Submitted:
14 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
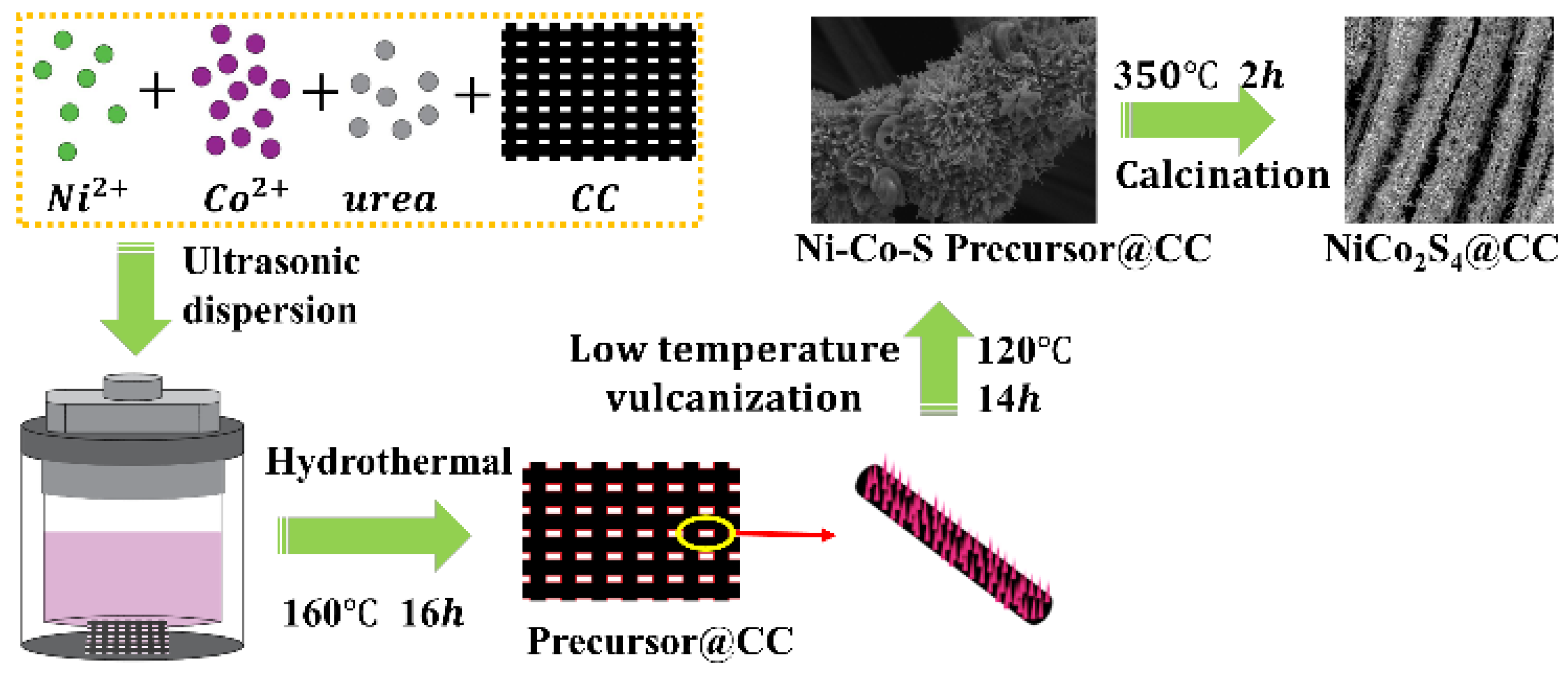
2.2. Synthesis of NiCo2O4@CC
2.3. Synthesis of NiCo2S4@CC
2.4. Characterization of Materials
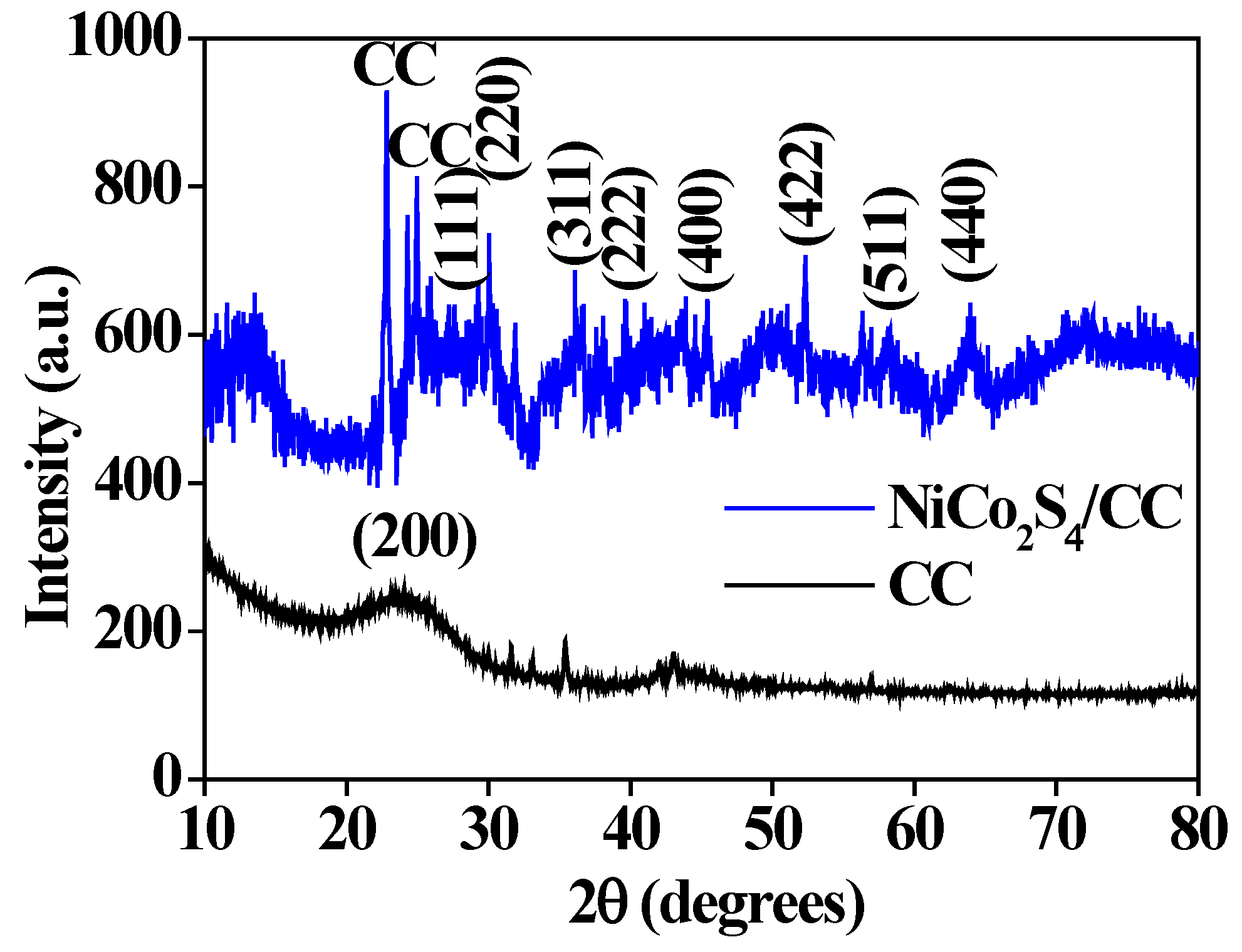
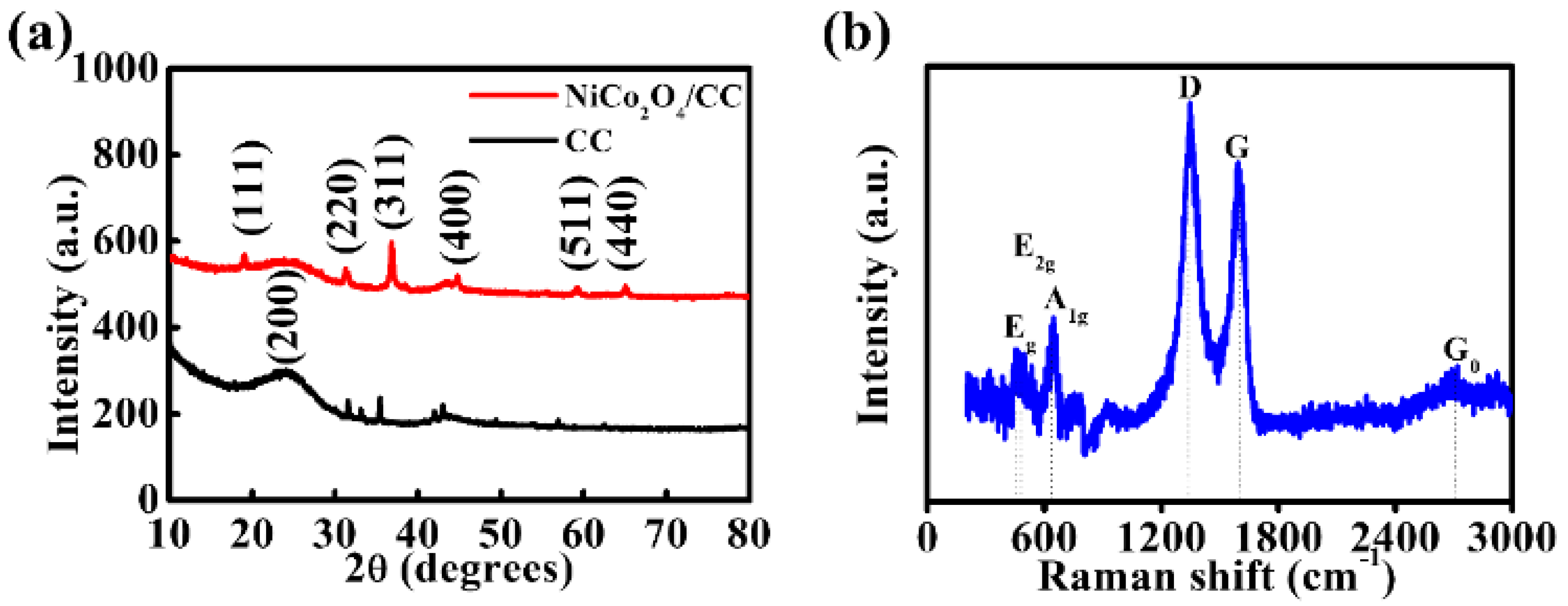
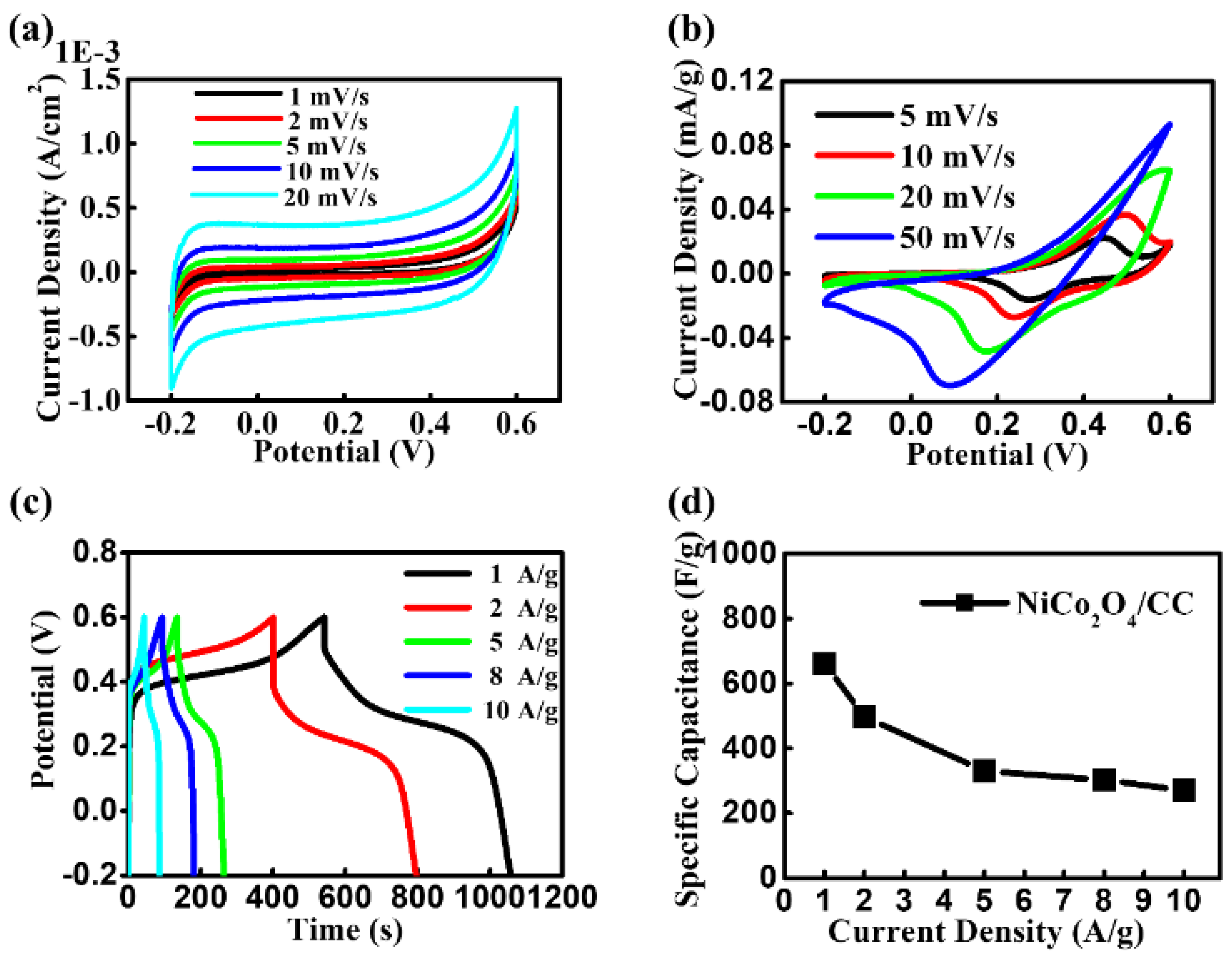
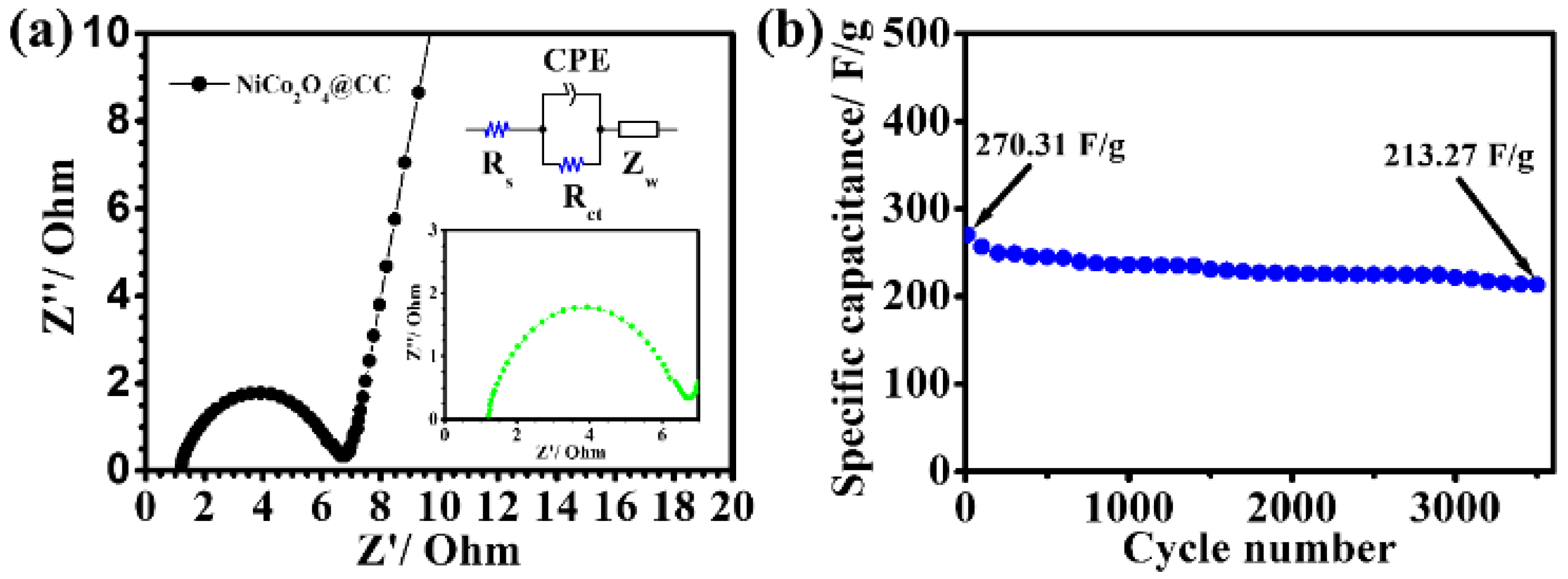
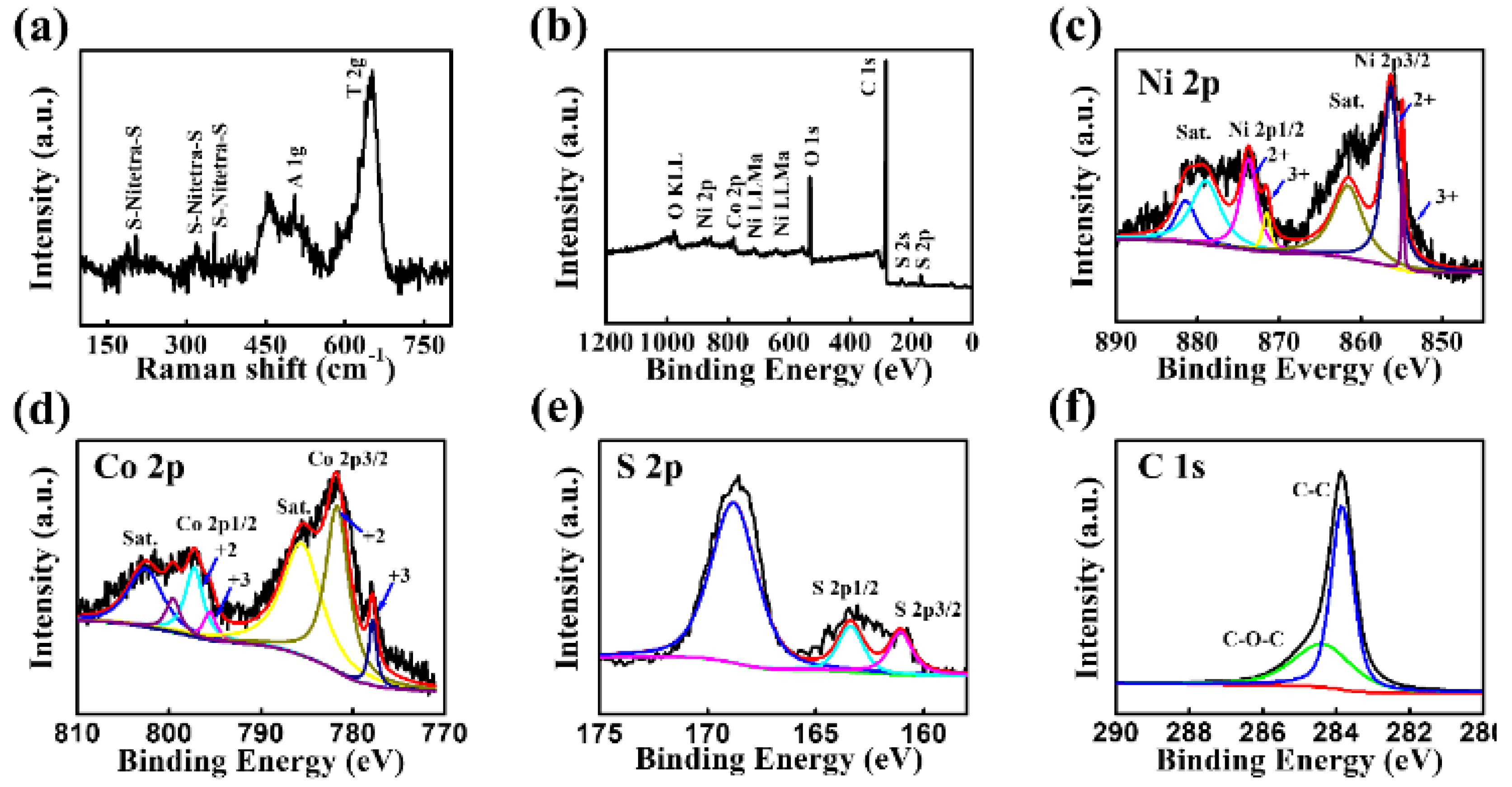
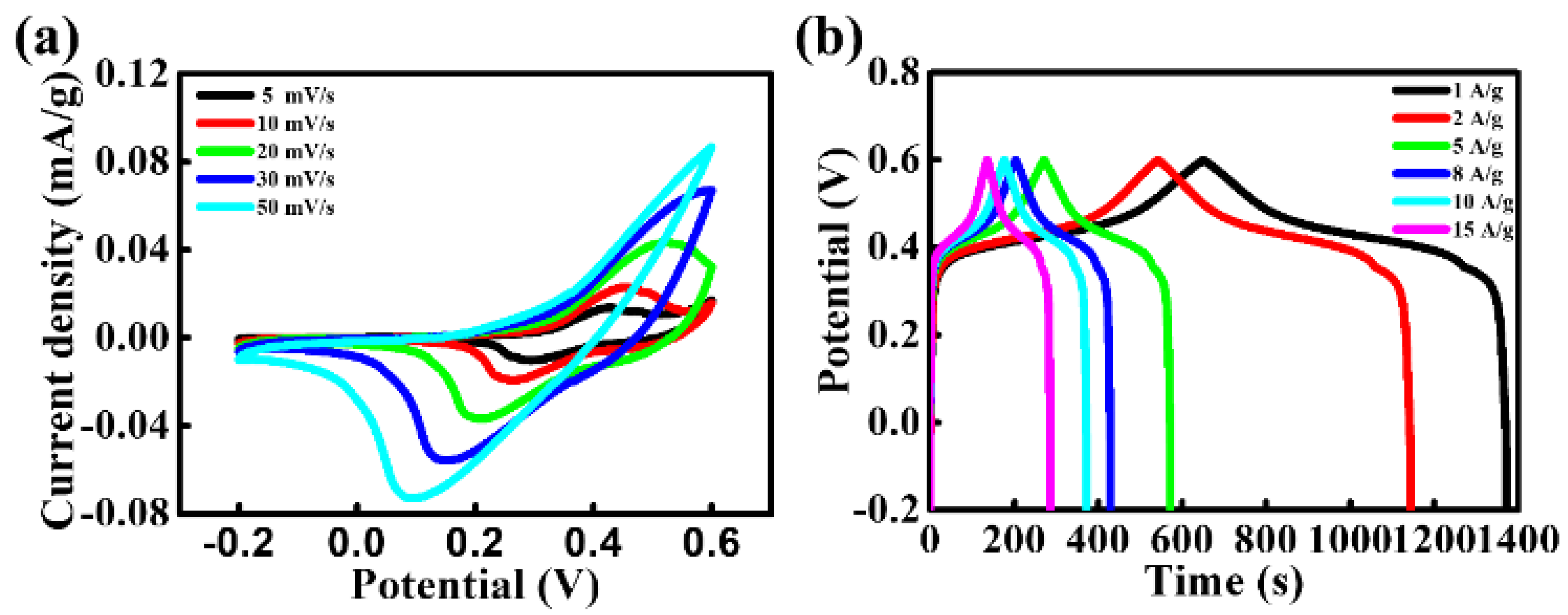
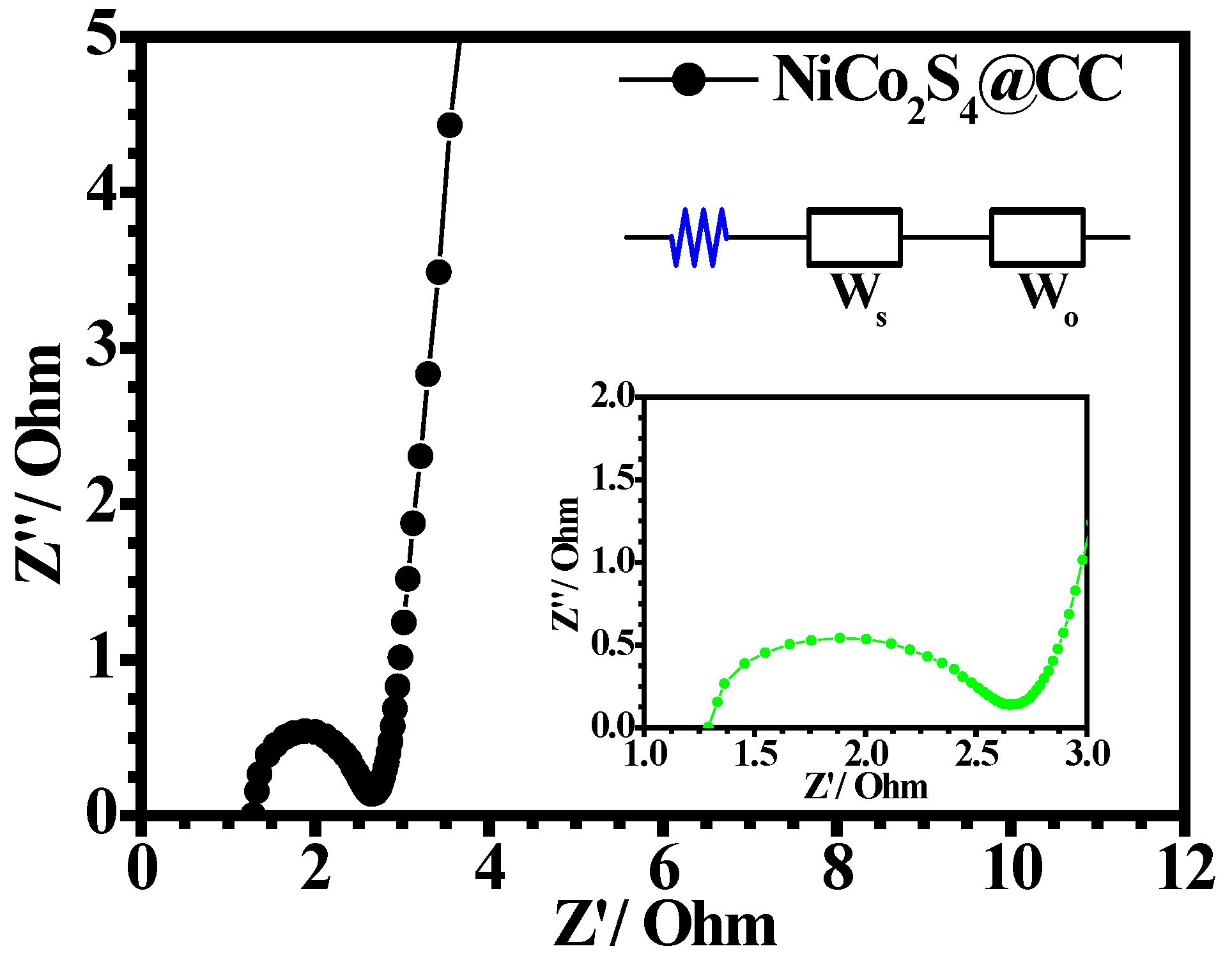
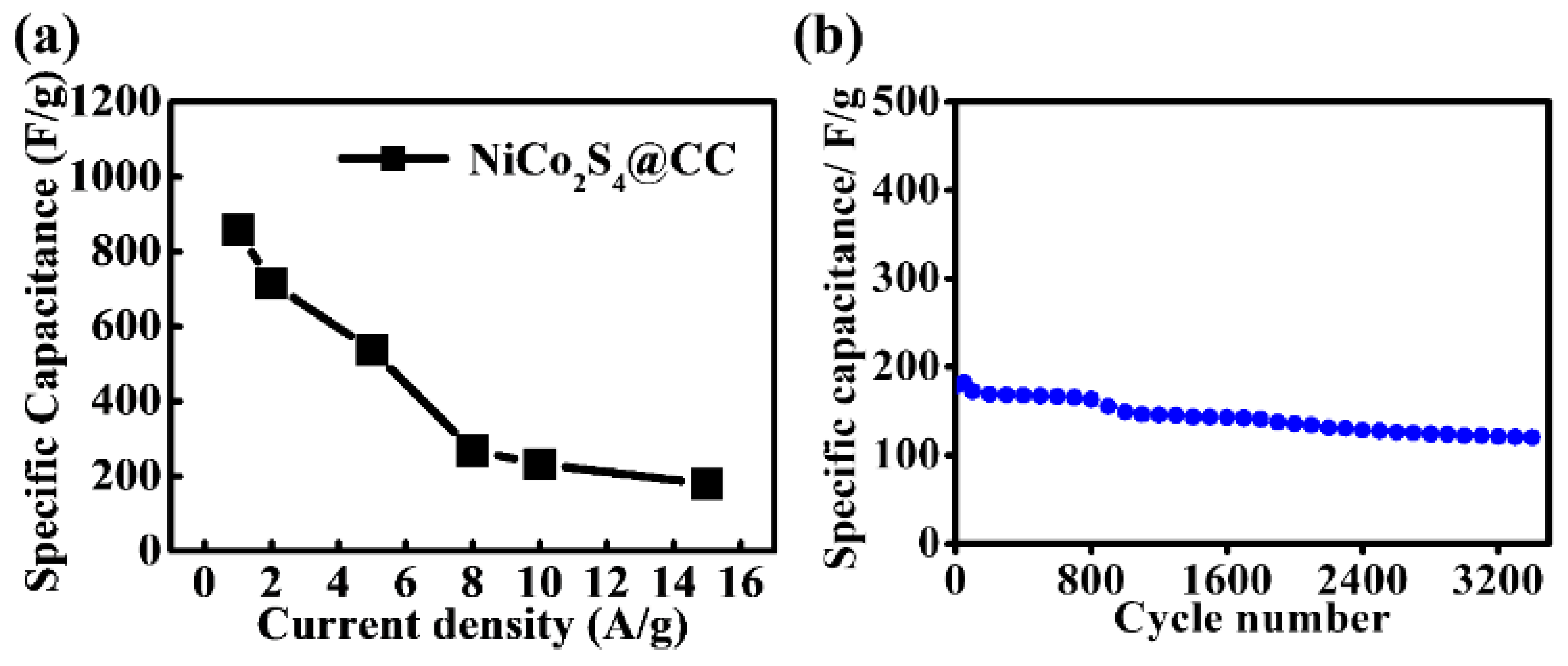
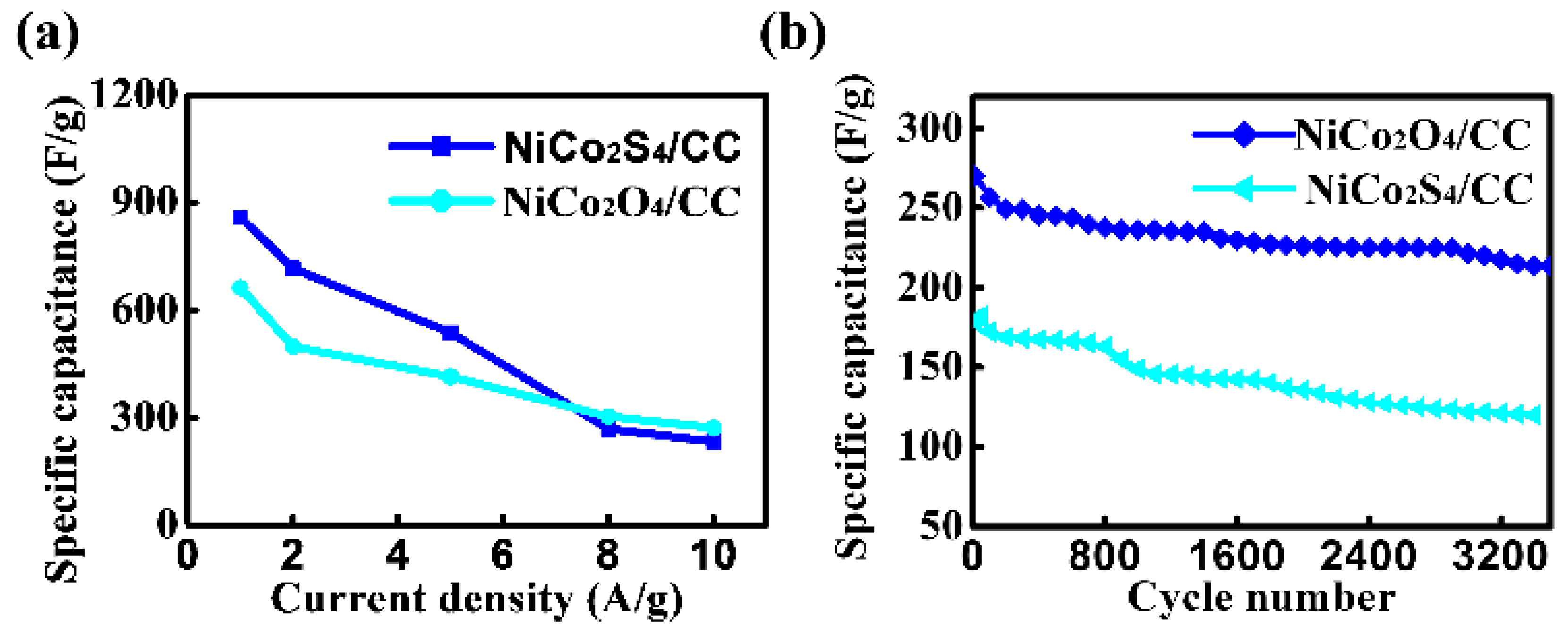
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Conway, B.E. Elements of Electrostatics Involved in Treatment of Double Layers and Ions at Capacitor Electrode Interphases. In Electrochemical supercapacitors: Scientific fundamentals and technological applications; Kluwer Academic/Plenum Press: New York, NY, USA, 1999; Volume 4, pp. 67–68. [Google Scholar]
- Lv, H.; Pan, Q.; Song, Y.; Liu, X.-X.; Liu, T. A Review on Nano-/Microstructured Materials Constructed by Electrochemical Technologies for Supercapacitors. Nano-Micro Lett. 2020, 12, 1–56. [Google Scholar] [CrossRef] [PubMed]
- Zaharaddeen, S. Iro.; Subramani, C.; S.S. A brief review on electrode materials for supercapacitor. Int. J. Electrochem. Sci 2016, 11, 10628–10643. [Google Scholar]
- Zhong, C.; Deng, Y. D.; Hu, W. B.; Qiao, J. L.; Zhang, L.; Zhang, J. J. A review of electrode materials and compositions for electrochemical supercapacitors. Chem. Soc. Rev 2015, 44, 7484–7539. [Google Scholar] [CrossRef]
- Bothe, A.; Pourhosseini, S.; Ratajczak, P.; Beguin, F.; Balducci, A. Towards understanding the impact of operating voltage on the stability of adiponitrile-based electrical double-layer capacitors. J. Power Sources 2021, 496. [Google Scholar] [CrossRef]
- Eskandari, M.; Malekfar, R.; Buceta, D.; Taboada, P. NiCo2O4-based nanostructured composites for high-performance pseudocapacitor electrodes. Colloids Surfaces A: Physicochem. Eng. Asp. 2020, 584. [Google Scholar] [CrossRef]
- Sun, P.; Zhang, J.; Huang, J.; Wang, L.; Wang, P.; Cai, C.; Lu, M.; Yao, Z.; Yang, Y. Bimetallic MOF-derived (CuCo)Se nanoparticles embedded in nitrogen-doped carbon framework with boosted electrochemical performance for hybrid supercapacitor. Mater. Res. Bull. 2021, 137. [Google Scholar] [CrossRef]
- Kötz, R.; Carlen, M. Principles and applications of electrochemical capacitors. Electrochim. Acta 2000, 45, 2483–2498. [Google Scholar] [CrossRef]
- Halper, M. S.; Ellenbogen, J. C. Supercapacitors: A Brief Overview; The MITRE Corporation: McLean, Virginia, USA, 2006; pp. 10–12. [Google Scholar]
- Kim, B. K.; Sy, S.; Yu, A. P.; Zhang, J. J. Handbook of Clean Energy Systems; John Wiley & Sons Press: Hoboken, New Jersey, USA, 2015; pp. 21–25. [Google Scholar]
- Wang, Y.; Diaz, D.F.R.; Chen, K.S.; Wang, Z.; Adroher, X.C. Materials, technological status, and fundamentals of PEM fuel cells—A review. Mater. Today 2020, 32, 178–203. [Google Scholar] [CrossRef]
- Sun, J.; Xu, C.; Chen, H. A review on the synthesis of CuCo2O4-based electrode materials and their applications in supercapacitors. J. Materiomics 2021, 7, 98–126. [Google Scholar] [CrossRef]
- Fischer, U.; Saliger, R.; Bock, V.; Petricevic, R.; Fricke, J. Carbon Aerogels as Electrode Material in Supercapacitors. J. Porous Mater. 1997, 4, 281–285. [Google Scholar] [CrossRef]
- Liu, C. F.; Liu, Y. C.; Yi, T. Y.; Hu, C. C. Carbon materials for high-voltage supercapacitors. Carbon 2019, 145, 529–548. [Google Scholar] [CrossRef]
- Jain, A.; Ghosh, M.; Krajewski, M.; Kurungot, S.; Michalska, M. Biomass-derived activated carbon material from native European deciduous trees as an inexpensive and sustainable energy material for supercapacitor application. J. Energy Storage 2021, 34. [Google Scholar] [CrossRef]
- Shirakawa, H.; Louis, E.J.; MacDiarmid, A.G.; Chiang, C.K.; Heeger, A.J. Synthesis of electrically conducting organic polymers: halogen derivatives of polyacetylene, (CH) x. J. Chem. Soc. Chem. Commun. 1977, 578–580. [Google Scholar] [CrossRef]
- MacDiarmid, A.G.; Mammone, R.J.; Kaner, R.B.; Porter, L.; Porter, S.J.; Pethig, R.; Heeger, A.J.; Rosseinsky, D.R. The concept of ‘doping’ of conducting polymers: the role of reduction potentials. Philos. Trans. R. Soc. London. Ser. A, Math. Phys. Sci. 1985, 314, 3–15. [Google Scholar] [CrossRef]
- Balint, R.; Cassidy, N.J.; Cartmell, S.H. Conductive polymers: Towards a smart biomaterial for tissue engineering. Acta Biomater. 2014, 10, 2341–2353. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhou, A.; Zhang, X.; Mu, J.; Che, H.; Wang, Y.; Wang, T.-T.; Zhang, Z.; Kou, Z. Fundamentals, advances and challenges of transition metal compounds-based supercapacitors. Chem. Eng. J. 2021, 412. [Google Scholar] [CrossRef]
- Hassan, I.U.; Salim, H.; Naikoo, G.A.; Awan, T.; Dar, R.A.; Arshad, F.; Tabidi, M.A.; Das, R.; Ahmed, W.; Asiri, A.M.; et al. A review on recent advances in hierarchically porous metal and metal oxide nanostructures as electrode materials for supercapacitors and non-enzymatic glucose sensors. J. Saudi Chem. Soc. 2021, 25. [Google Scholar] [CrossRef]
- Lin, C.; Ritter, J.A.; Popov, B.N. Development of Carbon-Metal Oxide Supercapacitors from Sol-Gel Derived Carbon-Ruthenium Xerogels. J. Electrochem. Soc. 1999, 146, 3155–3160. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Z.; Yu, Z.; Wei, L.; Guo, X. Mesoporous NiMoO4 microspheres decorated by Ag quantum dots as cathode material for asymmetric supercapacitors: Enhanced interfacial conductivity and capacitive storage. Appl. Surf. Sci. 2020, 505. [Google Scholar] [CrossRef]
- Ginting, R.T.; Ovhal, M.M.; Kang, J.-W. A novel design of hybrid transparent electrodes for high performance and ultra-flexible bifunctional electrochromic-supercapacitors. Nano Energy 2018, 53, 650–657. [Google Scholar] [CrossRef]
- Liu, X.Y.; Gao, Y.Q.; Yang, G.W. A flexible, transparent and super-long-life supercapacitor based on ultrafine Co3O4nanocrystal electrodes. Nanoscale 2016, 8, 4227–4235. [Google Scholar] [CrossRef]
- Shen, L.; Du, L.; Tan, S.; Zang, Z.; Zhao, C.; Mai, W. Flexible electrochromic supercapacitor hybrid electrodes based on tungsten oxide films and silver nanowires. Chem. Commun. 2016, 52, 6296–6299. [Google Scholar] [CrossRef] [PubMed]
- Sial, Q.A.; Javed, M.S.; Lee, Y.-J.; Duy, L.T.; Seo, H. Flexible and transparent graphene-based supercapacitors decorated with nanohybrid of tungsten oxide nanoflakes and nitrogen-doped-graphene quantum dots. Ceram. Int. 2020, 46, 23145–23154. [Google Scholar] [CrossRef]
- Barik, R.; Tanwar, V.; Kumar, R.; Ingole, P.P. A high energy density and high rate capability flexible supercapacitor based on electro-spun highly porous SnO2@carbon nanofibers. J. Mater. Chem. A 2020, 8, 15110–15121. [Google Scholar] [CrossRef]
- Rowlands, S.E.; Latham, R.J.; Schlindwein, W.S. Supercapacitor devices using porous silicon electrodes. Ionics 1999, 5, 144–149. [Google Scholar] [CrossRef]
- Dai, P.; Zhang, S.; Liu, H.; Yan, L.; Gu, X.; Li, L.; Liu, D.; Zhao, X. Cotton fabrics-derived flexible nitrogen-doped activated carbon cloth for high-performance supercapacitors in organic electrolyte. Electrochimica Acta 2020, 354. [Google Scholar] [CrossRef]
- Liu, T.; Zheng, Y.; Zhao, W.; Cui, L.; Liu, J. Uniform generation of NiCo2S4 with 3D honeycomb-like network structure on carbon cloth as advanced electrode materials for flexible supercapacitors. J. Colloid Interface Sci. 2019, 556, 743–752. [Google Scholar] [CrossRef]
- Zhao, W.; Zheng, Y.; Cui, L.; Jia, D.; Wei, D.; Zheng, R.; Barrow, C.; Yang, W.; Liu, J. MOF derived Ni-Co-S nanosheets on electrochemically activated carbon cloth via an etching/ion exchange method for wearable hybrid supercapacitors. Chem. Eng. J. 2019, 371, 461–469. [Google Scholar] [CrossRef]
- Ghosh, D.; Mandal, M.; Das, C.K. Solid State Flexible Asymmetric Supercapacitor Based on Carbon Fiber Supported Hierarchical Co(OH)xCO3 and Ni(OH)2. Langmuir 2015, 31, 7835–7843. [Google Scholar] [CrossRef]
- Horng, Y.-Y.; Lu, Y.-C.; Hsu, Y.-K.; Chen, C.-C.; Chen, L.-C.; Chen, K.-H. Flexible supercapacitor based on polyaniline nanowires/carbon cloth with both high gravimetric and area-normalized capacitance. J. Power Sources 2010, 195, 4418–4422. [Google Scholar] [CrossRef]
- Sekhar, S.C.; Nagaraju, G.; Yu, J.S. Conductive silver nanowires-fenced carbon cloth fibers-supported layered double hydroxide nanosheets as a flexible and binder-free electrode for high-performance asymmetric supercapacitors. Nano Energy 2017, 36, 58–67. [Google Scholar] [CrossRef]
- Shakir, I.; Shahid, M.; Rana, U.A.; Al Nashef, I.M.; Hussain, R. Nickel–Cobalt Layered Double Hydroxide Anchored Zinc Oxide Nanowires grown on Carbon Fiber Cloth for High-Performance Flexible Pseudocapacitive Energy Storage Devices. Electrochimica Acta 2014, 129, 28–32. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Q.; Liu, P. Poly(1,5-diaminoanthraquinone) coated carbon cloth composites as flexible electrode with extraordinary cycling stability for symmetric solid-state supercapacitors. J. Colloid Interface Sci. 2019, 546, 60–69. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, J.; Guo, H.; Tong, X. Hierarchically porous nickel oxide nanoflake arrays grown on carbon cloth by chemical bath deposition as superior flexible electrode for supercapacitors. Mater. Lett. 2014, 136, 198–201. [Google Scholar] [CrossRef]
- Liu, T.; Zheng, Y.; Zhao, W.; Cui, L.; Liu, J. Uniform generation of NiCo2S4 with 3D honeycomb-like network structure on carbon cloth as advanced electrode materials for flexible supercapacitors. J. Colloid Interface Sci. 2019, 556, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Rowlands, S.E.; Latham, R.J.; Schlindwein, W.S. Supercapacitor devices using porous silicon electrodes. Ionics 1999, 5, 144–149. [Google Scholar] [CrossRef]
- Dai, P.; Zhang, S.; Liu, H.; Yan, L.; Gu, X.; Li, L.; Liu, D.; Zhao, X. Cotton fabrics-derived flexible nitrogen-doped activated carbon cloth for high-performance supercapacitors in organic electrolyte. Electrochimica Acta 2020, 354. [Google Scholar] [CrossRef]
- Wu, P.; Cheng, S.; Yao, M.; Yang, L.; Zhu, Y.; Liu, P.; Xing, O.; Zhou, J.; Wang, M.; Luo, H.; et al. A Low-Cost, Self-Standing NiCo2O4@CNT/CNT Multilayer Electrode for Flexible Asymmetric Solid-State Supercapacitors. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef]
- Pu, J.; Cui, F.; Chu, S.; Wang, T.; Sheng, E.; Wang, Z. Preparation and Electrochemical Characterization of Hollow Hexagonal NiCo2S4 Nanoplates as Pseudocapacitor Materials. ACS Sustain. Chem. Eng. 2014, 2, 809–815. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




