Submitted:
11 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
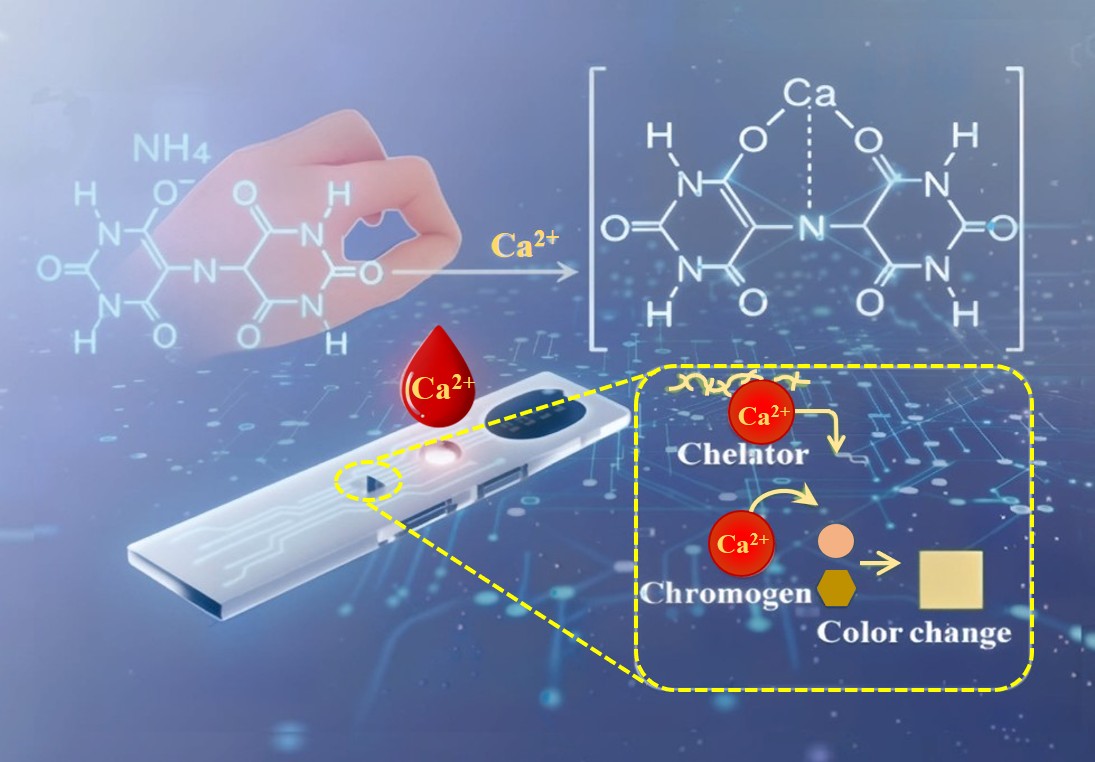
Keywords:
1. Introduction
2. Materials and Methods
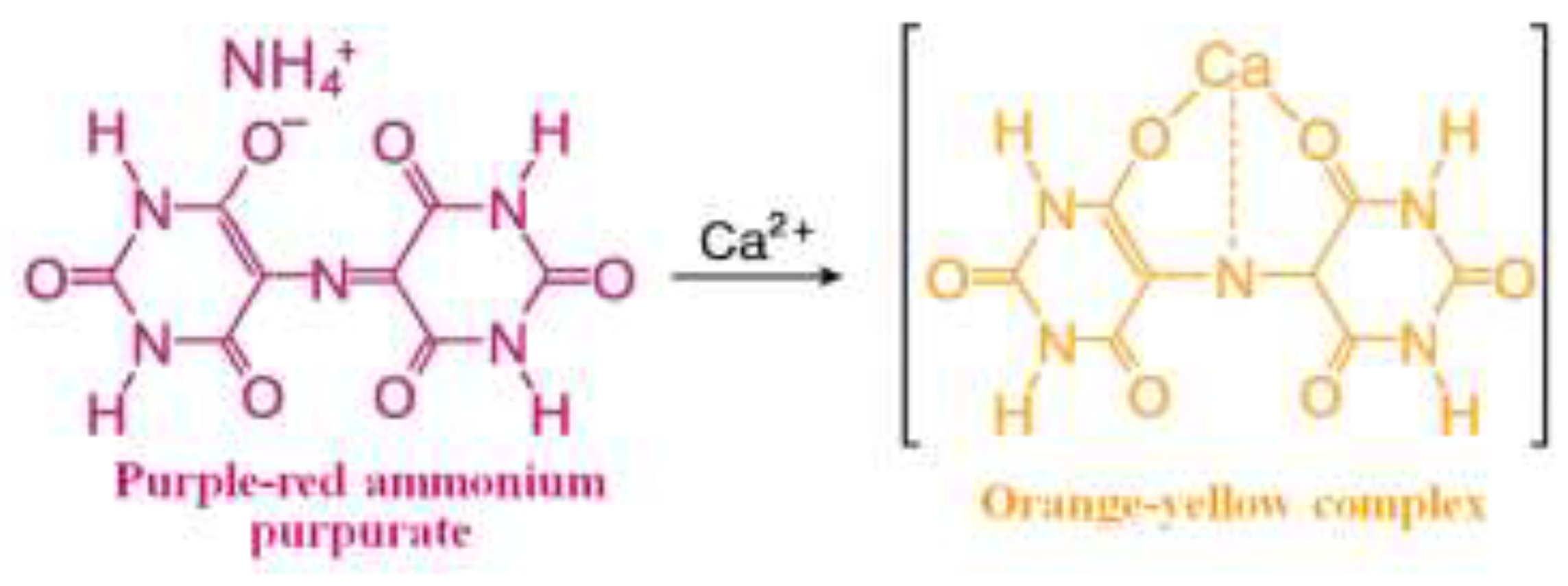
2.1. Sample and Reagent Preparation
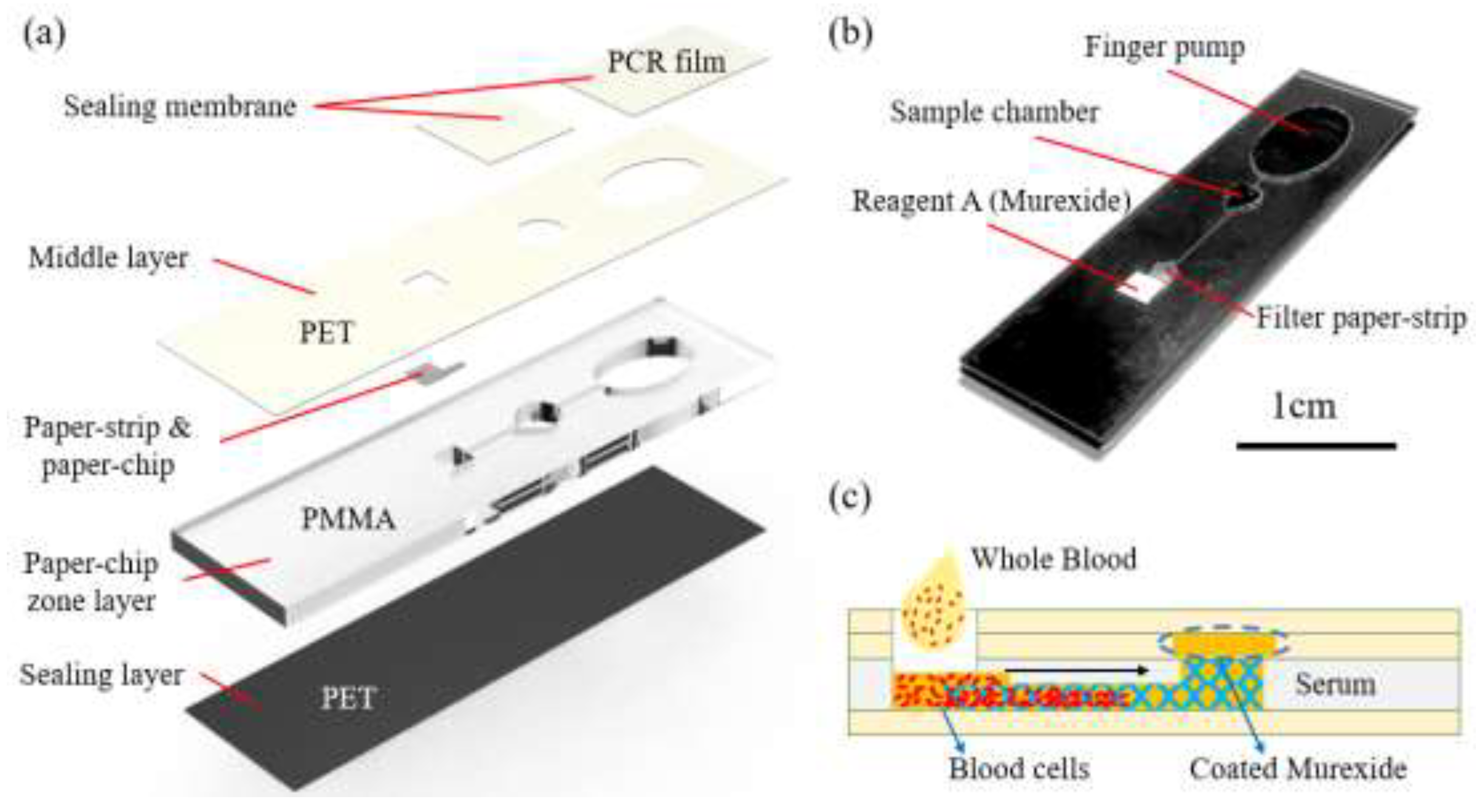
2.2. 3D Microfluidic Finger Pump Device
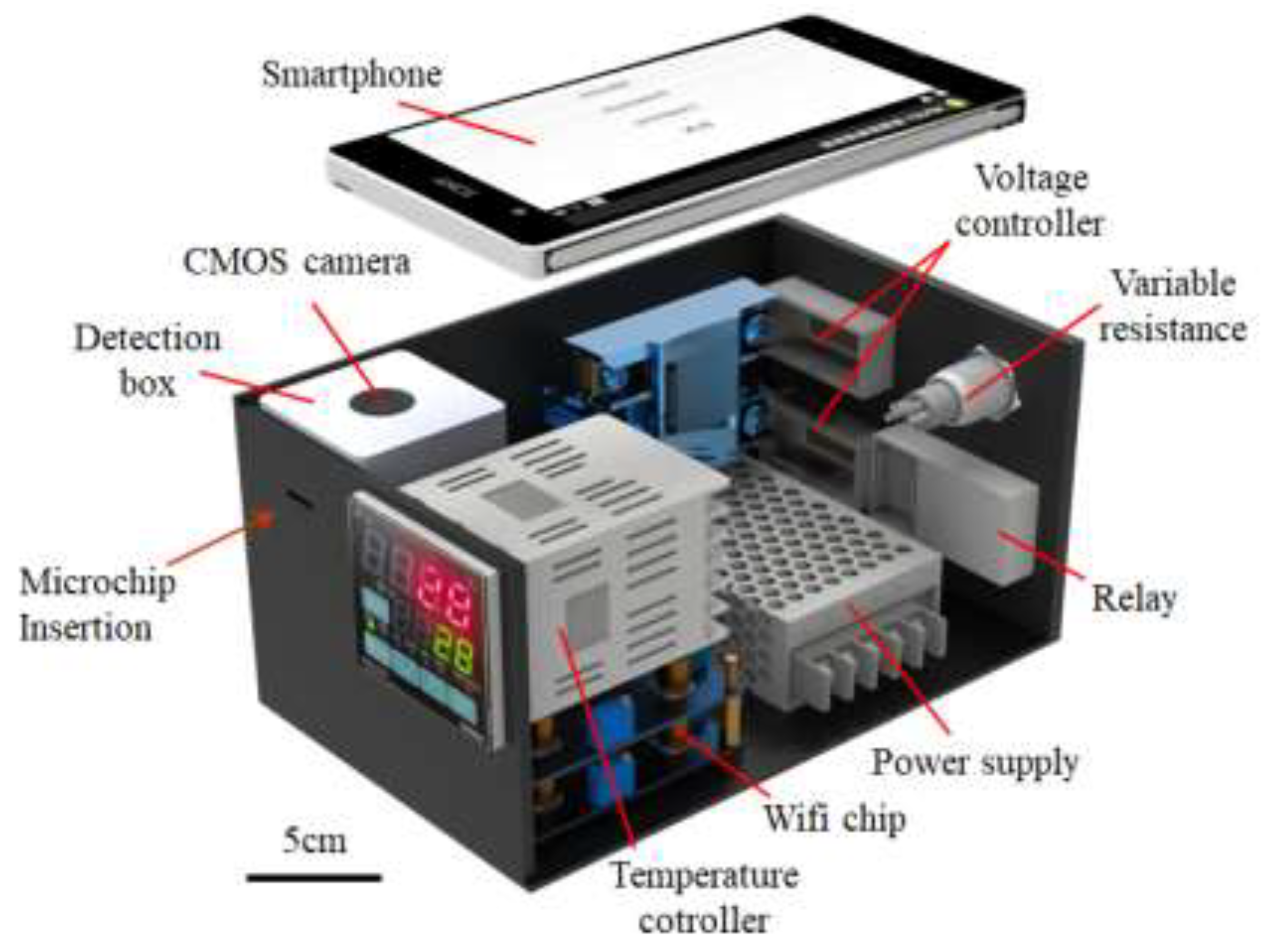
2.3. Hand-Held Detection System
3. Results
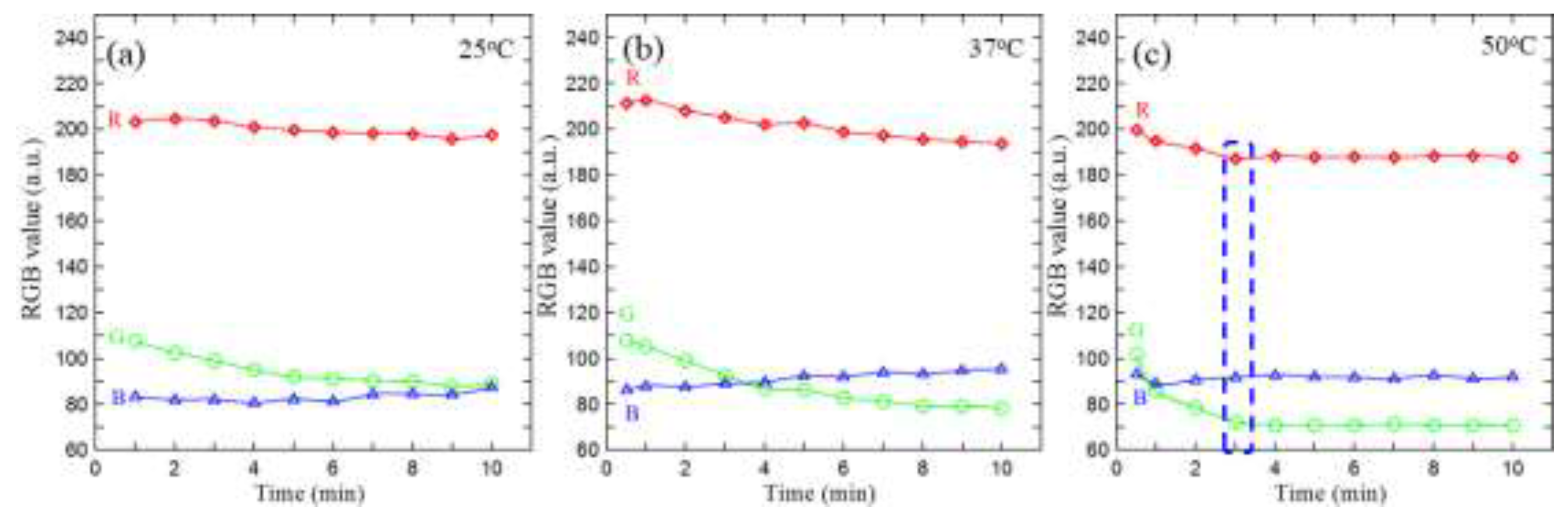
3.1. Effects of Reaction Time and Reaction Temperature
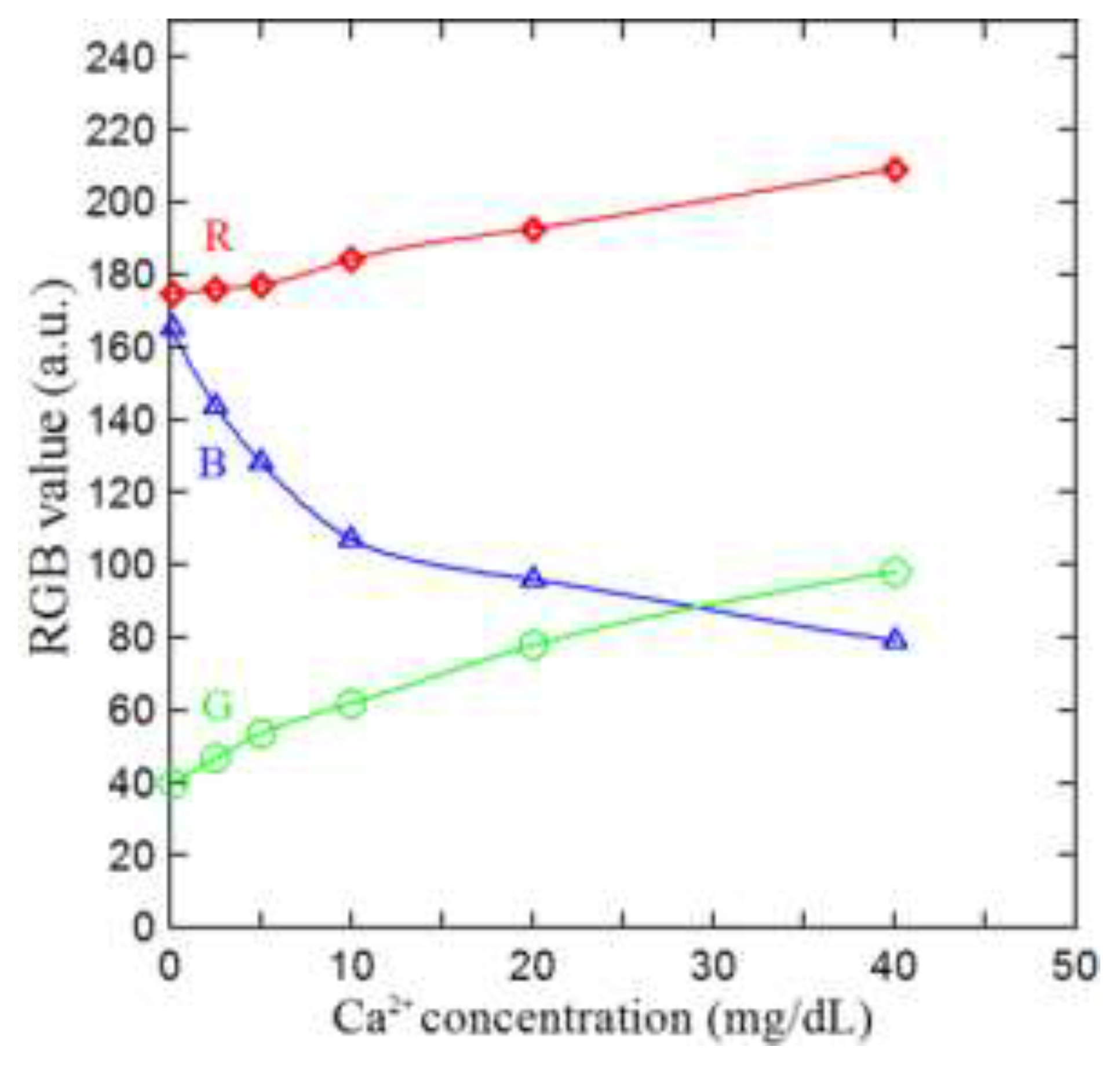
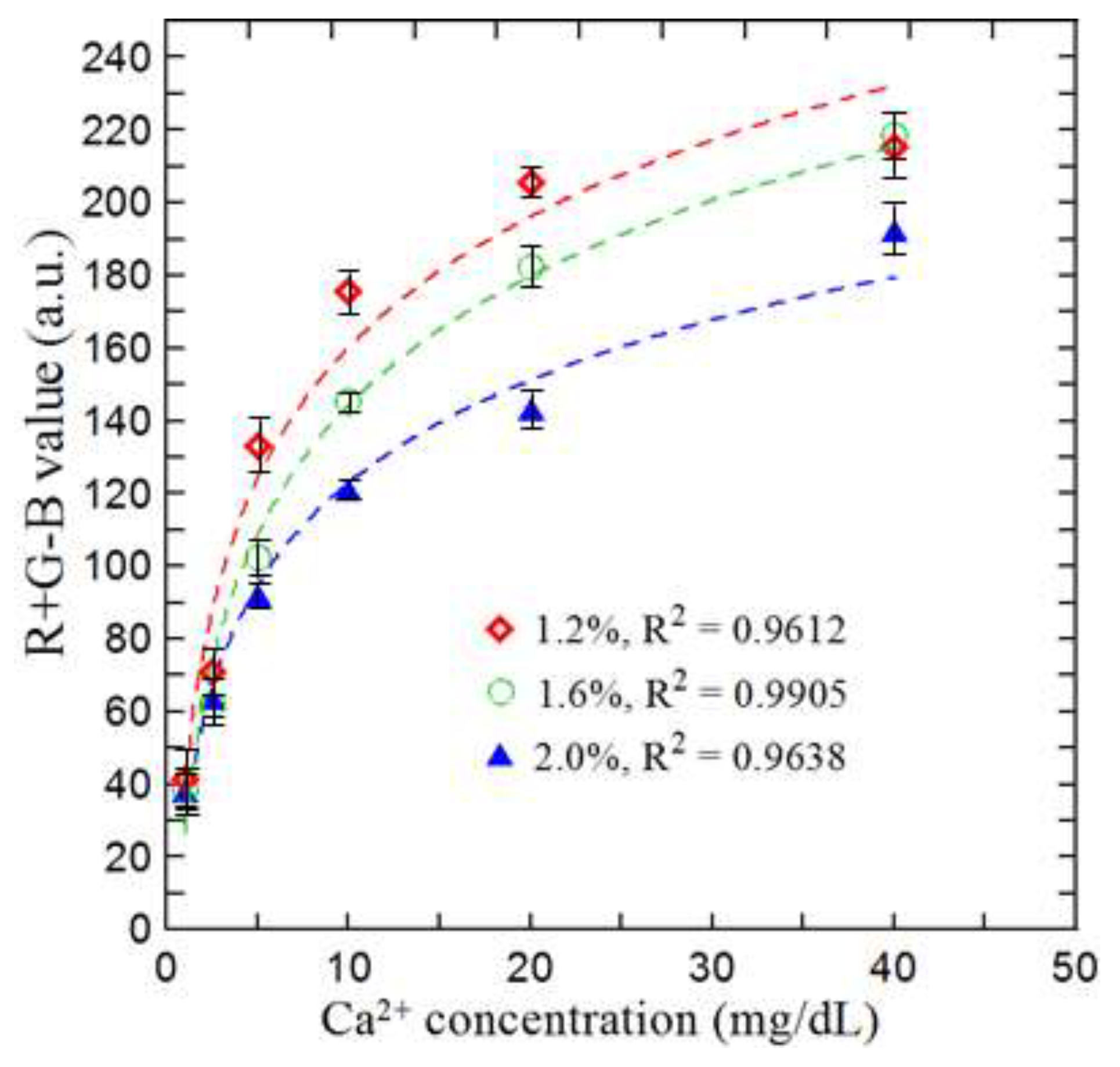
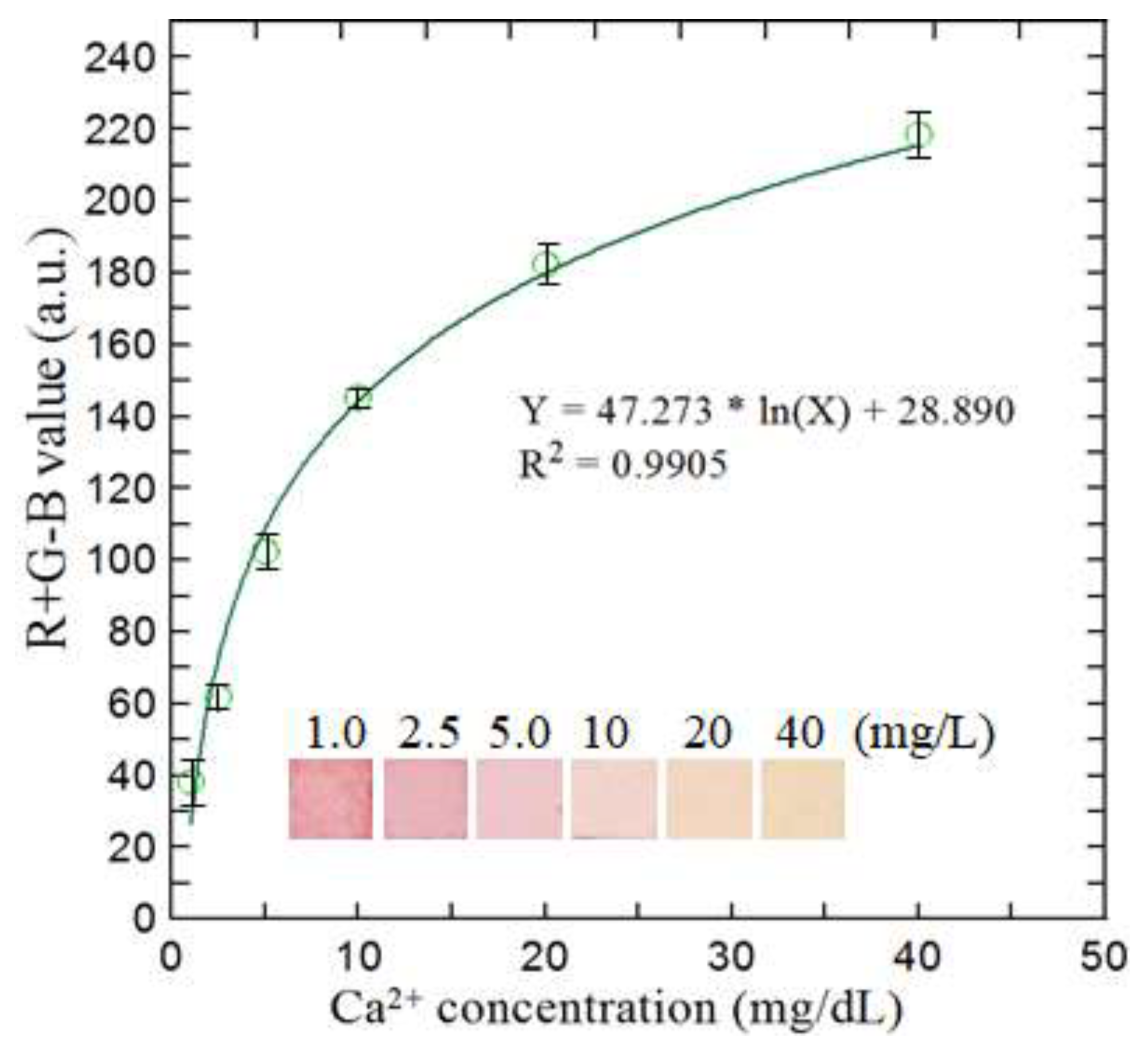
3.2. Effect of Reagent Concentration and Construction of Calibration Curve
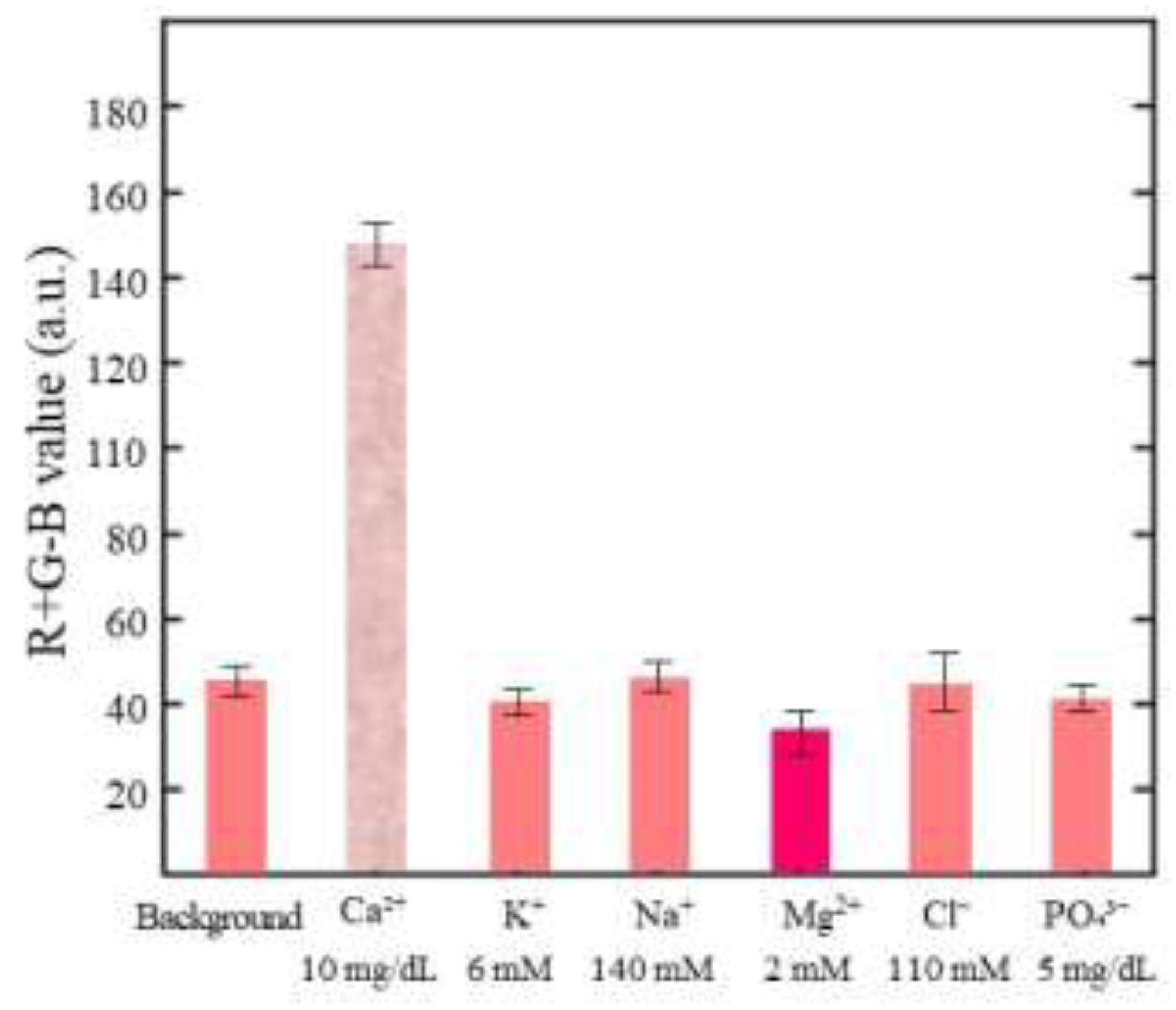
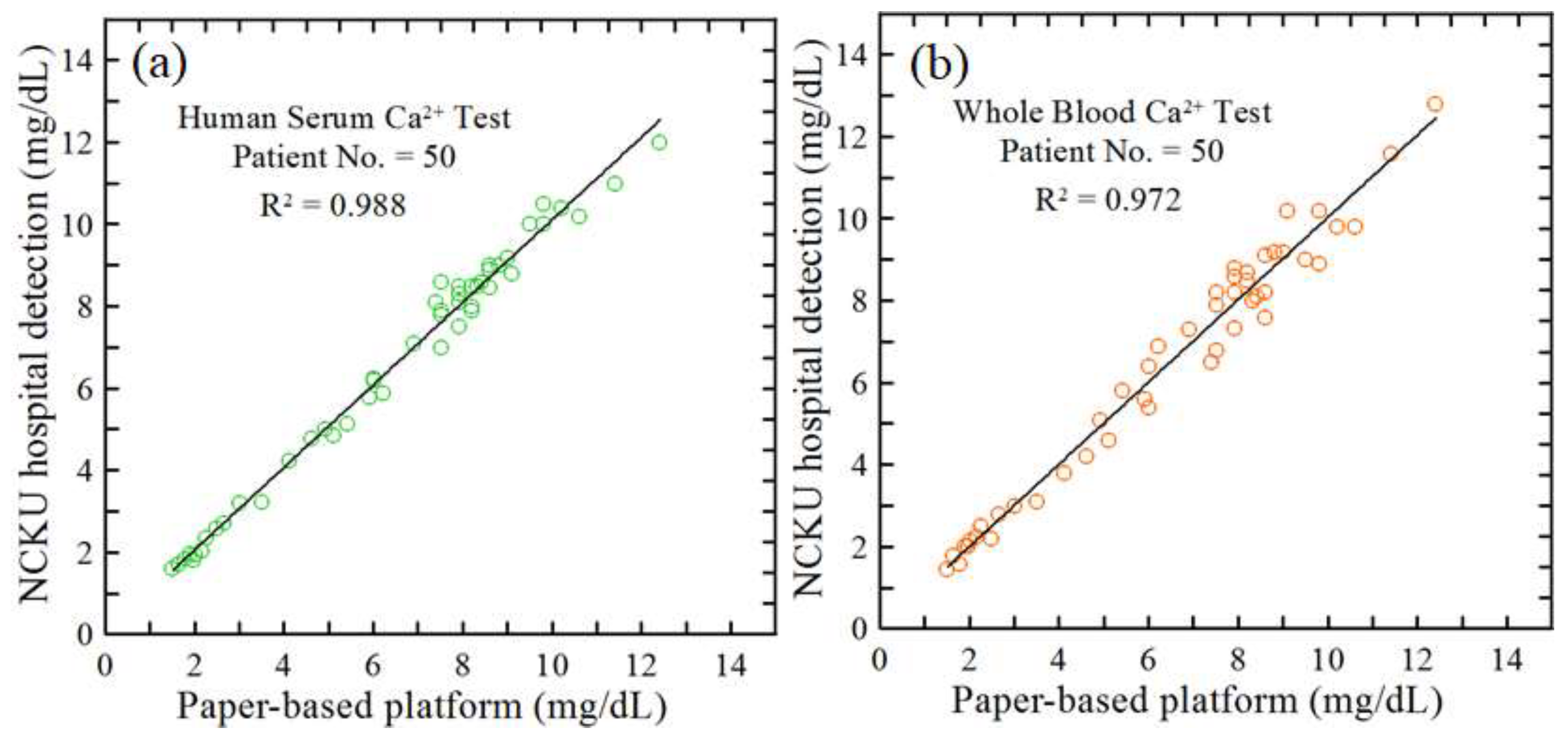
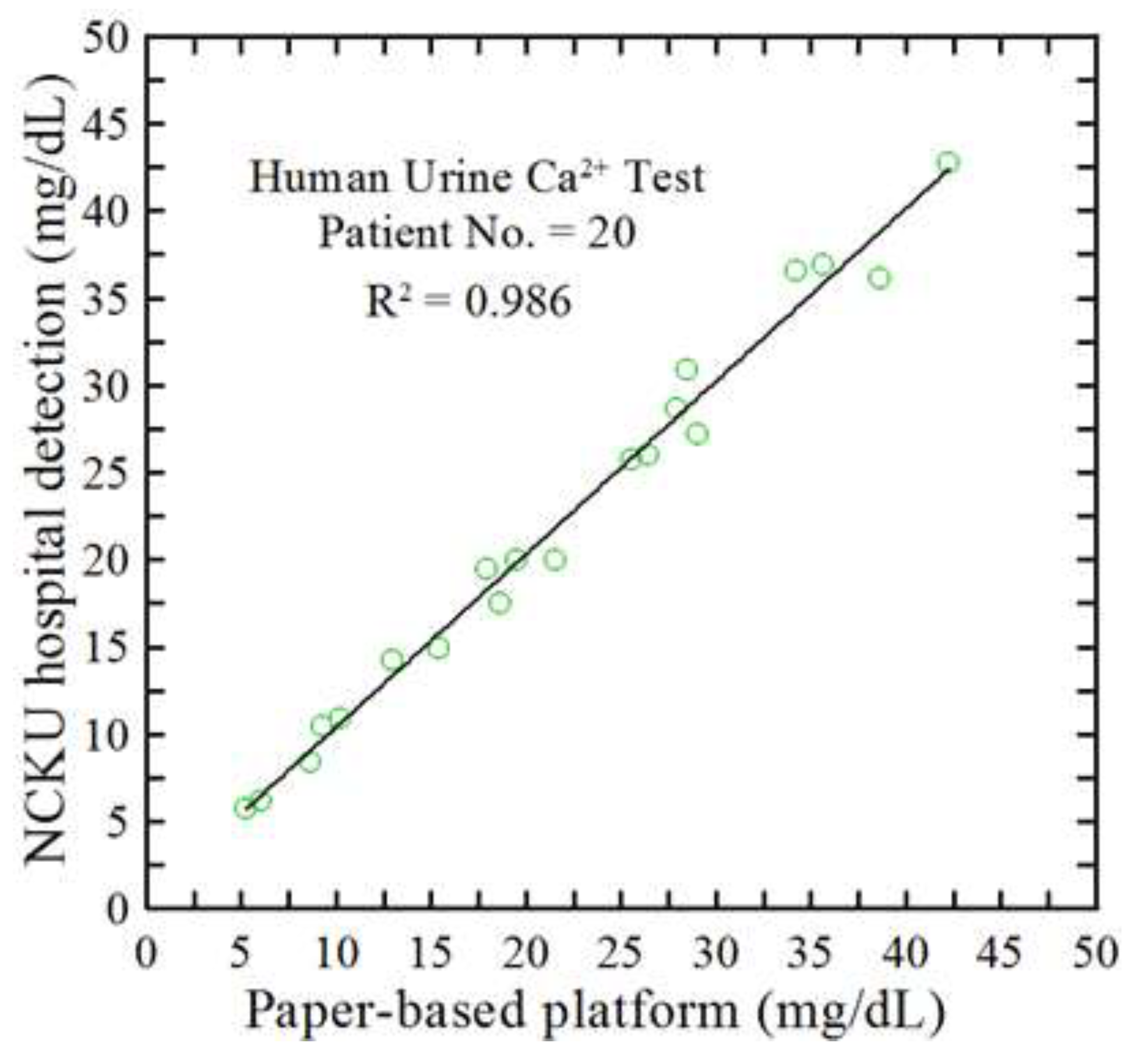
3.3. Ca²⁺ Determination in Real-World CKD Patient Samples
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Minutolo, R.; Lapi, F.; Chiodini, P.; Simonetti, M.; Bianchini, E.; Pecchioli, S.; Cricelli, I.; Cricelli, C.; Piccinocchi, G.; Conte, G. Risk of ESRD and death in patients with CKD not referred to a nephrologist: a 7-year prospective study. Clin. J. Am. Soc. Nephrol. 2014, 9, 1586. [Google Scholar] [CrossRef] [PubMed]
- Falodia, J.; Singla, M.K. CKD epidemiology and risk factors. Clin. Nephrol. 2012, 1, 249–252. [Google Scholar] [CrossRef]
- Anders, H.J.; Huber, T.B.; Isermann, B.; Schiffer, M. CKD in diabetes: diabetic kidney disease versus nondiabetic kidney disease. Nat. Rev. Nephrol. 2018, 14, 361–377. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Broadley, M.R. Biofortification of crops with seven mineral elements often lacking in human diets–iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 2009, 182, 49–84. [Google Scholar] [CrossRef]
- Peacock, M. Calcium metabolism in health and disease. Clin. J. Am. Soc. Nephrol. 2010, 5, S23–S30. [Google Scholar] [CrossRef]
- Moysés-Neto, M.; Guimarães, F.M.; Ayoub, F.H.; Vieira-Neto, O.M.; Costa, J.A.C.; Dantas, M. Acute renal failure and hypercalcemia. Ren. Fail. 2006, 28, 153–159. [Google Scholar] [CrossRef]
- Dell’Aquila, C.; Neal, A.L.; Shewry, P.R. Development of a reproducible method of analysis of iron, zinc and phosphorus in vegetables digests by SEC-ICP-MS. Food Chem. 2020, 308, 125652. [Google Scholar] [CrossRef]
- Ajayi, D.T.; Teepoo, S. Smartphone-based colorimetric microfluidic paper-based analytical device for on-site detection of calcium ions in milk samples. Anal. Lett. 2024, 58, 465–480. [Google Scholar] [CrossRef]
- Qiu, Y.; Ma, C.; Jiang, N.; Jiang, D.; Yu, Z.; Liu, X.; Zhu, Y.; Yu, W.; Li, F.; Wan, H.; Wang, P. A silicon-based field-effect biosensor for drug-induced cardiac extracellular calcium ion change detection. Biosensors 2024, 14, 16. [Google Scholar] [CrossRef]
- Ma, C.; Qiu, Y.; Liang, T.; Jiang, N.; Kong, L.; Wu, J.; Sun, X.; Ren, G.; Wei, X.; Wang, P.; Wan, H. Label-free and highly-sensitive detection of calcium ions using a silicon-on-sapphire light-addressable potentiometric sensor. Anal. Chim. Acta 2024, 1294, 342282. [Google Scholar] [CrossRef]
- Wu, H.; Wang, Y.; Wang, W.; Cai, Y.; Liao, X.; Yang, B.; Gao, C.; Yang, J. Functional fluorescent probe based on amide condensation of coumarin and melatonin for the turn-on detection of calcium ion in vitro and living cell. J. Mol. Liq. 2024, 401, 124646. [Google Scholar] [CrossRef]
- Cui, J.; Xia, Y.; Jin, L.; Chen, J.; Huo, Z.; Shen, F.; Mi, L. A highly sensitive and anti-interference Bio-QDs biosensor for electrochemical detection calcium ion in plant tissue culture medium. Microchem. J. 2025, 212, 113284. [Google Scholar] [CrossRef]
- Liu, Z.H.; Cai, X.; Dai, H.H.; Zhao, Y.H.; Gao, Z.W.; Yang, Y.F.; Liu, Y.Z.; Yang, M.; Li, M.Q.; Li, P.H.; Huang, X.J. Highly stable solid contact calcium ion-selective electrodes: Rapid ion–electron transduction triggered by lipophilic anions participating in redox reactions of CunS nanoflowers. Anal. Chem. 2024, 96, 9069–9077. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.; Kim, W.; Cho, H.; Han, J.; Sim, S.J.; Song, H.G.; Pak, Y.; Song, H.S. Label-free optical detection of calcium ion influx in cell-derived nanovesicles using a conical Au/PDMS biosensor. Lab Chip 2024, 24, 4138–4146. [Google Scholar] [CrossRef]
- Mettakoonpitak, J.; Chanthabun, A.; Hatsakhun, P.; Sirasunthorn, N.; Siripinyanond, A.; Henry, C.S. Microfluidic paper-based analytical devices for simple and nondestructive durian fruit maturity assessment. Anal. Chim. Acta 2024, 1329, 343252. [Google Scholar] [CrossRef]
- Farzin, M.A.; Naghib, S.M. Paper-based immunosensor integrated with bioinspired Cu-polydopamine nanozyme for voltammetric detection of CA-15-3 tumor marker. Sens. Actuators Rep. 2024, 8, 100209. [Google Scholar] [CrossRef]
- Huang, K.H.; Yu, C.X.; Lee, C.C.; Tseng, C.C.; Fu, L.M. Rapid microfluidic ion-exchange optode system for point-of-care determination of sodium concentration in serum. Biosensors 2025, 15, 104. [Google Scholar] [CrossRef]
- Li, X.; Liang, X.; Li, H.; Song, J.; Li, K.; Zhang, M.; Zhang, H.; Han, Z.; Chu, L.T.; Guo, W. Facile patterning of microfluidic paper-based analytical devices (μPADs) by dispensing propylene glycol methyl ether acetate (PGMEA). Sens. Actuators Rep. 2025, 9, 100323. [Google Scholar] [CrossRef]
- Mansor, M.A.; Jamrus, M.A.; Lok, C.K.; Ahmad, M.R.; Petrů, M.; Koloor, S.S.R. Microfluidic device for both active and passive cell separation techniques: A review. Sens. Actuators Rep. 2025, 9, 100277. [Google Scholar] [CrossRef]
- Hsueh, W.T.; Yu, C.X.; Cheng, H.C.; Chen, M.Y.; Wang, H.M.; Fu, L.M. A comprehensive review of wearable devices for non-invasive biosensing. TrAC-Trends Anal. Chem. 2025, 193, 118425. [Google Scholar] [CrossRef]
- Al Lawati, H.A.J.; Hassanzadeh, J.; Al-Maqbali, L.; Morsali, A. Fully integrated microfluidic paper-based analytical device for straightforward extraction and estimation of the total phenolic content of olive oil samples. Sens. Actuators B Chem. 2025, 431, 137419. [Google Scholar] [CrossRef]
- Ko, C.H.; Tseng, C.C.; Lu, S.Y.; Lee, C.C.; Kim, S.; Fu, L.M. Handheld microfluidic multiple detection device for concurrent blood urea nitrogen and creatinine ratio determination using colorimetric approach. Sens. Actuators B Chem. 2025, 422, 136585. [Google Scholar] [CrossRef]
- Adampourezare, M.; Asadpour-Zeynali, K.; de la Guardia, M.; Dolatabadi, J.E.N. The design of paper-based electroanalytical microfluidic device coupled with post-synthesized molecularly imprinted polymers (rGO/Au@Ag₂S/PANI/polyacrylamide) for the detection of streptomycin. Sens. Actuators Rep. 2025, 9, 100297. [Google Scholar] [CrossRef]
- Chiang, K.-H.; Chang, W.-J.; Liu, Y.-H.; Lin, H.-L.; Lin, C.-T.; Chiou, T.-W.; Wu, H.-M.; Tung, Y.-C. Quantification of oxygen tension variation in an in vitro tumor microenvironment model based on co-culture of tumor spheroid and endothelial cells in a microfluidic device. Sens. Actuators Rep. 2025, 10, 100396. [Google Scholar] [CrossRef]
- Farahinia, A.; Zhang, W.; Badea, I. Centrifugal microfluidic systems for cancer cell separation: Advances, challenges, and applications. Sens. Actuators Rep. 2025, 10, 100387. [Google Scholar] [CrossRef]
- Chen, S.J.; Lu, S.Y.; Tseng, C.C.; Huang, K.H.; Chen, T.L.; Fu, L.M. Rapid microfluidic immuno-biosensor detection system for point-of-care determination of high-sensitivity urinary C-reactive protein. Biosensors 2024, 14, 283. [Google Scholar] [CrossRef]
- Lu, S.Y.; Tseng, C.C.; Yu, C.X.; Huang, K.H.; Chen, T.L.; Fu, L.M.; Wu, P.H. Rapid microfluidic fluorescence detection platform for determination of whole blood sodium. Sens. Actuators B Chem. 2024, 400, 134839. [Google Scholar] [CrossRef]
- Rapier, C.E.; Jagadeesan, S.; Vatine, G.D.; Ben-Yoav, H. Impedance characteristics of microfluidic channels and integrated coplanar parallel electrodes as design parameters for whole-channel analysis in organ-on-chip micro-systems. Biosensors 2024, 14, 374. [Google Scholar] [CrossRef]
- Wang, M.; Pang, W.; Zhang, J.; Wang, L.; Wang, C.; Liu, Z.; Cheng, Z.; Luo, P.; Gao, Z.; Zhou, H. Integrated multi-channel microfluidic biosensing chip based on chain amplified enhanced fluorescence for determination of SEs. Sens. Actuators B Chem. 2025, 442, 138149. [Google Scholar] [CrossRef]
- Khachornsakkul, K.; Trakoolwilaiwan, T.; Del-Rio-Ruiz, R.; Friesen, E.; Dungchai, W.; Leelasattarathkul, T. Photothermal paper-based microfluidic analytical device integrated with carbon nanomaterials and molecularly imprinted polymers for sensitive perfluorooctanesulfonate quantification. ACS Sens. 2025, 10, 5008–5018. [Google Scholar] [CrossRef]
- Zhang, T.; Tang, M.; Yang, S.; Fa, H.; Wang, Y.; Huo, D.; Hou, C.; Yang, M. Development of a novel ternary MOF nanozyme-based smartphone-integrated colorimetric and microfluidic paper-based analytical device for trace glyphosate detection. Food Chem. 2025, 464, 141780. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.C.; Chen, T.L.; Wang, H.M.; Huang, K.H.; Fu, L.M. Simultaneous quantification of fructose and sucrose in beverages using microfluidic paper chip and colorimetric pixel-area analysis method. Food Chem. 2025, 496, 146702. [Google Scholar] [CrossRef] [PubMed]
- Zou, R.; Yu, Q.; Wang, Z.; Yang, P.; Zhao, Y.; Liu, Y.; Fu, Y.; Guo, Y. A portable 3D-printed lab-on-a-chip device for on-site monitoring of thiamethoxam residue in food samples. Food Chem. 2025, 486, 144594. [Google Scholar] [CrossRef] [PubMed]
- Alahmad, W.; Cetinkaya, A.; Kaya, S.I.; Ozkan, S.A. Innovative and cutting-edge approaches in microfluidic paper-based analytical devices for detection of food adulteration. TrAC-Trends Anal. Chem. 2024, 181, 118012. [Google Scholar] [CrossRef]
- Rath, R.J.; Giaretta, J.; Hoang, T.P.; Zulli, R.; Farajikhah, S.; Talebian, S.; Naficy, S.; Dehghani, F. Cascading chemiresistive paper-based enzymatic biosensor for urea detection. Sens. Actuators Rep. 2025, 9, 100330. [Google Scholar] [CrossRef]
- Qureshi, Y.Z.A.N.; Li, M.; Chang, H.; Song, Y. Microfluidic chip systems for color-based antimicrobial susceptibility test: a review. Biosens. Bioelectron. 2025, 273, 117160. [Google Scholar] [CrossRef]
- Chu, P.-Y.; Huang, P.-S.; Chen, C.-Y.; Tsai, K.-Y.; Chiu, S.-Y.; Fan, L.-W.; Cheng, Y.-C.; Lin, C.-J.; Hsieh, C.-H.; Wu, M.-H. Development of a point-of-care testing (POCT)-use paper-based device for recombinase polymerase amplification (RPA)-based bioassays: Demonstration of the detection of Neisseria gonorrhoeae. Sens. Actuators Rep. 2025, 9, 100307. [Google Scholar] [CrossRef]
- Huang, W.; Wu, T.; Shallan, A.; Kostecki, R.; Rayner, C.K.; Priest, C.; Ebendorff-Heidepriem, H.; Zhao, J. A multiplexed microfluidic platform toward interrogating endocrine function: Simultaneous sensing of extracellular Ca²⁺ and hormone. ACS Sens. 2020, 5, 490–499. [Google Scholar] [CrossRef]
- Tarara, M.; Tzanavaras, P.D.; Tsogas, G.Z. Development of a paper-based analytical method for the colorimetric determination of calcium in saliva samples. Sensors 2023, 23, 198. [Google Scholar] [CrossRef]
- Biswas, P.; Karan, P.; Pal, S.; Ghosh, A.K.; Chakrabort, S. Smartphone-interfaced serum calcium-level quantification on a simple paper strip assay for diagnostics at extreme point of care. IEEE J. Flexible Electron. 2023, 2, 336–343. [Google Scholar] [CrossRef]
- Aguiar, J.I.S.; Rangel, A.O.S.S.; Mesquita, R.B.R. Salivary calcium determination with a specially developed microfluidic paper-based device for point-of-care analysis. Talanta Open 2023, 8, 100254. [Google Scholar] [CrossRef]
- Lewińska, I.; Ścibisz, M.; Tymecki, Ł. Microfluidic paper-based analytical device for simultaneous determination of calcium and magnesium ions in human serum. Anal. Chim. Acta 2024, 1308, 342639. [Google Scholar] [CrossRef]
- Johnson, Z.T.; Ellis, G.; Pola, C.C.; Banwart, C.; McCormick, A.; Miliao, G.L.; Duong, D.; Opare-Addo, J.; Sista, H.; Smith, E.A.; Hu, H.; Gomes, C.L.; Claussen, J.C. Enhanced laser-induced graphene microfluidic integrated sensors for on-site biomedical and environmental monitoring. Small 2025, 21, 2500262. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Liu, Y.; Liu, G.; Tan, Q.; Dou, X.; Xie, Y.; Zhang, X. Development of a low-cost flexible potentiometric detector and its integrated system for electrochemical sensing of electrolytes in human sweat. Sens. Actuators Rep. 2025, 9, 100286. [Google Scholar] [CrossRef]
- Ostad, M.A.; Hajinia, A.; Heidari, T. A novel direct and cost effective method for fabricating paper-based microfluidic device by commercial eye pencil and its application for determining simultaneous calcium and magnesium. Microchem. J. 2017, 133, 545–550. [Google Scholar] [CrossRef]
- Chung, Y.; Green, W.H. New modified Arrhenius equation to describe the temperature dependence of liquid phase reaction rates. Chem. Eng. J. 2025, 516, 163300. [Google Scholar] [CrossRef]
- Janić, N.; Zhukouskaya, H.; Černoch, P.; Pánek, J.; Svoboda, J.; Hajná, M.; et al. BAPTA-based potentiometric polymer sensor: towards sensing inflammations and infections. J. Mater. Chem. B 2025, 13, 4157–4165. [Google Scholar] [CrossRef]
| Author and year | Sample type | Real sample | Detection reagent, method & time | Detection range & LOD | Strengths / limitations |
|---|---|---|---|---|---|
| Current work | Whole blood, serum, urine | B (30) S (30) U (20) |
Murexide Colorimetric 3 min |
1–40 mg/dL LOD: 0.2 mg/dL | On-chip filtration enables whole blood/urine; 6 μL; rapid heated endpoint readout. Requires compact heater/imager; endpoint (not continuous) measurement. |
| NCKU Hospital | Serum, urine | Serum Urine |
Colorimetric | S: 0.8-20 mg/dL U: 3.4-285 mg/dL |
Clinical-grade standardization and throughput. Requires benchtop analyzer and laboratory workflow; not field/decentralized. |
| [38] Huang et al. 2020 | Serum | − | Ca²⁺ indicator Fluorescence 3 min |
0.2–2 mM LOD: 0.2 mM |
Sensitive optical readout; potential multiplexing. Optical components and assay complexity limit low-resource deployment. |
| [39] Tarara et al. 2023 | Saliva | 5 | MTB Colorimetric 10 min |
3.07–8.42 mg/dL LOD: 0.29 mg/dL |
Simple colorimetric workflow; saliva-friendly sampling. Longer assay time; matrix-specific applicability (not whole blood). |
| [40] Biswas et al. 2023 | Serum | 50 | Arsenazo III Colorimetric 3 min |
8.8–10.4 mg/dL LOD: 4.2 mg/dL |
Smartphone-friendly readout. Narrower clinical window and performance constraints; serum-focused workflow. |
| [41] Aguiar et al. 2023 | Saliva | 10 | CPC Colorimetric 10 min |
1.1–18 mg/dL LOD: 0.32 mg/dL |
Low-cost paper microfluidics for noninvasive sampling. Saliva-specific; assay time longer than the proposed platform. |
| [42] Lewińska et al. 2024 | Serum | 10 | OCP Colorimetric 10 min |
0–40 mg/dL LOD: 0.36 mg/dL |
Paper-based multiplexing (Ca²⁺/Mg²⁺). Serum-centric; longer assay time; limited whole-blood handling. |
| [43] Johnson et al. 2025 | Saliva | 6 | LIGMIS Electrochemical 3 min |
0.004–3 mM LOD: 0.005 mM |
Fast electrochemical sensing; portable integration. Electrode drift/calibration and matrix dependence may complicate routine use. |
| [44] Li et al. 2025 | Sweat | – | ISM Potentiometric Real-time |
– LOD: 10 μM |
Real-time wearable monitoring. Not designed for blood/serum; requires long-term stability management. |
| [45] Ostad et al. 2017 | Water | 0 | Xylidyl Blue Colorimetric 10 min |
1–10 mg/dL LOD: 0.83 mg/dL |
Simple paper device for low-complexity samples. Not representative of biofluid matrices; limited translation to whole blood/urine. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




