conclusion
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of interest
Abbreviations
| ACT | Asthma Control Test |
| OCS | Oral Corticosteroids |
| SNOT-22 | Sino-Nasal Outcome Test-22 |
| NPS | Nasal Polyp Score |
| BMI | Body Mass Index |
| CRSwNP | Chronic Rhinosinusitis with Nasal Polyps |
| FEF25-75 | Forced Expiratory Flow at 25-75% of FVC |
| FeNO | Fractional exhaled Nitric Oxide |
| FEV1 | Forced Expiratory Volume in 1 second |
| FVC | Forced Vital Capacity |
| GERD | Gastroesophageal Reflux Disease |
| ICS | Inhaled Corticosteroids |
| ICUR | Incremental Cost-Utility Ratio |
| IgE | Immunoglobulin E |
| IL-4 | Interleukin-4 |
| IL-5 | Interleukin-5 |
| IL-13 | Interleukin-13 |
| IQR | Interquartile Ranges |
| LABA | Long-Acting Beta-Agonists |
| LAMA | Long-Acting Muscarinic Antagonists |
| NHS | National Health Service |
| NSAID-ERD | Non-Steroidal Anti-Inflammatory Drug-Exacerbated Respiratory Disease |
| QALY | Quality-Adjusted Life Year |
| RV | Residual Volume |
| SANI | Severe Asthma Network Italy |
| SS-I | Sniffin’ Sticks Identification test |
| T2 | Type 2 (inflammation) |
| TLC | Total Lung Capacity |
References
- Pelaia, C.; Pelaia, G.; Maglio, A.; et al. Pathobiology of Type 2 Inflammation in Asthma and Nasal Polyposis. J Clin Med. 2023, 12, 3371. [Google Scholar] [CrossRef]
- Sahnoon, L.; Bajbouj, K.; Mahboub, B.; et al. Targeting IL-13 and IL-4 in Asthma: Therapeutic Implications on Airway Remodeling in Severe Asthma. Clin Rev Allergy Immunol. 2025, 68, 44. [Google Scholar] [CrossRef]
- Le Floc’h, A.; Allinne, J.; Nagashima, K.; et al. Dual blockade of IL-4 and IL-13 with dupilumab, an IL-4Rα antibody, is required to broadly inhibit type 2 inflammation. Allergy 2020, 75, 1188–1204. [Google Scholar] [CrossRef]
- Castro, M.; Corren, J.; Pavord, I.D.; et al. Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. N Engl J Med. 2018, 378, 2486–2496. [Google Scholar] [CrossRef]
- Wechsler, M.E.; Ford, L.B.; Maspero, J.F.; et al. Long-term safety and efficacy of dupilumab in patients with moderate-to-severe asthma (TRAVERSE): an open-label extension study. Lancet Respir Med. 2022, 10, 11–25. [Google Scholar] [CrossRef]
- Gelardi, M.; Bocciolini, C.; Notargiacomo, M.; et al. Chronic rhinosinusitis with nasal polyps: how to identify eligible patients for biologics in clinical practice. Acta Otorhinolaryngol Ital. 2022, 42, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Nathan, R.A.; Sorkness, C.A.; Kosinski, M.; et al. Development of the asthma control test: a survey for assessing asthma control. J Allergy Clin Immunol. 2004, 113, 59–65. [Google Scholar] [CrossRef]
- Stanojevic, S.; Kaminsky, D.A.; Miller, M.R.; et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur Respir J. 2022, 60, 2101499. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, C.; Gillett, S.; Slack, R.; et al. Psychometric validity of the 22-item Sinonasal Outcome Test. Clin Otolaryngol. 2009, 34, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Lund, V.J.; Kennedy, D.W. Staging for rhinosinusitis. Otolaryngol Head Neck Surg. 1997, 117, S35–40. [Google Scholar] [CrossRef]
- Mauthe, T.; Ryser, F.S.; Brühlmann, C.; et al. Correlation of sino-nasal outcome test and nasal polyp score in dupilumab-treated chronic rhinosinusitis with nasal polyps. Eur Arch Otorhinolaryngol. 2025, 282, 207–218. [Google Scholar] [CrossRef]
- Canonica, G.W.; Blasi, F.; Carpagnano, G.E.; et al. Severe Asthma Network Italy Definition of Clinical Remission in Severe Asthma: A Delphi Consensus. J Allergy Clin Immunol Pract. 2023, 11, 3629–3637. [Google Scholar] [CrossRef]
- Fokkens, W.J.; De Corso, E.; Backer, V.; et al. EPOS2020/EUFOREA expert opinion on defining disease states and therapeutic goals in CRSwNP. Rhinology 2024, 62, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Masala, C.; Cavazzana, A.; Sanna, F.; et al. Correlation between olfactory function, age, sex, and cognitive reserve index in the Italian population. Eur Arch Otorhinolaryngol. 2022, 279, 4943–4952. [Google Scholar] [CrossRef]
- Bosi, A.; Lombardi, C.; Caruso, C.; et al. Clinical remission and control in severe asthma: agreements and disagreements. Drugs Context 2024, 13, 2024-7-2. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.; Papi, A.; Porsbjerg, C.; et al. Effect of dupilumab on exhaled nitric oxide, mucus plugs, and functional respiratory imaging in patients with type 2 asthma (VESTIGE): a randomised, double-blind, placebo-controlled, phase 4 trial. Lancet Respir Med. 2025, 13, 208–220. [Google Scholar] [CrossRef]
- De Corso, E.; Settimi, S.; Montuori, C.; et al. Effectiveness of dupilumab in the treatment of severe uncontrolled chronic rhinosinusitis with nasal polyps (CRSwNP): A multicentric real-life study. Int Forum Allergy Rhinol. 2022, 12, 1266–1275. [Google Scholar]
- De Corso, E.; Pasquini, E.; Trimarchi, M.; et al. Dupilumab in the treatment of severe uncontrolled chronic rhinosinusitis with nasal polyps (CRSwNP): A multicentric observational Phase IV real-life study (DUPIREAL). Allergy 2023, 78, 2683–2693. [Google Scholar] [CrossRef]
- Tajiri, T.; Suzuki, M.; Nishiyama, H.; et al. Dupilumab-induced remission in chronic rhinosinusitis with nasal polyps and comorbid asthma: a 24-month study. J Clin Med. 2025, 14, 3654. [Google Scholar] [CrossRef]
- De Corso, E.; Montuori, C.; Pipolo, C.; et al. Two-Year Turning Point With Dupilumab in CRSwNP: Control, Remission, and Tapering Dosage. Allergy 2025. [Google Scholar] [CrossRef]
- De Corso, E.; Canonica, G.W.; Heffler, E.; et al. Dupilumab versus omalizumab in patients with chronic rhinosinusitis with nasal polyps and coexisting asthma (EVEREST): a multicentre, randomised, double-blind, head-to-head phase 4 trial. Lancet Respir Med. 2025, 13, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Estravís, M.; Carreiras-Quintas, D.; Triviño, J.C.; et al. Identification of Dual Super-Response in Patients With Asthma and CRSwNP Treated With Mepolizumab. J Investig Allergol Clin Immunol. 2025. [Google Scholar] [CrossRef]
- Jommi, C.; Cipriani, F.; Fanelli, F.; et al. The effects of disease cross-coverage by dupilumab on the costs sustained by the Italian National Health Service. Glob Reg Health Technol Assess. 2020, 7, 33–39. [Google Scholar] [CrossRef] [PubMed]
- De Corso, E.; Furneri, G.; Salsi, D.; et al. Cost-Utility Analysis of Dupilumab for the Treatment of Chronic Rhinosinusitis with Nasal Polyps (CRSwNP) in Italy. J Pers Med. 2022, 12, 951. [Google Scholar] [CrossRef]
- Oh, S.H.; Rhee, C.K.; Bae, E.J.; et al. Cost-effectiveness analysis of dupilumab among patients with uncontrolled severe asthma using LIBERTY ASTHMA QUEST Korean data. Health Econ Rev. 2024, 14, 67. [Google Scholar] [CrossRef]
- Yong, M.; Kirubalingam, K.; Desrosiers, M.Y.; et al. Cost-effectiveness analysis of biologics for the treatment of chronic rhinosinusitis with nasal polyps in Canada. Allergy Asthma Clin Immunol. 2023, 19, 90. [Google Scholar] [CrossRef]
| Variable | Value (N=28) |
| Demographics | |
| Age, years | 53.50 (48.75 – 61.25) |
| Sex, n (%) | Male: 17 (61%) / Female: 11 (39%) |
| BMI, kg/m2 | 24.75 (22.75 – 28.00) |
| Disease history & severity | |
| Asthma severity, n (%) | |
| Severe asthma | 8 (29%) |
| Mild-Moderate asthma | 20 (71%) |
| Asthma duration, years | 12.00 (2.00 – 21.75) |
| CRSwNP duration, years | 12.50 (8.00 – 20.50) |
| Comorbidities (NSAID-ERD), n (%) | 12 (42.9%) |
| Baseline clinical metrics | |
| Blood eosinophils, cells/µL | 525.00 (400.00 – 652.50) |
| Total IgE, IU/mL | 164.50 (101.75 – 324.25) |
| FEV1, L | 3.12 (2.56 – 3.61) |
| ACT score | 19.00 (16.50 – 24.00) |
| SNOT-22 score | 67.00 (54.50 – 80.00) |
| Nasal Polyp Score (NPS) (0-8) | 6.00 (6.00 – 6.00) |
| Daily OCS, mg/day | 1.40 (0.00 – 2.90) |
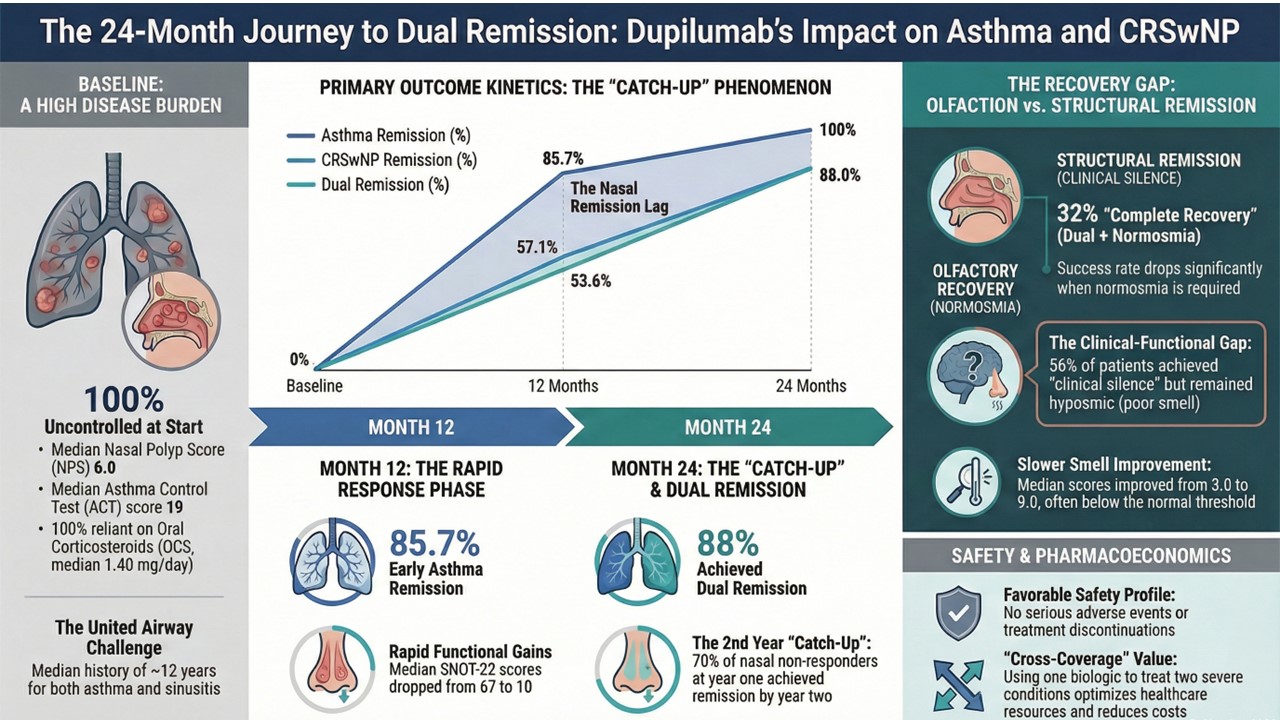
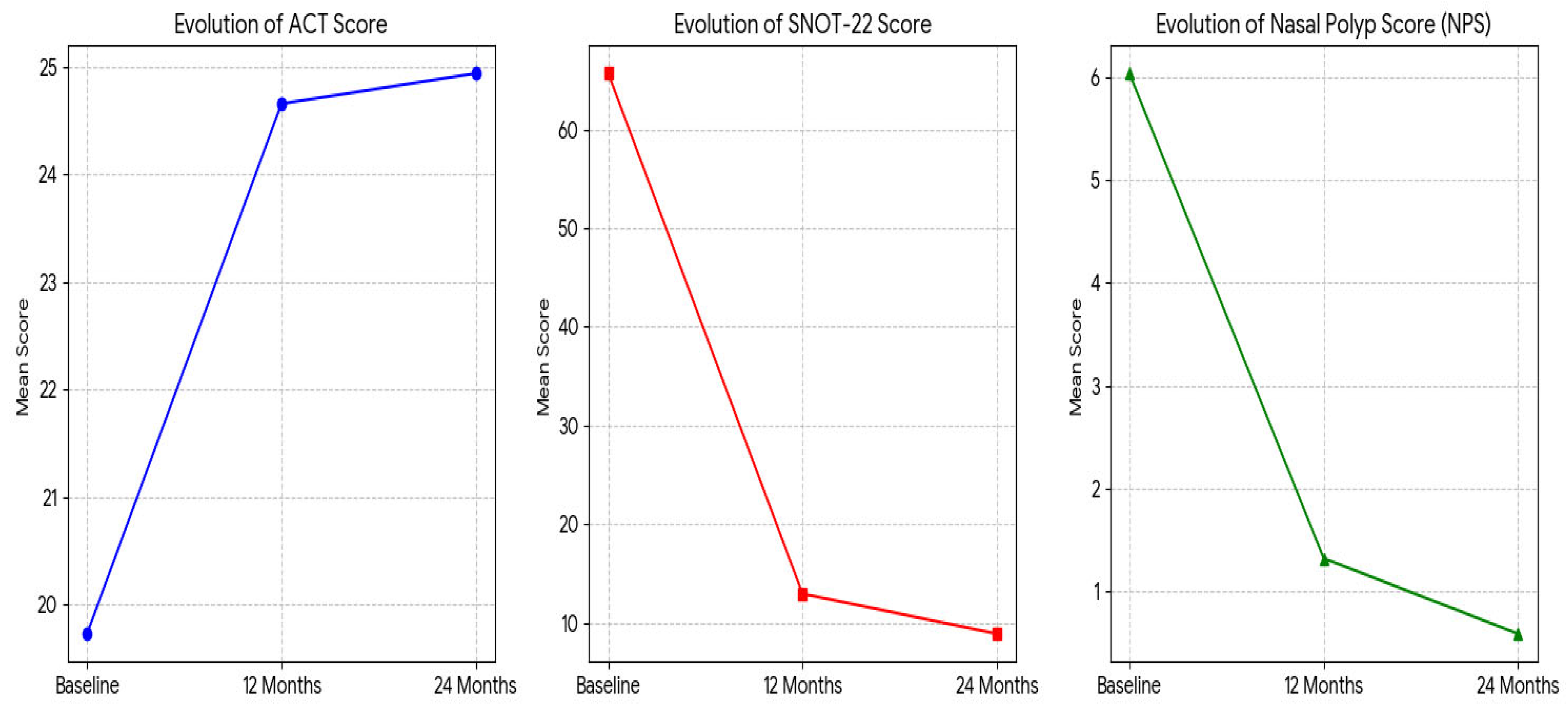
| Parameter | Baseline | 12 Months | 24 Months | p-value(12m vs baseline) | p-value(24m vs baseline) |
| Clinical control | |||||
| ACT score | 19 (16.5 – 24) | 25 (25 – 25) | 25 (25 – 25) | <0.001 | 0.007 |
| SNOT-22 score | 67 (54.5 – 80) | 10 (5– 15) | 5 (3 – 13.25) | <0.001 | <0.001 |
| NPS (0-8) | 6 (6 – 6) | 0.5 (0 – 2) | 0 (0 – 0) | <0.001 | <0.001 |
| Sniffin’ Stick score | 3 (3 – 3) | 7 (5.25 – 9.75) | 9 (5 – 11) | <0.001 | <0.001 |
| OCS (mg/day) | 1.4 (0.0 – 2.9) | 0.0 (0.0 – 0.0) | 0.0 (0.0 – 0.0) | <0.001 | <0.001 |
| Lung function | |||||
| FEV1 (L) | 3.12 (2.56 – 3.61) | 3.50 (3.05 – 3.83) | 3.50 (3.11 – 3.91) | 0.064 | 0.018 |
| FEV1 (% predicted) | 94.00 (84.00 – 111.8) | 102.50 (90.25 – 111.3) | 109.00 (101.5 – 117.8) | 0.127 | 0.017 |
| FVC (L) | 4.53 (3.87 – 5.21) | 4.42 (4.37 – 5.99) | 4.57 (4.13 – 5.40) | 0.433 | 0.330 |
| FVC (% predicted) | 112.00 (102.5 – 120.5) | 116.50 (109.3 – 121.5) | 117.50 (108.5 – 123.0) | 0.426 | 0.469 |
| FEF25-75 (L/s) | 1.76 (1.27 – 2.88) | 2.34 (1.45 – 2.83) | 2.74 (1.74 – 2.95) | 0.231 | 0.635 |
| FEF25-75 (% pred) | 54.00 (41.00 – 92.50) | 70.00 (51.50 – 88.75) | 88.00 (66.50 – 114.5) | 0.071 | 0.569 |
| Lung volumes | |||||
| RV (L) | 1.86 (1.35 – 2.56) | 1.79 (1.47 – 2.07) | 1.87 (1.46 – 2.17) | 0.022 | 1.000 |
| TLC (L) | 5.92 (4.84 – 6.73) | 6.15 (5.77 – 6.93) | 6.19 (5.85 – 7.38) | 0.241 | 0.042 |
| Biomarkers | |||||
| Blood eosinophils (/µL) | 525 (400 – 653) | 535 (405 – 1195) | 660 (140 – 1033) | 0.053 | 0.229 |
| FeNO (ppb) | 23.00 (22.00 – 90.00) | 37.00 (28.00 – 48.00) | 25.00 (19.00 – 30.00) | 0.500 | 1.000 |
| Parameter | Baseline (N=28) | 12 Months (N=28) | 24 Months (N=25) |
| Hypereosinophilia | |||
| Eosinophils ≥ 1,500 cells/µL, n (%) | 0 (0%) | 3 (11%) | 1 (6%) |
| Eosinophils > 3,000 cells/µL, n (%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Adverse events, n (%) | |||
| Serious infections* | 0 (0%) | 0 (0%) | 0 (0%) |
| Discontinuation due to AE | — | 0 (0%) | 0 (0%) |
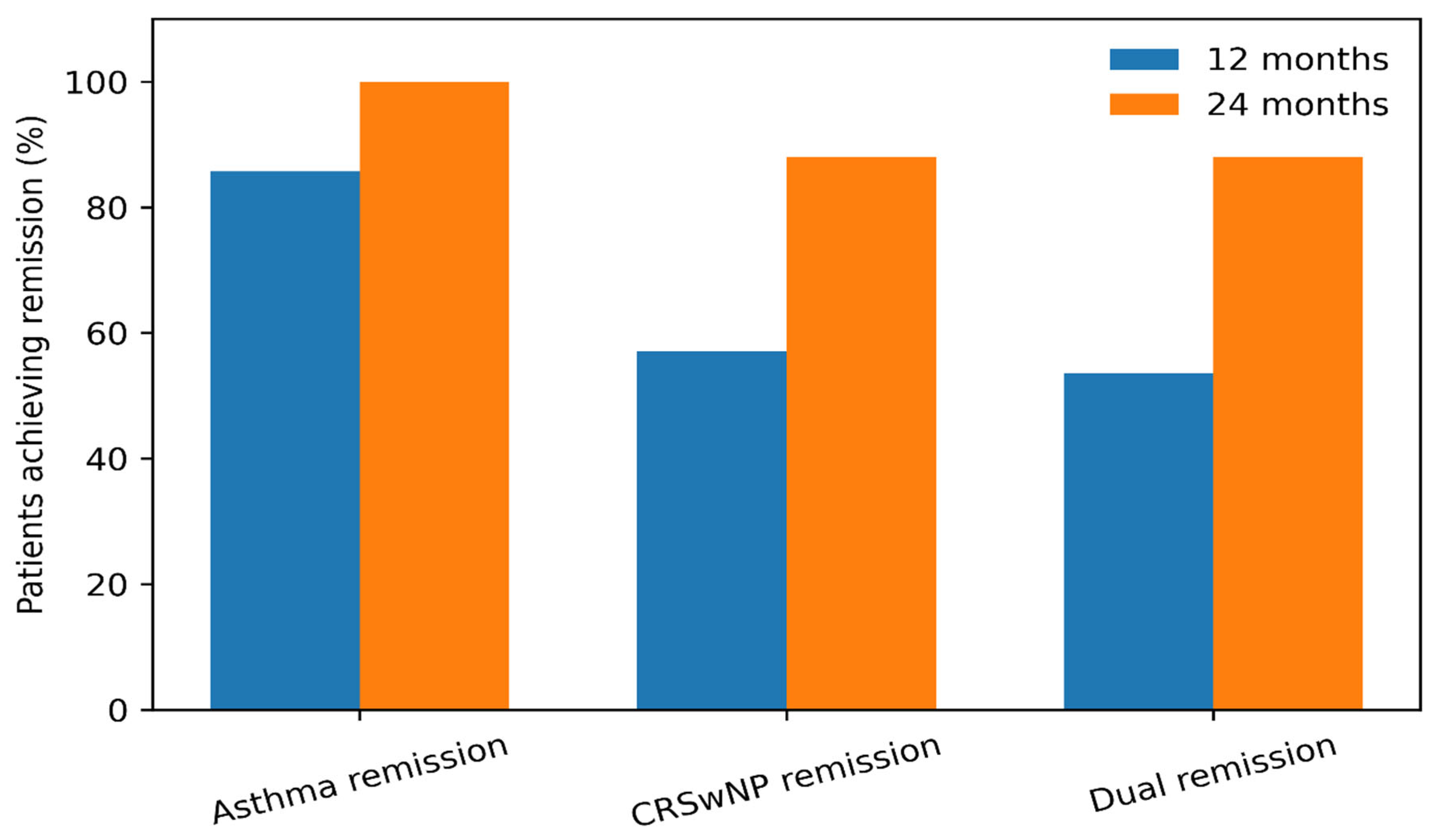
| Outcome measure | Baseline (N=28) | T12 (12 months) (N=28) | T24 (24 months) (N=25) | p-value (T12 vs baseline) | p-value (T24 vs baseline) |
| 1. Clinical remission outcomes | |||||
|
Asthma remission (ACT ≥ 20 + No OCS + No Exacerbations) |
0% | 24/28 (85.7%) | 25/25 (100%) | < 0.001 | < 0.001 |
|
CRSwNP remission (NPS ≤ 1 + SNOT-22 < 40) |
0% | 16/28 (57.1%) | 22/25 (88.0%) | < 0.001 | < 0.001 |
|
Dual Remission (Achieved both above) |
0% | 15/28 (53.6%) | 22/25 (88.0%) | < 0.001 | < 0.001 |
| 2. Olfactory outcomes | |||||
| Sniffin’ sticks score (Median) | 3.0 | 7.0 | 9.0 | < 0.001* | < 0.001* |
|
Olfactory recovery (Normosmia: Score ≥ 12) |
0% | < 10% (Est.) | 8 (32.0%) | ns | < 0.01 |
| 3. Combined “Complete” recovery | |||||
| Dual remission + normosmia | 0% | -- | 8 (32.0%) | -- | < 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





