Submitted:
07 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
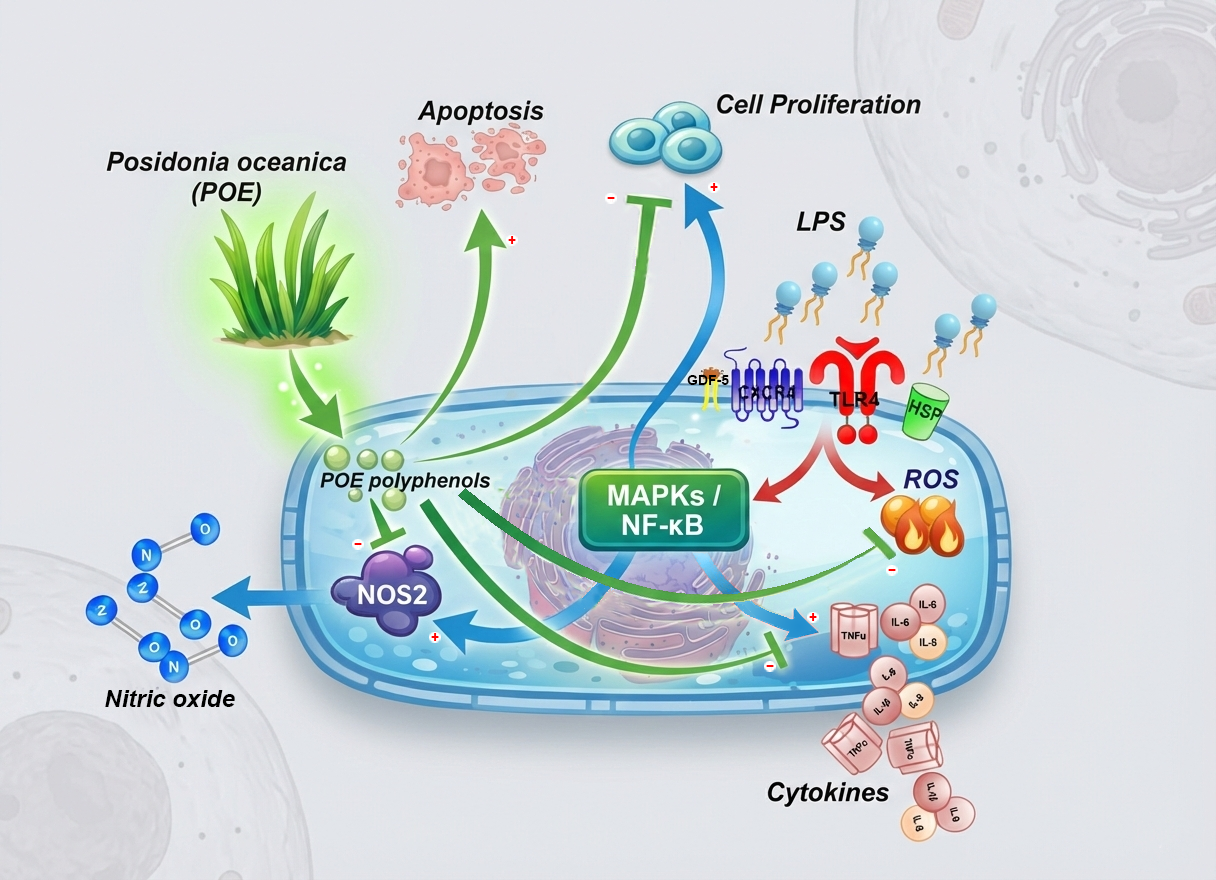
Keywords:
1. Introduction
2. Results
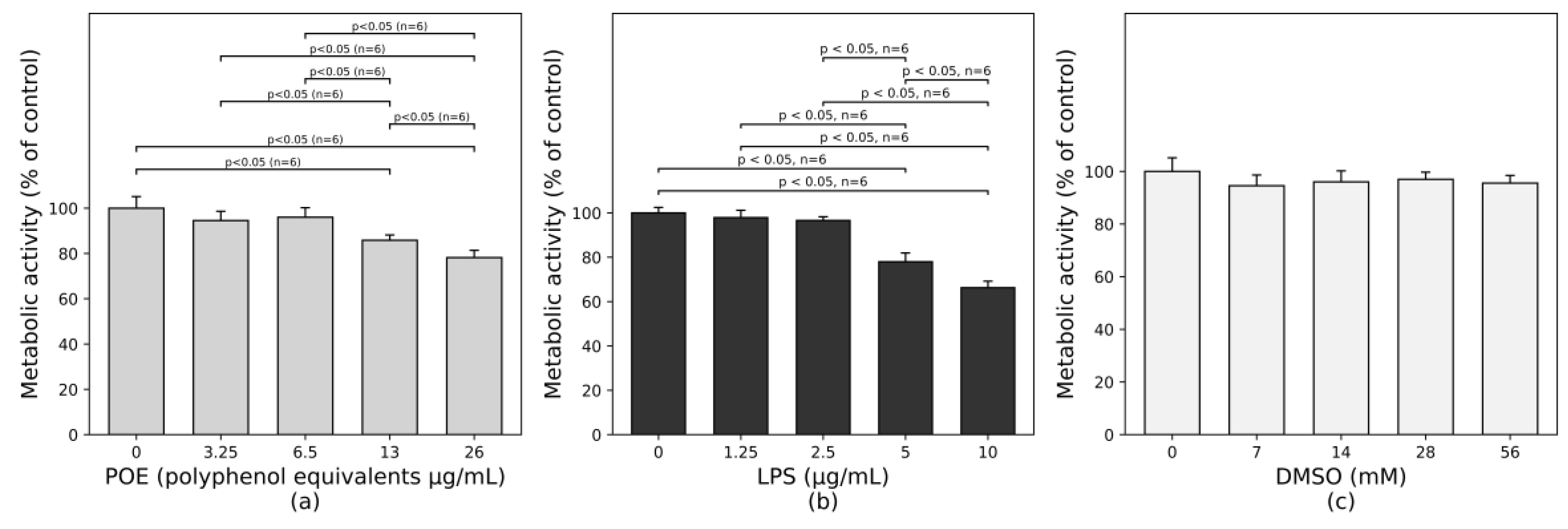
2.1. Determination of Safe Working Concentrations of POE and LPS
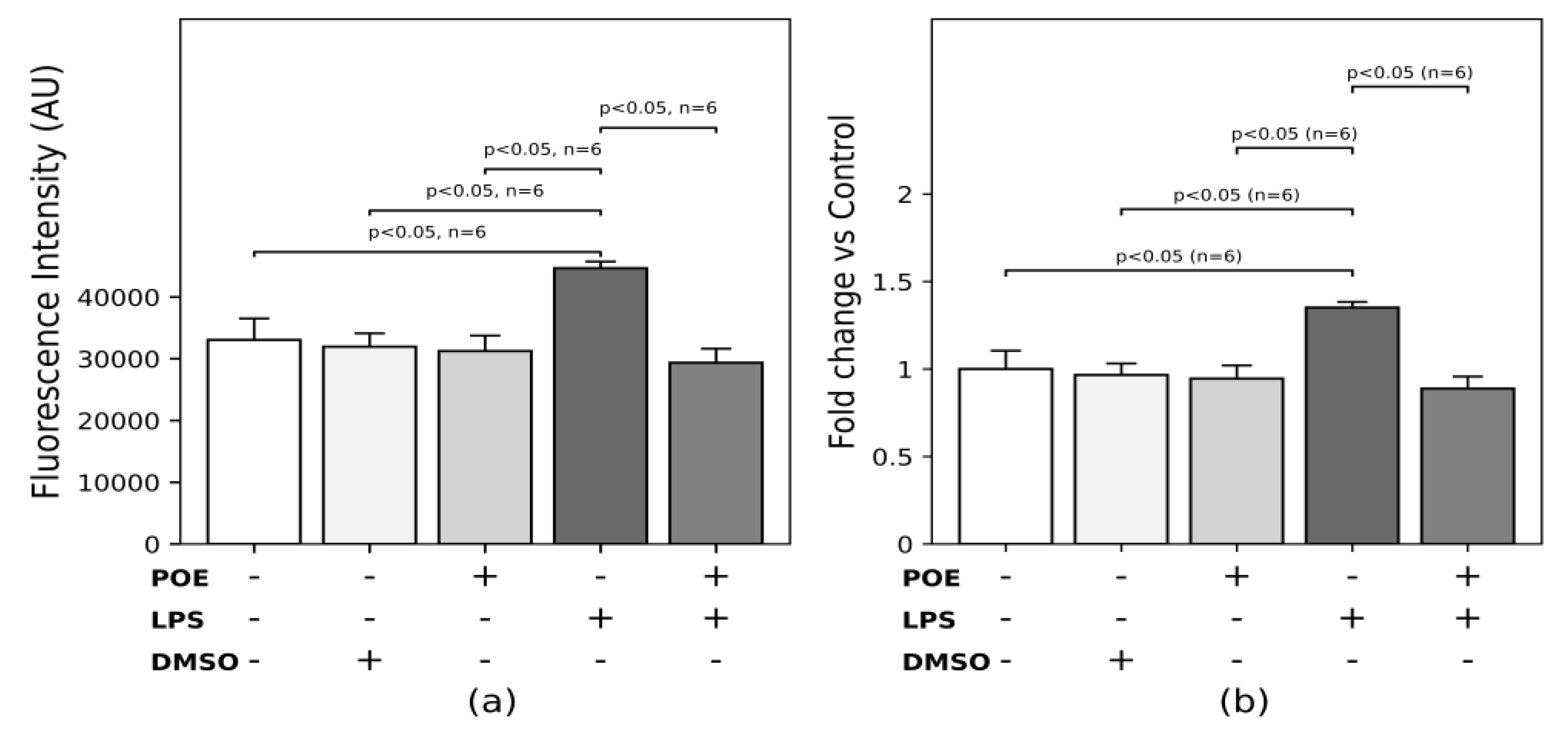
2.2. POE Mitigates the Stimulatory Effect of LPS on HaCaT Cell Proliferation
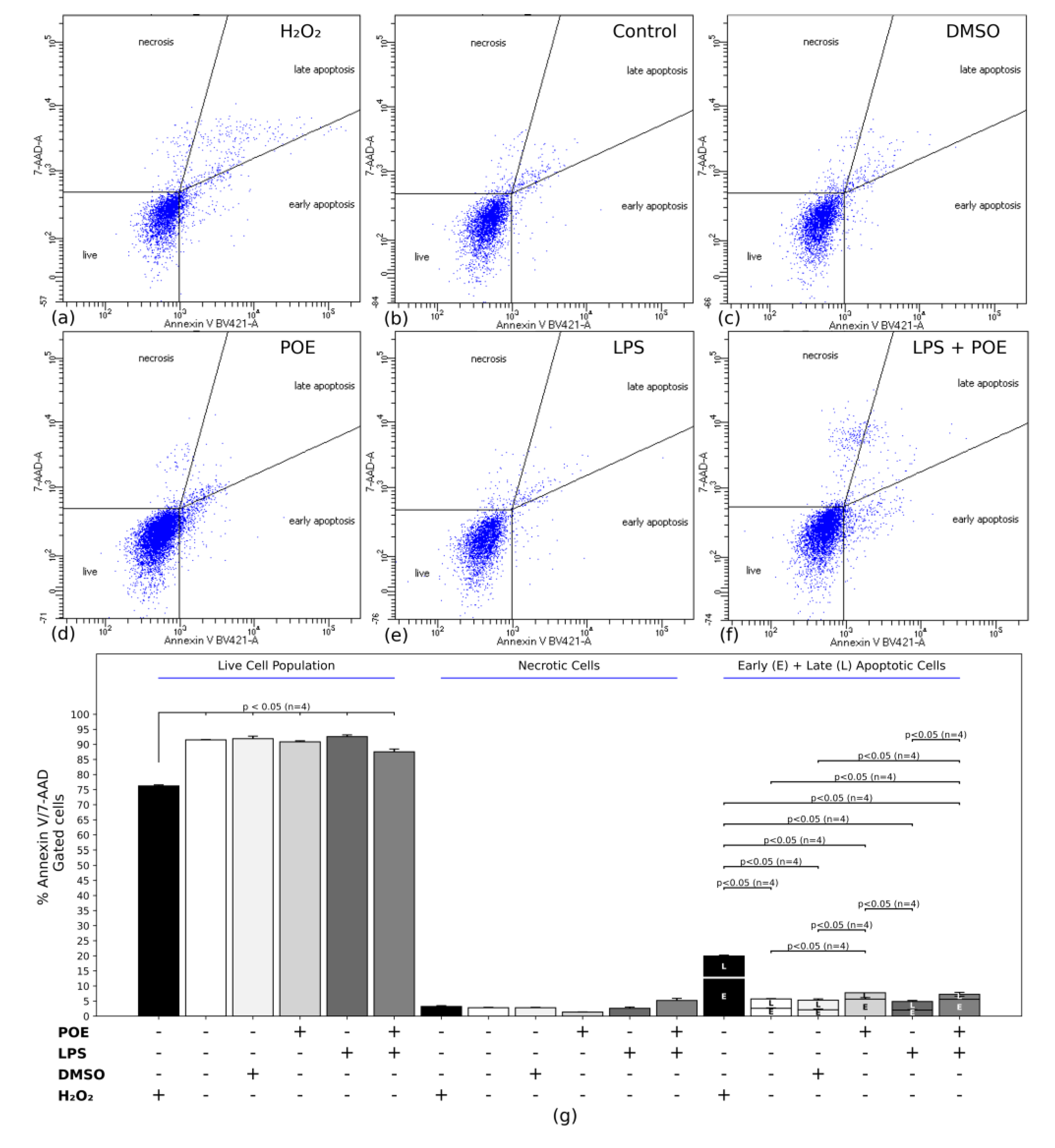
2.3. Analyses of Apoptosis During LPS and POE Stimulation
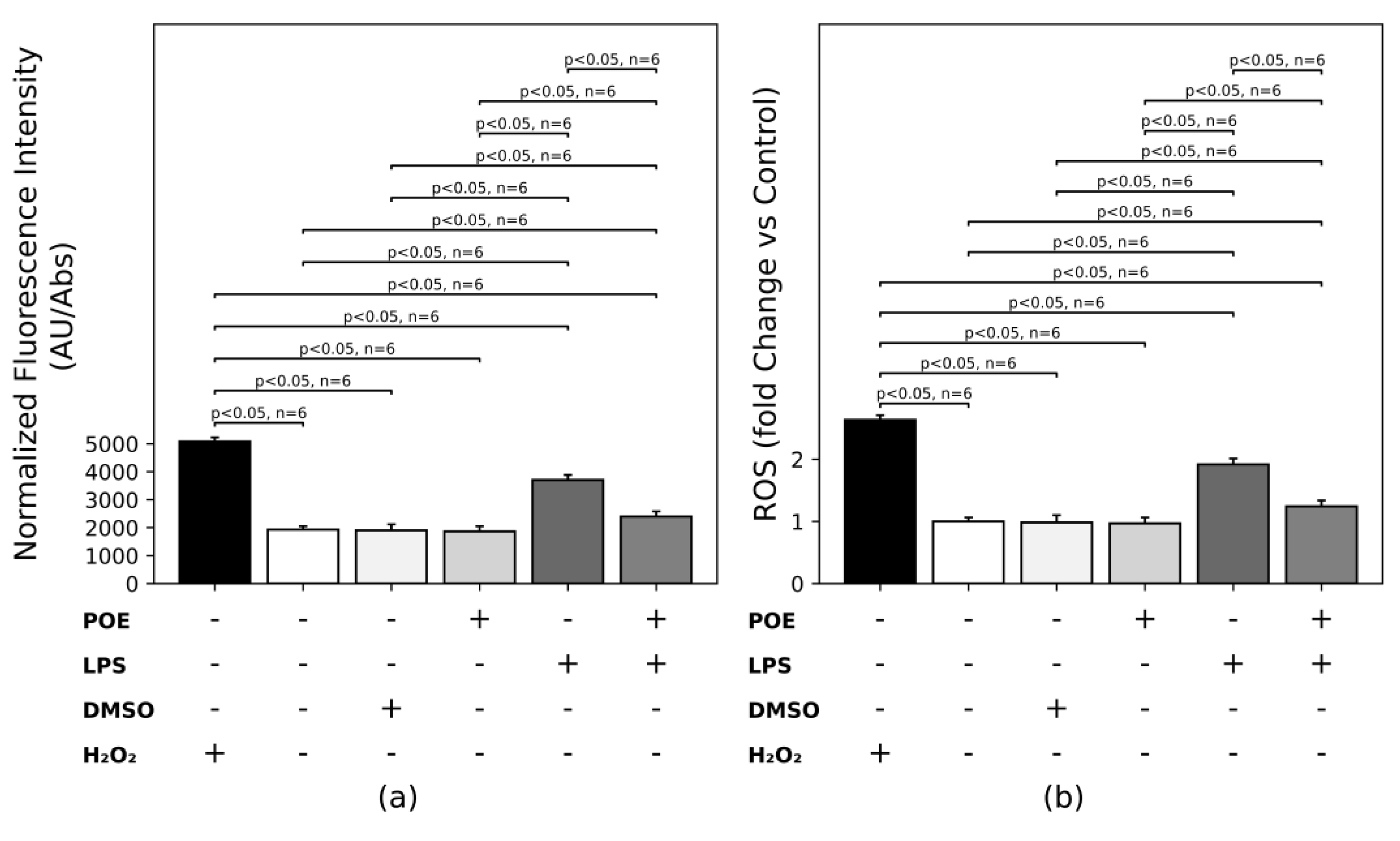
2.4. POE Reduces the ROS Increase Triggered by LPS Stimulation
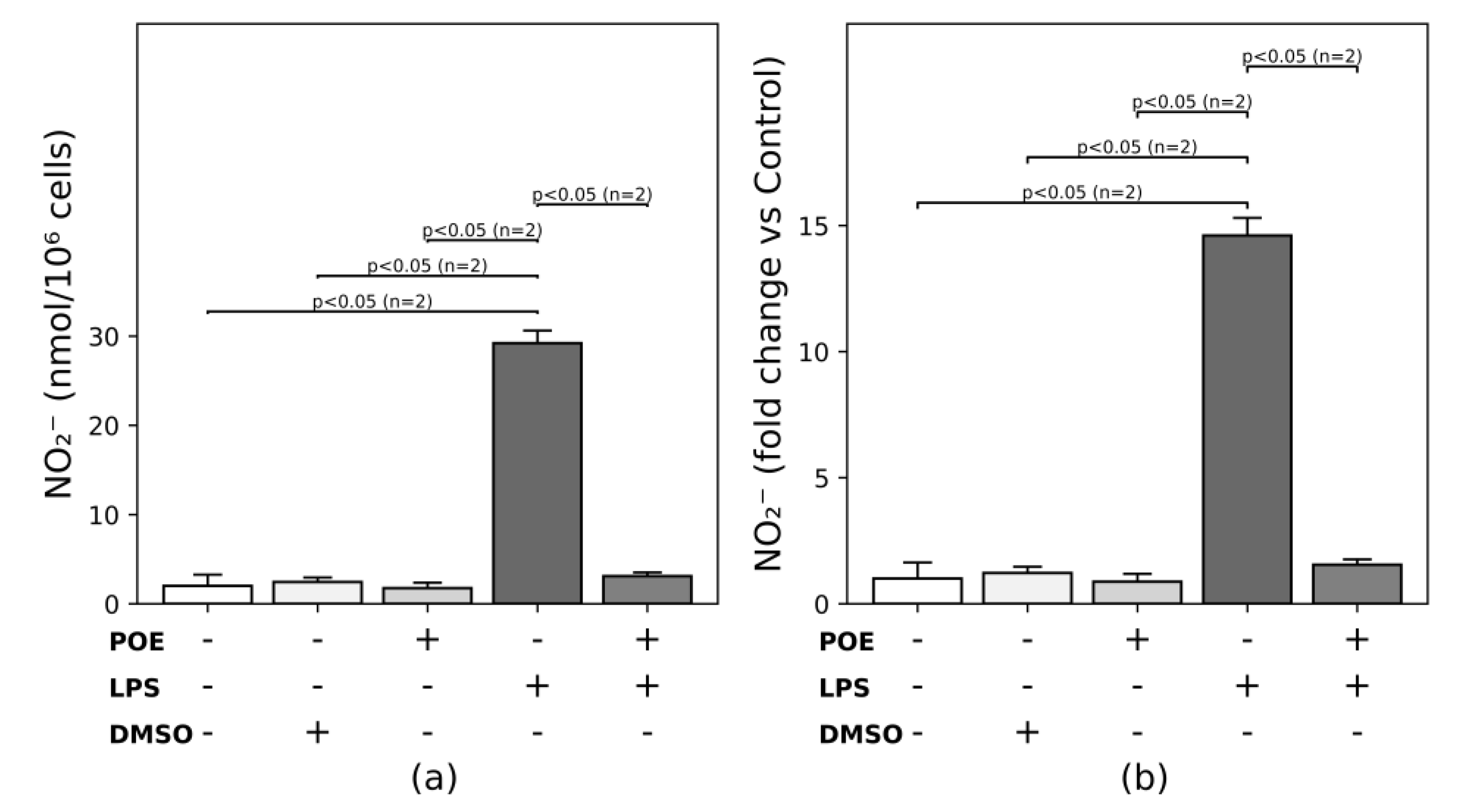
2.5. POE reduces the LPS-Induced Nitric Oxide Secretion
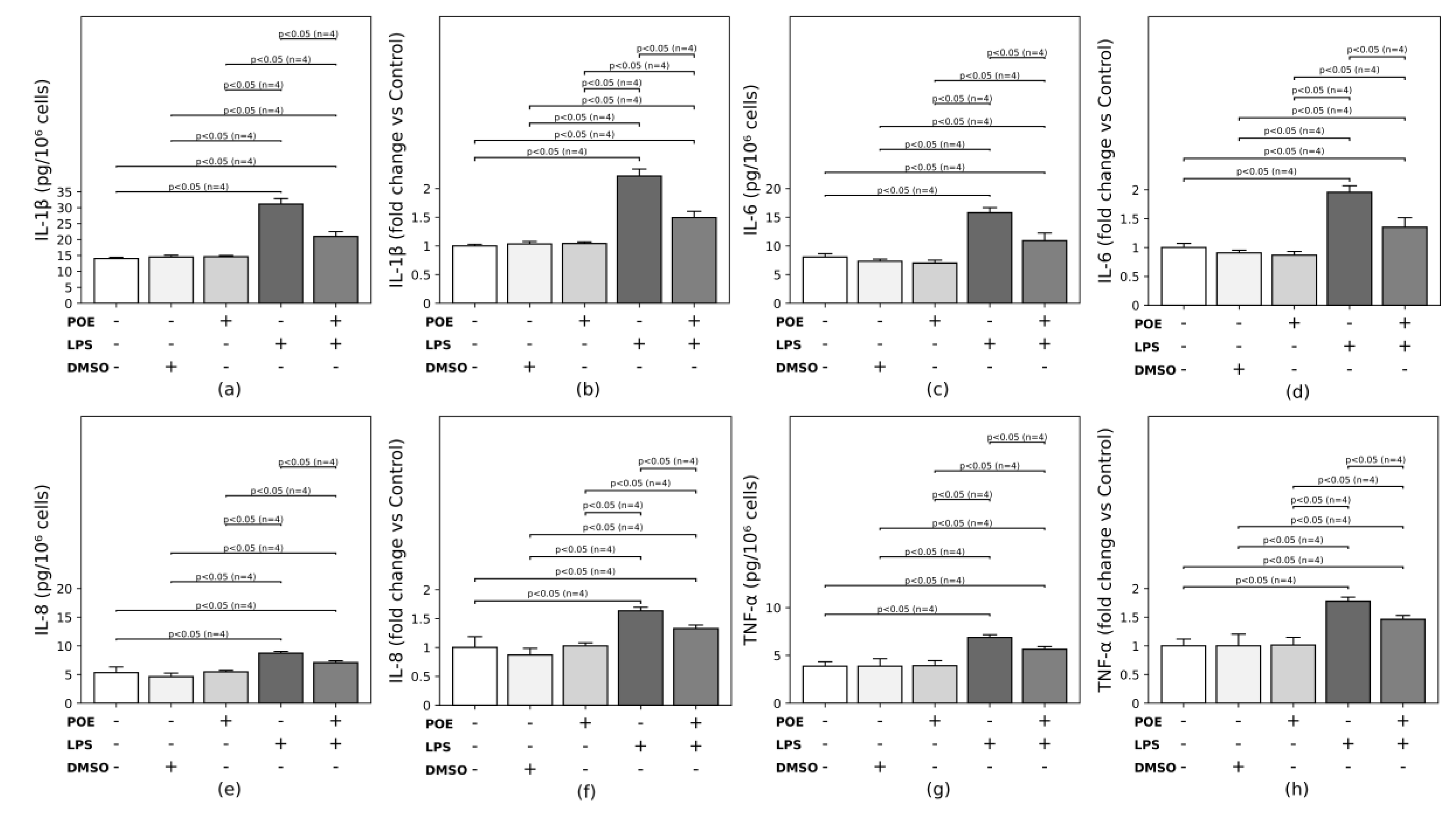
2.6. POE Reduces LPS-Induced Cytokine Secretion in HaCaT Cells
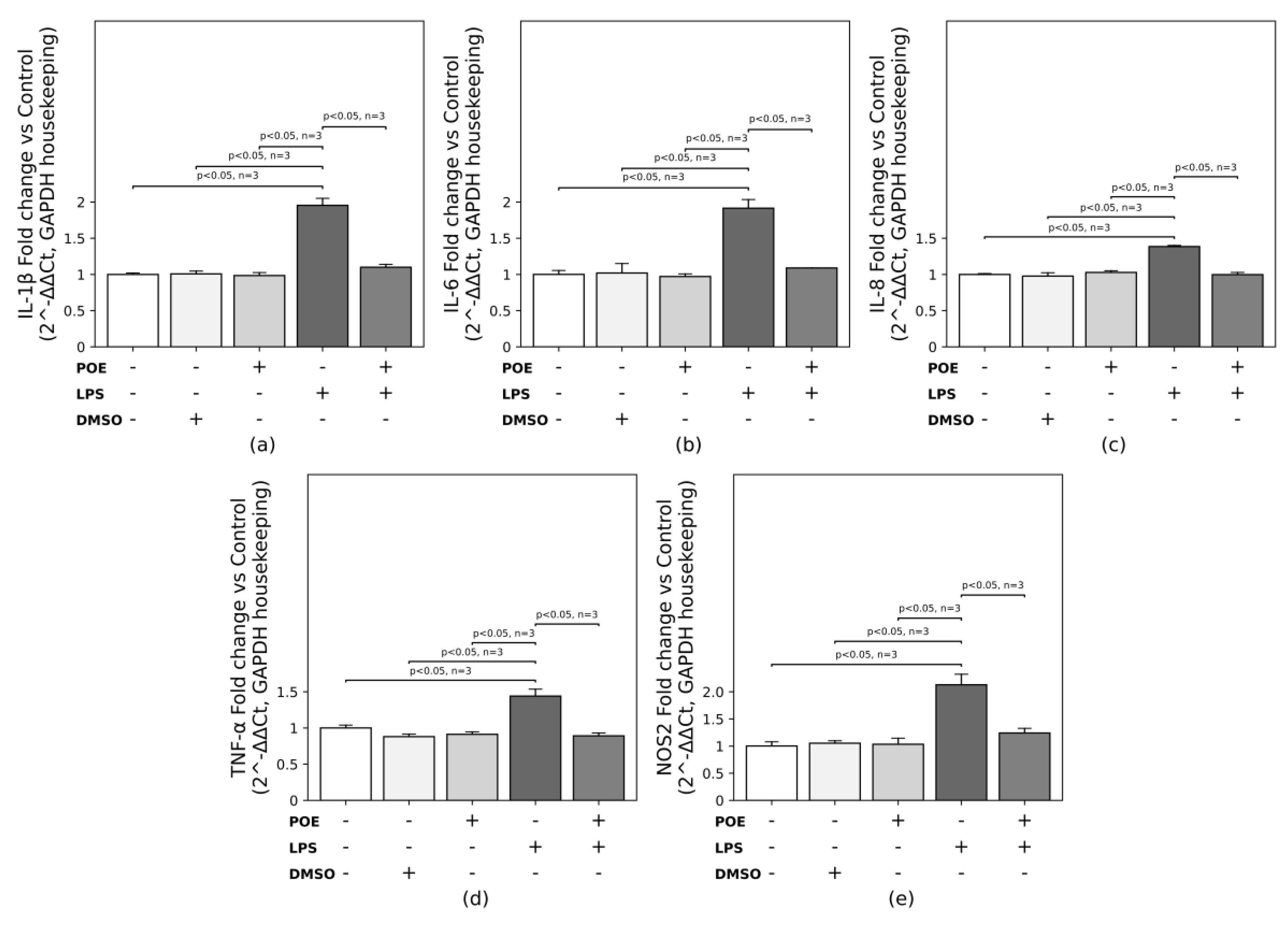
2.7. POE Suppresses Cytokine and Nitric Oxide Synthase 2 Expression Induced by LPS Stimulation
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Cell Line and Culture Conditions
4.3. Hydroalcoholic Extract of P. oceanica
4.4. Identification of Non-Toxic Doses of LPS and POE
4.5. Cell Proliferation Assay
4.6. Flow Cytometric Analysis of Apoptosis
4.7. ROS Detection
4.8. Assessment of Nitric Oxide Production
4.9. Assessment of Cytokine Secretion by ELISA
4.10. Cytokine and Nitric Oxide Synthase 2 Expression by qRT-PCR
4.11. Statistical Analysis
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Atopic dermatitis |
| Akt | Protein kinase B, Ak is the mouse strain and T is transforming ability |
| ANOVA | Analysis of variance |
| CD14 | Cluster of differentiation 14 |
| cDNA | Complementary DNA |
| CXCL10 | C-X-C motif chemokine ligand 10 |
| CXCL8/IL-8 | C-X-C motif chemokine ligand 8 / Interleukin-8 |
| CXCR4 | C-X-C chemokine receptor type 4 |
| DAMPs | Damage-associated molecular patterns |
| DMSO | Dimethyl sulfoxide |
| DMEM | Dulbecco’s modified Eagle’s medium |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| ELISA | Enzyme-linked immunosorbent assay |
| ERK1/2 | Extracellular signal-regulated kinases 1 and 2 |
| FBS | Fetal bovine serum |
| FRAP | Ferric reducing antioxidant power |
| GDF5 | Growth/Differentiation Factor 5 |
| H₂O₂ | Hydrogen peroxide |
| H₂DCF-DA | 2′,7′-Dichlorodihydrofluorescein diacetate |
| HaCaT | Human adult (keratinocytes) propagated under low Ca2+ and elevated Temperature |
| HSP70 | Heat shock protein 70 |
| HSP90 | Heat shock protein 90 |
| IFN-β | Interferon beta |
| IFN-γ | Interferon gamma |
| IL-1α | Interleukin-1 alpha |
| IL-1β | Interleukin-1 beta |
| IL-2 | Interleukin-2 |
| IL-6 | Interleukin-6 |
| IL-8 / CXCL8 | Interleukin-8 / C-X-C motif chemokine ligand 8 |
| IL-17A | Interleukin-17A |
| IL-17F | Interleukin-17F |
| IL-22 | Interleukin-22 |
| IL-23 | Interleukin-23 |
| IMQ | Imiquimod |
| LBP | LPS-binding protein |
| LCN-2 | Lipocalin-2 |
| LPS | Lipopolysaccharide |
| MAPKs | Mitogen-activated protein kinases |
| MD-2 | Myeloid differentiation factor 2 |
| MyD88 | Myeloid differentiation primary response 88 |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NO | Nitric oxide |
| NO₂⁻ | Nitrite |
| NOS2 (iNOS) | Inducible nitric oxide synthase 2 |
| PBS | Phosphate-buffered saline |
| PCR | Polymerase chain reaction |
| PI3K | Phosphoinositide 3-kinase |
| POE | Posidonia oceanica extract |
| P. oceanica | Posidonia oceanica |
| PAMPs | Pathogen-associated molecular patterns |
| RAW264.7 | Raschke William 264.7 Murine macrophage cell line |
| RNA | Ribonucleic acid |
| ROS | Reactive oxygen species |
| Th1 | Type 1 T helper lymphocytes |
| Th17 | Type 17 T helper lymphocytes |
| TNF-α | Tumor necrosis factor alpha |
| TLRs | Toll-like receptors |
| TLR4 | Toll-like receptor 4 |
| UPLC | Ultra-performance liquid chromatography |
| Rpm | Revolutions per minute |
Appendix A
Appendix A.1
| Year | Reference | Main effect of LPS stimulation |
| 2025 | Plata-Babula, A.; Kulej, W.; Ordon, P.; Gajdeczka, J.; Stefaniak, M.; Chwalba, A.; Gościniewicz, P.; Kulpok, T.; Grabarek, BO. Modulation of Nuclear Factor Kappa B Signaling and microRNA Profiles by Adalimumab in LPS-Stimulated Keratinocytes. DOI: 10.3390/ijms262010035 |
The effect of Adalimumab, a monoclonal antibody that inhibits tumor necrosis factor-alpha (TNF-α) used in psoriasis therapy, was evaluated on LPS-induced NF-κB–driven inflammation in HaCaT cells. |
| 2024 | Chen, L.; Liu, C.; Xiang, X.; Qiu, W.; Guo, K. miR-155 promotes an inflammatory response in HaCaT cells via the IRF2BP2/KLF2/NF-κB pathway in psoriasis. DOI: 10.3892/ijmm.2024.5415 | LPS induces inflammatory phenotype (↑IL-1β, ↑IL-6, ↑NF-κB). |
| 2023 | Kim, H.J.; Kim, S.Y.; Bae, H.J.; Choi, Y.Y.; An, J.Y.; Cho, Y.E.; Cho, S.Y.; Lee, S.J.; Lee, S.; Sin, M.; Yun, Y.M.; Lee, J.R.; Park, S.J. Anti-Inflammatory Effects of the LK5 Herbal Complex on LPS- and IL-4/IL-13-Stimulated HaCaT Cells and a DNCB-Induced Animal Model of Atopic Dermatitis in BALB/c Mice. DOI: 10.3390/pharmaceutics16010040 |
LPS induces keratinocyte inflammation for anti-inflammatory evaluation. |
| 2023 | Li, S.; Li, G.; Li, X.; Wu, F.; Li, L. Etanercept ameliorates psoriasis progression through regulating high mobility group box 1 pathway. DOI: 10.1111/srt.13329 | LPS triggers pro-inflammatory activation to test Etanercept effects. |
| 2023 | Wang, X.; Yao, Y.; Li, Y.; Guo, S.; Li, Y.; Zhang, G. Experimental study on the effect of luteolin on the proliferation, apoptosis and expression of inflammation-related mediators in lipopolysaccharide-induced keratinocytes. DOI: 10.1177/03946320231169175 | LPS induces inflammation, apoptosis, and proliferation changes in HaCaT. |
| 2023 | Kim, S.-Y.; Hong, M.; Deepa, P.; Sowndhararajan, K.; Park, S.J.; Park, S.; Kim, S. Carthamus tinctorius Suppresses LPS-Induced Anti-Inflammatory Responses by Inhibiting the MAPKs/NF-κB Signaling Pathway in HaCaT Cells. DOI: 10.3390/scipharm91010014 | LPS (1 μg/mL) stimulates NO, IL-6, IL-1β, NF-κB/MAPK activation. |
| 2022 | Zhang, Y.; Liu, P.; You, S.; Zhao, D.; An, Q.; Wang, D.; Zhang, J.; Li, M.; Wang, C. Anti-Inflammatory Effects of Opuntia Milpa Alta Polysaccharides Fermented by Lactic Acid Bacteria in Human Keratinocyte HaCaT Cells. DOI: 10.1002/cbdv.202100923 |
LPS triggers inflammatory phenotype to test polysaccharide effects. |
| 2021 | Takuathung, MN.; Potikanond, S.; Sookkhee, S.; Mungkornasawakul, P.; Jearanaikulvanich, T.; Chinda, K.; Wikan, N.; Nimlamool, W. Anti-psoriatic and anti-inflammatory effects of Kaempferia parviflora in keratinocytes and macrophage cells. DOI: 10.1016/j.biopha.2021.112229 |
LPS induces inflammatory stress in HaCaT cells. |
| 2021 | Yang X.; Dang X.; Zhang X.; Zhao S. Liquiritin reduces lipopolysaccharide-aroused HaCaT cell inflammation damage via regulation of microRNA-31/MyD88. DOI: 10.1016/j.intimp.2021.108283 |
LPS arouses inflammatory damage (↓viability, ↑IL-6/TNF-α/COX-2). |
| 2020 | Thatikonda, S.; Pooladanda, V.; Sigalapalli, DK.; Godugu, C. Piperlongumine regulates epigenetic modulation and alleviates psoriasis-like skin inflammation via inhibition of hyperproliferation and inflammation. DOI: 10.1038/s41419-019-2212-y |
LPS induces inflammation e hyperproliferation in HaCaT cells. |
References
- Shirley, S.N.; Watson, A.E.; Yusuf, N. Pathogenesis of Inflammation in Skin Disease: From Molecular Mechanisms to Pathology. Int J Mol Sci. 2024, 25:10152. [CrossRef]
- Hänel, K.H.; Cornelissen, C.; Lüscher, B.; Baron, J.M. Cytokines and the skin barrier. Int. J. Mol. Sci. 2013, 14, 6720–6745. [CrossRef]
- Luger, T.A.; Schwarz, T. Evidence for an epidermal cytokine network. J. Invest. Dermatol. 1990, 95, 100S–104S. [CrossRef]
- Zhou, X.; Chen, Y.; Cui, L.; Shi, Y.; Guo, C. Advances in the pathogenesis of psoriasis: from keratinocyte perspective. Cell Death Dis. 2022, 13, 81. [CrossRef]
- Sieminska, I.; Pieniawska, M.; Grzywa, T.M. The Immunology of Psoriasis—Current Concepts in Pathogenesis. Clin. Rev. Allergy Immunol. 2024, 66, 164–191. [CrossRef]
- Chen, C.H.; Wu, N.L.; Tsai, T.F. How Cells Die in Psoriasis? Int. J. Mol. Sci. 2025, 26, 3747. [CrossRef]
- Sun, L.; Liu, W.; Zhang, L.J. The Role of Toll-Like Receptors in Skin Host Defense, Psoriasis, and Atopic Dermatitis. J. Immunol. Res. 2019, 2019, 1824624. [CrossRef]
- Matsushima, Y.; Hwang, S.T.; Simon, S.I. Mechanobiology of neutrophil inflammasome signaling in psoriasis. Curr. Opin. Immunol. 2025, 97, 102680. [CrossRef]
- Ufomadu, P.; Gill, B.J.; Orengo, I.; Rosen, T.; Shimizu, I. The Efficacy of Complementary and Alternative Medicines in Medical Dermatology: A Comprehensive Review. J. Clin. Aesthet. Dermatol. 2025, 18, E61–E79. https://pmc.ncbi.nlm.nih.gov/articles/PMC12189221/.
- Landis, E.T.; Davis, S.A.; Feldman, S.R.; Taylor, S. Complementary and alternative medicine use in dermatology in the United States. J. Altern. Complement Med. 2014, 20, 392–398. [CrossRef]
- Chalupczak, N.V.; Lio, P.A. Complementary and Alternative Therapies for Psoriasis. Arch. Dermatol. Res. 2024, 316, 531. [CrossRef]
- Vasarri, M.; De Biasi, A.M.; Barletta, E.; Pretti, C.; Degl’Innocenti, D. An Overview of New Insights into the Benefits of the Seagrass Posidonia oceanica for Human Health. Mar. Drugs 2021, 19, 476. [CrossRef]
- Micheli, L.; Vasarri, M.; Degl’Innocenti, D.; Di Cesare Mannelli, L.; Ghelardini, C.; Emiliano, A.; Verdelli, A.; Caproni, M.; Barletta, E. Posidonia oceanica (L.) Delile Is a Promising Marine Source Able to Alleviate Imiquimod-Induced Psoriatic Skin Inflammation. Mar. Drugs 2024, 22, 300. [CrossRef]
- Boukamp, P.; Petrussevska, R.T.; Breitkreutz, D.; Hornung, J.; Markham, A.; Fusenig, N.E. Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J. Cell Biol. 1988, 106, 761–771. [CrossRef]
- Wilson, V.G. Growth and differentiation of HaCaT keratinocytes. Methods Mol. Biol. 2014, 1195, 33–41. [CrossRef]
- Kim, D.H.; Byamba, D.; Wu, W.H.; Kim, T.G.; Lee, M.G. Different characteristics of reactive oxygen species production by human keratinocyte cell line cells in response to allergens and irritants. Exp. Dermatol. 2012, 21, 99–103. [CrossRef]
- Mohamadzadeh, M.; Müller, M.; Hultsch, T.; Enk, A.; Saloga, J.; Knop, J. Enhanced expression of IL-8 in normal human keratinocytes and human keratinocyte cell line HaCaT in vitro after stimulation with contact sensitizers, tolerogens and irritants. Exp. Dermatol. 1994, 3, 298–303. [CrossRef]
- Wang, B.; Ruiz, N.; Pentland, A.; Caparon, M. Keratinocyte proinflammatory responses to adherent and nonadherent group A streptococci. Infect. Immun. 1997, 65, 2119–2126. [CrossRef]
- Lu, K.Y.; Cheng, L.C.; Hung, Z.C.; Chen, Z.Y.; Wang, C.W.; Hou, H.H. The Ethyl Acetate Extract of Caulerpa microphysa Promotes Collagen Homeostasis and Inhibits Inflammation in the Skin. Curr. Issues Mol. Biol. 2024, 46, 2701–2712. [CrossRef]
- Curzytek, K.; Maes, M.; Kubera, M. Immune-Regulatory and Molecular Effects of Antidepressants on the Inflamed Human Keratinocyte HaCaT Cell Line. Neurotox. Res. 2021, 39, 1211–1226. [CrossRef]
- An, J.Y.; Kim, S.Y.; Kim, H.J.; Bae, H.J.; Lee, H.D.; Choi, Y.Y.; Cho, Y.E.; Cho, S.Y.; Lee, S.J.; Lee, S.; Park, S.J. Geraniin from the methanol extract of Pilea mongolica suppresses LPS-induced inflammatory responses by inhibiting IRAK4/MAPKs/NF-κB/AP-1 pathway in HaCaT cells. Int. Immunopharmacol. 2024, 140, 112767. [CrossRef]
- Cals-Grierson, M.M.; Ormerod, A.D. Nitric oxide function in the skin. Nitric Oxide 2004, 10, 179–193. [CrossRef]
- Luo, R.; Yao, Y.; Chen, Z.; Sun, X. An examination of the LPS-TLR4 immune response through the analysis of molecular structures and protein-protein interactions. Cell Commun. Signal. 2025, 23, 142. [CrossRef]
- Kulms, D.; Schwarz, T. NF-kappaB and cytokines. Vitam. Horm. 2006, 74, 283–300. [CrossRef]
- Vasarri, M.; Leri, M.; Barletta, E.; Ramazzotti, M.; Marzocchini, R.; Degl’Innocenti, D. Anti-inflammatory properties of the marine plant Posidonia oceanica (L.) Delile. J. Ethnopharmacol. 2020, 247, 112252. [CrossRef]
- Burke, O.M.; Frerichs, V.R.; Garcia, D.F.; Stone, R.C.; Lev-Tov, H.; Czarnowicki, T.; Keane, R.W.; Ojeh, N.; Marjanovic, J.; Pastar, I.; Tomic-Canic, M.; de Rivero Vaccari, J.P.; Sawaya, A.P. The impact of innate immunity and epigenetics in the pathogenesis of hidradenitis suppurativa. Front. Immunol., 2025, 16, 1593253. [CrossRef]
- Ortiz-Lopez, L.I.; Choudhary, V.; Bollag, W.B. Updated perspectives on keratinocytes and psoriasis: keratinocytes are more than innocent bystanders. Psoriasis (Auckl.), 2022, 12, 73–87. [CrossRef]
- Chieosilaatham, P.; Kiatsurayanon, C.; Umehara, Y.; Trujillo-Paez, J.V.; Peng, G.; Yue, H.; Nguyen, L.T.H.; Niyonsaba, F. Keratinocytes: innate immune cells in atopic dermatitis. Clin. Exp. Immunol., 2021, 204, 296–309. [CrossRef]
- Morizane, S.; Mukai, T.; Sunagawa, K.; Tachibana, K.; Kawakami, Y.; Ouchida, M. Input/output cytokines in epidermal keratinocytes and the involvement in inflammatory skin diseases. Front. Immunol., 2023, 14, 1239598. [CrossRef]
- Miller, L.S. Toll-like receptors in skin. Adv. Dermatol., 2008, 24, 71–87. [CrossRef]
- Mao, H.; Zhao, X.; Sun, S.C. NF-κB in inflammation and cancer. Cell Mol. Immunol., 2025, 22, 811–839. [CrossRef]
- Salauddin, M.; Bhattacharyya, D.; Samanta, I.; Saha, S.; Xue, M.; Hossain, M.G.; Zheng, C. Role of TLRs as signaling cascades to combat infectious diseases: a review. Cell Mol. Life Sci., 2025, 82, 122. [CrossRef]
- Zhou, S.; Yao, Z. Roles of Infection in Psoriasis. Int. J. Mol. Sci., 2022, 23, 6955. [CrossRef]
- Ustuner, P.; Ceylan, A.N. Microbiological analysis of patients with inverse and plaque-type psoriasis. BMC Microbiol., 2025, 6, [Epub ahead of print]. [CrossRef]
- Ciesielska, A.; Matyjek, M.; Kwiatkowska, K. TLR4 and CD14 trafficking and its influence on LPS-induced pro-inflammatory signaling. Cell Mol. Life Sci., 2021, 78, 1233–1261. [CrossRef]
- Kuzmich, N.N.; Sivak, K.V.; Chubarev, V.N.; Porozov, Y.B.; Savateeva-Lyubimova, T.N.; Peri, F. TLR4 signaling pathway modulators as potential therapeutics in inflammation and sepsis. Vaccines (Basel), 2017, 5, 34. [CrossRef]
- Köllisch, G.; Kalali, B.N.; Voelcker, V.; Wallich, R.; Behrendt, H.; Ring, J.; Bauer, S.; Jakob, T.; Mempel, M.; Ollert, M. Various members of the Toll-like receptor family contribute to the innate immune response of human epidermal keratinocytes. Immunology, 2005, 114, 531–541. [CrossRef]
- Zheng, H.; Gu, L.; Wang, Z.; Zhou, H.; Zhang, C.; Teng, X.; Hu, Z.; Wei, X.; Liu, X.; Zeng, F.; Zhao, Q.; Hao, Y.; Hu, Y.; Wang, X.; Hu, J.; Yu, J.; Wu, W.; Zhou, Y.; Cui, K.; Huang, N.; Li, J. Establishing transcription profile of psoriasiform cutaneous in vitro using HaCaT cells stimulated with combination of cytokines. J. Vis. Exp., 2021, (169), [e61537]. [CrossRef]
- Triantafilou, M.; Triantafilou, K. Lipopolysaccharide recognition: CD14, TLRs and the LPS-activation cluster. Trends Immunol., 2002, 23, 301–304. [CrossRef]
- Triantafilou, M.; Lepper, P.M.; Briault, C.D.; Ahmed, M.A.; Dmochowski, J.M.; Schumann, C.; Triantafilou, K. Chemokine receptor 4 (CXCR4) is part of the lipopolysaccharide “sensing apparatus”. Eur. J. Immunol., 2008, 38, 192–203. [CrossRef]
- Micheli, L.; Vasarri, M.; Degl’Innocenti, D.; Di Cesare Mannelli, L.; Ghelardini, C.; Emiliano, A.; Verdelli, A.; Caproni, M.; Barletta, E. Posidonia oceanica (L.) Delile is a promising marine source able to alleviate imiquimod-induced psoriatic skin inflammation. Mar. Drugs, 2024, 22, 300. [CrossRef]
- Wang, J.; Luo, H.; Xiao, Y.; Wang, L. miR-125b inhibits keratinocyte proliferation and promotes keratinocyte apoptosis in oral lichen planus by targeting MMP-2 expression through PI3K/Akt/mTOR pathway. Biomed. Pharmacother., 2016, 80, 373–380. [CrossRef]
- Sangaran, P.G.; Ibrahim, Z.A.; Chik, Z.; Mohamed, Z.; Ahmadiani, A. Lipopolysaccharide pre-conditioning attenuates pro-inflammatory responses and promotes cytoprotective effect in differentiated PC12 cell lines via pre-activation of Toll-like receptor-4 signaling pathway leading to the inhibition of caspase-3/nuclear factor-κappa B pathway. Front. Cell Neurosci., 2021, 14, 598453. [CrossRef]
- Arya, P.; Sharma, V.; Singh, P.; Thapliyal, S.; Sharma, M. Bacterial endotoxin-lipopolysaccharide role in inflammatory diseases: an overview. Iran J. Basic Med. Sci., 2025, 28, 553–564. [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target Ther., 2017, 2, 17023. [CrossRef]
- Luo, J.L.; Kamata, H.; Karin, M. IKK/NF-kappaB signaling: balancing life and death--a new approach to cancer therapy. J. Clin. Invest., 2005, 115, 2625–2632. [CrossRef]
- Liu, D.; Zhong, Z.; Karin, M. NF-κB: A Double-Edged Sword Controlling Inflammation. Biomedicines, 2022, 10, 1250. [CrossRef]
- Bhat, I.A.; Bhat, A.M.; Tasduq, A.S. Apoptosis-Mechanisms, Regulation in Pathology, and Therapeutic. In , Biochemistrym, Cell Death Regulation in Pathology, Carafa, V., Ed; IntechOpen Limited: London, UK, 2025; [CrossRef]
- Mustafa, A.M.; Atwa, A.M.; Elgindy, A.M.; Alkabbani, M.A.; Ibrahim, K.M.; Esmail, M.M.; El-Shiekh, R.A.; Mohamed, E.M.; Kamel, K.M. Targeting psoriatic inflammation with natural compounds: mechanistic insights and therapeutic promise. Inflammopharmacology, 2025, 33, 3843–3870. [CrossRef]
- Patel, P.; Garala, K.; Bagada, A.; Singh, S.; Prajapati, B.G.; Kapoor, D. Phyto-pharmaceuticals as a safe and potential alternative in management of psoriasis: a review. Z. Naturforsch. C J. Biosci., 2024, 80, 409–430. [CrossRef]
- Marrassini, C.; Arcos, M.L.B.; Cogoi, L.; Alonso, M.R.; Anesini, C. Anti-Inflammatory Effect of Urtica urens on LPS-Challenged Keratinocytes. J. Pharmacopuncture, 2025, 28, 311–320. [CrossRef]
- An, J.Y.; Kim, S.Y.; Kim, H.J.; Bae, H.J.; Lee, H.D.; Choi, Y.Y.; Cho, Y.E.; Cho, S.Y.; Lee, S.J.; Lee, S.; Park, S.J. Geraniin from the methanol extract of Pilea mongolica suppresses LPS-induced inflammatory responses by inhibiting IRAK4/MAPKs/NF-κB/AP-1 pathway in HaCaT cells. Int. Immunopharmacol., 2024, 140, 112767. [CrossRef]
- Jin, C.; Cha, Y.J.; Kim, H.E.; Jin, Y.; Zhang, X.; Le, L.T.T.; Giang, N.N.; Choi, K.; Lee, Y.H.; Kim, W.H.; Choi, J.H.; Chien, P.N.; Yeong, H.C. A Natural Composite Extract Restores Skin Barrier Function by Modulating Inflammatory, Hydration, and Redox Pathways. J. Cosmet. Dermatol., 2025, 24, e70613. [CrossRef]
- Wang, X.; Yao, Y.; Li, Y.; Guo, S.; Li, Y.; Zhang, G. Experimental study on the effect of luteolin on the proliferation, apoptosis and expression of inflammation-related mediators in lipopolysaccharide-induced keratinocytes. Int. J. Immunopathol. Pharmacol., 2023, 37, 3946320231169175. [CrossRef]
- Klimitz, F.J.; Shen, Y.; Repetto, F.; Brown, S.; Knoedler, L.; Ko, C.J.; Abu Hussein, N.; Crisler, W.J.; Adams, T.; Kaminski, N.; Lian, C.G.; Murphy, G.F.; Hsia, H.C.; Pomahac, B.; Kauke-Navarro, M. Keratinocytes as active regulators of cutaneous and mucosal immunity: a systematic review across inflammatory epithelial disorders. Front. Immunol., 2025, 16, 1694066. [CrossRef]
- Bruch-Gerharz, D.; Fehsel, K.; Suschek, C.; Michel, G.; Ruzicka, T.; Kolb-Bachofen, V. A proinflammatory activity of interleukin 8 in human skin: expression of the inducible nitric oxide synthase in psoriatic lesions and cultured keratinocytes. J. Exp. Med. 1996, 184, 2007–2012. [CrossRef]
- Sirsjö, A.; Karlsson, M.; Gidlöf, A.; Rollman, O.; Törmä, H. Increased expression of inducible nitric oxide synthase in psoriatic skin and cytokine-stimulated cultured keratinocytes. Br. J. Dermatol., 1996, 134, 643–648. [CrossRef]
- Frank, S.; Kämpfer, H.; Podda, M.; Kaufmann, R.; Pfeilschifter, J. Identification of copper/zinc superoxide dismutase as a nitric oxide-regulated gene in human (HaCaT) keratinocytes: implications for keratinocyte proliferation. Biochem. J., 2000, 346, 719–728. [CrossRef]
- Khorsandi, K.; Hosseinzadeh, R.; Esfahani, H.; Zandsalimi, K.; Shahidi, F.K.; Abrahamse, H. Accelerating skin regeneration and wound healing by controlled ROS from photodynamic treatment. Inflamm. Regen., 2022, 42, 40. [CrossRef]
- Bito, T.; Nishigori, C. Impact of reactive oxygen species on keratinocyte signaling pathways. J. Dermatol. Sci., 2012, 68, 3–8. [CrossRef]
- Venza, I.; Venza, M.; Visalli, M.; Lentini, G.; Teti, D.; d’Alcontres, F.S. ROS as Regulators of Cellular Processes in Melanoma. Oxid. Med. Cell Longev., 2021, 2021, 1208690. [CrossRef]
- Greenbaum, D.; Colangelo, C.; Williams, K.; Gerstein, M. Comparing protein abundance and mRNA expression levels on a genomic scale. Genome Biol., 2003, 4, 117. [CrossRef]
- Maddur, M.S.; Miossec, P.; Kaveri, S.V.; Bayry, J. Th17 cells: biology, pathogenesis of autoimmune and inflammatory diseases, and therapeutic strategies. Am. J. Pathol., 2012, 181, 8–18. [CrossRef]
- Qin, J.Z.; Bacon, P.; Chaturvedi, V.; Nickoloff, B.J. Role of NF-kappaB activity in apoptotic response of keratinocytes mediated by interferon-gamma, tumor necrosis factor-alpha, and tumor-necrosis-factor-related apoptosis-inducing ligand. J. Invest. Dermatol., 2001, 117, 898–907. [CrossRef]
- Zhang, W.; Zhu, G.; Sun, H.; Jiang, C. NLRC3 affects the development of psoriasis by modulating the NF-κB signaling pathway mediated inflammatory response through its interaction with TRAF6. Immunol. Lett., 2025, 272, 106949. [CrossRef]
- Guo, J.; Zhang, H.; Lin, W.; Lu, L.; Su, J.; Chen, X. Signaling pathways and targeted therapies for psoriasis. Signal Transduct. Target Ther., 2023, 8, 437. Erratum in: Signal Transduct. Target Ther., 2024, 9, 25. https://doi.org/10.1038/s41392-023-01733-9. [CrossRef]
- Barletta, E.; Ramazzotti, M.; Fratianni, F.; Pessani, D.; Degl’Innocenti, D. Hydrophilic extract from Posidonia oceanica inhibits activity and expression of gelatinases and prevents HT1080 human fibrosarcoma cell line invasion. Cell Adh. Migr., 2015, 9, 422–431. [CrossRef]
- Medina, M.B. Simple and rapid method for the analysis of phenolic compounds in beverages and grains. J. Agric. Food Chem., 2011, 59, 1565–1571. [CrossRef]
- Pulido, R.; Bravo, L.; Saura-Calixto, F. Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J. Agric. Food Chem., 2000, 48, 3396–3402. [CrossRef]
- Fukumoto, L.R.; Mazza, G. Assessing antioxidant and prooxidant activities of phenolic compounds. J. Agric. Food Chem., 2000, 48, 3597–3604. [CrossRef]
- Leri, M.; Ramazzotti, M.; Vasarri, M.; Peri, S.; Barletta, E.; Pretti, C.; Degl’Innocenti, D. Bioactive Compounds from Posidonia oceanica (L.) Delile Impair Malignant Cell Migration through Autophagy Modulation. Mar. Drugs, 2018, 16, 137. [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods, 1983, 65, 55–63. [CrossRef]
- Jones, L.J.; Gray, M.; Yue, S.T.; Haugland, R.P.; Singer, V.L. Sensitive determination of cell number using the CyQUANT cell proliferation assay. J. Immunol. Methods, 2001, 254, 85–98. [CrossRef]
- LeBel, C.P.; Ischiropoulos, H.; Bondy, S.C. Evaluation of the probe 2’,7’-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem. Res. Toxicol., 1992, 5, 227-31. [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal. Biochem., 1982, 126, 131-8. [CrossRef]
| Gene product | Direction | Sequence (5’→3’) | Product size | Cat. No. |
| IL-1β | F | CCACAGACCTTCCAGGAGAATG | 131 bp | HP200544 |
| R | GTGCAGTTCAGTGATCGTACAGG | |||
| IL-6 | F | AGACAGCCACTCACCTCTTCAG | 132 bp | HP200567 |
| R | TTCTGCCAGTGCCTCTTTGCTG | |||
| IL-8 | F | GAGAGTGATTGAGAGTGGACCAC | 112 bp | HP200551 |
| R | CACAACCCTCTGCACCCAGTTT | |||
| TNF-α | F | CTCTTCTGCCTGCTGCACTTTG | 135 bp | HP200561 |
| R | ATGGGCTACAGGCTTGTCACTC | |||
| NOS2 | F | GCTCTACACCTCCAATGTGACC | 136 bp | HP200591 |
| R | CTGCCGAGATTTGAGCCTCATG | |||
| GAPDH | F | GTCTCCTCTGACTTCAACAGCG | 131 bp | HP205798 |
| R | ACCACCCTGTTGCTGTAGCCAA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




