1. Introduction
Lung cancer remains a leading cause of cancer-related mortality worldwide, largely because the disease is frequently diagnosed at an advanced stage, when curative treatment options are limited [
1]. Low-dose computed tomography (LDCT) screening in high-risk individuals has been shown to reduce lung cancer mortality by enabling earlier detection and treatment [
2,
3]. However, the real-world effectiveness of LDCT screening programs depends not only on imaging-based detection, but also on timely and complete downstream diagnostic pathway following a positive screening result [
4].
A positive LDCT finding does not necessarily indicate malignancy, as pulmonary nodules are common in high-risk populations [
5]. To balance early cancer detection against unnecessary procedures, standardized management algorithms such as Lung-RADS guide post-screening decision-making [
6]. Nevertheless, abnormal screening results, particularly higher-risk findings, often prompt multidisciplinary evaluation and further diagnostic work-up, including invasive procedures aimed at obtaining tissue confirmation. These procedures are essential for definitive diagnosis and treatment planning, but they also carry procedural risks, logistical demands, and challenge for healthcare system capacity [
7].
In real-world screening programs, a substantial proportion of individuals with abnormal LDCT findings do not complete recommended diagnostic evaluation [
8,
9]. Most prior studies describe this phenomenon broadly as loss to follow-up, without clearly distinguishing between delayed evaluation, system-related barriers, and explicit refusal of invasive diagnostic procedures [
10]. This lack of granularity limits understanding of where and why post-screening pathways fail in routine practice and constrain the development of targeted implementation strategies [
11].
Refusal of invasive diagnostic procedures represents a distinct and clinically relevant post-screening outcome, as it occurs after individuals have already engaged with screening and received a recommendation for further evaluation [
12]. Such refusal may be influenced by non-medical factors, including risk perception, prior healthcare experiences, communication quality, and organizational or geographic barriers to care [
13]. Importantly, refusal should not be viewed merely as an individual decision, but rather as the result of interactions between patients and healthcare systems [
14].
Understanding refusal of invasive diagnostics is particularly relevant in centralized screening models, where invasive diagnostic procedures are frequently performed at tertiary referral centers [
15]. In such settings, travel distance, coordination complexity, and system organization may affect diagnostic continuity and ultimately influence the overall effectiveness of screening programs [
16,
17].
The aim of this study was to characterize post-screening diagnostic pathways following positive LDCT findings within an organized lung cancer screening program, with a specific focus on refusal of recommended invasive diagnostic procedures. We further aimed to identify patient-related and system-related factors associated with refusal, providing real-world evidence to inform optimization of LDCT screening implementation.
2. Materials and Methods
2.1. Study Design and Setting
This retrospective observational study was conducted within an organized low-dose computed tomography (LDCT) lung cancer screening program in the Autonomous Province of Vojvodina, Serbia. The screening program was initiated on 20 September 2020 at the Institute for Pulmonary Diseases of Vojvodina (Novi Sad) and was subsequently expanded to additional screening sites in Subotica and Vrbas. All screening sites operated under a unified protocol for eligibility assessment, LDCT acquisition, structured reporting, and referral for further diagnostic evaluation.
The present analysis included consecutive participants screened between 20 September 2020 and 1 October 2025. Screening outcomes and downstream diagnostic events were assessed according to standardized post-screening diagnostic pathways routinely applied within the program.
2.2. Participants and Eligibility Criteria
Participants were eligible for inclusion in the LDCT screening program if they were aged 50–74 years at the time of screening, had no clinical signs or symptoms suggestive of lung cancer, and met predefined smoking exposure criteria. Eligible participants included current smokers with a cumulative smoking exposure of ≥30 pack-years, or ≥20 pack-years in the presence of additional lung cancer risk factors, such as chronic respiratory diseases (chronic obstructive pulmonary diseases, sarcoidosis), previous pneumonia (three or more confirmed infections in a one year period), prior malignancy other than lung cancer, a family history of lung cancer, or occupational exposure to carcinogens. Former smokers who had quit smoking within the preceding 10 years were also eligible, provided they met the same cumulative smoking exposure thresholds.
Participants were excluded from the screening program and from the present analysis if they had undergone chest computed tomography within the previous 12 months, had a prior diagnosis of lung cancer, were receiving long-term oxygen therapy, or had advanced comorbid conditions limiting life expectancy or the potential benefit of screening. Such conditions included advanced liver disease, severe chronic obstructive pulmonary disease with hypoventilation, or New York Heart Association class IV heart failure.
2.3. LDCT Acquisition and Image Interpretation
LDCT examinations were performed using site-specific multidetector CT scanners, following harmonized low-dose acquisition protocols designed to minimize radiation exposure while preserving diagnostic image quality. All LDCT scans were interpreted by radiologists experienced in thoracic imaging.
Structured reporting was applied using Lung-RADS version 2021, with standardized categorization and corresponding management recommendations assigned to each screening examination.
2.4. Definition of Screening Outcomes
A positive screening result was defined as Lung-RADS category 4A, 4B, or 4X, in accordance with Lung-RADS version 2021 recommendations. Lung-RADS category 3 was considered a suspicious finding requiring short-term imaging follow-up but was not classified as a positive screening result for the purposes of this analysis.
2.5. Post-Screening Diagnostic Pathway
Participants with a positive LDCT screening result entered a structured post-screening diagnostic pathway coordinated by a multidisciplinary team. Further evaluation could include follow-up imaging, positron emission tomography/computed tomography (PET/CT) when clinically indicated, and invasive diagnostic procedures aimed at obtaining tissue or cytological confirmation.
Diagnostic strategies were individualized based on lesion characteristics, anatomical accessibility, and clinical considerations. In general, bronchoscopic procedures were considered the first-line invasive diagnostic approach for lesions accessible via the bronchial tree, while transthoracic needle biopsy or surgical diagnostic procedures were employed when required related to lesion location or diagnostic feasibility. All diagnostic decisions were made within a multidisciplinary team framework, reflecting routine clinical practice rather than protocol-mandated pathways.
2.6. Invasive Diagnostic Procedures and Definition of Refusal
Invasive diagnostic work-up was defined as any procedure performed with the intent of obtaining histological or cytological sample for pathological confirmation. These procedures included bronchoscopic techniques (with or without endobronchial ultrasound guidance), transthoracic needle aspiration or biopsy (TTNA), and surgical diagnostic procedures.
Refusal of invasive diagnostic procedures was defined as an explicit decision by a participant to decline a recommended invasive diagnostic intervention following multidisciplinary team referral. Such decisions were documented in medical records and/or screening pathway documentation. Operationally, refusal was defined as the absence of any invasive diagnostic procedure within six months following the initial recommendation. Cases in which invasive procedures were postponed or deferred due to medical contraindications, ongoing clinical evaluation, or other physician-driven reasons were not classified as refusal.
2.7. Data Sources and Variables
Data were collected from LDCT screening registries, radiology reports, and institutional clinical records. Extracted variables included demographic characteristics (age and gender); sociodemographic indicators (education level, marital status, employment status, and place of residence categorized as rural or urban); screening site location; and smoking-related variables (current smoking status and cumulative smoking exposure expressed in pack-years).
Clinical variables included body mass index (BMI), comorbidities, multimorbidity status, history of prior malignancy, and self-reported concern regarding personal lung cancer risk. Sociodemographic characteristics were included to explore potential social and contextual influences on post-screening diagnostic decision-making. Geographic accessibility was approximated using the screening site as a proxy indicator of travel burden to the diagnostic center. Procedure-related complications and mortality were identified through review of institutional clinical records during post-procedural follow-up.
2.8. Study Outcomes
The primary study outcomes were the positive screening rate, the invasive diagnostic rate among participants with positive LDCT findings, and the refusal rate of recommended invasive diagnostic procedures. Secondary outcomes included timing of invasive diagnostic procedures, histopathological diagnoses, procedure-related complications, and mortality.
For time-to-event analyses, invasive diagnostic or therapeutic procedures occurring more than 300 days after the index LDCT examination were excluded, as such delays were considered unlikely to reflect the screening-related diagnostic pathway.
2.9. Statistical Analysis
Descriptive statistics were used to summarize participant characteristics and screening outcomes. Categorical variables were presented as counts and percentages, while continuous variables were summarized as mean ± standard deviation or median with interquartile range, as appropriate.
Time-to-procedure analyses for bronchoscopy and surgical interventions were performed among participants with positive LDCT findings. Comparisons of time intervals across screening centers and screening years were conducted using the Kruskal–Wallis test. Time-to-event analyses were restricted to procedures occurring within 300 days following the index LDCT examination.
Group comparisons were performed using the χ² test or Fisher’s exact test for categorical variables and appropriate parametric or non-parametric tests for continuous variables. Factors associated with refusal of invasive diagnostic procedures were explored using multivariable logistic regression analysis, with results presented as odds ratios (ORs) and 95% confidence intervals. A two-sided p-value < 0.05 was considered statistically significant.
All statistical analyses were performed using IBM SPSS Statistics (IBM Corp., Armonk, NY, USA).
2.10. Ethics Approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Institute for Pulmonary Diseases of Vojvodina, Sremska Kamenica, Serbia (approval number: No110-V/1). Written informed consent for participation in the LDCT screening program and for the use of anonymized data for research purposes was obtained from all participants.
2. Results
2.1. Study Population
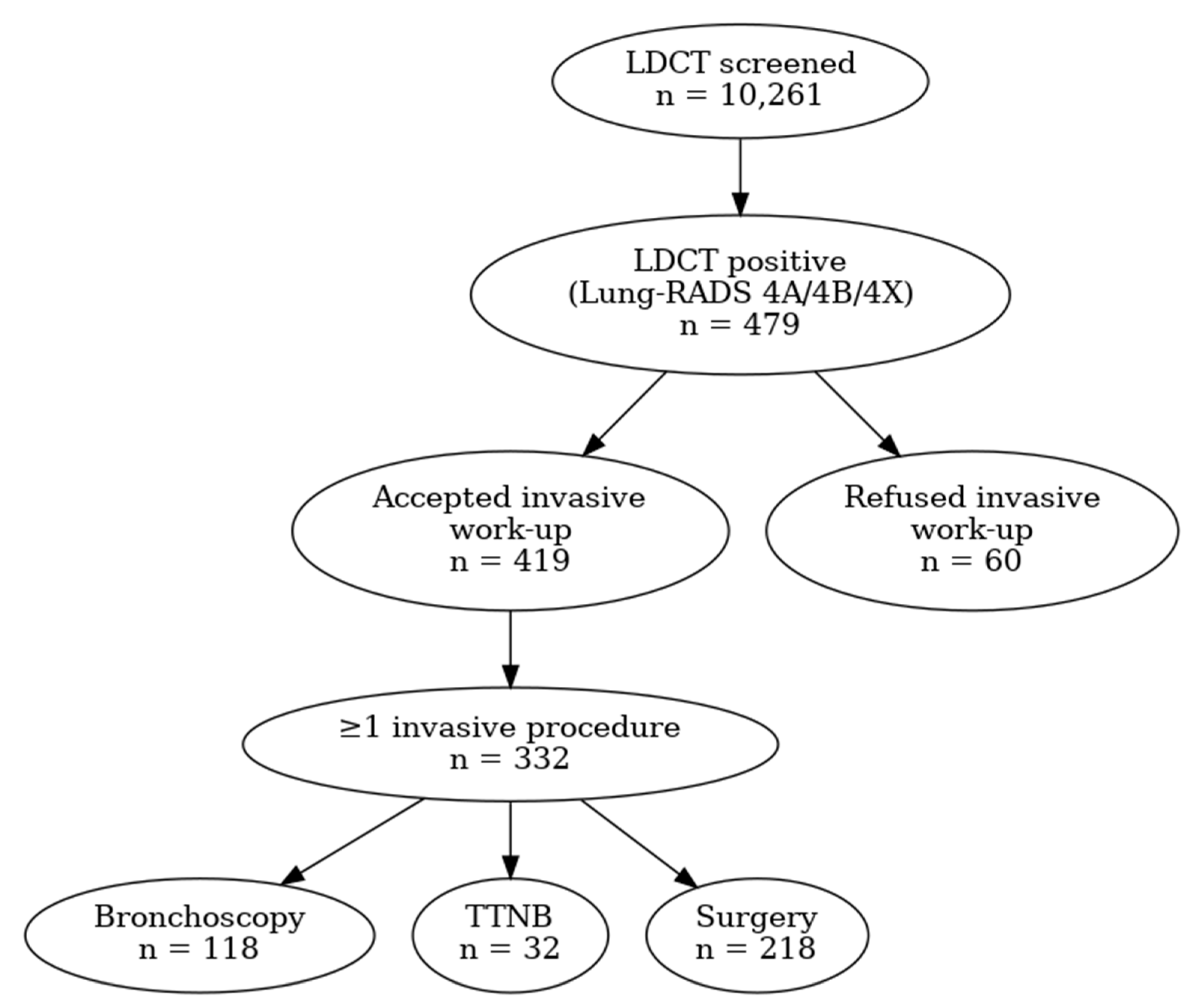
Between 1 October 2020 and 1 October 2025, a total of 10,261 individuals underwent low-dose computed tomography (LDCT) lung cancer screening at three screening sites (Novi Sad, Subotica, and Vrbas). Among the screened population, 479 individuals (4.7%) had positive LDCT findings classified as Lung-RADS 4A, 4B, or 4X and were included in the present analysis (
Figure 1).
The diagram illustrates the number of screened individuals, participants with positive LDCT findings (Lung-RADS 4A/4B/4X), acceptance and refusal of recommended invasive diagnostic procedures, and subsequent diagnostic and treatment steps. Participants may have undergone more than one invasive diagnostic procedure.
2.2. Diagnostic Pathway Following Positive LDCT Findings
Of the 479 LDCT-positive individuals, 419 (87.5%) proceeded with recommended invasive diagnostic evaluation, while 60 (12.5%) refused to undergo any invasive diagnostic procedure within six months following a multidisciplinary team (MDT) recommendation.
Overall, 332 patients (69.3%) underwent at least one invasive diagnostic procedure during follow-up. The distribution of invasive diagnostic procedures and procedure-related safety outcomes is summarized in
Table 1.
2.4. Timing of Invasive Diagnostic Procedures
Time intervals between LDCT screening and subsequent invasive diagnostic procedures were calculated for bronchoscopy and surgical interventions. All time-to-procedure analyses were restricted to a maximum follow-up of 300 days following LDCT screening. Overall, invasive diagnostic procedures were performed within clinically acceptable time frames, with variability observed across procedure types, screening centers, and screening years.
2.4.1. Time from LDCT to First Bronchoscopy
Among participants with positive LDCT findings who underwent bronchoscopy, the overall median time from LDCT screening to first bronchoscopy was 36.0 days (interquartile range [IQR] 25.0–47.0), with a mean of 42.9 ± 33.0 days (range 0–224 days;
n = 117) (
Table S1).
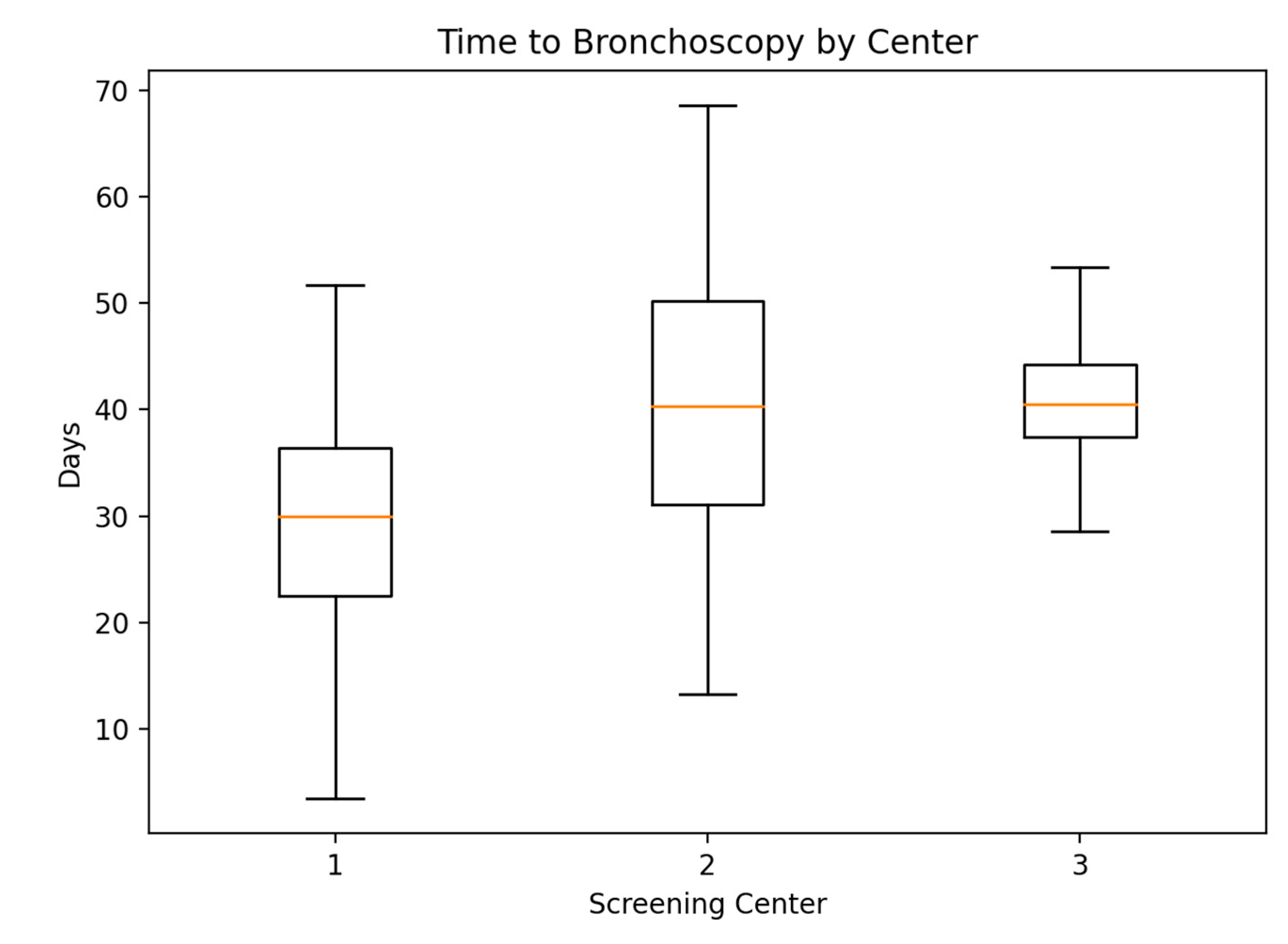
When stratified by screening center, statistically significant differences in time to bronchoscopy were observed (Kruskal–Wallis test:
H = 10.34,
p = 0.0057). Median time to bronchoscopy was shortest in Center 1 (29.0 days; IQR 20.0–42.0), compared with Center 2 (40.0 days; IQR 29.0–49.0) and Center 3 (41.0 days; IQR 35.0–44.0), although interquartile ranges partially overlapped (
Table S2;
Figure 2).
Analysis by LDCT screening year also demonstrated significant variation in time to bronchoscopy (Kruskal–Wallis test:
H = 14.65,
p = 0.012). Median time increased from 7.0 days in 2020 to values ranging between 35.0 and 40.0 days in subsequent years, with greater variability observed from 2022 onward (
Table S3). Despite these fluctuations, all observed time intervals remained within the predefined maximum follow-up period of 300 days.
2.4.2. Time from LDCT to Surgical Treatment
Overall, 223 participants underwent surgical treatment within 300 days following LDCT screening. The median time from LDCT screening to surgery was 52.0 days (IQR 36.5–76.5), with a mean of 63.4 ± 43.7 days (range 7–296 days) (
Table S4).
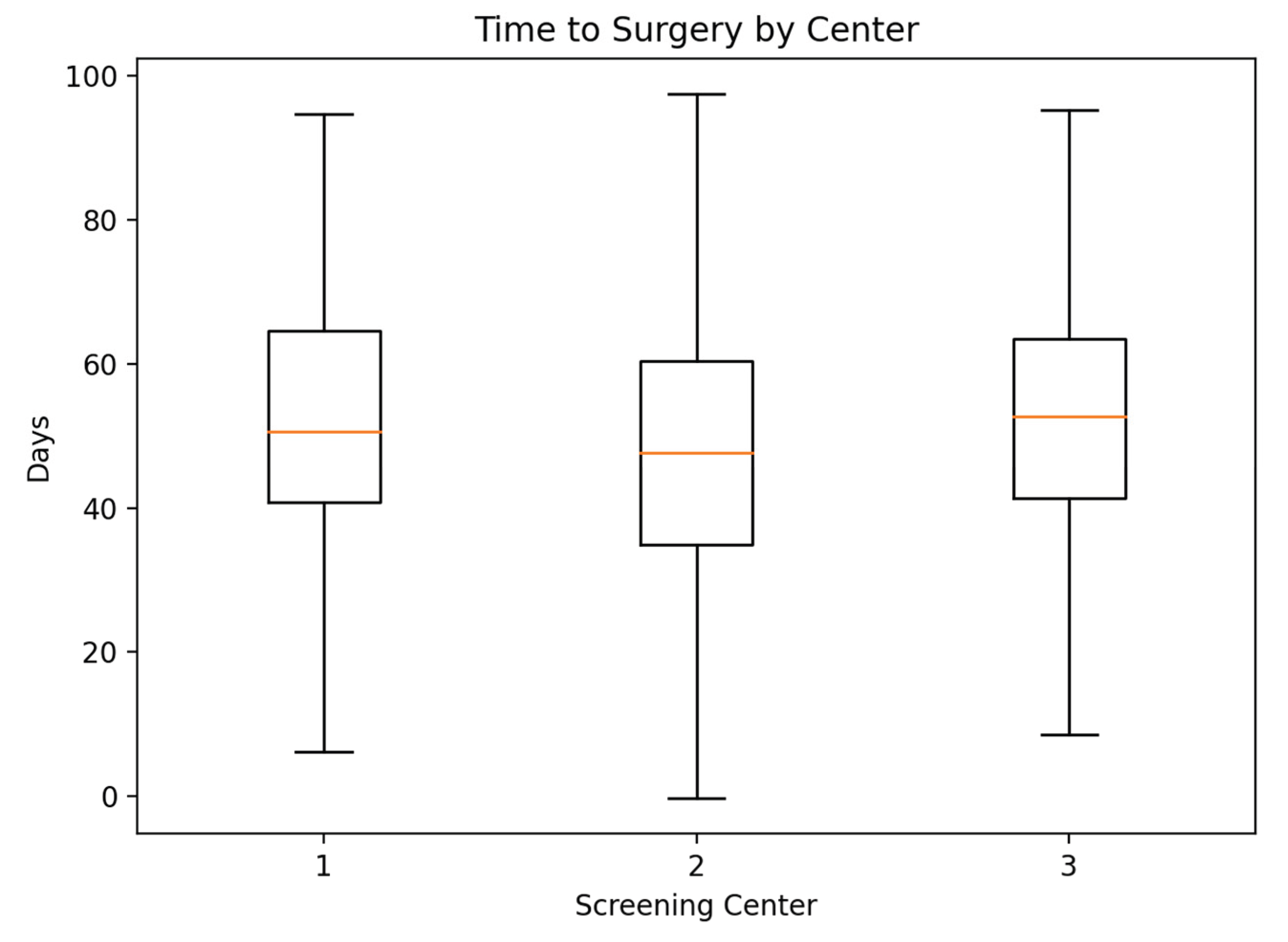
When analyzed by screening center, no statistically significant differences in time to surgery were observed (Kruskal–Wallis test:
H = 1.04,
p = 0.594). Median times to surgery were comparable across centers, ranging from 49.0 to 55.0 days, with overlapping interquartile ranges (
Table S5;
Figure 3).
Similarly, time from LDCT to surgery did not differ significantly across LDCT screening years (Kruskal–Wallis test: H = 9.59, p = 0.0877). Although some variability and a gradual increase in median time were observed in later years, these differences did not reach statistical significance within the 0–300 day interval (
Table S6).
2.5. Comparison of Individuals Who Refused Versus Accepted Invasive Diagnostic Procedures
Baseline demographic, clinical, and perceptual characteristics of individuals who refused invasive diagnostic procedures compared with those who accepted diagnostic work-up are summarized in
Table 2.
Individuals who refused invasive diagnostics had significantly higher cumulative smoking exposure, a higher prevalence of multimorbidity, and a higher prevalence of previous malignant disease. Refusal rates also differed significantly across screening sites and levels of perceived lung cancer risk. No statistically significant differences were observed between groups with respect to age, sex, body mass index, education level, employment status, living arrangement, or current smoking status.
2.6. Multivariable Analysis of Refusal of Invasive Diagnostic Procedures
In multivariable logistic regression analysis, multimorbidity, previous malignancy, cumulative smoking exposure (pack-years), screening site, and concern about personal lung cancer risk were included in the model (
Table 3).
Multimorbidity emerged as the strongest independent predictor of refusal, with participants without multimorbidity being significantly more likely to proceed with invasive diagnostic evaluation. A history of previous malignancy and higher cumulative smoking exposure were also independently associated with refusal. Screening site remained significantly associated with refusal at the overall level, with participants screened in Subotica being less likely to proceed with invasive diagnostic evaluation compared with the reference center.
Concern about personal lung cancer risk, recoded as low versus any concern, showed a directionally consistent but non-significant association with acceptance of invasive diagnostics after adjustment.
3. Discussion
In this real-world multicentre LDCT lung cancer screening program, refusal of recommended invasive diagnostic procedures occurred in a meaningful proportion of individuals with positive screening results. Importantly, refusal was observed among participants who had already engaged with screening and received a clear multidisciplinary recommendation for further diagnostic evaluation. This finding indicates that post-screening attrition cannot be explained solely by barriers to screening access, but rather represents a distinct outcome shaped by a combination of patient-related and system-level factors extending beyond the initial LDCT examination.
Unlike the broad concept of loss to follow-up commonly used in lung cancer screening studies to describe non-adherence across the screening continuum [
18], refusal reflects an active decision-making process rather than passive disengagement [
19]. By operationally defining refusal as the absence of invasive diagnostic procedures within six months following an MDT recommendation, our study provides a more granular understanding of where post-screening pathways may falter in routine practice. Recognizing refusal as a separate implementation outcome is essential, as it implies different corrective strategies than those aimed at improving attendance or follow-up alone [
20].
Several clinical characteristics were independently associated with refusal of invasive diagnostics.
Multimorbidity emerged as the strongest independent predictor, underscoring the role of cumulative health burden in shaping post-screening decision-making. Individuals with multiple chronic conditions may perceive invasive diagnostic procedures as disproportionately risky or burdensome, particularly in the context of competing health priorities and reduced physiological reserve [
21]. A history of prior malignancy was also independently associated with refusal, potentially reflecting accumulated treatment burden and heightened anticipatory distress related to further diagnostic procedures. Cancer survivors may experience fatigue, anxiety, and reduced tolerance for additional medical interventions, particularly when faced with renewed diagnostic uncertainty, which may influence their willingness to undergo invasive diagnostics in the post-screening setting [
22].
Notably, while individual comorbid conditions were not significant when examined separately, the composite measure of multimorbidity showed a strong association with refusal. This finding supports the use of aggregated comorbidity measures in screening research and suggests that cumulative health burden, rather than single diagnoses, may be more relevant to decision-making in complex care pathways [
21]. From an implementation perspective, multimorbidity may reflect not only biological vulnerability, but also increased healthcare workload, including frequent medical visits, multiple diagnostic procedures, and polypharmacy. In such contexts, the additional burden of invasive diagnostic evaluation following screening may be perceived as disproportionate, particularly within health systems characterized by fragmented care pathways and high organizational demands.
Smoking-related variables warrant particular attention. Although current smoking status was not independently associated with refusal, cumulative smoking exposure (pack-years) remained a significant predictor in multivariable analysis. This finding suggests that long-term smoking history, rather than current behavior alone, may shape perceptions of vulnerability, fatalism, or anticipated benefit from further diagnostic evaluation [
19]. Beyond its role as a marker of cumulative biological risk, long-term smoking exposure may also influence psychological responses to screening outcomes. Qualitative evidence suggests that smoking-related stigma, self-blame, and fear of judgment can shape how individuals interpret lung cancer risk information and engage with recommended diagnostic pathways. Such factors may contribute to anticipatory distress and reluctance to undergo invasive diagnostic procedures, particularly in the context of repeated medical encounters following a positive screening result [
24].
Perceived personal lung cancer risk was associated with refusal of invasive diagnostics in univariable analyses; however, this association did not remain statistically significant after adjustment for clinical and organizational factors. This attenuation suggests that the influence of risk perception may be partially mediated or overridden by accumulated health experiences, such as multimorbidity and prior cancer diagnosis, which appear to play a more dominant role in post-screening decision-making. Rather than diminishing the relevance of risk perception, this finding highlights its interaction with broader clinical context and underscores that cognitive and emotional responses to screening results are shaped not by perceived risk alone, but by lived experiences within the healthcare system.
Importantly, risk perception represents a potentially modifiable determinant and continues to hold relevance for intervention design. In the context of LDCT screening, perceived risk is not static and may evolve throughout the screening and diagnostic pathway, underscoring the role of healthcare professionals at multiple levels—from primary care providers and nursing staff to radiologists, pulmonologists, and surgeons—in supporting informed and context-sensitive decision-making.
Emerging evidence suggests that patient understanding of screening results and recommendations, perceived personal risk, and the quality of communication during shared decision-making substantially shape downstream diagnostic decisions following positive LDCT findings. Recent studies further indicate that both patient-facing decision-support tools and clinician-related factors—including training, confidence in shared decision-making, and referral practices—may influence whether recommended diagnostic pathways are pursued, underscoring refusal as a modifiable, system-mediated outcome rather than a purely individual choice [
26,
27,
28].
Although education level, employment status, and living arrangement were not statistically significant predictors of refusal in this cohort, their consideration remains relevant for contextual interpretation [
29]. The screened population predominantly consisted of older individuals, many of whom were retired and living with multiple chronic conditions. In such contexts, logistical burden, healthcare fatigue, and reliance on local healthcare infrastructure may influence decision-making, even when not captured as independent predictors in retrospective analyses. These factors underscore the multifactorial nature of refusal and caution against interpreting it as a purely individual choice.
Differences in refusal rates across screening sites further highlight the importance of system-level influences on diagnostic continuity.
Screening site remained independently associated with refusal after multivariable adjustment, suggesting that organizational and contextual factors may shape patient decisions beyond individual clinical characteristics. Screening site likely functions as a proxy for geographic accessibility, referral coordination, and local implementation practices, including communication pathways between screening and diagnostic services [
30]. While centralization of invasive diagnostic procedures in tertiary centers is clinically appropriate, it may inadvertently introduce additional access-related barriers for individuals residing farther from diagnostic facilities.
From an implementation perspective, refusal of invasive diagnostics may be considered a meaningful indicator of lung cancer screening program performance, particularly in centralized screening models. Rather than reflecting individual non-adherence alone, refusal may signal misalignment between patient expectations, provider communication, and system organization during the transition from screening to diagnostic evaluation [
20].
An important observation of this study is the contrast between relatively uniform time to surgical treatment across screening centers and greater variability in time to first bronchoscopy. Once patients entered definitive surgical pathways, timelines appeared largely standardized, suggesting that downstream treatment processes were robust to local organizational differences. In contrast, bronchoscopy represents an earlier and more operationally sensitive diagnostic step, potentially influenced by local infrastructure, availability of trained personnel, and referral logistics.
Importantly, this variability should not be interpreted as a direct cause of refusal, particularly given overall acceptable diagnostic timelines and low complication rates. Rather, it highlights early invasive diagnostics as a potential leverage point for system-level optimization. Ensuring timely, predictable, and clearly communicated access to minimally invasive diagnostic procedures may reduce uncertainty and support informed decision-making following positive screening results [
20,
26].
Taken together, our findings emphasize that LDCT lung cancer screening should be conceptualized as a longitudinal process extending beyond image acquisition. While patient education and risk communication remain central to improving diagnostic adherence, system-level factors shaping early diagnostic steps warrant equal attention. Strengthening coordination between screening sites and diagnostic services, enhancing patient navigation, and supporting workforce capacity where feasible may help reduce post-screening attrition without compromising safety [
20,
26]. Such measures should be viewed as supportive strategies aimed at improving continuity and equity of care, rather than as critiques of existing healthcare structures.
This study has limitations inherent to its retrospective design. Psychological, cognitive, and socioeconomic factors were not assessed using validated instruments, limiting insight into the mechanisms underlying refusal. The number of refusal events constrained more detailed subgroup analyses. Future prospective studies should incorporate validated measures of health literacy, decisional conflict, anxiety, and social support, as well as qualitative approaches to capture patient perspectives more comprehensively.