Submitted:
12 February 2026
Posted:
13 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Details
2.1. Materials and Solution Preparation
2.2. Thin Film Deposition
2.3. Schottky Diode Fabrication
2.4. Characterization Techniques
3. Data Analysis Methods
| Parameter | Selected Value | Purpose / Notes |
| Zinc acetate concentration | 0.5 M | Host precursor concentration |
| Solvent volume | 60 mL ethanol | Sol formation and viscosity control |
| Stabilizers | MEA 2 mL, Citric acid | Chelation; stabilizes sol and controls hydrolysis |
| Dopant concentrations | 0, 1, 3, 5 at.% (Er; Er-Yb) | Study of single and co-doping effects |
| Stirring temperature | 60 °C | Promote dissolution and homogenization |
| Stirring time | 2 h | Ensure complete sol formation |
| Aging time | 48 h | Improve precursor stability and homogeneity |
| Spin speed | 4000 rpm | Achieve uniform film thickness |
| Spin time | 40 s | Control film thickness |
| Preheat (soft-bake) | 350 °C, 10 min | Remove solvents between layers |
| Number of coating cycles | 3 | Achieve target thickness and uniformity |
| Annealing | 500 °C, 1 h (air) | Crystallization and removal of organics |
| Schottky contact | Pd, 100 nm, 0.6 mm diameter | Rectifying top contact |
| Ohmic contact | AuSb, 150 nm | Low-resistance back contact |
| Electrical measurement | HP 4140B, -2 to +2 V | Extraction of I-V device |
4. Results and Discussion
4.1. Structural Analysis (XRD)
4.1.1. Er-Doped ZnO Films
4.1.2. Er/Yb Co-Doped ZnO Films
4.2. Optical Analysis (UV–Vis)
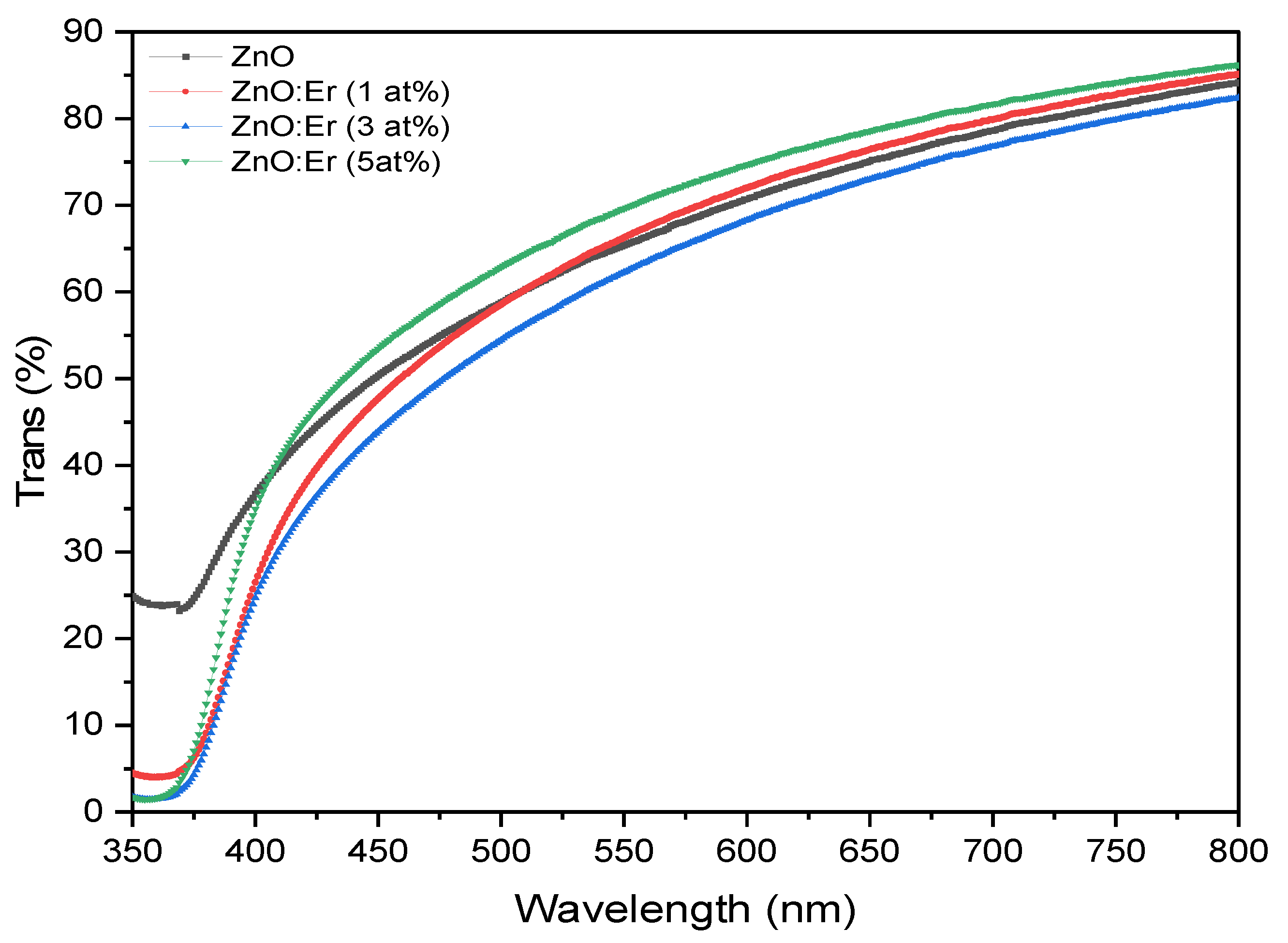
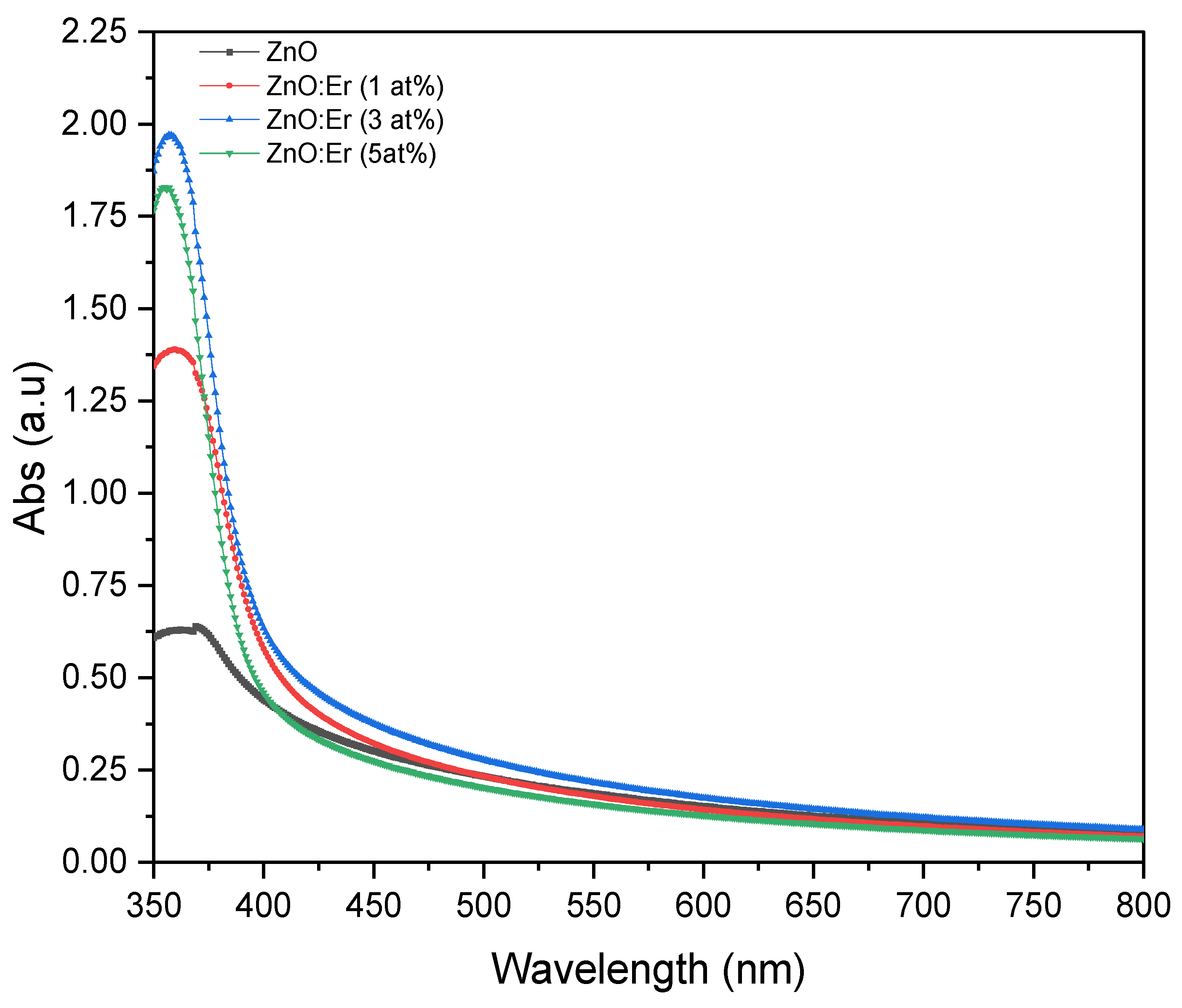
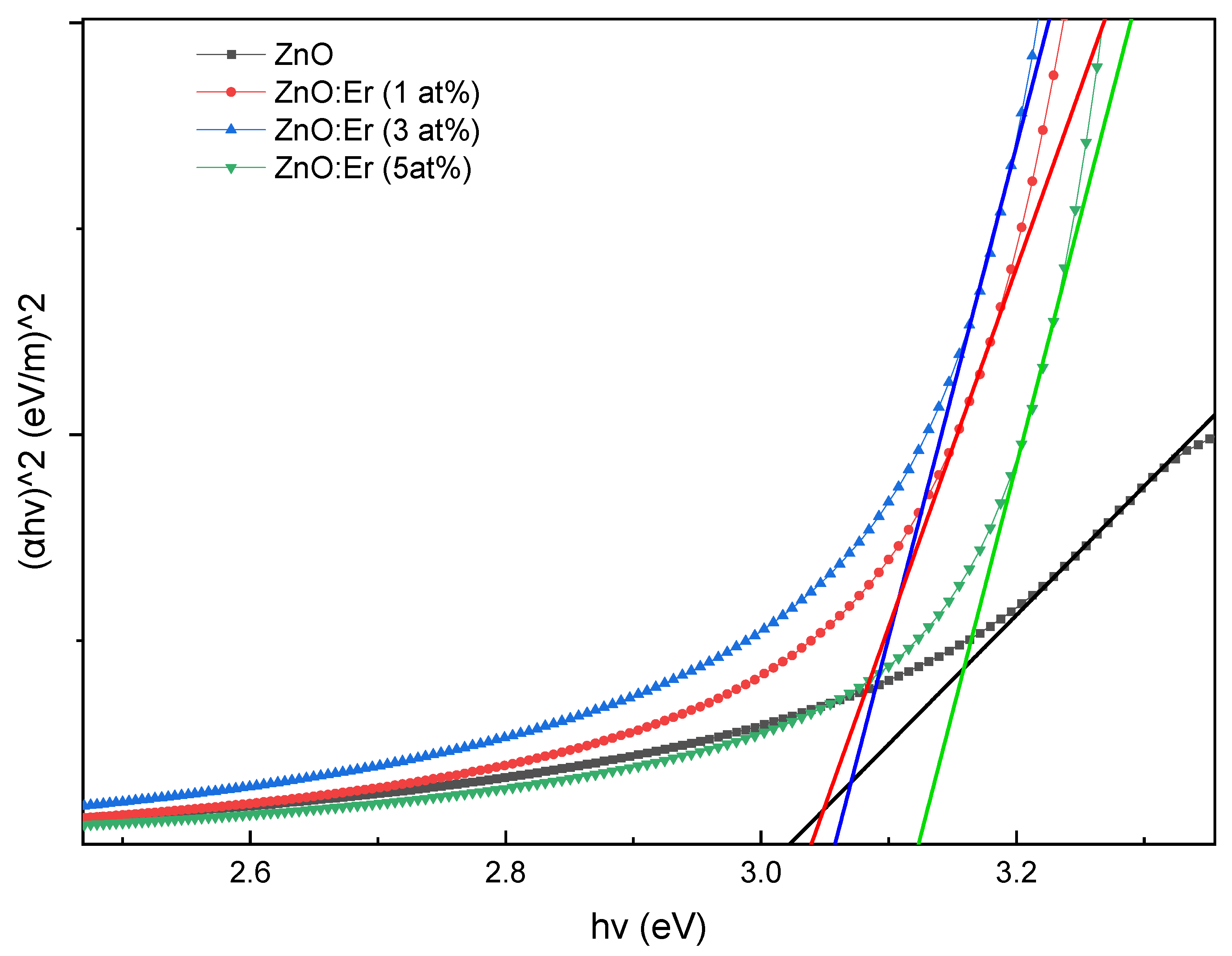
4.2.1. Er-Doped ZnO Films
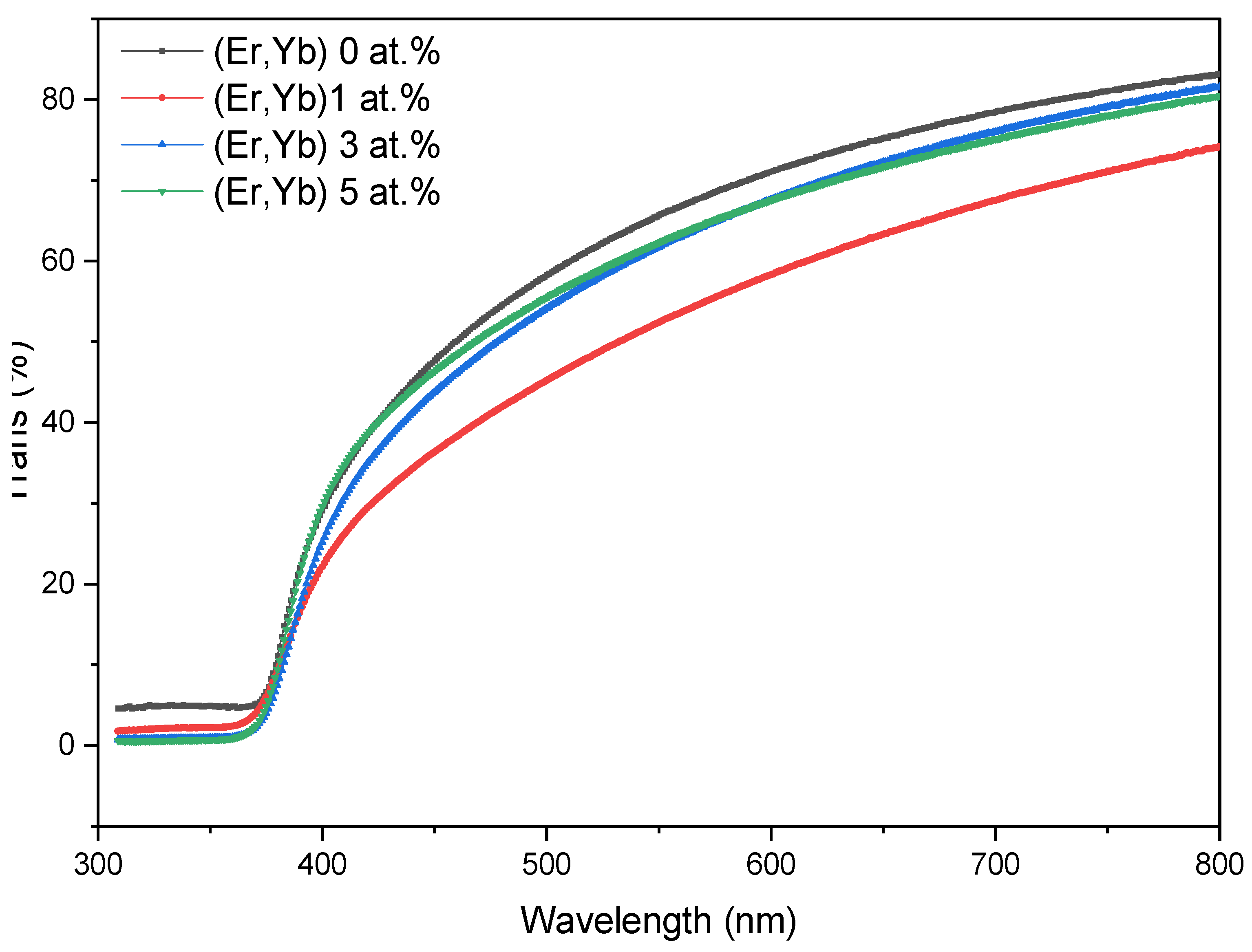
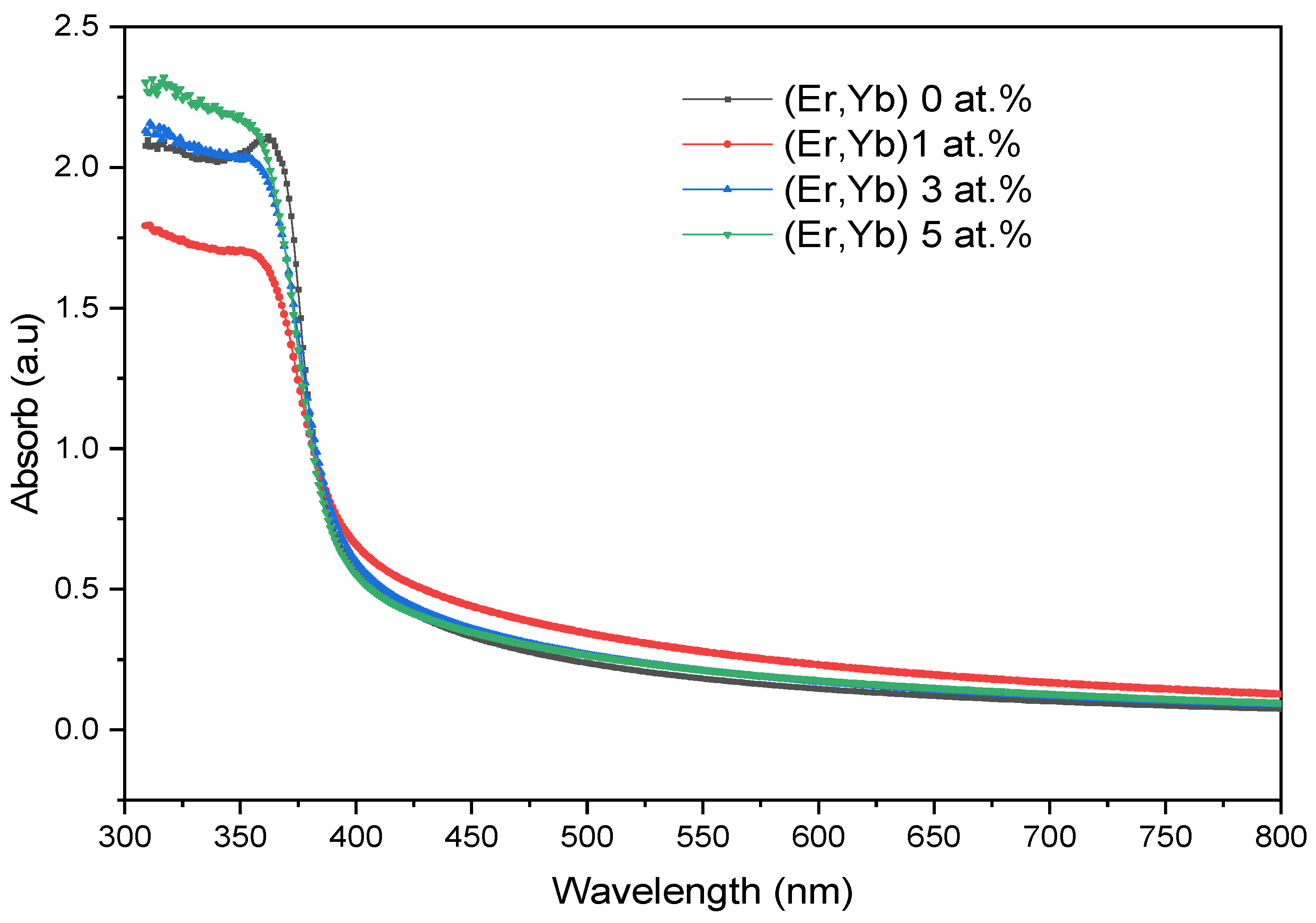
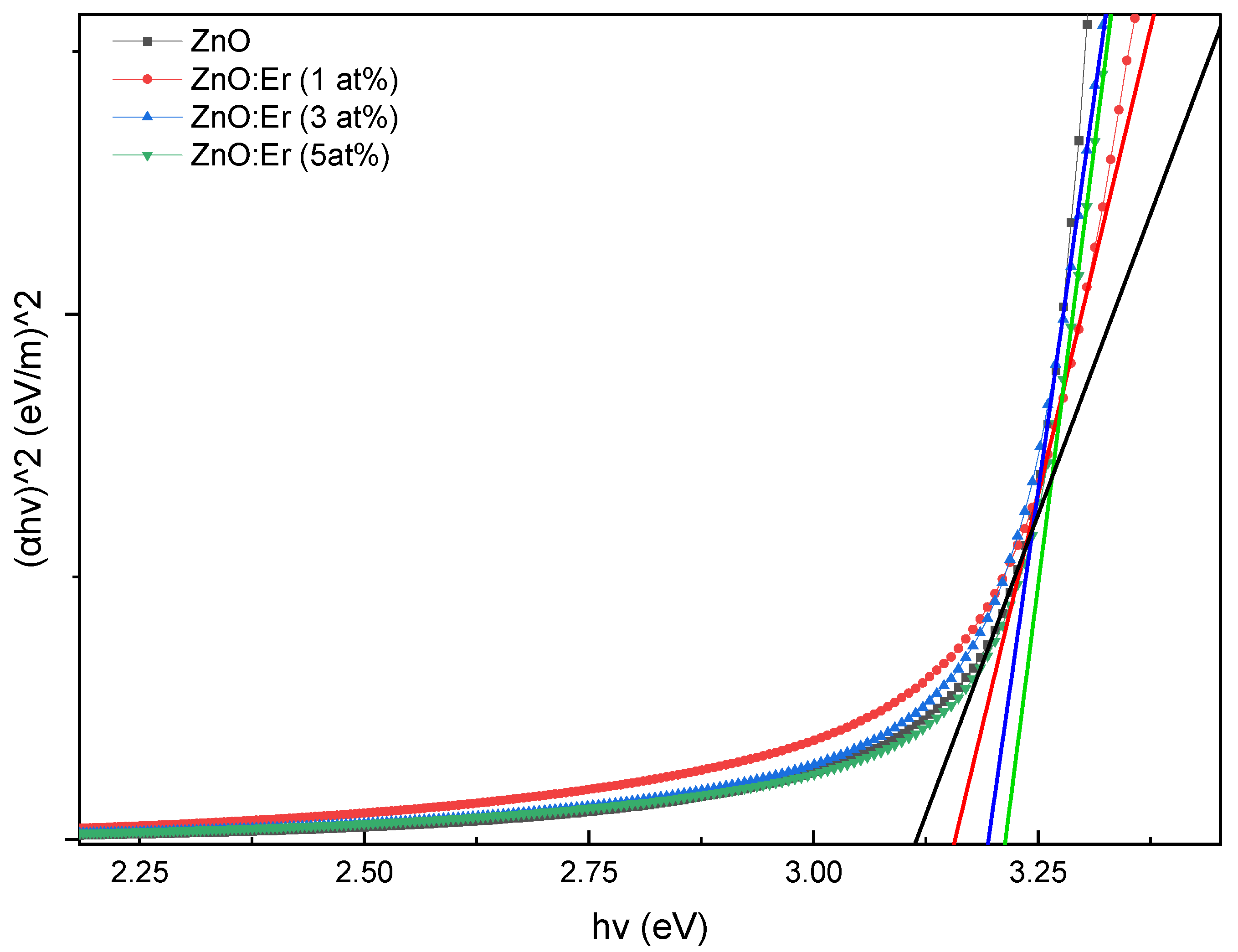
4.2.2. Er/Yb Co-Doped ZnO Films
4.2.3. Bandgap Comparison and Discussion
4.3. Electrical Properties and Schottky Diode Performance
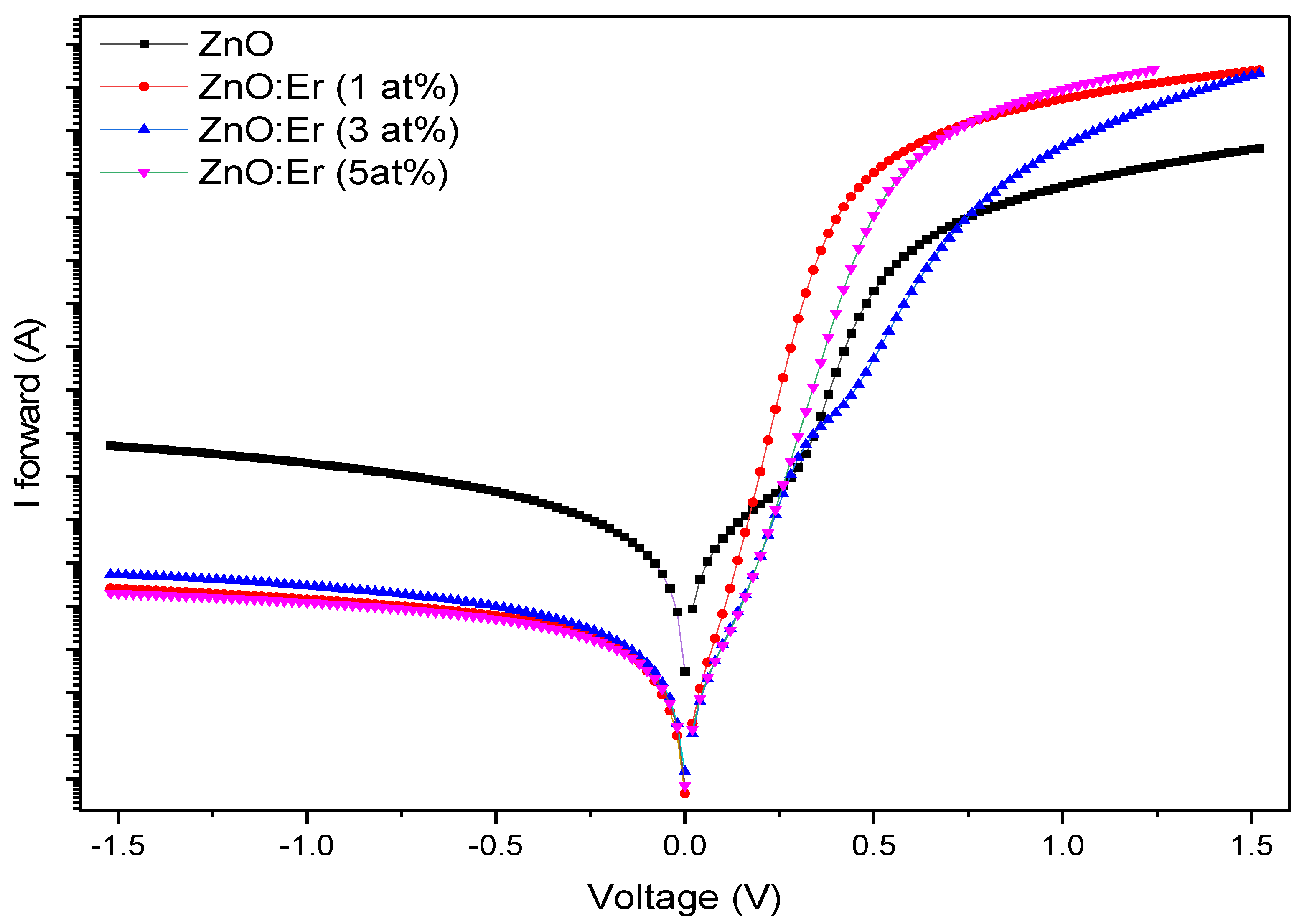
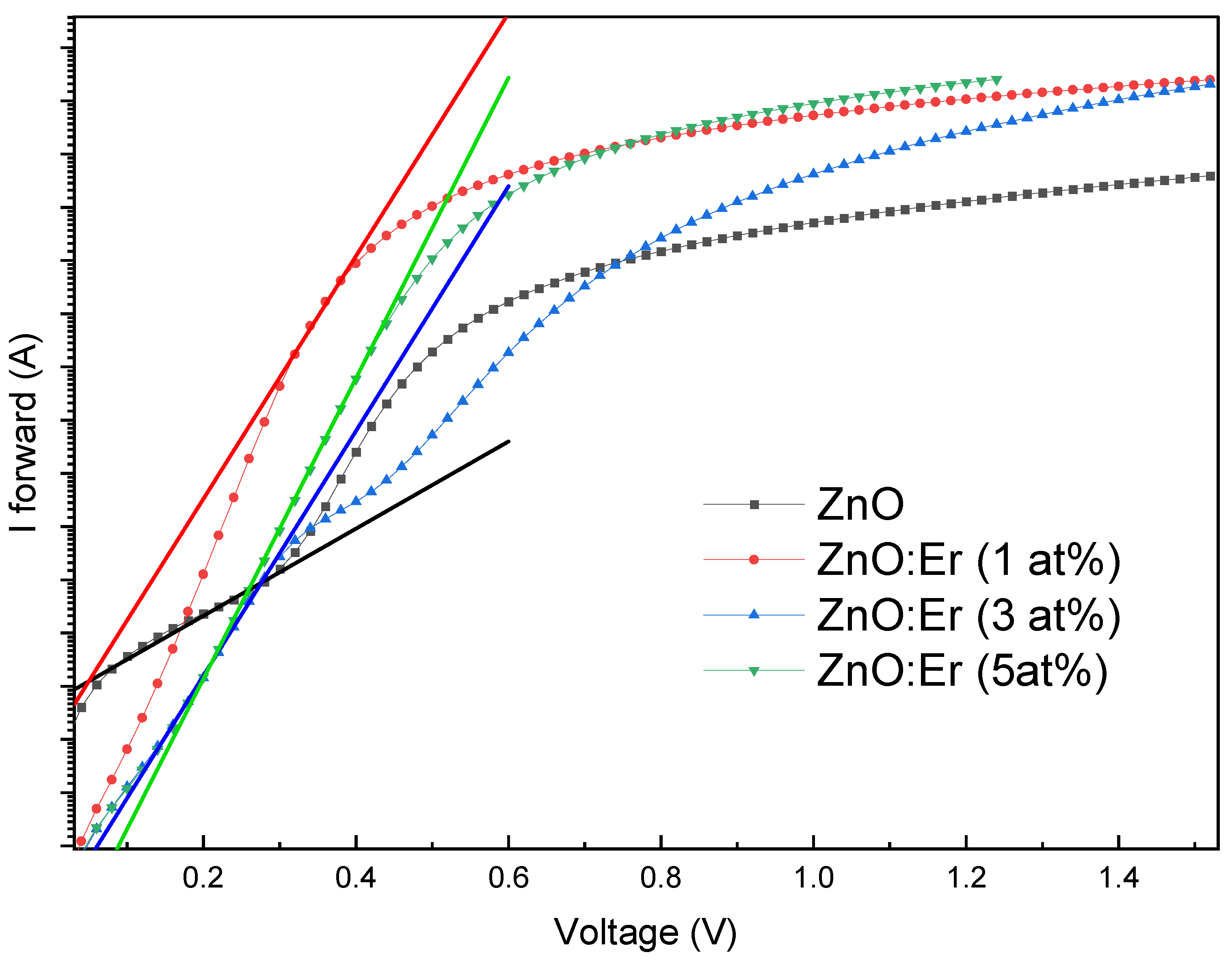
4.3.1. Er-Doped ZnO Schottky Diodes
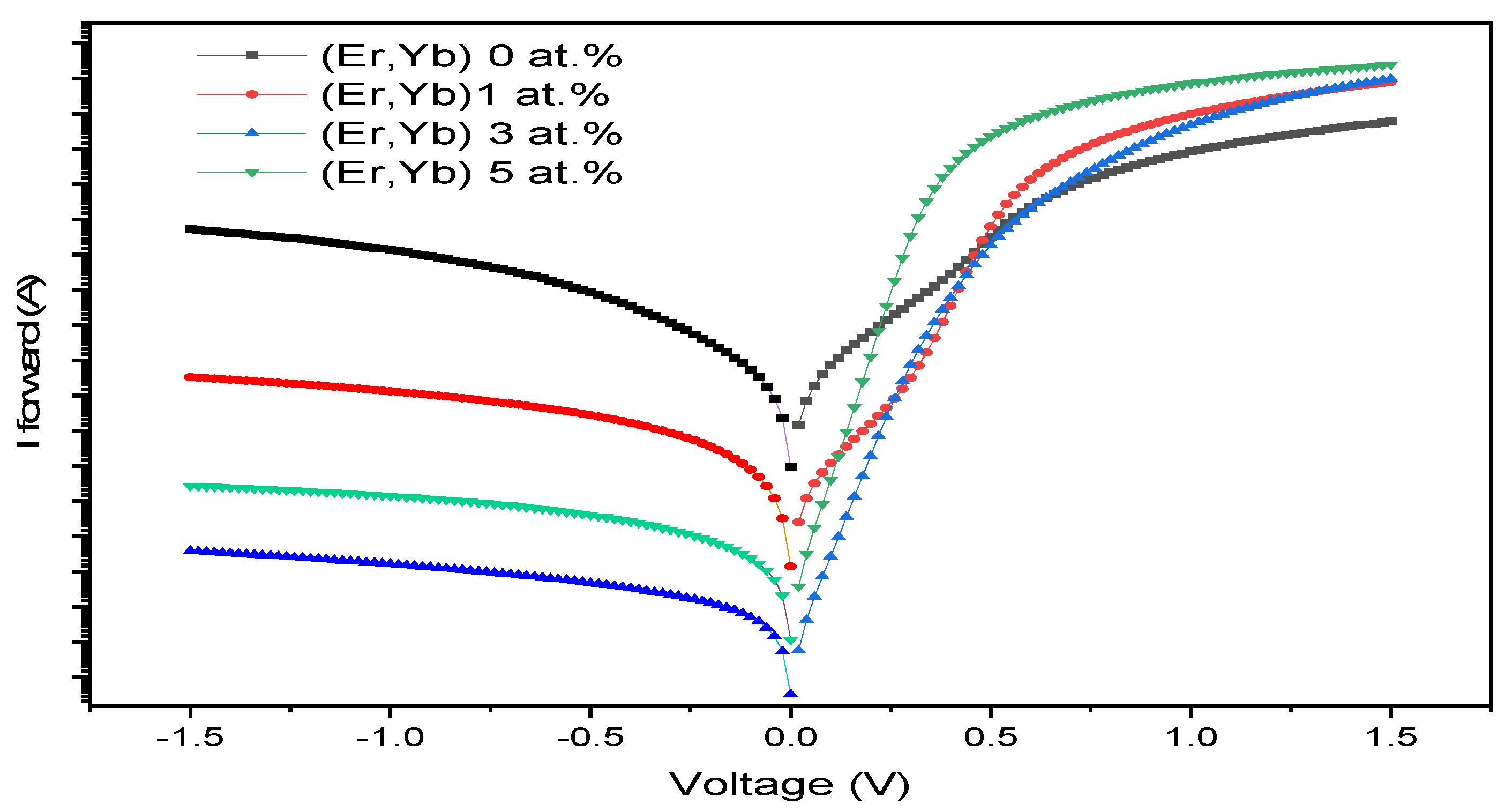
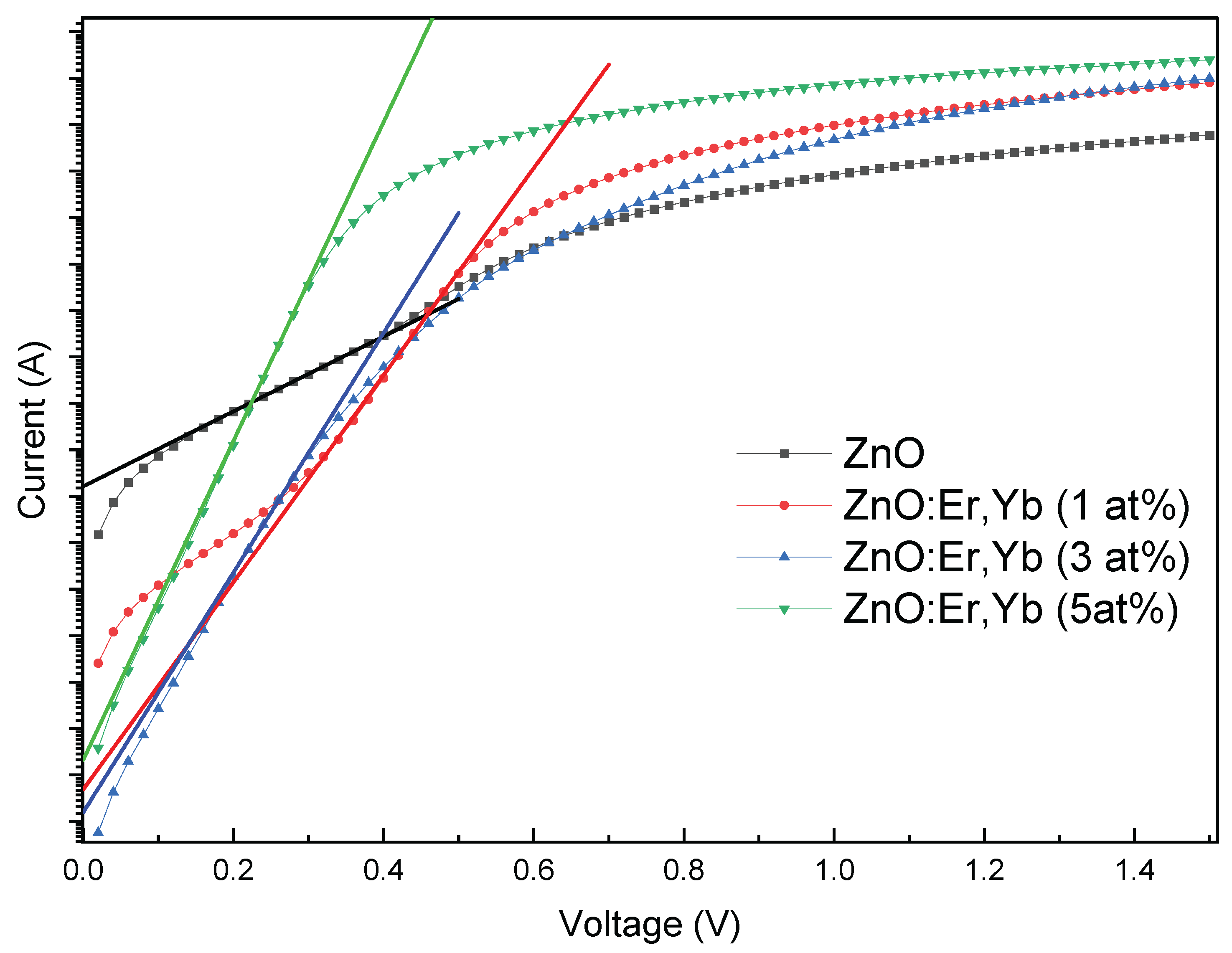
4.3.2. Er/Yb Co-Doped ZnO Schottky Diodes
4.3.3. Discussion of Electrical Performance
5. Conclusion
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tolera, D.; Senbeta, T.S.; Mesfin, B. Optical and temperature dependent electrical properties of Er-/Yb-doped ZnO Schottky diodes and thin films. Sci. Rep. 2025, 15, 1320. [Google Scholar] [CrossRef]
- Zheng, B.; et al. Rare-Earth Doping in Nanostructured Inorganic Materials. Chem. Rev. 2022, 122, 5519–5603. [Google Scholar] [CrossRef]
- Sharma, D.K.; et al. A review on ZnO: Fundamental properties and application. Mater. Today Proc. 2022, 49, 3028–3035. [Google Scholar] [CrossRef]
- Lopez-Mena, E.R.; et al. The effect of Yb doping on the ZnO thin films obtained via a low temperature spin coating method. J. Mater. Sci.: Mater. Electron. 2021, 31, 1–? (article number not provided). [Google Scholar] [CrossRef]
- Murali, K.R.; Subasri, R.; Vijila, M. Enhanced photoluminescence and electrical properties of Er-doped ZnO thin films. Mater. Sci. Semicond. Process. 2014, 18, 97–104. [Google Scholar] [CrossRef]
- Ahmed, M.A.M.; Ahmed, M.E.I. Structural, optical and electrical properties of the fabricated Schottky diodes based on ZnO, Ce and Sm doped and co-doped ZnO films prepared via wet chemical technique. Mater. Sci. Semicond. Process. 2023, 132, 105975. [Google Scholar] [CrossRef]
- Pearton, S.J.; et al. ZnO electronics for UV detection. J. Appl. Phys. 2003, 93, 1–13. [Google Scholar] [CrossRef]
- Ding, J.; Chen, H.; Fu, H. Defect-related photoluminescence emission from annealed ZnO films deposited on AlN substrates. Mater. Res. Bull. 2017, 95, 185–189. [Google Scholar] [CrossRef]
- Jalali, A.; et al. Synthesis of Zinc oxide thin films by sol-gel method and applications to improve the efficiency of silicon solar cells. Chem. Methodol. 2020, 15, 343–356. [Google Scholar] [CrossRef]
- Mouet, T.; et al. Growth and characterization of thin ZnO films deposited on glass substrates by electrodeposition techniques. Appl. Surf. Sci. 2010, 256, 4114–4120. [Google Scholar] [CrossRef]
- Sood, S.; et al. Enhancing Optoelectronic performance through rare-earth doped ZnO: Insights and applications. Photonics 2025, 12, 5454. [Google Scholar] [CrossRef]
- Periyasamy, A.P.; et al. Progress in Sol-Gel Technology for the Coatings of Fabrics. Materials 2020, 13, 1838. [Google Scholar] [CrossRef]
- Ahzan, et al. Synthesis and Characterization of ZnO Thin Layers using Sol-Gel Spin coating method. J. Penelit. Pengkaj. Ilmu Pendidik. 2021, 182–194. [Google Scholar] [CrossRef]
- Asghar, M.; et al. Electrical characterizations of Au/ZnO/Si Schottky contacts. J. Phys.: Conf. Ser. 2013, 439, 012030. [Google Scholar] [CrossRef]
- Tan, S.T.; et al. High-performance Pd/ZnO Schottky diodes. J. Appl. Phys. 2005, 98, 014502. [Google Scholar] [CrossRef]
- Chen, L.F.; et al. Formation of low-resistance AuSb ohmic contacts to n-type Si. IEEE Electron Device Lett. 1990, 11, 462–464. [Google Scholar] [CrossRef]
- Caglar, Y. Effects of rare earth doping on ZnO nanostructures. J. Alloys Compd. 2014, 617, 595–601. [Google Scholar] [CrossRef]
- Sira, K.; et al. Microstructure and optical properties of rare-earth doped ZnO thin films. Mater. Today Proc., 2015. [Google Scholar] [CrossRef]
- Wang, C.; et al. Metal oxide gas sensors: sensitivity and influencing factors. Sensors 2010, 10, 2088–2106. [Google Scholar] [CrossRef] [PubMed]
- Ip, K.P.; Heo, Y.W.; Baik, K.; Norton, D.P. Temperature-dependent characteristics of Pt Schottky contacts doped on n-type ZnO. Appl. Phys. Lett. 2004, 84, 2835–2837. [Google Scholar] [CrossRef]
- Sharma, A.; et al. Citric acid assisted sol–gel synthesis of ZnO for improved optoelectronic properties. J. Sol-Gel Sci. Technol. 2017, 81, 181–188. [Google Scholar] [CrossRef]
- Ghanem, M.G.; et al. Synthesis and characterization of undoped and Er-doped ZnO nano-structure thin films deposited by sol-gel spin coating technique. Mater. Res. Express 2019, 6, 085916. [Google Scholar] [CrossRef]
- Klingshirn, C. ZnO: From basics towards applications. Phys. Status Solidi B 2007, 244, 3027–3073. [Google Scholar] [CrossRef]
- Zamiri, R.; et al. Effects of rare earth (Er, La and Yb) Doping on morphology and structure Properties of ZnO Nanostructures prepared by wet chemical method. Ceram. Int. 2014, 40, 523–529. [Google Scholar] [CrossRef]
- Elgazzar, A.; et al. Impact of Pd and Sn Co-Doping ZnO Nanoflakes toward high performing Schottky Diodes based on the generation of intermediate bands within the energy gap. Mater. Sci.: Mater. Electron. 2024, 35, 13398–3. [Google Scholar] [CrossRef]
- Ye, Z.; et al. Co-Doping: an effective strategy for achieving stable p-type ZnO thin films. Nano Energy 2018, 52, 527–540. [Google Scholar] [CrossRef]
- Brillson, L.; Lu, Y. ZnO Schottky barriers and Ohmic Contacts. Appl. Phys. Rev. 2011, 109, 121301. [Google Scholar] [CrossRef]
- Ipek, Y.; et al. Rare earth element doped ZnO thin films and Applications. Int. J. Pure Appl. Phys. 2021, 7, 305–313. [Google Scholar] [CrossRef]
- Shen, M.; et al. Optimization of Copper Schottky Contacts on nanocrystalline ZnO thin films by atomic layer deposition. MRS Adv. 2016, 1, 3421–3427. [Google Scholar] [CrossRef]
- Mi, W.; et al. Study of the metal-semiconductor contact to ZnO films. Vacuum 2018, 155, 210–213. [Google Scholar] [CrossRef]
- Reisfeld, R.; Saraidarov, T. Innovative Materials based on sol gel technology. Opt. Mater. 2006, 28, 64–70. [Google Scholar] [CrossRef]
- Gartner, M.; et al. Towards advanced materials: Functional perspectives of co-doped ZnO thin films. Micromachines 2025, 16, 1179. [Google Scholar] [CrossRef] [PubMed]
- Ali, G.M.; et al. Interface properties and junction behavior of Pd contact on ZnO thin film grown by vacuum deposition technique. Phys. Status Solidi C 2010, 7, 252–255. [Google Scholar] [CrossRef]
- Boukhari, A.; et al. Thickness effect on the properties of 4% Mn-doped ZnO thin films grown by sol gel spin coating deposition. Macromol. Symp. 2021, 397, 1. [Google Scholar] [CrossRef]
- Mahalakshmi, S.; et al. Ytterbium-doped ZnO thin films for visible light photocatalysis. Ceram. Int. 2018, 44, 7777–7783. [Google Scholar] [CrossRef]
- Tsiarapas, C.; Girginoudi, D.; Nikolaos, N. Electrical characteristics of Pd Schottky contacts on ZnO thin films. Mater. Sci. Semicond. Process. 2014, 17, 199–206. [Google Scholar] [CrossRef]
- EI Hat, A.; et al. Growth and properties of (Yb-Er) Co-Doped ZnO thin films deposited via spray pyrolysis technique. Optics 2025, 6, 14. [Google Scholar] [CrossRef]
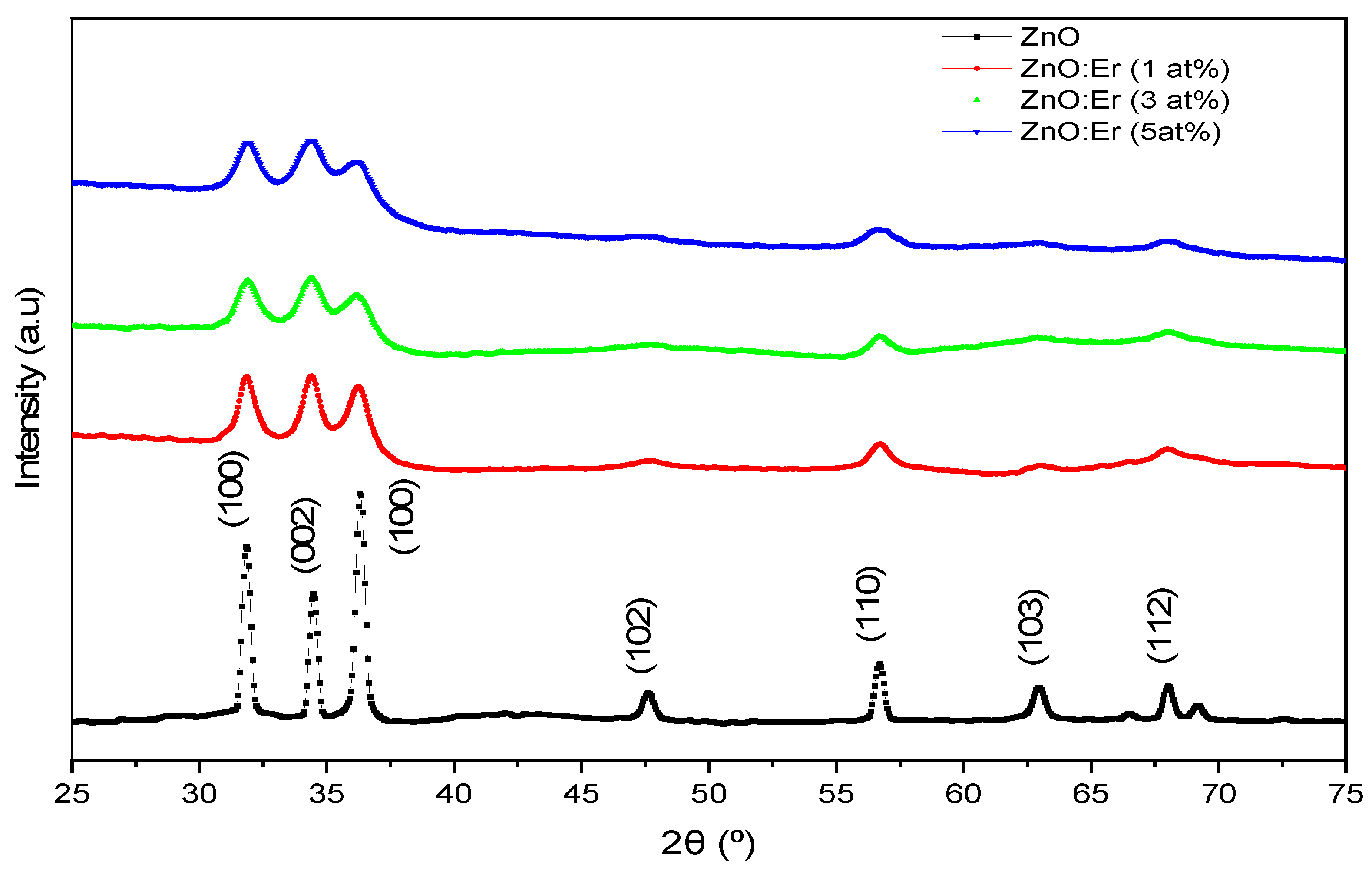
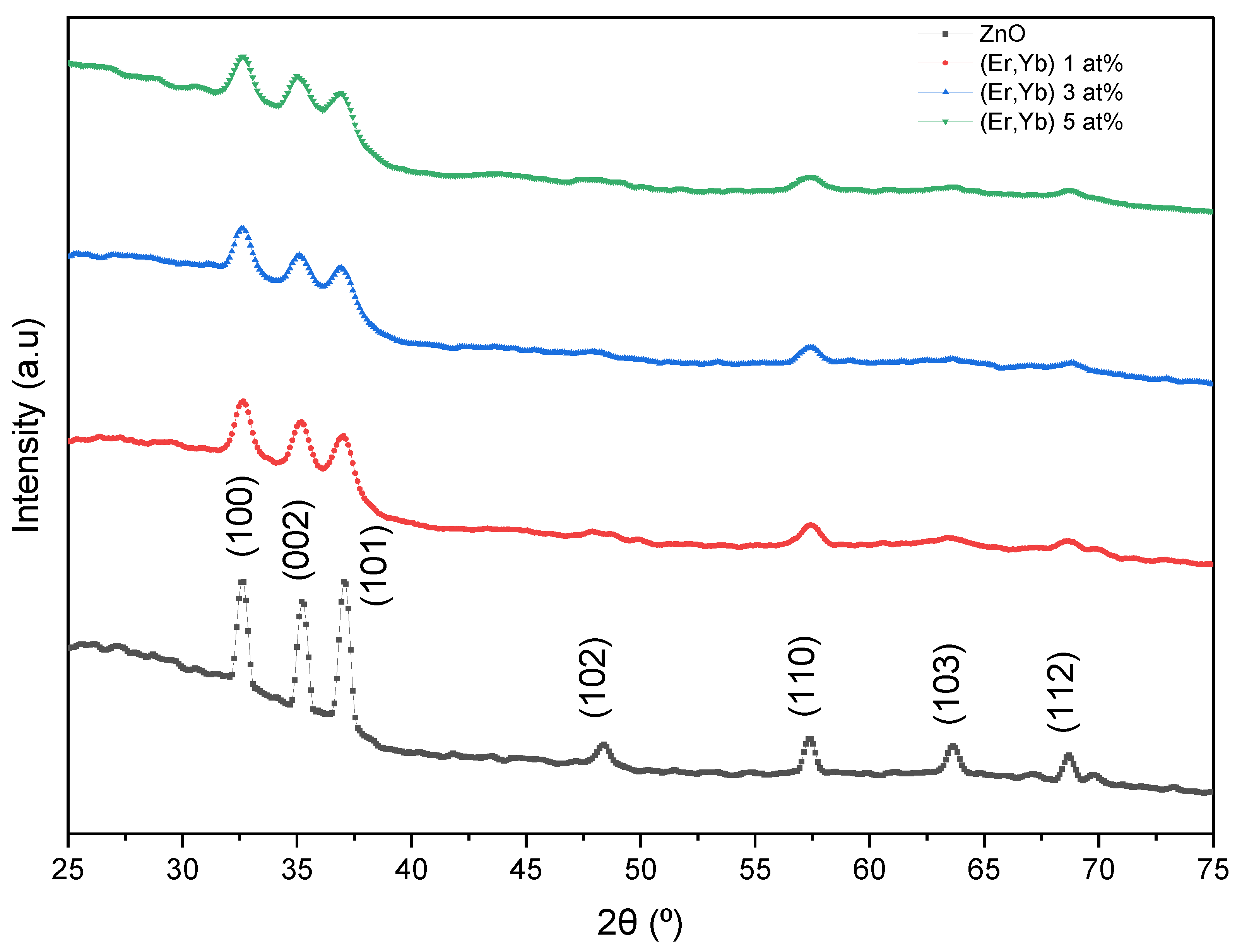
| Sample | Doping (at.%) | 2θ (°) | FWHM (°) | TC(002) | D (nm) | δ (1012 cm−2) | ε (10−3) | a (Å) | c (Å) |
| ZnO:Er | 0 | 34.47 | 0.368 | 1.226 | 22.6 | 0.20 | 5.18 | 3.243 | 5.200 |
| 1 | 34.31 | 1.487 | 1.164 | 5.6 | 3.20 | 21.0 | 3.234 | 5.223 | |
| 3 | 34.37 | 1.236 | 1.294 | 6.7 | 2.21 | 17.4 | 3.296 | 5.214 | |
| 5 | 34.34 | 1.461 | 1.164 | 5.7 | 3.09 | 20.6 | 3.245 | 5.219 | |
| ZnO:(Er-Yb) | 0 | 35.24 | 0.480 | 1.121 | 17.4 | 0.33 | 6.59 | 3.167 | 5.089 |
| 1 | 35.21 | 0.897 | 1.146 | 9.3 | 1.16 | 12.3 | 3.165 | 5.094 | |
| 3 | 35.12 | 1.616 | 1.145 | 5.2 | 3.76 | 22.3 | 3.161 | 5.106 | |
| 5 | 34.97 | 1.668 | 1.147 | 5.0 | 4.01 | 23.1 | 3.155 | 5.128 |
| Sample | Doping (at.%) | Avg. Trans (%) | Absorp. Edge (nm) | Bandgap (eV) |
| ZnO:Er | 0 | 67.80 | 410 | 3.023 |
| 1 | 67.74 | 403 | 3.040 | |
| 3 | 64.28 | 401 | 3.059 | |
| 5 | 70.99 | 393 | 3.125 | |
| ZnO:(Er, Yb) | 0 | 66.99 | 403 | 3.113 |
| 1 | 55.51 | 383 | 3.156 | |
| 3 | 63.82 | 386 | 3.195 | |
| 5 | 64.25 | 390 | 3.214 |
| Sample | Doping (at.%) | Ideality Fac. (n) | Saturation Current (A) | B. Height (eV) | Resistance Rs (Ω) | L. Current @ -1.0 V | Rect. Ratio @ ±1.0 V |
| ZnO:Er | 0 | 4.77 | 8.34×10−8 | 0.655 | 512 | 6.02×10−5 | 1.4×101 |
| 1 | 1.70 | 3.98×10−8 | 0.674 | 108 | 1.12×10−6 | 2.1×103 | |
| 3 | 1.68 | 1.38×10−9 | 0.761 | 105 | 8.45×10−9 | 2.3×105 | |
| 5 | 1.38 | 4.60×10−10 | 0.789 | 66 | 5.92×10−8 | 1.1×105 |
| Sample | Doping (at.%) | Ideality Factor (n) | Barrier Height ΦB (eV) | Series Resistance Rs (Ω) | Leakage Current @ -1.0 V (A) | Rectification Ratio @ ±1.0 V |
| ZnO:Er:Yb | 0 | 4.71 | 0.595 | 386 | 5.23×10−5 | 1.6×101 |
| 1 | 1.73 | 0.758 | 225 | 9.36×10−7 | 2.6×103 | |
| 3 | 1.50 | 0.771 | 183 | 6.94×10−9 | 2.6×105 | |
| 5 | 1.13 | 0.743 | 113 | 4.81×10−8 | 1.2×105 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




