Submitted:
14 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
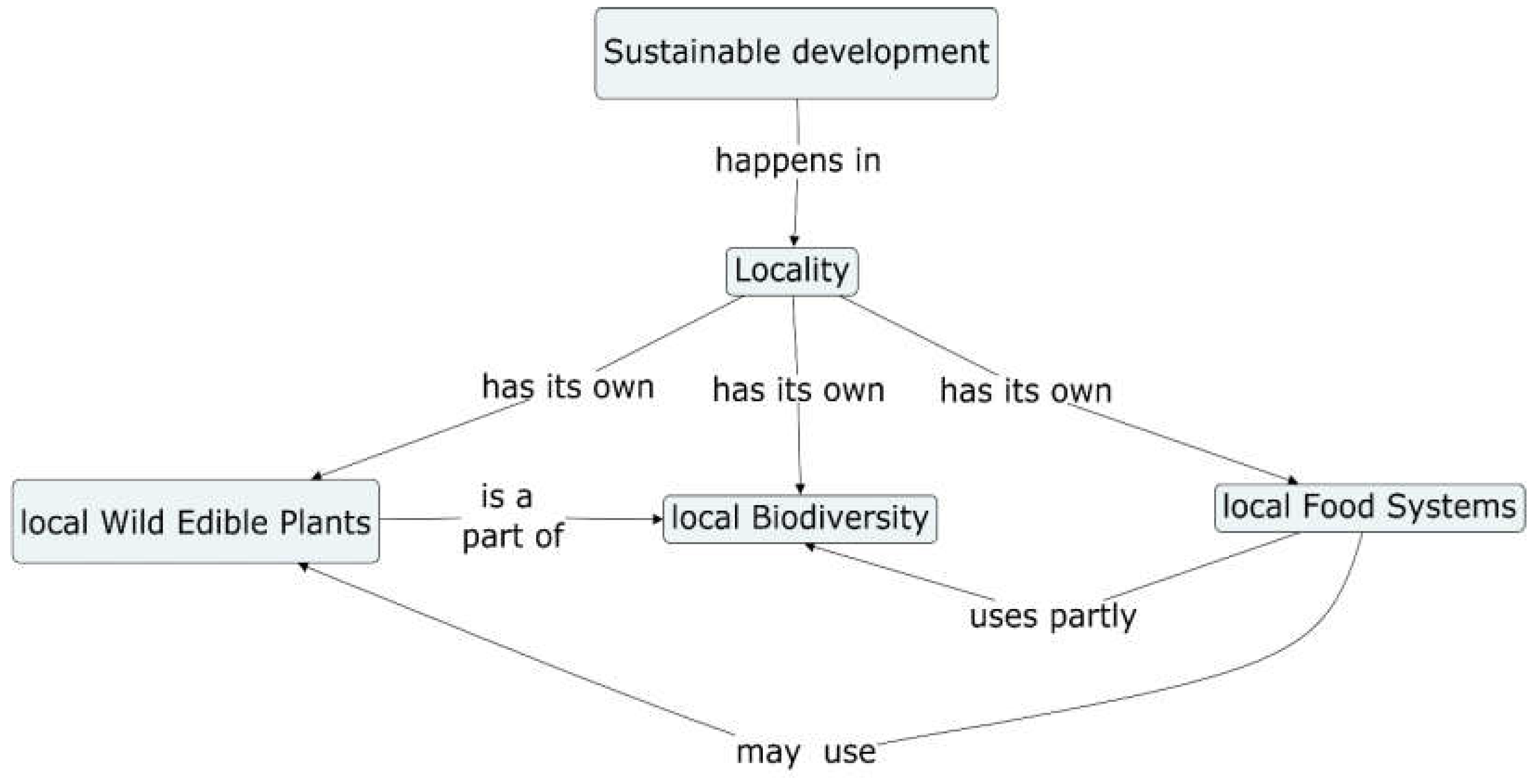
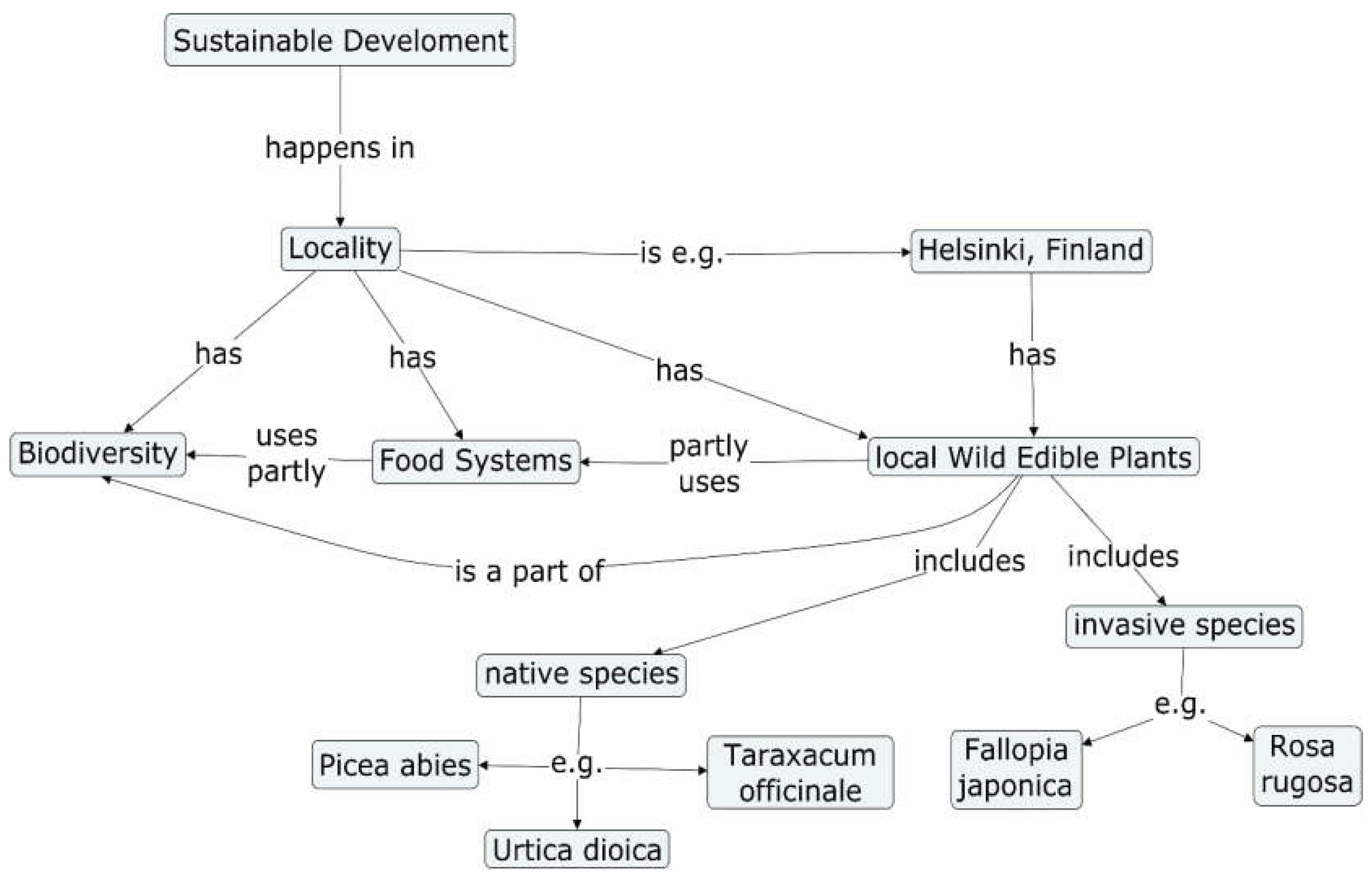
1. Introduction
- Locality
- Wild Edible Plants (WEPs)
- Biodiversity
- Food Systems
- Sustainable development.
- acetylcholine, 2) alpha-linolenic acid, 3) antheraxanthin, 4) ascorbic acid, 5) beta-carotene, 6) betasitosterol, 7) biotin, 8) caffeic acid, 9) calcium, 10) carotenoids, 11) chlorophylls, 12) chloride, 13) choline, 14) citric acid, 15) copper, 16) dietary fibers, 17) fatty acids, 18) flavonoids, 19) folic acid, 20) galactolipids, 21) glutathione, 22) iron, 23) lignins, 24) linoleic acid, 25) lutein, 26) manganese, 27) magnesium, 28) melatonin, 29) molybdenum, 30) neoxanthin, 31) niacin, 32) nickel, 33) nitrates, 34) oleic acid, 35) oxylipins, 36) pantothenate, 37) phenolic acids, 38) phenolic compounds, 39) phenylpropanoids, 40) phospholipids, 41) phosphorus, 42) phylloquinone, 43) phytic acid, 44) phytosterols, 45) plant fatty acids, 46) plant lipids, 47) plant proteins, 48) polyphenols, 49) polysaccharides, 50) potassium, 51) pyridoxine, 52) riboflavin, 53) salicylic acid, 54) selenium, 55) silicon, 56) sodium, 57) sphingolipids, 58) sulfur, 59) terpenoids, 60) thiamin, 61) tocopherols, 62) violaxanthin, 63) xanthophylls, 64) zeaxanthin, and 65) zinc.
| Box 1. Alkaloids. |
| FOR HUMANS: According to Singh et al. (2025), “Numerous studies have also highlighted the health benefits of FOR HUMANS: According to Singh et al. (2025), “Numerous studies have also highlighted the health benefits of these metabolites (alkaloids) for humans.” The authors do not use any examples of WEPs. Heinrich, Mah. Amirkia (2021, 8) gives a WEP example: Yarrow (Achillea millefolium) has an extensive distribution, contains fewer than 10 identified simple alkaloids, and is not a classical source of alkaloid-containing drugs. According to Åhlberg (2020a – 2022a), yarrow (Achillea millefolium) is an acknowledged health-promoting WEP. According to Chauhan. & al. (2010) reported that purple loosestrife (Lythrum salicaria) contains alkaloids with antidiabetic activity.metabolites (alkaloids) for humans.” The authors do not use any examples of WEPs. Heinrich, Mah. Amirkia (2021, 8) gives a WEP example: Yarrow (Achillea millefolium) has an extensive distribution, contains fewer than 10 identified simple alkaloids, and is not a classical source of alkaloid-containing drugs. According to Åhlberg (2020a – 2022a), yarrow (Achillea millefolium) is an acknowledged health-promoting WEP. According to Chauhan. & al. (2010) reported that purple loosestrife (Lythrum salicaria) contains alkaloids with antidiabetic activity. |
| IN PLANTS: According to Singh et al. (2025), alkaloids (1) play crucial roles in plant development, serving as (2) protectants against UV radiation, (2) defensive agents against pathogens, (3) structural components of the cell wall, and (4) pigments that facilitate plant-pollinator interactions. REFERENCES Chauhan, A., et al. 2010. Plants having potential antidiabetic activity: A review. Der Pharmacia Lettre, 2010, 2(3), 369–387. Heinrich, M., Mah, J., & Amirkia, V. (2021). Alkaloids used as medicines: Structural phytochemistry meets biodiversity—An update and forward look. Molecules 26,1836. https://doi.org/10.3390/molecules26071836 Singh, D. & al. 2025. Transcriptional regulation of secondary plant product biosynthesis: insights into flavonoid, alkaloid, and terpenoid pathways. Plant Cell, Tissue and Organ Culture (PCTOC) 160:6; https://doi.org/10.1007/s11240-024-02925-z |
| Box 2. Resveratrol. |
| According to Alesci & al. (2022), Wu & al. (2022), Zhu & al. (2022), Alauddin & al. (2021), Grinan-Ferre & al. (2021), Xiong & al. (2021), Kumar & al. (2020), Matsuno & al. (2020), Martínez & al. (2019) and Singh, A. & al (2019a), resveratrol has following health-promoting properties: 1) antioxidant, 2) anti-inflammatory, 3) anticancer, 4) antiviral, 5) antidiabetic, 6) anti-obesity, 7) antimetabolic syndrome, 8) cardiovascular protective, 9) antiplatelet, 10) anti-hypertension, 11) antiaging, 12) protects against neurodegenerative diseases, such as Alzheimer’s disease, 13) antistroke, 14) nephroprotective, 15) hepatoprotective, 16) delays the progression of osteoarthritis, and 17) maintains genome stability, promoting a longer and healthier life. According to Zhua et al. (2019), resveratrol has protective effects on stress-induced depression and anxiety. They present a molecular biological mechanism for it. According to Grinan-Ferre et al. (2021), resveratrol is a powerful antioxidant and “possesses pleiotropic actions, exerting its activity through various molecular pathways.” Kumar et al. (2020) state that resveratrol can cross the blood-brain barrier. Neuroinflammation is a part of Alzheimer’s disease. Resveratrol prevents neuroinflammation. |
| REFERENCES Alauddin, M., et al. (2021). Potential of nutraceuticals in preventing the risk of cancer and metabolic syndrome from the perspective of nutritional genomics. Cancer Plus, 3(2), 1–18. Alesci, A., et al. (2022). Resveratrol and immune cells: A link to improve human health. Molecules, 27(2), 24. https://doi.org/10.3390/molecules27020424 Grinan-Ferré, A., et al. (2021). The pleiotropic neuroprotective effects of resveratrol in cognitive decline and Alzheimer’s disease pathology: From antioxidant to epigenetic therapy. Aging Research Reviews, 67, Article 101271, 1–24. Kumar, S., et al. (2020). Resveratrol, a molecule with anti-inflammatory and anticancer activities: Natural product to chemical synthesis. Current Medicinal Chemistry, 27, 1–14. Matsuno, Y., et al. (2020). Resveratrol and related polyphenols help maintain genome stability. Scientific Reports, 10, Article 5388, 1–10. Wu, S., et al. (2022). Effects and mechanisms of resveratrol in the prevention and management of cancer: An updated review. Critical Reviews in Food Science and Nutrition. https://doi.org/10.1080/10408398.2022.2101428 Xiong, G., et al. (2021). Effect of resveratrol on abnormal bone remodelling and angiogenesis of subchondral bone in osteoarthritis. International Journal of Clinical and Experimental Pathology, 14(4), 417–425. Zhu, H., et al. (2022). Resveratrol protects against chronic alcohol-induced liver disease in a rat model. STEMedicine, 3(3), e133. https://doi.org/10.37175/stemedicine.v3i3.133 |
| Box 3. Resveratrol, polydatin, and piceid |
| According to Sirohi & al. (2021), Tang (2021), Wu & al. (2020b), Chen & al. (2015) and Du & al. (2013, 1347), polydatin is 1) anti-inflammatory, 2) antioxidant, 3) anti-cancer, 4) neuroprotective, 5) hepatoprotective, 6) nephroprotective, 7) immune regulating, immunostimulatory, 8) anti-platelet aggregation, 9) cardioprotective, 10) regulates lipid metabolism, 11) anti-atherosclerosis, 12) protects against Alzheimer’s disease. According to Wang et al. (2015), polydatin and resveratrol can interconvert; polydatin is a more potent antioxidant than resveratrol. According to Wu et al. (2020b), polydatin exerts its anti-atherosclerotic effects through three mechanisms: anti-inflammatory effects, regulation of lipid metabolism, and antioxidant activity. Wu et al. (2020b) present the pharmacological mechanism of polydatin in the treatment of atherosclerosis. According to PubChem (2021), polydatin and piceid are synonyms. According to Chen et al. (2015), polydatin (piceid) and resveratrol may interconvert in living organisms. Basholli-Salihu et al. (2016) describe in detail how this happens. |
| REFERENCES Cheng, C. K., Luo, J.-Y., Lau, C. W., Chen, Z.-Y., Tian, X. Y., & Huang, Y. (2019). Pharmacological basis and new insights of resveratrol action in the cardiovascular system. British Journal of Pharmacology. Advance online publication. https://doi.org/10.1111/bph.14801 PubChem. 2026. Resveratrol. https://pubchem.ncbi.nlm.nih.gov/compound/Resveratrol Sirohi, P., et al. (2021). The polyphenolic phytoalexin polydatin inhibits amyloid aggregation of recombinant human prion protein. RSC Advances, 11, 25901–25911. Tang, K. (2021). Protective effects of polydatin against dementia-related disorders. Current Neuropharmacology, 19(2), 127–135 Zhou, Y., et al. (2013). Synergistic anticancer effects of curcumin and resveratrol in Hepa1-6 hepatocellular carcinoma cells. Oncology Reports, 29(5), 1851–1858. https://doi.org/10.3892/or.2013.2310 (Note: Oncology Reports volume/issue inferred from page range; adjust if needed.) |
| Box 4. Ellagitannins. |
| According to Chen & al. (2022), García-Villalba & al. (2022), Gopalsamy & al. (2022), Al-Harbi & al. (2021), D’Amico & al. (2021), Hoseinynejad & al. (2021), Miloševic & al. (2021), Yüksel & al. (2021), Dreger & al. (2020), Li & al. (2020), Luca (2019, 17), Yoshida & al. (2018), Muthukumaran & al. (2017, 240 - 241), and Sangiovanni & al. (2013), ellagitannins have the following health-promoting properties: 1) antioxidant, 2) anti-inflammatory, 3) antimicrobial, 4) antiglycative, 4) hepato-protective, 5) beneficial effects on kidney diseases, 4) anti-virus, 5) cardioprotective, 6) neuroprotective, 7) prebiotic, 8) chronic disease prevention, 7) anticancer, 8) antidiabetic, 9) beneficial effects on chronic tissue inflammation, 10) beneficial effects on metabolic syndrome) 11) beneficial effects on obesity-mediated metabolic complications, 12) beneficial effects on gastrointestinal diseases, 13) beneficial effects on eye diseases, 14) beneficial effects on depression, 15) muscle mass protective effects, and 16) beneficial effects on Alzheimer’s disease and other neurodegenerative diseases. Schink et al. (2018) describe how ellagitannins prevent inflammation using molecular biology. |
| REFERENCES Al-Harbi, S.& al. 2021.Urolithins: The gut-based polyphenol metabolites of ellagitannins in cancer prevention, a review. Frontiers in Nutrition, Volume 8, article 647582, 1 – 15. Chen P. et al. (2022). Recent advances and perspectives on the health benefits of urolithin B, a bioactive natural product derived from ellagitannins. Frontiers in Pharmacology 13:917266 D’Amico, D., et al. (2021). Impact of the Natural Compound Urolithin A on Health, Disease, and Aging. Trends in Molecular Medicine 27(7), 687 – 699. Dreger, M. & al. 2020. Pharmacological properties of fireweed (Epilobium angustifolium L.) and bioavailability of ellagitannins. A review. Herba Polonica 66(1), 52 – 64. García-Villalba, R. et al. (2022). Ellagitannins, urolithins, and neuroprotection: Human evidence and the possible link to the gut microbiota. Molecular Aspects of Medicine. Available online 5 August 2022, 101109. In Press, Corrected Proof. https://doi.org/10.1016/j.mam.2022.101109 Gopalsamy, R., et al. (2022). Health functions and related molecular mechanisms of ellagitannin-derived urolithins, Critical Reviews in Food Science and Nutrition. https://doi.org/10.1080/10408398.2022.2106179 Li, Q. & al. 2020. Anti-renal fibrosis and anti-inflammation effect of urolithin B, ellagitannin-gut microbial-derived metabolites in unilateral ureteral obstruction rats. Journal of Functional Foods, Volume 65, article 103748, 1 – 13. Milošević M. et al. (2021). Memorable food: fighting age-related neurodegeneration by precision nutrition. Frontiers in Nutrition, Volume 8, article 688086, 1 – 13. Sangiovanni, E. et al. 2013. Ellagitannins from Rubus berries for the control of gastric inflammation: in vitro and in vivo studies. PLoS ONE, Volume 8(8), article e71762, 1 – 12. Yüksel, A. & al. 2021. Phytochemical, phenolic profile, antioxidant, anticholinergic, and antibacterial properties of Epilobium angustifolium (Onagraceae). Journal of Food Measurement and Characterization. Published online 12. 7. 2021, 1 – 10. Yoshida, T et al. 2018. The chemical and biological significance of oenothein B and related ellagitannin oligomers with macrocyclic structure. Molecules, Volume 23, article 552, 1–21. |
| Box 4. Tocopherols. |
| FOR HUMANS: According to Tkacz & al. (2021) The National Institutes of Health (2021) Arroz, & al. (2020) Azzi (2018) Morris, M. & al. (2015) and Kamal-Eldin & Appelqvist (1996) tocopherols (vitamin E) have the following health-promoting properties: 1) antioxidants, 2) prevent cardiovascular disease, 3) prevent neurodegenerative disease, 4) prevent macular degeneration, 5) prevent cancer, 6) anti-inflammatory, 7) immune-boosting compound, 8) protect against non-alcoholic hepato-steatosis, 9) regulate cell-signaling, 10) modulate gene transcription, and 11) the combination of dietary tocopherols, rather than individual tocopherols, has the strongest protective relation to the development of Alzheimer’s disease. |
| IN PLANTS, according to Munné-Bosch & Alegre (2002), tocopherols and tocotrienols are lipid-soluble molecules that perform several functions. Tocopherols and tocotrienols are essential to maintain plant cell membrane integrity. Alpha-tocopherol is the primary form found in plant green parts, while tocotrienols are found in seeds. All wild edible plants contain tocopherols. These compounds are antioxidants; thus, they protect the plant from oxygen toxicity. According to Ma et al. (2020), in green plants, tocopherols are vital to the plant’s stress response. |
| REFERENCES Arroz, A. et al. (2020). Comparative effects of alpha- and gamma-tocopherol on mitochondrial functions in Alzheimer’s disease in vitro model. Scientific Reports, volume 10, article 8962, 1 – 14. Kamal-Eldin, A. & Appelqvist, L. (1996). The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 31. Ma, J. & al. 2020. Diverse roles of tocopherols in response to abiotic and biotic stresses and strategies for genetic biofortification in plants. Molecular Breeding, volume 40(18) 1 – 15. Morris, M. et al. (2015). Brain tocopherols related to Alzheimer’s disease neuropathology in humans. Munné-Bosch, S. & Alegre, L. 2002. The function of tocopherols and tocotrienols in plants. Critical Reviews in Plant Sciences 21(1) 31 – 57 The National Institutes of Health. 2020. Tocopherols-Fact Sheet for Consumers. https://ods.od.nih.gov/ factsheets/VitaminE-HealthProfessional/ Tkacz, K. & al. 2021. Phytoprostanes, phytofurans, tocopherols, tocotrienols, carotenoids, free amino acids, and the biological potential of sea buckthorn juice. Journal of the Science of Food and Agriculture, First published online: 01 June 202, 1 – 13. https://doi.org/10.1002/jsfa.11345 |
| Box 4. Nitrates. |
| FOR HUMANS: According to Butler (2015), Bryan & Ivy (2015), Sindelar & Milkowski (2012), Bahra et al. (2012), and Bryan et al. (2012), plant-derived nitrates and nitrites are harmless to healthy adults and may even be indispensable nutrients. According to the reviews of Raubenheimer & al. (2019) and Lundberg & al (2018, 9) nitrates and nitrites that come from plant food have the following health-promoting properties: 1) lower blood pressure, 2) improve endothelial function, 3) increase exercise performance, 4) reverse of metabolic syndrome, 5) antidiabetic, 6) counteract inflammation and 7) promote homeostasis of the immune and vascular systems. Karwowska & Kononiuk (2020) present a balanced overview of the benefits and potential risks of nitrates in the human body. The threats mainly stem from the use of nitrates and nitrites in meat products. Avoiding processed meat products and using Mediterranean boiled mixtures of wild edible plants can optimize the benefits of nitrates and minimize risks. |
| IN PLANTS: According to Hou & al. (2020) Wang &al. (2019) Noguero & Lacombe (2016) Stitt & al. (2002) and Stitt (1999): 1) plant roots assimilate nitrate, nitrogen (N) from the soil; 2) nitrate is translocated to leaves, where nitrates take part in amino acid and protein synthesis; 3) nitrate (NO3-is an essential source of nitrogen for plant development and metabolism; 4) Nitrogen takes part in leaf photosynthesis. 5) Nitrogen takes part in the synthesis of numerous cellular components, such as a) chlorophyll and b) nucleic acids. 6) Nitrates take part in pH regulation. 7) Nitrates take part in the sugar supply of the leaves. 6) Nitrate is also a signal molecule in controlling many physiological processes, plant growth, and crop yield. 8) Nitrate participates in the regulation of lateral root development and architecture. 9) Nitrate participates in the regulation of leaf development. 9) Nitrate participates in the flowering induction, and 10) Nitrate participates in seed dormancy regulation. 10) Nitrate is a signal molecule that can trigger widespread changes in gene expression, resulting in changes in nitrogen and carbon metabolism to facilitate the uptake and assimilation of nitrate; 11) Nitrogen contributes to plant defence responses by the regulation of primary metabolites of plants, such as carbohydrates, amino acids, and lipids; 12) Nitrogen affects the mechanical strength of cell walls by changing the amount of plant cellulose and lignin to prevent pathogen penetration and limit disease development. |
| REFERENCES Hou, W., et al. (2020). Diagnosis of nitrogen nutrition in rice leaves influenced by potassium levels. Frontiers in Plant Science, volume 11, article 165, 1 – 13. Noguero, M., & Lacombe, B. (2016). Transporters involved in root nitrate uptake and sensing by Arabidopsis. Frontiers in Plant Science, 7, 1391. https://doi.org/10.3389/fpls.2016.01391 Stitt, M. (1999). Nitrate regulation of metabolism and growth. Current Opinion in Plant Biology 2(3) 178 – 186. Stitt, M., et al. (2002). Steps towards an integrated view of nitrogen metabolism. Journal of Experimental Botany 53(370) 959 – 970. Wang, M., et al. (2019). Plant primary metabolism regulated by nitrogen contributes to plant–pathogen interactions. Plant and Cell Physiology 60(2) 329–342. |
- What wild edible plants (WEPs) grow (a) in Helsinki, (b) in other parts of Finland?
- What wild edible plants grow (a) in Puerto de la Cruz and (b) in other parts of Tenerife?
- How do you recognize a WEP?
- What parts of these WEPs are used, and how to cook them?
- How to guarantee sustainable use of these WEPs?
2. Materials and Methods
- Åhlberg, M. K. (2019). Totuus syötävistä luonnonkasveista eli miksi uskallan syödä lähiluonnon kasveista kestävästi keräämääni ruokaa: Osa I: Tieteellisiä perusteita käytännönläheisesti [The truth about wild edible plants – why I am not afraid of eating food that I have made from plants in the local nature that I have foraged sustainably]. Eepinen Oy.
- Åhlberg, M. K. (2020a). Local Wild Edible Plants (WEP). Practical conclusions from the latest research: Healthy food from local nature. Helsinki: Oy Wild Edibles Ab. International distribution: Amazon.com.
- Åhlberg, M. K. (2020b). Field guide to local Wild Edible Plants (WEP): practical conclusions from the latest research: healthy food from local nature. Helsinki: Oy Wild Edibles Ab. International distribution: Amazon.com.
- Åhlberg, M. K. (2022a). Terveyttä lähiluonnosta [Health from local nature]. Helsinki: Readme. fi. (The book presents 75 common WEPs. Their health-promoting substances are in English.)
- Åhlberg, M. K. (2021). A profound explanation of why eating green (wild) edible plants promotes health and longevity. Food Frontiers, 2, 240–267.
- Åhlberg, M. K. (2022b). An update of Åhlberg (2021a): A profound explanation of why eating green (wild) edible plants promotes health and longevity. Food Frontiers, 3, 366–379.
- Åhlberg, M. K. (2025a). Wild edible plants: Ensuring sustainable food security in an era of climate change. Foods, 14(9), 1611. https://doi.org/10.3390/foods14091611
| IA WEP species | Suggested use |
| Acer platanoides | Flowers are tasty. Good for salads. |
| Achillea millefolium | This plant grows from the south of Finland to the shores of the North Atlantic ocean. |
| Aegopodium podagraria | Young leaves and shoots are tasty, especially in salads. |
| Alchemilla vulgaris | |
| Alliaria petiolata | It has a garlic scent when leaves are crushed. |
| Allium schoenoprasum | Leaves and flowers are good for salads. |
| Allium ursinum | |
| Angelica archangelica | Leaves are good for salads. |
| Angelica sylvestris | |
| Anthriscus sylvestris | |
| Arctous alpina | Berries are edible. |
| Argentina anserina, earlier Potentilla anserina | |
| Armoracia rusticana | |
| Artemisia vulgaris | |
| Barbarea vulgaris | Young shoots and flowers are tasty. Good fos salads. |
| Bellis perennis | |
| Betula pendula | |
| Betula pubescens | |
| Bidens pilosa | |
| Borago officinalis | |
| Cakile maritima | Young shoots and flowers are tasty. Good fos salads. |
| Calendula arvensis | |
| Calluna vulgaris | Flowers can be used for tea. |
| Capsella bursa-pastoris | |
| Cardamine pratensis | |
| Carum carvi | |
| Chenopodium album | |
| Cirsium arvense | |
| Cirsium heterophyllum | |
| Corylus avellana | |
| Empetrum nigrum | Berries are edible. |
| Epilobium angustifolium | Young shoots are tasty. |
| Epilobium hirsutum | |
| Equisetum arvense | A good tea can be cooked from the spring shoots. |
| Fallopia japonica | Has resveratrol. |
| Filipendula ulmaria | Dried flowers taste good in vanilla ice cream. |
| Fragaria vesca | Berries and young leaves are edible. |
| Geranium sylvaticum | Flowers are healthy. |
| Hippophaë rhamnoides | Berries and leaves are edible and healthy. |
| Humulus lupulus | Young shoots are tasty. Leaves and female flowers are edible. |
| Hylotelephium telephium | |
| Impatiens glandulifera | Seeds are tasty. Leaves and flowers can be used for health-promoting decoction. |
| Juniperus communis | Berries are tasty. |
| Lactuca alpina | Good for salads. |
| Lactuca muralis | |
| Lactuca serriola | |
| Lamium album | |
| Lamium purpureum | |
| Levisticum officinale | Good for salads. |
| Linaria vulgaris | Flowers are edible. |
| Lyhtrum salicaria | Flowers are tasty and healthy. |
| Malus species | Young leaves, flowers and fruits edible. |
| Matricaria discoidea | Good scent. Tasty. Good for salads. |
| Matteuccia struthiopteris | Fiddleheads, tightly coiled young fronds are tasty after steaming. |
| Mentha arvensis | Good for salads. |
| Myrrhis odorata | Good scent. Good for salads. |
| Origanum vulgare | |
| Oxalis acetosella | Good for salads. |
| Picea abies | The annual shoots are healthy and tasty. |
| Pimpinella saxifraga | |
| Pinus sylvestris | Female flowers are tasty. |
| Plantago lanceolata | |
| Plantago major | |
| Polygonum aviculare | |
| Prunella vulgaris | |
| Prunus padus | Berry pulp is edible. Seeds are toxic. |
| Ribes nigrum | Both berries and leaves are tasty and promote health. |
| Ribes rubrum | Berries are edible. Leaves are not edible. |
| Rosa rugosa | Petal and hips are tasty and promote health. |
| Rubus arcticus | Berries and leaves edible. |
| Rubus chamaemorus | Berries and leaves edible. |
| Rubus idaeus | Young shoots can be used in boiled mixtures of WEPs. |
| Rubus saxatilis | Berries are tasteless. Leaves can be used in boiled mixtures of WEPs. |
| Rumes acetosa | Good for salads. |
| Rumex acetosella | Good for salads. |
| Rumex obtusifolius | |
| Silene vulgaris | |
| Sonchus arvensis | |
| Sonchus asper | |
| Sonchus oleraceus | |
| Sorbus aucuparia | Buds, leaves, flowers and berries are edible. |
| Stellaria media | Good for salads. |
| Syringa vulgaris | Blossoms are edible. |
| Tanacetum vulgare | |
| Taraxacum officinale | Flowers and leaves are edible. Young leaves are good for salads. |
| Trifolium pratense | Flowers and leaves are edible. |
| Trifolium repens | Flowers and leaves are edible. |
| Urtica dioica | Leaves dry easily in shade. Crushed leaves make good green powder into all food. |
| Vaccinium myrtillus | Both berries and leaves are healthy. |
| Vaccinium oxycoccus | Berries are edible. |
| Vaccinium uloginosum | Berries are edible. |
| Vaccinium vitis-idea | |
| Veronica arvensis | |
| Veronica beccabunga | |
| Veronica chamaedrys | |
| Veronica longifolia | |
| Veronica officinalis | |
| Veronica persica | |
| Viburnum opulus | Ripe berries are edible after frost. |
| Viola arvensis | |
| Viola tricolor | Flowers are beautiful and tasty. |
| Acyranthes aspera | |
| Amaranthus viridis | |
| Antirrhinum majus | Flowers are tasty. |
| Arbutus canariensis | Fruits are tasty. |
| Asparagus acutifolius | |
| Beta macrocarpa | |
| Bidens pilosa | Flowers are tasty. |
| Brassica oleracea | Young shoots are tasty. |
| Calendula arvensis | |
| Capsella bursa-pastoris | |
| Carpobrotus edulis | |
| Chenopodium album | |
| Chenopodium murale | |
| Crithmum maritimum | |
| Emex spinosa | |
| Eriobotrya japonica | Fruits are tasty. |
| Erodium cicutarium | |
| Eruca vesicaria | |
| Ficus carica | Fruits are tasty. |
| Foeniculum vulgare | Leaves have good scent and they are tasty. |
| Fumaria officinalis | |
| Galinsoga quadriradiata | |
| Galium apare | |
| Malva neglecta | |
| Malva parviflora | |
| Malva sylvestris | |
| Mesembryanthemum crystallinum | |
| Opuntia ficus-indica | Fruits are tasty, but full of small spikes. |
| Oxalis pes-caprae | |
| Papaver rhoeas | |
| Plantago lanceolata | |
| Plantago major | |
| Portulaca oleracea | Leaves are good for salad. |
| Rubus ulmifolius | Berries are tasty. |
| Rumex crispus | |
| Rumex lunaria | |
| Rumex obtusifolius | |
| Sechium edule | Fruit is tasty when fried with salt and black pepper. |
| Silene vulgaris | |
| Sonchus asper | |
| Sonchus oleraceus | |
| Stellaria media | |
| Tagetes species | |
| Taraxacum officinale | |
| Tetragonia tetragonioides | Leaves are good for salad. |
| Trifolium pratense | |
| Tropaeolum majus | Flowers and leaves are tasty. |
| Tropaeolum majus | |
| Urtica urens | |
| Veronica persica |
| Bauhinia variegata ja Bauhinia purpurea. | Flowers are edible. |
| Carissa macrocarpa | Fruits are tasty. |
| Coccoloba uvifera | Fruits are edible. |
| Dovyalis caffra | Fruits are tasty. |
4. Discussion
Discussion and Conclusions
Funding
Data Availability Statement
Acknowledgments
Abbreviation
| GBIF | Global Biodiversity Information Facility |
References
- Åhlberg, M.K. The Number of Health-Promoting Substances That All Edible Vascular Plants Contain Compared to the Total Number of Health-Promoting Substances of Five Wild Edible Plants (WEPs). 2025. Submitted manuscript. Available online: https://www.preprints.org/manuscript/202410.1307.
- Åhlberg, M.K. Wild Edible Plants: Ensuring Sustainable Food Security in an Era of Climate Change. Foods 2025, 14, 1611. [Google Scholar] [CrossRef] [PubMed]
- knowledge from the Published Research Articles on the Internet. Int. J. Qual. Methods 2026. accepted.
- Pereira, A.G.; Fraga-Corral, M.; García-Oliveira, P.; Jiménez-López, C.; Lourenço-Lopes, C.; Carpena, M.; Otero, P.; Gullón, P.; Prieto, M.A.; Simal-Gandara, J. Culinary and Nutritional Value of Edible Wild Plants from Northern Spain, Rich in Phenolic Compounds with Potential Health Benefits. Food Funct. 2020, 11, 8493–8515. [Google Scholar] [CrossRef] [PubMed]
- Åhlberg, M.K. The Number of Health-Promoting Substances That All Edible Vascular Plants Contain Compared to the Total Number of Health-Promoting Substances of Five Wild Edible Plants (WEPs). 2025. Submitted manuscript. Available online: https://www.preprints.org/manuscript/202410.1307.
- Britannica Editors. Neolithic. 2026. Available online: https://www.britannica.com/event/Neolithic.
- Blakemore, E. What Was the Neolithic Revolution? Also Called the Agricultural Revolution, the Shift to Agriculture from Hunting and Gathering Changed Humanity Forever. Natl. Geogr. Mag. 2023. Available online: https://www.nationalgeographic.com/culture/article/neolithic-agricultural-revolution.
- Sadowski, R.F. Neolithic Revolution. In Encyclopedia of Food and Agricultural Ethics; Thompson, P., Kaplan, D., Eds.; Springer: Dordrecht, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Crozier, A.; Clifford, M.N.; Ashihara, H. (Eds.) Plant Secondary Metabolites: Occurrence, Structure and Role in the Human Diet; Blackwell Publishing: Oxford, UK, 2006. [Google Scholar] [CrossRef]
- Vanhakylä, S. Distribution of Foliar Plant Polyphenols across the Plant Phylogeny. Ph.D. Thesis, University of Turku, Department of Chemistry, Turku, Finland, 2025. [Google Scholar]
- Popper, K.R. Objective Knowledge: An Evolutionary Approach; Oxford University Press: Oxford, UK, 1972. [Google Scholar]
- Åhlberg, M.K. A Profound Explanation of Why Eating Green (Wild) Edible Plants Promotes Health and Longevity. Food Front. 2021, 2, 240–267. [Google Scholar] [CrossRef]
- Åhlberg, M.K. An Update of Åhlberg (2021a): A Profound Explanation of Why Eating Green (Wild) Edible Plants Promotes Health and Longevity. Food Front. 2022, 3, 366–379. [Google Scholar] [CrossRef]
- Åhlberg, M.K. Kasvatustavoitteiden Teoreettinen Kehikko ja Sen Empiiristä Koettelua [A Theoretical Framework for Educational Objectives and Its Empirical Testing]. Ph.D. Thesis, Helsingin Yliopisto, Kasvatustieteen Laitos, Tutkimuksia Nro 117, Helsinki, Finland, 1988. [Google Scholar]
- Åhlberg, M.K. Concept Mapping, Concept Matrices, Link Tables, and Argumentation Analysis Are Techniques for Educational Research on Textbooks and Educational Discourse, and as Tools for Teachers and Their Pupils in Their Everyday Work. In Research on Texts at School; Julkunen, M.-L., Selander, S., Author, M., Eds.; Research Reports of the Faculty of Education No. 37; University of Joensuu: Joensuu, Finland, 1991; pp. 89–154. [Google Scholar]
- Pohjanmies, T.; Jaskova, A.; Hotanen, J.-P.; Manninen, O.; Salemaa, M.; Tolvanen, A.; Merilä, P. Abundance and Diversity of Edible Wild Plants in Managed Boreal Forests. For. Ecol. Manag. 2021, 491, 119151. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




