Submitted:
10 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
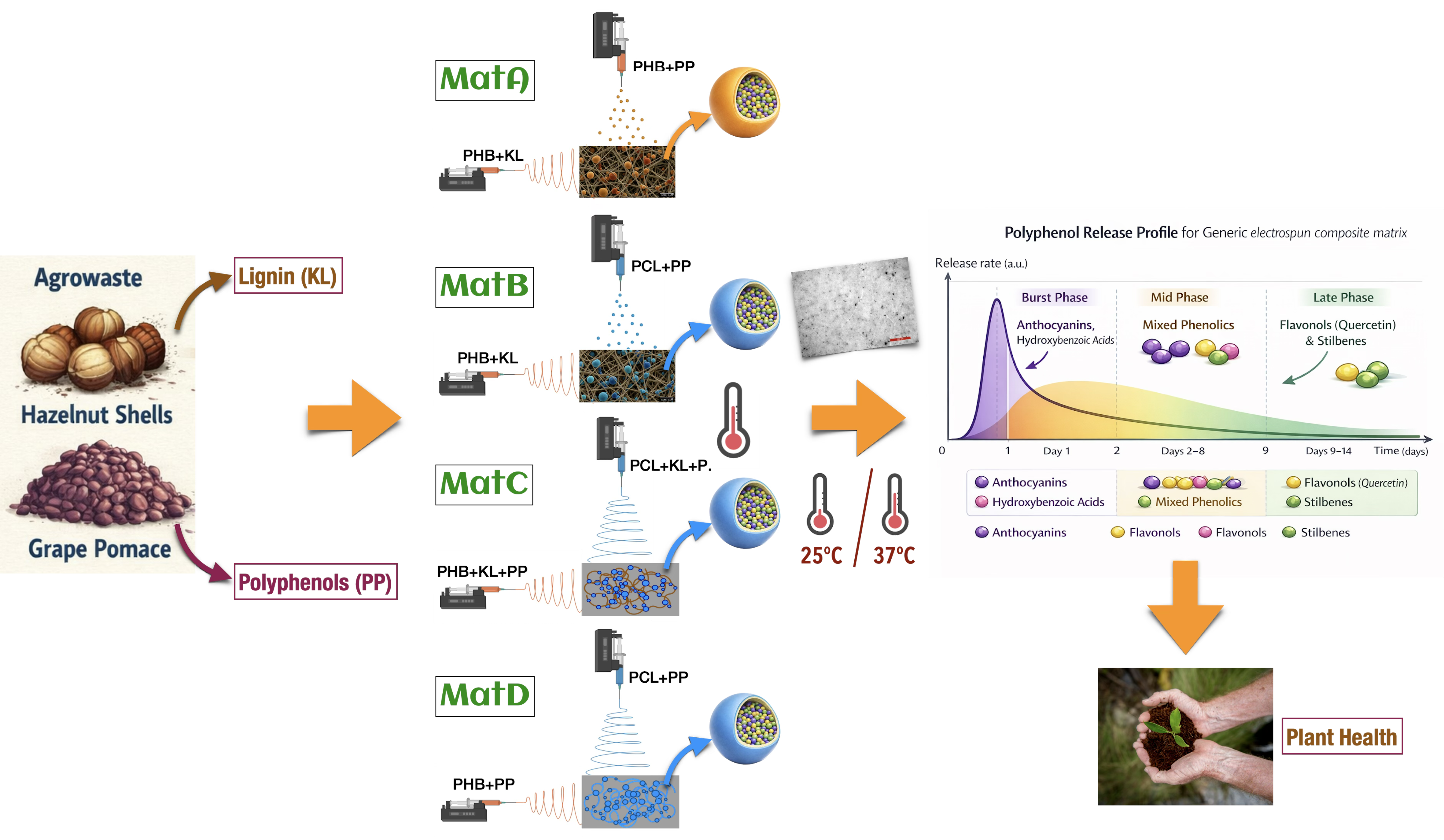
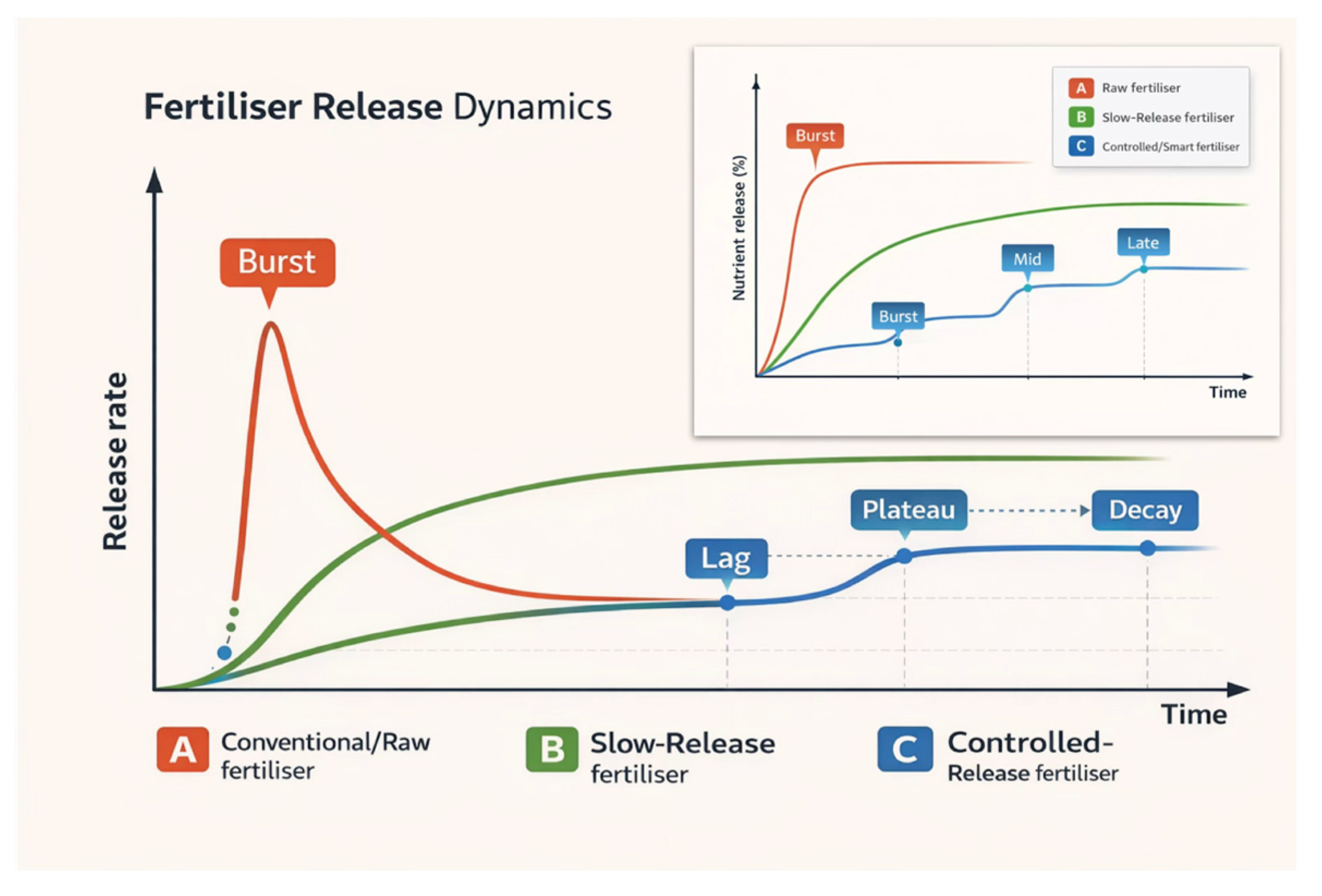
Electrospinning and electrospraying nanotechnologies were used to valorise agro-industrial residues into biohybrid controlled-release polyphenol (CRP) scaffolds. Four polyhydroxybutyrate ± polycaprolactone (PHB±PCL) architectures were fabricated that differed in polymer phase, Klason lignin from hazelnut-shell (HS-KL) presence vs absence and co-location with grape-pomace polyphenols (GP-PP), as well as distribution between fibres and bead-like depots. Scaffolds were characterised using optical microscopy/stereomicroscopy/SEM, FTIR, UV/VIS spectroscopy and dynamic water contact angle (absorption). GP-PP release was monitored for 14 days at ~25 °C and 37 °C, the latter representing shallow-soil hot-spell conditions in Mediterranean zones. All matrices exhibited multimodal release, with modest initial bursts and three phases (burst, mid, and late tail), analogous to controlled-release fertiliser profiles. At ~25 °C, the PHB/PCL matrix with HS-KL confined to PHB fibres and GP-PP in large PCL beads showed the highest total GP-PP release, whereas the architecture with HS-KL and GP-PP co-located in both PHB and PCL fibres and in PCL depots combined high total release with a smoother, well-metered late phase. At 37 °C, this HS-KL-GP-PP co-located scaffold was the most robust, retaining the highest total and late tail release. These results identify HS-KL-GP-PP co-located PHB/PCL architectures as promising carriers for temperature-resilient delivery of bioactive polyphenols in Mediterranean agrosystems.
Keywords:
1. Introduction
1.1. Nanotechnological Contributions to Agriculture — Electrodeposition Technologies
1.2. Lignin
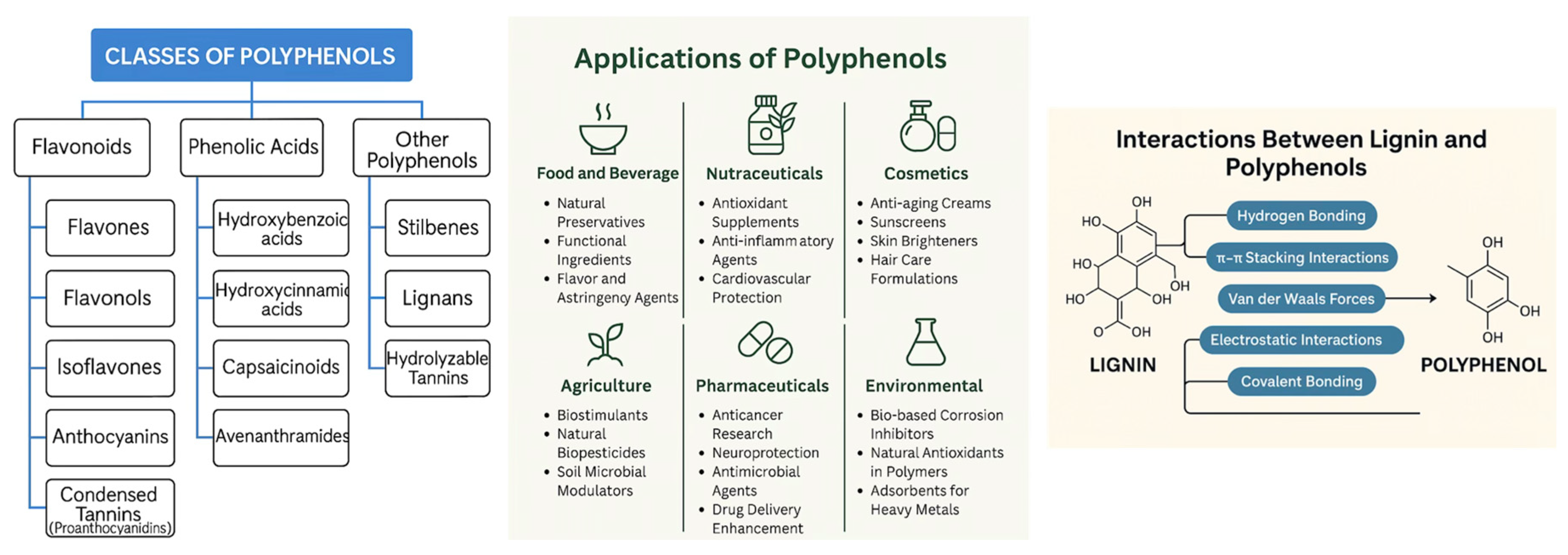
1.3. Polyphenols
1.4. Carriers
1.5. Aims
2. Materials and Methods
2.1. Materials
2.1.1. Lignin Extraction
2.1.2. Polyphenol Extraction
2.2. Electrospinning and Electrospraying Solutions
2.3. Electrospinning/Electrospraying of Nanostructured Frameworks
2.4. Morphological Characterisation
2.4.1. Stereomicroscopy and Optical Microscopy
2.4.2. Scanning Electron Microscopy and Image Analysis
2.5. Interactions of the Nanohybrid Scaffolds with Water
2.5.1. Water Contact Angle (WCA) Measurements
2.5.2. Water Absorption/Infiltration
2.6. Spectroscopic Characterisation
2.6.1. UV–VIS Spectroscopy
2.6.2. Fourier Transform Infrared Spectroscopy (FTIR-ATR)
2.7. Polyphenol Release from the Nanostructured Scaffolds
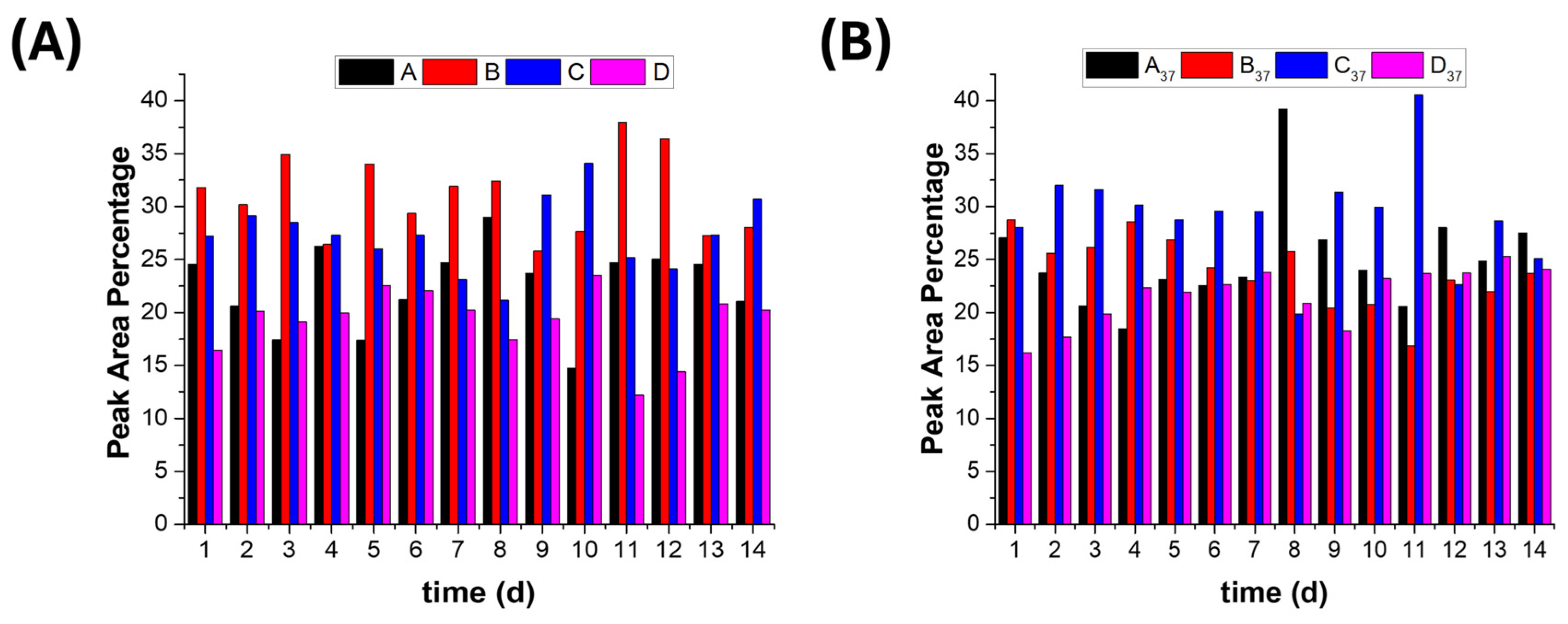
2.7.1. Daily Release Normalisation and Comparison
2.7.2. Polyphenol Release Rate Trend Analysis
3. Results and Discussion
3.1. Extraction and Characterisation of Valuable Bio-Based Compounds from Agro-Industrial Waste
3.1.1. Valuable Compounds from Agro-Industrial Waste for Functional Agricultural Purposes
3.1.2. Lignin Extraction
3.1.3. Polyphenol Extraction
3.1.4. Valuable Spectroscopic Characterisation of the Extracted Substances
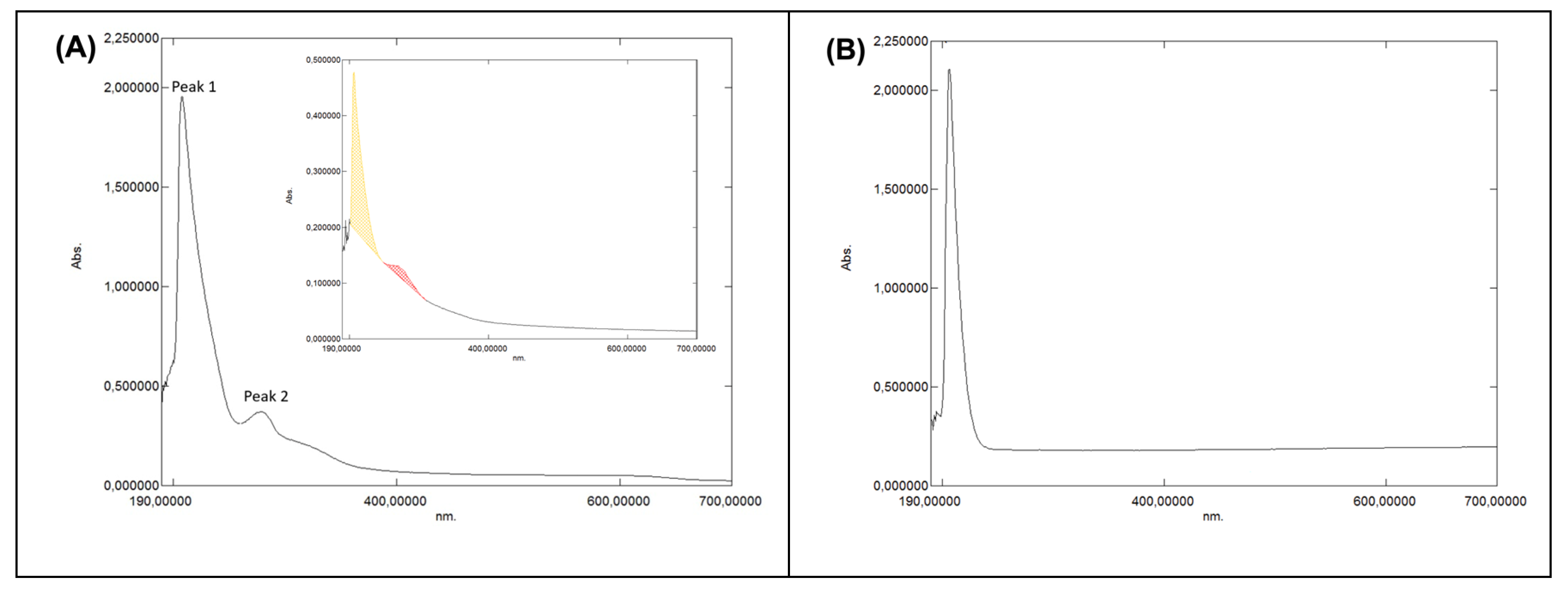
3.1.4.1. UV–VIS Characterisation of the Grape-Pomace Extract
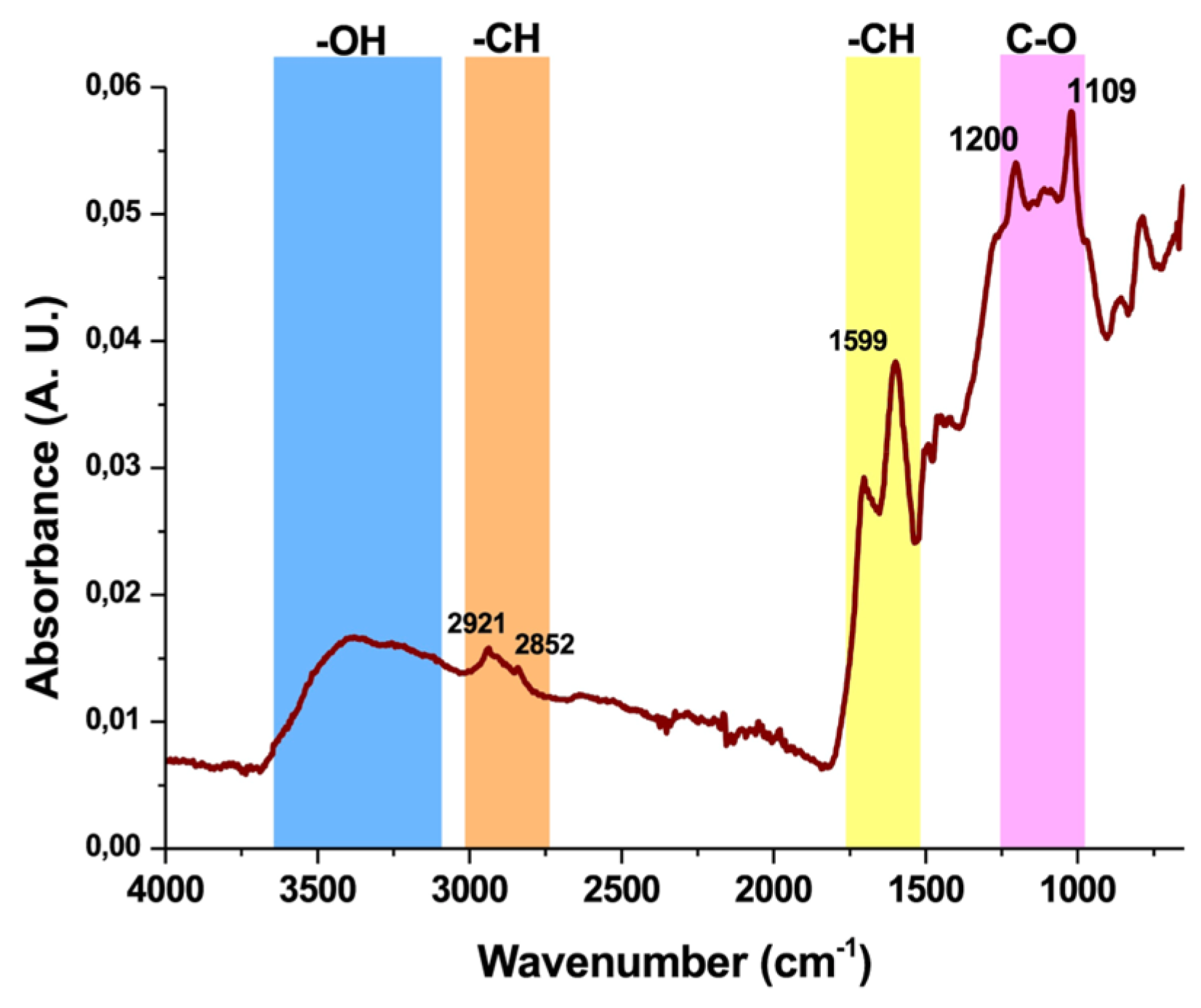
3.1.4.3. FTIR-ATR Characterisation of Klason Lignin from Hazelnut Shells
3.2. Structural Characterisation of Biohybrid Nanocomposites
3.2.1. Design Logic and Matrix Composition
- Polymer phase organisation: single-polymer fibrous networks (PHB) versus multiphase fibrous architectures obtained by the co-deposition of PHB and PCL from separate electrospinning nozzles;
- Localisation of HS-KL: confined to PHB fibres vs distributed across both PHB and PCL phases vs absent;
- Localisation of GP-PP: restricted to bead-like depots vs distributed between fibrous networks and bead-like structures
- Architecture: fibrous networks alone or combined with bead-type depots of different sizes and loading
- MatA is a PHB-only scaffold. HS-KL is confined to electrospun PHB fibres, whereas GP-PP are loaded into PHB particles generated by electrospraying (co-deposition). Lignin and polyphenols do not co-exist in the same domains.
- MatB is a PHB/PCL composite. PHB+HS-KL fibres form the structural network, whereas large PCL+GP-PP particles produced by electrospraying serve as the primary depots (co-deposition). Again, HS-KL and GP-PP reside in different polymer phases.
- MatC contains both PHB and PCL fibres and PCL bead-on-string segments (co-deposited) in which HS-KL and GP-PP are co-located. Both polymers, therefore, act as carriers for HS-KL-GP-PP microdomains distributed across fibres and beads.
- MatD has the same PHB/PCL architecture as MatC but contains GP-PP only, with no HS-KL. It provides a reference system in which polyphenols function solely as cargo and plasticisers, without lignin-mediated metering.
3.2.2. Polymer Phase and MAF/RAF Microstructure
3.2.3. Lignin as a Structural and Interfacial Modifier
3.2.4. Polyphenol Mixture and Expected Partitioning
3.2.5. Architecture, Porosity and Transport Pathways
3.3. Morphological Characterisation of the Bio-Based Nanohybrids
3.3.1. Stereomicroscopy
3.3.1. Optical Microscopy
3.3.3. SEM Analysis: Fibres and Particles Before and After Soaking
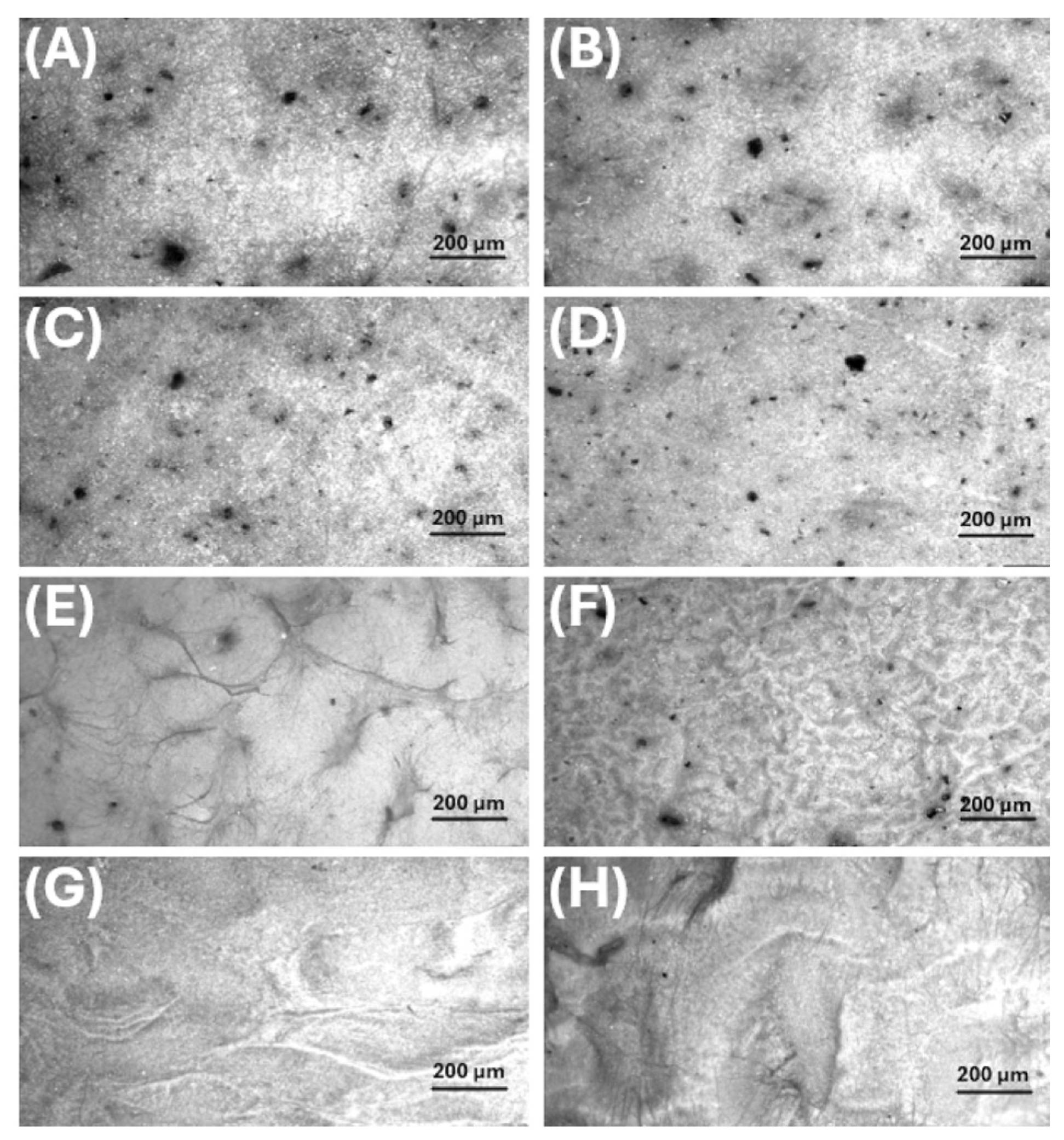
- MatA (PHB+HS-KL fibres, PHB+GP-PP beads). The pristine MatA consisted of a randomly oriented PHB fibre network (mean diameter ≈1.0 µm, CV ≈2%) decorated by relatively small PHB-based particles (mean area ≈1.0 µm2, CV ≈42%) distributed along fibres of different sizes (Figure 10A, 11A; Table 4). The overview images showed an open network with visible inter-fibre voids and bead-like elements anchored to both thicker fibres and very thin nanofibres, indicating strong adhesion between electrosprayed PHB particles and electrospun PHB fibres (Figure 8A inset). After 14 days in 0.11 M phosphate buffer (pH 7.4) at T37, fibres in MatA were clearly swollen: the mean diameter increased by ~32%, and inter-fibre spaces narrowed, consistent with water uptake and partial relaxation of the PHB network (Figure 9B). In contrast, the average particle area and particle roundness changed only minimally (~6%), and total particle area increased slightly (~3%), indicating that PHB beads behaved as soft, shape-preserving depots rather than densifying entities (Table 4). Morphologically, MatA thus evolves towards a more compact fibrous mesh with stable, small PHB depots.
- MatB (PHB+HS-KL fibres, large PCL+GP-PP beads). In MatB, PHB fibres (mean diameter ‚ ≈1.2 µm) formed a porous, multi-diameter network similar in scale to that of MatA but interspersed with large PCL-based spherical particles (mean area ≈7.6 µm2), often partially fused or embedded in a surrounding polymer matrix (Figure 10B, 11C; Table 4). At low magnification, these beads dominated the architecture, producing a pronounced fibre-bead multi-scale structure (Figure 8B inset). Very thin PCL nanofibres, originating from the electrosprayed PCL solution, were occasionally observed but were excluded from the diameter statistics (Figure 10B, 11C). Upon 14-day soaking in 0.11 M phosphate buffer (pH 7.4), PHB fibres in MatB swelled moderately (+17%), and some large fibres showed surface roughening or local damage, suggesting structural relaxation and mild degradation (Figure 9D; Table 4). The small PCL nanonet visible in the pristine material was no longer detectable, suggesting its collapse or dissolution. The most striking change concerned the PCL particles: their mean area and total particle area both decreased by ~36%, while roundness remained essentially constant. This pattern indicates significant in-place densification and shrinkage of PCL depots rather than their detachment. MatB, therefore, shifts from a fibre-supported network with large, soft depots to a structure with swollen fibres and more compact PCL beads.
- MatC (PHB+PCL fibres with HS-KL+GP-PP, PCL+HS-KL+GP-PP beads-on-string). MatC was produced by co-electrospinning PHB and PCL solutions containing both HS-KL and GP-PP, yielding a single, integrated fibrous architecture in which PHB and PCL fibres were physically entangled (Figure 10C, 11E). The mean fibre diameter was much smaller than in A and B (≈0.23 µm, 3-5-fold thinner), and the network appeared dense and homogeneous (CV ≈26%) (Table 4). Rounded and ellipsoidal features along PCL fibres were interpreted as bead-on-string structures (mean area ≈0.8 µm2, CV ≈58%), providing a fine population of depots embedded in the fibrous matrix. Top-view images emphasised a compact, membrane-like appearance with limited apparent surface porosity (Figure 8C inset). After 14-day soaking in 0.11 M phosphate buffer (pH 7.4), fibre diameter increased only slightly (+5%), and fibres showed limited coalescence and mild surface roughening, confirming a restrained swelling behaviour (Figure 9F). The average bead area remained essentially unchanged (+0.5%), but total bead area dropped by about 22%, indicating the loss or detachment of a fraction of bead-on-string segments rather than bead shrinkage (Table 4).
- MatD (PHB+GP-PP and PCL+GP-PP fibres, PCL+GP-PP beads, no lignin). MatD, co-electrospun from PHB+GP-PP and PCL+GP-PP solutions, comprised an open, highly entangled fibrous mesh with thin fibres (mean diameter ≈0.28 µm) and PCL-based beads (mean area ≈2.8 µm2) that were less embedded and more superficially located than in MatC (Figure 10D, 11G; Table 4). Low-magnification views revealed a looser network with greater apparent porosity and lower internal cohesion (Figure 8D inset). Prolonged soaking in 0.11 M phosphate buffer (pH 7.4) for 14 days induced the most dramatic changes among all scaffolds. Fibre diameters increased by about 175%, and the finer nanofibrous elements largely disappeared, replaced by thickened, swollen strands and partially collapsed bundles (Figure 9H; Table 4). The mean bead area and total bead area decreased by ~23%, and bead roundness dropped (~−7%), consistent with notable surface erosion and bead densification within a strongly plasticised network. Morphologically, MatD evolves into a swollen, partially collapsed structure with fewer, more irregular depots and a substantially altered pore architecture.
3.3.4. Role of Matrix Components in Scaffold Architecture and Stability
- Carrier polymers (PHB vs PCL): Comparing the four scaffolds highlights the distinct roles of PHB and PCL as carrier phases. PHB-based fibres in Mats A and B formed the thickest backbones and exhibited moderate swelling at T37, whereas the mixed PHB/PCL fibres in Mats C and D were initially much thinner and thus more prone to dimensional change when plasticised. However, the presence or absence of lignin and GP-PP strongly modulated this tendency: MatC, with HS-KL+GP-PP in both PHB and PCL phases, showed only minimal fibre thickening, while MatD, with GP-PP only, displayed extreme swelling. PCL-based depots were systematically larger than PHB-based ones (MatB > MatD > MatC > MatA) and, in MatB and MatD, underwent clear shrinkage on 14-day soaking in 0.11 M phosphate buffer (pH 7.4), indicating significant densification. In MatC, by contrast, bead size was preserved, and the reduction in total bead area resulted from loss rather than compaction.
- Role of lignin: Lignin influenced both morphology and stability. In fibres, its presence in MatA–MatC was associated with limited swelling upon 14-day soaking, whereas the lignin-free MatD exhibited pronounced fibre thickening and partial collapse of the nanofibrous network. This is consistent with lignin stiffening the amorphous phase of PHB and PCL and resisting water-driven expansion. In beads, the effect was more nuanced. PHB+GP-PP depots in MatA (no lignin) retain their size; PCL+GP-PP depots in MatB (no lignin) shrank markedly; PCL(HS-KL+GP-PP) beads in MatC largely retained their size but suffered some detachment.
- Role of polyphenols: Polyphenols behave as both cargo and internal plasticiser. When they are widely distributed in fibres and beads without lignin (MatD), their largely hydrophilic character favours water uptake and chain mobility, leading to strong fibre swelling, bead erosion and a looser, partly collapsed architecture (pore narrowing and increased tortuosity). When co-localised with lignin (MatC), they instead contributed to more compact fibres and beads, consistent with the formation of lignin–polyphenol complexes reported in the literature. In depots, GP-PP combined with PCL generates very large beads in MatB that subsequently densify on 14-day soaking at T37, whereas GP-PP co-loaded with lignin in PCL beads (MatC) yields smaller, dimensionally stable depots (for HS-KL-GP-PP complexes), whose main change is partial loss from the web rather than shrinkage, with bead loss providing an additional pathway for cargo release. In PHB+GP-PP beads (MatA), the GP-PP load does not drive significant compaction, and the depots are small and soft.
3.4. Interactions Between Nanohybrid Scaffolds and Water
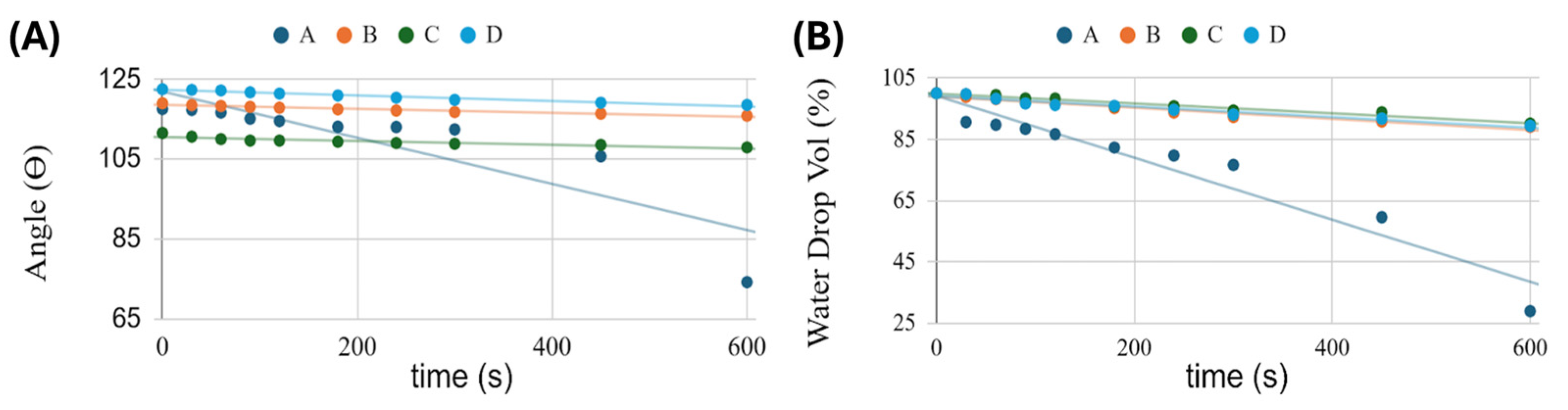
3.4.1. Early Wetting and Hydration (0–10 min, TA)
3.4.2. Long-Term Hydration at Ambient Temperature: Conceptual Extrapolation
- MatA - The PHB+HS-KL fibrous backbone with small PHB+GP-PP depots is expected to enter a moderately swollen state, with fibres thickening and inter-fibre pores narrowing, but without strong depot densification. The overall morphology should remain relatively open compared with its T37 counterpart, supporting a balanced burst-mid-late profile with a modest total release;
- MatB - Large PCL+GP-PP depots that sharply densify at T37 will hydrate and reorganise more slowly at TA, so secondary crystallisation and volume loss are likely incomplete after 14 days. PHB+HS-KL fibres will still swell to some extent, but pore constriction will be less pronounced than at T37. This is compatible with MatB showing the highest total and late release at TA: the depots are plasticised but not yet fully compacted, and a substantial mobile amorphous volume remains;
- MatC - At T37, MatC shows minimal fibre swelling and a reduction in total bead area mainly due to detachment of some PCL(HS-KL+GP-PP) bead-on-string segments. At TA, HS-KL-GP-PP complexes will still limit swelling and chain mobility, while mechanical stresses are milder; fibre diameters should increase only slightly, and bead detachment should be less extensive. The hydrated architecture after 14 days at TA is therefore expected to stay close to the original, compact state, with a well-preserved population of HS-KL-GP-PP depots. This matches the high total release and particularly strong late contribution observed for MatC at TA.
- MatD - The GP-PP-only PHB/PCL network that swells dramatically and partially collapses at T37 will also move towards a swollen, more tortuous morphology at TA, but with less extreme pore closure. Fibres will still thicken more than in the lignin-containing matrices, and PCL+GP-PP beads will densify, yet the lower temperature implies reduced swelling and compaction. This is consistent with MatD remaining the weakest releaser at both temperatures: even under gentler conditions, a sizeable fraction of GP-PP remains trapped in a highly plasticised, partially collapsed network
3.4.3. Morphological Evolution Under Prolonged Hydration (14 d, 37 °C)
3.5. Polyphenol Release from the Biohybrid Nanostructures
3.5.1. Release Profiles at Ambient Temperature (TA)
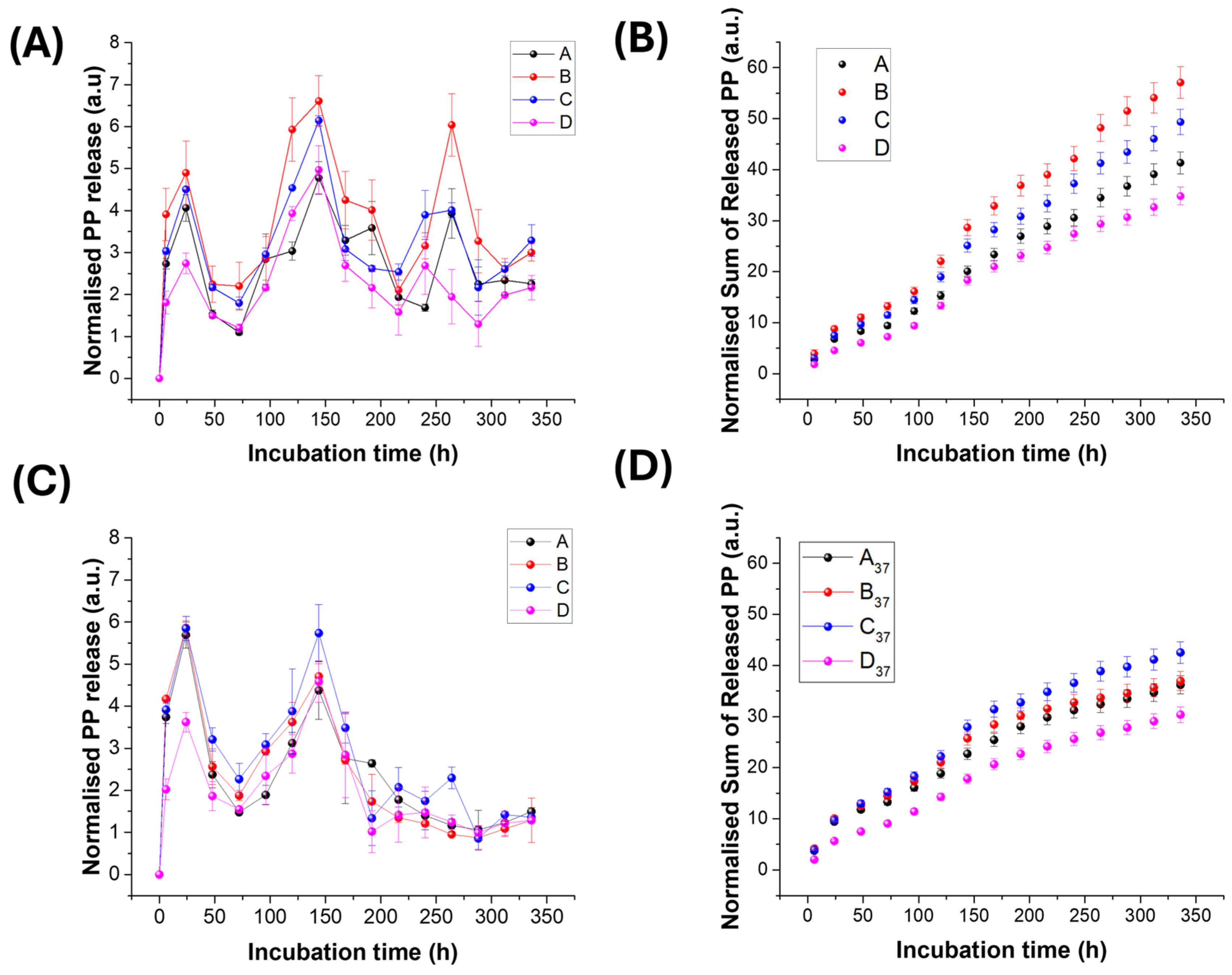
- MatA (PHB+HS-KL fibres + PHB+GP-PP depots, no HS-KL-GP-PP co-location). MatA delivered an intermediate total amount (41.4 a.u.; Table 5), with a modest burst, a moderate mid contribution and a medium late tail. The cumulative curve is clearly multimodal but with a relatively shallow slope (Figure 11B). The latest t50 (≈148 h; Table 5) of MatA reflects diffusion through a PHB-only network in which HS-KL stabilises the fibres but does not directly meter the GP-PP depots. Release is governed by slow diffusion from PHB beads and by the porosity set by fibre diameter and bead coverage. Overall, MatA behaves as a conservative, low-dose scaffold with a relatively delayed response.
- MatB (PHB+HS-KL fibres + large PCL+GP-PP depots, segregated HS-KL and GP-PP). At TA, MatB was the most productive matrix overall (total = 57.1 a.u.; Table 5), combining the largest burst (13.3 a.u.), mid (23.7 a.u.) and late (20.2 a.u.) areas. Although its burst was the strongest among the four matrices, it still represented only a minority of the total release, with most of the mass delivered in the mid and late windows. The cumulative curve for B therefore rises steeply but not explosively and clearly dominates those of the other scaffolds (Figure 11B). This behaviour reflects its architecture: PHB+HS-KL fibres provide a relatively stable supporting network, while the large PCL+GP-PP depots act as high-capacity, MAF-rich sources that are easily hydrated and drained at TA. Because HS-KL resides only in the PHB phase, it primarily stabilises the fibrous mesh rather than metering GP-PP within the PCL beads; as a result, MatB functions as a high-throughput system with strong mid and late contributions, suitable where a relatively high dose of polyphenols over 1-2 weeks is desired, even if fine control over tail "quality" is less critical.
- MatC (PHB+PCL fibres and PCL beads, all with HS-KL+GP-PP co-located). MatC released somewhat less GP-PP than B (49.4 a.u.; Table 5) but with a smoother profile and a particularly strong late contribution. Burst and mid phases were high, and the late tail was second only to MatB in absolute terms but largest in relative terms (late/total). The cumulative curve of MatC tracks just below that of MatB (Figure 11B). Here, HS-KL and GP-PP share the same microdomains in both PHB and PCL phases, so HS-KL-GP-PP complexes meter desorption and stabilise labile species (anthocyanins and flavonols). The architecture behaves as a system of parallel depots distributed in fibres and beads, providing a broad, well-structured burst-mid-late sequence. At TA, MatC is thus slightly less productive than MatB in total mass but more controlled and better suited when the quality and persistence of the late phase are important.
- MatD (PHB+PCL fibres and PCL beads, GP-PP only, no HS-KL). MatD, despite the highest nominal GP-PP loading, gave the lowest total release at TA (34.8 a.u.; Table 5), with the smallest burst and late areas. The cumulative curve lies clearly below those of the other systems (Figure 11B). The absence of HS-KL means that GP-PP primarily acts as a plasticiser for PHB and PCL; the network swells and softens rather than forming well-defined, stabilised depots. As a result, the scaffold is structurally quite responsive but functionally inefficient: pathways become tortuous, and a significant fraction of GP-PP remains trapped. MatD is therefore the least effective matrix at TA, despite its apparent "capacity".
3.5.2. Release Profiles at 37 °C
- MatA - At T37, MatA exhibited a modest reduction in total release (~−12 %) and a marked decrease in late-area (~−43.5 %), while its burst increased by 25.3% relative to TA, and t50 decreased from 148.3 h to 118.5 h (Table 5). PHB+HS-KL fibres swell and undergo secondary crystallisation, narrowing diffusion pathways, while PHB+GP-PP depots remain relatively soft but progressively less accessible. The profile becomes more front-loaded (larger early contribution, weaker tail), consistent with a scaffold that reacts quickly to warming but then self-limits further release.
- MatB - MatB was the most temperature-sensitive system. Total release dropped by about one-third, and the late tail decreased by roughly two-thirds compared with TA (Table 5). The burst and mainly mid phases were also weakened. t50 shifted from 143.6 to 103.2 h. Morphologically, PHB+HS-KL fibres swell, whereas large PCL+GP-PP beads densify and shrink, thereby reducing the accessible MAF volume. These changes push more releases earlier and sharply erode late-phase capacity. At T37, MatB therefore loses much of the advantage it had at TA and no longer dominates in either total or late delivery.
- MatC - MatC showed only a moderate reduction in total (≈−14%) and late (~−47%) areas, whereas both burst and mid peaks increased relative to TA (Table 5). t50 decreased but remained intermediate. Structurally, fibres swelled only slightly, preserving pore connectivity, and PCL(HS-KL+GP-PP) beads tended more to detach than to shrink in place. HS-KL-GP-PP co-location in both PHB and PCL phases continued to meter desorption and stabilise flavonols, stilbenes, and anthocyanins. As a result, even under stress conditions, MatC maintained the highest total release and the strongest late tail in absolute terms. It thus emerges as the most robust matrix at 37 °C.
- MatD - For MatD, total release decreased by only ~13%, but it delivered the lowest overall GP-PP, with the smallest relative drop in the mid area and moderate late suppression (Table 5). However, MatD still delivered the lowest overall GP-PP at T37. The extreme swelling of GP-PP-only fibres (~+174%) and densification of PCL+GP-PP beads (~-23%) (Table 4) produced a highly swollen, partially collapsed network with reduced porosity and high tortuosity. Release became largely access-limited and poorly controllable; the profile remained weak across all phases.
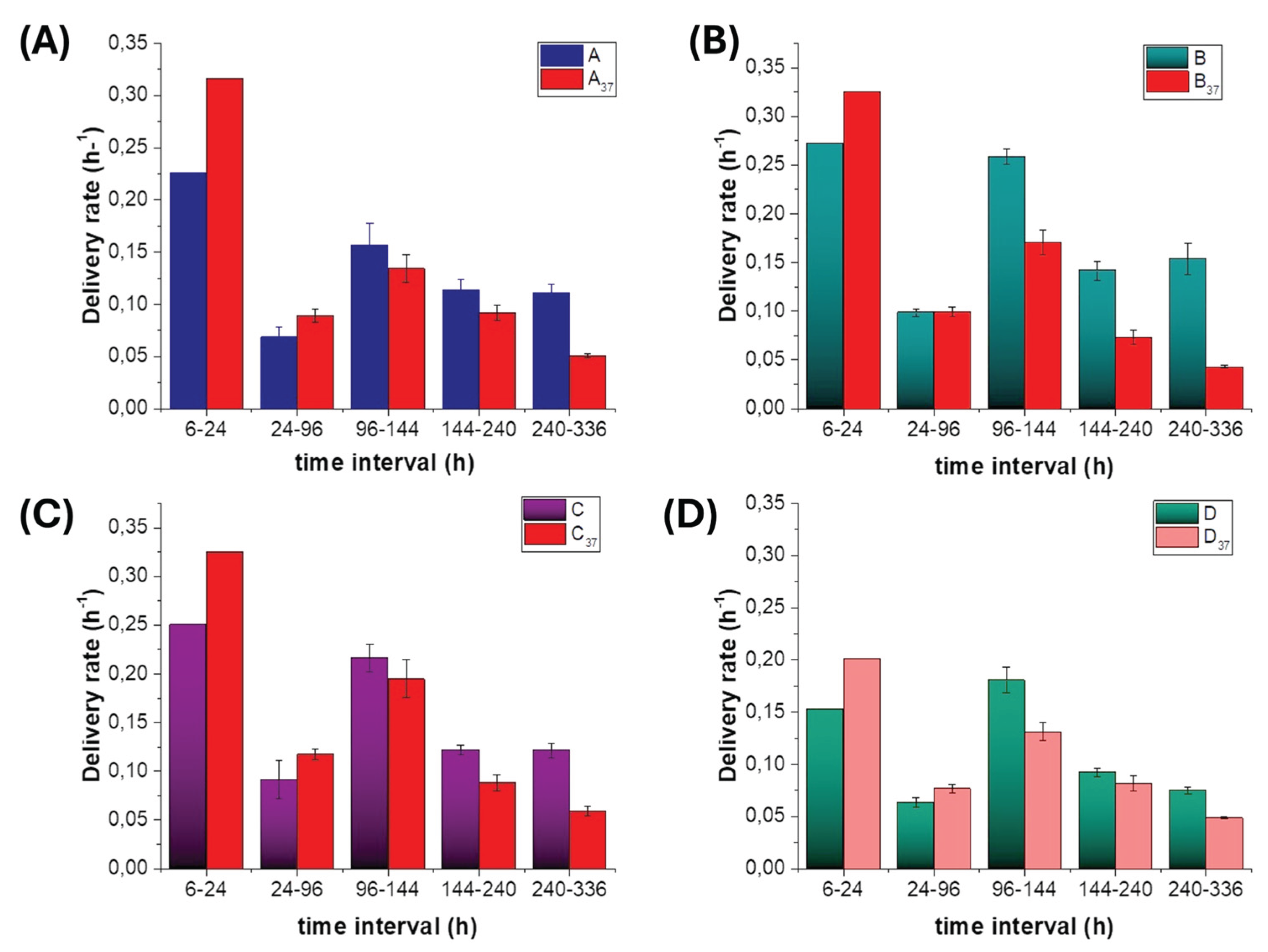
3.5.3. Polyphenol Release Rates at Ambient and 37 °C Temperatures.
3.5.4. Key Parameters Controlling Polyphenol Release
- Polymer phase and MAF/RAF microstructure: Both PHB and PCL are semicrystalline polyesters with crystalline lamellae embedded in an amorphous phase. Electrospinning produces oriented fibres in which the amorphous material can be separated conceptually into a mobile amorphous fraction (MAF), which hosts most of the diffusion pathways, and a rigid amorphous fraction (RAF) at crystal interfaces, which contributes more to mechanical stiffness than to transport. PHB typically crystallises more strongly and generates a larger RAF fraction, making it a slower, more gate-like carrier [86,87,93] . PCL, with a lower melting point and more mobile chains at both TA and T37, offers more continuous MAF pathways and behaves as a faster depot [118,119] . In the present architectures, PHB-rich regions therefore tend to set the overall permeability and late-phase quality, whereas PCL-rich depots largely control throughput in the burst and mid windows.
- Bead evolution: shrinkage vs detachment: At T37, PCL-based beads evolved in two main ways: shrink-in-place densification (MatB and MatD) and adhesion-limited detachment (MatC). Densification reduces depot volume and mobile amorphous pathways, thereby eroding mid/late capacity, even if it shortens diffusion paths locally. Detachment instead removes some depots from the fibrous network, generating an early-release pulse while preserving the internal structure of the remaining HS-KL–GP-PP depots, thereby sustaining optimal late-phase release. PHB+GP-PP beads in MatA, by contrast, remained largely unchanged in size, acting as soft but increasingly shielded sources as the surrounding PHB matrix tightened
- Fibre swelling and pore architecture: Fibres are not merely passive supports: they act as depots when loaded with GP-PP and, more importantly, as walls that define pore size, connectivity, and tortuosity. Swelling increases fibre diameter and reduces effective pore throats [120,121] , so a limited expansion can improve water access and wet the depots, whereas excessive swelling closes channels and throttles diffusion. In our system, co-electrospun PHB/PCL fibres in MatC swell least, PHB+HS-KL fibres in MatA and MatB show intermediate swelling, and GP-PP-only PHB/PCL fibres in MatD swell most, in line with their different GP-PP and HS-KL contents (Table 4), and the hydrophilic and H-bonding plasticising features of GP-PP. Thus, MatC preserves a relatively open, stable pore network that supports sustained mid/late release even at T37; MatA and MatB progressively tighten but retain usable pathways; and MatD evolves towards a highly swollen, tortuous mesh in which transport becomes access-limited and a large fraction of GP-PP remains trapped. Fibre swelling, therefore, acts as a second-level control on release, operating in parallel with bead densification/detachment and the chemical effects of HS-KL–GP-PP interactions.
- Lignin as a physical and chemical stabiliser: HS-KL plays a dual stabilising role. Physically, it forms π–π and H-bond networks with PHB and PCL, which limit fibre swelling and promote secondary crystallisation in a controlled manner, thereby preserving the pore architecture—especially in MatC, where swelling is minimal even at T37. Chemically, when HS-KL and GP-PP are co-located (MatC), lignin provides binding sites and hydration shells that stabilise polyphenols and slow their desorption, yielding better-structured mid/late release and a more chemically rich late tail. Where HS-KL and GP-PP are segregated (MatB) or HS-KL is absent (MatD), depots are more prone to uncontrolled densification, and sensitive GP-PP classes are less protected.
- Co-location vs segregation of HS-KL and GP-PP; GP-PP-only networks: The comparison between the matrices illustrates three regimes: i) Co-location (MatC) - HS-KL and GP-PP in the same PHB and PCL domains create genuine HS-KL-GP-PP depots: swelling is restricted, depots meter release, and labile species are better preserved. This configuration supports a broad burst-mid-late sequence that is relatively stable to temperature; ii) Segregation (MatA, MatB) - HS-KL in fibres and GP-PP in separate depots provide structural reinforcement and some gating, but do not directly co-stabilise the cargo. MatB exploits large PCL+GP-PP depots to maximise throughput at TA but loses much of this advantage at T37; MatA remains more conservative at both temperatures; iii) GP-PP-only networks (MatD) - Fibres and depots without HS-KL are strongly plasticised by GP-PP and water. Swelling and densification dominate, porosity collapses, and a substantial fraction of GP-PP remains trapped. This configuration performs poorly as a controlled-release system, even under high loading conditions.
3.6. Integrated Mechanistic Interpretation
3.6.1. Early Wetting (10 min, TA ) Versus Long-Term Release (14 days, TA)
3.6.2. Effect of Warming to 37 °C: How Temperature Reshapes Release
3.6.3. Final Synthesis and Design Rules
3.7. Potential Applications of the Biohybrid Nanostructures
- Ambient conditions (≈15-25 °C, irrigated or mild climates): i) MatB: best suited to fast, leafy crops (rocket, baby-leaf mixes, young brassicas) and short cycles, where a strong mass delivery over 1-2 weeks is desirable, and the late tail serves mainly to smooth the decline. ii) MatC: preferable for fruiting vegetables and perennial seedlings (tomato, pepper, aubergine, cucurbits, strawberry, vine/olive/citrus liners), where a well-metered, chemically "clean" late phase is advantageous for sustained antioxidant and signalling support. iii) MatA: conservative option when a gentle profile is required (e.g., sensitive seedlings, low-input systems), accepting a lower total release. iv) MatD: not recommended as a primary carrier due to its low effective release despite high loading.
- Warm or stressed beds (surface soil‚ ≈32-37 °C; shallow Mediterranean soils in hot periods): i) MatCremains the first choice because it preserves the highest total release and late tail under thermal stress, making it suitable for summer transplants of Solanaceae and cucurbits, or for shallow-rooted perennials on hot aspects. ii) MatA can be used where only a modest, self-limiting pulse is desired under heat (e.g., to avoid excessive stimulation or where other inputs are high). iii) MatB becomes temperature-sensitive: it can still be exploited in cooler or irrigated beds but is less appropriate in persistently hot, shallow soils unless reformulated to reduce depot densification. iv) MatD should be avoided in hot conditions, as its pronounced swelling and poor control lead to weak, poorly metered release.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgements
Conflicts of Interest
Abbreviations
| PHB | Poly-3-hydroxybutyric acid |
| PCL | Polycaprolactone |
| CRF | Controlled-release fertilisers |
| SRF | Slow-release fertilisers |
| CRP | Controlled-release polyphenols |
| HS | Hazelnut-shells |
| HS-KL | Hazelnut-shell Klason lignin (acid-insoluble fraction by Klason method); not Kraft lignin |
| GP | Grape-pomace |
| GP-PP | Grape-pomace polyphenols |
| (FE-)SEM | Field-emission Scanning electron microscopy |
| FTIR-ATR | Fourier Transform Infrared-Attenuated Total Reflectance |
| UV–VIS | Ultraviolet-Visible radiation |
| WCA | Water contact angle |
| WDV | Water droplet volume |
| RAF | Rigid amorphous fraction |
| MAF | Mobile amorphous fraction |
| HBA | Hydroxybenzoic acid |
References
- Prado-Acebo, I.; Cubero-Cardoso, J.; Lu-Chau, T.A.; Eibes, G. Integral Multi-Valorization of Agro-Industrial Wastes: A Review. Waste Manag. 2024, 183, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Kour, R.; Singh, S.; Sharma, H.B.; Naik, T.S.S.K.; Shehata, N.; N, P.; Ali, W.; Kapoor, D.; Dhanjal, D.S.; Singh, J.; et al. Persistence and Remote Sensing of Agri-Food Wastes in the Environment: Current State and Perspectives. Chemosphere 2023, 317, 137822. [Google Scholar] [CrossRef] [PubMed]
- Mondéjar-López, M.; García-Simarro, M.P.; Navarro-Simarro, P.; Gómez-Gómez, L.; Ahrazem, O.; Niza, E. A Review on the Encapsulation of “Eco-Friendly” Compounds in Natural Polymer-Based Nanoparticles as next Generation Nano-Agrochemicals for Sustainable Agriculture and Crop Management. Int. J. Biol. Macromol. 2024, 280, 136030. [Google Scholar] [CrossRef]
- Hazarika, A.; Yadav, M.; Yadav, D.K.; Yadav, H.S. An Overview of the Role of Nanoparticles in Sustainable Agriculture. Biocatal. Agric. Biotechnol. 2022, 43, 102399. [Google Scholar] [CrossRef]
- Joudeh, N.; Linke, D. Nanoparticle Classification, Physicochemical Properties, Characterization, and Applications: A Comprehensive Review for Biologists. J. Nanobiotechnology 2022, 20, 262. [Google Scholar] [CrossRef]
- Shekhawat, D.; Vauth, M.; Pezoldt, J. Size Dependent Properties of Reactive Materials. Inorganics 2022, 10, 56. [Google Scholar] [CrossRef]
- El-Kady, M.M.; Ansari, I.; Arora, C.; Rai, N.; Soni, S.; Verma, D.K.; Singh, P.; Mahmoud, A.E.D. Nanomaterials: A Comprehensive Review of Applications, Toxicity, Impact, and Fate to Environment. J. Mol. Liq. 2023, 370, 121046. [Google Scholar] [CrossRef]
- Belay, T.; Worku, L.A.; Bachheti, R.K.; Bachheti, A.; Husen, A. Advances in Smart Nanomaterials and Their Applications; 2023; pp. 1–21. [Google Scholar] [CrossRef]
- Ding, B.; Yu, J.; Wang, X. Electrospinning: Nanofabrication and Applications, 1st ed.; Ding, *!!! REPLACE !!!*, Yu, *!!! REPLACE !!!*, Bin, *!!! REPLACE !!!*, Wang, *!!! REPLACE !!!*, Jianyong, *!!! REPLACE !!!*, Xianfeng, *!!! REPLACE !!!*, Eds.; Elsevier Inc.; ISBN 9780323512701.
- Asmatulu, R.; Khan, W.S. Synthesis and Applications of Electrospun Nanofibers; Elsevier Inc.: Amsterdam, Netherlands, 2019; ISBN 978-0-12-813914-1. [Google Scholar]
- Keirouz, A.; Wang, Z.; Reddy, V.S.; Nagy, Z.K.; Vass, P.; Buzgo, M.; Ramakrishna, S.; Radacsi, N. The History of Electrospinning: Past, Present, and Future Developments. Adv. Mater. Technol. 2023, 8. [Google Scholar] [CrossRef]
- Cleeton, C.; Keirouz, A.; Chen, X.; Radacsi, N. Electrospun Nanofibers for Drug Delivery and Biosensing. Acs Biomater Sci Eng 2019, 5, 4183–4205. [Google Scholar] [CrossRef]
- Abdulhussain, R.; Adebisi, A.; Conway, B.R.; Asare-Addo, K. Electrospun Nanofibers: Exploring Process Parameters, Polymer Selection, and Recent Applications in Pharmaceuticals and Drug Delivery. J. Drug Deliv. Sci. Technol. 2023, 90, 105156. [Google Scholar] [CrossRef]
- Vatanpour, V.; Kose-Mutlu, B.; Koyuncu, I. Electrospraying Technique in Fabrication of Separation Membranes: A Review. Desalination 2022, 533, 115765. [Google Scholar] [CrossRef]
- Huang, S.; Mansouri, J.; Le-Clech, P.; Leslie, G.; Tang, C.Y.; Fane, A.G. A Comprehensive Review of Electrospray Technique for Membrane Development: Current Status, Challenges, and Opportunities. J. Membr. Sci. 2022, 646, 120248. [Google Scholar] [CrossRef]
- Jayaprakash, P.; Maudhuit, A.; Gaiani, C.; Desobry, S. Encapsulation of Bioactive Compounds Using Competitive Emerging Techniques: Electrospraying, Nano Spray Drying, and Electrostatic Spray Drying. J. Food Eng. 2023, 339, 111260. [Google Scholar] [CrossRef]
- Tang, J.; Wu, C.; Chen, S.; Qiao, Z.; Borovskikh, P.; Shchegolkov, A.; Chen, L.; Wei, D.; Sun, J.; Fan, H. Combining Electrospinning and Electrospraying to Prepare a Biomimetic Neural Scaffold with Synergistic Cues of Topography and Electrotransduction. ACS Appl. Bio Mater. 2020, 3, 5148–5159. [Google Scholar] [CrossRef]
- Soheili, S.; Dolatyar, B.; Adabi, M.R.; Lotfollahi, D.; Shahrousvand, M.; Zahedi, P.; Seyedjafari, E.; Mohammadi-Rovshandeh, J. Fabrication of Fiber-Particle Structures by Electrospinning/Electrospray Combination as an Intrinsic Antioxidant and Oxygen-Releasing Wound Dressing. J. Mater. Chem. B 2024, 12, 9074–9097. [Google Scholar] [CrossRef]
- He, M.-K.; He, Y.-L.; Li, Z.-Q.; Zhao, L.-N.; Zhang, S.-Q.; Liu, H.-M.; Qin, Z. Structural Characterization of Lignin and Lignin-Carbohydrate Complex (LCC) of Sesame Hull. Int. J. Biol. Macromol. 2022, 209, 258–267. [Google Scholar] [CrossRef] [PubMed]
- García-Fuentevilla, L.; Rubio-Valle, J.F.; Martín-Sampedro, R.; Valencia, C.; Eugenio, M.E.; Ibarra, D. Different Kraft Lignin Sources for Electrospun Nanostructures Production: Influence of Chemical Structure and Composition. Int. J. Biol. Macromol. 2022, 214, 554–567. [Google Scholar] [CrossRef]
- Luzi, F.; Yang, W.; Ma, P.; Torre, L.; Puglia, D. Lignin-Based Materials for Biomedical Applications; 2021; pp. 291–326. [Google Scholar] [CrossRef]
- Luzi, F.; Torre, L.; Puglia, D. Micro and Nanolignin in Aqueous Dispersions and Polymers; 2022; pp. 293–324. [Google Scholar] [CrossRef]
- Budnyak, T.M.; Slabon, A.; Sipponen, M.H. Lignin–Inorganic Interfaces: Chemistry and Applications from Adsorbents to Catalysts and Energy Storage Materials. ChemSusChem 2020, 13, 4344–4355. [Google Scholar] [CrossRef]
- Mateo, S.; Fabbrizi, G.; Moya, A.J. Lignin from Plant-Based Agro-Industrial Biowastes: From Extraction to Sustainable Applications. Polymers 2025, 17, 952. [Google Scholar] [CrossRef] [PubMed]
- Ariyanta, H.A.; Sari, F.P.; Sohail, A.; Restu, W.K.; Septiyanti, M.; Aryana, N.; Fatriasari, W.; Kumar, A. Current Roles of Lignin for the Agroindustry: Applications, Challenges, and Opportunities. 2023, 240, 124523. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, R.; Fardim, P. Lignin-Based Materials for Emerging Advanced Applications. Curr. Opin. Green Sustain. Chem. 2023, 41, 100834. [Google Scholar] [CrossRef]
- Shorey, R.; Salaghi, A.; Fatehi, P.; Mekonnen, T.H. Valorization of Lignin for Advanced Material Applications: A Review. RSC Sustain. 2024, 2, 804–831. [Google Scholar] [CrossRef]
- Yang, G.; Gong, Z.; Luo, X.; Chen, L.; Shuai, L. Bonding Wood with Uncondensed Lignins as Adhesives. Nature 2023, 621, 511–515. [Google Scholar] [CrossRef]
- Zagoskina, N.V.; Zubova, M.Y.; Nechaeva, T.L.; Kazantseva, V.V.; Goncharuk, E.A.; Katanskaya, V.M.; Baranova, E.N.; Aksenova, M.A. Polyphenols in Plants: Structure, Biosynthesis, Abiotic Stress Regulation, and Practical Applications (Review). Int. J. Mol. Sci. 2023, 24, 13874. [Google Scholar] [CrossRef] [PubMed]
- Koudoufio, M.; Desjardins, Y.; Feldman, F.; Spahis, S.; Delvin, E.; Levy, E. Insight into Polyphenol and Gut Microbiota Crosstalk: Are Their Metabolites the Key to Understand Protective Effects against Metabolic Disorders? Antioxidants 2020, 9, 982. [Google Scholar] [CrossRef]
- Panzella, L.; Napolitano, A. Condensed Tannins, a Viable Solution To Meet the Need for Sustainable and Effective Multifunctionality in Food Packaging: Structure, Sources, and Properties. J. Agric. Food Chem. 2022, 70, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Gourlay, G.; Hawkins, B.J.; Albert, A.; Schnitzler, J.; Constabel, C.P. Condensed Tannins as Antioxidants That Protect Poplar against Oxidative Stress from Drought and UV-B. Plant, Cell Environ. 2022, 45, 362–377. [Google Scholar] [CrossRef]
- Liu, Y.; Shi, Y.; Zhang, M.; Han, F.; Liao, W.; Duan, X. Natural Polyphenols for Drug Delivery and Tissue Engineering Construction: A Review. Eur. J. Med. Chem. 2024, 266, 116141. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, C.; Zou, Y.; Hu, J.; Li, Y.; Cheng, Y. Natural Polyphenols in Drug Delivery Systems: Current Status and Future Challenges. Giant 2020, 3, 100022. [Google Scholar] [CrossRef]
- Raju, N.N.; Sankaranarayanan, M.; Bharathiraja, B. Production of Polyhydroxybutyrate (PHB), a Biodegradable Polymer from Seaweed Biomass Using Novel Bacterial Isolates. J. Mol. Struct. 2024, 1303, 137511. [Google Scholar] [CrossRef]
- Balasubramanian, V.K.; Chellapandi, R.; Balakrishnan, M.; Murugan, K.; Kennedy, J.P.K.J.; Murugan, S.; Khumalo, M.V.; Sarangi, P.K.; Chou, J.-Y.; Muthuramalingam, J.B. Biosynthesis of Bioplastic Polyhydroxybutyrate (PHB) from Microbes Isolated from Paddy/Sugarcane Fields and Fabrication of Biodegradable Thin Film. Process Saf. Environ. Prot. 2024, 187, 1178–1188. [Google Scholar] [CrossRef]
- Price, S.; Kuzhiumparambil, U.; Pernice, M.; Ralph, P.J. Cyanobacterial Polyhydroxybutyrate for Sustainable Bioplastic Production: Critical Review and Perspectives. J. Environ. Chem. Eng. 2020, 8, 104007. [Google Scholar] [CrossRef]
- Mandragutti, T.; Jarso, T.S.; Godi, S.; Begum, S.S.; K, B. Physicochemical Characterization of Polyhydroxybutyrate (PHB) Produced by the Rare Halophile Brachybacterium Paraconglomeratum MTCC 13074. Microb. Cell Factories 2024, 23, 59. [Google Scholar] [CrossRef]
- Rathna, R.P.; Kishore, S.N.; Kulandhaivel, M. Statistical Design-Driven Optimization of Polyhydroxy-Butyrate (PHB) Production Using Sugarcane Bagasse by Brevibacterium Sp. (PP989436) Strain. J. Polym. Environ. 2025, 33, 910–927. [Google Scholar] [CrossRef]
- Rathna, R.P.; Kulandhaivel, M. Sustainable Synthesis of Biopolymer Polyhydroxybutyrate (PHB) from Agro-Residue by Brevibacterium Casei with Emphasis on Degradation Analysis. J. Pure Appl. Microbiol. 2024, 18, 347–366. [Google Scholar] [CrossRef]
- Kotcharat, P.; Chuysinuan, P.; Thanyacharoen, T.; Techasakul, S.; Ummartyotin, S. Development of Bacterial Cellulose and Polycaprolactone (PCL) Based Composite for Medical Material. Sustain. Chem. Pharm. 2021, 20, 100404. [Google Scholar] [CrossRef]
- Spizzirri, U.G.; Aiello, F.; Carullo, G.; Facente, A.; Restuccia, D. Nanotechnologies: An Innovative Tool to Release Natural Extracts with Antimicrobial Properties. Pharmaceutics 2021, 13, 230. [Google Scholar] [CrossRef] [PubMed]
- Bang, J.; Kim, J.-H.; Park, S.-W.; Kim, J.; Jung, M.; Jung, S.; Kim, J.-C.; Choi, I.-G.; Kwak, H.W. Effect of Chemically Modified Lignin Addition on the Physicochemical Properties of PCL Nanofibers. Int. J. Biol. Macromol. 2023, 240, 124330. [Google Scholar] [CrossRef] [PubMed]
- TAPPI Acid-Insoluble Lignin in Wood and Pulp. In 2002-2003 TAPPI Test Methods; Tappi Press: Atlanta, GA, USA, 2002.
- Agustin-Salazar, S.; Cerruti, P.; Medina-Juárez, L.Á.; Scarinzi, G.; Malinconico, M.; Soto-Valdez, H.; Gamez-Meza, N. Lignin and Holocellulose from Pecan Nutshell as Reinforcing Fillers in Poly (Lactic Acid) Biocomposites. Int. J. Biol. Macromol. 2018, 115, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Ritchey, J.G.; Waterhouse, A.L. A Standard Red Wine: Monomeric Phenolic Analysis of Commercial Cabernet Sauvignon Wines. Am. J. Enol. Vitic. 1999, 50, 91–100. [Google Scholar] [CrossRef]
- Torrent, J.; Pfeiffer, M.; Ibañez, J.J. Encyclopedia of Soils in the Environment (Second Edition). Pedology 2023, 319–331. [Google Scholar] [CrossRef]
- Marzaioli, R.; D’Ascoli, R.; Pascale, R.A.D.; Rutigliano, F.A. Soil Quality in a Mediterranean Area of Southern Italy as Related to Different Land Use Types. Appl. Soil Ecol. 2010, 44, 205–212. [Google Scholar] [CrossRef]
- Sadh, P.K.; Duhan, S.; Duhan, J.S. Agro-Industrial Wastes and Their Utilization Using Solid State Fermentation: A Review. Bioresour Bioprocess 2018, 5, 1. [Google Scholar] [CrossRef]
- Commission, E. Bioeconomy Research and Innovation. Available online: https://research-and-innovation.ec.europa.eu/research-area/environment/bioeconomy_en (accessed on 16 January 2026).
- WasteManaged Agricultural Waste Guide 2025. Available online: https://www.wastemanaged.co.uk/our-news/agriculture/agricultural-waste-guide/?utm_source=chatgpt.com (accessed on 18 January 2026).
- Šelo, G.; Planinić, M.; Tišma, M.; Tomas, S.; Komlenić, D.K.; Bucić-Kojić, A. A Comprehensive Review on Valorization of Agro-Food Industrial Residues by Solid-State Fermentation. Foods 2021, 10, 927. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.V.; Hamid, S.B.A.; Zain, S.K. Conversion of Lignocellulosic Biomass to Nanocellulose: Structure and Chemical Process. Sci. World J. 2014, 2014, 631013. [Google Scholar] [CrossRef]
- Srivastava, K.; Chaudhary, K.; Adil, M.; Rajora, A. Exploring the Potential of Lignin Nanoparticles: Synthesis, Applications, and Future Directions. Polym. Bull. 2025, 82, 6277–6306. [Google Scholar] [CrossRef]
- Singh, S.K.; Singh, A.; Mishra, S. Electrospun Nanofibers, Fabrication, Functionalisation and Applications. Springer Ser. Polym. Compos. Mater. 2021, 171–197. [Google Scholar] [CrossRef]
- Adam, A.A.; Dennis, J.O.; Al-Hadeethi, Y.; Mkawi, E.M.; Abdulkadir, B.A.; Usman, F.; Hassan, Y.M.; Wadi, I.A.; Sani, M. State of the Art and New Directions on Electrospun Lignin/Cellulose Nanofibers for Supercapacitor Application: A Systematic Literature Review. Polymers 2020, 12, 2884. [Google Scholar] [CrossRef] [PubMed]
- Kai, D.; Jiang, S.; Low, Z.W.; Loh, X.J. Engineering Highly Stretchable Lignin-Based Electrospun Nanofibers for Potential Biomedical Applications. J. Mater. Chem. B 2015, 3, 6194–6204. [Google Scholar] [CrossRef]
- Nainggolan, G.; Gea, S.; Marpongahtun; Harahap, M.; Dellyansyah; Situmorang, S.A. Promoting Electrospun Lignin/PEO Nanofiber for High-Performance CO Filtration. J. Nat. Fibers 2023, 20, 2160402. [Google Scholar] [CrossRef]
- Chutturi, M.; Kelkar, B.U.; Yadav, S.M.; Wibowo, E.S.; Bhuyar, P.; Naik, B.P.; Sinha, A.; Lee, S.H. Exploring Nanolignin as a Sustainable Biomacromolecule in Polymer Composites: Synthesis, Characterization, and Applications: A Review. Int. J. Biol. Macromol. 2025, 304, 140881. [Google Scholar] [CrossRef]
- Camargos, C.H.M.; Yang, L.; Jackson, J.C.; Tanganini, I.C.; Francisco, K.R.; Ceccato-Antonini, S.R.; Rezende, C.A.; Faria, A.F. Lignin and Nanolignin: Next-Generation Sustainable Materials for Water Treatment. ACS Appl. Bio Mater. 2025, 8, 2632–2673. [Google Scholar] [CrossRef]
- Saini, N.; Anmol, A.; Kumar, S.; Wani, A.W.; Bakshi, M.; Dhiman, Z. Exploring Phenolic Compounds as Natural Stress Alleviators in Plants- a Comprehensive Review. Physiol. Mol. Plant Pathol. 2024, 133, 102383. [Google Scholar] [CrossRef]
- Patil, J.R.; Mhatre, K.J.; Yadav, K.; Yadav, L.S.; Srivastava, S.; Nikalje, G.C. Flavonoids in Plant-Environment Interactions and Stress Responses. Discov. Plants 2024, 1, 68. [Google Scholar] [CrossRef]
- Kumar, G.A.; Kumar, S.; Bhardwaj, R.; Swapnil, P.; Meena, M.; Seth, C.S.; Yadav, A. Recent Advancements in Multifaceted Roles of Flavonoids in Plant–Rhizomicrobiome Interactions. Front. Plant Sci. 2024, 14, 1297706. [Google Scholar] [CrossRef]
- Hättenschwiler, S.; Vitousek, P.M.; Hättenschwiler, S.; Vitousek, P.M. The Role of Polyphenols in Terrestrial Ecosystem Nutrient Cycling. Trends Ecol. Evol. 2000, 15, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Min, K.; Freeman, C.; Kang, H.; Choi, S.-U. The Regulation by Phenolic Compounds of Soil Organic Matter Dynamics under a Changing Environment. Biomed Res Int 2015, 2015, 825098. [Google Scholar] [CrossRef]
- Shah, A.; Smith, D.L. Flavonoids in Agriculture: Chemistry and Roles in, Biotic and Abiotic Stress Responses, and Microbial Associations. Agronomy 2020, 10, 1209. [Google Scholar] [CrossRef]
- Mekapogu, M.; Vasamsetti, B.M.K.; Kwon, O.-K.; Ahn, M.-S.; Lim, S.-H.; Jung, J.-A. Anthocyanins in Floral Colors: Biosynthesis and Regulation in Chrysanthemum Flowers. Int. J. Mol. Sci. 2020, 21, 6537. [Google Scholar] [CrossRef]
- Buondonno, A.; Capra, G.F.; Coppola, E.; Dazzi, C.; Grilli, E.; Odierna, P.; Rubino, M.; Vacca, S. Aspects of Soil Phenolic Matter (SPM): An Explorative Investigation in Agricultural, Agroforestry, and Wood Ecosystems. Geoderma 2014, 213, 235–244. [Google Scholar] [CrossRef]
- Cortés-Ferré, H.E.; Arredondo-Ochoa, T.; Gaytán-Martínez, M. Polysaccharides-Polyphenolic Interactions: Formation, Functionality and Applications. Trends Food Sci. Technol. 2025, 163, 105117. [Google Scholar] [CrossRef]
- Guo, Y.; Sun, Q.; Wu, F.; Dai, Y.; Chen, X. Polyphenol-Containing Nanoparticles: Synthesis, Properties, and Therapeutic Delivery. Adv. Mater. 2021, 33, e2007356. [Google Scholar] [CrossRef]
- Xu, W.; Lin, Z.; Pan, S.; Chen, J.; Wang, T.; Cortez-Jugo, C.; Caruso, F. Direct Assembly of Metal-Phenolic Network Nanoparticles for Biomedical Applications. Angew. Chem. Int. Ed. 2023, 62, e202312925. [Google Scholar] [CrossRef] [PubMed]
- Huo, K.; Liu, W.; Shou, Z.; Wang, H.; Liu, H.; Chen, Y.; Zan, X.; Wang, Q.; Li, N. Modulating the Interactions of Peptide-Polyphenol for Supramolecular Assembly Coatings with Controllable Kinetics and Multifunctionalities. Adv. Sci. 2025, 12, 2412194. [Google Scholar] [CrossRef] [PubMed]
- Kaeswurm, J.A.H.; Scharinger, A.; Teipel, J.; Buchweitz, M. Absorption Coefficients of Phenolic Structures in Different Solvents Routinely Used for Experiments. Molecules 2021, 26, 4656. [Google Scholar] [CrossRef]
- Velho, P.; Rebelo, C.S.; Macedo, E.A. Extraction of Gallic Acid and Ferulic Acid for Application in Hair Supplements. Molecules 2023, 28, 2369. [Google Scholar] [CrossRef]
- Uivarasan, A.; Lukinac, J.; Jukić, M.; Šelo, G.; Peter, A.; Nicula, C.; Cozmuta, A.M.; Cozmuta, L.M. Characterization of Polyphenol Composition and Starch and Protein Structure in Brown Rice Flour, Black Rice Flour and Their Mixtures. Foods 2024, 13, 1592. [Google Scholar] [CrossRef] [PubMed]
- Favaro, L.; Balcão, V.; Rocha, L.; Silva, E.; Jr, J.O.; Vila, M.; Tubino, M. Physicochemical Characterization of a Crude Anthocyanin Extract from the Fruits of Jussara (Euterpe Edulis Martius): Potential for Food and Pharmaceutical Applications. J. Braz. Chem. Soc. 2018. [Google Scholar] [CrossRef]
- Boulet, J. -C.; Ducasse, M. -A.; Cheynier, V. Ultraviolet Spectroscopy Study of Phenolic Substances and Other Major Compounds in Red Wines: Relationship between Astringency and the Concentration of Phenolic Substances. Aust. J. Grape Wine Res. 2017, 23, 193–199. [Google Scholar] [CrossRef]
- Grasel, F. dos S.; Ferrão, M.F.; Wolf, C.R. Ultraviolet Spectroscopy and Chemometrics for the Identification of Vegetable Tannins. Ind. Crop. Prod. 2016, 91, 279–285. [Google Scholar] [CrossRef]
- Nakamura, K.; Shirato, M.; Ikai, H.; Kanno, T.; Sasaki, K.; Kohno, M.; Niwano, Y. Photo-Irradiation of Proanthocyanidin as a New Disinfection Technique via Reactive Oxygen Species Formation. PLoS ONE 2013, 8, e60053. [Google Scholar] [CrossRef]
- Chua, L.S.; Thong, H.Y.; Soo, J. Effect of pH on the Extraction and Stability of Anthocyanins from Jaboticaba Berries. 2024, 5, 100835. [Google Scholar] [CrossRef]
- Okello, A.; Owuor, B.O.; Namukobe, J.; Okello, D.; Mwabora, J. Influence of the pH of Anthocyanins on the Efficiency of Dye Sensitized Solar Cells. 2022, 8, e09921. [Google Scholar] [CrossRef]
- Ku, C.S.; Mun, S.P. Characterization of Proanthocyanidin in Hot Water Extract Isolated from Pinus Radiata Bark. Wood Sci. Technol. 2007, 41, 235. [Google Scholar] [CrossRef]
- Espina, A.; Sanchez-Cortes, S.; Jurašeková, Z. Vibrational Study (Raman, SERS, and IR) of Plant Gallnut Polyphenols Related to the Fabrication of Iron Gall Inks. Molecules 2022, 27, 279. [Google Scholar] [CrossRef]
- Argenziano, R.; Moccia, F.; Esposito, R.; D’Errico, G.; Panzella, L.; Napolitano, A. Recovery of Lignins with Potent Antioxidant Properties from Shells of Edible Nuts by a Green Ball Milling/Deep Eutectic Solvent (DES)-Based Protocol. Antioxidants 2022, 11, 1860. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Chen, Z.; Chen, H.; Chen, K.; Tao, W.; Ouyang, X.; Mei, L.; Zeng, X. Polyphenol-Based Hydrogels: Pyramid Evolution from Crosslinked Structures to Biomedical Applications and the Reverse Design. Bioact. Mater. 2022, 17, 49–70. [Google Scholar] [CrossRef]
- Lugoloobi, I.; Li, X.; Zhang, Y.; Mao, Z.; Wang, B.; Sui, X.; Feng, X. Fabrication of Lignin/Poly(3-Hydroxybutyrate) Nanocomposites with Enhanced Properties via a Pickering Emulsion Approach. Int. J. Biol. Macromol. 2020, 165, 3078–3087. [Google Scholar] [CrossRef]
- Weihua, K.; He, Y.; Asakawa, N.; Inoue, Y. Effect of Lignin Particles as a Nucleating Agent on Crystallization of Poly(3-hydroxybutyrate). J. Appl. Polym. Sci. 2004, 94, 2466–2474. [Google Scholar] [CrossRef]
- Vollrath, A.; Kretzer, C.; Beringer-Siemers, B.; Shkodra, B.; Czaplewska, J.A.; Bandelli, D.; Stumpf, S.; Hoeppener, S.; Weber, C.; Werz, O.; et al. Effect of Crystallinity on the Properties of Polycaprolactone Nanoparticles Containing the Dual FLAP/mPEGS-1 Inhibitor BRP-187. Polymers 2021, 13, 2557. [Google Scholar] [CrossRef] [PubMed]
- Govil, S.; Long, N.V.D.; Escribà-Gelonch, M.; Hessel, V. Controlled-Release Fertiliser: Recent Developments and Perspectives. Ind. Crop. Prod. 2024, 219, 119160. [Google Scholar] [CrossRef]
- Mansouri, H.; Said, H.A.; Noukrati, H.; Oukarroum, A.; youcef, H.B.; Perreault, F. Advances in Controlled Release Fertilizers: Cost-Effective Coating Techniques and Smart Stimuli-Responsive Hydrogels. Adv. Sustain. Syst. 2023, 7. [Google Scholar] [CrossRef]
- Lawrencia, D.; Wong, S.K.; Low, D.Y.S.; Goh, B.H.; Goh, J.K.; Ruktanonchai, U.R.; Soottitantawat, A.; Lee, L.H.; Tang, S.Y. Controlled Release Fertilizers: A Review on Coating Materials and Mechanism of Release. Plants 2021, 10, 1–26. [Google Scholar] [CrossRef]
- Swify, S.; Mažeika, R.; Baltrusaitis, J.; Drapanauskaitė, D.; Barčauskaitė, K. Review: Modified Urea Fertilizers and Their Effects on Improving Nitrogen Use Efficiency (NUE). Sustainability 2023, 16, 188. [Google Scholar] [CrossRef]
- Stephens, J.S.; Chase, D.B.; Rabolt, J.F. Effect of the Electrospinning Process on Polymer Crystallization Chain Conformation in Nylon-6 and Nylon-12. Macromolecules 2004, 37, 877–881. [Google Scholar] [CrossRef]
- Lizundia, E.; Sipponen, M.H.; Greca, L.G.; Balakshin, M.; Tardy, B.L.; Rojas, O.J.; Puglia, D. Multifunctional Lignin-Based Nanocomposites and Nanohybrids. Green Chem 2021, 23, 6698–6760. [Google Scholar] [CrossRef] [PubMed]
- Meenu, M.; Pujari, A.K.; Kirar, S.; Mansi; Thakur, A.; Garg, M.; Bhaumik, J. Development of Bionanocomposite Packaging Films Based on Lignin Nanoencapsulated Anthocyanins Extracted from Agro-Waste for Enhancing the Post-Harvest Shelf Life of Tomatoes. Sustain. Food Technol. 2025, 3, 414–424. [Google Scholar] [CrossRef]
- Lv, X.; Mu, J.; Wang, W.; Liu, Y.; Lu, X.; Sun, J.; Wang, J.; Ma, Q. Effects and Mechanism of Natural Phenolic Acids/Fatty Acids on Copigmentation of Purple Sweet Potato Anthocyanins. Curr. Res. Food Sci. 2022, 5, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Pajer, N.; Cestari, C.; Argyropoulos, D.S.; Crestini, C. From Lignin Self Assembly to Nanoparticles Nucleation and Growth: A Critical Perspective. npj Mater. Sustain. 2024, 2, 31. [Google Scholar] [CrossRef]
- Zhang, Z.; Yan, Y. The Effect of Surface Roughness on Sessile Droplet Evaporation Dynamics of Silica Nanofluid. Int. J. Heat Mass Transf. 2024, 234, 126156. [Google Scholar] [CrossRef]
- Bormashenko, E. Progress in Understanding Wetting Transitions on Rough Surfaces. Adv. Colloid Interface Sci. 2015, 222, 92–103. [Google Scholar] [CrossRef]
- Chen, X.; Ma, R.; Li, J.; Hao, C.; Guo, W.; Luk, B.L.; Li, S.C.; Yao, S.; Wang, Z. Evaporation of Droplets on Superhydrophobic Surfaces: Surface Roughness and Small Droplet Size Effects. Phys. Rev. Lett. 2012, 109, 116101. [Google Scholar] [CrossRef]
- Bico, J.; Thiele, U.; Quéré, D. Wetting of Textured Surfaces. Colloids Surf. A: Physicochem. Eng. Asp. 2002, 206, 41–46. [Google Scholar] [CrossRef]
- Brutin, D.; Starov, V. Recent Advances in Droplet Wetting and Evaporation. Chem. Soc. Rev. 2017, 47, 558–585. [Google Scholar] [CrossRef]
- Liu, X.; Liu, L.; Yang, Y.; Liu, B.; Duan, R. Evaporation Characteristics of Sessile Droplets on Flat Hydrophobic Surfaces in Non-Boiling Regime. Int. Commun. Heat Mass Transf. 2023, 142, 106614. [Google Scholar] [CrossRef]
- McHale, G.; Newton, M.I.; Shirtcliffe, N.J. Dynamic Wetting and Spreading and the Role of Topography. J. Phys.: Condens. Matter 2009, 21, 464122. [Google Scholar] [CrossRef]
- Subramani, M.; Balakrishnan, P. Nano-Enabled Phosphorus Fertilizers: Mechanisms, Applications, and Environmental Implications – A Critical Review. Soil Tillage Res. 2026, 257, 106959. [Google Scholar] [CrossRef]
- Ma, X.; Chen, J.; Yang, Y.; Su, X.; Zhang, S.; Gao, B.; Li, Y.C. Siloxane and Polyether Dual Modification Improves Hydrophobicity and Interpenetrating Polymer Network of Bio-Polymer for Coated Fertilizers with Enhanced Slow Release Characteristics. Chem. Eng. J. 2018, 350, 1125–1134. [Google Scholar] [CrossRef]
- Sanford, R.A.; Chee-Sanford, J.C.; Yang, W.H. Diurnal Temperature Variation in Surface Soils: An Underappreciated Control on Microbial Processes. Front. Microbiol. 2024, 15, 1423984. [Google Scholar] [CrossRef]
- Chen, H.; Huang, S.; Quan, C.; Chen, Z.; Xu, M.; Wei, F.; Tang, D. Effects of Different Colors of Plastic-Film Mulching on Soil Temperature, Yield, and Metabolites in Platostoma Palustre. Sci. Rep. 2024, 14, 5110. [Google Scholar] [CrossRef]
- Chicco, J.M.; Mandrone, G.; Vacha, D. Effects of Wildfire on Soils: Field Studies and Modelling on Induced Underground Temperature Variations. Front. Earth Sci. 2023, 11, 1307569. [Google Scholar] [CrossRef]
- Cheng, Q.; Zhang, M.; Jin, H.; Ren, Y. Spatiotemporal Variation Characteristics of Hourly Soil Temperature in Different Layers in the Low-Latitude Plateau of China. Front. Environ. Sci. 2022, 10, 1091985. [Google Scholar] [CrossRef]
- Hillel, D. Environmental Soil Physics; 1998; pp. 309–339. [Google Scholar] [CrossRef]
- SinghGill, M.; Gill, P.; Pal, R.; Singh, N. Mulching Effects on Soil Temperature and Yield of Pear [Pyrus Pyrifoila (Burm.) Nakai] in Humid Subtropical Climate of Punjab. 2022, 73, 941–948. [Google Scholar] [CrossRef]
- Ramakrishna, A.; Tam, H.M.; Wani, S.P.; Long, T.D. Effect of Mulch on Soil Temperature, Moisture, Weed Infestation and Yield of Groundnut in Northern Vietnam. 2006, 95, 115–125. [Google Scholar] [CrossRef]
- Calderisi, G.; Salaris, E.; Cogoni, D.; Rossetti, I.; Murtas, F.; Fenu, G. Relationship Between Post-Fire Vegetation Recovery and Soil Temperature in the Mediterranean Forest. Fire 2025, 8, 91. [Google Scholar] [CrossRef]
- Lozano-Parra, J.; Pulido, M.; Lozano-Fondón, C.; Schnabel, S. How Do Soil Moisture and Vegetation Covers Influence Soil Temperature in Drylands of Mediterranean Regions? Water 2018, 10, 1747. [Google Scholar] [CrossRef]
- Delgado-Capel, M.J.; Egea-Cariñanos, P.; Cariñanos, P. Assessing the Relationship between Land Surface Temperature and Composition Elements of Urban Green Spaces during Heat Waves Episodes in Mediterranean Cities. Forests 2024, 15, 463. [Google Scholar] [CrossRef]
- Costa, J.M.; Egipto, R.; Aguiar, F.C.; Marques, P.; Nogales, A.; Madeira, M. The Role of Soil Temperature in Mediterranean Vineyards in a Climate Change Context. Front. Plant Sci. 2023, 14, 1145137. [Google Scholar] [CrossRef]
- Serra, R.S. i; Kyritsis, A.; Ivirico, J.L.E.; Ribelles, J.L.G.; Pissis, P.; Salmerón-Sánchez, M. Molecular Mobility in Biodegradable Poly( -Caprolactone)/Poly(Hydroxyethyl Acrylate) Networks. Eur. Phys. J. E 2011, 34, 37. [Google Scholar] [CrossRef]
- Flamini, M.D.; Lima, T.; Corkum, K.; Alvarez, N.J.; Beachley, V. Annealing Post-Drawn Polycaprolactone (PCL) Nanofibers Optimizes Crystallinity and Molecular Alignment and Enhances Mechanical Properties and Drug Release Profiles. Mater. Adv. 2022, 3, 3303–3315. [Google Scholar] [CrossRef]
- Ko, S.W.; Lee, J.Y.; Lee, J.; Son, B.C.; Jang, S.R.; Aguilar, L.E.; Oh, Y.M.; Park, C.H.; Kim, C.S. Analysis of Drug Release Behavior Utilizing the Swelling Characteristics of Cellulosic Nanofibers. Polymers 2019, 11, 1376. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Sheardown, H.; Hoare, T. Reactive Electrospinning of Degradable Poly(Oligoethylene Glycol Methacrylate)-Based Nanofibrous Hydrogel Networks. Chem. Commun. 2015, 52, 1451–1454. [Google Scholar] [CrossRef] [PubMed]




| Scaffolds | Description | Ratio (w/w) | Rate and Voltage* | ES/ESP | Polymers (%Tot w/w) |
Lignin (%Tot w/w) |
Polyphenols (%Tot w/w) |
|---|---|---|---|---|---|---|---|
| MetA | Combination of: | -4.5 KV; (1) 7.3 KV; (2) 8.3 KV | |||||
| -Sol.1) PHB+HS-KL | 1:0.033 | 500 µL/h | ES | 64.77% PHB | 2.22% | ||
| -Sol.2) PHB+GP-PP | 1:0.088 | 1600 µL/h | ESP | 30.23% PHB | 2.78% | ||
| MetB | Combination of: | -10 KV; (1) 8.5 KV; (7) 14 KV | |||||
| -Sol.1) PHB+HS-KL | 1:0.033 | 500 µL/h | ES | 61.75% PHB | 2.02% | ||
| -Sol.7) PCL+GP-PP | 1:0.075 | 1600 µL/h | ESP | 33.70% PCL | 2.53% | ||
| MetC | Combination of: | -3.5 KV; (3) 7.3 KV; (4) 9.3 KV | |||||
| -Sol.3) PHB+HS-KL+GP-PP | 1:0.065:0.058 | 750 µL/h | ES | 41.36% PHB | 2.71% | 2.42% | |
| -Sol.4) PCL+HS-KL+GP-PP | 1:0.056:0.050 | 750 µL/h | ES | 48.38% PCL | 2.71% | 2.42% | |
| MetD | Combination of: | -5.5 KV; (5) 7.6 KV; (6) 10.3 KV | |||||
| -Sol.5) PHB+GP-PP | 1:0.058 | 750 µL/h | ES | 43.73% PHB | 0% | 2.56% | |
| -Sol.6) PCL+GP-PP | 1:0.050 | 750 µL/h | ES | 51.15% PCL | 0% | 2.56% | |
| Note | *In the voltage conditions, the first value corresponds to the negative potential applied to the collector, while the second and third values refer to the positive voltage applied to the needle during electrospinning and electrospraying. | ||||||
| Polyphenol classes | Compounds | Content (mg mL-1) | Percent of total | Percent within class |
|---|---|---|---|---|
| Anthocyanins | Total | 141.28 | 30.2 | — |
| Malvidin-3-O-glucoside | 40.88 | 8.7 | 28.9 | |
| Pelunidin-3-O-glucoside | 36.27 | 7.7 | 25.7 | |
| Cyanidin-3-O-glucoside | 32.68 | 7.0 | 23.1 | |
| Delphinidin-3-O-glucoside | 31.45 | 6.7 | 22.3 | |
| Flavan-3-ols | Total | 94.38 | 20.2 | — |
| Catechins | 33.18 | 7.1 | 35.2 | |
| Procyanidin dimer B3 | 31.67 | 6.8 | 33.6 | |
| Procyanidin dimer B1 | 29.53 | 6.3 | 31.3 | |
| Flavonols (quercetins) | Total | 116.61 | 24.9 | — |
| Quercetin | 62.3 | 13.3 | 53.4 | |
| Quercetin-3-O-glucoside | 49.31 | 10.5 | 42.3 | |
| Quercetin-3-O-rhamnoside | 5.0 | 1.1 | 4.3 | |
| Phenolic acids | Total | 53.98 | 11.5 | — |
| Hydroxybenzoic acids | 35.57 | 7.6 | 65.9 | |
| Gallic acid | 18.41 | 3.9 | 34.1 | |
| Stilbenes | Resveratrol | 62.12 | 13.3 | 100.0 |
| Scaffolds | Fibre diameterp(µm) | Fibre diameters(µm) | Fibre diameter variation (%) | Particle roundnessp | Particle roundnesss | Particle roundness variation (%) |
| MatA | 1.03±0.63 | 1.36±0.46 | 32.3% | 0.75±0.10 | 0.80±0.10 | 6.4% |
| MatB | 1.21±0.34 | 1.42±0.31 | 17.0% | 0.82±0.10 | 0.82±0.09 | 0% |
| MatC | 0.23±0.06 | 0.24±0.06 | 5.1% | 0.83±0.08 | 0.88±0.07 | 5.3% |
| MatD | 0.28±0.07 | 0.76±0.18 | 174.4% | 0.84±0.09 | 0.78±0.13 | -6.9% |
| Scaffolds | Particlep area(µm2) | Particles area(µm2) | Particle area variation (%) | Particlep areaTot(µm2) | Particles areaTot(µm2) | Particle AreaTot variation(%) |
| MatA | 1.03±0.43 | 1.05±0.34 | 1.3% | 51.7 | 53.4 | 3.4% |
| MatB | 7.31±2.77 | 4.70±2.09 | -35.7% | 365.7 | 235.2 | -35.7% |
| MatC | 0.81±0.47 | 0.81±0.41 | 0.5% | 39.5 | 30.8 | -22.1% |
| MatD | 2.83±0.96 | 2.18±1.07 | -22.8% | 141.4 | 111.4 | -22.5% |
| Notes |
p = pristine scaffolds s = scaffolds upon soaking |
|||||
| Ambient (TA) peak areas and t50 | ||||||
| Matrix | Burstregion | Midregion | Late tailregion | Totalarea | Latefraction | t50 (h) |
| MatA | 9.43 | 17.53 | 14.40 | 41.36 | 0,35 | 148 |
| MatB | 13.25 | 23.67 | 20.19 | 57.10 | 0,35 | 144 |
| MatC | 11.51 | 19.34 | 18.51 | 49.35 | 0,38 | 142 |
| MatD | 7.25 | 15.92 | 11.66 | 34.83 | 0,33 | 140 |
| 37 °C (T37) peak areas and t50 | ||||||
| Matrix | Burstregion | Midregion | Late tailregion | Totalarea | Late fraction | t50 (h) |
| MatA | 11.81 | 16.27 | 8.14 | 36.21 | 0,22 | 118 |
| MatB | 12.60 | 17.59 | 6.77 | 36.96 | 0,18 | 103 |
| MatC | 12.97 | 19.79 | 9.76 | 42.52 | 0,23 | 114 |
| MatD | 7.51 | 15.21 | 7.64 | 30.36 | 0,25 | 125 |
| Δpeak areas (absolute and percentage changes) | ||||||
| Matrix | ΔBurstregion | ΔMidregion | ΔLateregion | ΔTotalarea | ΔLate fraction | ∆ t50 (h) |
| MatA | +2.38 (+25.3%) | −1.27 (-7.2%) | −6.26 (-43.5%) | −5.14 (-12.4%) | −0.12 (-34.5%) | −29.84 (-20.1%) |
| MatB | −0.65 (-4.9%) | −6.08 (-25.7%) | −13.41 (-66.5%) | −20.14 (-35.3%) | −0.17 (-48.1%) | −40.50 (-28.2%) |
| MatC | +1.47 (+12.8) | +0.45 (+2.4%) | −8.75 (-47.3%) | −6.83 (-13.8%) | −0.15 (-40.0%) | −28.00 (-19.7%) |
| MatD | +0.26 (+3.5%) | −0.71 (-4.4%) | −4.02 (-34%) | −4.47 (-12.8%) | −0.08 (-23.9%) | −14.88 (-10.7%) |
| Causes of variations | ||||||
| Notes | Rises at 37 °C for MatA and MatC (faster hydration/solubility; in MatC also weaker bead–fibre adhesion), roughly flat/slightly down for MatB, small uptick for MatD | Largest drop in MatB (PCL bead shrinkage + narrowed pores); MatC is slightly up due to the detached-bead pulse; MatA/MatD down modestly | All down at 37 °C; the biggest fall is MatB , then MatC, MatA, MatD | All down at 37 °C; the biggest fall is MatB, then MatC, MatA, MatD | Earlier at 37 °C for every matrix (largest shift in MatB, meaning whatever can leave tends to do so before tightening/ageing fully develops. | Earlier at 37 °C for every matrix (largest shift in MatB, meaning whatever can leave tends to do so before tightening/ageing fully develops. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




