Submitted:
10 February 2026
Posted:
11 February 2026
You are already at the latest version
Abstract

Keywords:
1. Introduction
1.1. Study Area
2. Materials and Methods
2.1. Sampling
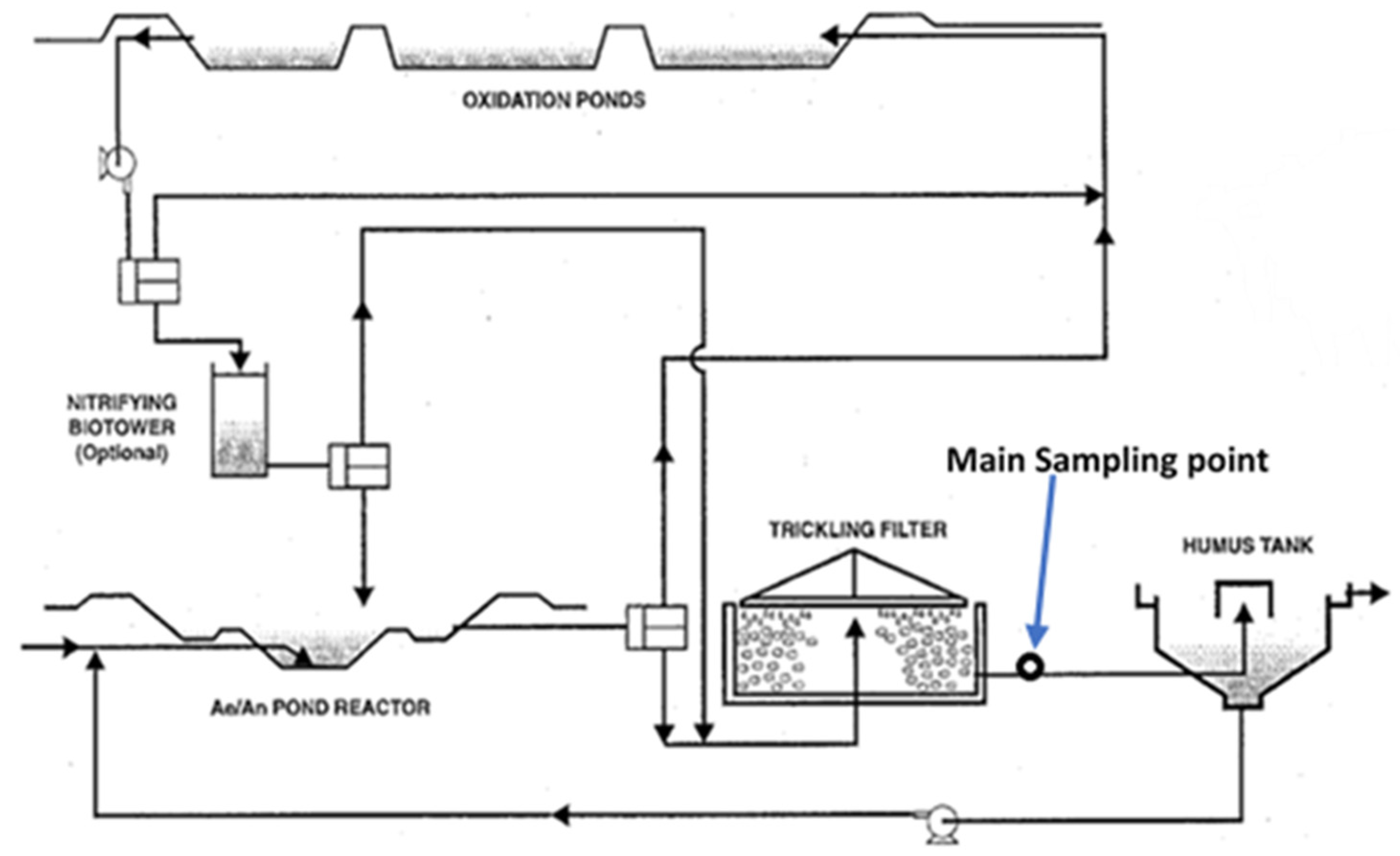
2.2. Materials
2.3. Statistical Analysis and Model Evaluation
2.4. Model Graphs
2.5. Quality Assurance and Replication
2.6. Final Equation in Terms of Coded and Actual Factors
2.7. Response Surface Methodology
3. Results
3.1. Preliminary Experiments
3.2. Response Surface Methodology
3.2.1. Descriptive and FIT Statistics
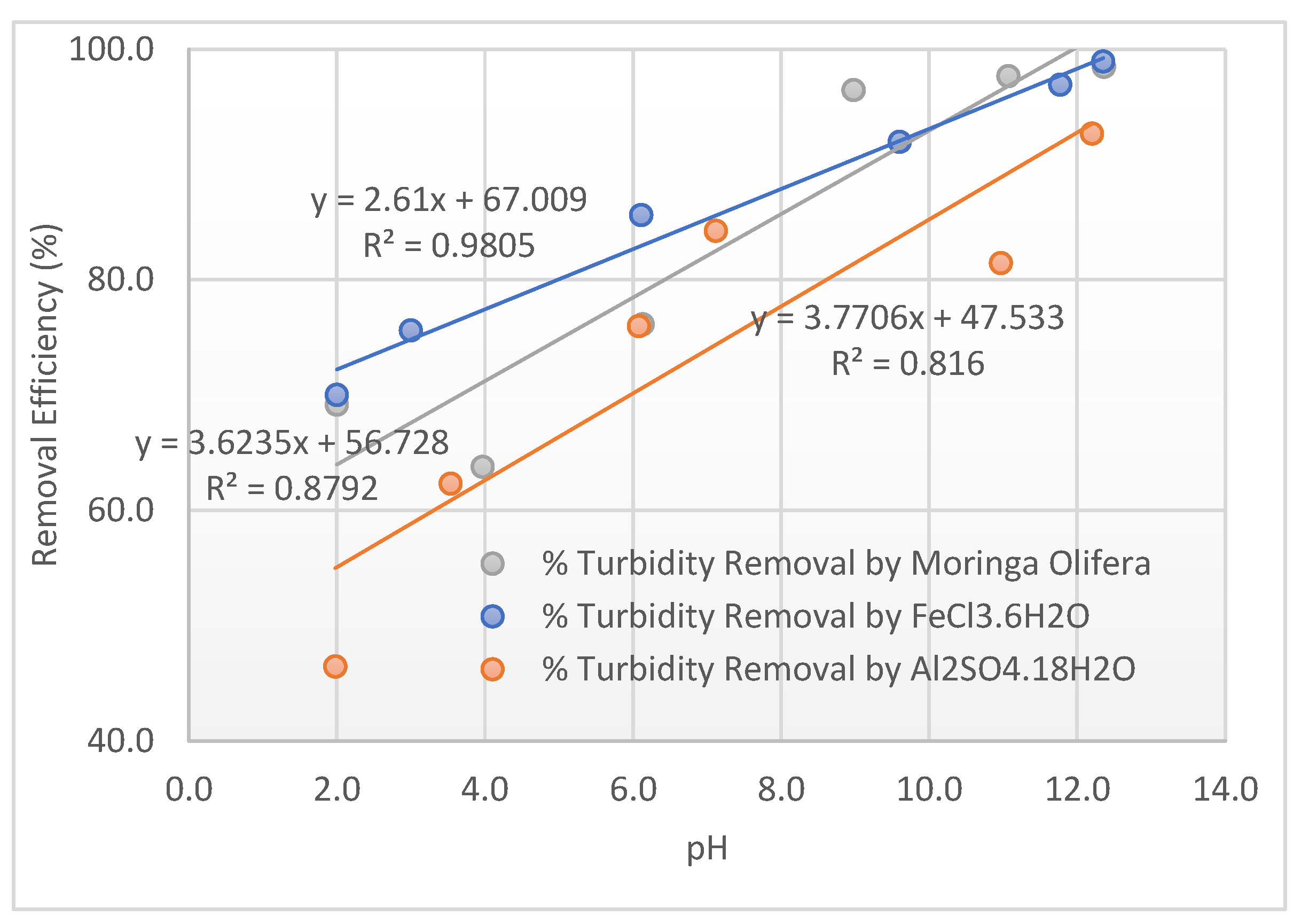
3.3. External Model Validation
4. Discussion
4.1. Interpretation of Coagulant Performance and Mechanisms
4.2. Insights from RSM Optimization and Model Validity
4.3. Regulatory Compliance and Economic Viability
4.4. Limitations and Recommendations for Future Work
5. Conclusions
- Ferric chloride (FeCl3·6H2O) emerged as the most effective coagulant, achieving the highest and most consistent multi-parameter removal, particularly for turbidity and TSS, under alkaline conditions (pH 12, 30 mg/L). Aluminum sulfate provided robust performance, while Moringa oleifera, though cost-effective at low doses, exhibited a narrow optimal dosage window beyond which performance deteriorated due to restabilization.
- Statistically significant quadratic models were developed with high predictive accuracy (R2 > 0.97 for key parameters). External validation with independent experimental runs confirmed the models’ reliability for forecasting treatment outcomes within the defined operational space.
- Most significantly, the optimized process using FeCl3 produces an effluent that meets Botswana’s discharge standards for TSS (≤ 30 mg/L), COD (≤ 75 mg/L), and turbidity (≤ 10 NTU), directly addressing the plant’s core compliance failure.
- The research provides a clear, data-driven pathway for implementation. Coagulant dosing can be integrated into existing infrastructure between the trickling filters and humus tanks with modest capital investment. A preliminary economic analysis indicates operational costs are manageable, with FeCl3 representing the optimal balance of performance and reliability for ensuring consistent compliance.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| WSP | Waste Stabilization Pond |
| TSS | Total Suspended Solids |
| TF | Trickling Filter |
| SS | Suspended Solids |
| RSM | Response Surface Methodology |
| RMSE | Root Mean Square Error |
| PWWTP | Palapye Wastewater Treatment Plant |
| PETRO | Pond Enhanced Treatment and Operation |
| OFAOT | One-Factor-at-a-Time |
| MRS | Multi-Response Surface |
| MP | Maturation Pond |
| HT | Humus Tank |
| FP | Facultative Pond |
| FeCl3·6H2O | Ferric chloride hexahydrate |
| DOE | Design of Experiment |
| COD | Chemical Oxygen Demand |
| CI | Confidence Interval |
| CCD | Central Composite Design |
| BOD | Biological Oxygen Demand |
| AS | Activated Sludge |
| AP | Anaerobic Pond |
| ANOVA | Analysis of Variance |
| Al2(SO4)3·18H2O | Aluminum sulfate octadecahydrate |
References
- Metcalf & Eddy, Inc. Wastewater Engineering: Treatment and Resource Recovery; McGraw-Hill: New York, NY, USA, 2014.
- Mara, D.D. Domestic Wastewater Treatment in Developing Countries; Earthscan: London, UK, 2004.
- Gopolang, O.P.; Letshwenyo, M.W. Performance evaluation of Palapye waste stabilization ponds. J. Water Sanit. Hyg. Dev. 2018, *8*, 123–134. [CrossRef]
- Botswana Department of Water Affairs. Effluent Standards for Wastewater Discharge; Government Printer: Gaborone, Botswana, 2012.
- Sarparastzadeh, H.; Saeedi, M.; Naeeni, M.A.; Faezipour, M. Use of chemical coagulation as a pretreatment for municipal wastewater. Iran. J. Environ. Health Sci. Eng. 2007, *4*, 265–270.
- Verma, A.K.; Dash, R.R.; Bhunia, P. A review on chemical coagulation/flocculation technologies for removal of colour from textile wastewater. J. Environ. Manag. 2012, *93*, 154–168. [CrossRef]
- Parmar, K.A.; et al. Coagulation-flocculation treatment of dairy wastewater using alum and lime. Int. J. Chem. Sci. 2011, *9*, 1365–1372.
- Irfan, M.; et al. Treatment of pulp and paper mill effluent using coagulation-flocculation process. J. Environ. Chem. Eng. 2017, *5*, 4026–4034. [CrossRef]
- Marañón, E.; Fernández, Y.; Fernández, Y.; Castrillón, L. Removal of dissolved organic carbon from landfill leachate by coagulation–flocculation. Waste Manag. 2010, *30*, 862–868. [CrossRef]
- Oubrayme, M.; et al. Use of natural coagulants for refinery wastewater treatment. J. Water Process Eng. 2016, *14*, 108–115. [CrossRef]
- Hendrawati, H.; et al. Utilization of Moringa oleifera seed as natural coagulant for textile wastewater treatment. J. Environ. Sci. Technol. 2016, *9*, 84–92. [CrossRef]
- Maurya, S.; Daverey, A. Evaluation of plant-based natural coagulants for municipal wastewater treatment. 3 Biotech 2018, *8*, 77. [CrossRef]
- Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF): Washington, DC, USA, 2017.
- Shaylinda, M.Z.N.; et al. Application of response surface methodology (RSM) for optimization of ammoniacal nitrogen removal from semi-aerobic landfill leachate using ion exchange resin. Desalination Water Treat. 2018, *128*, 334–343. [CrossRef]
- Machlor, A.; et al. Coagulation-flocculation process optimization using response surface methodology for landfill leachate treatment. Int. J. Environ. Sci. Dev. 2014, *5*, 312–317. [CrossRef]
- Fendri, I.; et al. Optimization of coagulation-flocculation process for landfill leachate treatment using response surface methodology. Environ. Technol. 2013, *34*, 1217–1224. [CrossRef]
- Wang, S.; et al. Optimization of coagulation process for water treatment using response surface methodology. J. Environ. Sci. 2011, *23*, 264–270. [CrossRef]
- Khayet, M.; et al. Optimization of reverse osmosis process for water treatment using response surface methodology. Desalination 2011, *268*, 89–98. [CrossRef]
- Wu, C.; et al. Interference of metal ions in COD determination during coagulation studies. Environ. Technol. 2012, *33*, 2453–2460. [CrossRef]
- Besson, F.; Guiraud, P. pH effect on coagulation performance of aluminum and iron salts. Water Sci. Technol. 2013, *68*, 1283–1289. [CrossRef]
- Pérez, A.G.; et al. pH-induced coagulation mechanisms in wastewater treatment. J. Water Process Eng. 2017, *19*, 120–128. [CrossRef]
- Botswana Department of Water Affairs. Effluent Standards for Wastewater Discharge; Government Printer: Gaborone, Botswana, 2012.
- Duan, J.; Gregory, J. Coagulation by hydrolysing metal salts. Adv. Colloid Interface Sci. 2003, *100-102*, 475–502. [CrossRef]
- Gan, Y.; Zhang, Y.; Song, Z.; Zheng, J.; Zhang, Z.; Wang, Y.; Zhang, Q. Ferric chloride versus alum: Which is more effective for coagulation? Water Sci. Technol. 2017, *76*, 289–297. [CrossRef]
- Ndabigengesere, A.; Narasiah, K.S.; Talbot, B.G. Active agents and mechanism of coagulation of turbid waters using Moringa oleifera. Water Res. 1995, *29*, 703–710. [CrossRef]
- Ndabigengesere, A.; Narasiah, K.S. Quality of water treated by coagulation using Moringa oleifera seeds. Water Res. 1998, *32*, 781–791. [CrossRef]
- Bratby, J. Coagulation and Flocculation in Water and Wastewater Treatment; IWA Publishing: London, UK, 2016.
- Montgomery, D.C. Design and Analysis of Experiments, 10th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2019.
- Derringer, G.; Suich, R. Simultaneous optimization of several response variables. J. Qual. Technol. 1980, *12*, 214–219. [CrossRef]
- Gregory, J. Particles in Water: Properties and Processes; IWA Publishing: London, UK, 2006.
- Sánchez-Martín, J.; Ghebremichael, K.; Beltrán-Heredia, J. Comparison of single-step and two-step purified Moringa oleifera coagulant for surface water treatment. J. Water Health 2010, *8*, 294–306. [CrossRef]
- Matilainen, A.; Vepsäläinen, M.; Sillanpää, M. Natural organic matter removal by coagulation during drinking water treatment: A review. Adv. Colloid Interface Sci. 2010, *159*, 189–197. [CrossRef]
- Pritchard, M.; Mkandawire, T.; O’Neill, J.G. Biological, chemical and physical drinking water quality from shallow wells in Malawi: The case of Moringa oleifera as a natural coagulant. Phys. Chem. Earth 2007, *32*, 1167–1172. [CrossRef]
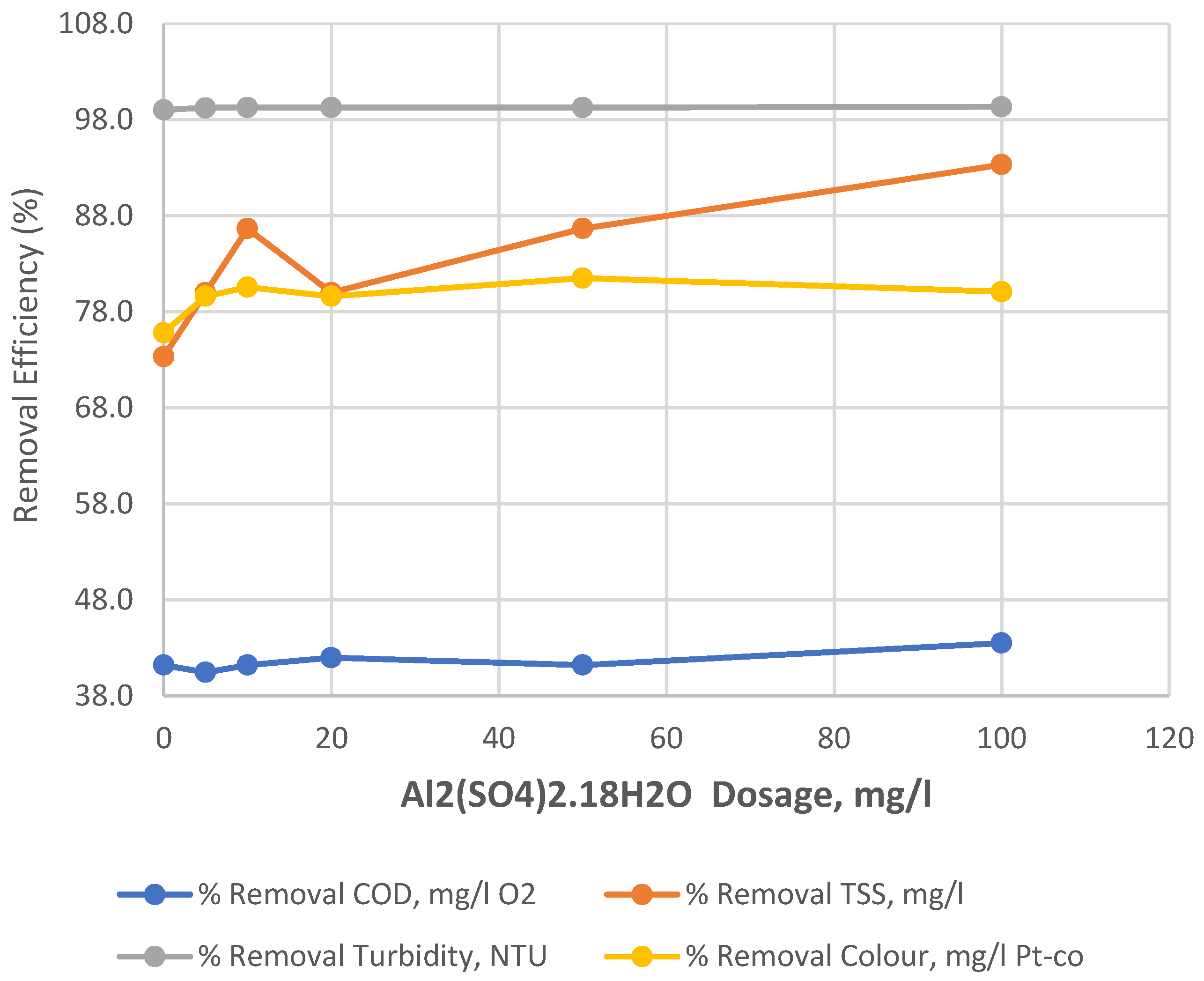
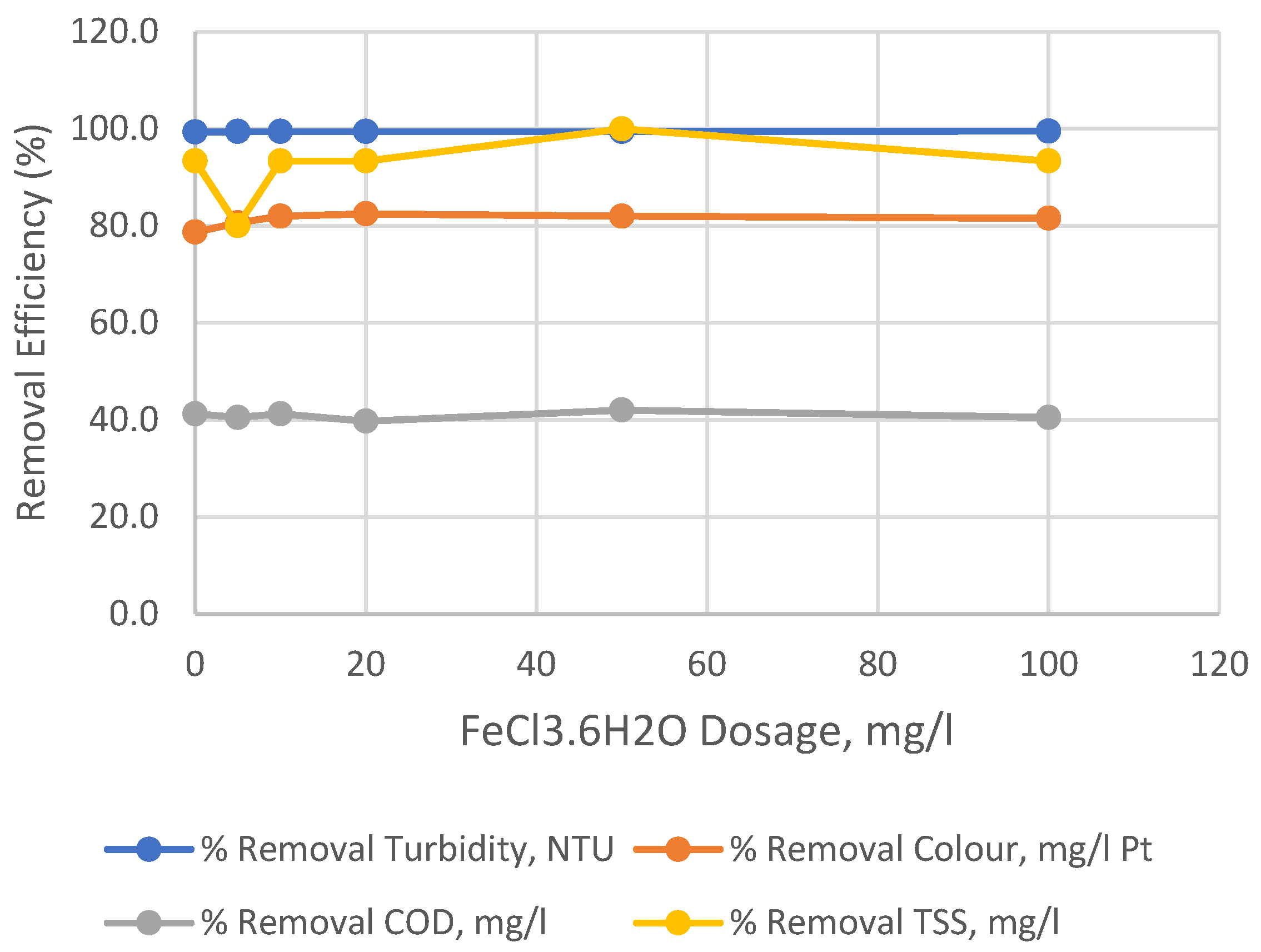
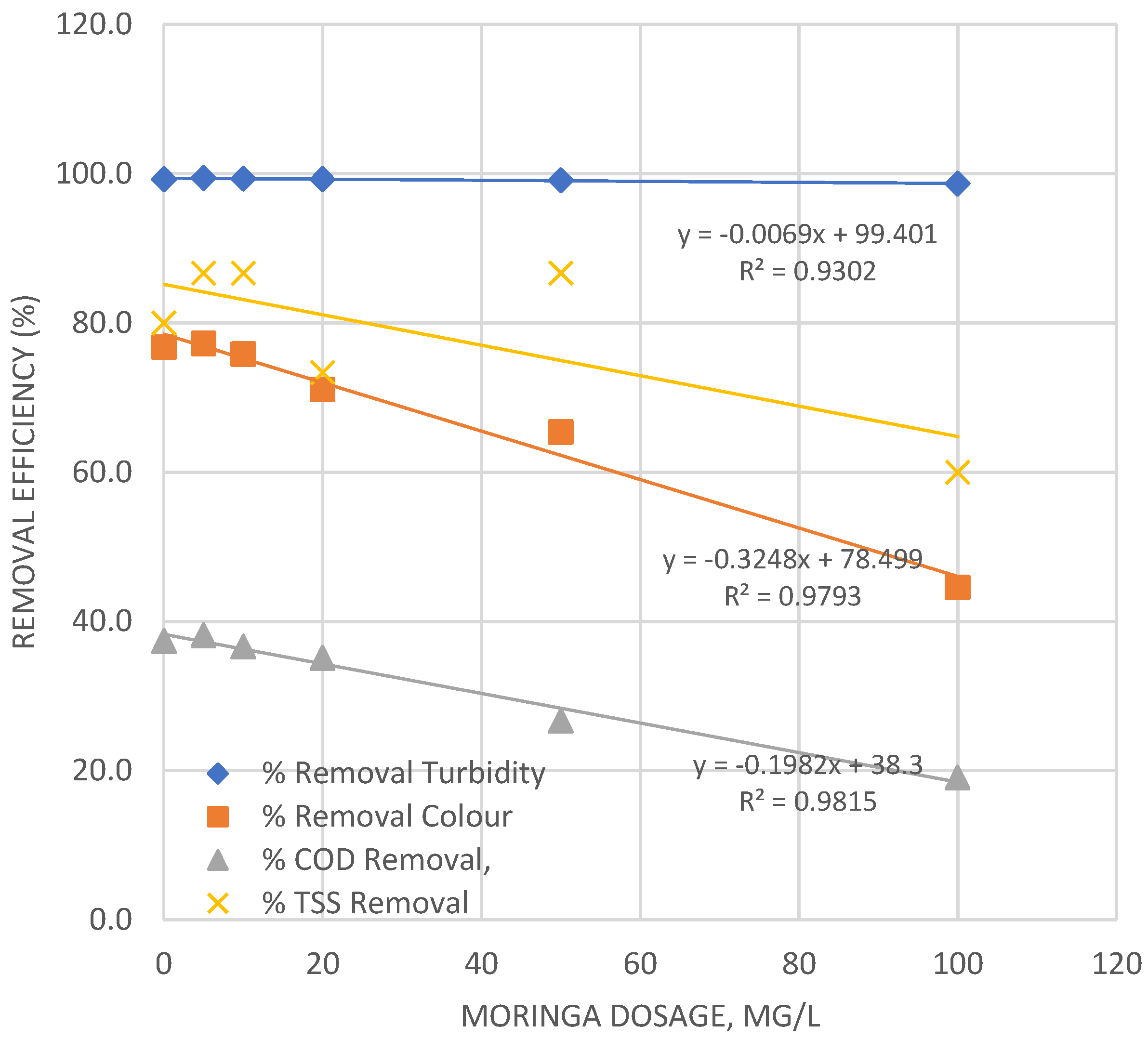
| Input variable | Numeric Factor Levels | ||||
| -1.414 | -1 | 0 | +1 | + 1.414 | |
| Lower | Low | Centre | High | Higher | |
| pH | 8.38 | 9 | 10.5 | 12 | 12.6 |
| Al2(SO4)3.18H2O, mg/L | 2.93 | 5 | 10 | 15 | 17.1 |
| FeCl3.6H2O, mg/L | 5.86 | 10 | 20 | 30 | 34.1 |
| Moringa, mg/L | 0.76 | 2 | 5 | 8 | 9.2 |
| Al2(SO4)3.18H2O | FeCl3.6H2O | Moringa | |||
| pH | % Turbidity Removal | pH | % Turbidity Removal | pH | % Turbidity Removal |
| 2.0 | 46.5 | 2.0 | 70.0 | 2.0 | 69.2 |
| 3.5 | 62.3 | 3.0 | 75.6 | 4.0 | 63.8 |
| 6.1 | 76.0 | 6.1 | 85.6 | 6.1 | 76.1 |
| 7.1 | 84.3 | 9.6 | 92.0 | 9.0 | 96.4 |
| 11.0 | 81.5 | 11.8 | 96.9 | 11.1 | 97.7 |
| 12.2 | 92.7 | 12.4 | 98.9 | 12.4 | 98.5 |
| Dosage, | Turbidity, | Colour, | COD, | TSS, | Removal COD, | Removal TSS, | Removal Turbidity, | Removal Colour, | % Removal COD, | % Removal TSS, | % Removal Turbidity | % Removal Colour, |
| mg/L | NTU | mg/L Pt-co | mg/L O2 | mg/L | mg/L O2 | mg/L | NTU | mg/L Pt-co | % | % | % | % |
| 0 | 0.12 | 51 | 77 | 4 | 54 | 11 | 12.28 | 160 | 41.2 | 73.3 | 99.0 | 75.8 |
| 5 | 0.095 | 43 | 78 | 3 | 53 | 12 | 12.305 | 168 | 40.5 | 80.0 | 99.2 | 79.6 |
| 10 | 0.09 | 41 | 77 | 2 | 54 | 13 | 12.31 | 170 | 41.2 | 86.7 | 99.3 | 80.6 |
| 20 | 0.09 | 43 | 76 | 3 | 55 | 12 | 12.31 | 168 | 42.0 | 80.0 | 99.3 | 79.6 |
| 50 | 0.09 | 39 | 77 | 2 | 54 | 13 | 12.31 | 172 | 41.2 | 86.7 | 99.3 | 81.5 |
| 100 | 0.08 | 42 | 74 | 1 | 57 | 14 | 12.32 | 169 | 43.5 | 93.3 | 99.4 | 80.1 |
| Dosage, mg/L | Turbidity, NTU | Colour, mg/L Pt-co | COD, mg/L O2 | TSS, mg/L | Removal COD, mg/L O2 | Removal TSS, mg/L | Removal Turbidity, NTU | Removal Colour, mg/L Pt-co | % Removal Turbidity, NTU | % Removal Colour, mg/L Pt | % Removal COD, mg/L O2 | % Removal TSS, mg/L |
| 0 | 0.085 | 45 | 77 | 1 | 54 | 14 | 12.315 | 166 | 99.3 | 78.7 | 41.2 | 93.3 |
| 5 | 0.07 | 41 | 78 | 3 | 53 | 12 | 12.33 | 170 | 99.4 | 80.6 | 40.5 | 80.0 |
| 10 | 0.07 | 38 | 77 | 1 | 54 | 14 | 12.33 | 173 | 99.4 | 82.0 | 41.2 | 93.3 |
| 20 | 0.07 | 37 | 79 | 1 | 52 | 14 | 12.33 | 174 | 99.4 | 82.5 | 39.7 | 93.3 |
| 50 | 0.07 | 38 | 76 | 0 | 55 | 15 | 12.33 | 173 | 99.4 | 82.0 | 42.0 | 100.0 |
| 100 | 0.06 | 39 | 78 | 1 | 53 | 14 | 12.34 | 172 | 99.5 | 81.5 | 40.5 | 93.3 |
| Dosage, mg/L | Turbidity, NTU | Colour, mg/L Pt | COD, mg/L | TSS, mg/L | Removal Turbidity, NTU | Removal Colour, mg/L Pt | COD Removal, mg/L | TSS Removal , mg/L | % Removal Turbidity |
% Removal Colour | % COD Removal, | % TSS Removal |
| 0 | 0.09 | 49 | 82 | 3 | 12.31 | 162 | 49 | 12 | 99.3 | 76.8 | 37.4 | 80.0 |
| 5 | 0.07 | 48 | 81 | 2 | 12.33 | 163 | 50 | 13 | 99.4 | 77.3 | 38.2 | 86.7 |
| 10 | 0.08 | 51 | 83 | 2 | 12.32 | 160 | 48 | 13 | 99.4 | 75.8 | 36.6 | 86.7 |
| 20 | 0.09 | 61 | 85 | 4 | 12.31 | 150 | 46 | 11 | 99.3 | 71.1 | 35.1 | 73.3 |
| 50 | 0.11 | 73 | 96 | 2 | 12.29 | 138 | 35 | 13 | 99.1 | 65.4 | 26.7 | 86.7 |
| 100 | 0.165 | 117 | 106 | 6 | 12.235 | 94 | 25 | 9 | 98.7 | 44.5 | 19.1 | 60.0 |
| Trickling filter effluent Characterization | Coagulant Stock Solution (10 g/l) | ||||
| Turbidity, NTU | 50.7 | Fil. Turb, NTU: | 4 | Coagulant Name | pH |
| Colour, mg/L, Pt-co | 762 | Fil. Colour, mg/L, Pt-co | 147 | Al2(SO4)3.18H2O | 3.7 |
| COD, mg/L | 202 | FeCl3.6H2O | 2.12 | ||
| TSS, mg/L | 65 | Moringa | 6.25 | ||
| Response | Model | Lack of FIT | FIT Statistics | |||||
| F values | P value |
F values | P value | R2 | Adj R2 | Pred. R2 | Adq. precision | |
| Turbidity | 114.47 | <0.0001 | 1.7 | 0.1789 | 0.9764 | 0.9676 | 0.9481 | 41.4541 |
| Colour | 244.43 | <0.0001 | 1.58 | 0.2161 | 0.9869 | 0.9828 | 0.9796 | 66.5861 |
| COD | 118.25 | <0.0001 | 1.71 | 0.1834 | 0.9718 | 0.9636 | 0.9456 | 47.2574 |
| TSS | 21.22 | <0.0001 | 0.9832 | 0.5234 | 0.8908 | 0.8488 | 0.7668 | 16.2008 |
| Run | Parameter | Predicted Value | Actual Value | Percent Error (%) | RMSE* |
| 1 | Turbidity (NTU) | 0.45 | 0.47 | 4.3 | 0.41 NTU |
| Colour (mg/L Pt-Co) | 42.1 | 44.5 | 5.4 | 8.7 mg/L | |
| COD (mg/L O2) | 58.3 | 62.8 | 7.2 | 5.2 mg/L | |
| TSS (mg/L) | 3.2 | 3.4 | 5.9 | 1.8 mg/L | |
| 2 | Turbidity (NTU) | 0.38 | 0.36 | 5.6 | 0.41 NTU |
| Colour (mg/L Pt-Co) | 38.7 | 40.2 | 3.7 | 8.7 mg/L | |
| COD (mg/L O2) | 61.5 | 65.1 | 5.5 | 5.2 mg/L | |
| TSS (mg/L) | 2.8 | 2.9 | 3.4 | 1.8 mg/L | |
| 3 | Turbidity (NTU) | 0.52 | 0.50 | 4.0 | 0.41 NTU |
| Colour (mg/L Pt-Co) | 45.3 | 47.8 | 5.2 | 8.7 mg/L | |
| COD (mg/L O2) | 55.8 | 60.3 | 7.5 | 5.2 mg/L | |
| TSS (mg/L) | 3.5 | 3.7 | 5.4 | 1.8 mg/L | |
| Average | All Parameters | - | - | 5.4 | - |
| Overall RMSE | - | - | - | - | See below |
| Parameter | Botswana Standard (Typical Limit) | Optimized Effluent Value (FeCl3) | Compliance Status |
| TSS | ≤ 30 mg/L | 1 mg/L | Fully Compliant |
| COD | ≤ 75 mg/L | 55 mg/L | Fully Compliant |
| Turbidity | ≤ 10 NTU | 0.07 NTU | Fully Compliant |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




