Submitted:
16 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Rosa canina L. is a medicinal and nutritionally valuable species with increasing industrial demand, yet its conventional propagation is limited by low rooting capacity and high genetic heterogeneity. In this study, a complete and reproducible in vitro micropropagation protocol was established, from explant introduction to plantlet acclimatization. Axillary buds were disinfected and introduced into Murashige and Skoog (MS) medium supplemented with 6-benzylaminopurine (BAP). Shoot multiplication was achieved using sequential cytokinin treatments, and shoot elongation was promoted by adding liquid MS medium containing activated charcoal (AC). The highest and fastest root induction percentage (up to 75%) was obtained on WPM with 2 mg·L-1 IBA and under 16 h light / 8 h dark photoperiod. Light promoted adventitious root formation depending on the nutrient formulation. Thereafter, shoots developed well-structured root systems in vitro, and plantlets fully survive to ex vitro acclimatization. This protocol provides an efficient platform for the large‑scale propagation of R. canina and offers new insights into the coordinated effects of mineral nutrition, hormones, and light conditions on adventitious rooting in woody species.
Keywords:
1. Introduction
2. Results and Discussion
2.1. Plants Introduction
2.2. Shoot Propagation and Elongation
2.3. Root Induction
2.3.1. Assay 1: Effect of AC
2.3.2. Assay 2: Effect of Basal Medium, Auxin and Photoperiod
2.4. Root Development
2.5. Acclimatization of Plantlets
3. Materials and Methods
3.1. Plant Material and Explant Establishment
3.2. Shoot Propagation and Elongation
3.3. Root Induction Experiments
3.3.1. Assay 1: Effect of Activated Charcoal
3.3.2. Assay 2: Effect of Basal Medium, Auxin and Photoperiod
3.4. Root Development
3.5. Acclimatization
3.6. Experimental Design and Statistical Analysis
4. Conclusions
Author Contributions
Data Availability Statement
Conflicts of Interest
Acknowledgments
Abbreviations
| MS | Murashige and Skoog |
| WPM | Woody Plant Medium |
| PGR | Plant Growth Regulator |
| BAP | 6-benzylaminopurine |
| IBA | Indole-3-butyric acid |
| GA3 | Gibberellic acid |
| NAA | Naphthaleneacetic acid |
| AC | Activated charcoal |
References
- Cunja, V.; Mikulic-Petkovsek, M.; Stampar, F.; Schmitzer, V. Compound Identification of Selected Rose Species and Cultivars: An Insight to Petal and Leaf Phenolic Profiles. Journal of American Society for Horticultural Science 2014, 139, 157–166. [CrossRef]
- D’Angiolillo, F.; Mammano, M.M.; Fascella, G. Pigments, Polyphenols and Antioxidant Activity of Leaf Extracts from Four Wild Rose Species Grown in Sicily. Not. Bot. Horti Agrobot. Cluj. Napoca. 2018, 46, 402–409. [CrossRef]
- Ouerghemmi, S.; Sebei, H.; Siracusa, L.; Ruberto, G.; Saija, A.; Cimino, F.; Cristani, M. Comparative Study of Phenolic Composition and Antioxidant Activity of Leaf Extracts from Three Wild Rosa Species Grown in Different Tunisia Regions: Rosa Canina L., Rosa Moschata Herrm. and Rosa Sempervirens L. Ind. Crops Prod. 2016, 94, 167–177. [CrossRef]
- Phetcharat, L.; Wongsuphasawat, K.; Winther, K. The Effectiveness of a Standardized Rose Hip Powder, Containing Seeds and Shells of Rosa Canina, on Cell Longevity, Skin Wrinkles, Moisture, and Elasticity. Clin. Interv. Aging 2015, 10, 1849–1856. [CrossRef]
- D’Espessailles, A.; Dossi, C.G.; Espinosa, A.; González-Mañán, D.; Tapia, G.S. Dietary Rosa Mosqueta (Rosa Rubiginosa) Oil Prevents High Diet-Induced Hepatic Steatosis in Mice. Food Funct. 2015, 6, 3109–3116. [CrossRef]
- Tapia, G.; Silva, D.; Romero, N.; Pettinelli, P.; Dossi, C.G.; de Miguel, M.; González-Mañán, D. Role of Dietary α- and γ-Tocopherol from Rosa Mosqueta Oil in the Prevention of Alterations Induced by High-Fat Diet in a Murine Model. Nutrition 2018, 53, 1–8. [CrossRef]
- Dossi, C.G.; Cadagan, C.; San Martín, M.; Espinosa, A.; González-Mañán, D.; Silva, D.; Mancilla, R.A.; Tapia, G.S. Effects of Rosa Mosqueta Oil Supplementation in Lipogenic Markers Associated with Prevention of Liver Steatosis. Food Funct. 2017, 8, 832–841. [CrossRef]
- Ercisli, S.; Güleryüz, M. Rose Hip Utilization in Turkey. Acta Hortic. 2005, 77–82. [CrossRef]
- Yildiz, O.; Alpaslan, M. Properties of Rose Hip Marmalades. Food Technol. Biotechnol. 2012, 1, 98–106.
- Hornero-Méndez, D.; Mínguez-Mosquera, M.I. Carotenoid Pigments in Rosa Mosqueta Hips, an Alternative Carotenoid Source for Foods. J. Agric. Food Chem. 2000, 48, 825–828. [CrossRef]
- Vossen, E.; Utrera, M.; De Smet, S.; Morcuende, D.; Estévez, M. Dog Rose (Rosa Canina L.) as a Functional Ingredient in Porcine Frankfurters without Added Sodium Ascorbate and Sodium Nitrite. Meat Sci. 2012, 92, 451–457. [CrossRef]
- Utrera, M.; Morcuende, D.; Ganhão, R.; Estévez, M. Role of Phenolics Extracting from Rosa Canina L. on Meat Protein Oxidation During Frozen Storage and Beef Patties Processing. Food Bioproc. Tech. 2015, 8, 854–864. [CrossRef]
- Armenteros, M.; Morcuende, D.; Ventanas, J.; Estévez, M. The Application of Natural Antioxidants via Brine Injection Protects Iberian Cooked Hams against Lipid and Protein Oxidation. Meat Sci. 2016, 116, 253–259. [CrossRef]
- Gudin, S.; Arene, L. Influence of Endocarp Thickness on Rose Achene Germination: Genetic and Environmental Factors. HortScience 1990, 25, 786–788.
- Hosafci, H.; Arslan, N. Propagation of Dogrose ( Rosa Canina L .) Plants by Softwood Cuttings. Acta Hortic. 2005, 690, 139–142. [CrossRef]
- Kazankaya, A.; Yörük, E.; Dogan, A. Effect of IBA on Rooting of Rosa Canina Hardwood Cuttings from Lake Van Region, Turkey. Acta Hortic. 2005, 153–158. [CrossRef]
- Fascella, G.; Giardina, G.; Maggiore, P.; Giovino, A.; Scibetta, S. Distribution, Habitats, Characterization and Propagation of Sicilian Rose Species. Acta Hortic. 2015, 1064, 31–38. [CrossRef]
- Carelli, B.P.; Echeverrigaray, S. An Improved System for the in Vitro Propagation of Rose Cultivars. Sci. Hortic. 2002, 92, 69–74. [CrossRef]
- Shirdel, M.; Motallebi-Azar, A.; Matloobi, M.; Zaare-Nahandi, F. Effects of Nodal Position and Growth Regulators on In Vitro Growth of Dog Rose (Rosa Canina). Journal of Ornamental and Horticultural Plants 2013, 3, 9–17.
- Singh, S.K.; Syamal, M.M. Anti-Auxin Enhance Rosa Hybrida L. Micropropagation. Biol. Plant. 2000, 43, 279–281.
- Davoudi Pahnekolayi, M.; Tehranifar, A.; Samiei, L.; Shoor, M. Optimization of the Micro-Propagation Protocol of Two Native Rose Species of Iran (Rosa Canina and Rosa Beggeriana). Acta Hortic. 2016, 1131, 87–96. [CrossRef]
- Dordas, C.; Brown, P.H. Boron Deficiency Affects Cell Viability, Phenolic Leakage and Oxidative Burst in Rose Cell Cultures. Plant Soil 2005, 268, 293–301. [CrossRef]
- Pan, M.J.; Van Staden, J. The Use of Charcoal in in Vitro Culture - A Review. Plant Growth Regul. 1998, 26, 155–163. [CrossRef]
- Constantin, M.J.; Henke, R.R.; Mansur, M.A. Effect of Activated Charcoal on Callus Growth and Shoot Organogenesis in Tobacco. In Vitro 1977, 13, 293–296. [CrossRef]
- Dencso, I. Factors Influencing Vitrification of Carnation and Conifers. In Proceedings of the Acta Horticulturae; International Society for Horticultural Science (ISHS), Leuven, Belgium, September 1 1987; pp. 167–176.
- Weatherhead, M.A.; Burdon, J.; Henshaw, G.G. Some Effects of Activated Charcoal as an Additive to Plant Tissue Culture Media. Zeitschrift für Pflanzenphysiologie 1978, 89, 141–147. [CrossRef]
- Lloyd, G.; McCown, B. Commercially-Feasible Micropropagation of Mountain Laurel, Kalmia Latifolia, by Use of Shoot-Tip Culture. Combined Proceedings, International Plant Propagators’Society 1980, 30, 421–427.
- Misra, P.; Chakrabarty, D. Clonal Propagation of Rosa Clinophylla Thory. through Axillary Bud Culture. Sci. Hortic. 2009, 119, 212–216. [CrossRef]
- Palacios-Arriaga, A.H.; Rout, N.P. Effect of Different Media and Rooting Substrates on the Hardening-off Process of Transgenic Rose. Agronomía Mesoamericana 2019, 30, 115–129. [CrossRef]
- Brunda, S.M.; Rani, C.L.; Rajendran, P.; Smitha, R.; Priya, L. In Vitro Propagation of Rosa Hybrida ‘Golden Fairy’ through Nodal Explants. Acta Hortic. 2017, 87–90. [CrossRef]
- Pati, P.K.; Rath, S.P.; Sharma, M.; Sood, A.; Ahuja, P.S. In Vitro Propagation of Rose—a Review. Biotechnol. Adv. 2006, 24, 94–114. [CrossRef]
- Toma, R.; Al-Mizory, L.S.M.; Faizy, H.S. Rooting Response of Rosa Canina and Cotoneaster Acuminatus to Different in Vitro Factors. American Journal of Experimental Agriculture 2014, 4, 724–731. [CrossRef]
- Wynne, J.; McDonald, M.S. Adventitious Root Formation in Woody Plant Tissue: The Influence of Light and Indole-3-Butyric Acid (IBA) on Adventitious Root Induction in Betula Pendula. In Vitro Cell.Dev.Biol.-Plant 2002, 210–212. [CrossRef]
- Van Gelderen, K.; Kang, C.; Pierik, R. Light Signaling, Root Development, and Plasticity. Plant Physiol. 2018, 176, 1049–1060. [CrossRef]
- Bao, Y.; Liu, X.; Feng, C.-H.; Niu, M.-X.; Liu, C.; Wang, H.-L.; Yin, W.; Xia, X. Light and Light Signals Regulate Growth and Development in Woody Plants. Forests 2024, 15, 523. [CrossRef]
- Han, J.; Li, Y.; Zhao, Y.; Sun, Y.; Li, Y.; Peng, Z. How Does Light Regulate Plant Regeneration? Front. Plant Sci. 2025, 15. [CrossRef]
- Dolcet-Sanjuan, R.; Claveria, E.; Gruselle, R.; Meier-Dinkel, A.; Jay-Allemand, C.; Gaspar, T. Practical Factors Controlling in Vitro Adventitious Root Formation from Walnut Shoot Microcuttings. J. Amer. Soc. Hort. Sci. 2004, 129, 198–203.
- Iglesias, I.; Vilardell, P.; Bonany, J.; Claveria, E.; Dolcet-Sanjuan, R. Micropropagation and Field Evaluation of the Pear (Pyrus Communis L.) `IGE 2002’, A New Selection of the Cultivar Dr. Jules Guyot. Journal of the American Society for Horticultural Science 2004, 129, 389–393. [CrossRef]
- Baas, R.; Berg, D.; Gislerød, H.R. Do Roots Of Rose Cuttings Suffer From Oxygen Deficiency During Propagation In Rockwool? (Refereed). Acta Hortic. 1997, 123–132. [CrossRef]
- Gislerød, H.R.; Baas, R.; Warmenhoven, M.; Berg, D. Effect Of Aeration On Rooting And Growth Of Roses (Refereed). Acta Hortic. 1997, 113–122. [CrossRef]
- Casanovas, M.; Claveria, E.; Dolcet-Sanjuan, R. Development of a Feasible and Efficient In Vitro Rescue Protocol for Immature Prunus Spp. Embryos. Plants 2024, 13. [CrossRef]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bioassays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 473–497. [CrossRef]







| Rooting treatment | |||||||||
| T1 | T2 | ||||||||
| NO AC | AC | ||||||||
| Weeks | Mean | SE | Mean | SE | |||||
| 2 | 22.2 a* | 2.8 | 16.7 a | 0.0 | |||||
| 3 | 25.0 a | 0.0 | 16.7 b | 0.0 | |||||
| 4 | 25.0 a | 0.0 | 16.7 b | 0.0 | |||||
| 5 | 25.0 a | 0.0 | 16.7 b | 0.0 | |||||
| 6 | 30.6 a | 2.8 | 16.7 b | 0.0 | |||||
| 7 | 36.1 a | 2.8 | 50.0 a | 4.8 | |||||
| 8 | 41.7 b | 0.0 | 58.3 a | 4.8 | |||||
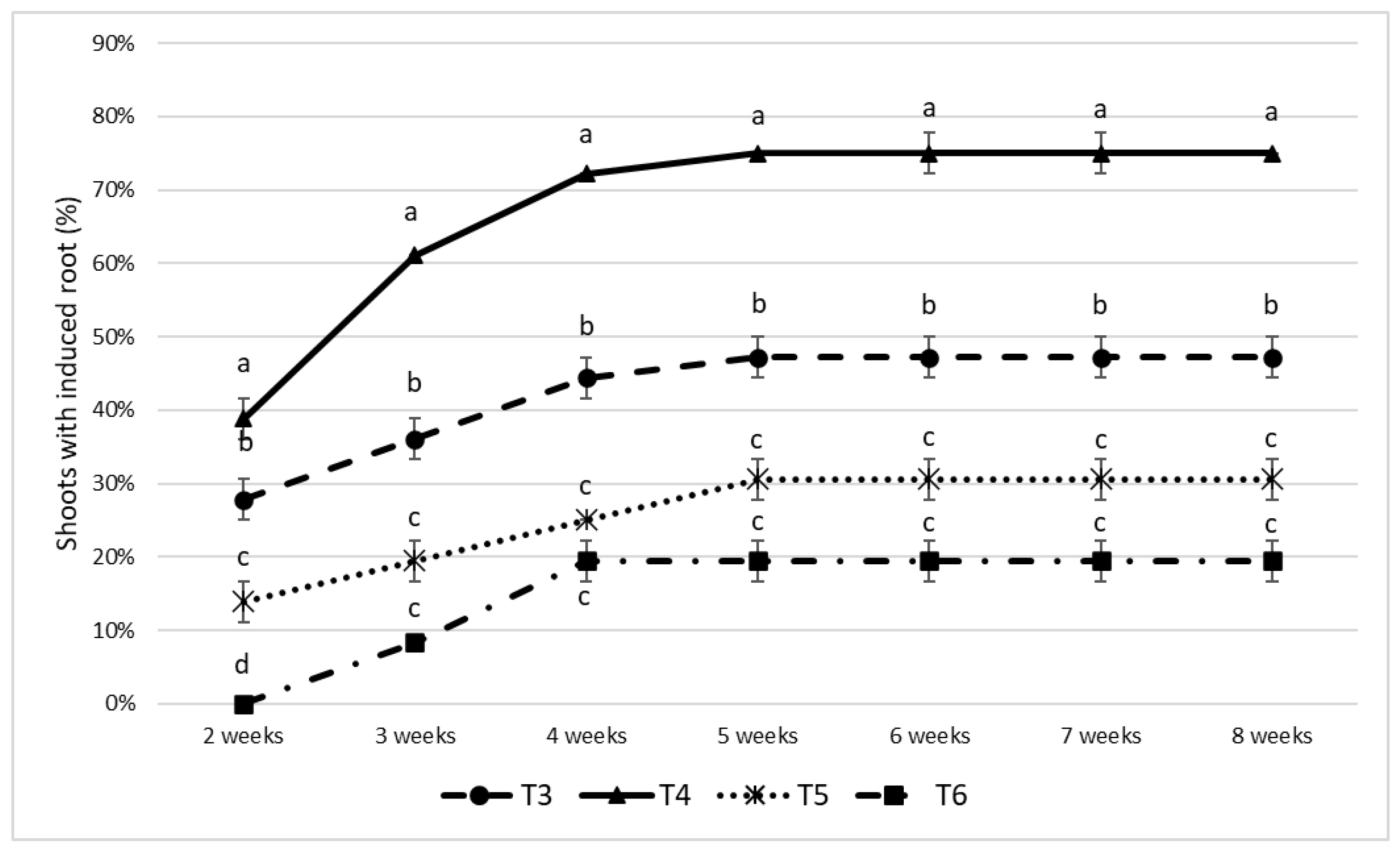
| Treatment | Basal medium | Additional Compounds | Photoperiod conditions |
| T1 | WPM | None | 16h light / 8h dark |
| T2 | WPM | Activated charcoal (3 g·L-1) | 16h light / 8h dark |
| Treatment | Basal medium | PGRS | Photoperiod conditions |
| T3 | WPM | IBA (2 mg·L⁻¹) | Continuous darkness (24h) |
| T4 | WPM | IBA (2 mg·L⁻¹) | 16 h light / 8 h dark |
| T5 | MS | IBA (2 mg·L⁻¹) | Continuous darkness (24h) |
| T6 | MS | IBA (2 mg·L⁻¹) | 16 h light / 8 h dark |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




