Submitted:
14 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Small-Scale Hydrogen Embrittlement
2.2. Contained Hydrogen Embrittlement
2.3. Large-Scale Hydrogen Embrittlement
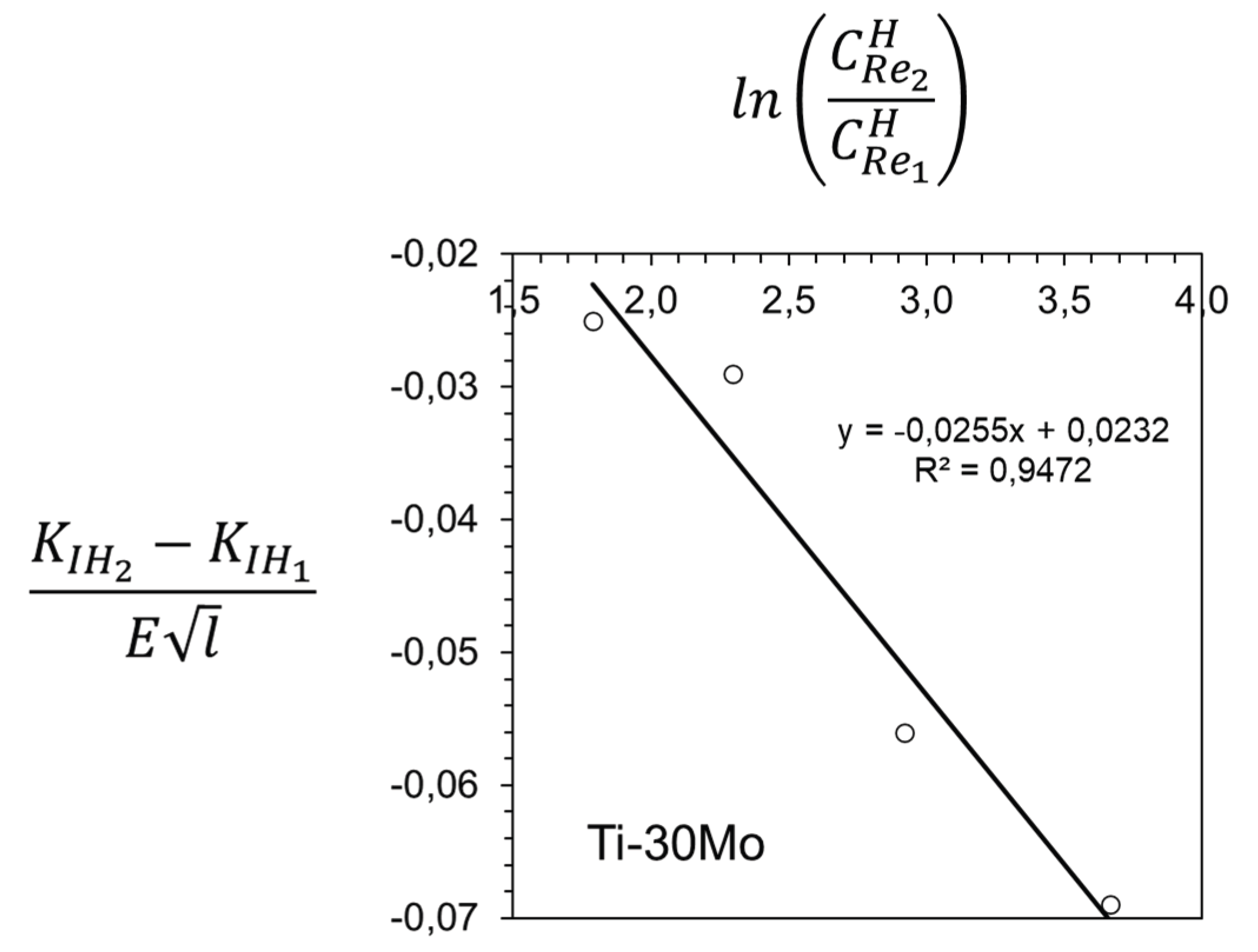
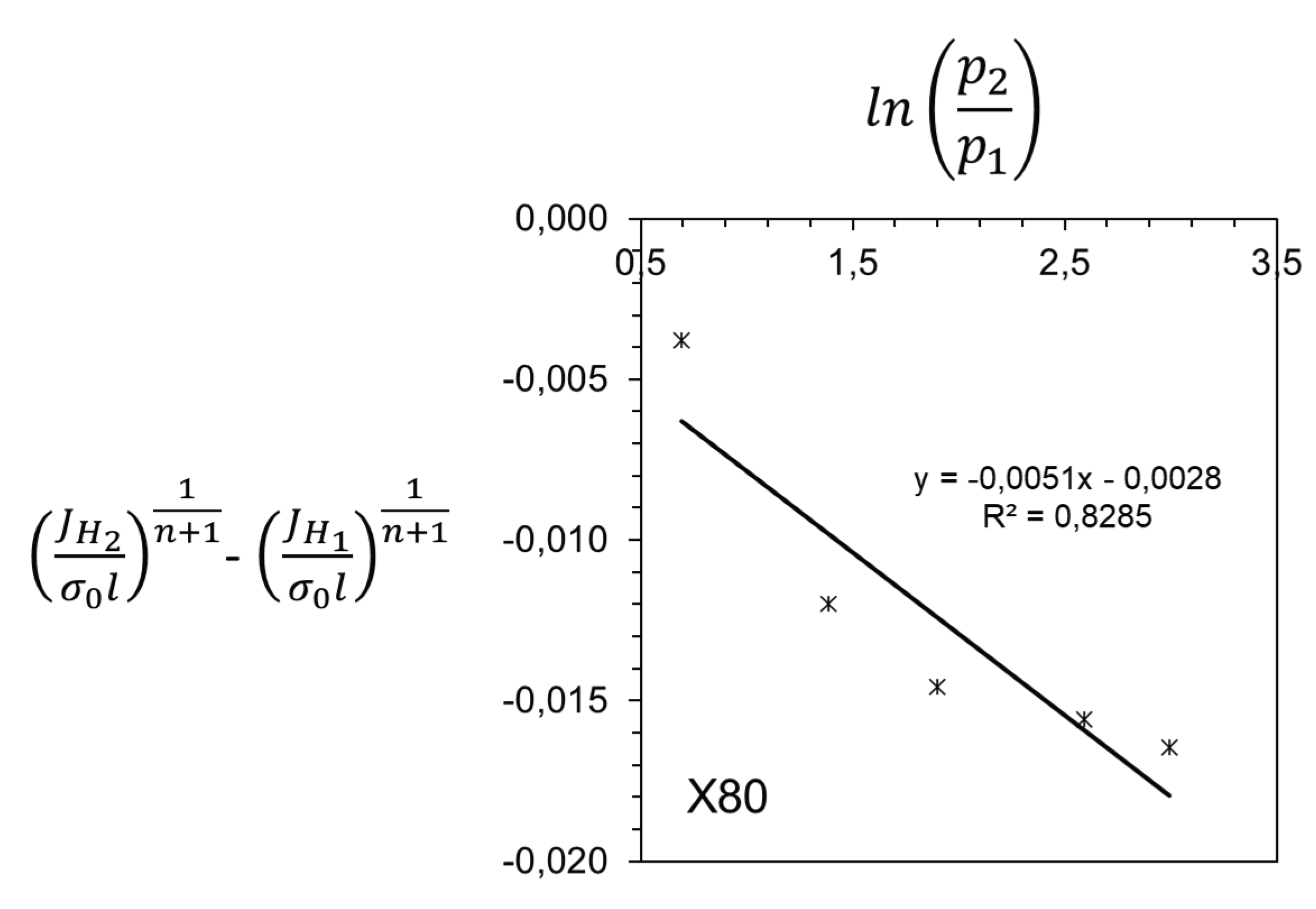
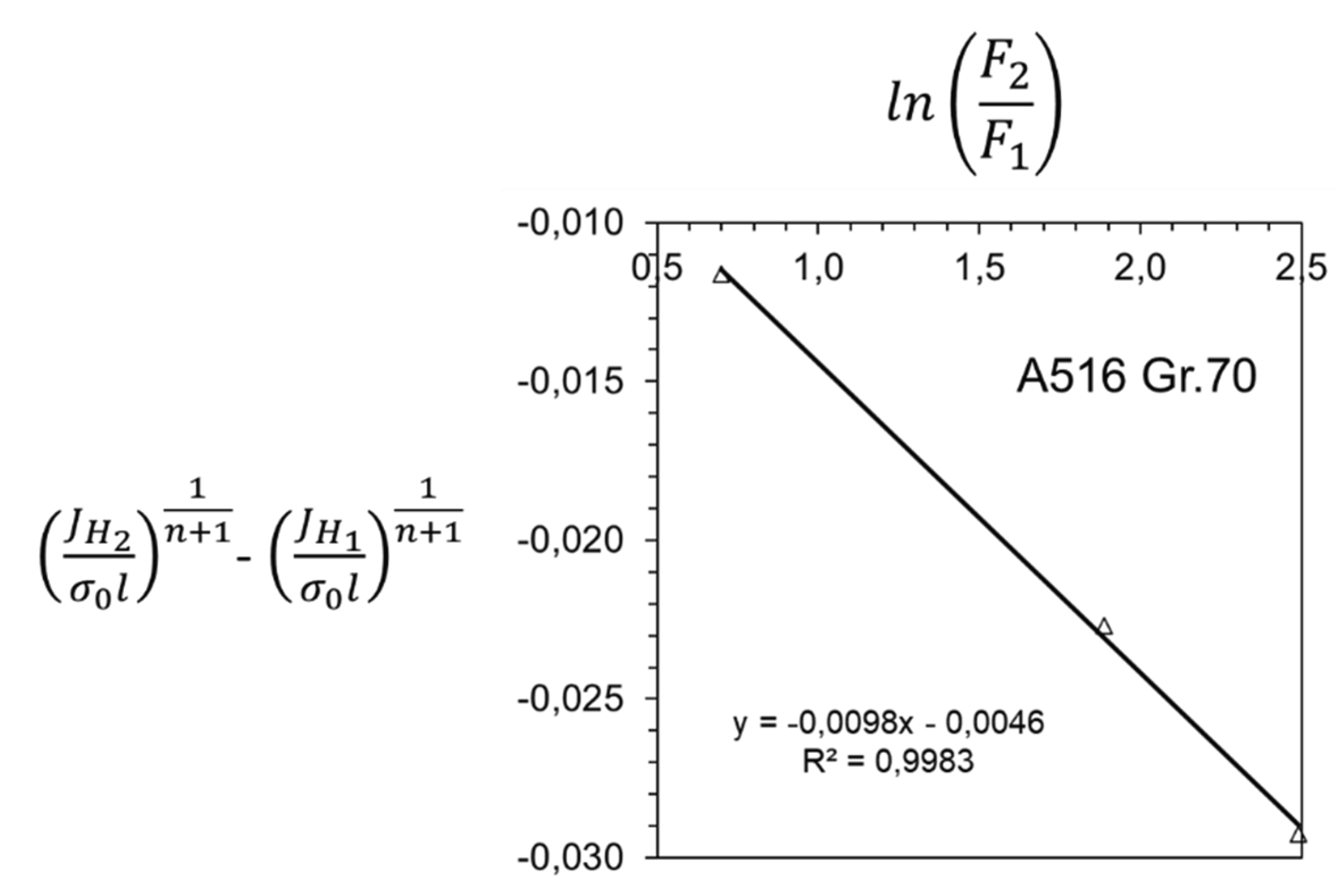
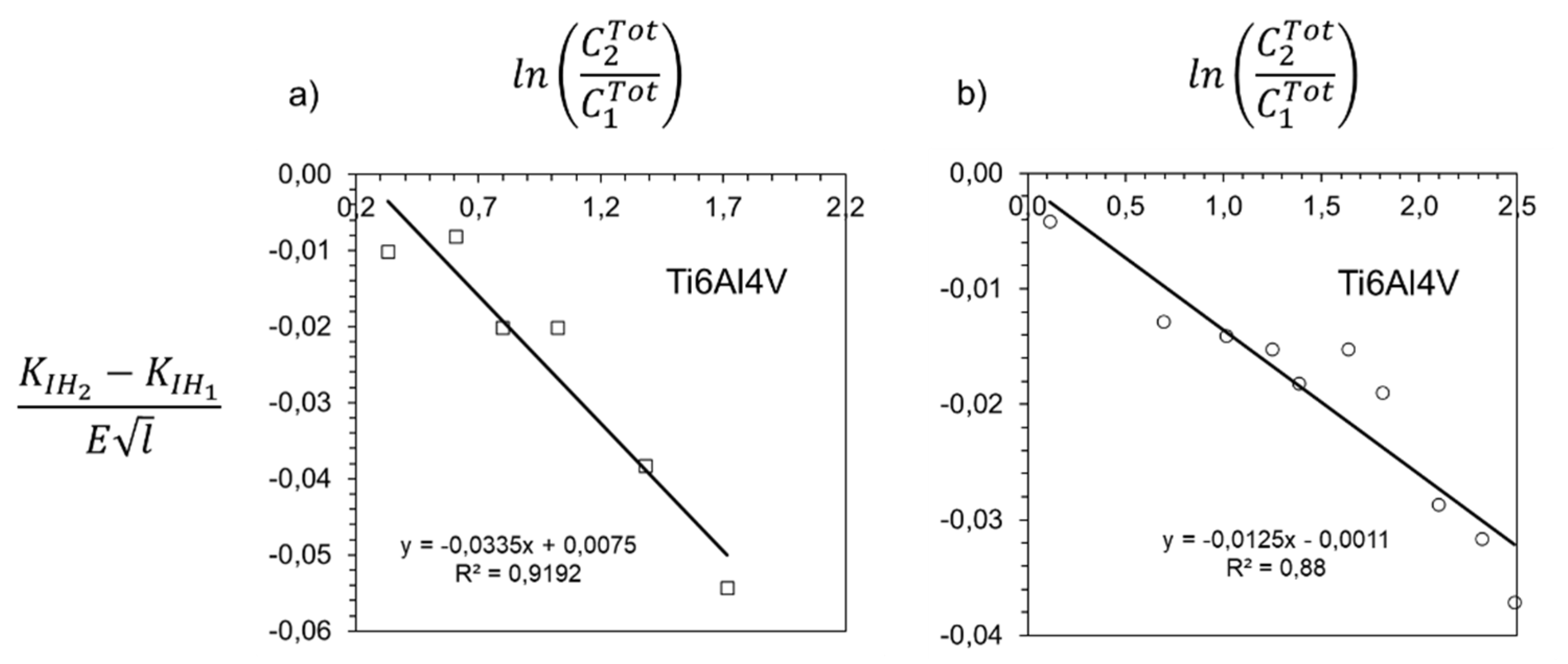
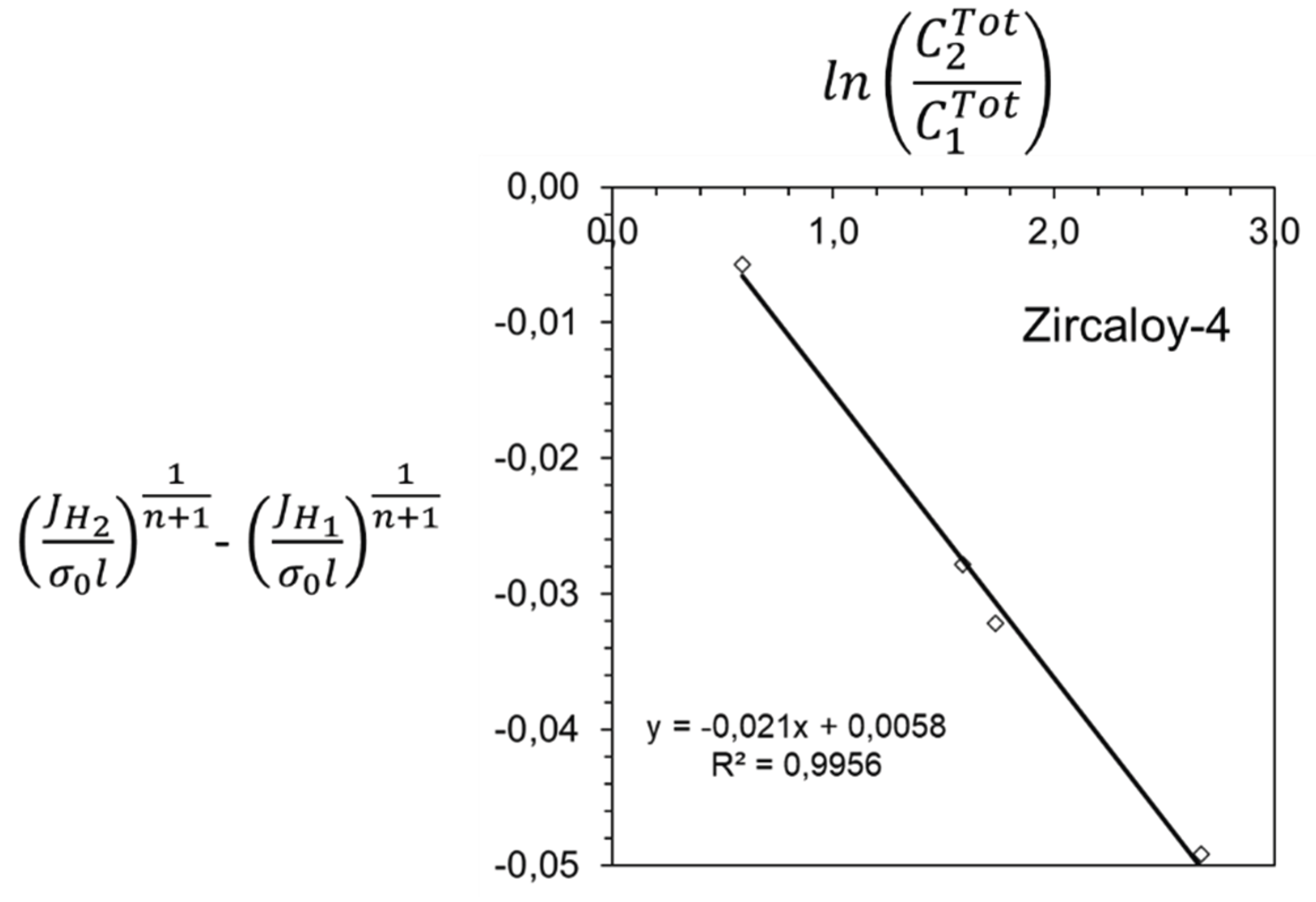
3. Results and Discussion
4. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Global hydrogen review 2025; International Energy Agency.
- Nuttal, WJ; Powel, JB; Anaya-Stucchi, KL; Bakenne, AT; Wilson, A. Insights into the new hydrogen economy; Springer Nature Switzerland: Cham, 2025. [Google Scholar]
- Quitzow, R; Zabanova, Y. The geopolitics of hydrogen. In European strategies in global perspective; Springer Nature Switzerland: Cham, 2024; Volume 1. [Google Scholar]
- Brewer, T. Climate change. An interdisciplinary introduction; Springer Nature Switzerland: Cham, 2023. [Google Scholar]
- Weichenhain, U. Hydrogen transportation. The key to unlocking the clean hydrogen economy; Roland Berger: Munich, 2021. [Google Scholar]
- Broberg, KB. Cracks and fracture; Academic Press: London, 1999. [Google Scholar]
- Kanninen, MF; Popelar, CH. Advanced fracture mechanics; Oxford University Press: Oxford, 1985. [Google Scholar]
- Hutchinson, JW. A course on nonlinear fracture mechanics; The Technical University of Denmark: Lyngby, 1979. [Google Scholar]
- Chen, Y-S; Huang, C; Liu, P-Y; Yen, H-W; Niu, R; Burr, P; Moore, KL; Martínez-Pañeda, E; Atrens, A; Cairney, JM. Hydrogen trapping and embrittlement in metals – A review. International Journal of Hydrogen Energy 2025, 136, 789–821. [Google Scholar] [CrossRef]
- Barrera, O; Bombac, D; Chen, Y; Daff, TD; Galindo-Nava, E; Gong, P; Haley, D; Horton, R; Katzarov, I; Kermode, JR; Liverani, C; Stopher, M; Sweeney, F. Understanding and mitigating hydrogen embrittlement of steels: a review of experimental, modelling and design progress from atomistic to continuum. Journal of Material Science Correction in Journal of Material Science. 53, 10593–10594. 2018, 53, 6251–6290. [Google Scholar] [CrossRef] [PubMed]
- Varias, AG. Elastic crack-tip field in hydride forming metals under hydrogen chemical equilibrium. International Journal of Fracture 2024, 245, 183–194. [Google Scholar] [CrossRef]
- Varias, AG. Elastic-plastic crack-tip field in hydride forming metals under hydrogen chemical equilibrium. International Journal of Fracture 2024, 246, 47–57. [Google Scholar] [CrossRef]
- Varias, AG. Hydride induced embrittlement and fracture of non-hardening metals under hydrogen chemical equilibrium. International Journal of Solids and Structures 2024, 305, 113073. [Google Scholar] [CrossRef]
- Behvar, A; Haghshenas, M; Djukic, MB. Hydrogen embrittlement and hydrogen-induced crack initiation in additively manufactured metals: A critical review on mechanical and cyclic loading. International Journal of Hydrogen Energy 2024, 58, 1214–1239. [Google Scholar] [CrossRef]
- Varias, AG. Chemical Equilibrium Fracture Mechanics – Hydrogen embrittlement application. Corrosion and Materials Degradation 2025, 6, 5. [Google Scholar] [CrossRef]
- Li, X; Zhong, Y; Li, H; Liu, Y; Shan, G; Qu, D; Zhang, J; Djukic, MB. Review of hydrogen embrittlement effect on fracture toughness of metallic materials: Influencing factors, and predictive models. Engineering Fracture Mechanics 2025, 327, 111392. [Google Scholar] [CrossRef]
- Coleman, CE. The metallurgy of zirconium; International Atomic Energy Agency: Vienna, 2022; Vol. 3. [Google Scholar]
- Varias, AG. Chemical Equilibrium Fracture Mechanics – Hydrogen embrittlement of two-phase hydride forming alloys. International Journal of Solids and Structures 2025, 323, 113635. [Google Scholar] [CrossRef]
- Williams, ML. On the stress distribution at the base of a stationary crack. Journal of Applied Mechanics 1957, 24, 109–114. [Google Scholar] [CrossRef]
- Hutchinson, JW. Singular behaviour at the end of a tensile crack in a hardening material. Journal of the Mechanics and Physics of Solids 1968, 16, 13–31. [Google Scholar] [CrossRef]
- Rice, JR; Rosengren, GF. Plane strain deformation near a crack tip in a power-law hardening material. Journal of the Mechanics and Physics of Solids 1968, 16, 1–12. [Google Scholar] [CrossRef]
- Rice, JR. A path independent integral and the approximate analysis of strain concentration by notches and cracks. Journal of Applied Mechanics 1968, 35, 379–387. [Google Scholar] [CrossRef]
- Oriani, RA. The diffusion and trapping of hydrogen in steel. Acta Metallurgica 1970, 18, 147–157. [Google Scholar] [CrossRef]
- Nagumo, M. Fundamentals of hydrogen embrittlement; Springer Nature Singapore: Singapore, 2023. [Google Scholar]
- Krom, AHM; Bakker, AD. Hydrogen trapping models in steel. Metallurgical and Materials Transactions B 2000, 31B, 1475–1482. [Google Scholar] [CrossRef]
- Nabarro, FRN. The strains produced by precipitation in alloys. Proceedings of the Royal Society 1940, A175, 519–538. [Google Scholar] [CrossRef]
- Eshelby, JD. The determination of the elastic field of an ellipsoidal inclusion, and related problems. Proceedings of the Royal Society 1957, A241, 376–396. [Google Scholar] [CrossRef]
- Lee, JK; Earmee, YY; Aaronson, HI; Russell, KC. Plastic relaxation of the transformation strain energy of a misfitting spherical precipitate: Ideal plastic behavior. Metallurgical Transactions A 1980, 11A, 1837–1847. [Google Scholar] [CrossRef]
- Earmee, YY; Johnson, WC; Lee, JK. Plastic relaxation of the transformation strain energy of a misfitting spherical precipitate: Linear and power-law strain hardening. Metallurgical Transactions A 1981, 12A, 1521–1530. [Google Scholar] [CrossRef]
- Ramberg, W; Osgood, WR. Description of stress-strain curves by three parameters. NACA, TN 902, 1943. [Google Scholar]
- Bai, JB; Francois, D. Some evidence of a brittle-ductile transition of zirconium hydride between 20 and 350oC. Journal of Nuclear Materials 1992, 187, 186–189. [Google Scholar] [CrossRef]
- Bai, JB; Prioul, C; Francois, D. Hydride embrittlement in zircaloy-4: part I. influence of microstructure on the hydride embrittlement in zircaloy-4 at 20oC and 350oC. Metallurgical and Materials Transactions A 1994, 25A, 1185–1197. [Google Scholar] [CrossRef]
- Arsene, S; Bai, JB; Bompard, P. Hydride embrittlement and irradiation effects on the hoop mechanical properties of pressurized water reactor (PWR) and boiling-water reactor (BWR) zircaloy cladding tubes: part I. hydride embrittlement in stress-relieved, annealed, and recrystallized zircaloys at 20oC and 300oC. Metallurgical and Materials Transactions A 2003, 34A, 553–566. [Google Scholar]
- Ebling, F; Bratsch, P; Wagner, S; Pundt, A; Preußner, J; Oesterlin, H; Wackermann, K; Michler, T. Assessing hydrogen embrittlement of alloy 718: Hollow and conventional tensile tests. Engineering Fracture Mechanics 2025, 319, 111028. [Google Scholar] [CrossRef]
- Hamed, A; Beschliesser, M; Fink, S; Mori, G. Assessment of the suitability of an existing L360NB natural gas pipeline for hydrogen transport using the hollow specimen technique. Journal of Materials Research and Technology 2025, 36, 8942–8952. [Google Scholar] [CrossRef]
- Symons, DM; Thompson, AW. The effect of hydrogen on the fracture of alloy X-750. Metallurgical and Materials Transactions A 1996, 27A, 101–110. [Google Scholar] [CrossRef]
- Gray, HR. Hydrogen environment embrittlement; TM X-68088; NASA, 1972. [Google Scholar]
- Rhythm, S; Ahmed, R; Ahmed, N; Gyaabeng, M; Teodoriu, C. Experimental study of the impact of hydrogen embrittlement on the ductility of natural gas pipeline steels. Gas Science and Engineering 2025, 143, 205746. [Google Scholar] [CrossRef]
- Hicks, PD; Altstetter, CJ. Hydrogen-enhanced cracking of superalloys. Metallurgical Transactions A 1992, 23A, 237–249. [Google Scholar] [CrossRef]
- Publication No. SMC-045; INCONEL® alloy 718, 2007. Special Metals Corporation.
- INCONEL® alloy 625. Special Metals Corporation, 2013.
- Katz, Y; Tymiak, N; Gerberich, WW. Nanomechanical probes as new approaches to hydrogen/deformation interaction studies. Engineering Fracture Mechanics 2001, 68, 619–646. [Google Scholar] [CrossRef]
- de Araújo, RO; Ayako Goto Donato, T; dos Santos Jorge Sousa, K; Akira Bazaglia Kuroda, P; Grandini, CR. Unveiling the effect of Mo on the crystal structure of β-BCC, microhardness, elastic modulus, and biocompatibility in titanium alloys. Journal of Alloys and Compounds Communications 2025, 6, 100076. [Google Scholar] [CrossRef]
- Martins, JR JRS; Araújo, RO; Nogueira, RA; Grandini, CR. Internal friction and microstructure of Ti and Ti-Mo alloys containing oxygen. Archives of Metallurgy and Materials 2016, 61, 25–30. [Google Scholar] [CrossRef]
- Jata, KV; Gerberich, WW; Beevers, CJ. Low-temperature fatigue crack propagation in a β-titanium alloy. ASTM STP 857In Fatigue at Low Temperatures; Stephens, RI, Ed.; American Society for Testing and Materials: Philadelphia, 1985; pp. 102–120. [Google Scholar]
- Denbigh, KG. The principles of chemical equilibrium; Cambridge University Press: Cambridge, 1955. [Google Scholar]
- San Marchi, C; Somerday, BP; Robinson, SL. Permeability, solubility and diffusivity of hydrogen isotopes in stainless steels at high gas pressures. International Journal of Hydrogen Energy 2007, 32, 100–116. [Google Scholar] [CrossRef]
- Kim, Y; Chao, YJ; Pechersky, MJ; Morgan, MJ. On the effect of hydrogen on the fracture toughness of steel. International Journal of Fracture 2005, 134, 339–347. [Google Scholar] [CrossRef]
- Wei, H; Tang, H; Xing, B; Shang, J; Qiu, S; Hua, Z; Gu, C. Research on the fracture toughness of pipeline steel X80 in a hydrogen environment. Engineering Failure Analysis 2025, 180, 109917. [Google Scholar] [CrossRef]
- API 5L; Specification for line pipe. American Petroleum Institute, 2018.
- Somerday, BP. Technical reference on hydrogen compatibility of materials. Plain carbon ferritic steels: C-Mn alloys (code 1100). Sandia National Laboratories, 2008; pp. SAND2008–1163.
- Efsing, P. Delayed hydride cracking in irradiated zircaloy. Ph.D. Thesis, Royal Institute of Technology, Stockholm, 1998. [Google Scholar]
- Kubo, T; Kobayashi, Y; Uchikoshi, H. Measurements of delayed hydride cracking propagation rate in the radial direction of Zircaloy-2 cladding tubes. Journal of Nuclear Materials 2012, 427, 18–29. [Google Scholar] [CrossRef]
- Nelson, HG; Williams, DP; Stein, JE. Environmental hydrogen embrittlement of an α-β titanium alloy: Effect of microstructure. Metallurgical Transactions 1972, 3, 469–475. [Google Scholar] [CrossRef]
- Varias, AG; Feng, JL. Simulation of hydride-induced steady-state crack growth in metals – Part I: Growth near hydrogen chemical equilibrium. Computational Mechanics 2004, 34, 339–356. [Google Scholar] [CrossRef]
- Senkov, ON; Dubois, M; Jonas, JJ. Elastic moduli of titanium-hydrogen alloys in the temperature range 20 oC to 1100 oC. Metallurgical and Materials Transaction A 1996, 27A, 3963–3970. [Google Scholar] [CrossRef]
- Gu, J; Hardie, D. Effect of hydrogen on the tensile ductility of Ti6Al4V - Part II Fracture of pre-cracked tensile specimens. Journal of Materials Science 1997, 32, 609–617. [Google Scholar] [CrossRef]
- Hardie, D; Ouyang, S. Effect of hydrogen and strain rate upon the ductility of mill-annealed Ti6Al4V. Corrosion Science 1999, 41, 155–177. [Google Scholar] [CrossRef]
- Bertolino, G; Meyer, G; Perez Ipiña, JP. In situ crack growth observation and fracture toughness measurement of hydrogen charged Zircaloy-4. Journal of Nuclear Materials 2003, 322, 57–65. [Google Scholar] [CrossRef]
- Rosinger, HE; Northwood, DO. The elastic properties of zirconium alloy fuel cladding and pressure tubing materials. Journal of Nuclear Materials 1979, 79, 170–179. [Google Scholar] [CrossRef]
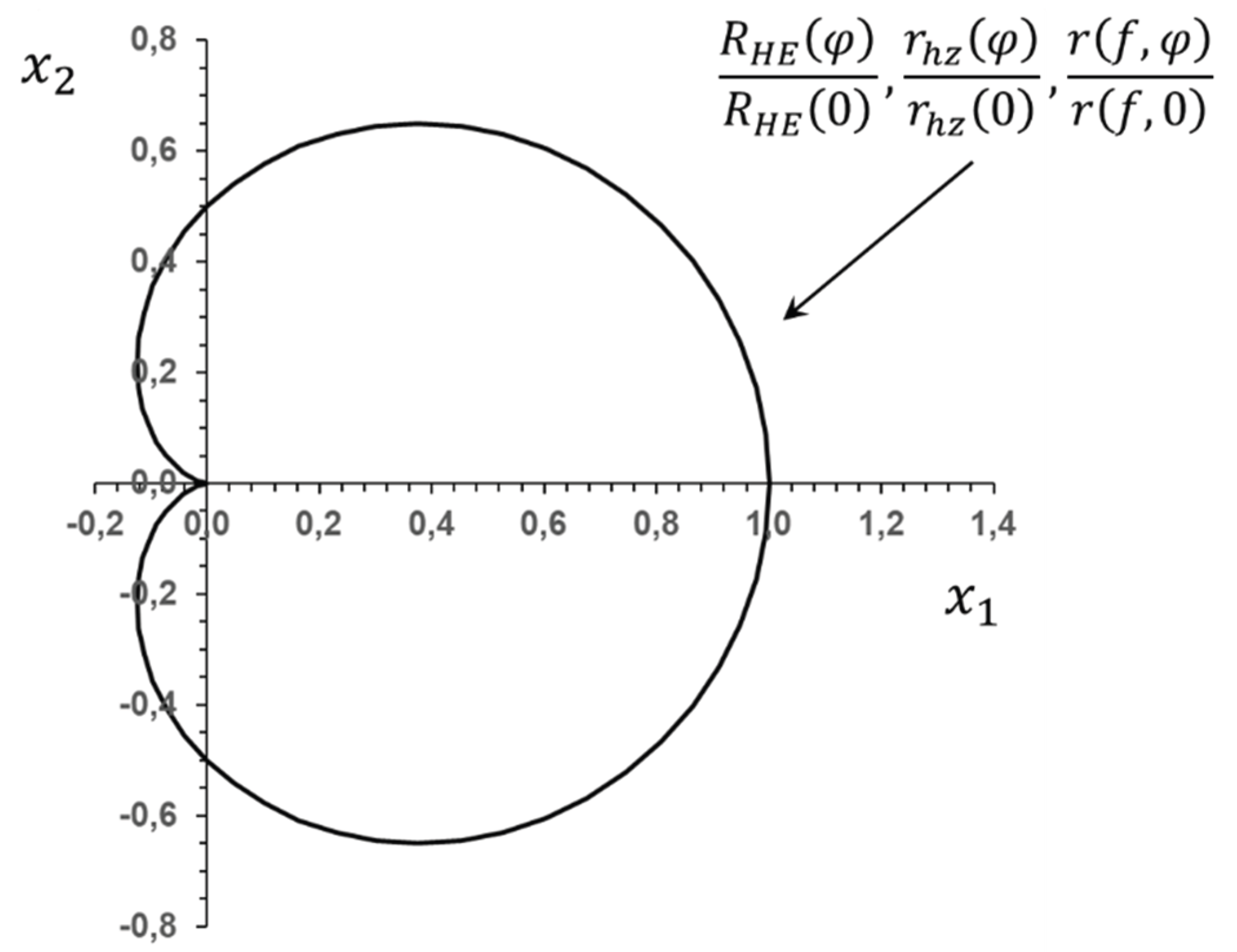
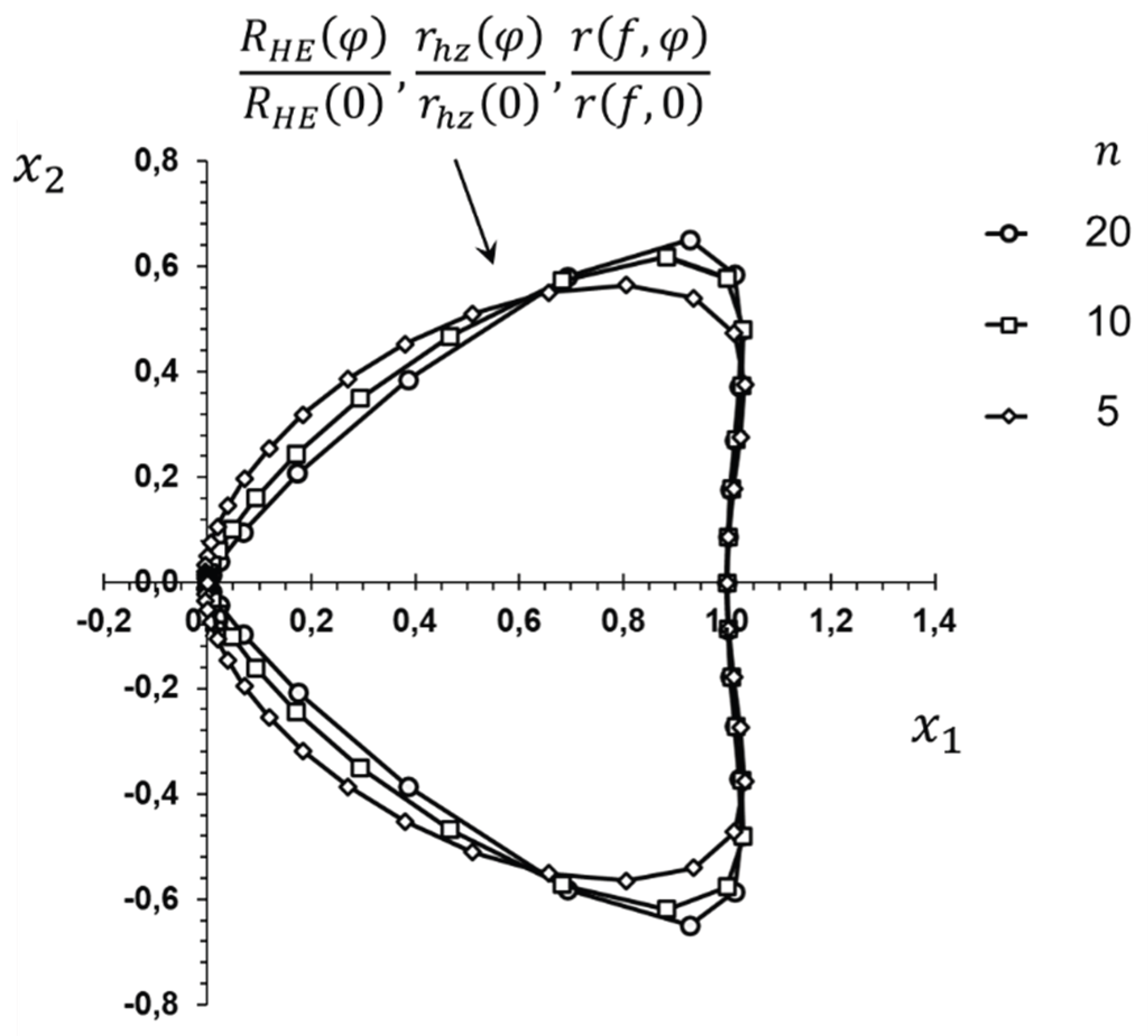
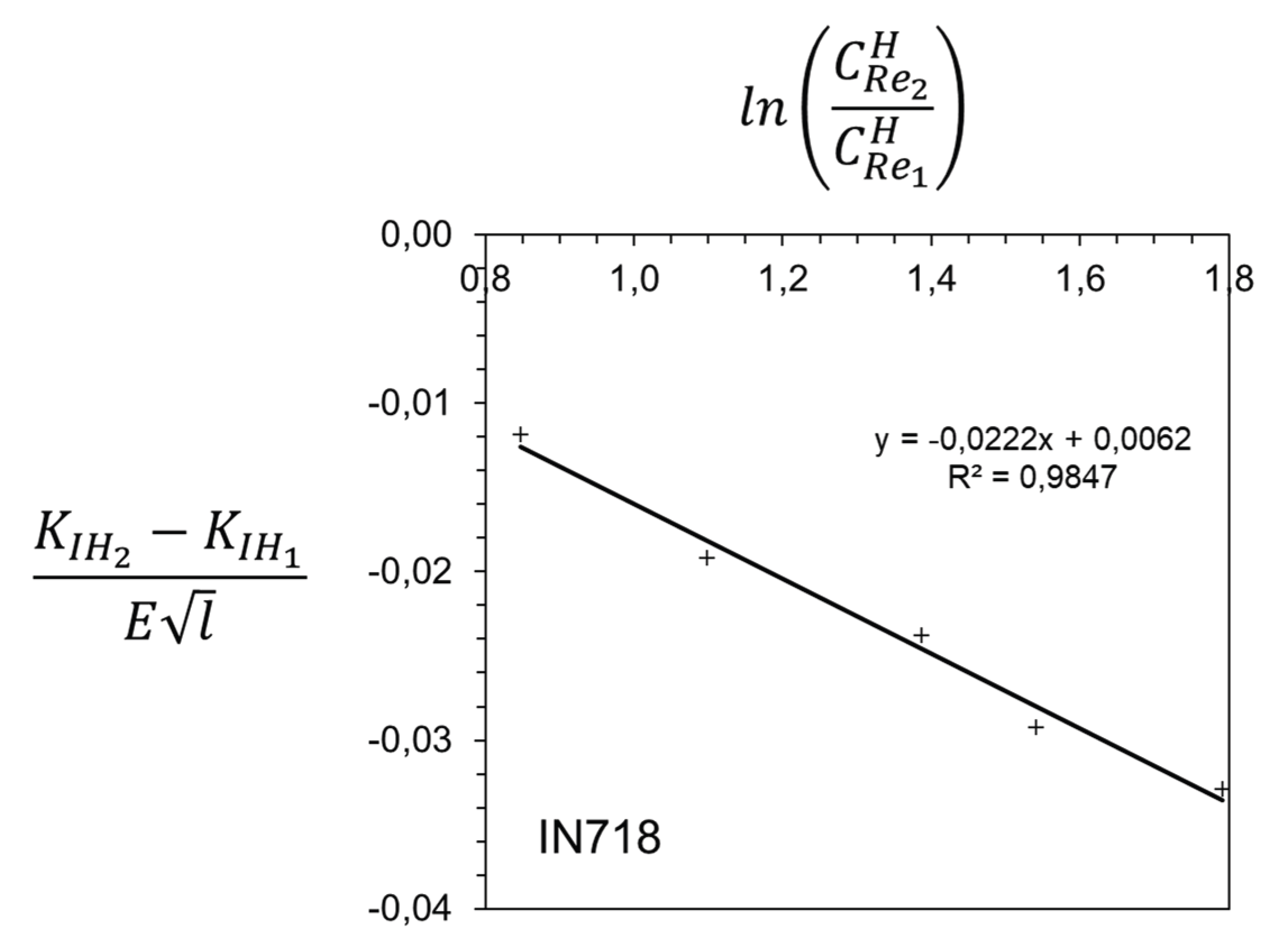
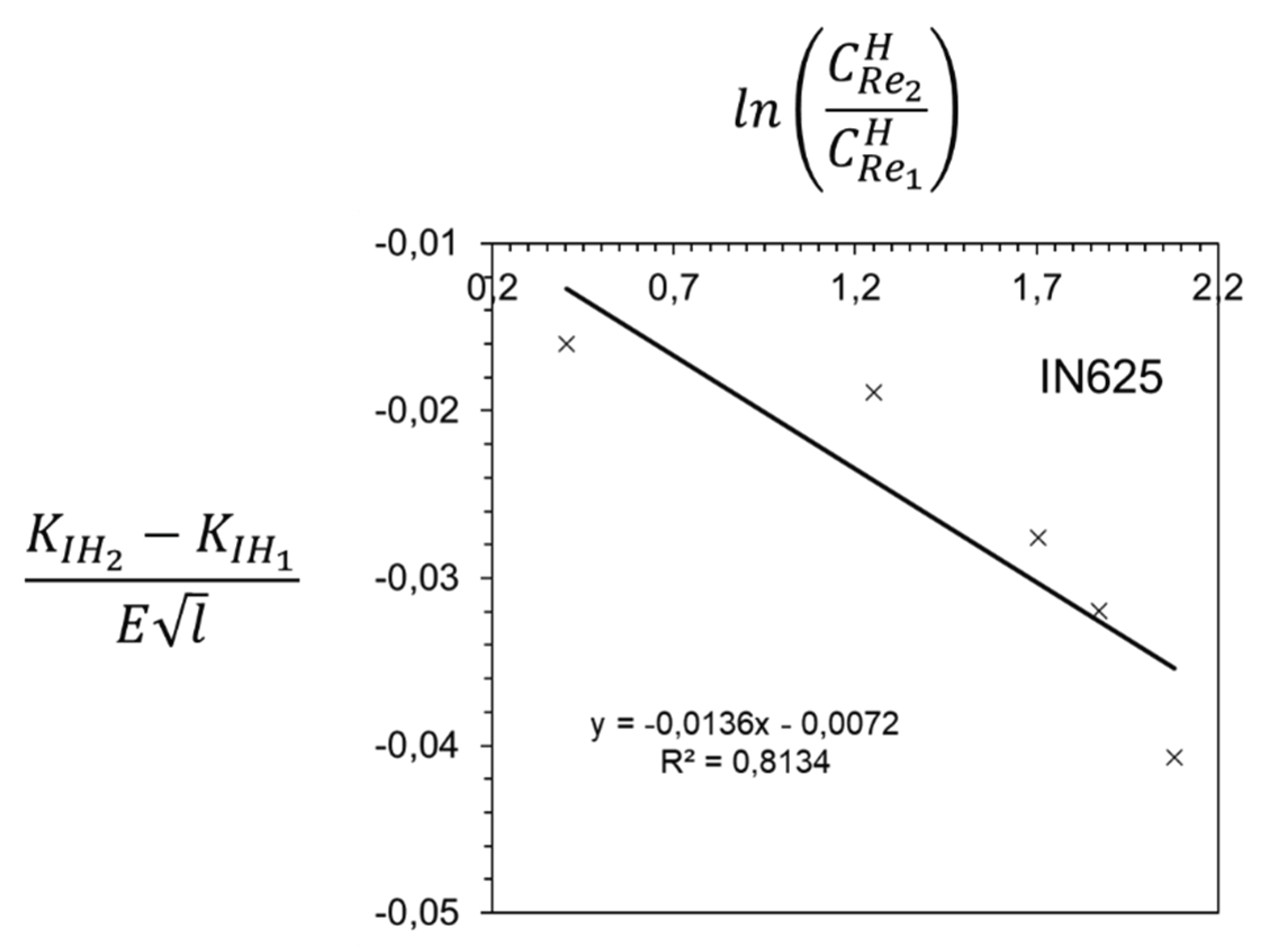
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




