Submitted:
06 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
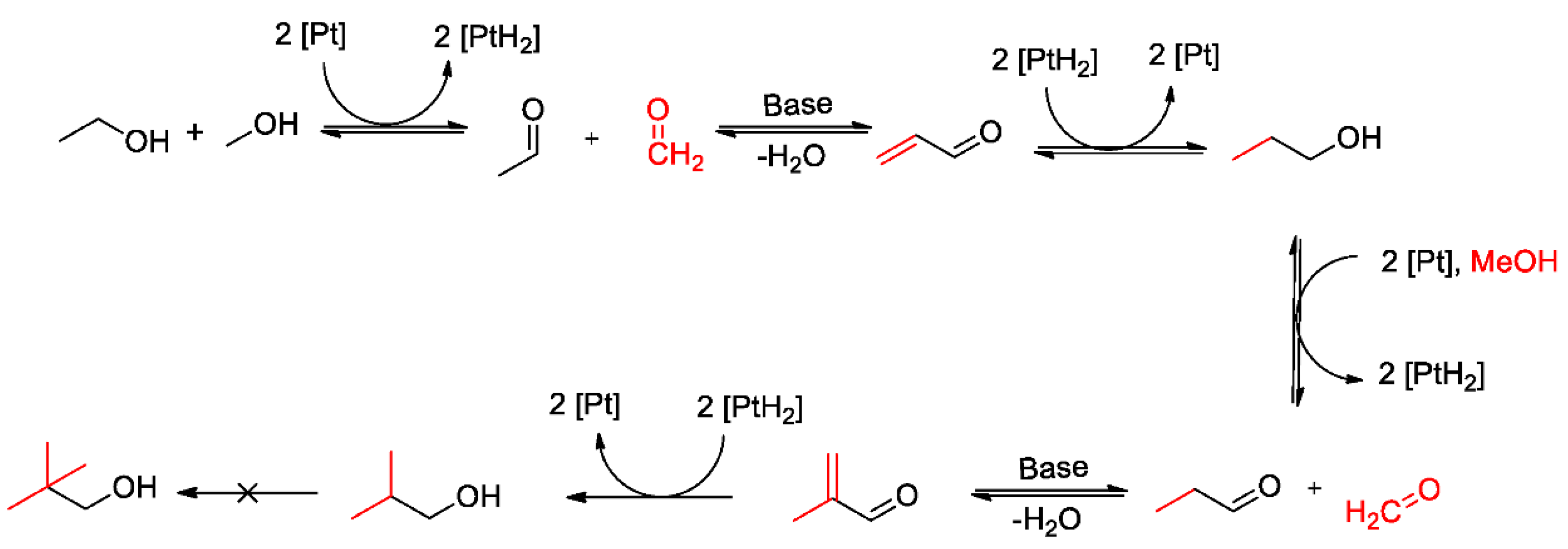
1. Introduction
- -
- to apply two different routes to the synthesis of Ni/Mg-Al mixed oxides catalysts. Many ways to synthesize hydrotalcites were already proposed in the past [28,29]. The methods chosen for this work are co-precipitation and urea hydrolysis precipitation due to their simplicity. The approach utilizing urea hydrolysis is intended to result in homogeneous distribution of the precipitation agent. This homogeneity should result in a narrower particle size distribution and enhanced crystallinity.
- -
- to investigate the influences of these syntheses’ routes on the structural properties and the catalytic activity of the synthesized catalysts for the above-presented Guerbet reaction, yielding iso-butanol.
2. Results and Discussion
2.1. Ni-Mg/Al Mixed Oxides Synthesized Via The Co-Precipitation Method
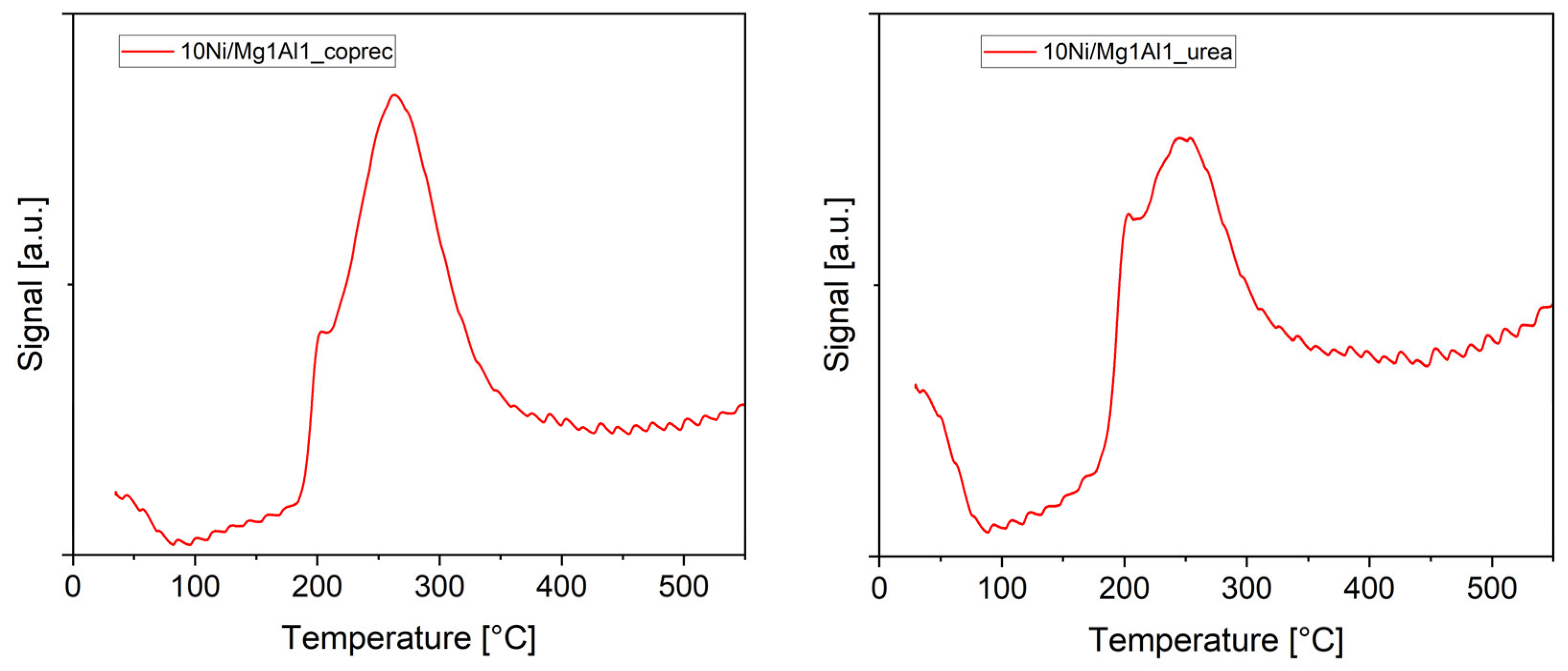
2.1.1. Temperature-Programmed Reduction (TPR)
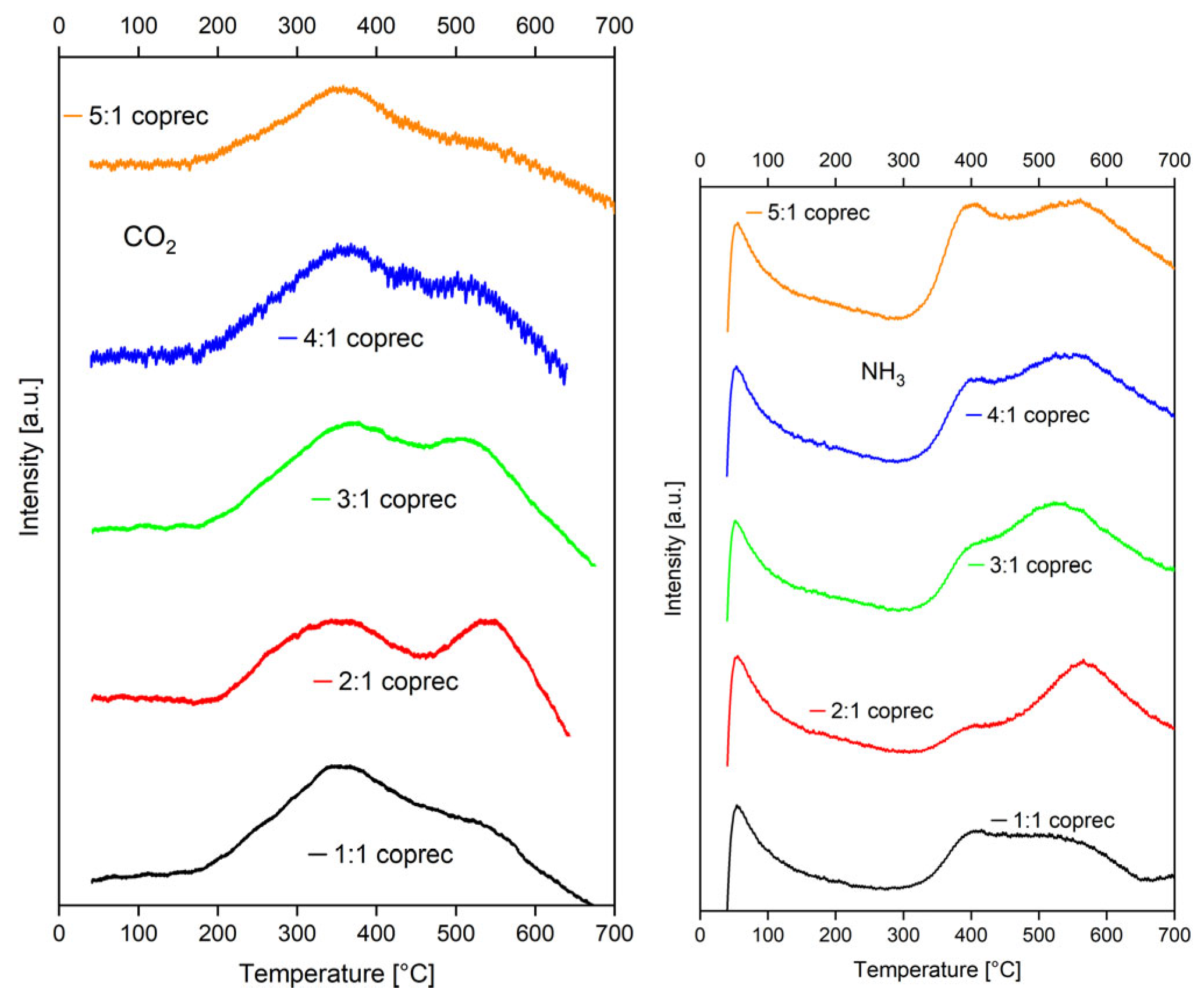
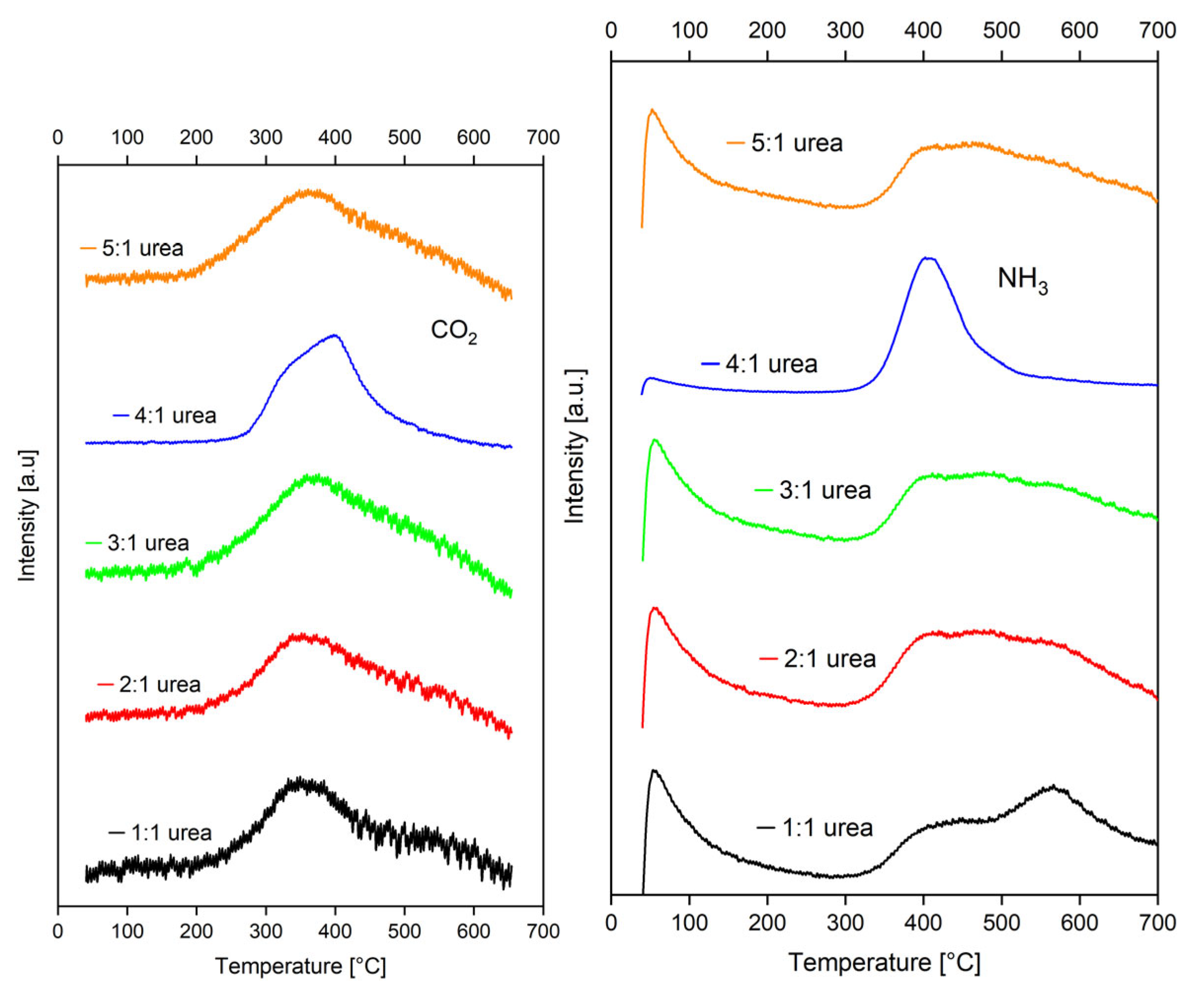
2.1.2. Temperature-Programmed Desorption (TPD) of CO2 and NH3
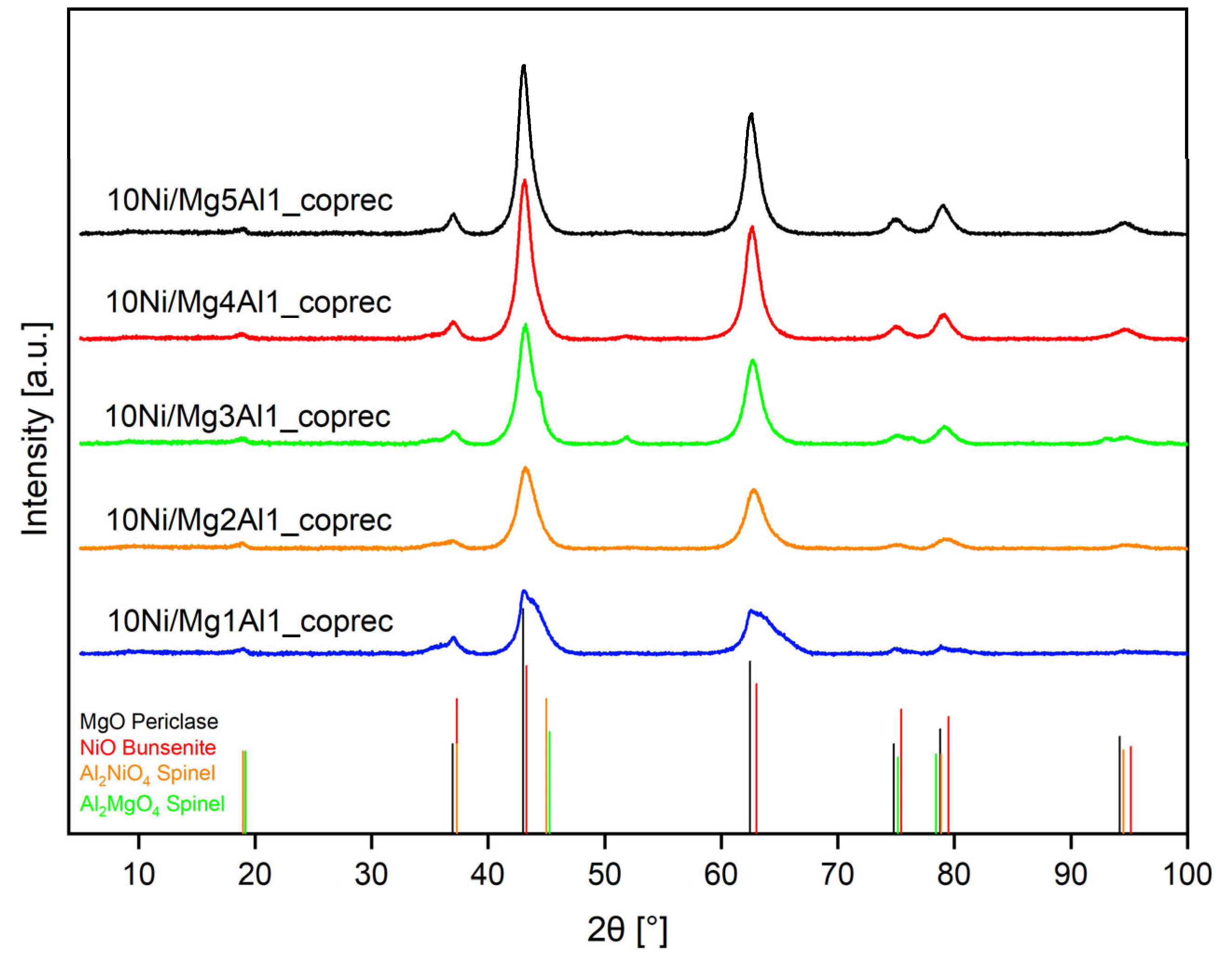
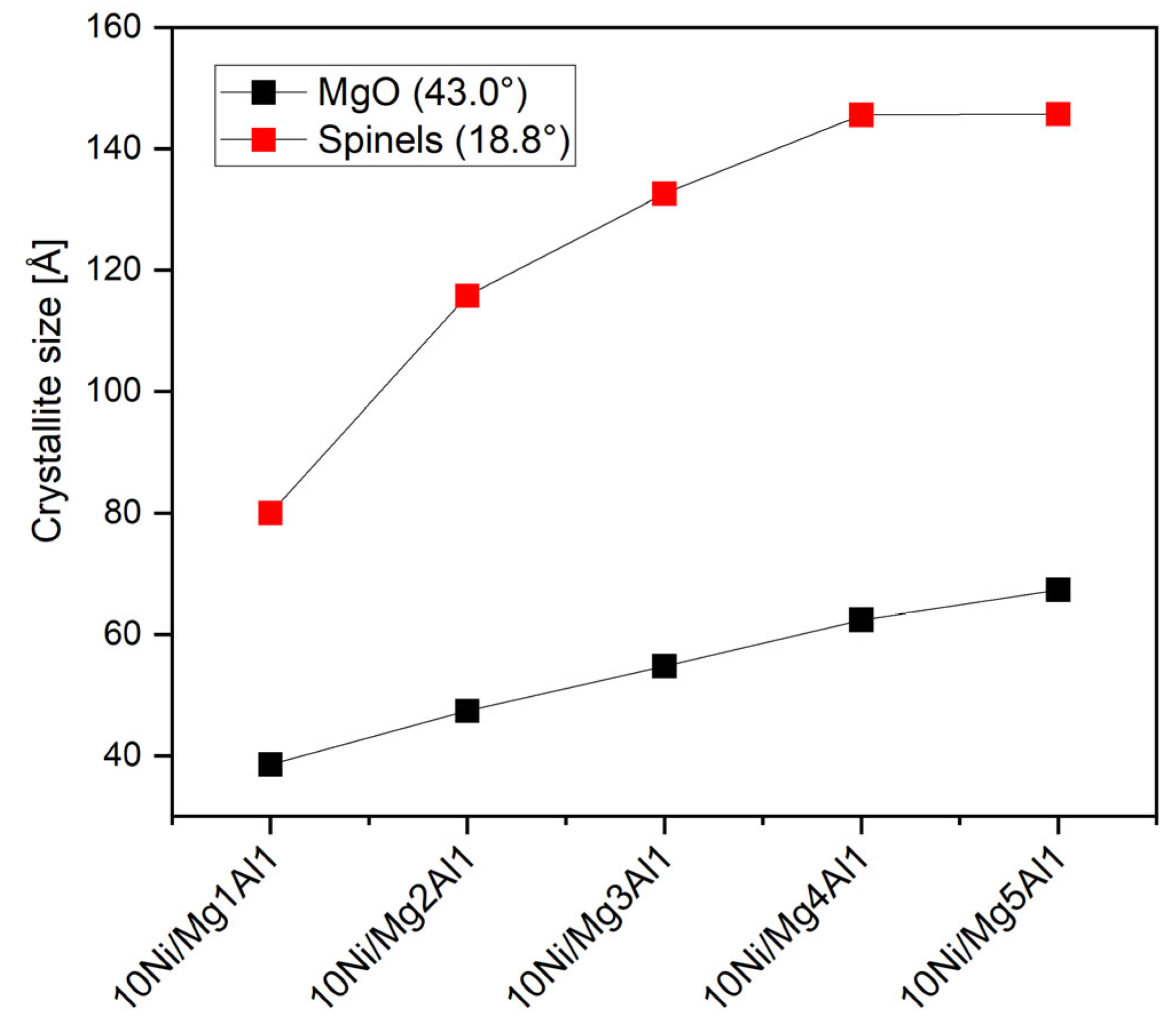
2.1.3. X-Ray Diffraction Analysis (XRD)
2.1.4. Inductively Coupled Plasma with Optical Emission Spectroscopy (ICP-OES)
2.1.5. Scanning Electron Microscopy (SEM)
2.1.6. N2 Sorption
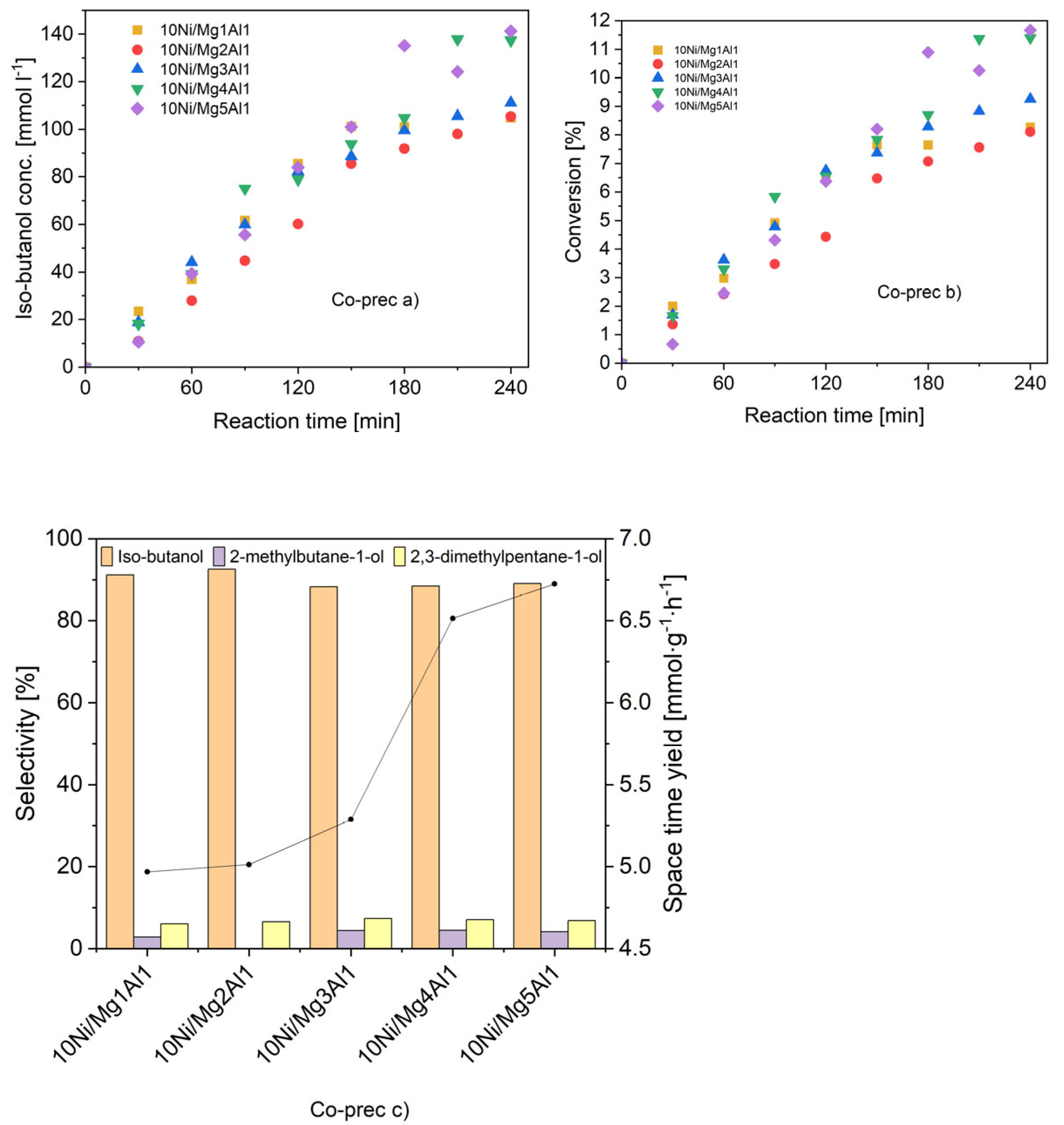
2.1.7. Catalytic Activity
- a)
- Concentration of iso-butanol
- b)
- Ethanol conversion
- c)
- Selectivities towards iso-butanol and other main by-products (left), space-time yield at 240 min reaction time
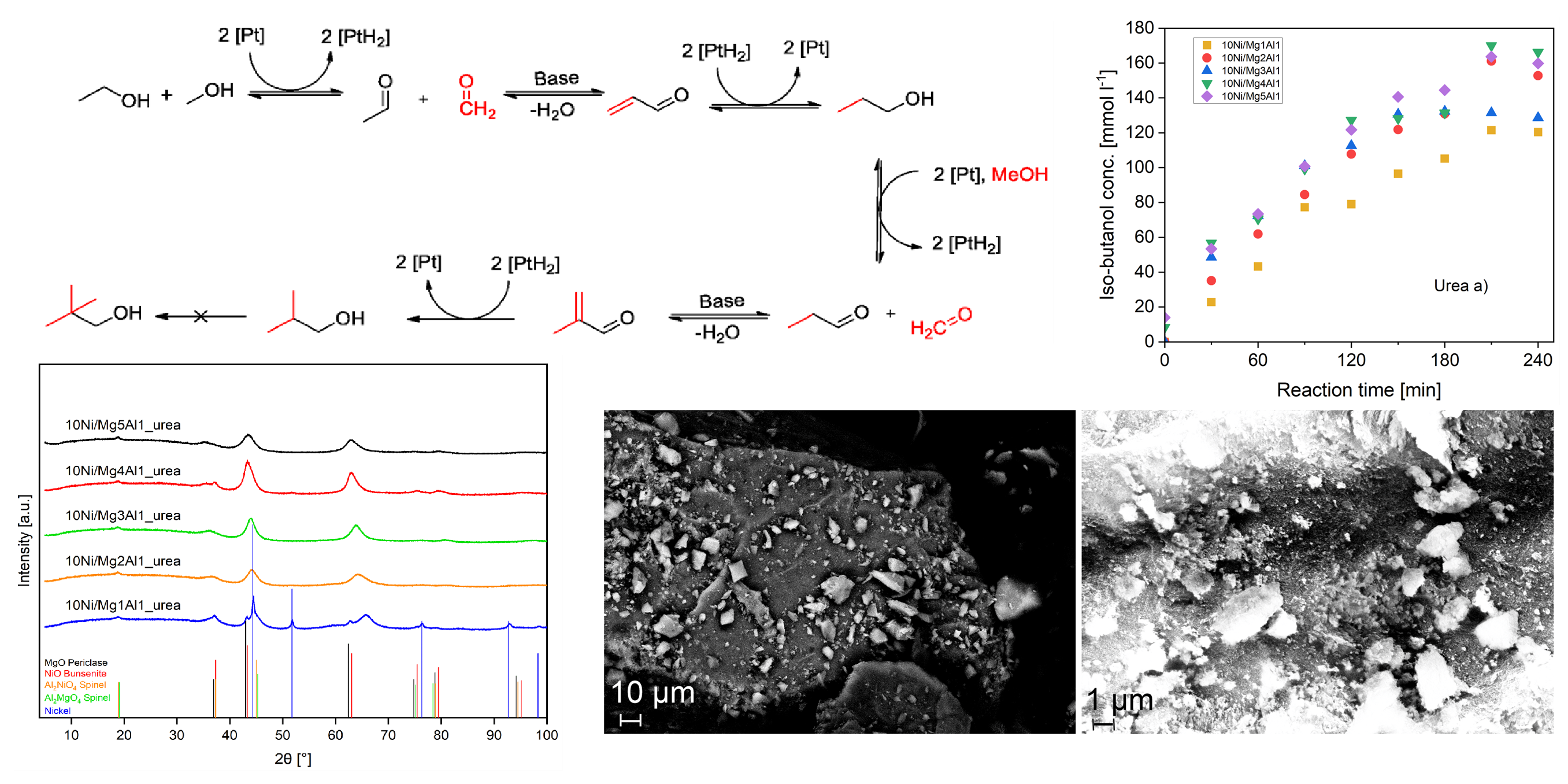
2.2. Ni-Mg/Al Mixed Oxides Synthesized Via the Urea Method
2.2.1. Temperature-Programmed Desorption (TPD) of CO2 and NH3
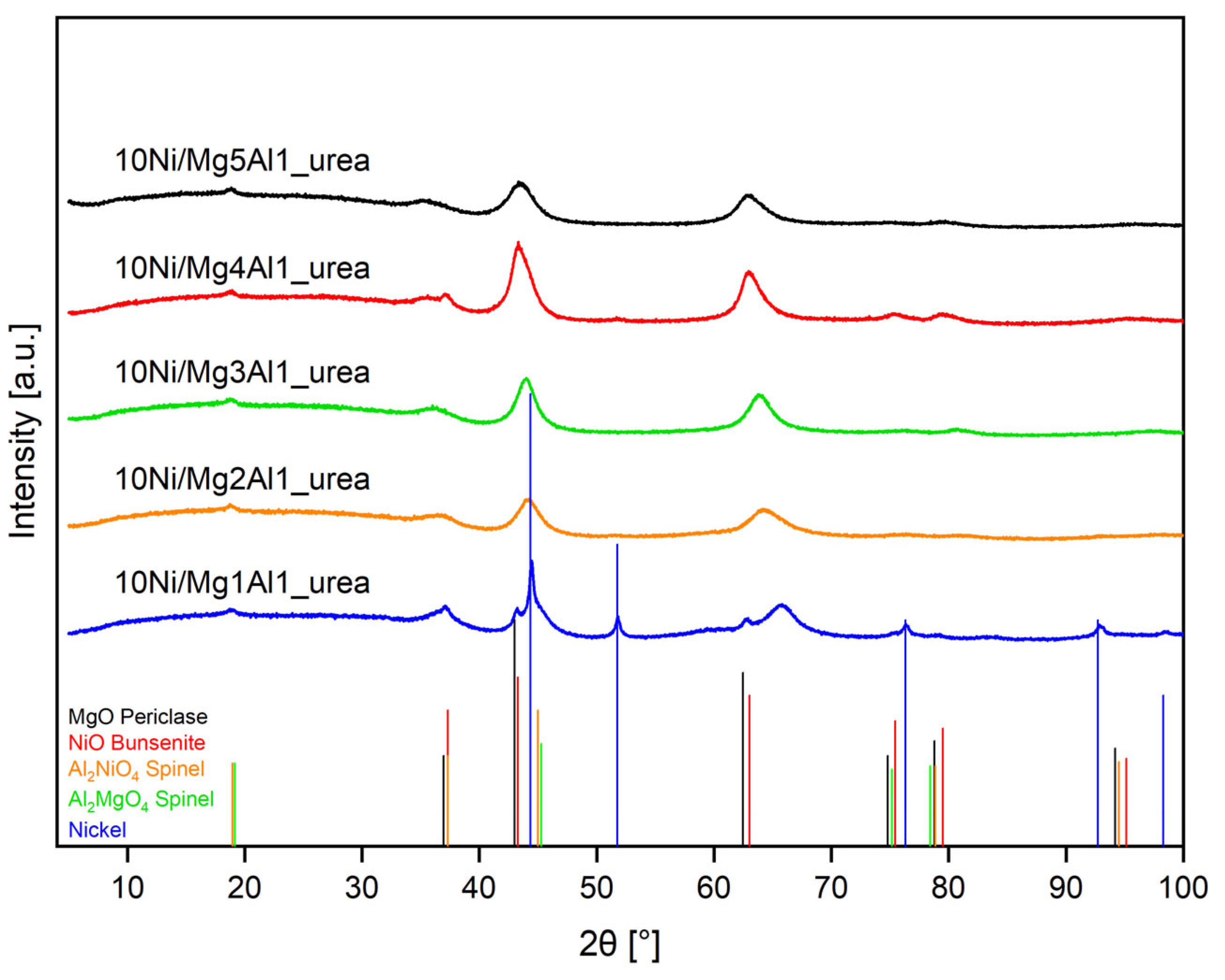
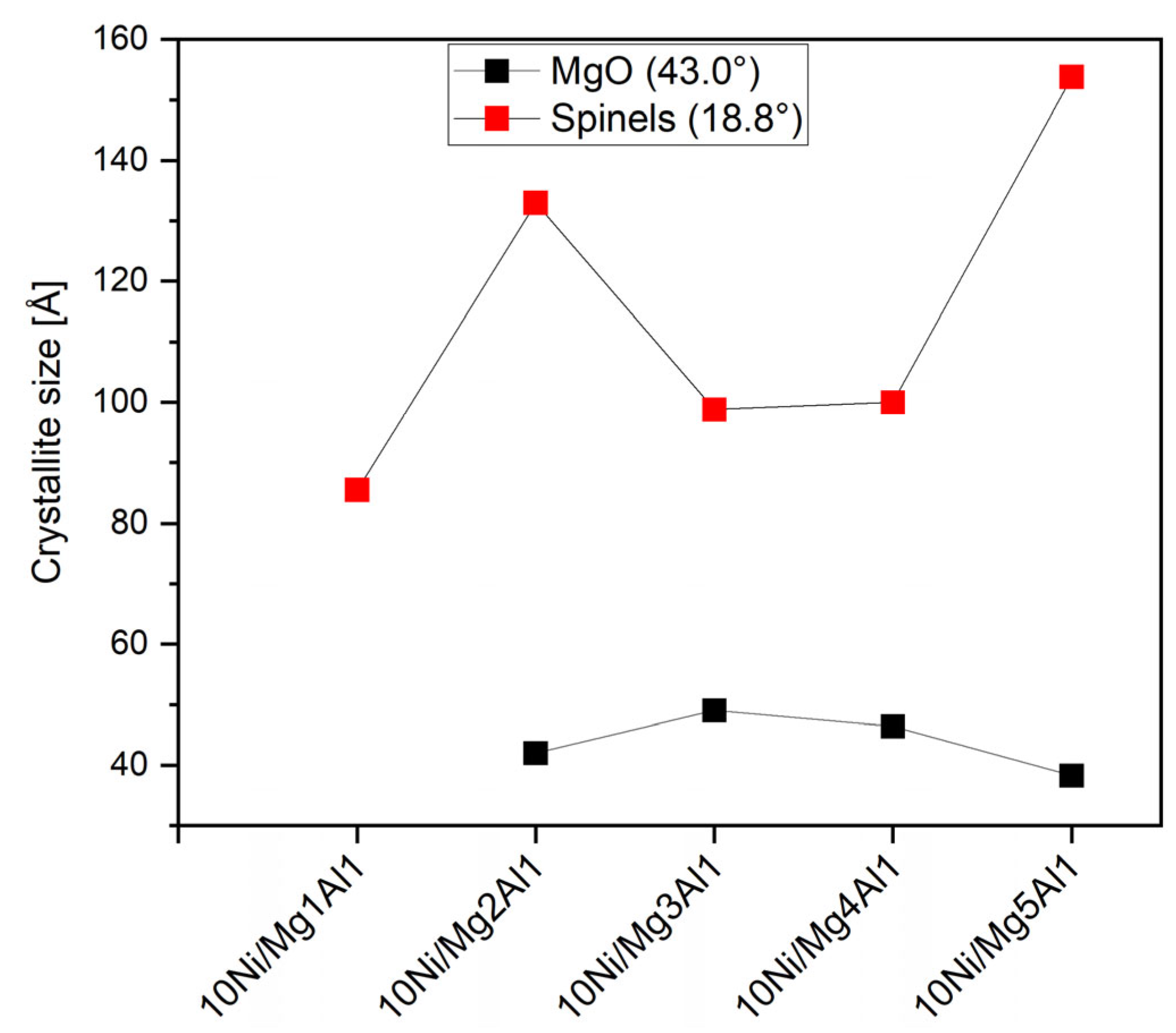
2.2.2. XRD Analysis
2.2.3. Inductively Coupled Plasma with Optical Emission Spectroscopy (ICP-OES)
2.2.4. Scanning Electron Microscopy (SEM)
2.2.5. N2 Sorption
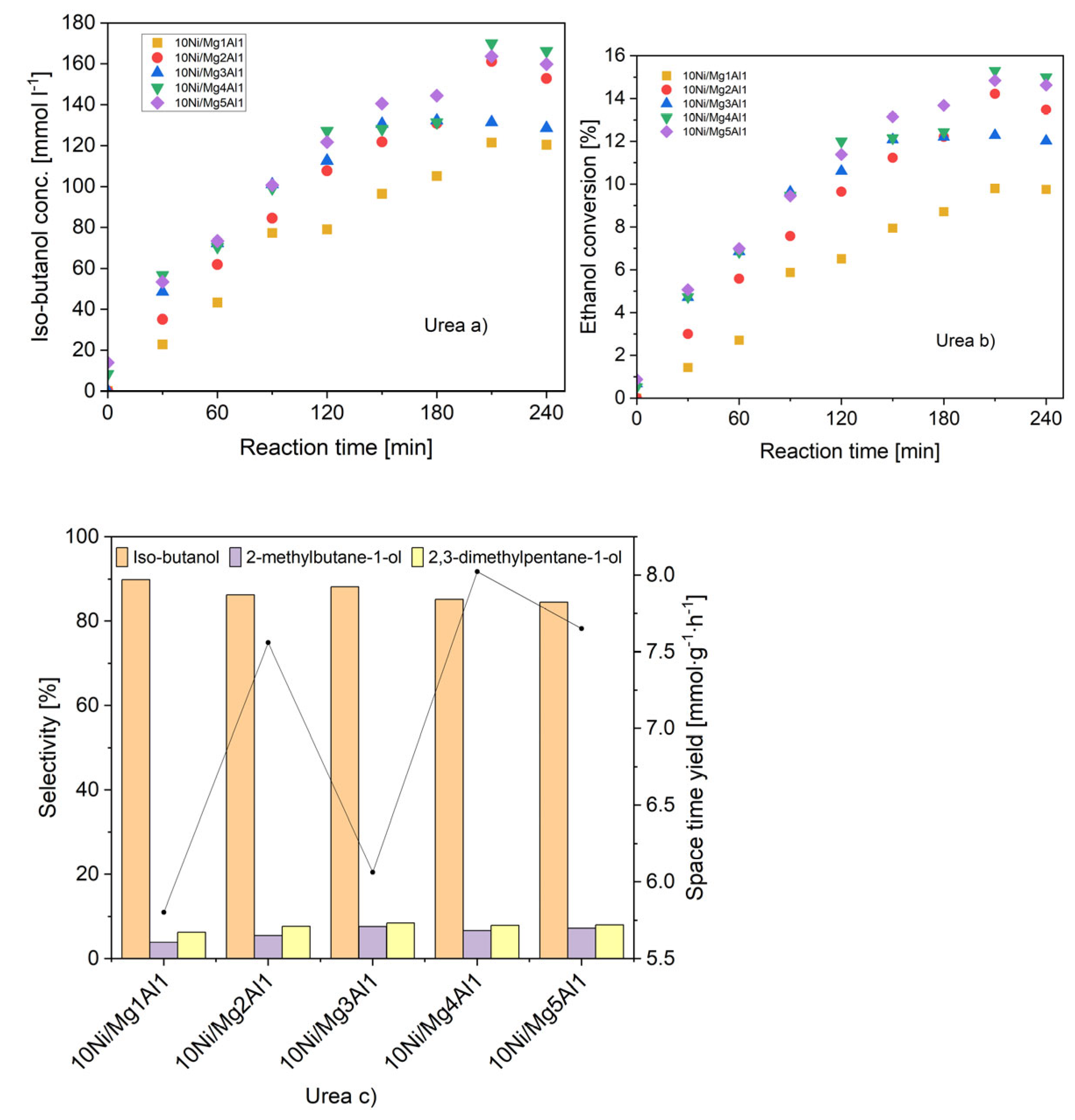
2.3.6. Catalytic Activity
- a)
- Concentration of iso-butanol
- b)
- Ethanol conversion
- c)
- Selectivities towards iso-butanol and other main by-products (left), space-time yield at 240 min reaction time
3. Materials and Methods
3.1. Synthesis of Hydrotalcite-Derived Mixed Oxide Supports
3.1.1. Synthesis Via Co-Precipitation
3.1.2. Synthesis Via Urea Hydrolysis
3.1.3. Wet Impregnation with Ni
3.2. Catalytic Experiments
3.2.1. Test of Catalytic Behavior
3.2.2. Analysis Via Gas-Chromatography And Mass Spectrometry (GC-MS)
3.3. Temperature-Programmed Methods
3.3.1 Temperature-Programmed Reduction (TPR)
3.3.2. Temperature-Programmed Desorption of CO2 (CO2-TPD)
3.3.3. Temperature.Programmed Desorption of NH3 (NH3-TPD)
3.4. Structural Analysis
3.4.1. X-Ray Diffraction (XRD)
3.4.2. Inductively Couple Plasma with Optical Emission Spectroscopy (ICP-OES)
3.4.3. Scanning Electron Microscopy (SEM)
3.4.4. N2 Sorption
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Q.; Xu, G.; Wang, X.; Mu, X. Selective upgrading of ethanol with methanol in water for the production of improved biofuel—isobutanol. Green Chemistry 2016, 18, 2811–2818. [Google Scholar] [CrossRef]
- Gabriëls, D.; Hernández, W.Y.; Sels, B.; Van Der Voort, P.; Verberckmoes, A. Review of catalytic systems and thermodynamics for the Guerbet condensation reaction and challenges for biomass valorization. Catalysis Science & Technology 2015, 5, 3876–3902. [Google Scholar]
- Li, X.; Li, X. Continuous upgrading of methanol and ethanol to isobutanol by heterogeneous catalysis over Cu-CeO2/AC catalyst and the combination. Molecular Catalysis 2024, 569, 114641. [Google Scholar] [CrossRef]
- Peters, M.; Menne, A.; Gielisch, H. Synthesis of Sustainable Fuels and Intermediates from Ethanol and Methanol. Chemie Ingenieur Technik 2022, 94, 1501–1508. [Google Scholar] [CrossRef]
- Wingad, R.L.; Bergström, E.J.E.; Everett, M.; Pellow, K.J.; Wass, D.F. Catalytic conversion of methanol/ethanol to isobutanol – a highly selective route to an advanced biofuel. Chemical Communications 2016, 52, 5202–5204. [Google Scholar] [CrossRef] [PubMed]
- Yusoff, M.N.A.M.; Zulkifli, N.W.M.; Masjuki, H.H.; Harith, M.H.; Syahir, A.Z.; Khuong, L.S.; Zaharin, M.S.M.; Alabdulkarem, A. Comparative assessment of ethanol and isobutanol addition in gasoline on engine performance and exhaust emissions. Journal of Cleaner Production 2018, 190, 483–495. [Google Scholar] [CrossRef]
- Pasel, J.; Häusler, J.; Schmitt, D.; Valencia, H.; Meledina, M.; Mayer, J.; Peters, R. Ethanol Dehydrogenation: A Reaction Path Study by Means of Temporal Analysis of Products. Catalysts 2020, 10, 1151. [Google Scholar] [CrossRef]
- Pizzoferrato, R.; Richetta, M. Layered Double Hydroxides (LDHs); Crystals, 2020. [Google Scholar]
- Trujillano, R.; Labajos, F.M.; Rives, V. Hydrotalcites, a rapid survey on the very recent synthesis and applications procedures. 2023, 238, 106927. [Google Scholar] [CrossRef]
- Bellotto, M.; Rebours, B.; Clause, O.; Lynch, J.; Bazin, D.; Elkaïm, E. Hydrotalcite Decomposition Mechanism: A Clue to the Structure and Reactivity of Spinel-like Mixed Oxides. The Journal of Physical Chemistry 1996, 100, 8535–8542. [Google Scholar] [CrossRef]
- Li, J.; Lin, L.; Tan, Y.; Wang, S.; Yang, W.; Chen, X.; Luo, W.; Ding, Y.-J. High Performing and Stable Cu/NiAlOx Catalysts for the Continuous Catalytic Conversion of Ethanol into Butanol. 2022, 14, e202200539. [Google Scholar] [CrossRef]
- Zhu, M.; Wang, Y.; Du, M.; Zhang, X.; Liu, H.; Qiao, Z.; Yang, J.; Peng, B.; Nie, L.; Li, Z. Cu-incorporated MgAl LDH Catalyst for Enhanced Bio-ethanol Upgrading to Butanol, n/a e01437. [CrossRef]
- Bravo-Suárez, J.J.; Subramaniam, B.; Chaudhari, R.V. Vapor-phase methanol and ethanol coupling reactions on CuMgAl mixed metal oxides. Applied Catalysis A: General 2013, 455, 234–246. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, P.; Wang, J.; Tan, T. Guerbet Reactions for Biofuel Production from ABE Fermentation Using Bifunctional Ni-MgO-Al2O3 Catalysts. Catalysts 2021, 414. [Google Scholar] [CrossRef]
- Cheng, F.-L.; Guo, H.-Q.; Cui, J.-L.; Hou, B.; Li, D.-B. Guerbet reaction of methanol and ethanol catalyzed by CuMgAlOx mixed oxides: Effect of M2+/Al3+ ratio. Journal of Fuel Chemistry and Technology 2018, 46, 1472–1481. [Google Scholar] [CrossRef]
- Cheng, F.-L.; Guo, H.; Cui, J.; Hou, B.; Xi, H.; Jia, L.; Li, D. Coupling of methanol and ethanol over CuMgAlOx catalysts: the roles of copper species and alkalinity. Reaction Kinetics, Mechanisms and Catalysis 2019, 126, 119–136. [Google Scholar] [CrossRef]
- Carlini, C.; Flego, C.; Marchionna, M.; Noviello, M.; Galletti, A.M.R.; Sbrana, G.; Basile, F.; Vaccari, A. Guerbet condensation of methanol with n-propanol to isobutyl alcohol over heterogeneous copper chromite/Mg–Al mixed oxides catalysts. Journal of Molecular Catalysis A: Chemical 2004, 220, 215–220. [Google Scholar] [CrossRef]
- Carlini, C.; Marchionna, M.; Noviello, M.; Galletti, A.M.R.; Sbrana, G.; Basile, F.; Vaccari, A. Guerbet condensation of methanol with n-propanol to isobutyl alcohol over heterogeneous bifunctional catalysts based on Mg–Al mixed oxides partially substituted by different metal components. Journal of Molecular Catalysis A: Chemical 2005, 232, 13–20. [Google Scholar] [CrossRef]
- Larina, O.V.; Valihura, K.V.; Kyriienko, P.I.; Vlasenko, N.V.; Balakin, D.Y.; Khalakhan, I.; Čendak, T.; Soloviev, S.O.; Orlyk, S.M. Successive vapour phase Guerbet condensation of ethanol and 1-butanol over Mg-Al oxide catalysts in a flow reactor. Applied Catalysis A: General 2019, 588, 117265. [Google Scholar] [CrossRef]
- Tang, X.; Song, C.; Li, H.; Liu, W.; Hu, X.; Chen, Q.; Lu, H.; Yao, S.; Li, X.-N.; Lin, L. Thermally stable Ni foam-supported inverse CeAlOx/Ni ensemble as an active structured catalyst for CO2 hydrogenation to methane. Nature Communications 2024, 15, 3115. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Y.; Sun, C.; Wang, Y.; Song, G.; Li, C.; Lim, K.H.; Ye, R.; Peng, Y.; Arandiyan, H.; Lu, Z.-H.; Feng, G.; Zhang, R.; Kawi, S. Lanthanum-mediated enhancement of nickel nanoparticles for efficient CO2 methanation. Fuel 2024, 371, 131998. [Google Scholar] [CrossRef]
- Häusler, J.; Pasel, J.; Wöllhaf, C.; Peters, R.; Stolten, D. Dilute Alloy Catalysts for the Synthesis of Isobutanol via the Guerbet Route: A Comprehensive Study. Catalysts 2024, 14, 215. [Google Scholar] [CrossRef]
- Häusler, J.; Pasel, J.; Woltmann, F.; Everwand, A.; Meledina, M.; Valencia, H.; Lipińska-Chwałek, M.; Mayer, J.; Peters, R. Elucidating the Influence of the d-Band Center on the Synthesis of Isobutanol. Catalysts 2021, 11. [Google Scholar] [CrossRef]
- Pasel, J.; Häusler, J.; Peters, R.; Stolten, D. Catalytic activity and stability of NiPt/C catalysts for the synthesis of iso-butanol from methanol/ethanol mixtures. Catalysis Science & Technology 2024. [Google Scholar]
- Pasel, J.; Häusler, J.; Peters, R.; Stolten, D. NiPt catalysts for the synthesis of iso-butanol: the influence of molar ratio and total metal loading on activity and stability. Catalysis Science & Technology 2025, 15, 2248–2260. [Google Scholar]
- Pasel, J.; Häusler, J.; Schmitt, D.; Valencia, H.; Mayer, J.; Peters, R. Aldol condensation of acetaldehyde for butanol synthesis: A temporal analysis of products study. Applied Catalysis B: Environmental 2023, 324, 122286. [Google Scholar] [CrossRef]
- Pasel, J.; Woltmann, F.; Häusler, J.; Peters, R. Surface Redox Reaction for the Synthesis of NiPt Catalysts for the Upgrading of Renewable Ethanol/Methanol Mixtures. Catalysts 2024, 14, 77. [Google Scholar] [CrossRef]
- Othman, M.; Helwani, Z.; Martunus; Fernando, W. Synthetic hydrotalcites from different routes and their application as catalysts and gas adsorbents: A review. Applied Organometallic Chemistry 2009, 23, 335–346. [Google Scholar] [CrossRef]
- Costantino, U.; Marmottini, F.; Nocchetti, M.; Vivani, R. New Synthetic Routes to Hydrotalcite-Like Compounds − Characterisation and Properties of the Obtained Materials. European Journal of Inorganic Chemistry 1998, 1998, 1439–1446. [Google Scholar] [CrossRef]
- Choudhary, V.R.; Rane, V.H. Acidity/basicity of rare-earth oxides and their catalytic activity in oxidative coupling of methane to C2-hydrocarbons. Journal of Catalysis 1991, 130, 411–422. [Google Scholar] [CrossRef]
- Bartholomew, C.H.; Farrauto, R.J. Catalyst Characterization and Selection. Fundamentals of Industrial Catalytic Processes 2005, 118–196. [Google Scholar]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. Journal of the American Chemical Society 1938, 60, 309–319. [Google Scholar] [CrossRef]


| Catalysts | Strong basic [%] |
Very strong basic [%] |
Weak acidic [%] |
Medium acidic [%] |
Strong acidic [%] |
|---|---|---|---|---|---|
| 10Ni/Mg1Al1_coprec | 83.3 | 16.7 | 28.5 | 19.0 | 52.5 |
| 10Ni/Mg2Al1_coprec | 65.8 | 34.2 | 32.5 | 4.8 | 62.7 |
| 10Ni/Mg3Al1_coprec | 72.3 | 27.7 | 28.2 | 1.7 | 70.1 |
| 10Ni/Mg4Al1_coprec | 79.0 | 21.0 | 26.9 | 6.7 | 66.4 |
| 10Ni/Mg5Al1_coprec | 100.0 | 0.0 | 33.1 | 7.9 | 58.9 |
| Catalysts | Ni mass fraction [wt%] ICP-OES calculated |
Molar ratios of Mg/Al [-] ICP-OES calculated |
||
|---|---|---|---|---|
| 10Ni/Mg1Al1_coprec | 9.20 ± 0.30 | 11.26 | 0.95 : 1 | 0.98 : 1 |
| 10Ni/Mg2Al1_coprec | 8.66 ± 0.11 | 11.15 | 1.97 : 1 | 2.00 : 1 |
| 10Ni/Mg3Al1_coprec | 8.85 ± 0.07 | 11.17 | 2.64 : 1 | 3.01 : 1 |
| 10Ni/Mg4Al1_coprec | 9.19 ± 0.05 | 11.48 | 3.03 : 1 | 4.00 : 1 |
| 10Ni/Mg5Al1_coprec | 9.76 ± 0.08 | 11.14 | 3.29 : 1 | 5.00 : 1 |
| Catalysts | Surface area [m2 g-1] |
Pore volume [cm3 g-1] |
Average pore size [Å] |
|---|---|---|---|
| 10Ni/Mg1Al1_coprec | 253.64 | 0.73 | 57.71 |
| 10Ni/Mg2Al1_coprec | 114.59 | 0.30 | 52.66 |
| 10Ni/Mg3Al1_coprec | 128.17 | 0.34 | 53.18 |
| 10Ni/Mg4Al1_coprec | 227.43 | 0.61 | 53.21 |
| 10Ni/Mg5Al1_coprec | 208.34 | 0.47 | 44.65 |
| Catalysts | Strong basic [%] |
Very strong basic [%] |
Weak acidic [%] |
Medium acidic [%] |
Strong acidic [%] |
|---|---|---|---|---|---|
| 10Ni/Mg1Al1_urea | 76.6 | 23.4 | 27.6 | 12.5 | 60.0 |
| 10Ni/Mg2Al1_urea | 100.00 | 0 | 27.8 | 18.6 | 53.5 |
| 10Ni/Mg3Al1_urea | 100.00 | 0 | 29.7 | 19.0 | 51.3 |
| 10Ni/Mg4Al1_urea | 100.00 | 0 | 1.7 | 98.3 | 0.0 |
| 10Ni/Mg5Al1_urea | 100.00 | 0 | 33.0 | 21.5 | 45.5 |
| Catalysts | Ni mass fraction [wt%] ICP-OES calculated |
Mg/Al molar ratios [-] ICP-OES calculated |
||
|---|---|---|---|---|
| 10Ni/Mg1Al1_urea | 8.96 ± 0.07 | 11.06 | 0.19 : 1 | 1.00 : 1 |
| 10Ni/Mg2Al1_urea | 8.60 ± 0.11 | 11.00 | 0.33 : 1 | 2.01 : 1 |
| 10Ni/Mg3Al1_urea | 10.24 ± 0.16 | 11.35 | 0.68 : 1 | 2.99 : 1 |
| 10Ni/Mg4Al1_urea | 14.48 ± 0.04 | 10.95 | 0.84 : 1 | 4.02 : 1 |
| 10Ni/Mg5Al1_urea | 10.40 ± 0.30 | 10.96 | 1.06 : 1 | 5.02 : 1 |
| Catalysts | Surface area [m2 g-1] |
Pore volume [cm3 g-1] |
Average pore size [Å] |
|---|---|---|---|
| 10Ni/Mg1Al1_urea | 186.5 | 0.42 | 45.3 |
| 10Ni/Mg2Al1_urea | 167.9 | 0.29 | 34.8 |
| 10Ni/Mg3Al1_urea | 245.0 | 0.31 | 25.1 |
| 10Ni/Mg4Al1_urea | 288.2 | 0.26 | 17.9 |
| 10Ni/Mg5Al1_urea | 210.7 | 0.24 | 22.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




