1. Introduction
Breast cancer remains the most frequently diagnosed malignancy and the leading cause of cancer-related mortality among women worldwide. According to recent global estimates, over 2.3 million new cases are diagnosed each year, representing approximately 11.7% of all cancers [
1]. Advances in screening programs, early detection, and multidisciplinary treatment approaches have significantly improved survival rates, shifting the focus toward optimizing oncologic safety while preserving quality of life and aesthetic outcomes.
Among surgical treatment options, nipple-sparing mastectomy (NSM) has gained increasing acceptance over the past two decades. This technique allows for complete removal of breast glandular tissue while preserving the skin envelope and nipple-areola complex (NAC) [
2], thereby offering superior cosmetic results compared to traditional mastectomy approaches. NSM is now considered a safe and effective procedure in selected patients with early-stage breast cancer or high-risk genetic mutations, provided appropriate oncologic criteria are met.
Breast reconstruction following mastectomy has also evolved considerably, particularly with the development of prepectoral and subpectoral implant-based techniques. Subpectoral reconstruction, the traditional approach, involves placing the implant beneath the pectoralis major muscle, which can provide enhanced soft tissue coverage but is often associated with increased postoperative pain, animation deformities, and longer recovery times. In contrast, prepectoral reconstruction—where the implant is placed above the muscle, directly beneath the skin flap and supported or not [
3] by acellular dermal matrices (ADM) or synthetic meshes —has emerged as a viable alternative. This technique offers several advantages, including reduced postoperative discomfort, preservation of muscle function, and more natural breast contour, especially when performed in carefully selected patients with good-quality skin flaps [
4].
The expanding indications and improved reconstructive techniques associated with NSM and implant-based reconstruction have underscored the importance of managing postoperative healing, minimizing complications, and supporting optimal tissue recovery—critical aspects for achieving both oncological and aesthetic goals [
5]. In this context, novel adjunctive therapies aimed at enhancing wound healing, reducing local inflammation, and preventing infection are being increasingly explored in the postoperative setting. At the UOC Breast Surgery of AOUI Verona, approximately 73% of patients who are candidates for mastectomy undergo immediate breast reconstruction. This high rate reflects the multidisciplinary approach and the centre’s commitment to combining oncologic safety with optimal aesthetic and functional outcomes, especially in nipple-sparing mastectomy cases.
A growing body of experimental evidence suggests that reactive oxygen species (ROS) are not merely cytotoxic metabolic by-products but also play crucial roles in cell signalling and regulation. This apparent paradox may be explained, at least in part, by the concentration-dependent effects of ROS, where low levels contribute to physiological processes while high concentrations are associated with cellular damage. Biochemical and metabolic studies of the microenvironment in chronic skin lesions have demonstrated the essential role of oxygen in tissue repair processes, including cell proliferation, angiogenesis, collagen synthesis, and defence against pathogenic microorganisms [
6]. Neutrophils and macrophages recruited by inflammatory mediators produce ROS at the wound site, which act as secondary messengers for both tissue-resident cells (such as fibroblasts) and immune cells involved in the healing process [
7].
Interestingly, similar beneficial effects have been observed following the exogenous application of ROS to wounds [
8]. These applications did not produce the expected oxidative damage, likely due to the upregulation of cytoprotective genes and the activation of endogenous antioxidant defence mechanisms [
7]. The clinical efficacy of oxygen therapies in chronic wound management is well established, with treatments such as hyperbaric oxygen therapy and oxygen-ozone therapy routinely used in practice. The mechanism of action of oxygen-enriched oil formulations includes not only the controlled release of ROS but also a pH-lowering effect caused by the micro release of carboxylic acids upon interaction with the aqueous component of wound exudate. Maintaining a slightly acidic wound environment is beneficial in preventing chronicity and creating unfavourable conditions for microbial proliferation.
Based on these biological premises, Novox® Cup, a topical medical device consisting of a polyurethane dome internally coated with an oxygen-enriched oleic matrix capable of releasing reactive oxygen species (ROS), has been developed. This innovative dressing is designed to maintain intimate contact with the surgical wound, creating a moist and slightly acidic microenvironment that promotes tissue regeneration, angiogenesis, and antimicrobial defence. Its anatomical shape allows for even distribution of ROS over the wound surface while providing a mechanical barrier against external contaminants.
2. Materials and Methods
Considering the growing use of nipple-sparing mastectomy combined with immediate implant-based reconstruction—either in prepectoral or subpectoral planes—and the importance of optimizing postoperative healing, we designed a single-centre observational study to evaluate the clinical utility of Novox® Cup.
Our prospective observational study aims to evaluate the efficacy of the NovoX® CUP medical device in the dressing of post-NSM surgical wounds, evaluating clinical-surgical outcomes and the impact on QoL.
To evaluate the efficacy of NovoX® CUP advanced dressing in promoting proper healing of the skin flap and nipple-areola complex (NAC) in patients undergoing nipple sparing mastectomy (NSM) with immediate reconstruction, by measuring the incidence of post-operative skin complications and the degree of flap viability according to the adopted standardized scale.
- 2.
Secondary Objectives
To evaluate the impact of the treatment on the quality of life (QoL) of patients, by administering the Wound-QoL17 questionnaire at the same follow-up intervals.
Monitor the incidence, type and severity of post-operative complications according to the Clavien-Dindo classification, within 90 days and up to 365 days after surgery.
Document the evolution of the surgical wound, skin flaps and scar throughout the follow-up through standardized digital imaging.
To explore the association between clinical and anamnestic variables (age, BMI, smoking habit, comorbidities, previous radiotherapy or breast surgery, duration of surgery) and skin healing outcomes.
The study was conducted at the UOC of Breast Surgery AOUI Verona and included a total of 54 patients treated between January 2025 and January 2026. Of these, 36 patients underwent bilateral prophylactic nipple-sparing mastectomy, while 18 patients underwent unilateral nipple-sparing mastectomy for histologically confirmed breast cancer.
A total of 90 Novox® Cup devices were applied in the immediate postoperative period following implant-based breast reconstruction, either in a prepectoral or subpectoral plane, depending on surgical indications and intraoperative assessment.
Study Population
Eligible participants were identified through a review of electronic medical records – EUSOMA DATABREAST- and operative reports.
Inclusion criteria were:
Female patients aged ≥18 years
Underwent nipple-sparing mastectomy for therapeutic or prophylactic indications
Received immediate breast reconstruction (prepectoral or subpectoral, with or without ADM)
Patients undergoing nipple-sparing mastectomy with intraoperative evidence of compromised skin flap perfusion, as assessed by clinical evaluation (e.g., pallor, delayed capillary refill, or reduced dermal bleeding).
Patients undergoing prophylactic mastectomy for high genetic risk (e.g., BRCA1/2 mutation carriers) receiving immediate implant-based breast reconstruction.
Application of the Novox® Cupdevice during the postoperative period
Regarding the application of the Novox® Cup, the medication was placed directly in the operating room at the end of the surgery. It was not fixed to the skin with any adhesive but was instead kept in place by a supportive post-surgical bra. The device remained in place for 48 hours post-surgery, after which the patient was instructed to replace it at home with a new device. The dressing was then removed after an additional 48-hour application time. This simple yet effective method contributed to a smooth postoperative recovery process, with minimal discomfort reported by the patients.
For each patient, information relevant to the study was collected. This includes age, BMI, gender, ethnicity, smoking habits, cardiovascular and/or rheumatologic comorbidities, and any usual therapies, within a complete remote medical history, duration of surgery, previous breast surgery, and prior radiation treatment.
All data were collected and securely stored in a dedicated database by the responsible investigators, in full compliance with current data protection regulations and best practices for clinical research conduct.
Digital photographs were obtained at each scheduled follow-up visit for all patients. These images provided qualitative documentation of wound healing progression, supporting the clinical assessments.
Data were collected retrospectively from the patients’ medical records, operative notes, follow-up visits, and nursing documentation.
During the peri-operative phase, the literature highlights the pivotal role of the breast care nurse in guiding, informing, and supporting the patient throughout the entire care pathway. Nursing interventions described in the selected articles focus primarily on the preoperative consultation, where effective communication becomes essential to ensure that patients clearly understand each stage of breast cancer treatment. The breast care nurse acts as a key point of reference, offering personalized information, addressing concerns, and fostering a trusting relationship through an empathetic and patient-centered approach.
In this context, the nurse provides not only technical and practical guidance—such as advice on preoperative preparation, the choice of supportive bras, options regarding breast implants, and access to support groups—but also continuous emotional and psychological support. Particular emphasis is placed on the nurse’s role in clarifying doubts related to surgical safety, medication management, and perioperative procedures, thereby reducing anxiety and promoting informed decision-making.
Furthermore, the development and use of educational tools, such as written guides or structured informational strategies, are strongly recommended to enhance patient understanding of the perioperative pathway. Through clear, consistent communication and ongoing assistance before and after surgery, the breast care nurse ensures continuity of care, empowers patients, and supports their preparation for both the surgical experience and postoperative recovery.
The post-discharge follow-up mode was organized as follows:
The patient was scheduled an outpatient visit 5-7 days after surgery, during which the outcomes from a clinical-surgical point of view were monitored to record data on the viability of the NAC/flap, on complications using Clavien-Dindo scale and the “Wound-QoL17” questionnaire will be administered.
The patient was scheduled an outpatient visit 30 days after surgery, during which the outcomes from a clinical-surgical point of view were monitored to record data on the viability of the NAC/flap, on complications using Clavien-Dindo scale and the “Wound-QoL17” questionnaire will be administered.
The patient was scheduled an outpatient visit 90 days after surgery, during which the outcomes from a clinical-surgical point of view were monitored to record data on the viability of the NAC/flap, on complications using Clavien-Dindo scale and the “Wound-QoL17” questionnaire will be administered.
The surgeon-s point of view
The follow-up modality from the surgeon’s point of view will be organized as follows:
The patient will have the advanced dressing in question placed in the operating room at the end of the surgery (0 day-post operation); this dressing will then be replaced by the medical-nursing staff in the ward after 48 hours or in II day-post operation.
Once discharged home, the patient will then replace her after a further 48 hours, then in IV day-post operation.
The patient will then be scheduled an outpatient visit 5-7 days after surgery.
The patient will be evaluated in the ward according to the flap viability rating scale (
Table 1) illustrated below in 0-1-2 day after surgery and at the first outpatient dressing or between V and VII day after surgery and any other complications will be recorded according to Clavien-Dindo (
Table 2).
The Skin Flap Viability Rating Scale (
Table 1) is based on criteria commonly used in the assessment of post-surgical skin perfusion (color, temperature, capillary refill, edema, integrity).
The patient’s point of view
Postoperative quality of life (QoL) was assessed using the validated
Wound-QoL17 questionnaire (Table 3), a 17-item patient-reported outcome measure specifically designed to assess the physical, psychological, and functional burden of wounds over the preceding 7 days. Each item was scored using a 5-point Likert scale:
0 = By no means
1 = Little
2 = Enough
3 = A lot
4 = Very much
Higher scores reflect greater perceived burden.
The questionnaire covers multiple dimensions of patient well-being, including:
Physical symptoms (e.g., pain, smell, secretion, sleep disruption)
Emotional impact (e.g., sadness, frustration, anxiety about healing)
Functional limitations (e.g., difficulty moving, climbing stairs, or participating in social and recreational activities)
Social and financial burden
Patients were instructed to complete the Wound-QoL17 on postoperative day 14, coinciding with the typical timeline for drain removal and wound stabilization. Responses were collected anonymously and analysed using descriptive statistics.
Data were collected from completed questionnaires available in the clinical record during early postoperative follow-up. In cases where the questionnaire was only partially completed, available responses were still included in domain-level analysis, provided at least 75% of items were answered.
These data were used to describe the subjective wound-related experience of patients and to explore possible associations with clinical outcomes such as complication rates, drainage duration, and healing time.
The following clinical and patient-reported outcomes were assessed:
-
Local Complications: The incidence of postoperative complications was recorded, including:
- ○
Infection (defined by clinical signs and/or need for antibiotics or surgical revision),
- ○
Seroma (requiring aspiration or delaying wound healing),
- ○
Wound dehiscence (partial or complete separation of surgical wound edges).
Healing Time: Defined as the time (in days) from surgery to complete wound closure, with no drainage or dressing required.
Drainage Duration: Duration of surgical drains was recorded in days; prolonged drainage was defined as >14 days postoperatively.
Reintervention: The need for surgical reintervention due to complications (e.g., implant removal, revision surgery) was documented.
Primary Endpoints
Incidence of skin flap impairment (NAC and mastectomy flaps) assessed according to the Skin Flap Viability Scale (scores 1–4) in the timeframes:
Overall incidence of early (within 30 days) surgical wound-related skin complications after NSM: flap distress, infection, seroma, dehiscence, partial/total NAC necrosis, etc.
Secondary Endpoints
Wound-QoL17 total score, detected in the same ranges, to quantify the impact on quality of life.
Incidence and severity of general post-operative complications graded according to Clavien-Dindo within 90 days and up to 365 days.
Time to complete wound healing (defined as complete closure and absence of signs of exudate, infection, or tissue distress).
Standardized photographic evolution of the wound and NAC, analyzed at follow-up times.
Statistical association between clinical/anamnestic variables and outcomes (complications, flap viability scores, Wound-QoL17).
Success criteria
Effective treatment is considered if all of the following criteria are met:
-
Flap vitality
Complications
No major wound-related complications (Clavien-Dindo ≥ III)
Absence of NAC necrosis (partial/total)
Absence of infection requiring IV antibiotics
-
Quality of life
Complete recovery within the expected time (30–45 days), with no clinically relevant delays.
Failure criteria
Treatment failure is considered if even one of the following occurs:
Significant flap suffering (score 3–4) persistent or progressive.
NAC necrosis (partial or total).
Major wound-related complications (Clavien-Dindo III–IV).
Severe infection requiring drainage or surgical revision.
Delayed healing > 60 days with persistence of exudate or dehiscence.
Significant worsening of QoL (clinically relevant increase in Wound-QoL17).
3. Results
A total of 54 patients who underwent nipple-sparing mastectomy with immediate breast reconstruction and postoperative application of the Novox® Cup device were included in this retrospective study. The mean age of the cohort was 51.5 years, and the mean body mass index (BMI) was 23,9 kg/m² (range18,6 -35,7). All patients were female, as per inclusion criteria. Smoking habits were recorded, with 5,4% identified as current smokers. Comorbidities, including cardiovascular and rheumatologic diseases, were present in 40,3% of patients. Usual therapies prior to surgery were also documented.
Of the total, no patient had previously undergone radiotherapy and only one patient had ipsilateral surgery.
The average duration of the intervention was 150 minutes. The most frequent surgical incisions (95%) were made at the breast sulcus or radial incisions at the upper-external quadrant.
All patients underwent nipple-sparing mastectomy, followed by immediate breast reconstruction, either prepectoral or subpectoral, with or without the use of Acellular Dermal Matrix (ADM). The Novox® Cup device was applied postoperatively in all cases. The initial application lasted for 48 hours immediately after surgery in the operating room, with no adhesives used to fix the device; instead, it was maintained in place using a supportive post-surgical bra. Patients were instructed to replace the device at home for an additional 48 hours, after which the dressing was removed.
- Results from Skin Flap Vitality Scale:
A total of 54 patients undergoing breast surgery with skin flap preservation were prospectively evaluated postoperatively using the Skin Flap Vitality Scale (grades 1–4). This standardized clinical scale assesses flap viability based on color, temperature, capillary refill, edema, and the presence of tissue suffering or necrosis.
One patient (1.85%) developed total nipple–areola complex (NAC) necrosis, corresponding to grade 4 vitality impairment. This represented a severe and irreversible vascular compromise, requiring surgical assessment and dedicated management.
Partial necrosis was observed in 8% of patients. These cases involved limited areas of tissue suffering without progression to full-thickness necrosis. All were successfully managed conservatively through advanced wound dressings and close outpatient follow-up, without the need for surgical revision.
Regarding flap vitality, 18% of patients were classified as grade 2 (reduced but acceptable vitality). These flaps exhibited mild and homogeneous color alterations, capillary refill times of 2–3 seconds, and mild to moderate edema, without signs of irreversible ischemia. In all grade 2 cases, a protocol of enhanced clinical monitoring combined with the application of Novox Cup dressings was implemented. Complete clinical resolution was achieved in 100% of these patients, with progressive normalization of flap color, temperature, and capillary refill, and no progression to higher-risk categories.
Only 1.84% of patients were classified as grade 3, characterized by evident signs of moderate flap suffering, including delayed or absent capillary refill, reduced temperature, and areas of dubious vitality. These patients underwent intensified surveillance and early therapeutic intervention, including optimization of local wound care and serial surgical evaluation, aimed at preventing progression toward necrosis.
Overall, the distribution of complications demonstrates a low incidence of severe flap-related adverse outcomes, with the majority of vascular alterations remaining mild and reversible when promptly identified and treated.
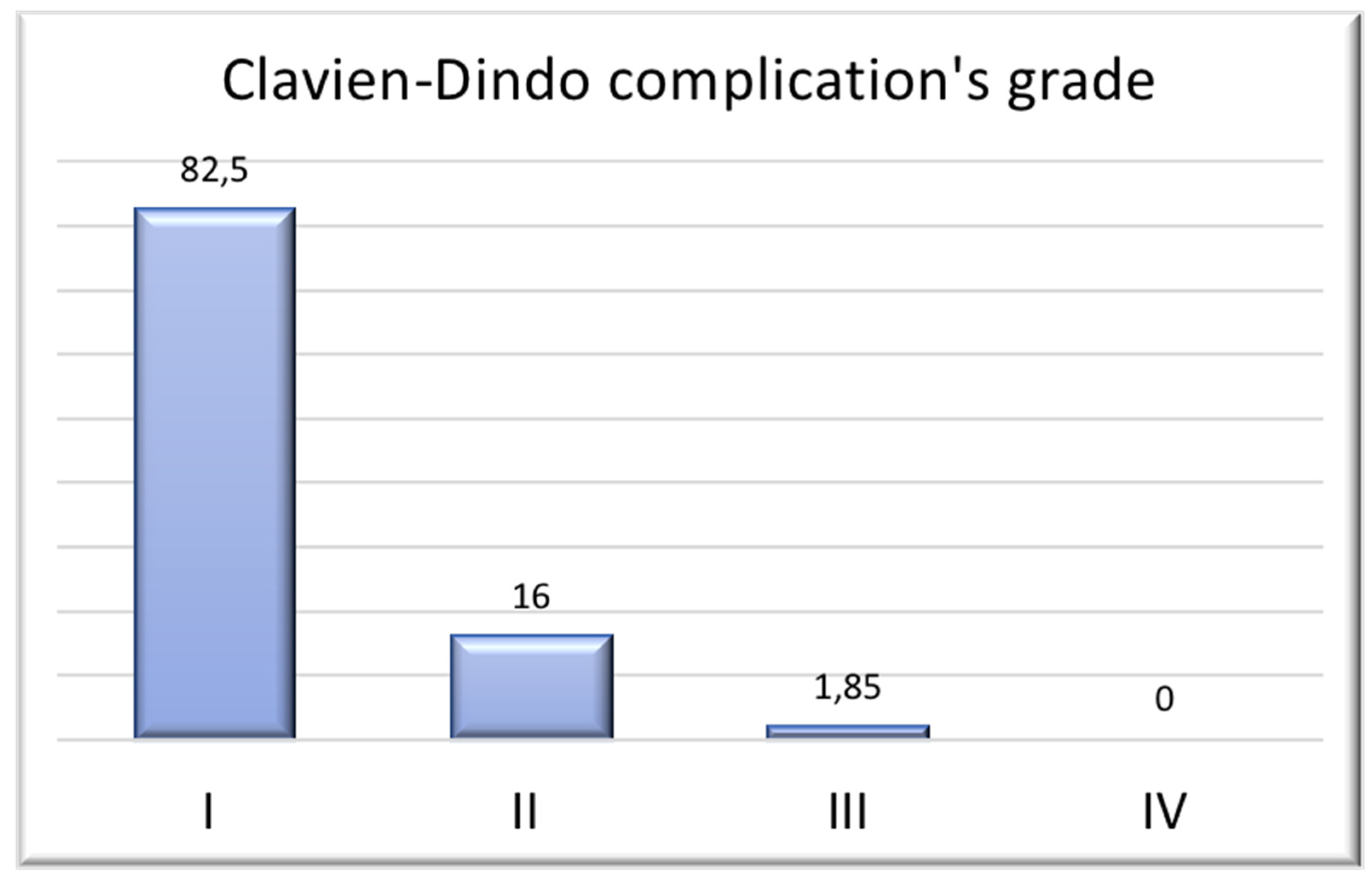
- Results from Clavien-Dindo classification’s complications:
Postoperative complications were carefully recorded. Local complications occurred in 30,4% of patients, including infections (12%) seromas requiring aspiration or delaying healing (4%) and wound dehiscence (10%). The mean healing time, defined as the interval from surgery to complete wound closure without the need for drainage or dressing, was 15 days. Notably, 87.4% of patients had their drainage removed by day 14, with a drainage output of less than 30 cc.
Surgical reintervention was required in only one patient. (
Table 4)
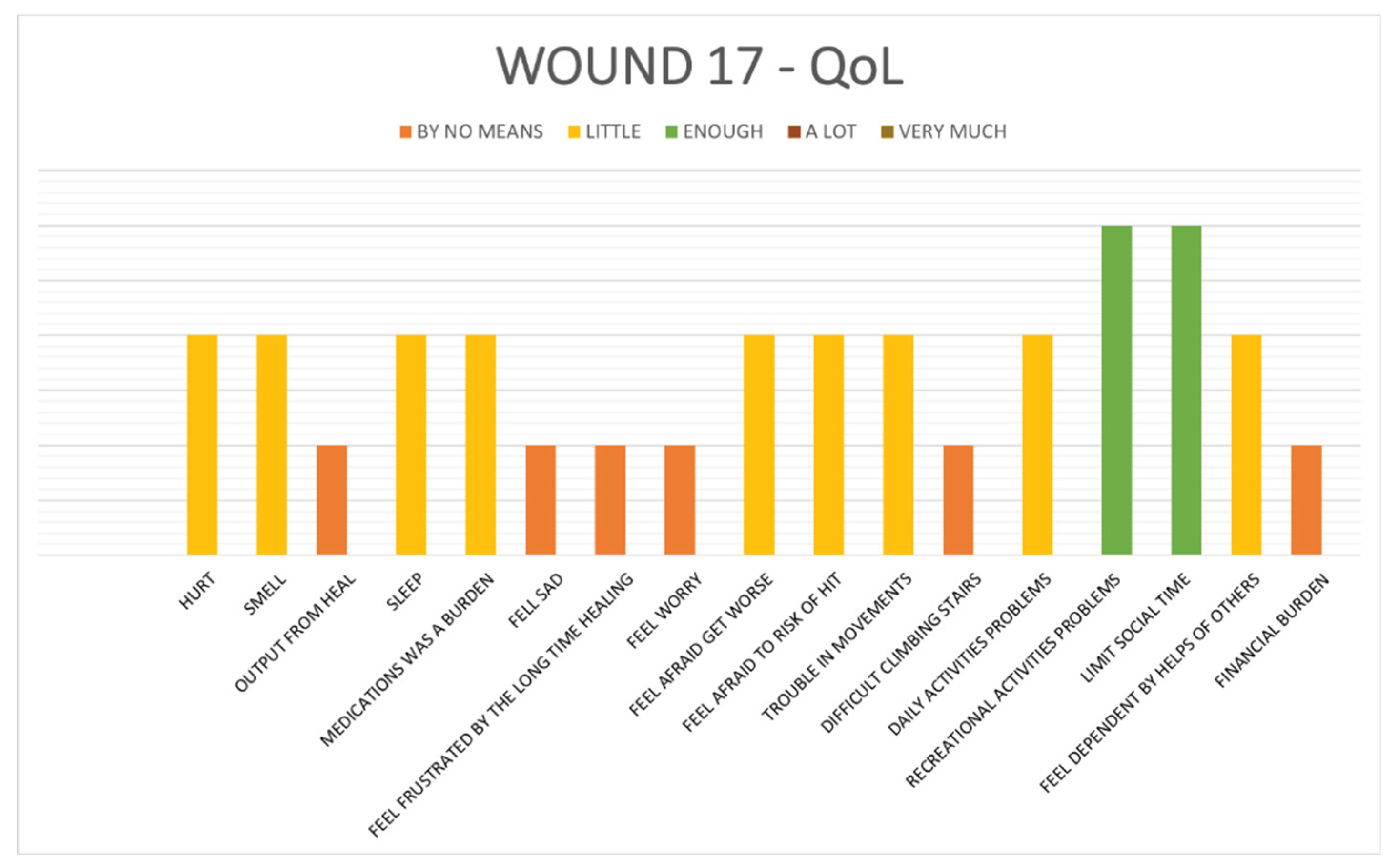
- Results on Wound-QoL17 questionnaire written by patients:
Patient-perceived quality of life related to wound burden was assessed through the
Wound-QoL17 questionnaire at postoperative days 7, 30, and 90. All patients completing the questionnaires (
Table 5).
Most patients selected responses within the “By no means” or “Little” categories for nearly all items, indicating that postoperative wound-related discomfort and limitations were minimal. This was particularly notable in areas related to pain, exudate, sleep disruption, emotional distress, mobility, and social interaction. The uniformity of these responses reflects a broadly favorable postoperative experience and suggests that the recovery process was well-tolerated across the patient population.
From a clinical and surgical perspective, early observations corroborated the subjective data. In the immediate postoperative phase, it was possible to appreciate the beneficial effects of Novox® Cup on the visual appearance of the mastectomy flaps. Surgeons noted a visibly improved appearance of the cutaneous tissue, with the flaps demonstrating optimal perfusion, minimal edema, and favorable color and texture within the first 48 hours after surgery. This early stabilization of the tissue is critical in procedures such as nipple-sparing mastectomy, where vascular integrity and skin viability are essential for both functional and aesthetic success. Furthermore, the use of the Novox® Cup dressing appears to have contributed to a reduction in localized complications, including ischemic changes and delayed healing of the nipple-areola complex and surrounding flaps. The dressing’s bioactive and antioxidant properties, combined with its pressure-distributing design, created a favorable environment for tissue regeneration and minimized mechanical stress on the surgical site. This facilitated not only a faster healing trajectory but also more predictable and uniform cosmetic outcomes.
The consistently high satisfaction scores observed in the patient-reported outcomes, paired with early and clearly observable clinical benefits, suggest a strong synergistic effect between the robotic surgical technique and the postoperative wound management protocol. These results underscore the importance of integrating advanced technologies not only in the intraoperative phase but also in the postoperative continuum of care.
In this cohort of patients undergoing nipple-sparing mastectomy with immediate breast reconstruction, the application of the Novox® Cup device was associated with favourable clinical outcomes and a positive impact on patient-reported wound-related quality of life. The device’s ease of use and tolerability contributed to a smooth postoperative recovery with minimal discomfort. Further studies are warranted to confirm these findings and to explore the potential of the Novox® Cup in reducing local complications and improving the overall postoperative experience.
4. Discussion
In this retrospective observational study, the use of the Novox® Cup device in the immediate postoperative period following nipple-sparing mastectomy (NSM) with immediate breast reconstruction proved to be safe, well-tolerated, and potentially effective in improving both clinical outcomes and patients perceived wound-related quality of life.
Novox® Cup is characterized by the combination of a gel-like oil matrix resulting from the oxygen-enrichment of natural oils, which is embedded in a polyester and polyurethane backing with a conformation that adapts in the most appropriate way to the treatment area (for example, as a breast cup or bra cup) [
9].
The mechanism of action of Novox® Cup, is based on a synergistic combination of ROS and Carboxylic Acid micro release and the oily nature of the matrix, which allow:
the facilitation of the healing process by creating a microenvironment favourable to the activation of the microcirculation and consequently of the natural reparative processes of cell proliferation
the reduction of microbial contamination thanks to the ability to create a local microenvironment unfavourable to the proliferation of pathogens.
the establishment of a protective barrier film with additional soothing action [
9].
Skin flap and NAC viability remains a critical determinant of surgical outcomes in breast surgery, particularly in breast procedures where both oncologic safety and aesthetic preservation are paramount. Early identification of vascular compromise and timely intervention are essential to minimize complications and avoid secondary surgical procedures.
In the present cohort, the incidence of total NAC necrosis (1.85%) was low and consistent with rates reported in the breast surgery literature. Importantly, partial necrosis cases were successfully managed conservatively, highlighting the effectiveness of structured postoperative surveillance and advanced wound care strategies in preventing progression to more severe tissue loss.
A particularly relevant finding of this study is the outcome observed in grade 2 flaps, representing reduced but still acceptable vitality. These cases are clinically significant, as they constitute a potentially reversible condition that may either resolve or deteriorate depending on postoperative management. The systematic application of Novox Cup dressings, combined with closer clinical monitoring, resulted in complete recovery in all grade 2 patients, with no evolution toward grade 3 or 4 impairment.
This suggests that Novox Cup dressings may play a meaningful role in supporting microcirculation, reducing edema, and promoting tissue recovery in compromised yet viable flaps. Their use appears particularly beneficial in the early postoperative phase, when vascular instability is most pronounced and therapeutic intervention can alter the natural course of flap suffering.
Conversely, the small percentage of
grade 3 cases underscores the importance of early recognition and aggressive management of moderate flap suffering. Prompt intervention in these patients is crucial to halt further ischemic damage and may explain the limited progression toward frank necrosis observed in this series. These findings are consistent with existing evidence regarding oxygen-enriched oil matrix-based dressings, which promote local microcirculation, reduce oxidative stress, and stimulate reparative cell proliferation [
10].
The adoption of a standardized vitality scale proved useful not only for postoperative assessment but also for guiding treatment decisions and stratifying follow-up intensity. This approach enhances reproducibility, facilitates communication within the multidisciplinary breast team, and allows for more objective comparison of outcomes across studies.
In conclusion, our findings support the value of early, standardized flap vitality assessment in breast surgery and suggest that targeted conservative interventions, including the use of Novox Cup dressings, may significantly improve outcomes in patients with reduced but salvageable flap vitality.
Novox® Cup dressing, with its bioactive formulation and ergonomic design, plays a dual role in enhancing clinical wound healing and improving the patient’s recovery experience. When applied according to a standardized protocol—involving initial intraoperative placement, followed by patient-managed replacement at home—the device has been associated with smoother, faster, and more comfortable recoveries. Most patients resumed daily activities with minimal disruption, indicating benefits not only in physical rehabilitation but also in psychological well-being. Clinically, its use was linked to a reduction in complications such as flap necrosis or NAC ischemia—known challenges in breast-conserving mastectomy techniques.
Furthermore, the device’s capacity to establish a stable, antioxidant-rich microenvironment promotes more effective healing dynamics. This was reflected in both objective and subjective outcomes: patients consistently reported higher comfort levels, reduced scarring with more pliable and aesthetically favourable tissue, and better preservation of the NAC’s pigmentation and contour. These aspects are central to overall satisfaction, emphasizing the importance of integrating advanced technologies not only intraoperatively but throughout the postoperative care continuum [
11].
Patient-reported outcomes, assessed using the validated
Wound-QoL17 questionnaire, confirmed minimal perceived burden in domains such as pain, exudate, sleep disruption, mobility, and emotional impact. The overall scores remained low throughout follow-up, suggesting that wound-related discomfort was limited. Statistical analysis demonstrated a significant correlation between higher Wound-QoL17 scores and the presence of postoperative complications, reinforcing the relevance of this tool in evaluating recovery trajectories. Importantly, only a small proportion of patients (1.6%) experienced delays of ≥2 weeks in initiating planned adjuvant therapies due to local wound complications. In our cohort, only one patient required a delay in adjuvant treatment, following the need to explant the prosthesis and replace it with a tissue expander due to a postoperative complication. This highlights the potential role of optimized postoperative management in maintaining oncologic timelines — a critical objective in the multidisciplinary treatment of breast cancer [
12].
In summary, the combined clinical and patient-reported findings suggest that the Novox® Cup dressing contributes positively to postoperative healing, reduces complications, and enhances patient comfort and satisfaction. These preliminary results warrant further validation through prospective controlled studies, but they provide a strong rationale for incorporating bioactive dressings into standardized postoperative protocols following NSM and immediate reconstruction.
5. Conclusions
These observations suggest that Novox® Cup extends well beyond passive wound coverage—it actively contributes to the preservation of mastectomy flap integrity and the viability of the nipple–areola complex (NAC), both of which are essential in prophylactic procedures where oncologic safety must be balanced with aesthetic and psychological restoration.
Preliminary clinical results support the integration of advanced postoperative wound care into the multidisciplinary treatment pathway. The targeted use of devices like Novox® Cup appears to enhance wound healing dynamics, reduce local complications, and improve the overall patient experience following nipple-sparing mastectomy. These findings underscore the importance of combining surgical innovation with effective postoperative strategies to achieve optimal functional and aesthetic outcomes.
Author Contributions
For Conception and design: F Pellini, G Deguidi; (II) Administrative support: All Authors; (III) Provision of study materials or patients: G Deguidi; (IV) Collection and assembly of data: F Pellini, G Deguidi; (V) Data analysis and interpretation: F Pellini, G Deguidi; (VI) Manuscript writing: G Deguidi; (VII) Final approval of manuscript: All authors.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of AOUI of Verona (protocol code 2.0; 31/01/2024).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request. Due to patient confidentiality and institutional policies, raw clinical data are not publicly accessible.
Acknowledgments
All authors thanks Moss SpA corporation for the assistance about Novox Cup.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| NSM |
Nipple Sparing Mastectomy |
| NAC |
nipple-areola complex |
| ADM |
acellular dermal matrices |
| ROS |
reactive oxygen species |
| QOL |
quality of life |
References
- Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [Internet]. 1 July 2025. Available online: https://acsjournals.onlinelibrary.wiley.com/doi/epdf/10.3322/caac.21492.
- De La Cruz, L; Moody, AM; Tappy, EE; Blankenship, SA; Hecht, EM. Overall Survival, Disease-Free Survival, Local Recurrence, and Nipple–Areolar Recurrence in the Setting of Nipple-Sparing Mastectomy: A Meta-Analysis and Systematic Review. Ann Surg Oncol 2015, 22(10), 3241–9. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, VJ; Toyserkani, NM. Prepectoral Breast Reconstruction without Acellular Dermal Matrix: Have We Come Full Circle? J Pers Med. 2022, 12(10), 1619. [Google Scholar] [CrossRef] [PubMed]
- Urban, C; González, E; Fornazari, A; Berman, G; Spautz, C; Kuroda, F; et al. Prepectoral Direct-to-Implant Breast Reconstruction without Placement of Acellular Dermal Matrix or Mesh after Nipple-Sparing Mastectomy. Plast Reconstr Surg 2022, 150(5), 973. [Google Scholar] [CrossRef] [PubMed]
- Vidya, R; Berna, G; Sbitany, H; Nahabedian, M; Becker, H; Reitsamer, R; et al. Prepectoral implant-based breast reconstruction: a joint consensus guide from UK, European and USA breast and plastic reconstructive surgeons. ecancermedicalscience 2019, 13, 927. [Google Scholar] [CrossRef] [PubMed]
- Jakubczyk, K; Dec, K; Kałduńska, J; Kawczuga, D; Kochman, J; Janda, K. Reactive oxygen species - sources, functions, oxidative damage. Pol Merkur Lek Organ Pol Tow Lek 2020, 48(284), 124–7. [Google Scholar]
- Dunnill, C; Patton, T; Brennan, J; Barrett, J; Dryden, M; Cooke, J; et al. Reactive oxygen species (ROS) and wound healing: the functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int Wound J 2015, 14(1), 89–96. [Google Scholar] [CrossRef] [PubMed]
- Valacchi, G; Fortino, V; Bocci, V. The dual action of ozone on the skin. Br J Dermatol 2005, 153(6), 1096–100. [Google Scholar] [CrossRef] [PubMed]
- Casella, D; Palumbo, P; Sandroni, S; Caponi, C; Littori, F; Capuano, F; et al. Positive ROS (Reactive Oxygen Species) Modulator Engineered Device Support Skin Treatment in Locally Advanced Breast Cancer (LABC) Enhancing Patient Quality of Life. J Clin Med. 2021, 11(1), 126. [Google Scholar] [CrossRef] [PubMed]
- Oxygen-enriched oleic matrix medical dressings for the treatment of extensive burns and related acute and post-burn complications in children: a case series – Wounds International [Internet]. 7 July 2025. Available online: https://woundsinternational.com/journal-articles/oxygen-enriched-oleic-matrix-medical-dressings-for-the-treatment-of-extensive-burns-and-related-acute-and-post-burn-complications-in-children-a-case-series/.
- Djohan, R; Gage, E; Gatherwright, J; Pavri, S; Firouz, J; Bernard, S; et al. Patient Satisfaction following Nipple-Sparing Mastectomy and Immediate Breast Reconstruction: An 8-Year Outcome Study [Outcomes Article]. Plast Reconstr Surg 2010, 125(3), 818. [Google Scholar] [CrossRef] [PubMed]
- Lorentzen, T; Heidemann, LN; Möller, S; Bille, C. Impact of neoadjuvant chemotherapy on surgical complications in breast cancer: A systematic review and meta-analysis. Eur J Surg Oncol 2022, 48(1), 44–52. [Google Scholar] [CrossRef] [PubMed]
Table 1.
Skin Flap Vitality Scale (1–4). Assess: color, temperature, turgidity/capillary refill, presence of suffering or necrosis.
Table 1.
Skin Flap Vitality Scale (1–4). Assess: color, temperature, turgidity/capillary refill, presence of suffering or necrosis.
| 1 — Optimal vitality |
|---|
| ∙ Color: Pink, Uniform |
| ∙ Temperature: warm and symmetrical with respect to the surrounding tissues |
| ∙ Capillary refill: < 2 seconds |
| ∙ Edema: minimal or absent |
| ∙ No signs of skin distress=> Perfused and stable flap
|
| |
| 2 — Reduced but Acceptable Vitality |
| ∙ Colour: slightly pale or hyperemic, but homogeneous |
| ∙ Temperature: slightly reduced or increased, but not critical |
| ∙ Capillary refill: 2–3 seconds |
| ∙ Edema: mild-moderate |
| ∙ No necrosis, any small areas of reversible distress=> Flap to be monitored more frequently, but vital |
| |
| 3 — Moderate Flap Suffering |
| ∙ Colour: marbled, cyanotic or greyish in patches |
| ∙ Temperature: clearly colder |
| ∙ Capillary refill: > 3 seconds or no |
| ∙ Edema: marked |
| ∙ Areas of dubious vitality=> High risk of negative evolution, requires timely intervention
|
| |
| 4 — Necrosis or Severe Impairment |
| ∙ Color: black, brown, dark purplish, or ischemic white |
| ∙ Temperature: cold |
| ∙ Capillary refill: absent |
| ∙ Loss of sensation, non-viable tissue |
∙ Frank necrosis (partial or total)
= > Compromised flap: surgical evaluation required |
Table 2.
CLAVIEN-DINDO classification.
Table 2.
CLAVIEN-DINDO classification.
| Grade I |
| Complications that do not require pharmacological, surgical, endoscopic or radiological interventions. |
| ∙ Treatment with conservative therapies (e.g., analgesic, antipyretics, physiotherapy). |
| ∙ It does not include antibiotics, medications, etc. |
| ∙ Example: post-operative fever treated with antipyretics. |
| Grade II |
| ∙ Complications that require specific pharmacological interventions. |
| ∙ It includes antibiotics, blood transfusions, and parenteral nutrition. |
| ∙ Example: Infection treated with antibiotics. |
| Grade III |
| Complications requiring surgical, endoscopic or radiological intervention. |
| ∙ IIIa: Surgery performed without general anesthesia. Example: Drainage of an infected abscess/seroma/subcutaneous hematoma. |
| ∙ IIIb: Surgery performed under general anesthesia. Example: re-surgery for hemorrhage. |
| Grade IV |
| Complications that endanger the patient’s life and require intensive management. |
Table 3.
The “Wound-QoL17” questionnaire is divided into 17 questions, as follows.
Table 3.
The “Wound-QoL17” questionnaire is divided into 17 questions, as follows.
| In the last seven days ... |
Question |
BY NO MEANS |
LITTLE |
ENOUGH |
A LOT |
VERY MUCH |
| 1 |
My wound hurt |
|
|
|
|
|
| 2 |
My wound smelled bad |
|
|
|
|
|
| 3 |
There was a leak of disturbing material from the wound |
|
|
|
|
|
| 4 |
The injury affected my sleep |
|
|
|
|
|
| 5 |
Wound treatment was a burden on me |
|
|
|
|
|
| 6 |
The injury made me miserable |
|
|
|
|
|
| 7 |
I felt frustrated because the wound is taking so long to heal |
|
|
|
|
|
| 8 |
I worried about my injury |
|
|
|
|
|
| 9 |
I was afraid that the wound would get worse or that new ones would appear. |
|
|
|
|
|
| 10 |
I was afraid to hit the wound |
|
|
|
|
|
| 11 |
I had trouble moving because of the injury |
|
|
|
|
|
| 12 |
Climbing the stairs was difficult due to the injury |
|
|
|
|
|
| 13 |
I had problems with daily activities due to the injury |
|
|
|
|
|
| 14 |
The injury limited my recreational activities |
|
|
|
|
|
| 15 |
The injury forced me to limit my activities with others |
|
|
|
|
|
| 16 |
I felt dependent on the help of others because of the injury |
|
|
|
|
|
| 17 |
The injury was a financial burden on me |
|
|
|
|
|
Table 4.
Results from Clavien-Dindo classification’s complications: most patients had no complications or had minor complications; 16% of patients had seromas requiring outpatient needle aspiration; only one patient had a complication requiring surgery (necrosis of the NAC with its subsequent surgical removal).
Table 4.
Results from Clavien-Dindo classification’s complications: most patients had no complications or had minor complications; 16% of patients had seromas requiring outpatient needle aspiration; only one patient had a complication requiring surgery (necrosis of the NAC with its subsequent surgical removal).
Table 5.
Results from “Wound 17 QoL”: most patients selected responses within the “By no means” or “Little” categories for nearly all items, indicating that postoperative wound-related discomfort and limitations were minimal.
Table 5.
Results from “Wound 17 QoL”: most patients selected responses within the “By no means” or “Little” categories for nearly all items, indicating that postoperative wound-related discomfort and limitations were minimal.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).