1. Introduction
1.1. Global Biosecurity Context, Military Relevance, and Threat Landscape
Viral hemorrhagic fevers (VHFs) are a group of severe, often fatal zoonotic diseases caused by enveloped RNA viruses that belong predominantly to the families
Filoviridae,
Arenaviridae,
Nairoviridae,
Phenuiviridae,
Flaviviridae, and
Hantaviridae. Despite substantial virological diversity, these pathogens share convergent clinical and operational characteristics that place them among the highest-priority biological threats to global health security and military medical systems. These include the capacity for rapid human-to-human transmission, high case fatality rates, the absence or limited availability of licensed therapeutics or vaccines for many agents, and stringent biosafety requirements for laboratory handling and diagnosis [
1,
2,
3].
From a military medicine perspective, VHFs occupy a unique position at the intersection of infectious disease epidemiology, biodefense, operational readiness, and international security. Armed forces are disproportionately exposed to VHF risks due to deployment in endemic or outbreak-prone regions, to humanitarian and peacekeeping missions, and to disaster response operations, where healthcare infrastructure may be degraded or overwhelmed. Moreover, the deliberate misuse of VHF agents as biological weapons, while historically limited, remains a persistent concern within national and multinational defense planning frameworks [
4,
5].
The strategic relevance of VHFs is underscored by their consistent inclusion in high-consequence pathogen lists maintained by the World Health Organization (WHO), the U.S. Centers for Disease Control and Prevention (CDC), and military alliances such as NATO. Filoviruses, including Ebola virus and Marburg virus, and arenaviruses such as Lassa virus, are classified as Risk Group 4 agents due to their extreme pathogenicity and lack of widely available countermeasures [
2,
6]. Several arboviral VHFs, including dengue virus, yellow fever virus, and Crimean–Congo hemorrhagic fever virus, pose additional challenges due to vector-borne transmission, geographic expansion driven by climate change, and the potential for simultaneous civilian and military exposure during outbreaks [
7,
8,
9].
Historically, VHF outbreaks have destabilized healthcare systems, disrupted military operations, and generated significant political and societal consequences. The 2014–2016 West African Ebola virus disease epidemic exemplified these dynamics, resulting in more than 28,000 cases, over 11,000 deaths, and the deployment of military medical assets from multiple nations to support outbreak containment, diagnostics, and logistics [
10,
11]. Similar operational lessons have emerged from outbreaks of Lassa fever in West Africa, Crimean–Congo hemorrhagic fever in Eastern Europe and Central Asia, and yellow fever in sub-Saharan Africa and South America [
12,
13,
14].
The biological characteristics of VHF agents complicate both clinical recognition and laboratory diagnosis. Early symptoms are typically nonspecific and include fever, malaise, headache, and myalgia, and can closely resemble malaria, bacterial sepsis, or other endemic febrile illnesses. Hemorrhagic manifestations, when present, often occur late in the disease course and are neither universal nor pathognomonic [
15]. Consequently, delayed clinical suspicion remains a major contributor to nosocomial transmission, laboratory exposure incidents, and delayed outbreak detection [
16].
In military and austere settings, diagnostic challenges are amplified by limited laboratory infrastructure, constraints on specimen transport, and the need to protect medical personnel and maintain operational continuity. The requirement for rapid, accurate, and biosafe diagnostic workflows is therefore not merely a clinical concern but a strategic imperative. Delays or failures in diagnosis can lead to inappropriate patient management, uncontrolled transmission within military units, and mission degradation [
17,
18].
Biosecurity considerations extend beyond individual patient diagnosis to encompass the entire diagnostic chain, from exposure assessment and specimen collection to laboratory testing, data reporting, and decision-making. VHFs demand a risk-based approach to biosafety that integrates technical containment measures, procedural controls, personnel training, and command-level oversight. International standards, such as the WHO Laboratory Biosafety Manual and the CDC/NIH Biosafety in Microbiological and Biomedical Laboratories (BMBL), provide the foundation for these practices, but their implementation must be adapted to military operational realities [
2,
6].
The dual-use nature of diagnostic technologies further complicates VHF biosecurity [
19]. Molecular platforms capable of detecting high-consequence pathogens can support outbreak response and surveillance, but also raise concerns about misuse, data security, and pathogen information hazards. Military medical systems must therefore balance transparency and international collaboration with the need to safeguard sensitive capabilities and information [
20].
Advances in diagnostic technologies over the past two decades have significantly transformed the detection and management of VHFs. Real-time reverse transcription polymerase chain reaction (RT-PCR) has become the cornerstone of acute-phase diagnosis, offering high sensitivity and specificity when performed on appropriately collected specimens [
21]. Next-generation sequencing has enhanced outbreak investigation, pathogen characterization, and attribution, while rapid diagnostic tests and portable molecular platforms have expanded diagnostic capacity in field settings [
22,
23,
24].
Despite these advances, diagnostic inequities persist, particularly in regions where VHFs are endemic. Limited access to high-containment laboratories, shortages of trained personnel, and fragile supply chains continue to impede timely diagnosis and outbreak control. For military forces operating in such environments, reliance on host-nation diagnostic capacity may be insufficient, necessitating deployable laboratory assets and multinational cooperation [
25].
Military doctrine increasingly recognizes infectious disease threats as integral components of the operational environment. Modern concepts of force health protection emphasize early detection, situational awareness, and rapid response to biological hazards, including naturally occurring outbreaks and deliberate biological incidents. VHFs, due to their severity and complexity, serve as critical test cases for integrating medical intelligence, diagnostics, and command decision-making [
5,
26].
This review aims to provide a comprehensive, strategically oriented analysis of the biosecurity and diagnostic dimensions of viral hemorrhagic fevers, with a particular focus on military medical relevance. It synthesizes current international guidance, peer-reviewed evidence, and operational experience to outline best practices for diagnosis, laboratory biosafety, and deployment of diagnostic capabilities. By integrating virological, clinical, and biosecurity perspectives, the review seeks to support informed decision-making by military medical planners, laboratory leaders, and policymakers.
The subsequent sections will examine virus-specific diagnostic strategies, comparative performance of diagnostic platforms, biosafety level requirements and laboratory containment, and the integration of centralized and deployable diagnostic systems. An overarching diagnostic and biosecurity workflow is presented to contextualize these elements within a coherent operational framework applicable to both civilian outbreak response and military operations.
1.2. Virus-Specific Diagnostic Strategies and Operational Platforms
Accurate diagnosis of viral hemorrhagic fevers depends on a precise understanding of the causative virus, the stage of infection at which clinical suspicion arises, and the biosafety constraints under which diagnostic testing is performed. Unlike many common infectious diseases, VHFs demand a diagnostic approach that integrates molecular sensitivity with stringent containment requirements, particularly in military and field environments where laboratory infrastructure may be limited. Virus-specific diagnostic strategies, therefore, represent a cornerstone of both clinical management and biosecurity risk mitigation.
1.2.1. Filoviridae: Ebola Virus and Marburg Virus
Filoviruses remain the archetypal VHF pathogens due to their high lethality, epidemic potential, and historical role in shaping global outbreak response doctrine. Diagnostic confirmation of Ebola virus disease (EVD) and Marburg virus disease relies primarily on real-time reverse transcription polymerase chain reaction (RT-PCR) targeting conserved genomic regions such as the nucleoprotein (NP), glycoprotein (GP), or RNA-dependent RNA polymerase (L) genes. RT-PCR assays demonstrate high analytical sensitivity during the acute viremic phase, often detectable within the first three days of symptom onset [
28,
29,
30].
Antigen detection assays, including lateral flow immunoassays targeting viral nucleoproteins, have been deployed in outbreak settings to support rapid triage and isolation decisions. While these assays offer operational advantages in speed and minimal equipment requirements, their reduced sensitivity compared with RT-PCR necessitates confirmatory molecular testing, particularly for low-viral-load samples or early infection [
31].
Serological diagnostics play a limited role in acute filovirus infection due to delayed antibody responses and high mortality. IgM and IgG enzyme-linked immunosorbent assays (ELISA) are primarily used for retrospective diagnosis, survivor follow-up, and seroepidemiological studies [
32]. Virus isolation and culture are restricted to biosafety level 4 (BSL-4) laboratories and are not routinely performed for diagnostic purposes because of the extreme risk posed by handling live virus [
2].
From a military medical standpoint, filovirus diagnostics underscore the need for deployable molecular platforms that can operate under high-containment field conditions. Mobile laboratories equipped with negative-pressure gloveboxes and portable RT-PCR systems have proven effective in reducing diagnostic turnaround times during outbreaks and supporting force health protection in deployed settings [
33].
1.2.2. Arenaviridae: Lassa Virus and South American Arenaviruses
Arenaviruses, particularly Lassa virus, pose a persistent threat in West Africa and cause significant morbidity among civilian populations and deployed personnel. Diagnostic confirmation relies on RT-PCR detection of viral RNA in blood, serum, or plasma during the acute phase of illness. Lassa virus exhibits genetic diversity across lineages, necessitating careful assay design to ensure broad coverage and minimize false-negative results [
34].
Antigen detection assays for Lassa virus have been developed but are not widely available or standardized. Serological assays, including IgM and IgG ELISA, are useful for diagnosing infection in later stages and for surveillance. However, cross-reactivity with other arenaviruses and delayed seroconversion limit their utility as standalone diagnostic tools [
35].
South American arenaviruses, such as Junín, Machupo, and Guanarito viruses, are geographically restricted but pose a severe disease risk with high case-fatality rates. Diagnostic principles parallel those for Lassa virus, with molecular detection as the primary modality and serology as a complementary approach. As with filoviruses, virus isolation is confined to BSL-4 laboratories and is rarely indicated outside research or reference settings [
36].
1.2.3. Nairoviridae: Crimean–Congo Hemorrhagic Fever Virus
Crimean–Congo hemorrhagic fever virus (CCHFV) presents unique diagnostic challenges due to its wide geographic distribution, tick-borne transmission, and potential for nosocomial outbreaks. RT-PCR remains the gold standard for acute diagnosis, with assays targeting the small (S) segment of the viral genome commonly employed [
37].
Serological testing, including IgM capture ELISA, becomes useful after the first week of illness and is particularly valuable in settings with limited molecular testing capacity. Unlike filoviruses, CCHFV may be processed under biosafety level (BSL) 3 (BSL-3) conditions for non-propagative diagnostic procedures, although enhanced precautions are recommended due to the risk of laboratory-acquired infections [
38].
For military forces operating in endemic regions, particularly in Eastern Europe, the Middle East, and Central Asia, integrating CCHFV diagnostics into routine febrile illness algorithms is essential. Rapid recognition and laboratory confirmation can prevent secondary transmission among healthcare workers and deployed units.
1.2.4. Flaviviridae: Dengue Virus and Yellow Fever Virus
Flaviviral VHFs, including dengue virus (DENV) and yellow fever virus (YFV), differ from filoviruses and arenaviruses in their transmission dynamics [
39] and diagnostic profiles [
40]. Dengue virus infection is characterized by high viremia early in illness, allowing for RT-PCR detection within the first five days of symptom onset. Non-structural protein 1 (NS1) antigen detection assays are a rapid, widely used diagnostic option in both civilian and military contexts [
41].
Serological assays for dengue, while essential for diagnosis after the acute phase, are complicated by extensive cross-reactivity among flaviviruses [[ ] and coronaviruses [
42], particularly in individuals with prior vaccination or infection. This limitation has significant implications for military personnel deployed to regions where multiple flaviviruses co-circulate [
43].
Yellow fever virus diagnosis similarly relies on RT-PCR during early infection and serology thereafter. Vaccination history must be carefully considered when interpreting serological results, as vaccine-induced antibodies can confound diagnostic interpretation [
44].
1.2.5. Hantaviridae and Phenuiviridae
Hantaviruses and phleboviruses, including Rift Valley fever virus, present additional diagnostic considerations. RT-PCR and serology are both employed, with the choice of modality guided by disease phase and specimen availability. Rift Valley fever virus, due to its zoonotic and epizootic nature, is of particular relevance to military operations involving livestock or vector exposure [
45].
Table 1 summarizes virus-specific diagnostic approaches, specimen types, biosafety requirements, and primary use cases. This comparative framework supports informed diagnostic selection and risk assessment across diverse operational scenarios.
2. Centralized Versus Deployable Diagnostic Platforms
The operational context strongly influences the selection of a diagnostic platform. Centralized reference laboratories offer comprehensive diagnostic capabilities, including confirmatory testing, viral sequencing, and quality assurance. However, they are limited by specimen transport times and dependency on secure logistics [
46].
Deployable diagnostic platforms, including mobile laboratories and point-of-care molecular systems, provide rapid preliminary diagnosis in outbreak or combat settings. While these systems may lack the full analytical breadth of centralized laboratories, they play a critical role in early detection, isolation, and force protection [
47].
Table 2 compares centralized and deployable diagnostic platforms in terms of performance, biosafety, and operational suitability.
3. Biosafety, Containment Doctrine, and Biosecurity Risk Management
The diagnosis of VHF is inseparable from biosafety and biosecurity considerations. Unlike routine clinical microbiology, VHF diagnostics must balance analytical performance with strict containment measures designed to protect laboratory personnel, healthcare workers, and surrounding communities. In military medical systems, these requirements are further shaped by operational constraints, expeditionary environments, and the potential use of VHFs as biological threats.
3.1. Biosafety Levels and Containment Principles
International biosafety frameworks, including those established by the WHO, the United States CDC, and military medical doctrine, classify laboratory activities by BSL based on pathogen transmissibility, disease severity, and the availability of countermeasures. VHFs span multiple biosafety categories, with filoviruses and certain arenaviruses requiring the highest levels of containment.
BSL-4 laboratories are required for work involving the Ebola virus, Marburg virus, Lassa virus, and related agents. These facilities incorporate maximum-containment features, including positive-pressure suits, dedicated air-supply systems, chemical decontamination of effluent, and controlled access. While indispensable for viral culture, pathogenesis research, and countermeasure development, BSL-4 laboratories are not designed for routine diagnostic throughput and are limited in number globally [
48].
In contrast, non-propagative diagnostic procedures, such as nucleic acid extraction from inactivated specimens, may be performed under BSL-3 conditions, provided that validated inactivation protocols are strictly followed. This distinction has enabled the expansion of molecular diagnostic capacity for VHFs beyond a small number of high-containment reference centers, particularly during large-scale outbreaks [
49].
3.2. Biosafety in Molecular Diagnostics
Molecular diagnostics represent the backbone of modern VHF detection, but they introduce specific biosafety challenges related to specimen handling, aerosol generation, and waste management. Pre-analytical steps, including blood collection, centrifugation, and nucleic acid extraction, pose the highest risk of exposure. Accordingly, WHO and CDC guidelines emphasize the use of sealed centrifuge rotors, class II or III biological safety cabinets, and chemical or thermal inactivation prior to downstream processing [
1,
50].
For military laboratories, especially those deployed forward, adherence to these principles requires careful selection of platforms and training of personnel. Compact molecular systems with closed-cartridge designs reduce operator exposure and simplify biosafety compliance, albeit sometimes at the expense of assay flexibility or throughput [
51].
3.3. Occupational Risk and Laboratory-Acquired Infections
A historical analysis of laboratory-acquired infections underscores the dangers of VHF diagnostics when biosafety measures are inadequate. Crimean–Congo hemorrhagic fever virus, in particular, has been associated with multiple laboratory and healthcare worker infections, often linked to needlestick injuries or unrecognized exposure to infectious blood products [
52,
53]
Filoviruses, while less commonly associated with laboratory-acquired infections, present catastrophic consequences in the event of containment failure. As a result, military medical doctrine emphasizes redundancy in engineering controls, procedural safeguards, and medical surveillance of laboratory personnel handling high-risk specimens [
54].
3.4. Biosecurity and Dual-Use Considerations
Beyond biosafety, VHF diagnostics intersect with broader biosecurity concerns, particularly the potential misuse of pathogens or diagnostic knowledge for hostile purposes. VHFs are recognized under international frameworks, including the Biological Weapons Convention, as agents of concern due to their lethality and psychological impact.
Diagnostic laboratories, especially those operating in conflict zones or politically unstable regions, must therefore implement robust biosecurity measures. These include controlled access to specimens and data, chain-of-custody documentation, and coordination with military intelligence and public health authorities when unusual patterns of disease are detected [
55].
3.5. Military-Specific Containment Doctrine
Military medical systems adopt a layered approach to biosafety and biosecurity, integrating civilian public health standards with force protection requirements. This approach recognizes that deployed forces may encounter VHFs in environments lacking established healthcare infrastructure, necessitating self-sufficient diagnostic and containment capabilities.
Mobile high-containment laboratories, often deployed under military or joint civilian–military command, exemplify this doctrine. These units combine modular laboratory design, negative-pressure workspaces, and on-site waste decontamination to enable safe diagnostics in austere settings [
56]. While not equivalent to fixed BSL-4 facilities, such platforms provide a critical bridge between frontline clinical care and centralized reference laboratories.
Table 3 summarizes biosafety levels, containment requirements, and laboratory capabilities for major VHF agents, harmonized with WHO, CDC, and military doctrine.
3.6. Waste Management and Environmental Decontamination
Effective waste management is a critical but often underemphasized component of VHF diagnostics. Liquid waste, sharps, and disposable PPE must be decontaminated with validated chemical disinfectants or by autoclaving prior to disposal. Environmental persistence of certain VHF agents necessitates rigorous surface decontamination protocols, particularly in field laboratories where infrastructure may be limited [
57,
58].
Military guidelines further emphasize the need for contingency planning in the event of laboratory compromise, including evacuation procedures, environmental remediation, and, where available, post-exposure prophylaxis.
3.7. Ethical and Legal Dimensions
The handling of VHF specimens raises ethical and legal considerations related to informed consent, data sharing, and international cooperation. During multinational military operations or humanitarian deployments, diagnostic activities must comply with host-nation regulations while adhering to international standards. Transparent communication between military medical units, civilian health authorities, and international organizations is essential to maintain trust and ensure effective outbreak response [
59].
3.8. Integration into Outbreak Response and Force Protection
Ultimately, biosafety and biosecurity measures are not ends in themselves but integral components of a broader outbreak response strategy. Timely diagnosis, safe specimen handling, and secure data management enable rapid clinical decision-making, targeted infection control, and effective communication with command structures.
For military forces, integrating diagnostic biosafety into operational planning enhances resilience against both naturally occurring outbreaks and deliberate biological threats. The lessons learned from recent VHF outbreaks highlight the necessity of sustained investment in training, infrastructure, and international collaboration [
60].
4. Diagnostic Timelines, Clinical Algorithms, and Operational Workflows
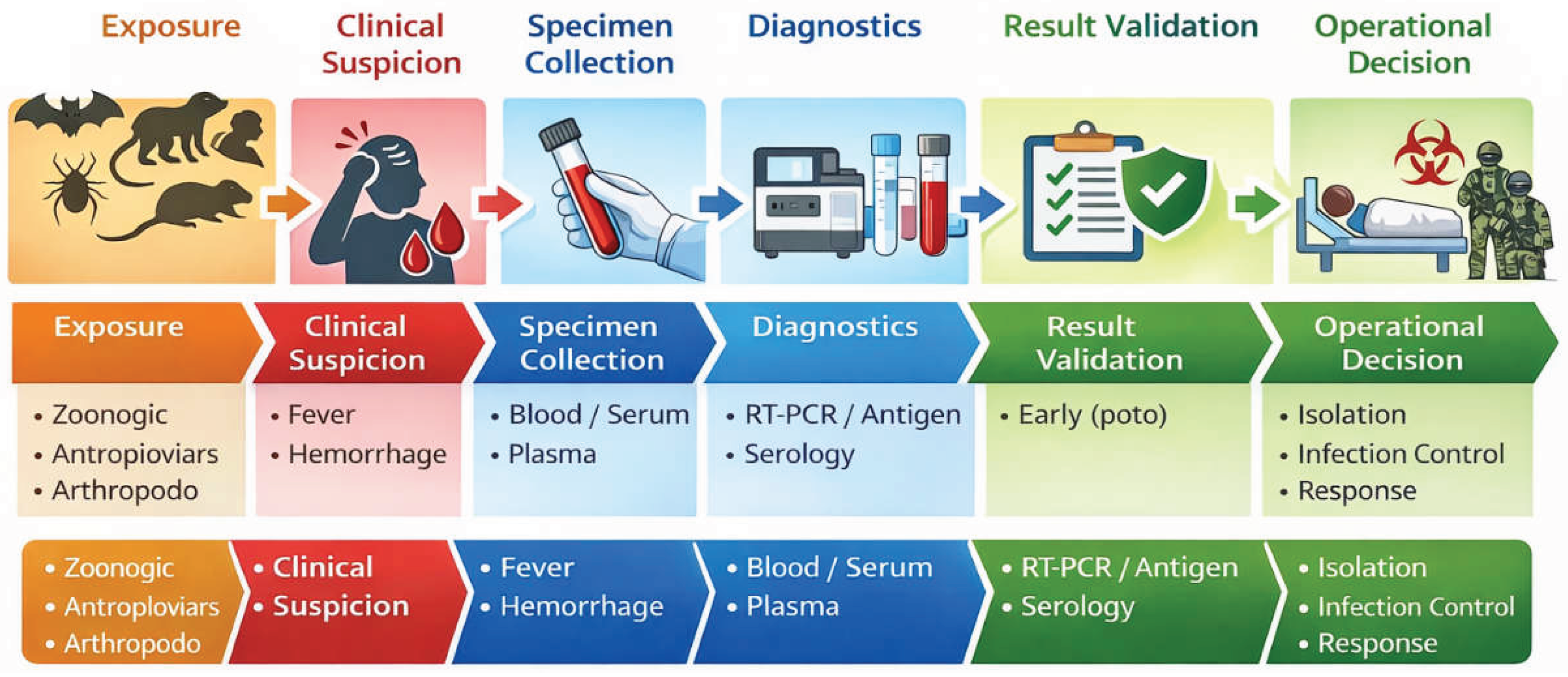
Timely diagnosis of VHFs is a decisive factor in patient outcomes, outbreak containment, and force protection. Unlike many infectious diseases, VHFs present a narrow window in which early clinical suspicion must be translated into laboratory confirmation under heightened biosafety conditions. Diagnostic delays not only increase mortality risk but also amplify the potential for secondary transmission among healthcare workers and military personnel. Consequently, modern VHF diagnostic doctrine emphasizes time-structured workflows that integrate exposure history, clinical evolution, specimen selection, and laboratory capacity.
4.1. Temporal Dynamics of VHF Infection
The incubation periods of VHFs vary by virus family but typically range from 2 to 21 days. During this phase, individuals are asymptomatic, and diagnostic testing is generally uninformative. Once symptoms emerge, viral kinetics diverge significantly across pathogens, shaping the optimal diagnostic approach.
Filoviruses and arenaviruses are characterized by rapidly increasing viremia shortly after symptom onset, rendering molecular diagnostics highly sensitive during the first week of illness. In contrast, flaviviral VHFs such as dengue may exhibit an earlier but shorter window of detectable viremia, followed by a rapid transition to antibody-mediated diagnosis. These temporal differences necessitate pathogen-specific diagnostic algorithms that are responsive to both disease stage and operational context [
61].
4.2. Clinical Suspicion and Case Definition
Initial diagnostic pathways begin with clinical suspicion based on non-specific symptoms—fever, malaise, headache, myalgia—that overlap with numerous endemic diseases. In military deployments, differential diagnosis must also consider malaria, typhoid fever, leptospirosis, and other febrile illnesses common in austere environments.
Standardized case definitions, such as those issued by WHO and CDC, provide structured criteria for suspect, probable, and confirmed VHF cases. While essential for surveillance and reporting, these definitions are intentionally conservative and may delay laboratory testing if applied rigidly. Military medical doctrine, therefore, advocates a lower threshold for initiating diagnostic testing when exposure risk is credible, particularly following contact with wildlife, ticks, livestock, or known outbreak zones [
1,
62].
4.3. Specimen Selection and Handling over Time
Specimen type and timing are critical determinants of diagnostic yield. Whole blood, plasma, or serum collected during the acute febrile phase provides the highest sensitivity for RT-PCR–based assays. Improper timing or specimen choice can lead to false negatives, particularly early in the disease, when viral loads may be near the detection threshold.
As infection progresses, serological assays gain diagnostic value, with IgM antibodies typically detectable from day 5 to 7 onward. However, reliance on serology alone is discouraged in high-consequence settings due to delayed seroconversion and cross-reactivity. Sequential testing strategies combining molecular and serological assays are therefore recommended to maximize diagnostic confidence [
63].
4.4. Diagnostic Turnaround Time and Decision-Making
In outbreak and military settings, diagnostic turnaround time directly influences isolation decisions, resource allocation, and command-level risk assessments. Centralized laboratories offer high analytical performance but may require days for specimen transport and result reporting. Deployable diagnostic platforms reduce this delay, enabling same-day or next-day results that support rapid clinical and operational decisions.
However, rapid diagnostics introduce trade-offs between speed, sensitivity, and biosafety. Field-deployed molecular platforms must be rigorously validated and integrated into broader laboratory networks to ensure confirmatory testing and epidemiological oversight [
64].
Figure 1 illustrates the integrated diagnostic and biosecurity workflow from exposure to result, emphasizing critical control points where clinical, laboratory, and biosafety decisions intersect.
4.5. Clinical Algorithms for Military Settings
Military diagnostic algorithms prioritize simplicity, robustness, and adaptability. Unlike civilian tertiary care centers, deployed medical units must operate with limited personnel, equipment, and time. Algorithms, therefore, emphasize early molecular testing for high-risk cases, coupled with conservative infection-control measures until VHF is excluded.
Decision trees commonly integrate exposure risk, symptom severity, and diagnostic availability to guide escalation from field testing to centralized confirmation. Importantly, negative initial results do not automatically exclude VHF, particularly if testing occurs early in the disease. Repeat testing at 24–48 h intervals is recommended when clinical suspicion remains high [
65].
4.6. Communication, Reporting, and Learned from Outbreak Responses
Effective communication of diagnostic results is essential for outbreak control and military command decision-making. Laboratories must establish secure, redundant reporting channels to ensure the timely dissemination of results to clinicians, infection control teams, and operational leadership. Delays or ambiguity in result communication can undermine trust and impede coordinated response efforts.
Recent VHF outbreaks have highlighted recurring diagnostic challenges, including delayed testing, insufficient biosafety training, and fragmented laboratory networks. Conversely, successful responses demonstrate the value of pre-established diagnostic workflows, mobile laboratory integration, and continuous training of medical personnel [
66].
For military medical systems, these lessons reinforce the need for preparedness throughout the diagnostic timeline from exposure assessment to definitive confirmation. Diagnostic excellence in VHFs is not merely a technical achievement but a strategic capability that underpins biosecurity and force protection.
5. Strategic Preparedness, Future Diagnostic Directions, and Military Biosecurity Integration
The sustained threat posed by VHFs necessitates a forward-looking diagnostic and biosecurity strategy that transcends reactive outbreak response. For military medical systems, preparedness for VHFs represents both a public health imperative and a core element of biological defense. Advances in diagnostics, coupled with evolving operational environments and emerging technologies, require continuous adaptation of doctrine, infrastructure, and training.
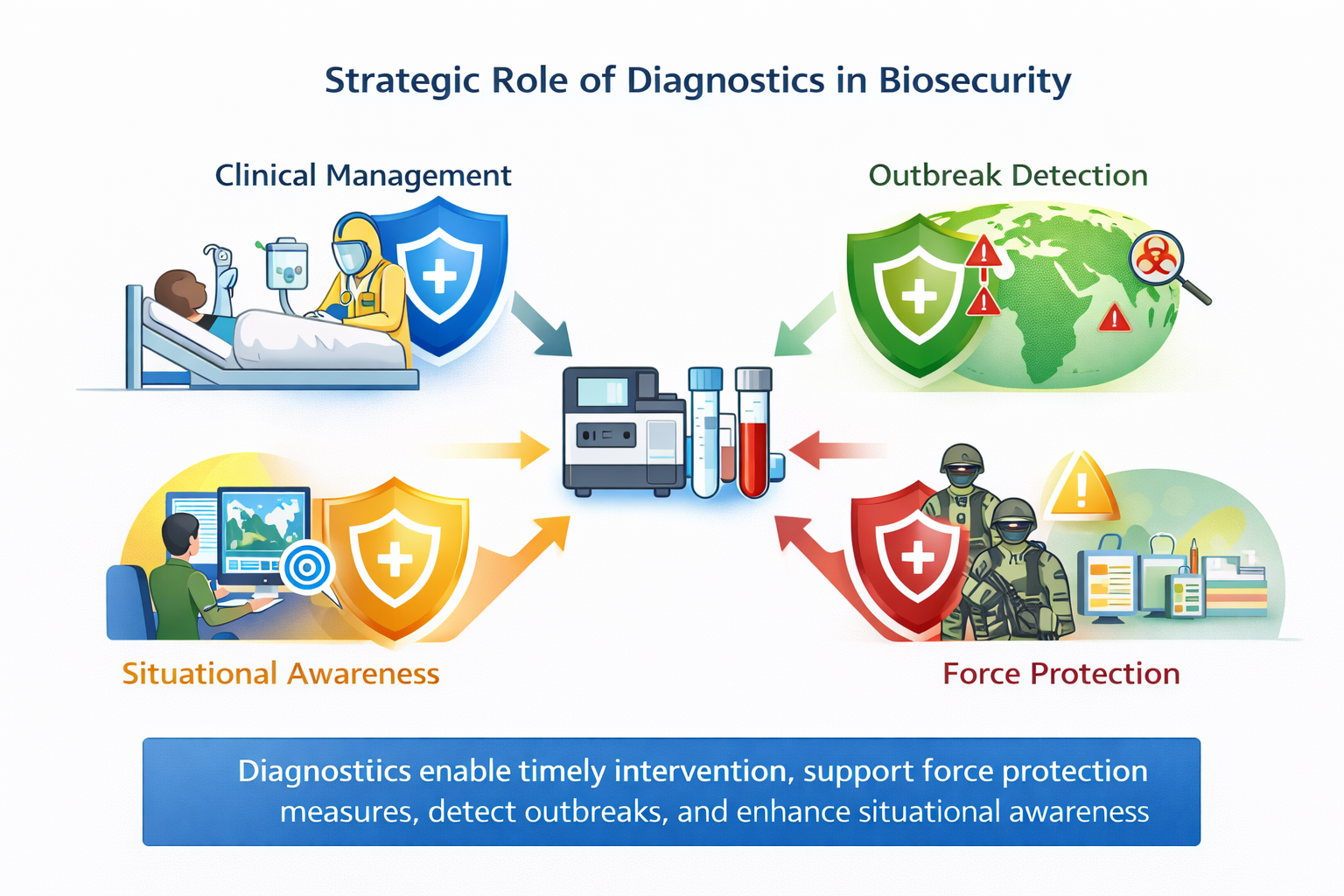
5.1. Strategic Role of Diagnostics in Biosecurity
Diagnostics occupy a central position in the biosecurity architecture surrounding VHFs. Early detection enables timely clinical intervention, limits secondary transmission, and supports strategic decision-making at both tactical and operational levels. In military contexts, diagnostic confirmation of a VHF case may trigger force protection measures, mission reconfiguration, or international coordination with civilian health authorities.
Importantly, diagnostics also serve as surveillance tools that contribute to situational awareness beyond individual patient management. Syndromic surveillance systems, when integrated with laboratory confirmation, provide early warning of outbreaks that may threaten deployed forces or destabilize regions of strategic interest [
67].
5.2. Preparedness Frameworks and Doctrine Alignment
Preparedness for VHF diagnostics is guided by a convergence of civilian public health frameworks and military medical doctrine. WHO and CDC guidelines establish foundational principles for laboratory readiness, while military-specific doctrines emphasize scalability, redundancy, and interoperability.
Key components of preparedness include pre-positioning of diagnostic reagents, maintaining deployable laboratory units, and establishing referral pathways to regional or international reference laboratories. Exercises and simulations play a critical role in validating these systems and in exposing gaps in logistics, communication, and biosafety compliance before real-world crises occur [
68].
5.3. Training and Workforce Development
Human expertise remains a limiting factor in VHF diagnostics, particularly in high-containment laboratory operations. Military medical personnel must be proficient not only in technical diagnostic procedures but also in biosafety practices, risk assessment, and emergency response.
Training programs increasingly emphasize cross-disciplinary competencies, integrating laboratory science with clinical medicine, epidemiology, and biosecurity. Simulation-based training, including mock specimen handling and containment breach scenarios, enhances preparedness and reduces the risk of laboratory-acquired infections [
69].
5.4. Emerging Diagnostic Technologies
Technological innovation is reshaping the diagnostic landscape for VHFs. Next-generation sequencing (NGS) platforms enable comprehensive pathogen identification and genomic surveillance, supporting outbreak tracing and detection of novel or engineered variants. While currently confined largely to centralized laboratories, miniaturized sequencing platforms are increasingly being evaluated for field deployment [
70,
71].
Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)-based diagnostics represent another promising avenue, offering rapid, highly specific detection with minimal equipment requirements. Early studies demonstrate potential applicability to filoviruses and other high-consequence pathogens, although validation under field conditions remains ongoing [
72].
Digital health technologies, including artificial intelligence [
73,
74,
75] driven decision support systems, further enhance diagnostic workflows by integrating clinical data, exposure risk, and laboratory results into coherent risk assessments. Such systems may prove particularly valuable in resource-limited or high-tempo military operations.
5.5. Integration with Global Health Security
Military medical capabilities play a unique role in global health security [
76], often bridging gaps in diagnostic capacity during international outbreaks. Deployable laboratories and trained personnel have supported civilian health systems during Ebola outbreaks in West Africa and elsewhere, illustrating the dual-use nature of military diagnostic assets.
This integration requires careful coordination to ensure alignment with international norms, respect for sovereignty, and ethical use of military resources in public health emergencies. Transparent collaboration enhances trust and strengthens collective resilience against VHF threats [
77].
5.6. Biosecurity and Counterproliferation Considerations
The diagnostic domain intersects with counterproliferation efforts aimed at preventing the misuse of biological agents. Surveillance data derived from diagnostic testing may provide early indicators of deliberate release or laboratory accidents. Military medical intelligence units, therefore, increasingly collaborate with diagnostic laboratories to contextualize unusual epidemiological patterns [
78].
At the same time, dissemination of diagnostic methodologies must be balanced against the risk of dual-use knowledge. Responsible publication practices and controlled access to sensitive protocols form part of a broader biosecurity strategy that safeguards both scientific progress and national security.
5.7. Ethical and Challenges
Preparedness for VHF diagnostics extends beyond technical considerations to encompass ethical and legal dimensions. Informed consent, data privacy, and equitable access to diagnostics are critical issues, particularly during multinational military operations or humanitarian interventions.
Military medical systems must navigate complex legal frameworks governing specimen transport, data sharing, and international cooperation. Adherence to ethical standards not only protects individual rights but also reinforces legitimacy and trust in military-led health interventions [
79].
The evolving threat landscape for VHFs includes climate-driven expansion of vector ranges, increased human–animal interface, and the potential emergence of novel pathogens. These trends underscore the necessity for adaptable diagnostic platforms and flexible biosafety frameworks. Investment in research and development, coupled with sustained international collaboration, will be essential to address these challenges. For military medical systems, maintaining diagnostic readiness for VHFs is not a static goal but an ongoing process that must evolve alongside scientific and geopolitical realities [
80,
81].
5.8. Strategy for Biological Defense 82-90
Viral hemorrhagic fevers and convergent threats such as the Nipah virus must be recognized as high-consequence biological risks that can simultaneously affect public health, military operational capability, and national and international stability [
82,
83]. Their high case-fatality rates, potential for dissemination, early diagnostic uncertainty, and stringent biosafety requirements justify their prioritization in biological defense frameworks [
84], regardless of their specific clinical classification [
85].
In defense contexts, safe and timely diagnostic capability constitutes a critical element of operational readiness. Rapid and reliable laboratory confirmation informs strategic decisions related to force protection, asset deployment, continuity of operations, and crisis management. The absence or fragility of such capability undermines evidence-based decision-making, increases the risk of intra-organizational transmission, and may result in destabilizing sanitary, economic, and geopolitical effects [
86,
87].
The integration of molecular diagnostics, particularly RT-PCR, into deployable medical structures and command-and-control systems should be treated as a strategic priority. A layered diagnostic architecture combining advanced forward-deployed capabilities with centralized reference laboratories ensures operational resilience, redundancy, and strategic oversight. This model applies to both classical VHFs [
88] and high-consequence emerging viruses, such as NiV [
87,
89].
Biosafety and biosecurity constitute inseparable pillars of biological defense. Doctrines and policies should emphasize multilayered containment, protocol standardization, continuous training, and an organizational culture of safety to reduce the risks of occupational exposure, containment failures, and the misuse of biological agents. In addition, diagnostic information must be treated as a strategic asset, requiring robust governance, data protection, and close coordination among defense, health, intelligence, and science and technology sectors.
Finally, the incorporation of emerging technologies such as genomic sequencing, rapid diagnostic platforms, CRISPR-based assays, and artificial intelligence [
73,
74,
75]-enabled decision-support systems enhances early warning and anticipatory response capabilities. Readiness for VHFs and convergent threats such as the NiV directly reflects the level of national biological defense maturity, in which robust diagnostic capabilities, firmly anchored in biosafety and biosecurity, function as decisive force multipliers for force protection, institutional resilience, and global security.
Table 4.
Comparison between Nipah virus and viral hemorrhagic fevers from a biosafety and preparedness perspective.
Table 4.
Comparison between Nipah virus and viral hemorrhagic fevers from a biosafety and preparedness perspective.
| Domain |
Nipah Virus (NiV) |
Viral Hemorrhagic Fevers (VHFs) |
| Virological family |
Paramyxoviridae [90,91] |
Filoviridae, Arenaviridae, Nairoviridae, Flaviviridae [92] |
| Natural reservoirs |
Fruit bats (Pteropus spp.) [93] |
Rodents, bats, ticks (virus-dependent) [3] |
| Human-to-human transmission |
Documented, mainly healthcare/household settings [94] |
Common, especially in healthcare and funerals [93,94] |
| Case fatality rate |
High, often >40% [91,92] |
High, virus-dependent [94,95] |
| Initial clinical features |
Nonspecific; respiratory or neurological signs [91] |
Nonspecific; may progress to hemorrhage [93,96] |
| Diagnostic methods |
RT-PCR; serology (late phase) [92,96] |
RT-PCR; serology; virus isolation [93,97] |
| Biosafety requirements |
BSL-3 (diagnostics); BSL-4 (isolation) [98,99] |
BSL-3 or BSL-4, depending on agent [98,99] |
| Medical countermeasures |
Mostly experimental [91] |
Available for some VHFs (e.g., Ebola) [96] |
| Strategic relevance |
Emerging high-consequence pathogen [92] |
Established high-consequence pathogens [93,95] |
6. Conclusions
Viral hemorrhagic fevers constitute a persistent biological threat to military forces, civilian populations, and international security. Their high mortality rates, epidemic potential, diagnostic ambiguity, and demanding biosafety requirements position VHFs as pathogens of strategic concern within biodefense and military medical planning. This review examines current diagnostic approaches and biosecurity frameworks for VHF agents, emphasizing their relevance to military operational environments.
Within defense contexts, VHF diagnosis is not merely a clinical task but a critical operational capability. Rapid and reliable laboratory confirmation directly influences force health protection, operational tempo, and command decision-making. Diagnostic failure or delay may degrade mission effectiveness, facilitate intra-unit transmission, and contribute to broader regional destabilization.
Molecular diagnostics, particularly RT-PCR, remain the primary tools for VHF detection across virus families. Their operational value depends on integration into deployable medical assets, time-critical decision pathways, and validated biosafety workflows. Forward-deployable diagnostic platforms enhance situational awareness in austere and contested environments, while centralized reference laboratories provide confirmatory capacity and strategic oversight.
Biosafety and biosecurity are inseparable from diagnostic operations involving high-consequence pathogens. Military medical doctrine emphasizes layered containment, redundancy, and sustained training to mitigate the risk of laboratory-associated exposure or loss of containment. Beyond laboratory settings, diagnostic information supports surveillance, threat attribution, and counterproliferation efforts, requiring secure data handling and coordination with defense, intelligence, and public health authorities.
As environmental change, population mobility, and technological diffusion alter the threat landscape, emerging diagnostic technologies—including genomic sequencing, CRISPR-based assays, and AI-enabled decision support offer potential advantages for early warning and response. Ultimately, preparedness for VHFs reflects overall biological defense readiness, in which robust diagnostic capability anchored in biosafety and biosecurity serves as a decisive force multiplier for force protection and global stability.
Author Contributions
Conceptualization and Investigation: S.G.D.-S.; Methodology: A.C.S., M.M.M., and F.R.S.; Funding: S.G.D-S. Writing—original draft: S.G.D.-S.; Review and editing: S.G.D-S and F.R.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by MCTI (PNIPE)-FINEP under grant number #0918/25, the Brazilian Council for Scientific Research (CNPq #30515-2020-5), and the Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro (FAPERJ #200.960-2022).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
For detailed data, please contact the author directly.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| BMBL |
Microbiological and Biomedical Laboratories |
| BSL |
Biosafety Level |
| CCHFV |
Crimean–Congo hemorrhagic fever virus |
| CDC |
Centers for Disease Control and Prevention |
| CRISPR |
Clustered Regularly Interspaced Short Palindromic Repeat |
| DENV |
dengue virus |
| ELISA |
enzyme-linked immunosorbent assays |
| EVD |
Ebola virus disease |
| GP |
glycoprotein |
| NiV |
Nipah Virus |
| NS1 |
Non-structural protein 1 |
| NP |
nucleoprotein |
| RT-PCR |
Real-time reverse transcription polymerase chain reaction |
| VHFs |
Viral Hemorrhagic Fevers |
| YFV |
yellow fever virus |
| WHO |
World Health Organization |
References
- World Health Organization. Managing epidemics: key facts about major deadly diseases. WHO, 2018. Available on https://www.who.int/publications/i/item/managing-epidemics-key-facts-about-major-deadly-diseases (Acessed on 11/11/2025).
- World Health Organization. Laboratory biosafety manual. 4th ed. Geneva: WHO; 2020. Available on: https://www.who.int/ publications/i/item/9789240011311 (Accessed on 112/11/2025).
- Centers for Disease Control and Prevention (CDC). Viral hemorrhagic fevers (VHFs). U.S. Department of Health and Human Services; 2023. Available from: https://www.cdc.gov/viral-hemorrhagic-fevers/about/index.html. (Accessed on 13/12/2025).
- Kortepeter, M.G.; Parker, G.W. Viral hemorrhagic fevers as biological weapons. Biosecurity and Bioterrorism: Biodefense Strategy, Practice, and Science. 2010, 8, 157–168.
- NATO Standardization Office. AMedP-8.1: documentation relative to initial medical treatment and evacuation. NATO; 2023. Available https://www.coemed.org/files/stanags/03_AMEDP/AMedP-8.1_EDB_V1_E_2132.pdf (Accessed on 11/11/2025).
- Centers for Disease Control and Prevention (CDC) and National Institutes of Health (NIH). Biosafety in Microbiological and Biomedical Laboratories (BMBL), 6th ed. U.S. Department of Health and Human Services; 2020. https://www.cdc.gov/labs/pdf/SF __19_308133-A_ BMBL6_00-BOOK-WEB-final-3.pdf (Accessed on 15/12/2025).
- Messina, J.P.; Brady, O.J.; Scott, T.W.; Zou, C.; Pigott, D.M.; Duda, K.A.; Bhatt, S.; Katzelnick, L.; Howes, R.E.; Battle, K.E.; et al., Global spread of dengue virus types: mapping the 70-year history. Trends Microbiol. 2014, 22, 138-46. [CrossRef]
- Hawman, D.W.; Feldmann, H. Crimean-Congo haemorrhagic fever virus. Nat Rev Microbiol. 2023, 21, 463-477. [CrossRef]
- Gonzalez, E.; Gutierrez-Lopez, R. Editorial: Global movement and the spread of vector-borne diseases: challenges and strategies. Front. Trop. Dis. 2025, 6,1717661. [CrossRef]
- . WHO Ebola Response Team; Aylward, B.; Barboza, P.; Bawo, L.; Bertherat, E.; Bilivogui, P.; Blake, I.; Brennan, R.; Briand, S.; Chakauya, J.M.; Chitala, K.; et al., Ebola virus disease in West Africa--the first 9 months of the epidemic and forward projections. N Engl J Med. 2014, 371, 1481-1495. [CrossRef]
- Dembek, Z.; Hadeed, S.; Tigabu, B.; Schwartz-Watjen, K.; Glass, M.; Dressner, M.; Frankel, D.; Blaney, D.; Eccles III, T.G.; Military Medicine, Volume 2024, 189, e1470–e1478. [CrossRef]
- Moore, K.A.; Ostrowsky, J.T.; Mehr, A.J.; Johnson, R.A.; Ulrich, A.K.; Moua, N.M.; Fay, P.C.; Hart, P.J.; Golding, J.P.; Benassi, V.; et al., Lassa fever research priorities: towards effective medical countermeasures by the end of the decade. Lancet Infect Dis. 2024, 24, e696-e706. [CrossRef]
- Grahn, E.; Picard, J.; Henning, L Yellow fever. An old foe with new developments. Aust J Rural Health. 2024, 32, 455-461. [CrossRef]
- Papa, A.; Tsergouli, K.; Mirazimi, A. Crimean-Congo hemorrhagic fever: Tick–host–virus interactions. Antiviral Research. 2017, 143, 33–40. [CrossRef]
- Nicastri, E.; Kobinger, G.; Vairo, F.; Montaldo, C.; Mboera, L.E.G.; Ansunama, R.; Zumla, A.; Ippolito, G. Ebola virus disease: epidemiology, clinical features, management, and prevention. Infect Dis Clin North America. 2019, 33, :953–976. [CrossRef]
- Lau, K.A.; Theis, T.; Gray, J.; Rawlinson, W.D. Ebola preparedness: diagnosis improvement using rapid approaches for proficiency testing. J Clin Microbiol. 2017, 55, 783–790. [CrossRef]
- Takada, A. Filovirus diagnostics. Viruses. 2012, 4, 134–146.
- Turbett, S.E.; Lazarusm, J.E.; Nardini, M.A.; Braidt, J.E.; Lane, S.A.; Searle, E.F.; Biddinger, P.D.; Shenoy, E.S. Enabling laboratory readiness and preparedness for the evaluation of suspected viral hemorrhagic fevers: development of a laboratory toolkit. Infect Control Hosp Epidemiol. 2024, 45, 1-7. [CrossRef]
- Brizee, S.; Budeski, K.; James, W.; Nalabandian, M.; Bleijs, D.A.; Becker, S.J.; Wallace-Sankarsinghm, S.; Ahumibe, A.; Agogo, E.; Ihekweazu, C.; et al., Accelerating action in Global Health Security: Global biosecurity dialogue as a model for advancing the global health security agenda. Health Secur. 2019, 17(6):495-503. [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Dual Use Research of Concern in the Life Sciences: Current Issues and Controversies. Washington, DC: The National Academies Press; 2017. [CrossRef]
- Letafati, A.; Salahi Ardekani, O.; Karami, H.; Soleimani, M. Ebola virus disease: A narrative review. Microb Pathog. 2023, 181, 106213. [CrossRef]
- Gire, S.K.; Goba. A.; Andersen, K.G.; Sealfon, R.S.; Park, D.J.; Kanneh, L.; Jalloh, S.; Momoh, M.; Fullah, M.; Dudas, G.; et al., Genomic surveillance elucidates Ebola virus origin and transmission during the 2014 outbreak. Science. 2014, 345, 1369-72. [CrossRef]
- Bettini, A.; Lapa, D.; Garbuglia, A.R. Diagnostics of Ebola virus. Front Public Health. 2023, 11, 1123024. [CrossRef]
- Wilson, A.; Poeck-Goux, H.; Ruschaupt, M.; Olschner, S.; Ricks, K.M.; Smith, D.R. Development of a rapid diagnostic test to distinguish between emerging viruses that cause hemorrhagic fever. Am J Trop Med Hyg. 2025,113, 1097-1105. [CrossRef]
- Garami, A.; Agreiter, I.; Woo, J.; Stephenn Hernandez, S.; Boland, M.; Brown, K.; Connell, J.; Keogan, B.; Hare, D.; De Gascun, C.F.; et al. Laboratory diagnostic pathway for viral haemorrhagic fevers in Ireland: a review of existing guidelines and gap analysis. Discov. Viruses 2025, 2, 6. [CrossRef]
- Lee F. Diagnostics and laboratory role in outbreaks. Curr Opin Infect Dis. 2017, 30, 419-424. [CrossRef]
- United Nations Office for Disarmament Affairs (UNODA). Biological Weapons Convention: Implementation Support Unit. United Nations, 2022. Available from: https://www.un.org/disarmament/biological-weapons/ (Accessed on 9 November 2025).
- Letafati, A.; Salahi Ardekani, O.; Karami, H.; Soleimani, M. Ebola virus disease: A narrative review. Microb Pathog. 2023,181, 106213. [CrossRef]
- Gire, S.K.; Goba, A.; Andersen, K.G.; Sealfon, R.S.G.; Park, D.J.; Kanneh, L.; Jalloh, S.; Momoh, M.; Fullah, M.; Dudas, G.; et al., Genomic surveillance elucidates Ebola virus origin and transmission during the 2014 outbreak. Science. 2014, 345, 1369–1372. [CrossRef]
- Ball, A.; Wong, T.A.S.; Lehrer, A.T. Detection of serum antibodies targeting the Marburg Virus glycoprotein using a multiplex immunoassay platform. Methods Mol Biol. 2025, 2877, 345-354. [CrossRef]
- Bettini, A.; Lapa, D.; Garbuglia, A.R. Diagnostics of Ebola virus. Front Public Health. 2023, 11, 1123024. [CrossRef]
- Semancik, C.S.; Whitworth, H.S.; Price, M.A.; Yun, H.; Postler, T.S.; Zaric, M.; Kilianski, A.; Cooper, C.L.; Kuteesa, M.; Talasila, S.; et al. Seroprevalence of Antibodies to Filoviruses with Outbreak Potential in Sub-Saharan Africa: A Systematic Review to Inform Vaccine Development and Deployment. Vaccines (Basel) 2024,12, 1394. [CrossRef]
- Presser, L.D.; Coffin, J.; Koivogui, L.; Campbell, A.; Campbell, J.; Barrie, F.; Ngobeh, J.; Souma, Z.; Sorie, S.; Harding, D.; et al. The deployment of mobile diagnostic laboratories for Ebola virus disease diagnostics in Sierra Leone and Guinea. Afr J Lab Med. 2021, 10, a1414. [CrossRef]
- Luo, X.L.; Zhang, X.D.; Li, B.J.; Qin, T.; Cao, Z.J.; Fan, Q.J.; Yang, J.; Jin, D.; Lu, S.; Zheng, Y.Y.; et al. Comparative evaluation of standard RT-PCR assays and commercial real-time RT-PCR kits for detection of Lassa Virus. Microbiol Spectr. 2023, 11, e0501122. [CrossRef]
- Boisen, M.L.; Hartnett, J.N.; Shaffer, J.G.; Goba, A.; Momoh, M.; Sandi, J.D.; Fullah, M.; Nelson, D.K.S.; Bush, D.J.; Rowland, M.M.; et al., Field validation of recombinant antigen immunoassays for diagnosis of Lassa fever. Scientific Reports. 2018, 8,:5939. [CrossRef]
- Frank, M.G.; Beitscher, A.; Webb, C.M.; Raabe, V.; members of the medical countermeasures working group of the National Emerging Special Pathogens Training and Education Center’s (NETEC’s) special pathogens research network (SPRN). South American Hemorrhagic Fevers: A summary for clinicians. Int J Infect Dis. 2021,105, 505-515. [CrossRef]
- Papa, A.; Tsergouli, K.; Mirazimi, A. Crimean-Congo hemorrhagic fever: Advances in diagnosis and treatment. Clin Microbiol Infect. 2017, 23, 293–299. [CrossRef]
- Aradaib, I.E.; Erickson, B.R.; Mustafa, M.E.; Khristova, M.L.; Saeed, N.S.; Elageb, R.M.; et al. Nosocomial outbreak of Crimean-Congo hemorrhagic fever, Sudan. Emerg Infect Dis. 2010, 16, 837–839. [CrossRef]
- Kalimuddin, S.; Chia, P.Y.; Low JG, Ooi EE. Dengue and severe dengue. Clin Microbiol Rev. 2025, 38, e0024424. [CrossRef]
- Frazer, J.L.; Norton, R. Dengue: A review of laboratory diagnostics in the vaccine age. J Med Microbiol. 2024, 73, 001833. [CrossRef]
- Thergarajan, G.; Sekaran, S.D. Diagnostic approaches for dengue infection. Expert Rev Mol Diagn. 2023, 23, 643-651. [CrossRef]
- De-Simone, S.G.; Napoleão-Pêgo, P.; Lechuga, G.C.; Carvalho, J.P.R.S.; Monteiro, M.E.; Morel, C.M.; Provance, D.W. Jr. Mapping IgA epitope and cross-reactivity between severe acute respiratory syndrome-associated coronavirus 2 and DENV. Vaccines (Basel). 2023, 11, 1749. [CrossRef]
- Sarker, A.; Dhama, N.; Gupta, R.D. Dengue virus neutralizing antibody: a review of targets, cross-reactivity, and antibody-dependent enhancement. Front Immunol. 2023, 14, 200195. [CrossRef]
- Waggoner, J.J.; Rojas, A.; Pinsky, B.A. Yellow Fever Virus: diagnostics for a persistent arboviral threat. J Clin Microbiol. 2018, 56,:e00827-18. [CrossRef]
- Kimble, J.B.; Noronha, L.; Trujillo, J.D.; Mitzel, D.; Richt, J.A.; Wilson, W.C. Rift valley fever. Vet Clin North Am Food Anim Pract. 2024, 40, 293-304. [CrossRef]
- Kelly-Cirino, C.D.; Nkengasong, J.; Kettler, H.; Tongio, I.; Gay-Andrieu, F.; Escadafal, C.; Piot, P.; Peeling, R.W.; Gadde, R.; Boehme, C. Importance of diagnostics in epidemic and pandemic preparedness. BMJ Global Health. 2019, 4, e001179. [CrossRef]
- Liu L, Benyeda Z, Zohari S, Yacoub A, Isaksson M, Leijon M, LeBlanc N, Benyeda J, Belák S. Assessment of preparation of samples under the field conditions and a portable real-time RT-PCR assay for the rapid on-site detection of Newcastle disease virus. Transbound Emerg Dis. 2016, 63, e245-50. [CrossRef]
- World Health Organization (WHO). Laboratory biosafety manual. 4th ed. Geneva: World Health Organization; 2020. ISBN: 978-92-4-001131-1. Available from: https://www.who.int/publications/i/item/9789240011311. (Accessed on 12 September 2025).
- In Biosafety in Microbiological and Biomedical Laboratories, 6th ed.; Meechan, P.J.; Potts, J. (Eds.). Biosafety in Microbiological and Biomedical Laboratories (6th ed.). U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention & National Institutes of Health, 2020. https://stacks.cdc.gov/view/cdc/97733.
- World Health Organization. Laboratory guidance for the diagnosis of Ebola virus disease: interim recommendations (WHO/EVD/GUIDANCE/LAB/14.1). World Health Organization, 2014. https://iris.who.int/handle/10665/134009.
- Broadhurst, M.J.; Kelly, J.D.; Miller, A.; Semper, A.; Bailey, D.; Groppelli, E.; Simpson, A.; Brooks, T.; Hula, S.; Nyoni, W.; et al., ReEBOV Antigen Rapid Test kit for point-of-care and laboratory-based testing for Ebola virus disease: a field validation study. Lancet 2015, 386, 867–874. [CrossRef]
- Berche, P. Laboratory-associated infections and biosafety. Presse Med. 2025, 55, 104277. [CrossRef]
- Ergönül, Ö. Crimean-Congo haemorrhagic fever. Lancet Infect Dis 2006, 6, 203–214. [CrossRef]
- U.S. Department of Defense. Medical Management of Biological Casualties Handbook. 8th ed. Fort Detrick, MD: U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID), 2022. (Accessed on 11 July 2025).
- National Research Council (US) Committee on Research Standards and Practices to Prevent the Destructive Application of Biotechnology. 2004. Biotechnology Research in an Age of Terrorism. Washington, DC: National Academies Press. [CrossRef]
- Rao, V.; Bordelon, E. Mobile high-containment biological laboratories deployment: Opportunities and challenges in expeditionary deployments to outbreak response. Appl Biosafety. 2019, 24, 20–29. [CrossRef]
- Cook, B.W.; Cutts, T.A.; Nikiforuk, A.M.; Leung, A.; Kobasa, D.; Theriault, S.S. The disinfection characteristics of Ebola Virus outbreak variants. Sci Rep. 2016, 6, 38293. [CrossRef]
- Carney, T. Public health emergencies of international concern: Global, regional, and local responses to risk. Med Law Rev 2017,25, 223–239. [CrossRef]
- Heymann, D.L.; Chen, L.; Takemi, K.; Fidler, D.P.; Tappero, J.W.; Thomas, M.J.; Kenyon, T.A.; Frieden, T.R.; Yach, D.; Nishtar, S.; et al., Global health security: the wider lessons from the West African Ebola virus disease epidemic. Lancet 2015, 385,1884–1901. [CrossRef]
- Feldmann, H.; Geisbert, T.W. Ebola haemorrhagic fever. Lancet. 2011;377:849–862. [CrossRef]
- World Health Organization. Case definition recommendations for Ebola or Marburg virus diseases: interim guideline (WHO/EVD/CaseDef/14.1). World Health Organization 2014. Available on: https://www.who.int/publications/i/item/WHO-EVD-CaseDef-14.1 (Accessed on 19 November 2025.
- Ashenafi, A.; Sule, O.; Peter, T.; Mashate, S.; Otieno, O.; Kebede, A.; Oio, J.; Kao, K.; Carter, J.; Whistler, T.; et al., Diagnostics for detection and surveillance of priority epidemic-prone diseases in Africa: an assessment of testing capacity and laboratory strengthening needs. Front Public Health. 2024,12, 1438334. [CrossRef]
- Zarei, M. Advances in point-of-care technologies for molecular diagnostics. Biosens Bioelectron. 2017, 98, 494-506. [CrossRef]
- Centers for Disease Control and Prevention (CDC). Recommendations for Organizations Sending U.S.-based Personnel to Areas with VHF Outbreaks. National Center for Emerging and Zoonotic Infectious Diseases, Viral Hemorrhagic Fevers Program, U.S. Department of Health and Human Services. 2025. Available from: https://www.cdc.gov/viral-hemorrhagic-fevers/php/partners/recommendations-for-vhf-outbreaks.html. (Accessed on 10 October 2025).
- Moon, S.; Sridhar, D.; Pate, M.A.; Jha, A.K.; Clinton, C.; Delaunay, S.; Edwin, V.; Fallah, M.; Fidler, D.P.; Garrett, L.; et al., Will Ebola change the game? Ten essential reforms before the next pandemic. The report of the Harvard-LSHTM Independent Panel on the Global Response to Ebola. Lancet. 2015, 386, 2204-22421. [CrossRef]
- Ndjomou, J.; Shearrer, S.; Karlstrand, B.; Asbun, C.; Coble, J.; Alam, J.S.; Mar, M.P.; Presser, L.; Poynter, S.; Michelotti, J.M.; et al., Sustainable laboratory capacity building after the 2014 Ebola outbreak in the Republic of Guinea. Front Public Health. 2021, 9, 659504. [CrossRef]
- Jacobsen, K.H.; Aguirre, A.A.; Bailey, C.L.; Baranova, A.V.; Crooks, A.T.; Croitoru, A.; Delamater, P.L.; Gupta, J.; Kehn-Hall, K.; Narayanan, A.; et al., Lessons from the Ebola outbreak: Action items for emerging infectious disease preparedness and response. Ecohealth. 2016,13, 200-212. [CrossRef]
- Wolicki, S.B.; Nuzzo, J.B.; Blazes, D.L.; Pitts, D.L.; Iskander, J.K.; Tappero, J.W. Public health surveillance: At the Core of the Global Health Security agenda. Health Secur. 2016,14, 185-188. [CrossRef]
- Antonio, E.; Pulik, N.; Ibrahim, S.K.; Adenipekun, A.; Levanita, S.; Foster, I.; Chepkirui, D.; Harriss, E.; Sigfrid, L.; Norton, A. Research prioritisation in preparedness for and response to outbreaks of high-consequence pathogens: a scoping review. BMC Med. 2025, 23, 147. [CrossRef]
- Kelly, C.D.; Egan, C.; Cirino, N.M. The CODE RED Solution: biothreat response training for first responders. Biosecur Bioterror. 2006, 4, 391-6. [CrossRef]
- Brunker, K. Rapid pathogen surveillance: field-ready sequencing solutions. Nat Rev Genet 2024, 25, 532. [CrossRef]
- Vennis, I.M.; Boskovic M, Bleijs DA, Rutjes SA. Complementarity of international instruments in the field of biosecurity. Front Public Health 2022,10, 894389. [CrossRef]
- Kaminski, M.M.; Abudayyeh, O.O.; Gootenberg, J.S.; Zhang, F.; Collins, J.J. CRISPR-based diagnostics. Nat Biomed Eng. 2021, 5, 643-656. [CrossRef]
- Brogan, D.J.; Akbari, O.S. CRISPR Diagnostics: Advances toward the Point of Care. Biochemistry 2023, 62, 3488-3492. [CrossRef]
- Al-Antari, M.A. Artificial intelligence for medical diagnostics: existing and future AI technology! Diagnostics (Basel). 2023,13,688. [CrossRef]
- Sohmer, J.S.; Fridman, S.; Peters, D.; Jacomino, M.; Luck, G. Winning the Lottery: A Simulation study comparing scarce resource allocation protocols in crisis scenarios. Cureus. 2025, 17, e76977. [CrossRef]
- Dehury, R.K. Relevance of the World Health Organization in a multipolar world in solving global health challenges. Front Public Health. 2022, 10, 1037734. [CrossRef]
- Ayala, A.; Brush, A.; Chai, S.; Fernandez, J.; Ginsbach, K.; Gottschalk, K.; Halabi, S.; Hosangadi, D.; Mapatano, D.; Monahan, J.; et al., Advancing legal preparedness through the global health security agenda. J Law Med Ethics. 2022, 50, 200-203. [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Biodefense in the Age of Synthetic Biology. Washington, DC: The National Academies Press, 2018. [CrossRef]
- Emanuel, E.J.; Rid, A.; Miller, F.G. Ethical considerations of experimental interventions in the Ebola outbreak. Lancet. 2014, 384, 1897–1905. [CrossRef]
- Edwards, K.M.; Kochhar, S. Ethics of conducting clinical research in an outbreak setting. Annu Rev Virol. 2020, 7, 475-494. [CrossRef]
- Chala, B.; Hamde, F. Emerging and re-emerging vector-borne infectious diseases and the challenges for control: A Review. Front Public Health. 2021, 9, 715759. [CrossRef]
- Joshi J, Shah Y, Pandey K, Ojha RP, Joshi CR, Bhatt LR, Dumre SP, Acharya PR, Joshi HR, Rimal S, et al., Possible high risk of transmission of the Nipah virus in South and Southeast Asia: a review. Trop Med Health. 2023,51,44. [CrossRef]
- Hao R, Liu Y, Shen W, Zhao R, Jiang B, Song H, Yan M, Ma H. Surveillance of emerging infectious diseases for biosecurity. Sci China Life Sci. 2022, ;65,1504-1516. [CrossRef]
- Rahim, A.A.; Chandran, P.; Bindu, V.; Radhakrishnan, C.; Moorkoth, A.P.; Ramakrishnan, L.V. Recurrent Nipah outbreaks in Kerala: implications for health policy and preparedness. Front Public Health. 2024, 12, 1356515. [CrossRef]
- World Health Organization. Laboratory Biosafety Manual, 4th ed.; WHO: Geneva, Switzerland, 2020. Available from: https://www.who.int/publications/i/item/9789240011311 (Accessed on 05/02/2026).
- World Health Organization. WHO R&D Blueprint: Priority Diseases. WHO: Geneva, Switzerland, 2023. Available from https://www.emro.who.int/pandemic-epidemic-diseases/news/list-of-blueprint-priority-diseases.html (Accessed on 05 February 2026).
- Bausch, D.G.; Rollin, P.E. Ebola and Marburg virus infections. Lancet 2021, 397, 189–201.
- Yadav, P.D.; Baid, K.; Patil, D.Y.; Shirin, T.; Rahman, M.Z.; Peel, A.J.; Epstein, J.H.; Montgomerym J.M.; Plowright, R.K.; Salje, H.; et al. A One Health approach to understanding and managing Nipah virus outbreaks. Nat Microbiol. 2025, 10, 1272-1281. [CrossRef]
- Garbuglia, A.R.; Lapa, D.; Pauciullo, S.; Raoul, H.; Pannetier, D. Nipah Virus: An overview of the current status of diagnostics and their role in preparedness in endemic countries. Viruses. 2023,15, 2062. [CrossRef]
- Ganguly, A.; Mahapatra, S.; Ray, S.; Chattopadhyay, S.; Islam, M.J.; Garai, S.; Dutta, T.K.; Chattaraj, M.; Chattaraj, S. The rising threat of Nipah virus: a highly contagious and deadly zoonotic pathogen. Virology J. 2025, 22,139. [CrossRef]
- Ang, B.S.P.; Lim, T.C.C.; Wang, L. Nipah virus infection. J. Clin. Microbiol. 2018, 56, e01875-17. [CrossRef]
- Garbuglia, A.R.; Lapa, D.; Pauciullo, S.; Raoul, H.; Pannetier, D. Nipah virus diagnostics and preparedness. Viruses 2023, 15, 2062. [CrossRef]
- World Health Organization. Managing Epidemics; WHO: Geneva, Switzerland, 2018.
- Luby, S.P.; Gurley, E.S.; Hossain, M.J. Nipah virus transmission. Clin. Infect. Dis. 2009, 49, 1743–1748.
- Paessler, S.; Walker, D.H. Pathogenesis of the viral hemorrhagic fevers. Annu Rev Pathol. 2013, 8, 411-440. [CrossRef]
- Wilson, A.; Poeck-Goux, H.; Ruschaupt, M.; Olschner, S.; Ricks, K.M.; Smith, D.R. Development of a rapid diagnostic test to distinguish between emerging viruses that cause hemorrhagic fever. Am J Trop Med Hyg. 2025, 18,113,1097-1105. [CrossRef]
- World Health Organization. Laboratory Biosafety Manual, 4th ed.; WHO: Geneva, Switzerland, 2020. (Accessed on 19 January 2026).
- Centers for Disease Control and Prevention. BMBL, 6th ed.; CDC: Atlanta, GA, USA, 2020 (Accessed on 19 January 2026).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).