Submitted:
17 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- population: preterm infants, ranging from extremely extremely preterm (<28 weeks gestational age) to late preterm (<34-37 weeks gestational age);
- diagnostic tools: HUS, MRI, or head circumference (HC) measurements;
- assessments: correlate imaging findings with standardized functional tools, such as GMs Prechtl analysis;
- timeframe: studies from the last 5 years.
- Studies were excluded if they met any of the following:
- technical standards: use of outdated imaging technology or non-standard protocols;
- publication type: case reports, conference abstracts, or non-English language papers.
3. Results
3.1. Imaging Modalities and Diagnostic Sensitivity (Table 2)
| Metric | HUS | MRI | Source |
|---|---|---|---|
| CP | High specificity (96-98%), but low sensitivity (12-27%) | PPV: 60-67%. Superior for detecting subtle WM injury | Inder, T.E., et al. [9], McLean, G., et al. [13] |
| Cognitive Outcomes | Low correlation; poor at identifying diffuse injury | High negative predictive value. Low positive predictive value | Guillot, M., et al. [8], Inder, T.E., et al. [9] |
| Severe Lesions | Optimal for Grade III/IV IVH and cystic PVL | Equivalent to HUS but offers more anatomical detail | Zhang, X.H., et al. [10], Chevallier, M., et al. [15] |
| Subtle Injuries | Often missed unless a specific scoring system is used | Superior sensitivity for non-cystic WM injury and cerebellar bleeds | Guillot, M., et al. [8], Inder, T.E., et al. [9] |
| Early Predictors | Early scans (first 2 weeks) have lower predictive power | Not typically used in the acute phase for routine prognosis | Zhang, X.H., et al. [10], Helderman, J., et al. [11] |
3.2. Anthropometric and Structural Predictors of Neurodevelopment
3.3. Lesion Characteristics and Motor Outcomes
3.4. Functional Assessments and the Role of Timing
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADHD | Attention Deficit Hyperactivity Disorder |
| NICU | Neonatal Intensive Care Unit |
| VLBW | very low birth weight |
| HUS | head ultrasound |
| IVH | intraventricular hemorrhage |
| MRI | Magnetic Resonance Imaging |
| PVL | periventricular leukomalacia |
| TEA | term-equivalent age |
| AI | Artificial Intelligence |
| CP | Cerebral Palsy |
| GMs | General Movements |
| HC | head circumference |
| WM | white matter |
References
- Fletcher, J. M.; Landry, S. H.; Bohan, T. P.; Davidson, K. C.; Brookshire, B. L.; Lachar, D.; Francis, D. J. Effects of intraventricular hemorrhage and hydrocephalus on the long-term neurobehavioral development of preterm very-low-birthweight infants. Dev. Med. Child Neurol. 1997, 39 (9), 596–606.
- Davies, M. W.; Swaminathan, M.; Chuang, S. L.; Betheras, F. R. Reference ranges for the linear dimensions of the intracranial ventricles in preterm neonates. Arch. Dis. Child. Fetal Neonatal Ed. 2000, 82 (3), F218–F223.
- Leijser, L. M.; Srinivasan, L.; Rutherford, M. A.; Counsell, S. J.; Allsop, J. M.; Cowan, F. M. Structural linear measurements in the newborn brain: accuracy of cranial ultrasound compared to MRI. Pediatr. Radiol. 2007, 37 (7), 640–648.
- Inder, T. E.; Wells, S. J.; Mogridge, N. B.; Spencer, C.; Volpe, J. J. Defining the nature of the cerebral abnormalities in the premature infant: a qualitative magnetic resonance imaging study. J. Pediatr. 2003, 143 (2), 171–179.
- Mutlu, A.; Livanelioğlu, A.; Korkmaz, A. Assessment of “general movements” in high-risk infants by Prechtl analysis during early intervention period in the first year of life. Turk. J. Pediatr. 2010, 52 (6), 630–637.
- Toma, A. I. Paediatric neurology: standardization of neonatal assessment in Romania. Enfance 2023, 4 (4), 333–338.
- Kwong, A. K.; Eeles, A. L.; Spittle, A. J. Early neurodevelopmental screening: Parent perspectives from the neonatal intensive care unit. Acta Paediatr. 2021, 110 (1), 359–360.
- Guillot, M.; Sebastianski, M.; Lemyre, B. Comparative performance of head ultrasound and MRI in detecting preterm brain injury and predicting outcomes: a systematic review. Acta Paediatr. 2021, 110 (5), 1425–1432.
- Inder, T. E.; de Vries, L. S.; Ferriero, D. M.; Grant, P. E.; Ment, L. R.; Miller, S. P.; Volpe, J. J. Neuroimaging of the preterm brain: review and recommendations. J. Pediatr. 2021, 237, 276–287.
- Zhang, X. H.; Chen, W. J.; Gao, X. R.; Li, Y.; Cao, J.; Qiu, S. J. Predicting the developmental outcomes of very premature infants via ultrasound classification: a CONSORT-clinical study. Medicine 2021, 100 (15), e25421.
- Helderman, J.; O’Shea, T. M.; Dansereau, L.; Check, J.; Hofheimer, J. A.; Smith, L. M.; et al. Association of abnormal findings on neonatal cranial ultrasound with neurobehavior at neonatal intensive care unit discharge in infants born before 30 weeks’ gestation. JAMA Network Open 2022, 5 (4), e226561.
- Kumar, N.; Kumar, D.; Priyadarshi, M.; Kumar, A.; Bharti, A. K.; Nath, K. S.; Ahmad, G. S. Role of neuroimaging in preterm infants to predict neurological outcomes. Int. J. Acad. Med. Pharm. 2023, 5 (3), 660–663.
- McLean, G.; Razak, A.; Ditchfield, M.; Lombardo, P.; Malhotra, A. Evaluation of a Cranial Ultrasound Scoring System for Prediction of Abnormal Early Neurodevelopment in Preterm Infants. J. Paediatr. Child Health 2023, DOI: 10.1111/jpc.16432 (In Press).
- Toma, A. I.; Dima, V.; Alexe, A.; Rusu, L.; Nemeș, A. F.; Gonț, B. F.; et al. Correlations between head ultrasounds performed at term-equivalent age in premature neonates and general movements neurologic examination patterns. Life 2023, 14 (1), 46.
- Chevallier, M.; Barrington, K. J.; Church, P. T.; Luu, T. M.; Janvier, A. Decision-making for extremely preterm infants with severe hemorrhages on head ultrasound: Science, values, and communication skills. Semin. Fetal Neonatal Med. 2023, 28 (3), 101444.
- Mayrink, M. L. D. S.; Villela, L. D.; Méio, M. D. B. B.; Soares, F. V. M.; Abranches, A. D. D.; Nehab, S. R. G.; et al. The trajectory of head circumference and neurodevelopment in very preterm newborns during the first two years of life: a cohort study. J. Pediatr. (Rio J.) 2024, 100, 483–490.
- Toma, A. I.; Dima, V.; Rusu, L.; Nemeș, A. F.; Gonț, B. F.; Arghirescu, A.; et al. Cerebral Ultrasound at Term-Equivalent Age: Correlations with Neuro-Motor Outcomes at 12–24 Months Corrected Age. Children 2024, 12 (1), 30.
- Toma, A. I.; Dima, V.; Alexe, A.; Bojan, C.; Nemeș, A. F.; Gonț, B. F.; et al. Early Intervention Guided by the General Movements Examination at Term Corrected Age—Short Term Outcomes. Life 2024, 14 (4), 480.
- Necula, A. I.; Stoiciu, R.; Radulescu Botica, R.; Durdu, C. E.; Bohiltea, R. Neurological outcomes in late preterm infants: an updated review of recent research and clinical insights. Diagnostics 2025, 15 (12), 1514.
- Hintz, S. R.; Barnes, P. D.; Bulas, D.; Slovis, T. L.; Finer, N. N.; Wrage, L. A.; Das, A.; Tyson, J. E.; Stevenson, D. K.; Carlo, W. A.; Walsh, M. C.; Laptook, A. R.; Yoder, B. A.; Van Meurs, K. P.; Faix, R. G.; Rich, W.; Newman, N. S.; Higgins, R. D. Neuroimaging and neurodevelopmental outcome in extremely preterm infants. Pediatrics 2015, 135 (1), e32–e42.
- Ballabh, P.; de Vries, L. S. White matter injury in infants with intraventricular haemorrhage: mechanisms and therapies. Nat. Rev. Neurol. 2021, 17 (4), 199–214.
- Inder, T. E.; Anderson, N. J.; Spencer, C.; Wells, S.; Volpe, J. J. White matter injury in the premature infant: a comparison between serial cranial sonographic and MR findings at term. Am. J. Neuroradiol. 2003, 24 (5), 805–809.
- Li, H.; Liu, M.; Zhang, J.; Liu, S.; Fang, Z.; Pan, M.; Xu, Y.; Ge, X. The effect of preterm birth on thalamic development based on shape and structural covariance analysis. NeuroImage 2024, 297, 120708.
- Kidokoro, H.; Neil, J. J.; Inder, T. E. New MR imaging assessment tool to define brain abnormalities in very preterm infants at term. Am. J. Neuroradiol. 2013, 34 (11), 2208–2214.
- Nongena, P.; Ederies, A.; Azzopardi, D. V.; Edwards, A. D. Confidence in the prediction of neurodevelopmental outcome by cranial ultrasound and MRI in preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 2010, 95 (6), F388–F390.
- Hintz, S. R.; Vohr, B. R.; Bann, C. M.; Taylor, H. G.; Das, A.; Gustafson, K. E.; et al. Preterm neuroimaging and school-age cognitive outcomes. Pediatrics 2018, 142 (1), e20174058.
- Yum, S. K.; Im, S. A.; Seo, Y. M.; Sung, I. K. Enlarged subarachnoid space on cranial ultrasound in preterm infants: Neurodevelopmental implication. Sci. Rep. 2019, 9 (1), 19072.
- Li, Y. T.; Chen, L. W.; Koh, C. L.; Lin, Y. C.; Huang, C. C. Functional connectivity as a prognostic biomarker for neurodevelopmental outcomes in preterm infants without severe brain injury. Brain Commun. 2025, 7 (6), fcaf476.
- Cizmeci, M. N.; de Vries, L. S.; Ly, L. G.; van Haastert, I. C.; Groenendaal, F.; Kelly, E. N.; Govaert, P.; Leijser, L. M. Periventricular hemorrhagic infarction in very preterm infants: characteristic sonographic findings and association with neurodevelopmental outcome at age 2 years. J. Pediatr. 2020, 217, 79–85.
- De Vries, L. S.; Benders, M. J.; Groenendaal, F. Progress in neonatal neurology with a focus on neuroimaging in the preterm infant. Neuropediatrics 2015, 46 (04), 234–241.
- Whyte, H. E.; Blaser, S. Limitations of routine neuroimaging in predicting outcomes of preterm infants. Neuroradiology 2013, 55 (Suppl 2), 3–11.
- Skiöld, B.; Hallberg, B.; Vollmer, B.; Ådén, U.; Blennow, M.; Horsch, S. A novel scoring system for term-equivalent-age cranial ultrasound in extremely preterm infants. Ultrasound Med. Biol. 2019, 45 (3), 786–794.
- Sarkar, S.; Shankaran, S.; Laptook, A. R.; Sood, B. G.; Do, B.; Stoll, B. J.; Das, A.; Guillet, R.; Higgins, R. D.; Barks, J. Screening cranial imaging at multiple time points improves cystic periventricular leukomalacia detection. Am. J. Perinatol. 2015, 32 (10), 973–979.
- Ibrahim, J.; Mir, I.; Chalak, L. Brain imaging in preterm infants <32 weeks gestation: a clinical review and algorithm for the use of cranial ultrasound and qualitative brain MRI. Pediatr. Res. 2018, 84 (6), 799–806.
- Hintz, S. R.; O’Shea, M. Neuroimaging and neurodevelopmental outcomes in preterm infants. Semin. Perinatol. 2008, 32 (1), 11–19.
- Bayram, E.; Bayram, M. T.; Topcu, Y.; Hız, S.; Kayserili, E. Long term neurodevelopmental outcome of preterm infants with periventricular-intraventricular hemorrhage. J. Clin. Exp. Invest. 2012, 3 (3), 331–336.
- Dorner, R. A.; Burton, V. J.; Allen, M. C.; Robinson, S.; Soares, B. P. Preterm neuroimaging and neurodevelopmental outcome: a focus on intraventricular hemorrhage, post-hemorrhagic hydrocephalus, and associated brain injury. J. Perinatol. 2018, 38 (11), 1431–1443.
- Muehlbacher, T.; Dudink, J.; Steggerda, S. J. Cerebellar Development and the Burden of Prematurity. Cerebellum 2025, 24 (2), 39.
- Drayne, J. P.; Mella, A. E.; McLean, M. M.; Ufkes, S.; Chau, V.; Guo, T.; Synnes, A.; Miller, S. P.; Grunau, R. E.; Weber, A. M. Long-range temporal correlation development in resting-state fMRI signal in preterm infants: scanned shortly after birth and at term-equivalent age. PLOS Complex Syst. 2024, 1 (4), e0000024.
- Guha, A.; Hunter, S. K.; Legget, K. T.; McHugo, M.; Tregellas, J. R. Greater Increase in Hippocampal Activity During the Early Postnatal Period After Preterm Birth Is Associated With Better Cognitive and Motor Outcomes at 18 Months. Dev. Neurobiol. 2026, 86 (1), e23018.
- Gande, N.; Bloching, M.; Hochmayr, C.; Staudt, A.; Peglow, U. P.; Kiechl-Kohlendorfer, U.; Griesmaier, E. Developmental Trajectories of Very Preterm Infants and Implications for Routine Neurodevelopmental Follow-Up. Acta Paediatr. 2026, 115 (1), 217–226.
- Serrano-Gómez, M. E.; Massó-Ortigosa, N.; Castellanos-Garrido, A. L.; Acuña De La Rosa, E.; García-Barriga, V. M.; López-Dóriga, A.; Carballo-Santiesteban, G.; Guerra-Balic, M. General movements in infants with neurological risk: Associations with motor development and referral patterns for brain magnetic resonance imaging. Children 2025, 12 (5), 590.
- Doyle, L. W.; Spittle, A.; Anderson, P. J.; Cheong, J. L. Y. School-aged neurodevelopmental outcomes for children born extremely preterm. Arch. Dis. Child. 2021, 106 (9), 834–838.
- Hintz, S. R.; Newman, J. E.; Vohr, B. R. Changing definitions of long-term follow-up: should “long term” be even longer? Semin. Perinatol. 2016, 40 (6), 398–409.
- Duan, Y.; Sun, F. Q.; Li, Y. Q.; Que, S. S.; Yang, S. Y.; Xu, W. J.; et al. Prognosis of psychomotor and mental development in premature infants by early cranial ultrasound. Ital. J. Pediatr. 2015, 41 (1), 30.
- Hagmann, C. F.; Robertson, N. J.; Acolet, D.; Nyombi, N.; Ondo, S.; Nakakeeto, M.; Cowan, F. M. Cerebral measurements made using cranial ultrasound in term Ugandan newborns. Early Hum. Dev. 2011, 87 (5), 341–347.
- Ortega-Leon, A.; Urda, D.; Turias, I. J.; Lubián-López, S. P.; Benavente-Fernández, I. Machine learning techniques for predicting neurodevelopmental impairments in premature infants: a systematic review. Front. Artif. Intell. 2025, 8, 1481338.
- Travis, K. E.; Lazarus, M. F.; Scala, M.; Marchman, V. A.; Bruckert, L.; Velasco Poblaciones, R.; Arnam, J. S.; Ben-Shachar, M.; Yeom, K. W.; Marchuk, J. C.; Feldman, H. M. Skin-to-skin holding in relation to white matter microstructure in infants born preterm. Neurology 2025, 105 (8), e214138.
- Tudor, S.; Bhatia, R.; Liem, M.; Wani, T. A.; Boyd, J.; Raza Khan, U. Opportunities and challenges of using artificial intelligence in predicting clinical outcomes and length of stay in neonatal intensive care units: Systematic review. J. Med. Internet Res. 2025, 27, e63175.
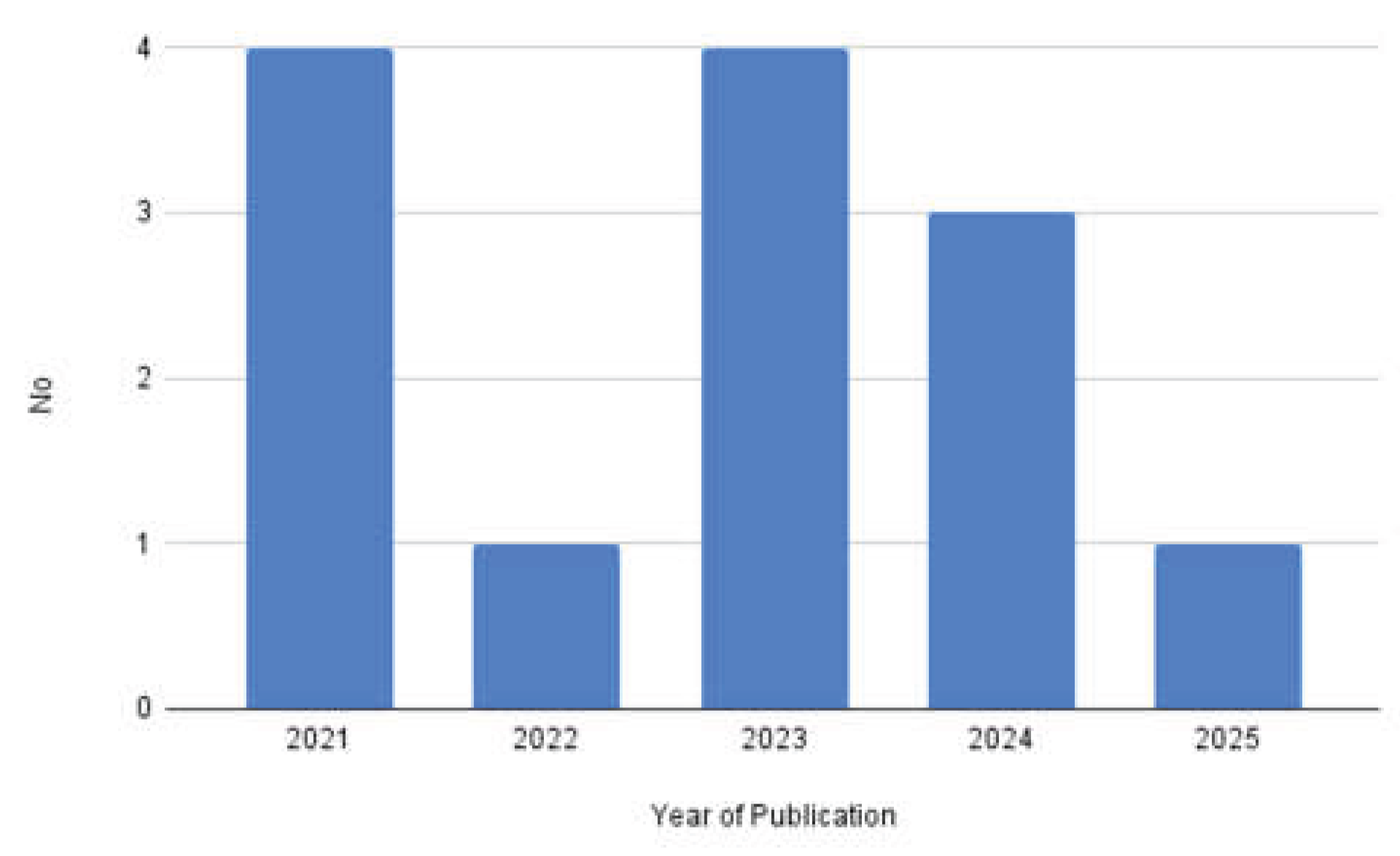
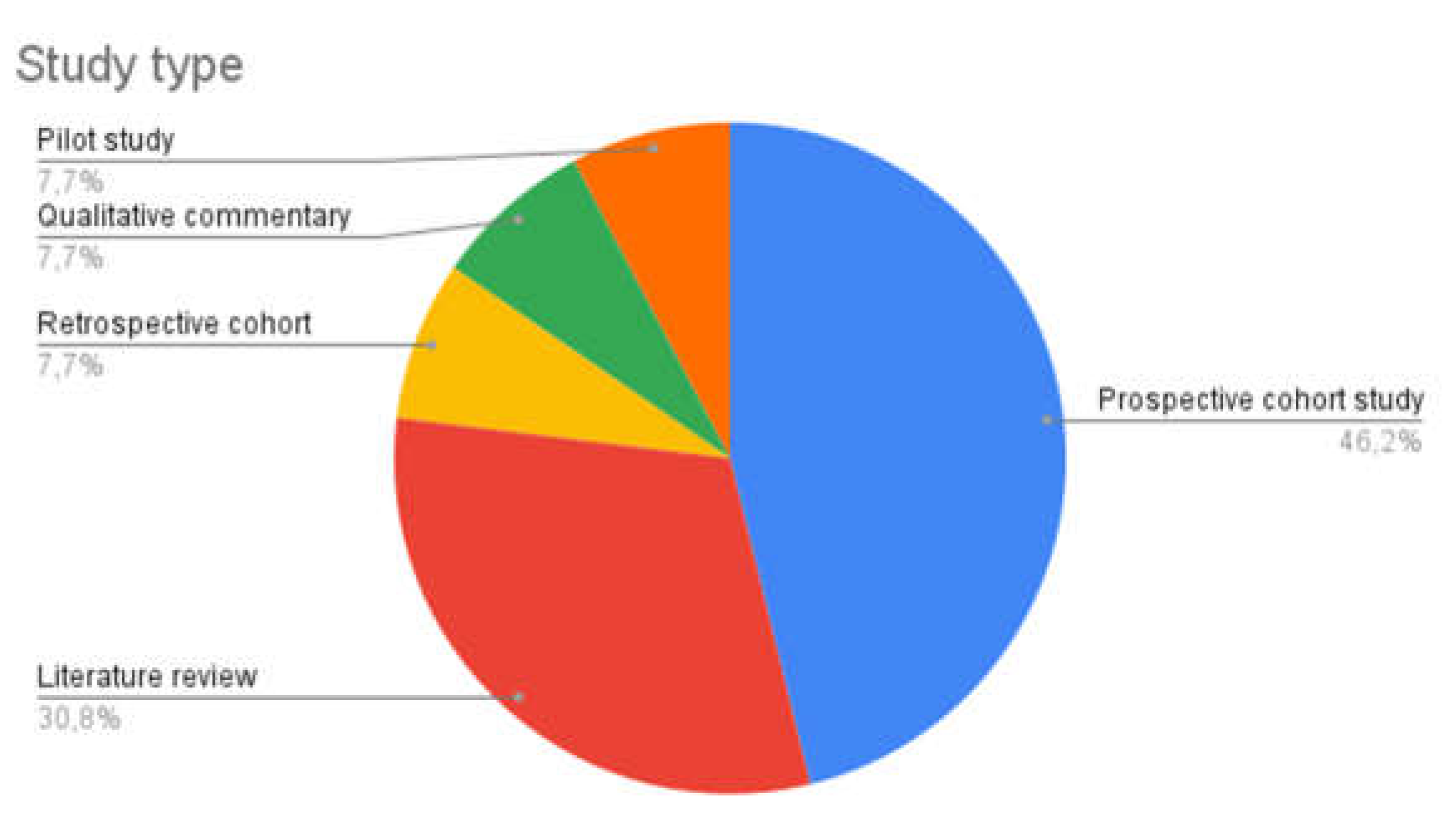
| Reference | Title | Year | Study Type | Country | Population | Key findings |
|---|---|---|---|---|---|---|
| Kwong, A.K., et al. [7] | Early neurodevelopmental screening: Parent perspectives from the neonatal intensive care unit | 2021 | qualitative commentary | USA | 19 parents in Level III and Level IV NICU | to ensure follow-up compliance, parents require structured emotional and professional support; a lack of information prevents parents from understanding the necessity of long-term neurological re-examinations |
| Guillot, M., et al. [8] | Comparative performance of head ultrasound and MRI in detecting preterm brain injury and predicting outcomes: a systematic review | 2021 | literature review | Canada | preterm neonates | a shift from overt cystic WM injury to diffuse, non-cystic patterns has been captured by MRI; while HUS and MRI reliably predicts motor deficits (CP), predicting cognitive outcomes remains a significant challenge; TEA imaging yields high Negative Predictive Value, making it highly effective at ruling out severe impairment |
| Inder, T.E., et al. [9] | Neuroimaging of the Preterm Brain: Review and Recommendations | 2021 | literature review | USA | preterm neonates | accurate prognosis depends on matching the neuropathology to the modality: HUS is effective for apparent hemorrhagic lesions, but MRI is superior for detecting cerebellar hemorrhages and diffuse WM injury; MRI growth metrics provide objective measures of brain development that help characterize the impact of preterm birth on future cognitive and behavioral outcomes |
| Zhang, X.H., et al. [10] | Predicting the developmental outcomes of very premature infants via ultrasound classification A CONSORT- clinical study | 2021 | prospective cohort study | China | 129 very preterm infants ( <28weeks GA) | birth weight is the most significant protective factor against poor outcomes; while early scans do not correlate with long-term development, serial HUS classification ending at TEA demonstrates strong associations with mental and psychomotor development indices |
| Helderman, J., et al. [11] | Association of abnormal findings on neonatal cranial ultrasound with neurobehavior at neonatal intensive care unit discharge in infants born before 30 weeks’ gestation | 2022 | prospective cohort study | USA | 704 infants (<30 weeks GA) | WM injury detected within the first 2 weeks of life is significantly associated with poor attention and movement quality at discharge; early HUS serves as safe, cost-effective bedside triage tool to identify infants who require immediate therapeutic intervention to regulate motoric agitation and muscle tone. |
| Kumar, N., et al. [12] | Role of neuroimaging in preterm infants to predict neurological outcomes | 2023 | prospective cohort study | India | 56 preterm infants (<34 weeks) | intracranial complications are significantly associated with lower GA and birth weight; on the contrary, a higher-than- expected incidence of abnormalities (44.4%) was found in infants >2kg |
| McLean, G., et al. [13] | Evaluation of a Cranial Ultrasound Scoring System for Prediction of Abnormal Early Neurodevelopment in Preterm Infants | 2023 | retrospective cohort study | Australia | 242 preterm infants (median GA 26.5 weeks) | a formalized HUS system including subtle markers (corpus callosum thinning, delayed folding) is more sensitive (57%) for predicting CP than standard “severe abnormality” reporting (27%); late screening (6 weeks postnatal or TEA) is essential to detect WM injury and brain atrophy that are not visible on initial early-life scans |
| Toma, A.I., et al. [14] | Correlations between Head Ultrasounds Performed at Term-Equivalent Age in Premature Neonates and General Movements Neurologic Examination Patterns | 2023 | prospective cohort study | Romania | 44 preterm neonates (mean GA, 33.59 weeks (+2.43 weeks)) | atypical GM patterns, such as “Poor Repertoire” and “Cramped-Synchronized”, are strongly associated with TEA-HUS markers of white matter volume loss and dysmaturation (reduced basal ganglia width and immature gyration); these structural findings together are significant predictors of long-term motor deficits |
| Chevallier, M., et al. [15] | Decision-making for extremely preterm infants with severe hemorrhages on head ultrasound: Science, values, and communication skills | 2023 | literature review | Canada | extremely preterm infants | bilateral involvement (laterality) and post-hemorrhagic ventricular dilatation are far more reliable predictors of CP than the specific anatomical location of the hemorrhage; the need for surgical shunting significantly worsens the motor prognosis, whereas traditional grading systems have limited utility compared to assessing the total extent of the injury |
| Mayrink, M.L.D.S., et al. [16] | The trajectory of head circumference and neurodevelopment in very preterm newborns during the first two years of life: a cohort study | 2024 | prospective cohort study | Brazil | 95 newborns (<32 weeks or 1500g) | HC is a practical, high-value clinical marker for neurodevelopment; higher HC growth at 5 months corrected age correlates positively with cognitive, motor, and language at 18 months; the window between discharge and 1 month corrected age is a critical period for catch-up growth and prediction of functional status |
| Toma, A.I., et al. [17] | Cerebral Ultrasound at Term-Equivalent Age: Correlations with Neuro-Motor Outcomes at 12–24 Months Corrected Age | 2024 | prospective cohort study | Romania | 34 premature infants (30-34 weeks GA) followed to 24 months | abnormal motor acquisitions at 24 months correlate with structural markers at TEA, specifically increased ventricular midbody size, decreased basal ganglia width, decreased cortical depth, and immature gyration; the association is strong enough to support the potential development of a simplified clinical scoring system for motor deficit prediction via HUS |
| Toma, A.I., et al. [18] | Early Intervention Guided by the General Movements Examination at Term Corrected Age—Short Term Outcomes | 2024 | pilot study | Romania | 44 premature neonates (mean GA 33.59 weeks (+2.43 weeks)) examined at term-equivalent age | lesion severity determines the success of neuroplasticity; while many infants benefit from early intervention, severe lesions like Grade III cystic PVL may lead to permanent motor pathways destruction that cannot be compensated for; this highlights that early identification and therapy may have variable efficacy depending on the specific substrate of the injury |
| Necula, A.I., et al. [19] | Neurological Outcomes in Late Preterm Infants: An Updated Review of Recent Research and Clinical Insights | 2025 | literature review | Romania | late preterm infants | structural alterations in the posterior region of the corpus callosum are linked to motor integration deficits and cognitive delays that persist into school age |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




