1. Introduction
In present-day military conflicts, extremity combat wounds (ECW) represent the most common type of injury, largely due to the widespread use of personal protective equipment that reduces fatal torso trauma. Musculoskeletal injuries account for approximately 50% of all combat wounds, with explosive mechanisms responsible for more than 75% of casualties reported during military operations in Iraq and Afghanistan [
1,
2]. In a large cohort comprising 3575 ECW from these conflicts, penetrating soft-tissue injuries constituted 53% of cases, while fractures accounted for 26%, of which 82% were open fractures. Fractures were equally distributed between the upper and lower limbs, with nearly half of lower extremity fractures involving the tibia and fibula [
2].
Similar patterns have been observed in the ongoing armed conflict in Ukraine. According to recent reports, approximately 40% of combat injuries involve fractures of long tubular bones, 30% affect the soft tissues of the extremities, and the remaining 30% involve other anatomical regions. Among casualties with extremity injuries, approximately 10% experience massive hemorrhage requiring urgent hemostatic intervention [
3]. A recent retrospective study analyzing data from 2014 to 2022 and including 2496 casualties with limb injuries requiring tourniquet application reported lower extremity involvement in 84.4% of cases. Tourniquet application time (TAT) ranged from 50 to 380 minutes, with a mean duration of 205.9 minutes. Notably, reported mortality and limb amputation rates were relatively low at 4.0% and 3.7%, respectively [
4].
Critical bleeding in ECW is defined as rapid blood loss leading to progressive and potentially fatal hypovolemic shock. Such bleeding most commonly results from injury to major arterial vessels or complex multiple vascular damage. Immediate and effective hemorrhage control is therefore essential. For example, a casualty with femoral artery injury may lose consciousness within one minute and die shortly thereafter if effective hemostasis is not achieved [
5].
Hemostatic tourniquets represent a reliable and effective method for rapid control of life-threatening extremity hemorrhage. In the Ukrainian Armed Forces, each soldier is equipped with a tourniquet and trained in its application for self-aid or buddy-aid, allowing hemorrhage control to be initiated directly on the battlefield. This practice has significantly reduced immediate combat fatalities. However, prolonged tourniquet application is associated with substantial secondary tissue injury. Experimental and clinical data indicate that irreversible ischemic tissue damage may begin approximately three hours after complete interruption of blood flow [
6].
According to established military medical procedures, tourniquet conversion or temporary loosening is recommended every 2–3 hours to mitigate ischemic injury when feasible. Information regarding tourniquet placement time and any conversion attempts is documented on a casualty card accompanying the wounded soldier throughout evacuation and treatment. During the study period, however, tourniquet manipulation was permitted exclusively for physicians, particularly in cases of prolonged TAT. As a result, recommended periodic tourniquet conversion was frequently not performed due to delayed evacuation, limited availability of medical personnel, and ongoing combat conditions. Consequently, continuous tourniquet application often persisted for several hours and in some cases exceeded six hours.
Prolonged limb ischemia results in critical disturbances of oxygen delivery and metabolic waste elimination, triggering inflammatory cascades and cellular injury. Severe hypoperfusion with hypoxia and hypercapnia leads to increased concentrations of vasoactive mediators, including histamine, serotonin, and tumor necrosis factor, with subsequent endothelial dysfunction and increased vascular permeability [
7]. Fluid shifts into the interstitium cause tissue edema, while hypoxia, oxidative stress, and metabolic derangements disrupt cellular osmotic balance. These processes culminate in cellular swelling, membrane dysfunction, necrosis, and increased intracompartmental pressure, ultimately leading to acute extremity compartment syndrome (AECS) [
8].
In combat settings, additional factors may exacerbate tissue injury. Although soldiers are trained in tourniquet application, they are not medical professionals, and improper application is common. In many cases, insufficient arterial occlusion with preserved venous inflow occurs, resulting in venous congestion and increased intrafascial pressure [
9]. Combined with external compression commonly used in combat wound management, these factors further promote AECS, muscle necrosis, and the development of rhabdomyolysis (RM). This initiates a vicious cycle leading to acute kidney injury (AKI), limb amputation, and potentially death.
Timely fasciotomy reduces intracompartmental pressure, improves tissue perfusion, facilitates drainage of toxic edema fluid, and mitigates ischemic injury. According to current regulations in the Ukrainian Armed Forces, fasciotomy is performed upon first contact with qualified surgical personnel, typically at advanced surgical units (ASU). Tourniquet removal is subsequently performed, resulting in limb reperfusion.
Reperfusion of ischemic muscle leads to systemic release of muscle breakdown products, including myoglobin, potassium, phosphate, and inflammatory mediators. This process may result in metabolic acidosis, electrolyte disturbances, myoglobinemia, myoglobinuria, and multiple organ dysfunction, with acute kidney injury representing the most clinically significant complication [
1,
10]. Rhabdomyolysis has been reported in approximately 25–30% of wounded soldiers during recent military operations, with injured soldiers being three to four times more likely to develop RM than civilian trauma patients [
11,
12]. Identified risk factors include extremity injury, explosive mechanisms, high injury severity scores, shock index greater than 0.9, and delayed evacuation. Importantly, the presence of RM more than doubles the risk of AKI [
12].
Free circulating myoglobin is the principal pathological mediator of RM-induced AKI. Its nephrotoxic effects include direct tubular cytotoxicity, intratubular cast formation with uromodulin, oxidative injury mediated by free iron, and renal vasoconstriction [
2,
7,
8]. Prevention of AKI therefore remains the cornerstone of RM management and is primarily achieved through early restoration of intravascular volume, maintenance of renal perfusion, and dilution of intratubular myoglobin. However, the optimal timing for initiating preventive therapy, particularly in the absence of overt clinical signs of RM, remains uncertain [
9,
10].
In current Ukrainian military practice, intensive infusion therapy—including crystalloids, osmotically active solutions, and urine alkalization—is initiated once clinical signs of RM become evident. Prior to tourniquet removal, however, such signs are typically absent, and clear indications for therapy initiation are lacking. Whether initiating anti-rhabdomyolysis infusion therapy before reperfusion and before the appearance of clinical RM manifestations confers clinical benefit remains an open and clinically relevant question.
Therefore, the aim of this study was to evaluate the impact of early initiation of infusion therapy relative to tourniquet removal on the risk of death, dialysis-requiring acute kidney injury, and limb amputation in casualties with lower extremity combat wounds complicated by rhabdomyolysis.
2. Materials and Methods
2.1. Study Design and Setting
This retrospective single-center observational study was conducted at a military medical facility providing surgical and intensive care to casualties with combat-related injuries. The study period included patients treated during active hostilities. This study analyzed the medical records of 120 wounded Ukrainian Army soldiers who were treated in medical units of the Zaporizhzhia Military Hospital from 01/06/2023 to 31/08/2023. The assumptions and plan of the research were discussed and approved by the Ethics Committee of the Ternopil National Medical University, Ukraine, on May 25, 2023.
The study analyzed medical records of casualties with lower extremity combat wounds requiring prolonged tourniquet application and subsequent surgical management. Patients were managed according to local clinical protocols under wartime conditions.
2.2. Study Population
Eligible patients met the following inclusion criteria:
combat-related injury to the lower extremity;
application of a hemostatic tourniquet prior to hospital admission;
prolonged tourniquet application time (TAT);
surgical treatment including fasciotomy and tourniquet removal;
availability of clinical and laboratory data for analysis.
Patients with incomplete medical records or missing key outcome data were excluded from the analysis.
2.3. Treatment Strategy
Patients were divided into two groups according to the infusion strategy applied prior to tourniquet removal.
In the standard treatment group, infusion therapy was initiated after tourniquet removal and the appearance of clinical or laboratory signs of rhabdomyolysis.
In the early anti-rhabdomyolysis treatment group, intensive infusion therapy was initiated before tourniquet removal, during the ischemic phase, as a preventive measure. The infusion protocol included crystalloids, osmotically active solutions, and urine alkalization according to local practice.
Allocation to treatment groups reflected differences in organizational and protocol-based practice between comparable medical units and time periods, rather than individual patient selection.
Importantly, baseline demographic characteristics, tourniquet application time, and shock index at admission did not differ significantly between groups, suggesting comparable initial injury severity.
2.4. Data Collection
Demographic, clinical, and laboratory data were extracted from medical records. Collected variables included age, tourniquet application time, shock index at admission (SI-1) and after initial resuscitation (SI-2), laboratory markers of rhabdomyolysis and renal function, including urinary hemoglobin/myoglobin and serum creatinine at 24 hours (Cr-24). Urinary hemoglobin was assessed at admission (Hb-1), 30 minutes (Hb-2), and 24 hours (Hb-3). Urinary pH was recorded at admission (pH-1), and at subsequent time points within the first 24 hours (pH-2 and pH-3).
2.5. Outcomes
The primary outcomes were:
Secondary outcomes included changes in shock index and laboratory markers associated with rhabdomyolysis.
2.6. Statistical Analysis
Continuous variables were tested for normality using visual inspection and distribution assessment (histograms and Q–Q plots). Statistical analysis was performed using Python (statsmodels and scikit-learn libraries).
Between-group comparisons for continuous variables were performed using the Mann–Whitney U test.
Categorical variables are presented as counts and percentages and were compared using the χ² test or Fisher’s exact test, as appropriate.
Univariable logistic regression was used to estimate crude odds ratios (OR) with 95% confidence intervals (CI) for the association between early infusion therapy and each clinical outcome. Multivariable logistic regression models were constructed to adjust for baseline physiological severity and included treatment group, shock index at admission, and baseline urinary pH.
Model performance was assessed using the Akaike Information Criterion (AIC), with lower values indicating improved model fit. Discriminative ability was evaluated using receiver operating characteristic (ROC) curves and the area under the curve (AUC).
As a sensitivity analysis to address potential confounding by indication, propensity scores were estimated using baseline covariates (age, tourniquet application time, shock index at admission, baseline urine pH, and baseline urinary hemoglobin). Stabilized inverse probability of treatment weights (IPTW) were applied to achieve covariate balance (standardized mean differences < 0.10). Weighted logistic regression models were then fitted to estimate the association between early infusion therapy and each outcome.
All statistical tests were two-sided, and a p-value < 0.05 was considered statistically significant.
3. Results
3.1. Baseline Characteristics
Baseline demographic and clinical characteristics of the study groups are presented in
Table 1. The groups were comparable in terms of age, duration of tourniquet application (TAT), and shock index at admission (SI-1), indicating a similar initial severity of injury. No statistically significant differences were observed in baseline urinary pH (pH-1).
After initiation of treatment and prior to reperfusion, patients receiving early anti-rhabdomyolysis infusion therapy demonstrated significantly lower shock index values after initial resuscitation (SI-2) compared with the standard treatment group (p < 0.001). Laboratory dynamics revealed significantly higher urinary hemoglobin levels at 30 minutes (Hb-2) in the early-treatment group (p = 0.027), while no significant differences were observed at 24 hours (Hb-3). Importantly, serum creatinine measured 24 hours after admission (Cr-24) was significantly lower in the early-treatment group (p = 0.007).
3.2. Clinical Outcomes
Clinical outcomes are summarized in
Table 2. Patients treated with the early infusion protocol had lower rates of dialysis-requiring acute kidney injury, limb amputation, and death compared with those receiving standard treatment.
3.3. Logistic Regression Analysis
Results of univariable and multivariable logistic regression analyses are presented in
Table 3. In crude (unadjusted) models, early anti-rhabdomyolysis infusion therapy was not significantly associated with dialysis or limb amputation, while a non-significant trend toward reduced mortality was observed.
After adjustment for baseline physiological severity, including shock index at admission and baseline acid–base status, early infusion therapy emerged as an independent protective factor for all studied outcomes. Adjusted analysis demonstrated a significant reduction in the odds of dialysis, amputation, and death compared with standard treatment.
Model performance improved after adjustment, as reflected by lower Akaike Information Criterion (AIC) values across all outcomes, indicating better model fit.
As a sensitivity analysis to address potential confounding by indication, propensity score–based inverse probability of treatment weighting (IPTW) was applied using baseline covariates. The IPTW analysis yielded effect estimates consistent with the primary multivariable models, supporting the robustness of the observed associations.
3.4. Model Discrimination
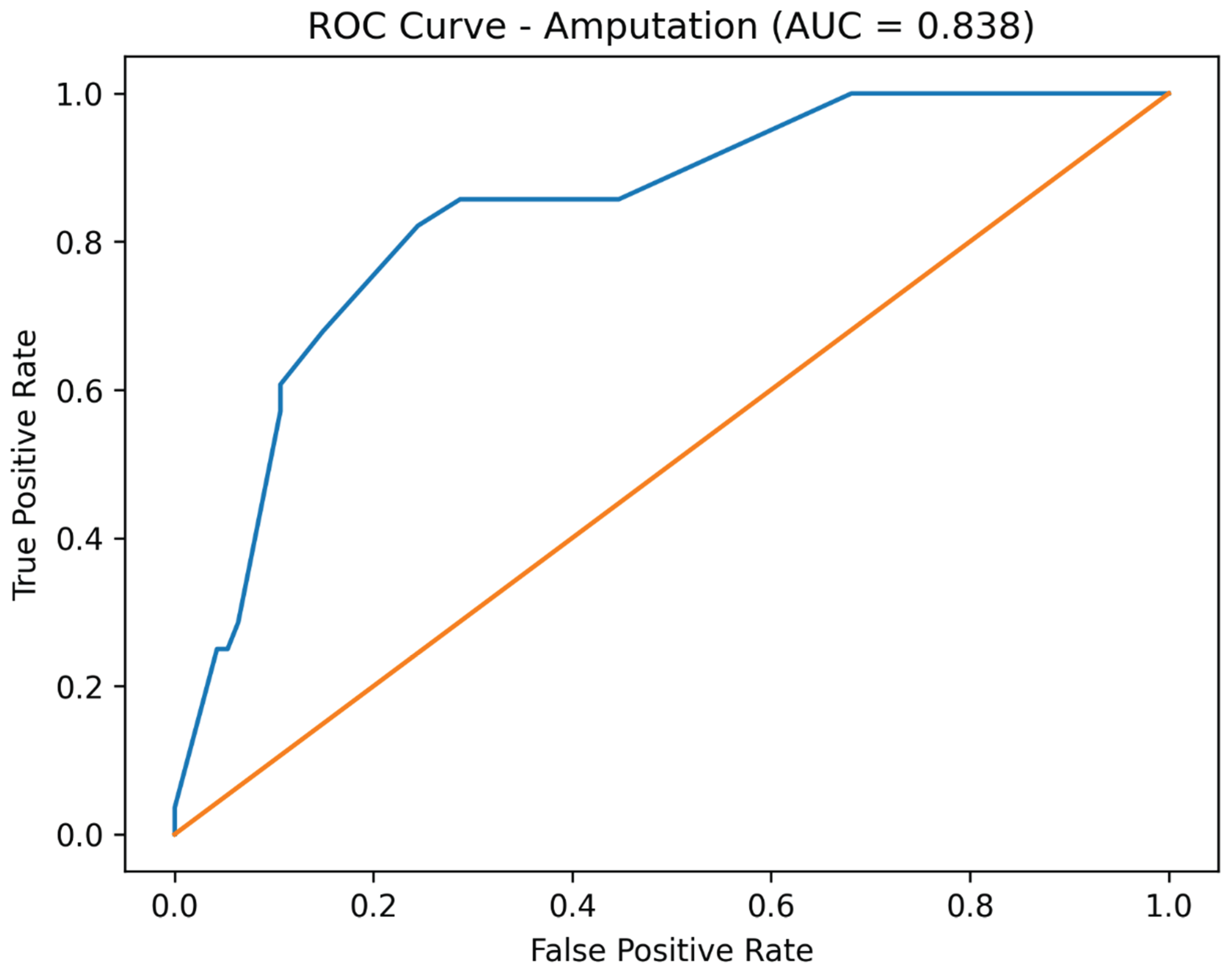
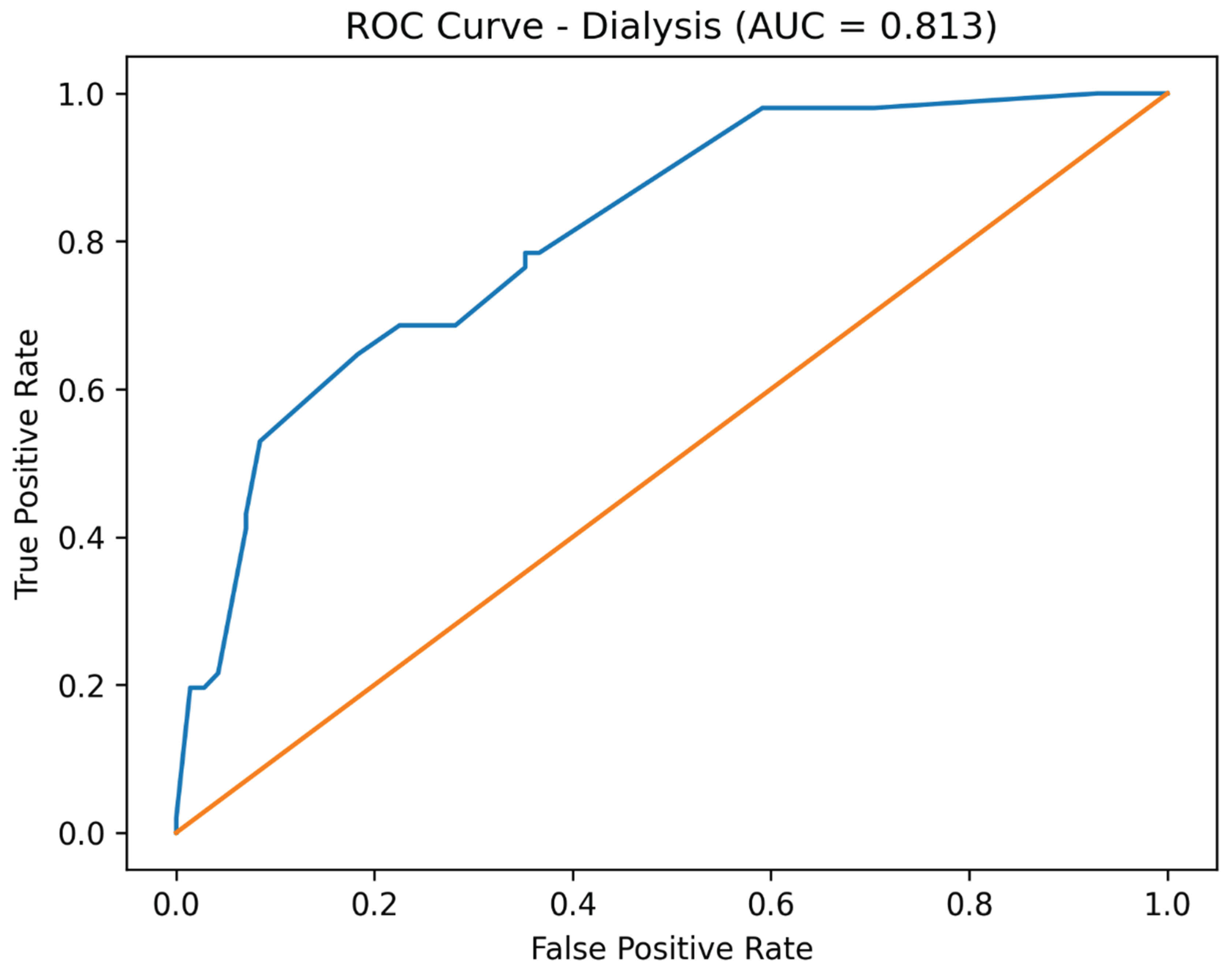
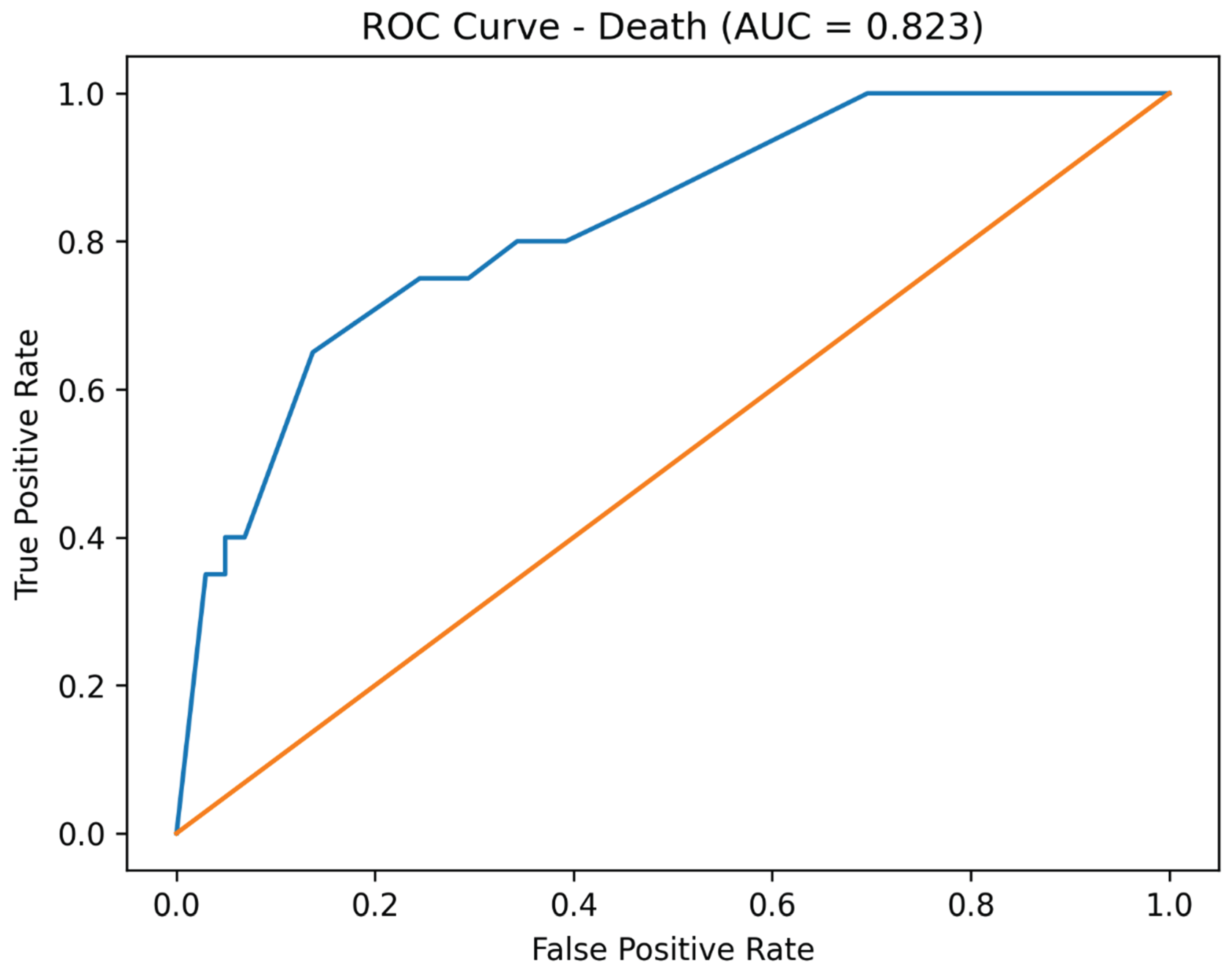
Receiver operating characteristic (ROC) curve analysis demonstrated good discriminative performance of the adjusted logistic regression models. The area under the curve (AUC) was 0.813 for dialysis, 0.838 for amputation, and 0.823 for mortality (
Figure 1,
Figure 2 and
Figure 3).
4. Discussion
The present study provides additional evidence supporting the clinical relevance of early targeted anti-rhabdomyolysis infusion therapy in patients with combat-related lower extremity injuries requiring prolonged tourniquet application. Using updated statistical modeling and expanded outcome analysis, we demonstrate that initiation of infusion therapy prior to tourniquet removal is independently associated with reduced odds of dialysis-requiring acute kidney injury, limb amputation, and mortality after adjustment for baseline physiological severity.
Rhabdomyolysis remains a frequent and potentially devastating complication of severe trauma, particularly in military and disaster settings characterized by extensive muscle injury, blast mechanisms, and prolonged ischemia [
1,
2,
3]. In contrast to civilian trauma, combat casualties are exposed to a combination of massive soft-tissue destruction, delayed evacuation, and prolonged tourniquet use, all of which significantly increase the risk of muscle necrosis and systemic toxicity [
4,
5,
6]. Acute kidney injury represents the most clinically significant systemic consequence of rhabdomyolysis and is consistently associated with increased morbidity, mortality, and resource utilization [
2,
7].
The pathophysiology of rhabdomyolysis-associated AKI is complex and multifactorial. Myoglobin released from damaged skeletal muscle plays a central role through direct tubular toxicity, intratubular cast formation with uromodulin, oxidative injury mediated by free iron, and renal vasoconstriction leading to medullary hypoxia [
2,
6,
7,
8,
9]. Experimental studies have demonstrated that acidic urinary conditions promote myoglobin precipitation and amplify oxidative stress, thereby exacerbating tubular injury [
8]. These mechanisms provide a strong biological rationale for early interventions aimed at optimizing renal perfusion, urine flow, and urinary pH before systemic exposure to myotoxic metabolites.
In the context of prolonged tourniquet application, the ischemic limb initially functions as a relatively isolated compartment, limiting systemic dissemination of myoglobin and other intracellular breakdown products [
4,
10]. While this effect may transiently protect vital organs, reperfusion following tourniquet release results in an abrupt systemic surge of myoglobin, potassium, lactate, and inflammatory mediators. Clinical and experimental data suggest that ischemia exceeding 3–6 hours substantially increases the likelihood of irreversible muscle injury and severe reperfusion-associated toxicity [
4,
11,
12,
13].
Consistent with previous observations, overt clinical signs of rhabdomyolysis were often absent prior to tourniquet removal in our cohort. Urinary discoloration and laboratory abnormalities typically appeared only after reperfusion, underscoring the limited value of relying on visible clinical markers to guide preventive interventions [
1,
2]. This temporal dissociation supports the concept that prophylactic therapy initiated before reperfusion may be more effective than reactive treatment after systemic injury has already occurred.
In unadjusted analyses, differences in adverse outcomes between treatment groups were not uniformly statistically significant. However, after adjustment for baseline shock index and metabolic status, early infusion therapy emerged as an independent protective factor across all studied outcomes. This finding highlights the importance of accounting for physiological severity when evaluating treatment effects in heterogeneous trauma populations. Similar masking of therapeutic effects by baseline instability has been described in other trauma and critical care studies [
6,
7].
Importantly, model performance improved substantially after adjustment, as demonstrated by marked reductions in AIC values and acceptable-to-good discrimination on ROC analysis. These results suggest that the observed associations are not merely statistical artifacts but reflect meaningful prognostic information captured by the multivariable models.
The reduction in dialysis-requiring AKI observed in the early-treatment group is of particular clinical significance. Acute kidney injury not only worsens short-term outcomes but also increases the risk of long-term renal dysfunction and mortality [
7,
14]. In wartime and disaster settings, where access to renal replacement therapy may be limited or delayed, prevention of severe AKI represents a critical therapeutic objective [
5,
14]. Our findings support the concept that early renal protective strategies may reduce the burden of dialysis-dependent renal failure in such environments.
The role of bicarbonate and mannitol therapy in rhabdomyolysis remains controversial. While aggressive crystalloid resuscitation is universally recommended as the cornerstone of management [
1,
9], systematic reviews and meta-analyses have produced inconsistent conclusions regarding the additional benefit of urine alkalization and osmotic diuresis [
9,
10]. Some authors report no clear advantage beyond early fluid therapy, whereas others emphasize potential benefits in selected high-risk patients with severe ischemia-reperfusion injury [
8,
15]. The protocol evaluated in the present study does not allow isolation of the individual contribution of each component; however, the observed associations suggest that the combined early approach may be beneficial when initiated before reperfusion.
Current trauma and military guidelines emphasize hemorrhage control and early volume resuscitation but do not uniformly address prophylactic strategies for rhabdomyolysis-associated AKI in the setting of prolonged tourniquet use [
15,
16]. In this context, the treatment approach evaluated in our study reflects an adaptation driven by frontline clinical experience and pathophysiological reasoning rather than formal guideline recommendations.
Several limitations must be acknowledged. This was a retrospective single-center study with a relatively limited sample size, and residual confounding cannot be fully excluded despite multivariable adjustment. Follow-up was limited to the early post-injury period, and long-term renal outcomes were not assessed. Additionally, variations in evacuation time and prehospital care could not be fully accounted for. Nevertheless, the consistency of findings across clinical endpoints, regression modeling, and predictive performance metrics strengthens the internal validity of the results.
In conclusion, this study suggests that in combat-related lower extremity injuries requiring prolonged tourniquet application, early initiation of targeted anti-rhabdomyolysis infusion therapy prior to reperfusion is associated with reduced renal complications, limb loss, and mortality. These findings support further prospective, multicenter investigation to confirm efficacy and to refine optimal preventive strategies in both military and civilian mass-casualty settings.
Limitations
This study has several limitations. Its retrospective single-center design precludes causal inference and may be subject to residual confounding despite multivariable adjustment and sensitivity analyses. The sample size was relatively limited, which may have affected the precision of some estimates. In addition, treatment allocation was not randomized and reflected organizational differences in clinical practice. Finally, long-term renal and functional outcomes were not assessed.
5. Conclusions
In this retrospective cohort study of combat-related lower extremity injuries requiring prolonged tourniquet application, early initiation of anti-rhabdomyolysis infusion therapy prior to tourniquet release was associated with reduced risks of dialysis-requiring acute kidney injury, limb amputation, and death. These associations remained consistent after adjustment for baseline physiological severity and in propensity score–based sensitivity analyses.
The findings highlight the potential clinical importance of the timing of infusion therapy relative to reperfusion in patients at high risk of ischemia–reperfusion injury and rhabdomyolysis. Early preventive strategies aimed at renal protection before systemic release of myotoxic metabolites may represent a feasible and clinically relevant approach in military and mass-casualty settings.
Further prospective, multicenter studies are warranted to confirm these observations and to define optimal protocols for the prevention of rhabdomyolysis-associated complications following prolonged tourniquet application.
Funding
This research was supported by the statutory subsidy of Wroclaw Medical University, Poland, no. SUBZ.A120.26.014.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki. Due to the retrospective design and the use of anonymized clinical data collected under wartime conditions, formal ethical approval was waived according to local regulations.
Informed Consent Statement
Patient consent was waived due to the retrospective nature of the study and the use of anonymized data.
Data Availability Statement
The data presented in this study are available on reasonable request from the corresponding author.
Acknowledgments
The authors acknowledge the use of an artificial intelligence–based language tool (ChatGPT, OpenAI, San Francisco, CA, USA) for assistance with English translation. The authors reviewed and approved all content.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Huerta-Alardín AL, Varon J, Marik PE. Bench-to-bedside review: Rhabdomyolysis—an overview for clinicians. Crit Care. 2005;9(2):158–169. [CrossRef]
- Bosch X, Poch E, Grau JM. Rhabdomyolysis and acute kidney injury. N Engl J Med. 2009;361(1):62–72. [CrossRef]
- McMahon GM, Zeng X, Waikar SS. A risk prediction score for kidney failure or mortality in rhabdomyolysis. JAMA Internal Medicine. 2013;173(19):1821–1828. [CrossRef]
- Gibney RN, Sever MS, Vanholder RC. Disaster nephrology: crush injury and beyond. Kidney Int. 2014;85(5):1049–1057. [CrossRef]
- Sever MS, Vanholder R. Recommendations for the management of crush victims in mass disasters. Nephrol Dial Transplant. 2012;27(Suppl 1):i1–i67. [CrossRef]
- Zutt R, van der Kooi AJ, Linthorst GE, Wanders RJ, de Visser M. Rhabdomyolysis: review of the literature. Neuromuscul Disord. 2014;24(8):651–659. [CrossRef]
- Petejova N, Martinek A. Acute kidney injury due to rhabdomyolysis and renal replacement therapy: a critical review. Crit Care. 2014;18(3):224. [CrossRef]
- Zager RA. Rhabdomyolysis and myohemoglobinuric acute renal failure. Kidney Int. 1996;49(2):314–326. [CrossRef]
- Brown CV, Rhee P, Chan L, Evans K, Demetriades D, Velmahos GC. Preventing renal failure in patients with rhabdomyolysis: do bicarbonate and mannitol make a difference? J Trauma. 2004;56(6):1191–1196.
- Scharman EJ, Troutman WG. Prevention of kidney injury following rhabdomyolysis: a systematic review. Ann Pharmacother. 2013;47(1):90–105.
- Sawhney S, Fraser SD, Brar S, et al. Long-term prognosis after acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis. 2015;65(4):551–567. [CrossRef]
- Melli G, Chaudhry V, Cornblath DR. Rhabdomyolysis: an evaluation of 475 hospitalized patients. Medicine (Baltimore). 2005;84(6):377–385. [CrossRef]
- Better OS. The crush syndrome revisited. Kidney Int. 1997;51(6):1901–1910. [CrossRef]
- Rossaint R, Afshari A, Bouillon B, Cerny V, Cimpoesu D, Curry N, et al. The European guideline on management of major bleeding and coagulopathy following trauma: sixth edition. Crit Care. 2023;27(1):80. [CrossRef]
- American College of Surgeons Committee on Trauma. ATLS® Advanced Trauma Life Support Student Course Manual. 10th ed. Chicago (IL): American College of Surgeons; 2018.
- U.S. Department of Defense. Tactical Combat Casualty Care (TCCC) Guidelines. Washington (DC): Department of Defense; 2024.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).