Submitted:
05 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Method
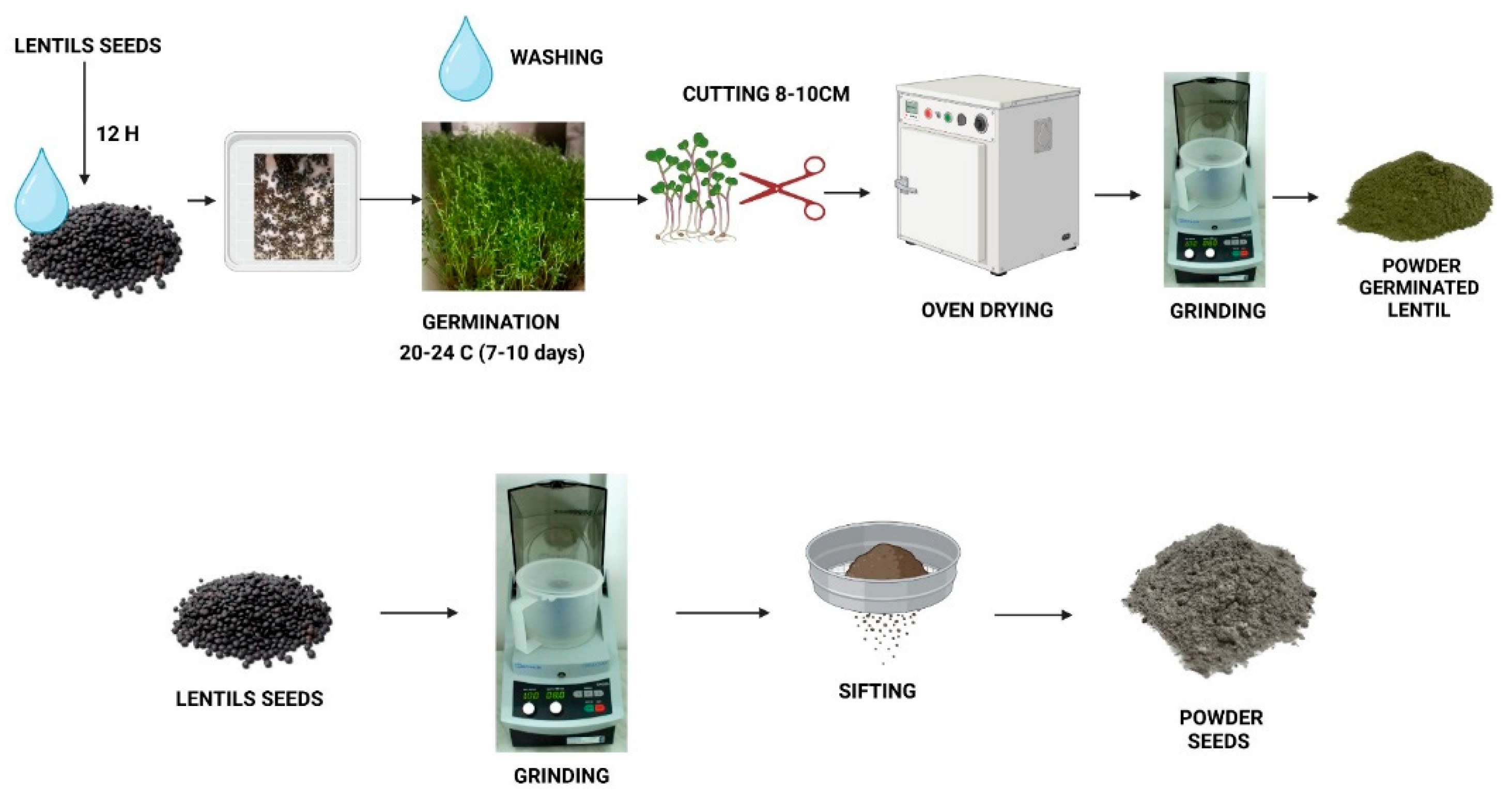
2.1. Preparation of Powder Seeds and Germinated Lentil as Microgreen
2.2. Flours and Breads Preparation
2.3. Evaluation of the Physical Properties of Bread Samples
2.4. Determination of Nutritional Composition
2.5. Determination of Antinutritional Compounds – Phytic Acid
2.6. Determination of Total Polyphenols Content (TPC)
2.7. Determination of Color Parameters
2.8. Statistical Analysis
3. Results and Discussion
3.1. Physical Characteristics of Bread Samples
3.2. Nutritional Composition of Bread Samples
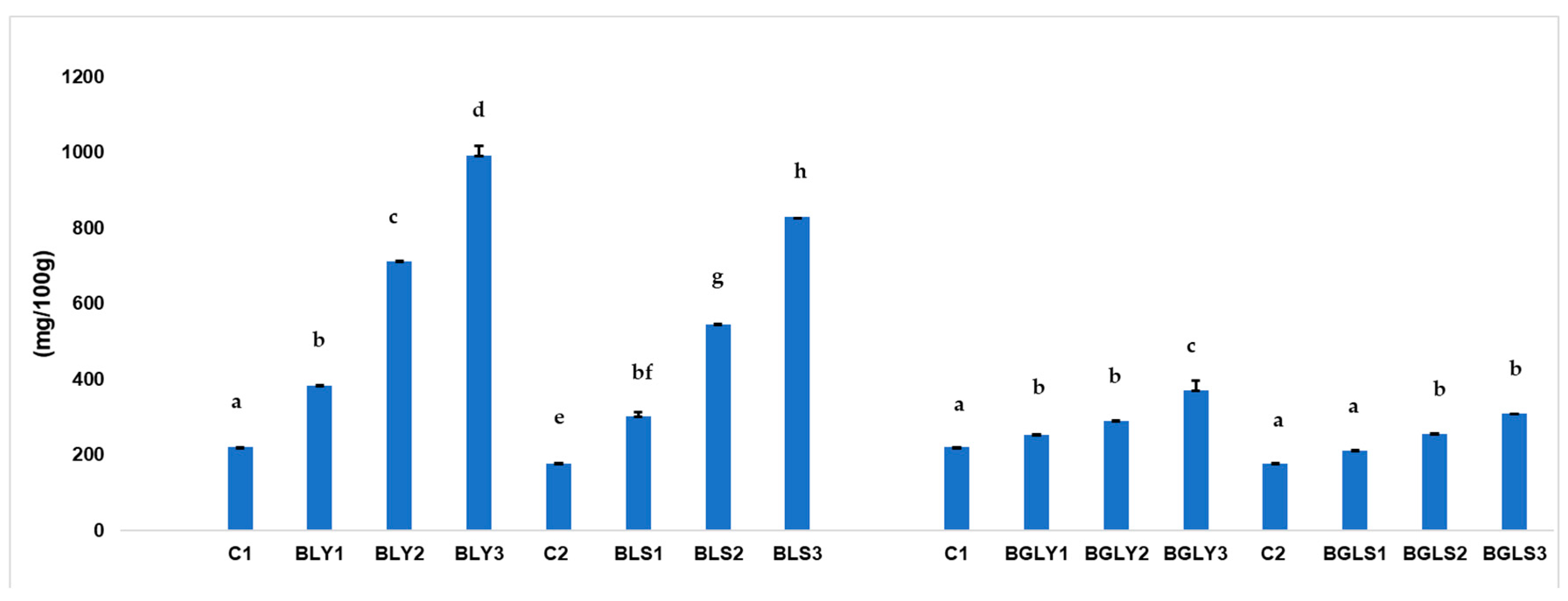
3.3. Antinutritional Compounds of Bread Samples(Phytic Acid)
3.4. Color Parameters of Bread Samples
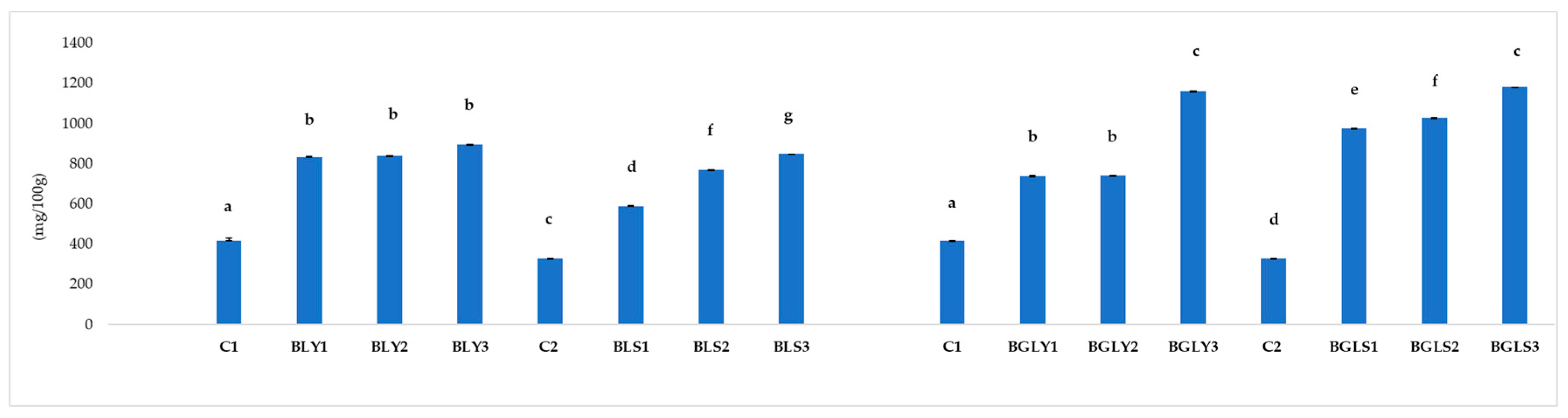
3.5. Total Phenols Content (TPC) of Bread Samples
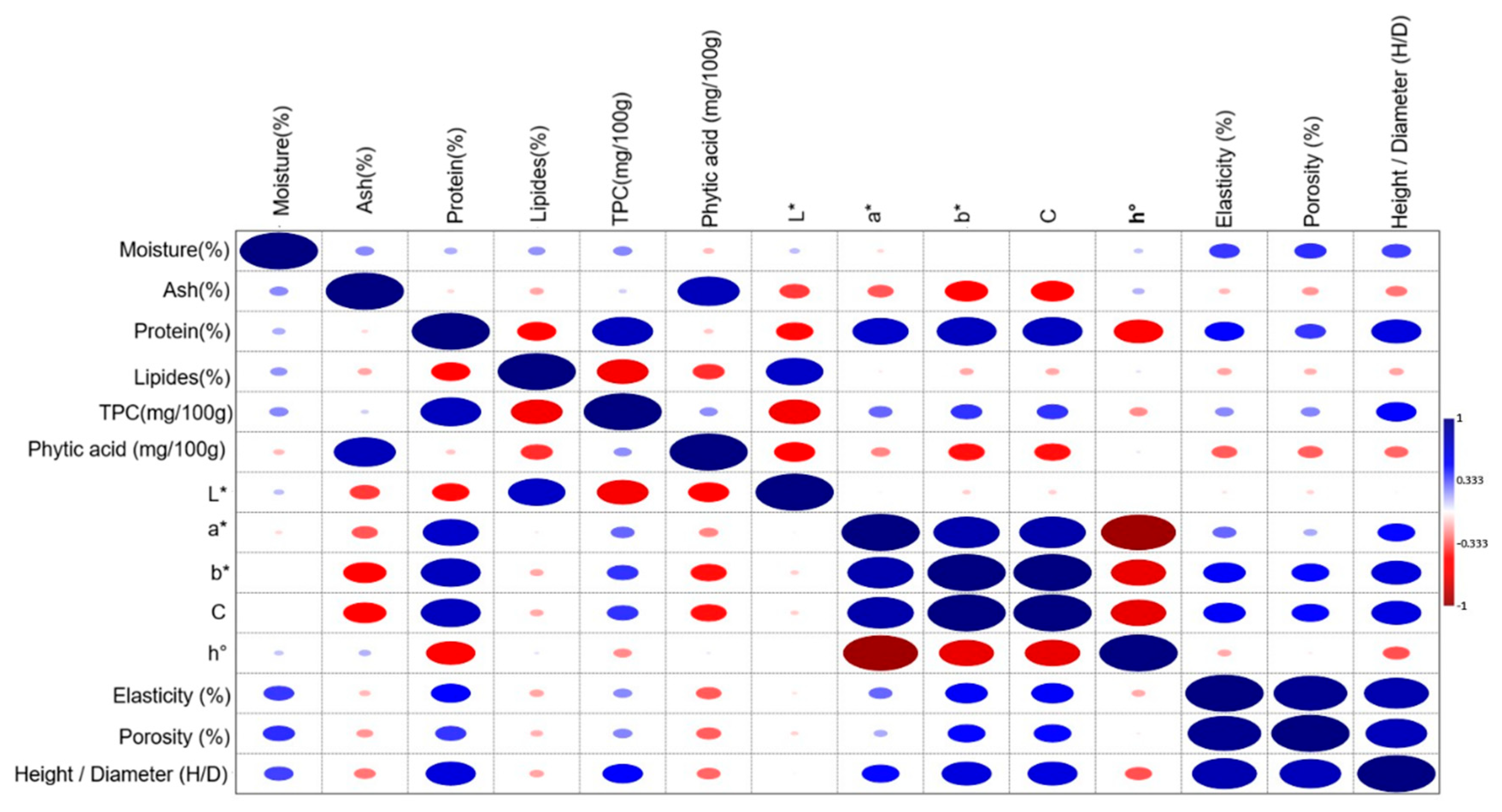
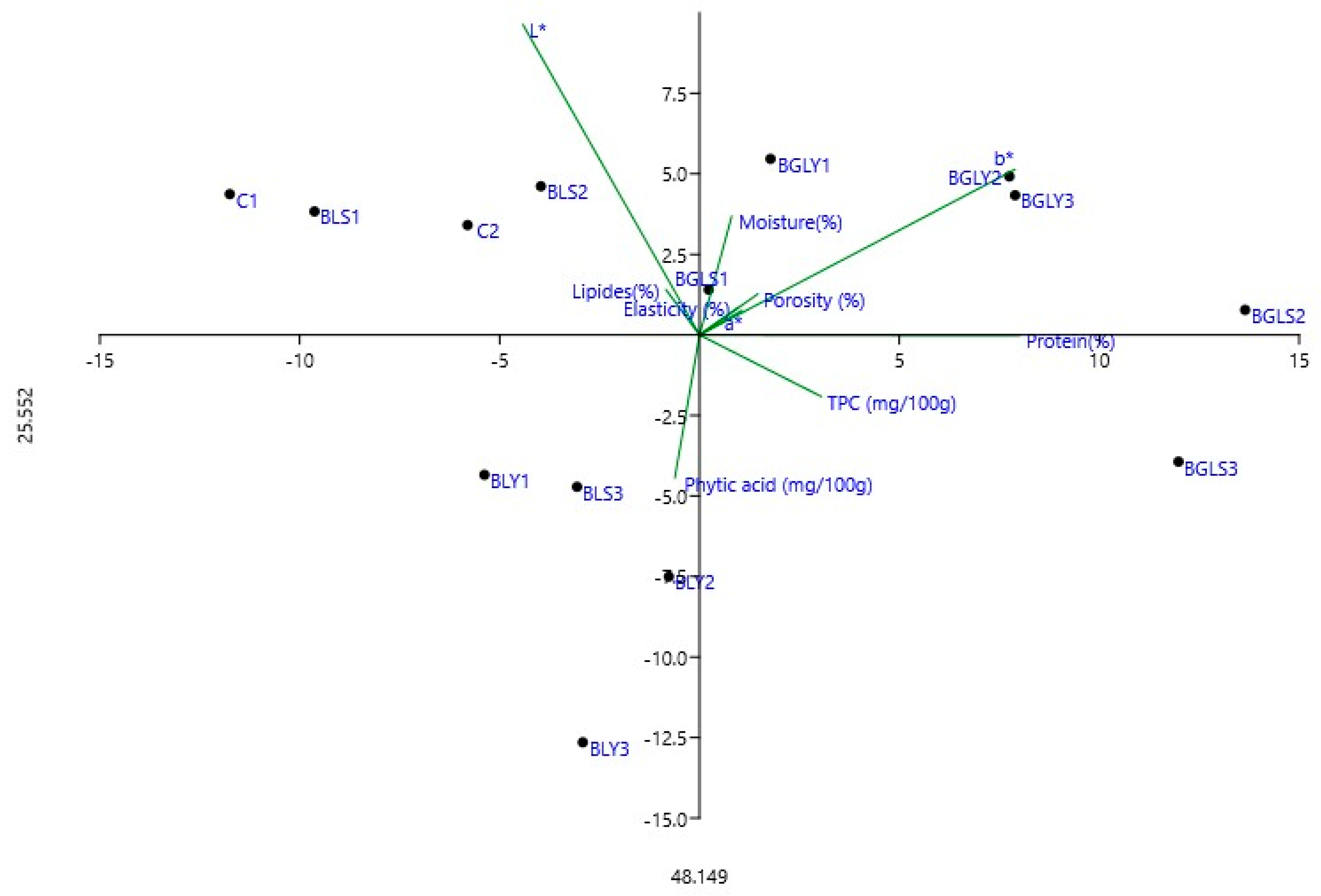
3.6. Pearson Correlations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pontoni S, Garzón A, Heinen G, Drago S. Lentil Flours as Sustainable Source of Proteins for the Food Industry. Sustainable Food Proteins. 2025;3(3):e70027. [CrossRef]
- Yılmaz Tuncel N, Polat Kaya H, Sakarya FB, Andaç AE, Korkmaz F, Ozkan G, Tuncel NB, Capanoglu E. The effect of germination on antinutritional components, in vitro starch and protein digestibility, content, and bioaccessibility of Phenolics and Antioxidants of Some Pulses. Food Science & Nutrition. 2025;13(5):e70103. [CrossRef]
- Cacak-Pietrzak G, Sujka K, Księżak J, Bojarszczuk J, Ziarno M, Studnicki M, Krajewska A, Dziki D. Assessment of physicochemical properties and quality of the breads made from organically grown wheat and legumes. Foods. 2024;13(8):1244. [CrossRef]
- Park YR, Kwon S-J, Kim JH, Duan S, Eom SH. Light-induced antioxidant phenolic changes among the sprouts of lentil cultivar. Antioxidants. 2024;13(4):399. [CrossRef]
- Dragomir C, Dossa S, Jianu C, Cocan I, Radulov I, Berbecea A, Radu F, Alexa E. Composite flours based on black lentil seeds and sprouts with nutritional, phytochemical and rheological impact on bakery/pastry products. Foods. 2025;14(2):319. [CrossRef]
- Islam MA, Islam S. Sourdough bread quality: Facts and Factors. Foods. 2024;13(13):2132.
- Alkay Z, Falah F, Cankurt H, Dertli E. Exploring the Nutritional Impact of Sourdough Fermentation: Its Mechanisms and Functional Potential. Foods. 2024;13(11):1732. [CrossRef]
- Reffai YM, Fechtali T. A critical review on the role of lactic acid bacteria in sourdough nutritional quality: Mechanisms, potential, and challenges. Applied Microbiology. 2025;5(3):74. [CrossRef]
- Leenhardt F, Levrat-Verny M-A, Chanliaud E, Rémésy C. Moderate decrease of pH by sourdough fermentation is sufficient to reduce phytate content of whole wheat flour through endogenous phytase activity. Journal of agricultural and food chemistry. 2005;53(1):98-102. [CrossRef]
- Plustea L, Negrea M, Cocan I, Radulov I, Tulcan C, Berbecea A, Popescu I, Obistioiu D, Hotea I, Suster G. Lupin (Lupinus spp.)-fortified bread: A sustainable, nutritionally, functionally, and technologically valuable solution for bakery. Foods. 2022;11(14):2067. [CrossRef]
- Amini Khoozani A, Kebede B, Birch J, Bekhit AE-DA. The effect of bread fortification with whole green banana flour on its physicochemical, nutritional and in vitro digestibility. Foods. 2020;9(2):152.
- Đurović V, Radovanović M, Mandić L, Knežević D, Zornić V, Đukić D. Chemical and microbial evaluation of biscuits made from wheat flour substituted with wheat sprouts. Food Science and Technology International. 2021;27(2):172-83. [CrossRef]
- Ungureanu-Iuga M, Atudorei D, Codină GG, Mironeasa S. Rheological approaches of wheat flour dough enriched with germinated soybean and lentil. Applied Sciences. 2021;11(24):11706. [CrossRef]
- Dossa S, Dragomir C, Plustea L, Dinulescu C, Cocan I, Negrea M, Berbecea A, Alexa E, Rivis A. Gluten-Free Cookies Enriched with Baobab Flour (Adansonia digitata L.) and Buckwheat Flour (Fagopyrum esculentum). Applied Sciences. 2023;13(23):12908. PubMed PMID: doi:10.3390/app132312908. [CrossRef]
- Dossa S, Neagu C, Lalescu D, Negrea M, Stoin D, Jianu C, Berbecea A, Cseh L, Rivis A, Suba M. Evaluation of the Nutritional, Rheological, Functional, and Sensory Properties of Cookies Enriched with Taro (Colocasia esculenta) Flour as a Partial Substitute for Wheat Flour. Foods. 2025;14(20):3526. [CrossRef]
- SR 91:2007; Romanian Standard for Bread CaBSMoAARSAB, Romania, 2007.
- Methods No. 110/1; ICC: International Association for Cereal Science and Technology: Vienna A, 2003.
- ISO 2171:2023; Cereals PaB-PDoTAYbIIOfSG, Switzerland, 2007.
- Latimer Jr GW. Official methods of analysis of AOAC International. (No Title). 2012.
- https://prod-docs.megazyme.com/documents/Assay_Protocol/K-PHYT_DATA.pdf.
- Dossa S, Negrea M, Cocan I, Berbecea A, Obistioiu D, Dragomir C, Alexa E, Rivis A. Nutritional, physico-chemical, phytochemical, and rheological characteristics of composite flour substituted by baobab pulp flour (Adansonia digitata L.) for bread making. Foods. 2023;12(14):2697. [CrossRef]
- Plustea L, Dossa S, Dragomir C, Cocan I, Negrea M, Obistioiu D, Poiana M-A, Voica D, Berbecea A, Alexa E. Comparative study of the nutritional, phytochemical, sensory characteristics and glycemic response of cookies enriched with lupin sprout flour and lupin green sprout. Foods. 2024;13(5):656. [CrossRef]
- https://www.konicaminolta.com/instruments/knowledge/color/pdf/color_communication.pdf.
- Fluerasu D, Negrea M, Neagu C, Dossa S, Jianu C, Lalescu D, Berbecea A, Cseh L, Cocan I, Misca C, Suba M, Muresan V, Tanislav A, Alexa E. Whey Valorization in Functional Jellies: A Nutritional and Technological Approach. Foods. 2025;14(18):3193. PubMed PMID: doi:10.3390/foods14183193. [CrossRef]
- Zhang L, Li Z, Qiao Y, Zhang Y, Zheng W, Zhao Y, Huang Y, Cui Z. Improvement of the quality and shelf life of wheat bread by a maltohexaose producing α-amylase. Journal of cereal science. 2019;87:165-71. [CrossRef]
- Atudorei D, Mironeasa S, Codină GG. Effects of germinated lentil flour on dough rheological behavior and bread quality. Foods. 2022;11(19):2982. [CrossRef]
- Klopsch R, Baldermann S, Voss A, Rohn S, Schreiner M, Neugart S. Bread enriched with legume microgreens and leaves—ontogenetic and baking-driven changes in the profile of secondary plant metabolites. Frontiers in chemistry. 2018;6:322. [CrossRef]
- Cardone G, D’Incecco P, Pagani MA, Marti A. Sprouting improves the bread-making performance of whole wheat flour (Triticum aestivum L.). Journal of the Science of Food and Agriculture. 2020;100(6):2453-9. [CrossRef]
- Sharma N, Sahu JK, Joshi S, Khubber S, Bansal V, Bhardwaj A, Bangar SP, Bal LM. Modulation of lentil antinutritional properties using non-thermal mediated processing techniques–A review. Journal of Food Composition and Analysis. 2022;109:104498. [CrossRef]
- Ouazib M, Dura A, Zaidi F, Rosell CM. Effect of partial substitution of wheat flour by processed (germinated, toasted, cooked) chickpea on bread quality2016. [CrossRef]
- Poutanen K, Flander L, Katina K. Sourdough and cereal fermentation in a nutritional perspective. Food microbiology. 2009;26(7):693-9. [CrossRef]
- Huchet V, Pavan S, Lochardet A, Divanac’h M, Postollec F, Thuault D. Development and application of a predictive model of Aspergillus candidus growth as a tool to improve shelf life of bakery products. Food microbiology. 2013;36(2):254-9. [CrossRef]
- Lim SM, Choo JM, Li H, O’rielly R, Carragher J, Rogers GB, Searle I, Robertson SA, Page AJ, Muhlhausler B. A high amylose wheat diet improves gastrointestinal health parameters and gut microbiota in male and female mice. Foods. 2021;10(2):220. [CrossRef]
- Benayad A, Taghouti M, Benali A, Aboussaleh Y, Benbrahim N. Nutritional and technological assessment of durum wheat-faba bean enriched flours, and sensory quality of developed composite bread. Saudi Journal of Biological Sciences. 2021;28(1):635-42. [CrossRef]
- Gan R-Y, Lui W-Y, Wu K, Chan C-L, Dai S-H, Sui Z-Q, Corke H. Bioactive compounds and bioactivities of germinated edible seeds and sprouts: An updated review. Trends in Food Science & Technology. 2017;59:1-14. [CrossRef]
- Katina K, Salmenkallio-Marttila M, Partanen R, Forssell P, Autio K. Effects of sourdough and enzymes on staling of high-fibre wheat bread. LWT-Food Science and Technology. 2006;39(5):479-91. [CrossRef]
- Graça C, Lima A, Raymundo A, Sousa I. Sourdough fermentation as a tool to improve the nutritional and health-promoting properties of its derived-products. Fermentation. 2021;7(4):246. [CrossRef]
- Priti, Mishra GP, Dikshit HK, T V, Tontang MT, Stobdan T, Sangwan S, Aski M, Singh A, Kumar RR. Diversity in phytochemical composition, antioxidant capacities, and nutrient contents among mungbean and lentil microgreens when grown at plain-altitude region (Delhi) and high-altitude region (Leh-Ladakh), India. Frontiers in Plant Science. 2021;12:710812. [CrossRef]
- Lopez HW, Leenhardt F, Coudray C, Remesy C. Minerals and phytic acid interactions: is it a real problem for human nutrition? International journal of food science and technology. 2002;37(7):727-39. [CrossRef]
- Greiner R, Konietzny U. Phytase for food application. Food Technology & Biotechnology. 2006;44(2).
- Gunjal M, Singh J, Kaur S, Nanda V, Ullah R, Iqbal Z, Ercisli S, Rasane P. Assessment of bioactive compounds, antioxidant properties and morphological parameters in selected microgreens cultivated in soilless media. Scientific Reports. 2024;14(1):23605. [CrossRef]
- Pathare PB, Opara UL, Al-Said FA-J. Colour measurement and analysis in fresh and processed foods: A review. Food and bioprocess technology. 2013;6(1):36-60. [CrossRef]
- Wrolstad RE, Smith DE. Color analysis. Food analysis: Springer; 2010. p. 573-86.
- Xue H, Zhao J, Wang Y, Shi Z, Xie K, Liao X, Tan J. Factors affecting the stability of anthocyanins and strategies for improving their stability: A review. Food Chemistry: X. 2024;24:101883. [CrossRef]
- Pirsaheb M, Fattahi N, Rahimi R, Sharafi K, Ghaffari HR. Evaluation of abamectin, diazinon and chlorpyrifos pesticide residues in apple product of Mahabad region gardens: Iran in 2014. Food Chemistry. 2017;231:148-55. doi: https://doi.org/10.1016/j.foodchem.2017.03.120. [CrossRef]
- Visciano P, Schirone M, Paparella A. An Overview of Histamine and Other Biogenic Amines in Fish and Fish Products. Foods. 2020;9(12):1795. PubMed PMID: doi:10.3390/foods9121795. [CrossRef]
- Wolter A, Hager AS, Zannini E, Galle S, Gänzle MG, Waters DM, Arendt EK. Evaluation of exopolysaccharide producing Weissella cibaria MG1 strain for the production of sourdough from various flours. Food Microbiology. 2014;37:44-50. doi: https://doi.org/10.1016/j.fm.2013.06.009. [CrossRef]
- Vilarinho F, Sendón R, van der Kellen A, Vaz MF, Silva AS. Bisphenol A in food as a result of its migration from food packaging. Trends in Food Science & Technology. 2019;91:33-65. doi: https://doi.org/10.1016/j.tifs.2019.06.012. [CrossRef]
- Duenas M, Hernandez T, Estrella I, Fernandez D. Germination as a process to increase the polyphenol content and antioxidant activity of lupin seeds (Lupinus angustifolius L.). Food chemistry. 2009;117(4):599-607. [CrossRef]
- Fernández-Peláez J, Paesani C, Gómez M. Sourdough technology as a tool for the development of healthier grain-based products: An update. Agronomy. 2020;10(12):1962. [CrossRef]
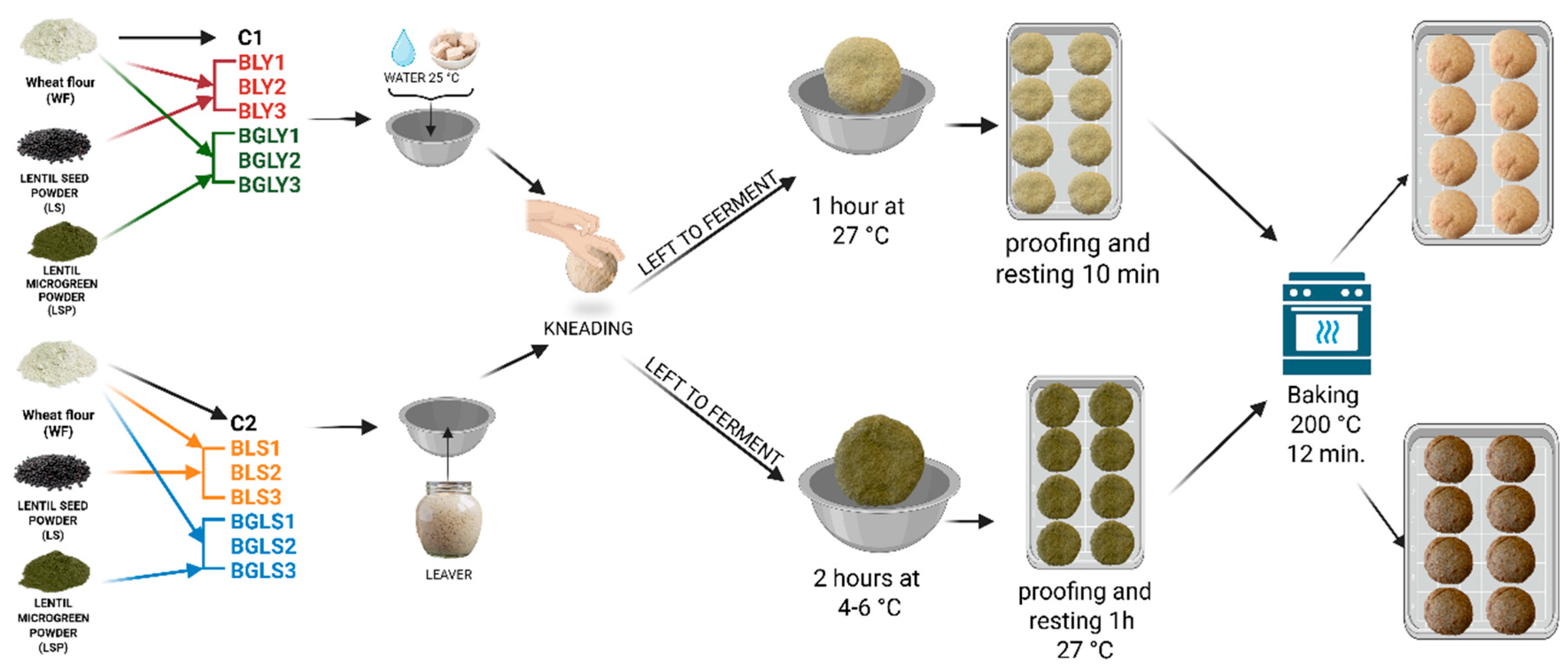
| Bread sample | Flour | Cod sample |
|---|---|---|
| Yeast control bread | 100% wheat flour | C1 |
| Sourdough control bread | 100% wheat flour | C2 |
| Yeast lentil seeds bread 10% | 10% lentils seeds + 90% wheat flour | BLY1 |
| Yeast lentil seeds bread 20% | 20% lentils seeds + 80% wheat flour | BLY2 |
| Yeast lentil seeds bread 30% | 30% lentils seeds + 70% wheat flour | BLY3 |
| Sourdough lentil seeds bread 10% | 10% lentils seeds + 90% wheat flour | BLS1 |
| Sourdough lentil seeds bread 20% | 20% lentils seeds + 80% wheat flour | BLS2 |
| Sourdough lentil seeds bread 30% | 30% lentils seeds + 70% wheat flour | BLS3 |
| Yeast germinated lentil bread 2.5% | 2.5% germinated lentilss + 97,5% wheat flour | BGLY1 |
| Yeast germinated lentil bread 5% | 5% germinated lentils + 95% wheat flour | BGLY2 |
| Yeast germinated lentil bread 7.5% | 7.5% l germinated lentils + 92,5% wheat flour | BGLY3 |
| Sourdough germinated lentil bread2.5% | 2.5% germinated lentils + 97,5% wheat flour | BGLS1 |
| Sourdough germinated lentil bread 5% | 5% germinated lentils + 95% wheat flour | BGLS2 |
| Sourdough germinated lentil bread 7.5% | 7.5% germinated lentils + 92,5% wheat flour | BGLS3 |
| Ingredients | Wheat flour (kg) | Lentil flour (kg) | Germinated lentil powder (kg) | Active dry baker’s yeast (kg) | Sourdough (kg) | Salt (Kg) | Water (mL) |
|---|---|---|---|---|---|---|---|
| C1 | 1 | - | - | 0.030 | - | 0.020 | 500 |
| BLY1 | 0.900 | 0.100 | - | 0.030 | - | 0.020 | 500 |
| BLY2 | 0.800 | 0.200 | - | 0.030 | - | 0.020 | 500 |
| BLY3 | 0.700 | 0.300 | - | 0.030 | - | 0.020 | 500 |
| BGLY1 | 0.975 | - | 0.025 | 0.030 | - | 0.020 | 500 |
| BGLY2 | 0.950 | - | 0.050 | 0.030 | - | 0.020 | 500 |
| BGLY3 | 0.925 | - | 0.075 | 0.030 | - | 0.020 | 500 |
| C2 | 1 | - | - | - | 0.200 | 0.020 | 500 |
| BLS1 | 0.900 | 0.100 | - | - | 0.200 | 0.020 | 500 |
| BLS2 | 0.800 | 0.200 | - | - | 0.200 | 0.020 | 500 |
| BLS3 | 0.700 | 0.300 | - | - | 0.200 | 0.020 | 500 |
| BGLS1 | 0.975 | - | 0.025 | - | 0.200 | 0.020 | 500 |
| BGLS2 | 0.950 | - | 0.050 | - | 0.200 | 0.020 | 500 |
| BGLS3 | 0.925 | - | 0.075 | - | 0.200 | 0.020 | 500 |
| Steps | Process | Parameters | |
|---|---|---|---|
| Yeast fermentation | Active dry baker’s yeast (Saccharomyces cerevisiae) was dissolved in water prior to mixing | Yeast pH : 4.25; water temperature : 25 °C Time : 30 min |
|
| Soudough fermentation | Sourdough-fermented samples were prepared using an active natural sourdough starter containing lactic acid bacteria (mainly Lactobacillus spp., Lactiplantibacillus, Levilac-tobacillus). | Sourdough pH : 4.3 Sourdough Total Titratable Acidity (TTA): 5.1 mL NaOH/10 g Sourgdough Temperature : 25 °C Time : 24 h |
|
| Mixing | Dough mixing was carried out in two stages: initial mixing to ensure homogenization, followed by intensive mixing to promote gluten development. Salt was added during the second mixing stage. | 1rst stage | 2nd Stage |
| Speed : 80 rpm Time : 4 min |
Speed : 160 rpm Time : 4 min |
||
| Dough fermentation with yeast | Yeast-fermented doughs were processed according to a conventional fermentation procedure (direct method without sourdough | Time : 1h Temperature : 27 °C humidity : 70% |
|
| Dough fermentation with soudough | Sourdough doughs underwent a prolonged fermentation step to enhance microbial activity and biochemical transformations. | Time :2h Temperature : 4-5 °C humidity : 70% |
|
| Proofing | After fermentation, the doughs were kneaded, divided, scaled, and manually molded. Final proofing was performed at different parameters for yeast and sourdough | Yeast samples | Sourdough samples |
| Time : 1h Temperature :27 °C humidity : 65% |
Time : 10min Temperature : 27 °C humidity : 70% |
||
| Baking | Baking was carried out in bakery oven, followed by cooling at room temperature prior to analysis. | Temperature : 200 °C Time : 12min |
|
| Parameters | Methods | References |
|---|---|---|
| Moisture | ICC Standard Methods (2003) | [17] |
| Protein content* (%) | ICC Standard Methods (2003) | [17] |
| Ash content* (%) | ISO Method No. 2171:2007 | [18] |
| Fat content* (%) | AOAC Official Method (2000) | [19] |
| Carbohydrate content (g/100 g) | Calculated by difference: 100 − (moisture + ash + proteins + fats) | [10] |
| Energy value (kcal/100 g) | Calculated using the formula: Energy value = 4 x proteins + 4 x carbohydrates + 9 x lipids | [14] |
| Bread samples | Elasticity (%) | Porosity (%) | Height/Diameter (H/D) |
|---|---|---|---|
| Bread fermented with yeast | |||
| C1 | 91.220 ± 0.200a | 66.087 ± 0.153a | 0.526 ± 0.011a |
| BLY1 | 91.857 ± 0.153a | 67.790 ± 0.173a | 0.555 ± 0.009a |
| BLY2 | 92.267 ± 0.050a | 68.100 ± 0.106a | 0.569 ± 0.005a |
| BLY3 | 91.440 ± 0.115a | 66.177 ± 0.055a | 0.534 ± 0.014a |
| BGLY1 | 93.073 ± 0.058a | 68.340 ± 0.265a | 0.578 ± 0.012a |
| BGLY2 | 94.153 ± 0.153a | 69.680 ± 0.178a | 0.588 ± 0.004a |
| BGLY3 | 91.625 ± 0.132a | 66.610 ± 0.204a | 0.577 ± 0.011a |
| Bread fermented with sourdough | |||
| C2 | 92.140 ± 0.132a | 67.607 ± 0.202a | 0.546 ± 0.007a |
| BLS1 | 92.827 ± 0.101a | 68.640 ± 0.150a | 0.570 ± 0.010a |
| BLS2 | 93.647 ± 0.252a | 71.173 ± 0.153b | 0.586 ± 0.008a |
| BLS3 | 92.493 ± 0.050a | 67.220 ± 0.100a | 0.558 ± 0.011a |
| BGLS1 | 93.727 ± 0.095a | 69.760 ± 0.115a | 0.588 ± 0.004a |
| BGLS2 | 94.920 ± 0.075a | 72.607 ± 0.202b | 0.590 ± 0.007a |
| BGLS3 | 92.763 ± 0.104a | 68.763 ± 0.056a | 0.574 ± 0.020a |
| Samples | Nutritional characteristics | ||||||
|---|---|---|---|---|---|---|---|
| Moisture | Ash | Protein | Fat | Carbohydrates | Energy value (kcal/100g) |
||
| (%) | (%) | (%) | (%) | (g/100g) | |||
| Composite bread with yeast fermentation | |||||||
| C1 | 35.01±0.68a | 0.89±0.02a | 12.71±0.99a | 2.61±0.03 a | 48.77±0.29a | 269.47±2.77a | |
| BLY1 | 32.69±0.45b | 1.18±0.08b | 17.40±2.34b | 1.07±0.03b | 47.66±2.78a | 269.87±1.97a | |
| BLY2 | 29.45±0.23c | 1.52±0.12b | 18.16±3.00b | 0.75±0.53b | 50.12±2.82a | 279.87±1.92b | |
| BLY3 | 26.28±0.11b,c | 2.23±0.28c | 19.72±0.11b | 0.45±0.10b | 51.32±0.40a | 288.21±1.95b | |
| BGLY1 | 33.53±0.60b | 0.69±0.42a | 21.33±0.51c | 1.22±0.06b | 43.23±1.12a | 269.22±3.06a | |
| BGLY2 | 32.80±5.59b | 1.23±0.20b | 24.44±0.41c | 1.82±0.09c | 39.71±5.77a | 272.98±22.68b | |
| BGLY3 | 31.64±0.32b | 1.99±0.21c | 26.53±0.87c | 1.93±0.04c | 37.91±0.80a | 275.13±0.58b | |
| Composite bread with sourdough fermentation | |||||||
| C2 | 29.31±0.56a | 1.13±0.06a | 16.81±2.95a | 2.85±0.05a | 49.89±2.93a | 292.52±2.65a | |
| BLS1 | 39.70±0.29b | 1.46±0.46b | 14.50±0.13a | 3.91±0.07b | 40.42±0.78b | 254.90±2.61b | |
| BLS2 | 41.72±0.46c | 1.47±0.25b | 18.23±0.29b | 2.08±0.03a | 36.50±0.47c | 237.71±2.52c | |
| BLS3 | 41.03±1.03c | 2.67±0.25c | 20.57±0.05c | 1.07±0.97c | 34.66±2.10c | 230.57±3.16c | |
| BGLS1 | 35.72±0.63bc | 1.14±0.07a | 23.00±0.40c | 0.56±0.06c | 47.96±13.51a | 288.93±55.02a | |
| BGLS2 | 38.23±0.29b | 1.15±0.12a | 26.81±2.67d | 0.89±0.02c | 33.22±2.92c | 248.13±1.30c | |
| BGLS3 | 38.53±0.13b | 1.19±0.18a | 29.18±0.50d | 1.03±0.07c | 30.37±0.44c | 247.47±1.67c | |
| Samples | L* | a* | b* | C* | h° |
|---|---|---|---|---|---|
| Composite bread with yeast fermentation | |||||
| C1 | 64.58±0.21a | 0.50±0.02 a | 13.25±0.78 a | 13.26±0.13 a | 87.84±0.10 a |
| BLY1 | 56.09±0.45a | 0.31±0.04a | 12.21±0.41 a | 12.21±0.04 a | 88.55±0.14 a |
| BLY2 | 52.89±1.12a | 1.07±0.01 a | 17.70±1.12 a | 17.73 ±0.21a | 86.54±0.20 a |
| BLY3 | 50.13±1.41a | 0.54±0.04 a | 11.29±1.47 a | 11.30 ±0.07a | 87.26±0.13a |
| BGLY1 | 61.96±0.47a | 1.77±0.14 a | 22.77±1.96 a | 22.84±0.09 a | 85.56±0.20 a |
| BGLY2 | 60.18±0.05a | 3.16±0.69 a | 24.77±1.65 a | 24.97±0.15 a | 82.73±0.08 a |
| BGLY3 | 60.07±0.31a | 2.74±0.12 a | 24.95±1.34 a | 25.10±0.23 a | 83.73±0.10 a |
| Mean | 57.99 | 1.44 | 18.13 | 18.20 | 86.03 |
| SD | 5.16 | 1.14 | 6.03 | 6.10 | 2.15 |
| Composite bread with sourdough fermentation | |||||
| C2 | 62.32±0.10a | 1.22±0.7a | 17.81±0.12a | 17.85±0.10a | 86.08±0.45a |
| BLS1 | 62.70±1.02a | 0.17±0.9b | 12.22±0.98a | 12.22±0.35a | 89.20±0.12a |
| BLS2 | 62.35±1.32a | 1.04±0.10a | 15.76±0.65a | 15.79±1.25a | 86.22±1.98a |
| BLS3 | 54.89±0.14b | 0.67±1.54a | 11.46±0.10a | 11.48±2.14a | 86.65±2.79a |
| BGLS1 | 59.67±0.21c | 0.37±2.10b | 15.91±0.96a | 15.91±1.98a | 88.67±1.22a |
| BGLS2 | 51.34±0.28d | 1.53±1.87a | 27.71±0.10b | 27.75±2.73b | 86.84±1.63a |
| BGLS3 | 49.30±0.05d | 1.66±1.65a | 21.90±0.32b | 21.96±1.32b | 85.67±2.36a |
| Mean | 57.51 | 0.95 | 17.54 | 17.57 | 87.05 |
| SD | 5.63 | 0.57 | 5.68 | 5.70 | 1.35 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





