1. Introduction
Understanding the molecular architecture of oncogenic progression remains a central challenge in cancer biology. Transcriptional programs shape the molecular processes underlying the Hallmarks of Cancer, enabling non-mutational epigenetic reprogramming and deregulated cellular energetics. While conventional tumor promoters and suppressors foster clonal expansion through established signaling cascades, emerging evidence indicates that modulation of deuterium concentration—particularly depletion— may represent a distinct, yet underexplored, potential regulatory layer. Shifts in deuterium concentration have been suggested to influence mitochondrial function, redox balance [
1], and signal transduction [
2], with potential consequences for cell-cycle progression, the emergence of drug-resistance [
3], and the selective metabolic targeting of malignant cells [
4].
The role of deuterium as a regulator of cellular physiology was first proposed in the early 1990s, when Somlyai et al. demonstrated that reducing naturally occurring deuterium content interferes with tumor cell growth [
5]. Subsequent studies extended these findings by reporting that deuterium-depleted water (DDW) reduces the expression of cancer-related genes, including c-myc, Ha-ras and p53, in carcinogen-treated mice [
6], and as well as Kras, Bcl2, and Myc in lung cancer models [
7].
These observations raise the question of whether such gene-level effects extend to human cancer models. In this context, Cong et al. [
8] demonstrated that 50±5 ppm DDW inhibited A549 lung adenocarcinoma (LUAD) cell growth and induced apoptosis in vitro—supported by electron microscopy and flow-cytometric analysis—and also reduced tumor growth in a mouse xenograft model in vivo. However, despite these phenotypic observations, the broader transcriptomic landscape underlying these metabolic and survival effects remains insufficiently characterized.
Kovács et al. [
9] conducted a comprehensive
in vitro study using the A549 LUAD cells to examine how altered deuterium concentrations affect gene expression. Prior screening had shown that A549 cells are particularly sensitive to deuterium modulation compared to other cell lines [
1], supporting their use as a sentinel model. Cells were cultured in media containing 40, 80, 150 (control), and 300 ppm deuterium, and transcriptional output of technical duplicates was quantified with NanoString technology [
10] across 236 cancer-related and 536 kinase genes. Expression changes were evaluated under permissive thresholds (≥30% deviation from control; ≥30 mRNA copies) given the noise level in the results. The study revealed that 97.3% of cancer-related genes were upregulated at 300 ppm, whereas only five genes were downregulated under depletion, highlighting strong transcriptional activation under enrichment and modest suppression under depletion.
In our previous work [
11], we reanalyzed the same A549 dataset with a focus on data quality. Recognizing that raw NanoString counts are prone to stochastic noise, especially at low expression levels, we exploited technical duplicates to calculate the coefficient of variation (CV) for each gene, thereby quantifying measurement precision. Genes with high CVs or low (<20) counts were excluded, ensuring that subsequent conclusions were drawn only from reliable, high-confidence signals. This refined analysis revealed that deuterium concentration not only drives broad transcriptional activation but also specifically modulates oncogenic and drug-resistance networks across nine distinct expression patterns—observations that had been obscured in the noisier raw dataset.
In our current work, we build on this foundation but adopt a different filtering strategy. The rationale for choosing 40, 80, 150, and 300 ppm deuterium concentration is grounded in prior experimental evidence demonstrating that deuterium concentration directly influences cancer cell proliferation and metabolic signaling as detailed in section 4.1. Rather than relying on CV and robust Z-score thresholds, we calculated propagated relative errors across the three non-control concentrations (40, 80, and 300 ppm) and retained only genes with a total relative error < 0.55. Threshold selection (0.55 summed relative error) was empirically guided by the distribution of error values in this dataset and may vary depending on dataset size, measurement precision, and error structure. As a result, 102 genes were included compared to 87 in our previous study, with 37 biologically relevant additions—such as TP53, NRAS, IL6, and PTEN—and 22 removals due to excessive propagated error. This refinement yields a gene set that is both more accurate and biologically comprehensive, providing a stronger basis for downstream clustering and pathway analysis.
Crucially, this study employed the A549 cell line, a sentinel model of
KRAS-driven LUAD characterized by co-occurring
KRAS G12S,
STK11 (
LKB1) loss, and
KEAP1 G333C mutations on a wild-type
TP53 background [
12,
13]. Collectively, these alterations constitute a high-risk genetic constellation in which
KRAS activation and
STK11 deficiency cooperate to promote proliferative signaling and metabolic deregulation, while
KEAP1 inactivation reinforces oxidative-stress resilience through constitutive
NRF2 activation. Importantly, the retention of wild-type
TP53 preserves DNA-damage signaling competence, positioning A549 as a robust model for dissecting how
KRAS-mutant LUAD integrates metabolic adaptation, drug resistance programs, and stress-response circuitry. This framework supports the use of A549 as a platform for mapping deuterium-linked regulation in aggressive,
KRAS-driven disease. Thus, our results specifically characterize the
KRAS/STK11/KEAP1 co-mutant LUAD subtype and should not be generalized to genetically distinct forms of LUAD.
The present study introduces a refined analytical pipeline that integrates symbolic clustering, trajectory mapping, and rigorous data cleaning. Using density-based spatial clustering (DBSCAN) to detect sentinel outliers and Gaussian mixture modeling (GMM-6) to resolve coherent transcriptional modules, this framework advances prior work [
9,
11] by resolving the gene regulatory networks governing metabolic plasticity and defining the specific transcriptomic signatures associated with invasive and drug-resistant phenotypes.
2. Results
2.1. The Oncogenic Core Network
To define the baseline architecture of the study, we first mapped the 102 selected consensus cancer genes onto a canonical oncogenic core network (
Figure 1). This network illustrates how the selected genes organize into interconnected functional modules that collectively sustain malignant phenotypes. RTKs & Adaptors (e.g.,
EGFR,
ERBB2,
MET) act as upstream hubs, transmitting extracellular growth signals into the RAS–MAPK core and PI3K–AKT axis. The RAS–MAPK module—constitutively locked in an active state by the gain-of-function
KRAS G12S mutation—links directly to Cell Cycle & Replication (e.g.,
CCND1,
CDK4) and Transcriptional Amplifiers include canonical oncogenes (e.g.
MYC) and contextual promoters (e.g.,
STAT3,
JUN), collectively enforcing the Hallmarks of Sustaining Proliferative Signaling and Enabling Replicative Immortality.
In parallel, the PI3K–AKT axis functions as a critical survival node. In the specific context of A549 cells, this axis connects to Cell Survival to override the apoptotic potential of wild-type TP53 while engaging anti-apoptotic effectors like BCL3. This active suppression ensures Resisting Cell Death and Deregulating Cellular Energetics, maintaining viability despite oncogenic stress. Cytokine/Inflammatory Signaling (e.g., IL6, TGFB1— contextual tumor promoters) feeds directly into transcriptional amplification and Invasion & ECM Remodeling (e.g., CD44, MMP9), linking Tumor-Promoting Inflammation to Activating Invasion & Metastasis—a connection that is particularly amplified by the STK11 deficiency inherent to this cell line.
Finally, DNA Repair & Stress Tolerance (e.g., ERCC2, MSH6) anchors the hallmark of Genome Instability. Critically, this module is shaped by the loss-of-function KEAP1 G333C mutation found in A549 cells. By disabling the "brake" on oxidative stress responses, this mutation creates a constitutive antioxidant shield that protects the tumor against therapeutic insults, maintaining a permissive state for oncogenic evolution.
2.2. Classification of Genes by Pathway and Global Centroid Dynamics
To map the transcriptional landscape of A549 cells under deuterium stress, we plotted the 102 consensus genes in a three-dimensional expression space defined by their ratios at 40, 80, and 300 ppm relative to the 150 ppm control (
Figure 2). Genes were color-coded by pathway, and their collective behavior was quantified by calculating pathway-specific centroids (
Table 1). To identify transcriptional outliers, we ranked all genes by their Euclidean distance from the global center. The top six genes—
TGFBR2,
IL6,
FGFR4,
ABCB1,
MYCN, and
PTK7—were selected based on their pronounced divergence and a clear inflection point in the distance distribution (highlighted in red in
Figure 2).
The quantitative analysis of pathway centroids (
Table 1) reveals a monotonic increase in transcriptional activity correlating with deuterium concentration. The
Global Center coordinates (0.884 → 0.940 → 1.436) serve as the system-wide baseline:
At 40 ppm, the global centroid (0.884) is well below unity, indicating that extreme deuterium depletion exerts a broad suppressive effect on the oncogenic transcriptome.
At 80 ppm, expression remains dampened (0.940), reinforcing the concept of depletion as a "metabolic brake."
At 300 ppm, the global centroid surges to 1.436, reflecting a robust, system-wide activation (~44% increase).
Using this global 1.436 value as a benchmark reveals distinct pathway sensitivities. Pathways exceeding this threshold at 300 ppm—including Cytokine/Inflammation (1.567), Cell Survival (1.476), and Invasion & ECM Remodeling (1.469)—are hyper-responsive to deuterium enrichment. In contrast, RTKs & Adaptors (1.377) and Cell Cycle & Replication (1.371) respond more modestly, falling below the global average. Notably, Drug Metabolism and Transport (ABCB1) exhibits a unique trajectory with extreme suppression at 80 ppm (0.585), diverging sharply from the global trend. Together, these results indicate that A549 cells adapt to deuterium stress by suppressing energetically costly programs (like drug efflux) under depletion, while enrichment drives a coordinated reprogramming that favors survival, invasion, and inflammation.
Centroid values represent the mean relative expression ratios at 40, 80, and 300 ppm compared to the 150 ppm control. The Global center corresponds to the overall average across all pathways and serves as a reference baseline. Pathways are annotated with representative genes.
2.3. Density-Based Spatial Clustering
To complement centroid-based identification of transcriptionally sensitive genes, we applied DBSCAN to the 3D expression space defined by relative expression ratios at 40 ppm, 80 ppm, and 300 ppm deuterium as shown in
Figure 3. A 5-distance plot (
Figure S1) was used to select the ε parameter, consistent with the standard practice of setting
min_samples = 5. This choice balances sensitivity to small transcriptional modules with robustness against noise, enabling detection of biologically meaningful outliers in the 102-gene dataset. Unlike centroid-based methods that emphasize spherical pathway-level coherence, DBSCAN identifies density-based outliers with unique expression ratios, independent of canonical pathway clustering. These outliers may represent sentinel responders or context-specific regulators whose behavior diverges from pathway averages, offering additional insight into deuterium-dependent transcriptional heterogeneity.
To visually summarize the directional behavior of gene expression ratios across three deuterium concentrations (40, 80, 300 ppm), we assigned each outlier a pattern symbol triplet (e.g., → ↓ ↗) based on its relative expression values. These symbols reflect the magnitude and direction of change using a biologically interpretable scale: ↑ for strong upregulation (>1.37), ↗ for moderate upregulation (1.17–1.37), → for stable expression (0.82–1.17), ↙ for moderate downregulation (0.70–0.82), and ↓ for strong downregulation (<0.70). The thresholds were rigorously selected based on statistical distributions and centroid benchmarks (e.g., the 1.37 upper threshold aligns with the lowest centroid of upregulated clusters at 300 ppm).
2.4. Pathway-Specific Analysis of DBSCAN Outlier Genes
To elucidate the biological relevance of the 11 outlier genes identified by DBSCAN clustering (ε = 0.13), we grouped them by functional pathway and analyzed their expression dynamics.
2.5. Drug Metabolism and Transport
ABCB1 (→ ↓ ↗) encodes P-glycoprotein, a member of the ATP-Binding Cassette (ABC) transporter superfamily. It functions as an ATP-powered efflux pump that actively exports a wide range of hydrophobic molecules, including chemotherapeutic agents, making it a central mediator of multidrug resistance (MDR). In our dataset, ABCB1 exhibited a non-linear response: mild induction at 40 ppm (+12%), suppression at 80 ppm (−42%) (with small 7% error margin), and reactivation at 300 ppm (+27%). This distinct, significant suppression at 80 ppm is particularly significant and identifies ABCB1 as a sentinel gene highly sensitive to moderate deuterium depletion.
2.6. Cell Survival
BCL3 (↗ → ↑) is an IκB-like protein that modulates NF-κB signaling, promoting transcription of anti-apoptotic genes and enabling tumor survival under stress. It is frequently upregulated in hematologic and solid malignancies. In this dataset, BCL3 exhibited progressive upregulation across all deuterium concentrations, consistent with a robust survival response. Its low total error reinforces the reliability of this measurement and supports its role as a transcriptional amplifier of cell survival under deuterium concentration change.
2.7. RTKs and Adaptors
This functional group highlights a striking divergence in receptor sensitivity. FGFR1 (→ → ↑) exhibited stable expression at physiological levels but surged at 300 ppm (+57%), indicating a threshold-dependent response to deuterium enrichment. In sharp contrast, FGFR4 (↙ ↙ →) was consistently suppressed at 40 and 80 ppm (~−19%), suggesting that deuterium depletion dampens specific growth factor receptivity. PTK7 (↗ → ↑), an orphan receptor involved in Wnt/Planar Cell Polarity signaling, displayed a progressive upward trend across all concentrations. Together, these profiles demonstrate that while enrichment fuels broad receptor activation (FGFR1, PTK7), depletion selectively compromises specific signaling nodes (FGFR4).
2.8. Cytokine and Inflammation
IL6 (→ → ↑), PTGS2 (↗ → ↑), TGFBR2 (→ → ↑), and TGFBR3 (↓ → ↗) are key regulators of inflammation and immune modulation. IL6 and PTGS2 promote tumor growth, angiogenesis, and immune suppression, with IL6 showing strong induction at 300 ppm (+99%). TGFBR2 and TGFBR3 mediate TGF-β signaling, which plays context-dependent roles in cancer progression. TGFBR2 was strongly induced at 300 ppm (+105%), while TGFBR3 showed distinct suppression at 40 ppm (−36%) followed by recovery. These patterns suggest that deuterium enrichment amplifies inflammatory signaling, contributing to Tumor-Promoting Inflammation.
2.9. Invasion and ECM Remodeling
MMP9 (↓ → ↑) encodes a matrix metalloproteinase that degrades extracellular matrix components, facilitating tumor invasion. It was suppressed at 40 ppm (−31%) and progressively induced at higher concentrations. As a driver of Activating Invasion & Metastasis, its suppression under depletion reflects a dose-dependent inhibition of invasive behavior.
2.10. Transcriptional Amplifiers
MYCN (→ ↙ →) is a member of the MYC family, regulating cell cycle and metabolism. It showed mild suppression at 80 ppm (−24%) followed by stability at 300 ppm (+1%). As a master regulator, MYCN’s modulation under deuterium stress may have cascading effects on downstream oncogenic programs.
2.11. Core Pathway Stability
In the current dataset of 102 genes, four pathways—Cell cycle & replication, DNA repair & stress tolerance, PI3K–AKT axis, and RAS–MAPK core—contained no outlier genes. This consistency shows that all genes in these core oncogenic programs responded in a moderate, coordinated manner to deuterium concentration changes, indicating that these pathways act as stable, coherent modules. In contrast, heterogeneous pathways like Cytokine and Inflammation showed larger variations, producing multiple outliers.
2.12. Symbolic Clustering of Gene Expression Patterns
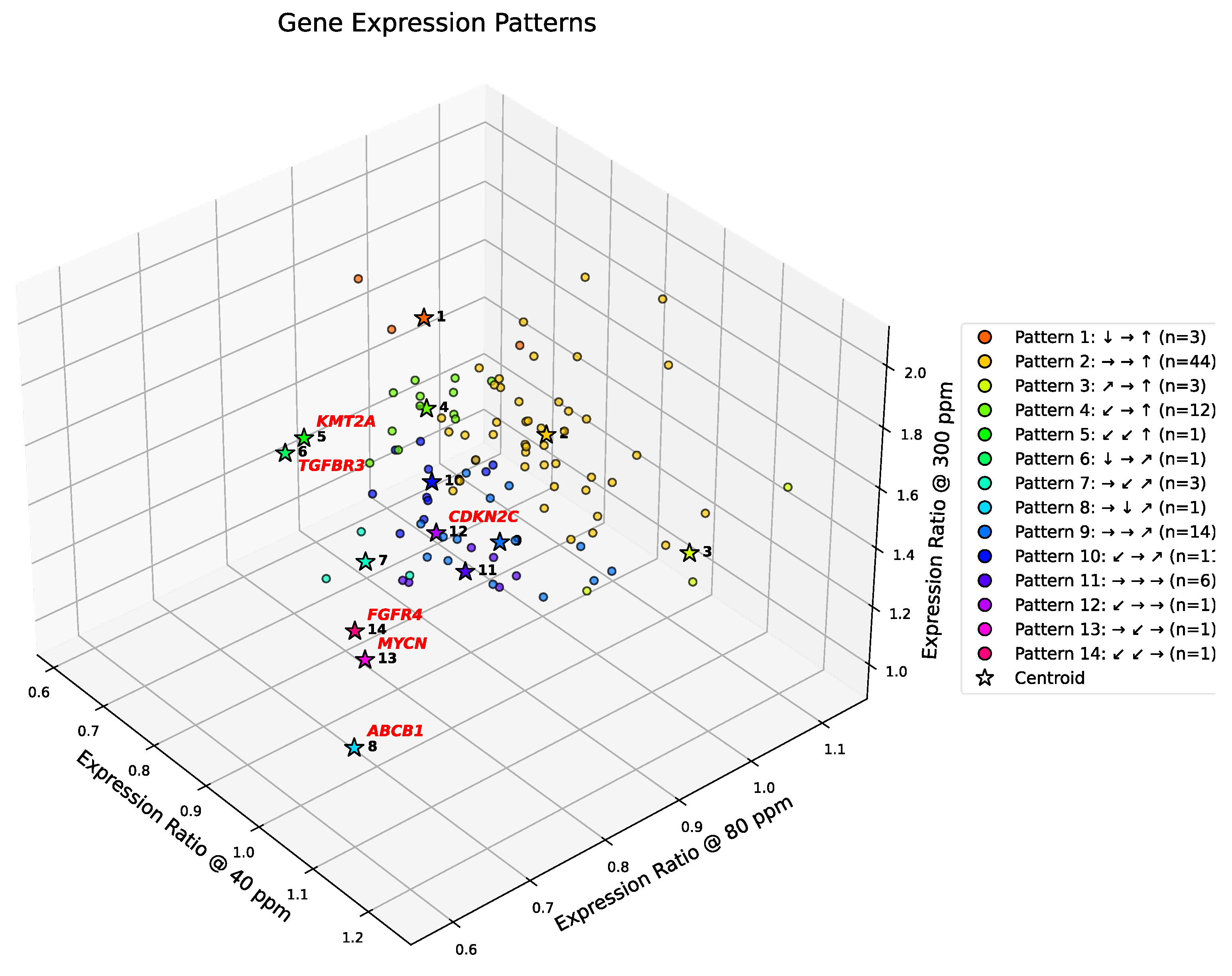
In this section, we analyze gene expression patterns in the 102-gene dataset using the discretized thresholds introduced above. Symbolic clustering across deuterium concentrations reveals a striking sparsity in realized transcriptional patterns. Although the symbolic encoding scheme allows for 125 theoretical combinations, only 14 distinct patterns were observed (cf.
Figure 4). This >90% reduction in theoretical complexity reflects strong biological constraints, suggesting that A549 cells converge onto a limited set of coordinated regulatory programs to manage deuterium stress.
2.13. Dominant Transcriptional Trajectories
The distribution of genes across these 14 patterns is highly skewed. Most genes converge into just three dominant clusters, with Pattern 2 emerging as the overarching transcriptional program.
Pattern 2 (→ → ↑): This dominant group (n=44) represents genes that remain stable under depletion but surge under enrichment. It encapsulates the "Enrichment as Accelerator" phenotype, comprising nearly half the dataset and driving the Hallmarks of Resisting Cell Death, Sustaining Proliferative Signaling, and Tumor-Promoting Inflammation.
Minor Clusters: Two other patterns (Patterns 4 and 9) contain between 12–14 genes, representing alternative regulatory modes. Patterns 1, 3, 7, and 11 contain between 3–6 genes.
2.14. Singleton Outliers and Methodological Consensus
In contrast to the dominant groups, six genes—
ABCB1, CDKN2C, FGFR4, KMT2A, MYCN, and
TGFBR3—exhibit unique symbolic patterns not shared by any other gene. Importantly, four of these six singleton genes align with the outliers identified by DBSCAN, reinforcing their status as biologically distinct transcriptional entities (
Figure 4). For instance,
ABCB1 and
FGFR4 consistently diverge from the population mean, spanning critical hallmarks of Multidrug Resistance and Growth Factor Signaling.
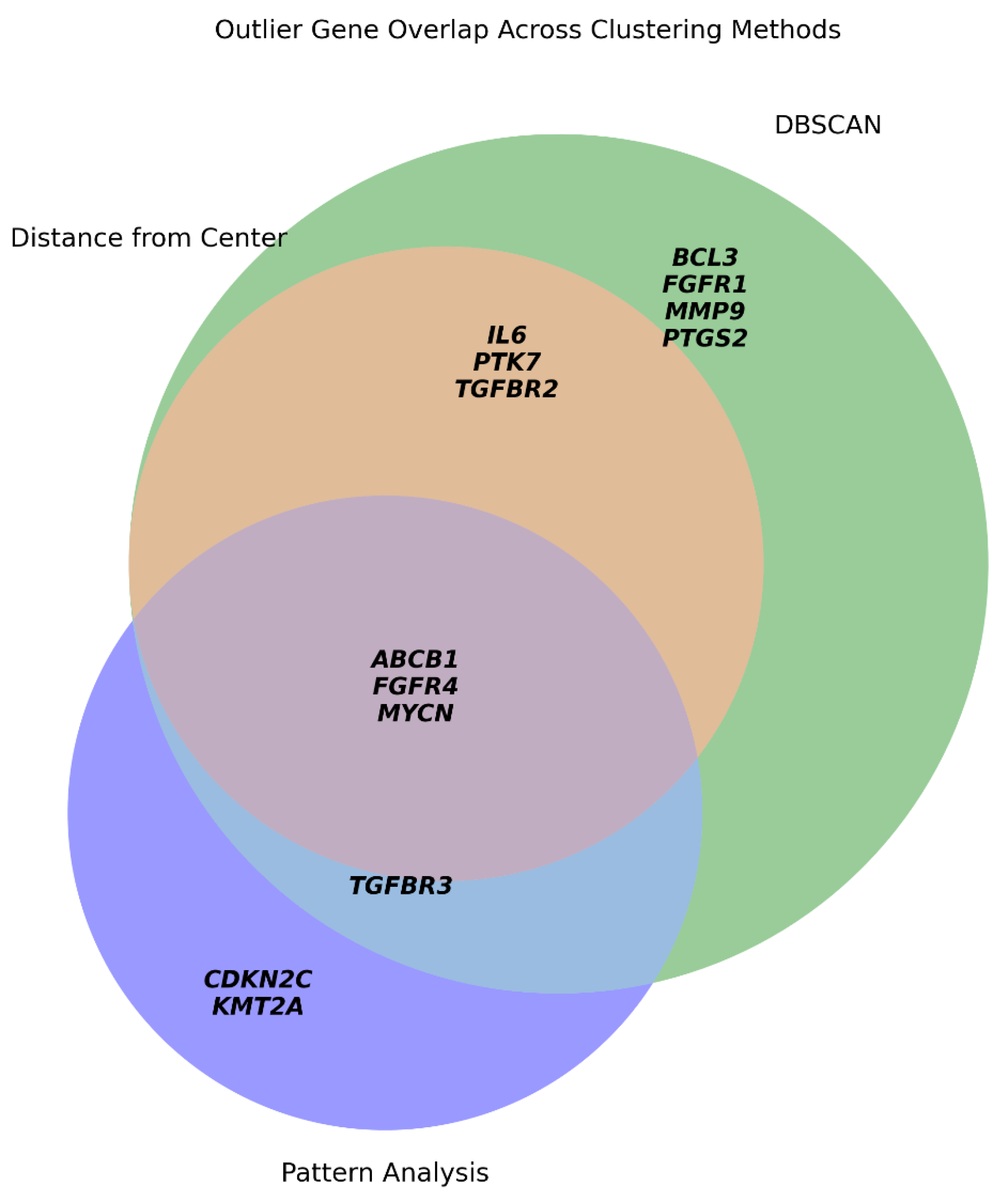
Comparative Outlier Detection and Dataset Refinement
We evaluated transcriptional outliers using three complementary approaches: global centroid distance, DBSCAN clustering, and symbolic pattern analysis (
Figure 5).
Centroid-based: Flagged six genes, all of which were re-confirmed by DBSCAN.
Symbolic Analysis: Overlapped with four DBSCAN-defined genes and identified unique trajectories for CDKN2C and KMT2A.
DBSCAN: Proved the most sensitive method, identifying the "core" outliers captured by other methods plus five additional context-specific regulators.
This comparison demonstrates that while distance-based metrics provide baseline selectivity, DBSCAN offers superior sensitivity in detecting transcriptional divergence. Accordingly, for the subsequent GMM clustering, we excluded the full set of DBSCAN-defined outliers to improve cluster resolution and biological interpretability, reducing the dataset to 91 genes.
In summary, the symbolic clustering framework delineates a biologically constrained transcriptional landscape, characterized by a limited set of dominant high-expression patterns and punctuated by distinct outliers. The sparsity of realized patterns, the centrality of Pattern 2, and the functional relevance of singleton genes highlight the importance of integrated clustering and pathway-level interpretation in future analyses. Nonetheless, propagated measurement error in the current dataset may reach 10–15%, potentially causing genes near classification boundaries to overlap with adjacent patterns. This ambiguity (see Figure S2) highlights the need for complementary unsupervised clustering approaches. Moreover, the five-category classification scheme may be overly rigid for capturing extreme expression levels—for example, it assigns the same symbol (↑) to 40% and 100% overexpression. To complement the symbolic classification, we next applied unsupervised clustering using Gaussian Mixture Models (GMM), which provide a probabilistic framework for identifying continuous transcriptional variation and resolving ambiguous boundary assignments. Gaussian Mixture Models are particularly suited to this dataset because they treat expression ratios as overlapping probability distributions, allowing genes with propagated error near symbolic boundaries to be assigned probabilistically rather than rigidly. This flexibility captures subtle transcriptional shifts and resolves the ambiguity inherent in discrete symbolic categories.
2.15. GMM Clustering 91 Genes with Tied Covariance Ellipsoids
We evaluated Gaussian Mixture Models (GMMs) using silhouette analysis across seven candidate cluster counts (
k = 2 to 9), where
k denotes the number of components the algorithm attempts to identify (cf.
Figure S3). Each cluster ideally captures a distinct gene expression pattern under varying deuterium concentrations.
2.16. Statistical Model Selection
A critical observation from the silhouette analysis is that all scores remained below 0.5. In clustering topology, a coefficient < 0.5 indicates that the data structure is not characterized by distinct, widely separated "islands," but rather represents a continuous biological gradient with overlapping boundaries. Consequently, the silhouette score alone was insufficient to determine the optimal partition, requiring a balance between statistical separation and biological resolution.
Rejection ofk= 2: Although the tied covariance model with k = 2 yielded the highest numerical silhouette score, inspection revealed this solution was uninformative, providing only a trivial binary partition (high vs. low expression at 300 ppm) that masked pathway-specific dynamics.
Selection ofk= 4 andk= 6: The tied covariance model with k = 4 yielded the second-highest score, closely followed by k = 6. This configuration achieved a favorable Bayesian Information Criterion (BIC = −438) and log-likelihood (LogLik = 267), suggesting an optimal balance between model fit and complexity.
2.17. Model Robustness and Sensitivity Analysis
While full covariance models produced higher LogLik values, their substantially worse BIC scores pointed to overfitting, justifying the use of tied covariance. Overall, GMM clustering (silhouette = 0.320) outperformed k-means (silhouette = 0.255), confirming that the probabilistic, ellipsoid-based boundaries of GMM better capture the complex covariance structure of oncogenic gene expression than the spherical assumptions of k-means.
To assess clustering robustness, we performed a leave-one-out sensitivity analysis comparing GMM models with 4 and 6 clusters (GMM-4 and GMM-6, cf.
Table S2). For each gene, we computed the Adjusted Rand Index (ARI) after its removal. The average ARI difference was −0.043, indicating that GMM-6 is marginally more stable overall. However, gene-specific impacts varied:
Stabilizers: Genes such as
BIRC5 and
MUC1 stabilized the GMM-4 solution, reflecting their roles in core cell cycle/survival modules, cf.
Table S2.
Destabilizers: Genes like
AKT2 and
BCL2A1 destabilized GMM-4, suggesting they sit at the boundaries of broader clusters and require the higher resolution of GMM-6 to be accurately partitioned, cf.
Table S2.
Instability ofk= 9: Notably, the 9-cluster solution proved unstable; removal of a single gene triggered complete cluster rearrangement, confirming that k = 9 over-fragmented the data into artificial groups.
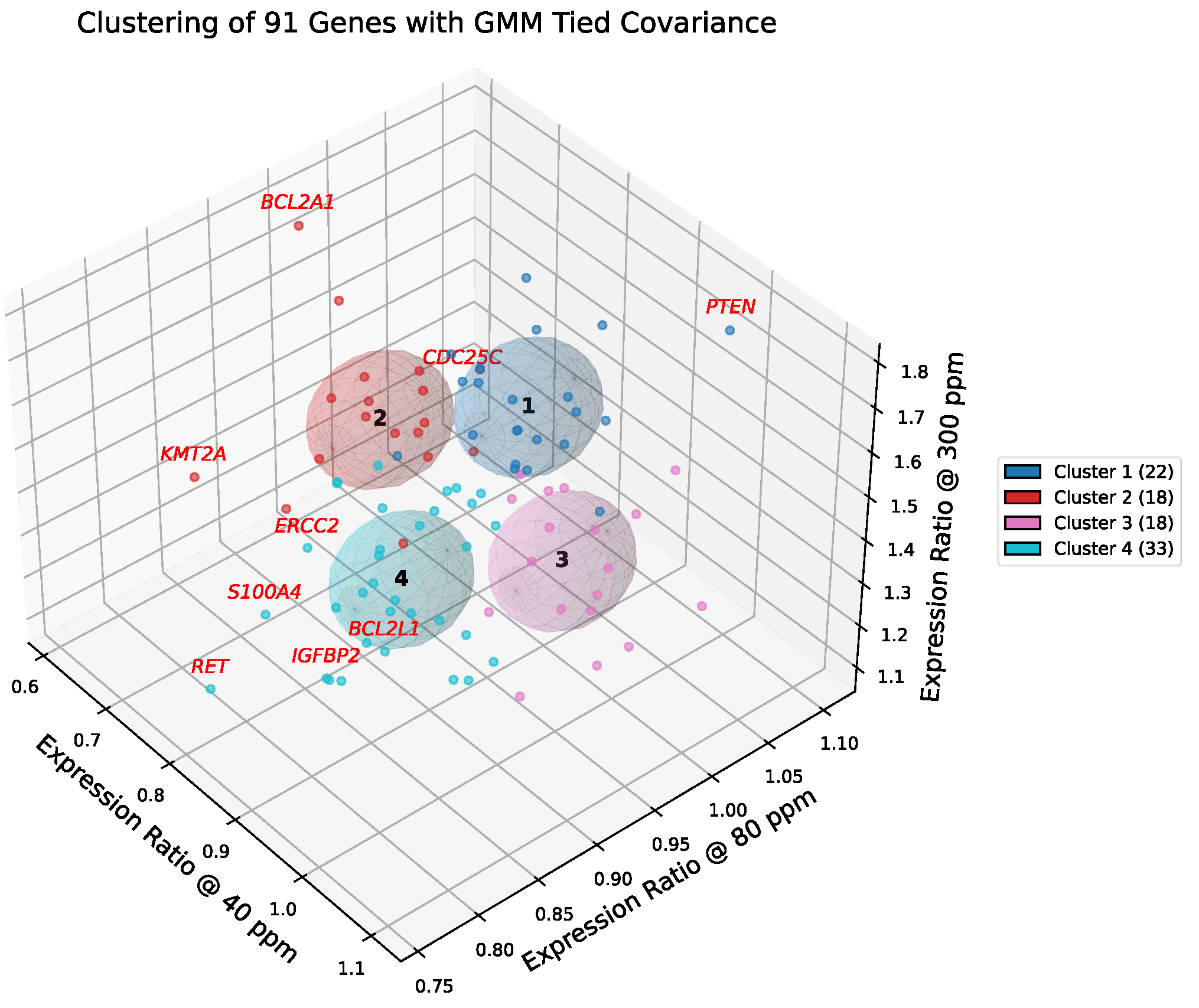
2.18. Cluster Visualization and Boundary Drivers
Figure 6 and
Figure 7 present the results of GMM-4 and GMM-6 applied to the curated set of 91 cancer-related genes. In the selected model, each cluster is represented by a shared covariance ellipsoid centered at its coordinate mean. To assess the confidence of assignments, we computed the Mahalanobis distance of each gene from its cluster centroid, cf.
Table S2. Unlike Euclidean distance, Mahalanobis distance accounts for the covariance structure, penalizing directions of low variance. Genes with high Mahalanobis distances (e.g.,
PTEN, CDC25C, ERBB3, BCL2A1) were identified as boundary drivers—genes that are statistically peripheral to their cluster yet biologically critical for defining the cluster's functional edge (e.g., balancing tumor suppression with amplification).
2.19. Cluster Transitions and Heterogeneity (GMM-4 to GMM-6)
To visualize how increasing model granularity redistributes genes, we mapped the transitions from GMM-4 to GMM-6 using a Sankey diagram (cf.
Figure 8). The analysis revealed clear patterns of stability and fragmentation:
Stable Modules: Genes assigned to GMM-4 clusters 4-1 and 4-2 (22 and 18 genes, respectively) largely remained together, mapping almost exclusively into GMM-6 clusters 6-1 and 6-2. This indicates that these transcriptional programs (associated with Cellular Regulation and KRAS Effector signaling) are robust across model resolutions.
Fragmented Heterogeneity: In contrast, the largest GMM-4 cluster (4-4, n = 33) was partitioned into multiple GMM-6 clusters (6-4, 6-5, and 6-6). This dissociation unmasked critical heterogeneity: it separated the Invasive/Plasticity drivers (TGFB1, now Cluster 5) and Growth Factors (PDGFA, now Cluster 4) from the Basal Stability genes (BIRC5, now Cluster 6). The proportional widths of the flows emphasize that while some transcriptional states are robust, others fragment into finer substructures, proving that the six-cluster resolution is necessary to distinguish Invasion programs from general survival mechanisms.
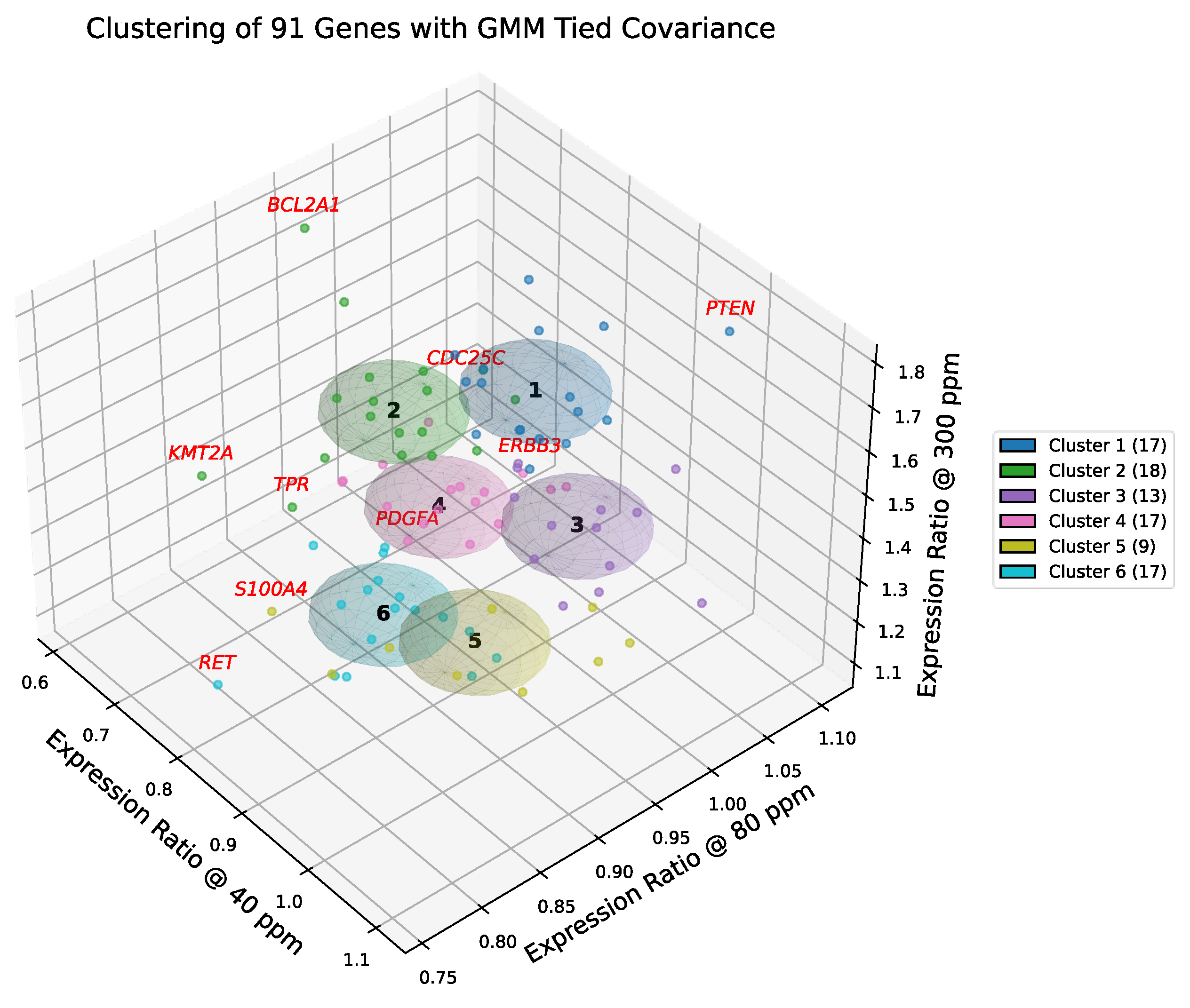
2.20. Composition and Biological Identity of GMM-6 Clusters
Gaussian mixture modeling with tied covariance ellipsoids (k = 6), validated by silhouette analysis, delineated six distinct gene groups whose centroid trajectories across deuterium concentrations define the transcriptional landscape of A549 cells.
Clusters 1 and 2 showed the strongest induction at 300 ppm (cf. Figure 7), representing stress-responsive proliferative and survival modules:
Cluster 1 integrated cell cycle regulators and DNA repair factors, with boundary outliers (PTEN, CDC25C, ERBB3) marking the tension between tumor suppression and receptor-driven proliferation. Canonical oncogenes (MYC, STAT3, TERT) reinforced its proliferative bias within the context of a wild-type TP53 axis.
Cluster 2 encompassed transcriptional amplifiers and apoptosis regulators, with outliers (BCL2A1, KMT2A, TPR) highlighting survival stability. Importantly, this cluster contained the core RAS effector RAF1 alongside EGFR, underscoring its role as the primary signaling engine driven by the G12S mutation.
Cluster 3 was cohesive, balancing oncogenic drivers (ERBB2, JUN, IGF1) with the tumor suppressor TP53. This module reflects a signaling core where proliferative drive is counterweighted by suppressive control. While basal transcriptional load is driven by oncogenic RAS, the co-clustering of TP53 with homeostatic regulators confirms that A549 cells retain a competent, stress-responsive p53 axis. This module preserves canonical regulatory architecture, enabling a coordinated—though ultimately overwhelmed—response to deuterium modulation.
Cluster 4 contained accessory growth factor and stress response genes, with PDGFA defining its proliferative edge. Oncogenes (NRAS, MET, LYN) were present alongside apoptosis regulators (BCL2, FAS), highlighting a dual role in growth signaling and stress adaptation distinct from the primary RAS driver in Cluster 2.
Cluster 5 was enriched for ECM remodeling and invasion, with S100A4 marking the invasive boundary. Key members included the oncogene HRAS alongside the tumor promoter TGFB1. The enrichment of these plasticity drivers is consistent with the phenotype of STK11 (LKB1) deficiency inherent to A549 cells, identifying this cluster as the primary engine of microenvironmental adaptation and metastatic potential.
Cluster 6 grouped cell cycle and repair genes that remained relatively stable across conditions, with RET delineating its invasion-linked outlier. Oncogenes (AKT2, CDK4, FGFR3) were central, together with survival factors such as BIRC5 (Survivin). This defines a “Basal Maintenance” cluster that sustains viability even when more plastic modules (Cluster 5) are suppressed.
Together, these clusters reveal a structured landscape in which proliferative and survival modules dominate the stress response, while invasion and repair pathways provide adaptive support. Outlier genes stretch cluster boundaries, identifying the most stress-responsive drivers of instability. Notably, several highlighted genes—including TP53, EGFR, and RET—are clinically relevant in A549 LUAD cell line, underscoring the translational importance of these clustering results.
3. Discussion
3.1. Deuterium as a Dual Regulator of Oncogenic Physiology
Recent work has highlighted that intracellular deuterium concentration is not a passive variable but an active regulator of cellular physiology [
14], with both depletion and enrichment exerting measurable effects on cancer biology. Reviews have shown that deuterium enrichment can drive oxidative stress, metabolic rewiring, and transcriptional activation [
14], while deuterium depletion has been reported to suppress tumor growth in vitro [
9], in vivo [
6], and in clinical settings [
9,
15]. Our findings that A549 cells exhibit global downregulation under depletion (40–80 ppm) but strong transcriptional activation under enrichment (300 ppm) are consistent with these observations. We extend this knowledge by providing pathway-level resolution of the adaptive programs engaged by cancer cells. In particular, the preferential upregulation of
Tumor-Promoting Inflammation, PI3K–AKT survival, and invasive remodeling pathways under enrichment suggests that deuterium availability couples directly to redox balance, metabolic flux, and stress-response transcriptional networks, thereby shaping tumor cell fitness.
3.2. Clinical Resonance and the A549 Sentinel Model
Our cell-level observations resonate with clinical and translational studies of deuterium-depleted water (DDW), which consistently demonstrate antiproliferative effects. However, these results must be contextualized within the specific genetic architecture of the A549 model. As a KRAS/STK11 co-mutant line with functional TP53, A549 cells represent an aggressive, “immune-cold” phenotype often resistant to standard therapies.
The suppression of ABCB1 (P-glycoprotein) and MMP9 observed under depletion provides mechanistic evidence that deuterium concentration change can bypass these hardwired defenses. This implies that the clinical benefits of DDW reported in systematic reviews may arise not only from general stress but also from selective dismantling of resistance networks—such as NRF2-driven antioxidant shields—that aggressive, metabolically rewired tumors rely on for survival.
3.3. Mechanistic Bypass of KEAP1-Mediated Resistance via an Active Concentration Window
The suppression of ABCB1 reveals a highly specific, concentration-dependent vulnerability in the A549 antioxidant defense system. Critically, this response is not linear: while ABCB1 is suppressed by 42% at 80 ppm, it is slightly induced (+12%) at 40 ppm. This non-linearity defines a distinct active therapeutic window for deuterium depletion.
In the context of the A549 KEAP1 G333C loss-of-function mutation, which drives constitutive NRF2 activity, this “V-shaped” response suggests distinct adaptive thresholds. We hypothesize that moderate depletion (80 ppm) functions as a metabolic brake sufficient to destabilize the energetic support for NRF2-driven transcription, effectively bypassing the genetic defect. By contrast, extreme depletion (40 ppm) appears to exceed a stress threshold, potentially triggering a compensatory survival alarm that re-engages drug resistance mechanisms despite the metabolic constraint. This finding challenges the “lower is better” paradigm, suggesting that maximizing therapeutic efficacy against multidrug resistance requires targeting the specific deuterium concentration window (e.g., 80 ppm) where the metabolic cost of resistance exceeds the cell’s adaptive capacity without triggering a rebound response.
3.4. Resolving Latent Heterogeneity: The Biological Necessity of GMM-6
While the four-component model (GMM-4) captured broad biological programs such as proliferation, the transition to the six-component model (GMM-6) was essential to resolve latent phenotypic heterogeneity. The statistical stability (ARI) of both models masks a critical biological trade-off: GMM-4 obscures the distinction between basal maintenance and active plasticity.
Sankey transition analysis demonstrates that this refinement is not random. The fragmentation of the large GMM-4 Cluster 4 into GMM-6 Clusters 4, 5, and 6 unmasked distinct adaptive strategies:
Cluster 4 retained growth factor-driven proliferation (PDGFA, NRAS).
Cluster 5 emerged as a distinct module driving phenotypic plasticity (TGFB1, S100A4).
Cluster 6 isolated stable, basal survival machinery (AKT2, BIRC5).
This separation is biologically crucial, particularly given the STK11-deficient status of A549 cells, which drives invasion. It reveals that the invasive phenotype (Cluster 5) is transcriptionally distinct from basal survival (Cluster 6) and, importantly, is far more sensitive to deuterium depletion. By resolving these modules, we demonstrate that deuterium stress does not simply suppress the cell uniformly; rather, it selectively disengages the high-energy programs required for Activating Invasion and Metastasis while leaving the core survival machinery relatively intact.
3.5. Biological Coherence of GMM-Derived Clusters and Pathway Enrichment
In contrast to widely used transcriptomic stratification methods such as consensus clustering, non-negative matrix factorization (NMF), or single-cell trajectory inference, our symbolic clustering approach is not intended to define stable molecular subtypes or extract latent transcriptional programs. Instead, symbolic clustering provides a coarse-grained, categorical representation of within-gene trajectories across variable deuterium concentrations. Each gene’s expression profile is compressed into a triplet of intuitive symbols (e.g., ↓ → ↑), serving as a visual shorthand for directional behavior.
This symbolic encoding complements the probabilistic assignments generated by Gaussian mixture model (GMM) clustering by highlighting how expression changes unfold across conditions, rather than only which cluster a gene belongs to. The result is a compact, biologically interpretable map of transcriptional dynamics that facilitates rapid recognition of coordinated regulatory programs, identification of sentinel outliers, and cross-pathway comparability. Thus, symbolic clustering functions as a visualization and interpretive overlay, offering accessibility and interpretability beyond conventional clustering frameworks.
The six GMM clusters exhibit distinct transcriptional profiles across exposure levels, with centroid coordinates stratified by decreasing expression at 300 ppm. This stratification reflects biologically meaningful modules, as confirmed by Kyoto Encyclopedia of Genes and Genomes (KEGG) and Reactome pathway enrichment.
The pathway categories shown in Figs. 1 and 2 represent canonical bins used for visualization, where genes are grouped into broad functional classes such as PI3K–AKT axis, RAS–MAPK signaling, cell cycle and replication, or invasion/ECM remodeling. In contrast, the pathway labels assigned to clusters below derive from enrichment analysis of the specific gene sets within each cluster. This approach sometimes introduces more granular or mechanistic terms (e.g., JAK–STAT signaling, FoxO signaling, EMT, hypoxia, focal adhesion) that refine but do not replace the canonical categories. For example, STAT3 in Cluster 1 supports annotation as JAK–STAT signaling, even though in Figure 2 it was grouped under cytokine/inflammatory signaling. Similarly, TGFB1 and S100A4 in Cluster 5 justify annotation as Epithelial–Mesenchymal Transition (EMT), which is a mechanistic subdivision of invasion/ECM remodeling. Thus, the cluster nomenclature should be interpreted as functional refinements of the broader bins in Figure 2, providing higher-resolution modules that capture context-specific behavior under deuterium concentration change.
3.6. Pathway and Hallmark Coherence Among High-Confidence Cluster Members
Cluster 1: Cellular Regulation and Genomic Stability
Pathways: Cell Cycle, PI3K–AKT axis, DNA Repair, JAK–STAT signaling
Hallmarks: Genome Instability & Mutation, Enabling Replicative Immortality
This cluster exhibits the most dramatic response to deuterium enrichment, rising to an expression ratio of 1.66 at 300 ppm. It integrates the TP53-responsive phosphatase CDC25C and the tumor suppressor PTEN with potent transcriptional amplifiers from the Core Network (Figure 1), specifically MYC and STAT3. In A549 cells, which retain a wild-type TP53 axis, this cluster represents a functional checkpoint response coupled with a proliferative surge. The co-clustering of ERBB3 (RTK adaptor) with MYC implies that deuterium enrichment simultaneously fuels upstream growth signals and downstream replication programs, engaging the p53 machinery (PTEN, CDC25C) to maintain genomic integrity under proliferative stress.
Cluster: Numerical identifier for each cluster (1–6) as determined by GMM-6 clustering ordered by the 300 ppm centroid coordinate. Function: Biological role associated with the cluster. 40 ppm, 80 ppm, 300 ppm: Centroid coordinates representing the average normalized gene expression in the cluster at each deuterium concentration, relative to the 150 ppm control. Genes: List of gene symbols assigned to the cluster. A more complete summary of the GMM-6 clusters—including detailed pathway and hallmark associations—is provided in
Table S2.
Cluster 2: KRAS Effector and Hypoxic Response Module
Pathways: RAS–MAPK signaling, Hypoxia, Focal Adhesion
Hallmarks: Sustaining Proliferative Signaling, Activating Invasion & Metastasis
Cluster 2 reveals a critical metabolic vulnerability in the KRAS-driven engine. It displays the steepest suppression at 40 ppm (0.792). Biologically, this cluster contains RAF1 (c-Raf), the direct downstream effector of mutant KRAS, alongside the upstream receptor EGFR. While the G12S mutation constitutively activates KRAS, the monotonic response of this cluster indicates that the effector machinery (RAF1, HSP90AB1) remains highly sensitive to deuterium deprivation. The inclusion of HIF1A and the invasion marker CD44 suggests that depletion imposes a powerful metabolic brake on proliferation and hypoxic adaptation, with suppression magnitude directly correlating with depletion level.
Cluster 3: TP53 Homeostatic Hub
Pathways: p53 Signaling, Cell Cycle, FoxO Signaling
Hallmarks: Evading Growth Suppressors, Deregulating Cellular Energetics
Cluster 3 is characterized by stability at 40 and 80 ppm, acting as a homeostatic core. The central node is TP53. Unlike p53-mutant subtypes where DNA damage responses are uncoupled from growth arrest, the clustering of TP53 with growth drivers—including CCND1, ERBB2, and IGF1—confirms that A549 cells retain a competent, stress-responsive p53 axis. While basal transcriptional load is driven by oncogenic KRAS, this module preserves canonical regulatory architecture, balancing growth signaling (IGF1, JUN) with suppression until overwhelmed at 300 ppm. The observed stability suggests that moderate depletion is perceived not as genotoxic crisis but as metabolic shift, allowing basal p53 surveillance to persist.
Cluster 4: Accessory Growth Signals (“Co-Conspirators”)
Pathways: MAPK signaling, Apoptosis, Nucleotide Excision Repair
Hallmarks: Sustaining Proliferative Signaling, Resisting Cell Death
This cluster is suppressed under depletion (0.820 at 40 ppm), representing the accessory signaling arm. It contains NRAS and MET. In KRAS-mutant tumors, wild-type isoforms like NRAS often act as co-conspirators to support the phenotype. The sensitivity of this cluster highlights synthetic vulnerability: while mutant KRAS is constitutively active, supporting growth factor networks (MET) and anti-apoptotic reserves (BCL2) are energetically costly. Their downregulation under depletion suggests they can be decoupled from the primary driver when energetics are constrained. The presence of XPC (DNA repair) and ETS1 (transcription factor) further links this cluster to maintenance of the oncogenic state outlined in Figure 1.
Cluster 5: Invasion and Phenotypic Plasticity (STK11-Linked)
Pathways: TGF-β signaling, EMT
Hallmarks: Unlocking Phenotypic Plasticity, Activating Invasion & Metastasis
This cluster is critical for the STK11-deficient phenotype of A549 cells, associated with aggressive invasion. Enriched for TGFB1 (contextual tumor promoter) and the metastasis driver S100A4, this module drives EMT. Its trajectory is distinct, showing a dip at 80 ppm relative to 40 ppm, suggesting non-linear sensitivity where specific concentrations disrupt signaling thresholds for plasticity. The presence of HRAS and BCL2L1 (Bcl-xL) indicates that this invasive program is tightly coupled to survival mechanisms distinct from basal machinery.
Cluster 6: Basal Maintenance and Survival Reserve
Pathways: PI3K–AKT signaling, Cell Cycle, DNA Repair
Hallmarks: Resisting Cell Death, Deregulating Cellular Energetics
Cluster 6 exhibits the lowest induction at 300 ppm (1.180), defining it as a basal maintenance reserve. It contains key Core Network hubs: AKT2 and CDK4. In the context of STK11 deficiency, the mTOR/AKT pathway is often hyperactivated. The relative stability of this cluster suggests that constitutive survival signaling is robust. However, suppression at 40 ppm (0.830) indicates that even basal reserves are not immune to extreme depletion. The presence of BIRC5 (Survivin)—typically repressed by wild-type TP53—suggests that oncogenic signaling overrides the p53 brake to ensure viability, while ERCC2 maintains background DNA repair.
Table 2 summarizes the key characteristics of each gene cluster identified by Gaussian Mixture Model (GMM-6) analysis of A549 lung adenocarcinoma cells under varying deuterium concentrations. Each row represents a distinct cluster, defined by its functional annotation, centroid coordinates (mean relative gene expression at 40, 80, and 300 ppm deuterium), and the list of genes assigned to that cluster.
3.7. Conclusions
Our study identifies deuterium concentration as a distinct modulator of the oncogenic transcriptome in A549 lung adenocarcinoma cells. We observe that deuterium enrichment amplifies gene signatures linked to inflammation and invasion, whereas moderate depletion correlates with the systematic downregulation of key survival networks. Specifically, the suppression of ABCB1, FGFR4, and MMP9 under depletion uncovers a potential transcriptomic vulnerability in the multidrug resistance and invasive machinery driven by the KRAS/STK11/KEAP1 genotype. By resolving these dynamics through GMM-6 clustering and DBSCAN outlier detection, we define a transcriptional window where metabolic constraints appear to counteract constitutive oncogenic drivers. Consequently, these data support a model in which deuterium depletion functions not merely as a general stressor, but as a candidate metabolic intervention, offering a transcriptomic rationale for re-sensitizing aggressive, drug-resistant tumors.
At the same time, several limitations constrain the scope of these conclusions. The analysis is based on a single LUAD model (A549) with a specific KRAS/STK11/KEAP1 genotype, and the findings may not generalize to other molecular subtypes such as EGFR-driven, ALK-rearranged, or KEAP1-wildtype tumors. Furthermore, this study is purely transcriptomic; the inferred biological outcomes regarding proliferation, drug response, and invasion require direct functional validation. Moreover, the in vitro conditions used here do not capture the metabolic and microenvironmental constraints present in vivo.
To address these limitations and extend these insights, systematic evaluation across additional LUAD cell lines with diverse mutational constellations is warranted, together with comparative studies in other malignancies such as prostate and ovarian cancers. Parallel profiling of healthy epithelial and stromal cells will be critical for distinguishing tumor-specific vulnerabilities from general metabolic responses. Expanding the range and increasing the number of tested deuterium concentrations will help define threshold effects, dose–response relationships, and context-dependent metabolic rewiring. Finally, integrating transcriptomic datasets with proteomic analyses will provide a deeper mechanistic understanding of how deuterium concentration reshapes signaling networks—potentially by modulating the metabolic substrates required for non-mutational epigenetic reprogramming—thereby refining its potential as a therapeutic axis in oncology.
Figure 1.
Oncogenic core network architecture of selected A549 lung adenocarcinoma genes. The schematic illustrates the functional organization of 102 consensus cancer genes into nine interconnected pathways. Nodes represent functional modules (e.g., RAS–MAPK core, PI3K–AKT axis), while edges depict canonical signaling dependencies. Upstream receptor tyrosine kinases (e.g., EGFR, MET) transmit growth signals to proliferative and survival cascades; Cytokine signaling integrates with transcriptional amplification and invasion; and DNA repair mechanisms anchor the stress tolerance response. Pathways are annotated with representative Hallmarks of Cancer. Though not all hallmarks are displayed to avoid visual overcrowding. The Cell Survival module functionally subdivides into intrinsic apoptosis regulators (BCL2, BCL2A1, BCL2L1), NF-κB/IAP survival signaling (BCL3, BIRC5), and DNA damage checkpoints (TP53). Key drivers such as EGFR and TP53 are positioned as central hubs, reflecting their roles in mediating sustaining Proliferative Signaling and Genome Instability, respectively. Interconnections between pathways (e.g., RTKs → RAS–MAPK, RTKs → PI3K–AKT, RAS–MAPK → cell cycle, PI3K–AKT → apoptosis evasion, cytokine/inflammation → transcriptional amplification and invasion) are shown as canonical specific pathway-oriented knowledge edges. The biological rationale for each edge is detailed in Table S1.
Figure 1.
Oncogenic core network architecture of selected A549 lung adenocarcinoma genes. The schematic illustrates the functional organization of 102 consensus cancer genes into nine interconnected pathways. Nodes represent functional modules (e.g., RAS–MAPK core, PI3K–AKT axis), while edges depict canonical signaling dependencies. Upstream receptor tyrosine kinases (e.g., EGFR, MET) transmit growth signals to proliferative and survival cascades; Cytokine signaling integrates with transcriptional amplification and invasion; and DNA repair mechanisms anchor the stress tolerance response. Pathways are annotated with representative Hallmarks of Cancer. Though not all hallmarks are displayed to avoid visual overcrowding. The Cell Survival module functionally subdivides into intrinsic apoptosis regulators (BCL2, BCL2A1, BCL2L1), NF-κB/IAP survival signaling (BCL3, BIRC5), and DNA damage checkpoints (TP53). Key drivers such as EGFR and TP53 are positioned as central hubs, reflecting their roles in mediating sustaining Proliferative Signaling and Genome Instability, respectively. Interconnections between pathways (e.g., RTKs → RAS–MAPK, RTKs → PI3K–AKT, RAS–MAPK → cell cycle, PI3K–AKT → apoptosis evasion, cytokine/inflammation → transcriptional amplification and invasion) are shown as canonical specific pathway-oriented knowledge edges. The biological rationale for each edge is detailed in Table S1.
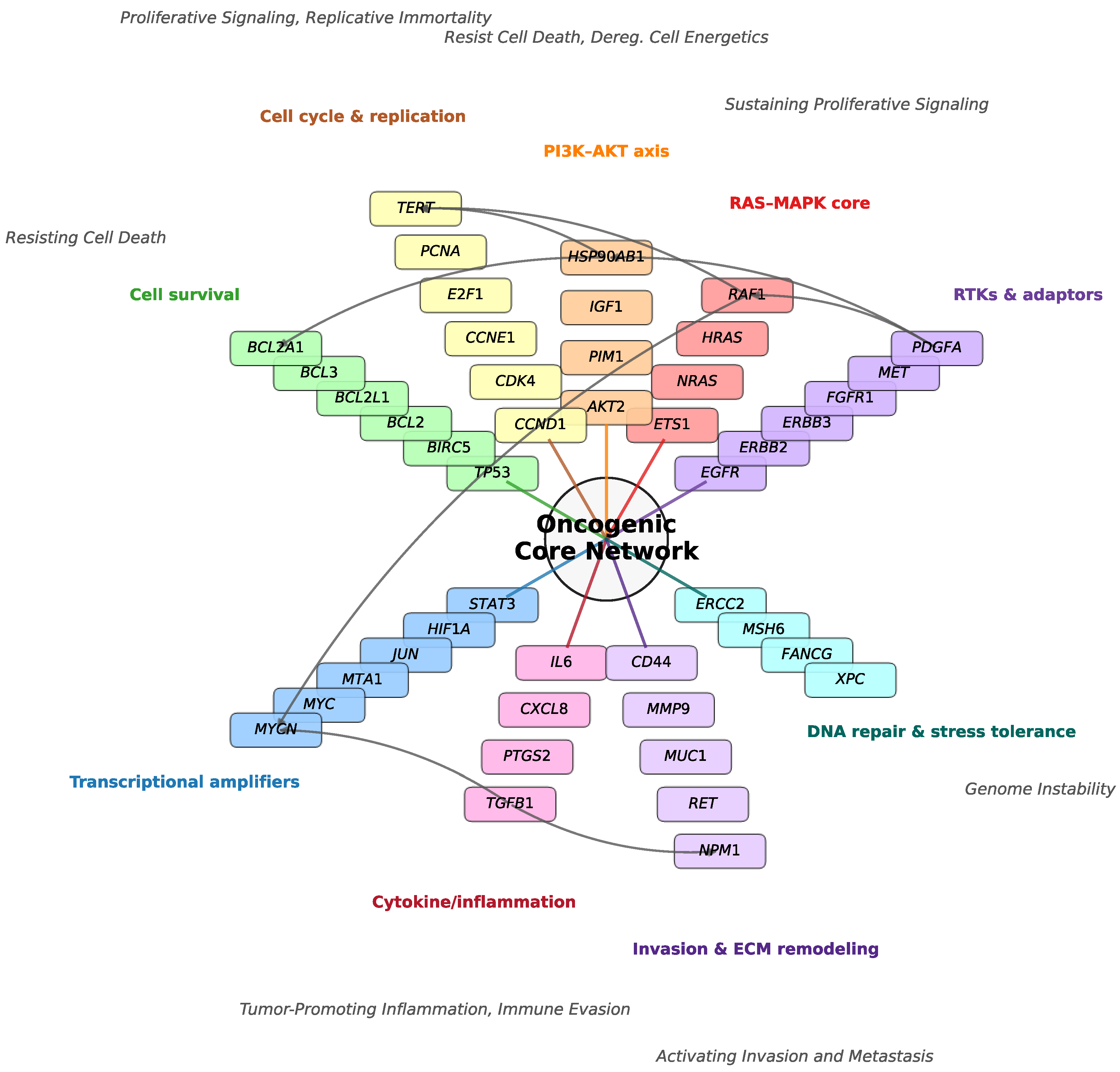
Figure 2.
Three-dimensional classification of consensus cancer genes in A549 cells under variable deuterium concentrations. Scatter plot of 102 genes, each positioned by its expression ratios at 40 ppm (x-axis), 80 ppm (y-axis), and 300 ppm (z-axis), normalized to copy numbers at the 150 ppm control. Genes are color-coded by functional pathway, with the number of genes indicated in parentheses: drug metabolism and transport (n = 1), PI3K–AKT axis (n = 11), cell survival (n = 11), RAS–MAPK signaling (n = 11), cell cycle and replication (n = 16), invasion and extracellular matrix remodeling (n = 12), transcriptional amplifiers (n = 14), receptor tyrosine kinases and adaptors (n = 14), DNA repair and stress response (n = 5), cytokine/inflammatory signaling (n = 7). Genes highlighted in red (e.g., TGFBR2, IL6) are positioned farthest from the global centroid, marking the strongest transcriptional responses to altered deuterium concentration. Pathway centroids are provided in Table 1. This visualization illustrates both the segregation of canonical oncogenic versus stress-response pathways and the identification of sentinel genes with heightened sensitivity to deuterium concentration change.
Figure 2.
Three-dimensional classification of consensus cancer genes in A549 cells under variable deuterium concentrations. Scatter plot of 102 genes, each positioned by its expression ratios at 40 ppm (x-axis), 80 ppm (y-axis), and 300 ppm (z-axis), normalized to copy numbers at the 150 ppm control. Genes are color-coded by functional pathway, with the number of genes indicated in parentheses: drug metabolism and transport (n = 1), PI3K–AKT axis (n = 11), cell survival (n = 11), RAS–MAPK signaling (n = 11), cell cycle and replication (n = 16), invasion and extracellular matrix remodeling (n = 12), transcriptional amplifiers (n = 14), receptor tyrosine kinases and adaptors (n = 14), DNA repair and stress response (n = 5), cytokine/inflammatory signaling (n = 7). Genes highlighted in red (e.g., TGFBR2, IL6) are positioned farthest from the global centroid, marking the strongest transcriptional responses to altered deuterium concentration. Pathway centroids are provided in Table 1. This visualization illustrates both the segregation of canonical oncogenic versus stress-response pathways and the identification of sentinel genes with heightened sensitivity to deuterium concentration change.
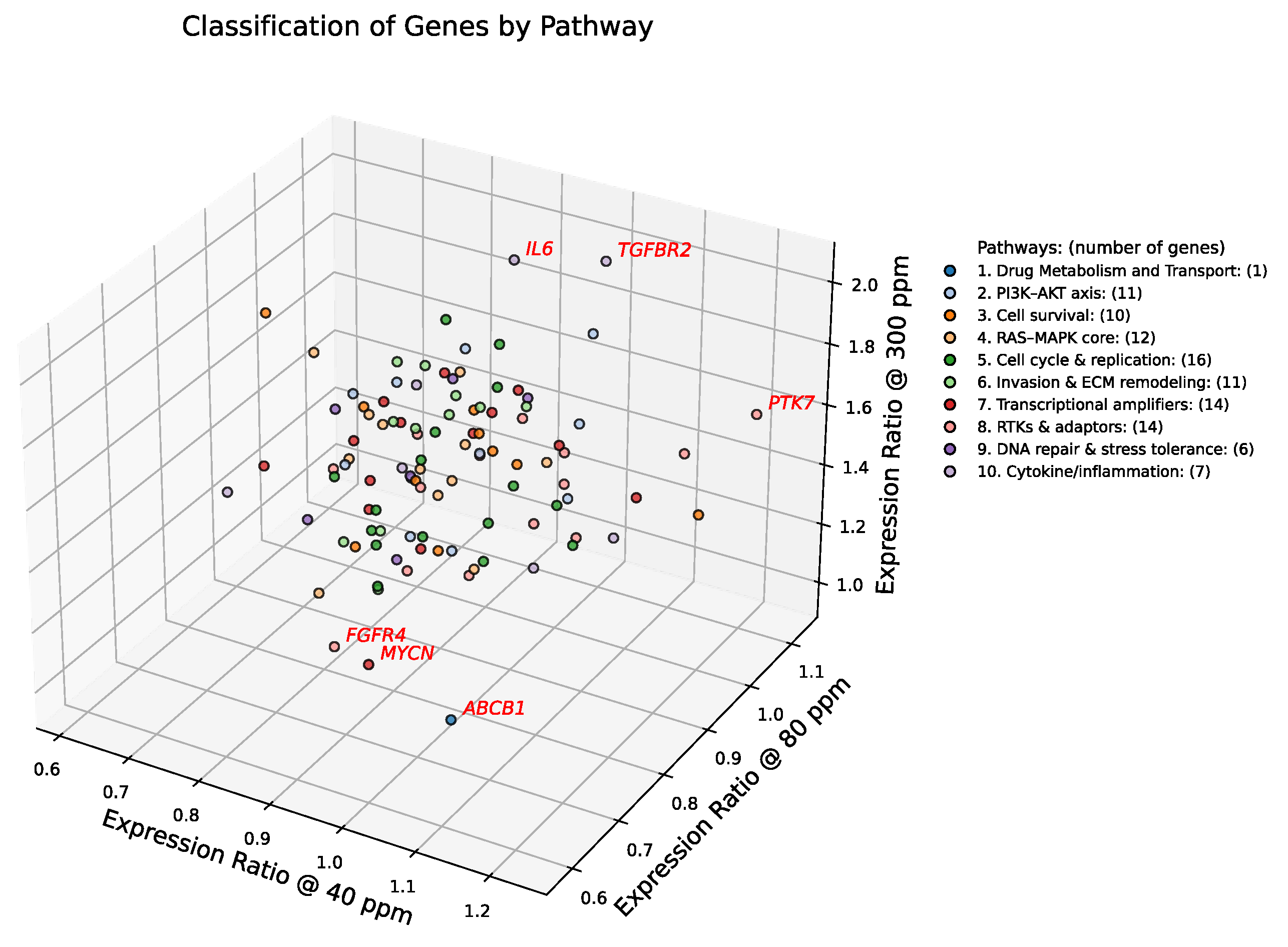
Figure 3.
DBSCAN clustering of deuterium-responsive gene expression in A549 cells. Three-dimensional scatter plot showing expression ratios of 102 consensus cancer genes at 40, 80, and 300 ppm deuterium, normalized to the 150 ppm control. Each axis represents the expression ratio at one concentration. DBSCAN analysis (ε = 0.13, min_samples = 5) separates the dense transcriptional core (blue circles) from 11 sentinel outlier genes (gray X symbols, labeled in italics). Outliers exhibit trajectories that diverge strongly from pathway averages, highlighting stress-sensitive regulators and context-dependent modulators. Notably, these outliers map to distinct Hallmarks of Cancer: ABCB1 (drug resistance; Resisting Cell Death), FGFR4 (growth factor signaling; Sustaining Proliferative Signaling), MYCN (transcriptional amplification; Enabling Replicative Immortality), IL6 and TGFBR2 (inflammation and invasion; Tumor-Promoting Inflammation and Activating Invasion & Metastasis). Cells were cultured for 72 h under the indicated conditions.
Figure 3.
DBSCAN clustering of deuterium-responsive gene expression in A549 cells. Three-dimensional scatter plot showing expression ratios of 102 consensus cancer genes at 40, 80, and 300 ppm deuterium, normalized to the 150 ppm control. Each axis represents the expression ratio at one concentration. DBSCAN analysis (ε = 0.13, min_samples = 5) separates the dense transcriptional core (blue circles) from 11 sentinel outlier genes (gray X symbols, labeled in italics). Outliers exhibit trajectories that diverge strongly from pathway averages, highlighting stress-sensitive regulators and context-dependent modulators. Notably, these outliers map to distinct Hallmarks of Cancer: ABCB1 (drug resistance; Resisting Cell Death), FGFR4 (growth factor signaling; Sustaining Proliferative Signaling), MYCN (transcriptional amplification; Enabling Replicative Immortality), IL6 and TGFBR2 (inflammation and invasion; Tumor-Promoting Inflammation and Activating Invasion & Metastasis). Cells were cultured for 72 h under the indicated conditions.
Figure 4.
Symbolic clustering of gene expression trajectories under graded deuterium concentrations. Three-dimensional scatter plot showing expression ratios of 102 consensus cancer genes at 40, 80, and 300 ppm deuterium, normalized to the 150 ppm control. Genes are color-coded by their symbolic triplet pattern (e.g., ↓ → ↑), with cluster centroids indicated by stars. Patterns are ordered by their expression ratio at 300 ppm (highest to lowest). The legend specifies each triplet and the number of genes (n) per group. Only 14 of 125 theoretical triplets are represented, with Pattern 2 (→ → ↑; n = 44) emerging as the dominant trajectory. See Results for detailed interpretation. Genes highlighted in red (e.g., ABCB1, FGFR4, MYCN) represent unique singleton trajectories, diverging from canonical clusters and marking sentinel regulators of drug resistance, growth factor signaling, and transcriptional amplification.
Figure 4.
Symbolic clustering of gene expression trajectories under graded deuterium concentrations. Three-dimensional scatter plot showing expression ratios of 102 consensus cancer genes at 40, 80, and 300 ppm deuterium, normalized to the 150 ppm control. Genes are color-coded by their symbolic triplet pattern (e.g., ↓ → ↑), with cluster centroids indicated by stars. Patterns are ordered by their expression ratio at 300 ppm (highest to lowest). The legend specifies each triplet and the number of genes (n) per group. Only 14 of 125 theoretical triplets are represented, with Pattern 2 (→ → ↑; n = 44) emerging as the dominant trajectory. See Results for detailed interpretation. Genes highlighted in red (e.g., ABCB1, FGFR4, MYCN) represent unique singleton trajectories, diverging from canonical clusters and marking sentinel regulators of drug resistance, growth factor signaling, and transcriptional amplification.
Figure 5.
Overlap of transcriptional outlier genes identified by three clustering strategies. Venn diagram showing the intersection of genes classified as outliers by three independent approaches: (1, orange) Euclidean distance from the global centroid; (2, green) DBSCAN clustering; and (3, violet) unique singleton symbolic patterns. The overlap defines a consensus core of highly divergent genes (e.g., ABCB1, MYCN, FGFR4), while also illustrating that DBSCAN captures the broadest set of sentinel responders.
Figure 5.
Overlap of transcriptional outlier genes identified by three clustering strategies. Venn diagram showing the intersection of genes classified as outliers by three independent approaches: (1, orange) Euclidean distance from the global centroid; (2, green) DBSCAN clustering; and (3, violet) unique singleton symbolic patterns. The overlap defines a consensus core of highly divergent genes (e.g., ABCB1, MYCN, FGFR4), while also illustrating that DBSCAN captures the broadest set of sentinel responders.
Figure 6.
Gaussian mixture clustering of deuterium-responsive genes in A549 cells. Three-dimensional scatter plot of 91 genes, each positioned by its expression ratios at 40, 80, and 300 ppm relative to the 150 ppm control. Gaussian mixture modeling with tied covariance ellipsoids (k = 4) partitions the dataset into four clusters, shown with color-coded membership and corresponding covariance ellipsoids. The legend reports the number of genes per cluster. Genes with the greatest Mahalanobis distance from their cluster centroids (e.g., BCL2A1, PTEN) are highlighted in red, denoting boundary outliers with divergent transcriptional trajectories.
Figure 6.
Gaussian mixture clustering of deuterium-responsive genes in A549 cells. Three-dimensional scatter plot of 91 genes, each positioned by its expression ratios at 40, 80, and 300 ppm relative to the 150 ppm control. Gaussian mixture modeling with tied covariance ellipsoids (k = 4) partitions the dataset into four clusters, shown with color-coded membership and corresponding covariance ellipsoids. The legend reports the number of genes per cluster. Genes with the greatest Mahalanobis distance from their cluster centroids (e.g., BCL2A1, PTEN) are highlighted in red, denoting boundary outliers with divergent transcriptional trajectories.
Figure 7.
Gaussian mixture clustering of deuterium-responsive genes in A549 cells. Three-dimensional scatter plot of 91 genes positioned by their relative expression ratios at 40, 80, and 300 ppm compared to the 150 ppm control. Gaussian mixture modeling with tied covariance ellipsoids (k = 6) partitions the dataset into six optimal clusters, selected based on silhouette analysis and biological coherence. Genes and covariance ellipsoids are color-coded by cluster membership, with cluster sizes reported in the legend. Genes exceeding a Mahalanobis distance of 2.42 from their cluster centroid (e.g., BCL2A1, PTEN) are highlighted in red, denoting boundary outliers that extend beyond the canonical definition of the transcriptional module.
Figure 7.
Gaussian mixture clustering of deuterium-responsive genes in A549 cells. Three-dimensional scatter plot of 91 genes positioned by their relative expression ratios at 40, 80, and 300 ppm compared to the 150 ppm control. Gaussian mixture modeling with tied covariance ellipsoids (k = 6) partitions the dataset into six optimal clusters, selected based on silhouette analysis and biological coherence. Genes and covariance ellipsoids are color-coded by cluster membership, with cluster sizes reported in the legend. Genes exceeding a Mahalanobis distance of 2.42 from their cluster centroid (e.g., BCL2A1, PTEN) are highlighted in red, denoting boundary outliers that extend beyond the canonical definition of the transcriptional module.
Figure 8.
Sankey diagram of gene cluster transitions between GMM-4 and GMM-6 models. Sankey diagram illustrating how gene memberships redistribute when Gaussian mixture modeling is expanded from four clusters (left) to six clusters (right). Each node represents a cluster, labeled by ID (e.g., 4-1) and gene count. Colors correspond to the original GMM-4 assignments, and flow widths are proportional to the number of genes transitioning between clusters. Increasing model granularity partitions the large, heterogeneous GMM-4 Cluster 4 (blue) into three distinct functional submodules (GMM-6 Clusters 4, 5, and 6), thereby separating invasive and proliferative drivers from basal maintenance genes.
Figure 8.
Sankey diagram of gene cluster transitions between GMM-4 and GMM-6 models. Sankey diagram illustrating how gene memberships redistribute when Gaussian mixture modeling is expanded from four clusters (left) to six clusters (right). Each node represents a cluster, labeled by ID (e.g., 4-1) and gene count. Colors correspond to the original GMM-4 assignments, and flow widths are proportional to the number of genes transitioning between clusters. Increasing model granularity partitions the large, heterogeneous GMM-4 Cluster 4 (blue) into three distinct functional submodules (GMM-6 Clusters 4, 5, and 6), thereby separating invasive and proliferative drivers from basal maintenance genes.
Table 1.
Centroid coordinates of pathway clusters in A549 lung adenocarcinoma cells cultured for 72 h under graded deuterium concentrations.
Table 1.
Centroid coordinates of pathway clusters in A549 lung adenocarcinoma cells cultured for 72 h under graded deuterium concentrations.
| Pathway |
40 ppm |
80 ppm |
300 ppm |
Example Genes |
| Cytokine/inflammation |
0.908 |
0.955 |
1.567 |
IL6, CXCL8, PTGS2 |
| Cell survival |
0.874 |
0.936 |
1.476 |
TP53, BCL2, BIRC5 |
| Invasion & ECM remodeling |
0.847 |
0.923 |
1.469 |
CD44, MMP9, MUC1 |
| PI3K–AKT axis |
0.880 |
0.971 |
1.468 |
AKT2, PIM1, IGF1 |
| Transcriptional amplifiers |
0.868 |
0.931 |
1.442 |
MYC, MYCN, STAT3 |
| RAS–MAPK core |
0.842 |
0.954 |
1.440 |
HRAS, NRAS, RAF1 |
| Global center |
0.884 |
0.940 |
1.436 |
|
| DNA repair & stress tolerance |
0.842 |
0.940 |
1.402 |
ERCC2, MSH6, XPC |
| RTKs & adaptors |
0.959 |
0.948 |
1.377 |
EGFR, ERBB2, FGFR1 |
| Cell cycle & replication |
0.885 |
0.940 |
1.371 |
CCND1, TERT, CDK4 |
| Drug Metabolism and Transport |
1.122 |
0.585 |
1.268 |
ABCB1 |
Table 2.
Summary of GMM-6 Cluster Centroids, Functional Annotations, and Gene Composition in A549 Cells Under Variable Deuterium Concentrations.
Table 2.
Summary of GMM-6 Cluster Centroids, Functional Annotations, and Gene Composition in A549 Cells Under Variable Deuterium Concentrations.
| Cluster |
Function |
40 ppm |
80 ppm |
300 ppm |
Genes |
| 1 |
Cellular Regulation and
Genomic Stability |
0.906 |
0.984 |
1.666 |
CDC25C, ERBB3, ITGB1, LAMB1, MSH6, MYC, NQO1, PPARG, PTEN, RB1, STAT3, TERT, TFRC, TGFBI, TIMP2, TOP1, XRCC5 |
| 2 |
KRAS Effector and Hypoxic
Response Module |
0.792 |
0.929 |
1.565 |
BCL2A1, CCNE1, CD44, EGFR, ETV6, FAT1, GADD45A, HIF1A, HSP90AB1, CXCL8, KMT2A, PGK1, PLAUR, RAD54L, RAF1, SFPQ, TPR, YY1 |
| 3 |
TP53 Homeostatic Hub |
0.982 |
0.975 |
1.460 |
CAV1, CCND1,
CEBPA, ERBB2, FOSL2, IGF1, JUN, MST1R, PCNA, PIM1, RARA, SIAH1, TP53
|
| 4 |
Accessory Growth Signals (“Co-Conspirators”) |
0.820 |
0.952 |
1.343 |
BCL2, BCR, CDC25B, ETS1, ETS2, FAS, FGF2, IGFBP3, IRF1, LYN, MET, NRAS, PDGFA, PTPRG, SERPINE1, TYMS, XPC |
| 5 |
Invasion and Phenotypic
Plasticity (STK11-Linked) |
0.957 |
0.903 |
1.264 |
BCL2L1, CCND3, CSK, HRAS, IGFBP2, NUMA1, S100A4, TGFB1, TYRO3 |
| 6 |
Basal Maintenance and Survival Reserve |
0.830 |
0.897 |
1.180 |
AKT2, BIRC5, CDK2, CDK4, CDKN1A, CDKN2C, E2F1, ERCC2, FANCG, FGFR3, MTA1, MUC1, MYBL2, NPM1, CDK16, RET, TFE3 |