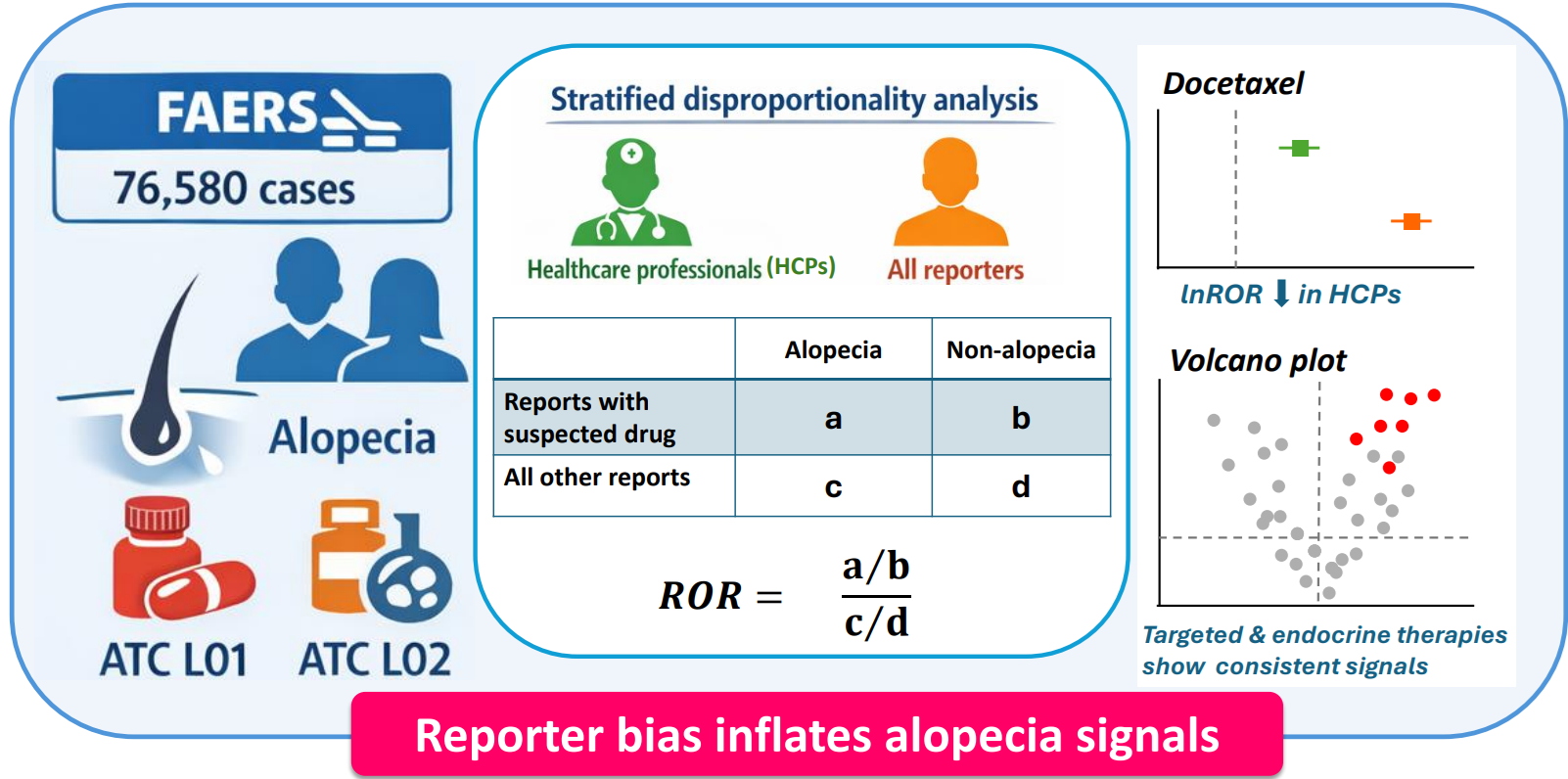
Background/Objectives: Cancer therapy–induced alopecia (CTIA) profoundly affects patients’ quality of life. This study conducted a disproportionality analysis of CTIA using the FDA Adverse Event Reporting System (FAERS) database to provide an overview of drug-specific signal distributions by systematically evaluating the impact of reporter type on CTIA signal detection. Methods: FAERS data from January 2004 to September 2024 were analyzed to extract alopecia-related Preferred Terms included under the Medical Dictionary for Regulatory Activities High Level Term “Alopecias.” Reporting odds ratios (RORs) were calculated to assess disproportionality. A primary analysis including all reports and a stratified analysis restricted to reports submitted by healthcare professionals (HCPs) were performed. No individual case-level clinical review was conducted. Results: Approximately 90% of alopecia reports were associated with female patients, and approximately 40% of these reports were linked to breast cancer. In the disproportionality analysis including all reporters, the highest ROR [95% confidence interval (CI)] was observed for docetaxel [58.31 (57.46–59.17)]. In the analysis restricted to HCP reports, the highest ROR was observed for vismodegib [23.92 (21.86–26.17)], whereas that for docetaxel markedly decreased to 3.68 (3.48–3.89). For molecular targeted agents, statistically significant signals were maintained even in the HCP-restricted analysis. Conclusions: Reporter characteristics substantially influence the detection of alopecia signals, with patients amplifying signals reflecting psychological harm and HCPs amplifying signals reflecting pharmacological plausibility. These findings should be interpreted as hypothesis-generating and warrant further validation using prospective or clinical datasets.