Submitted:
06 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mechanisms of Biosurfactant-Based MEOR in Reservoirs
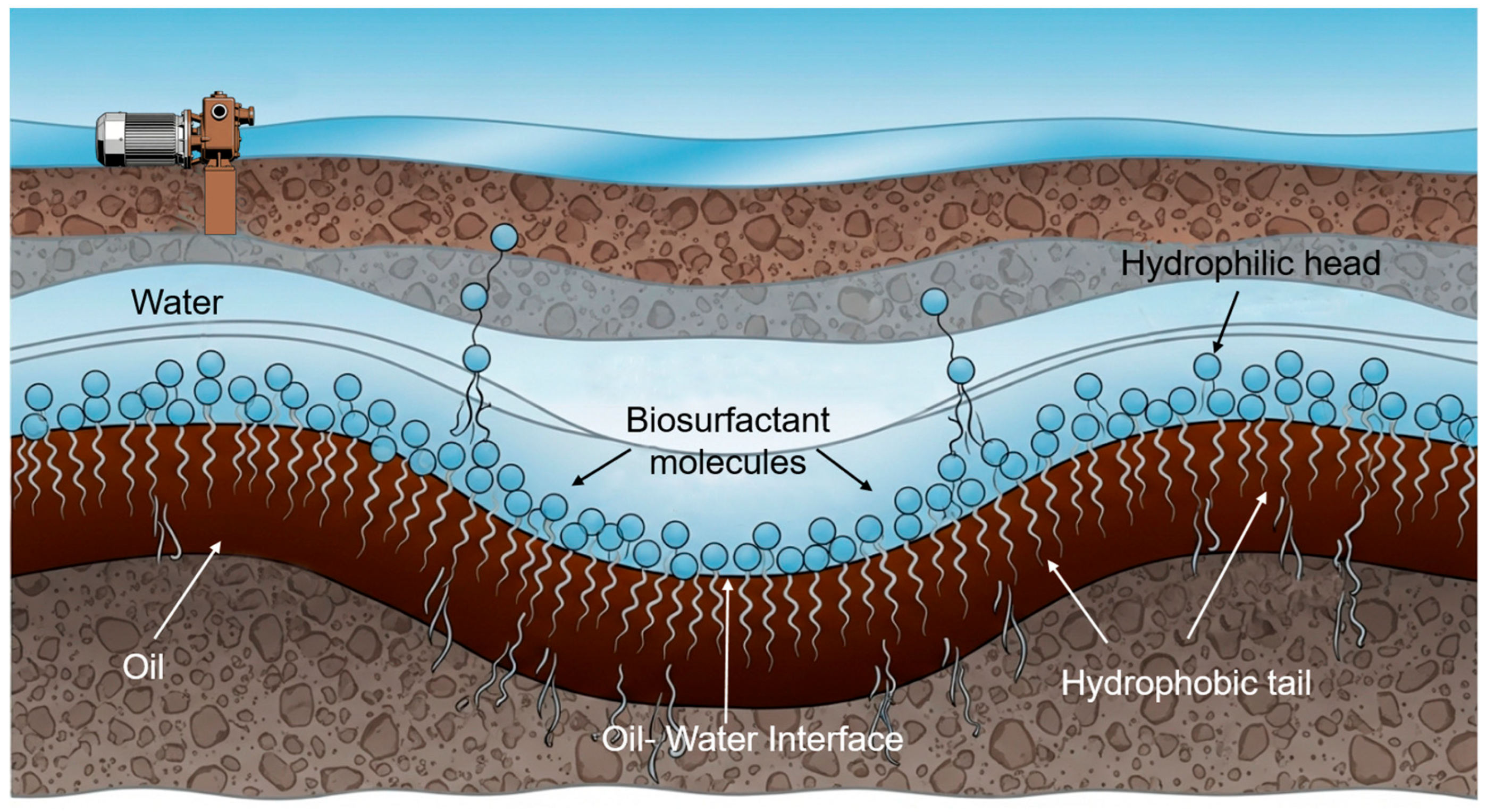
2.1. Interfacial Tension Reduction
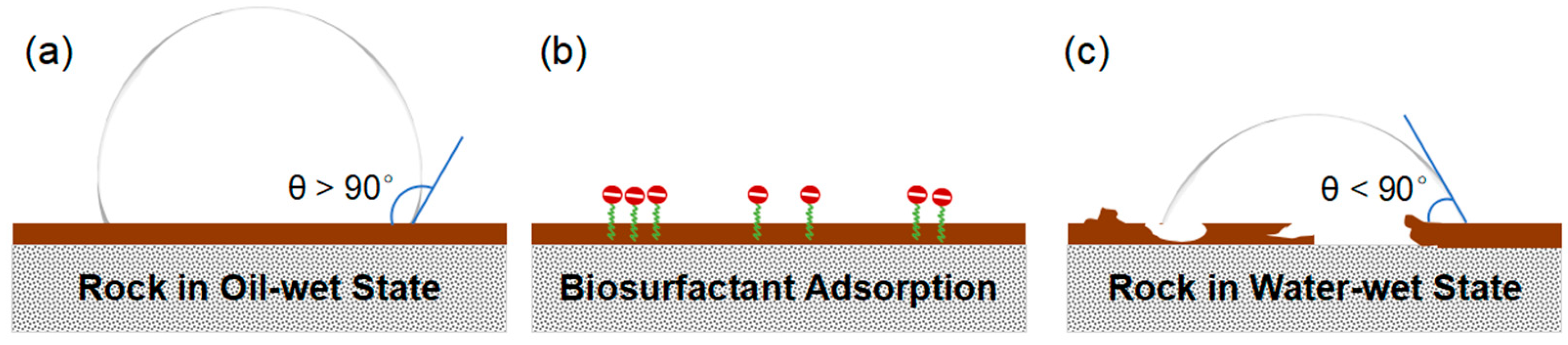
2.2. Wettability Alteration: The Primary Driver of Recovery
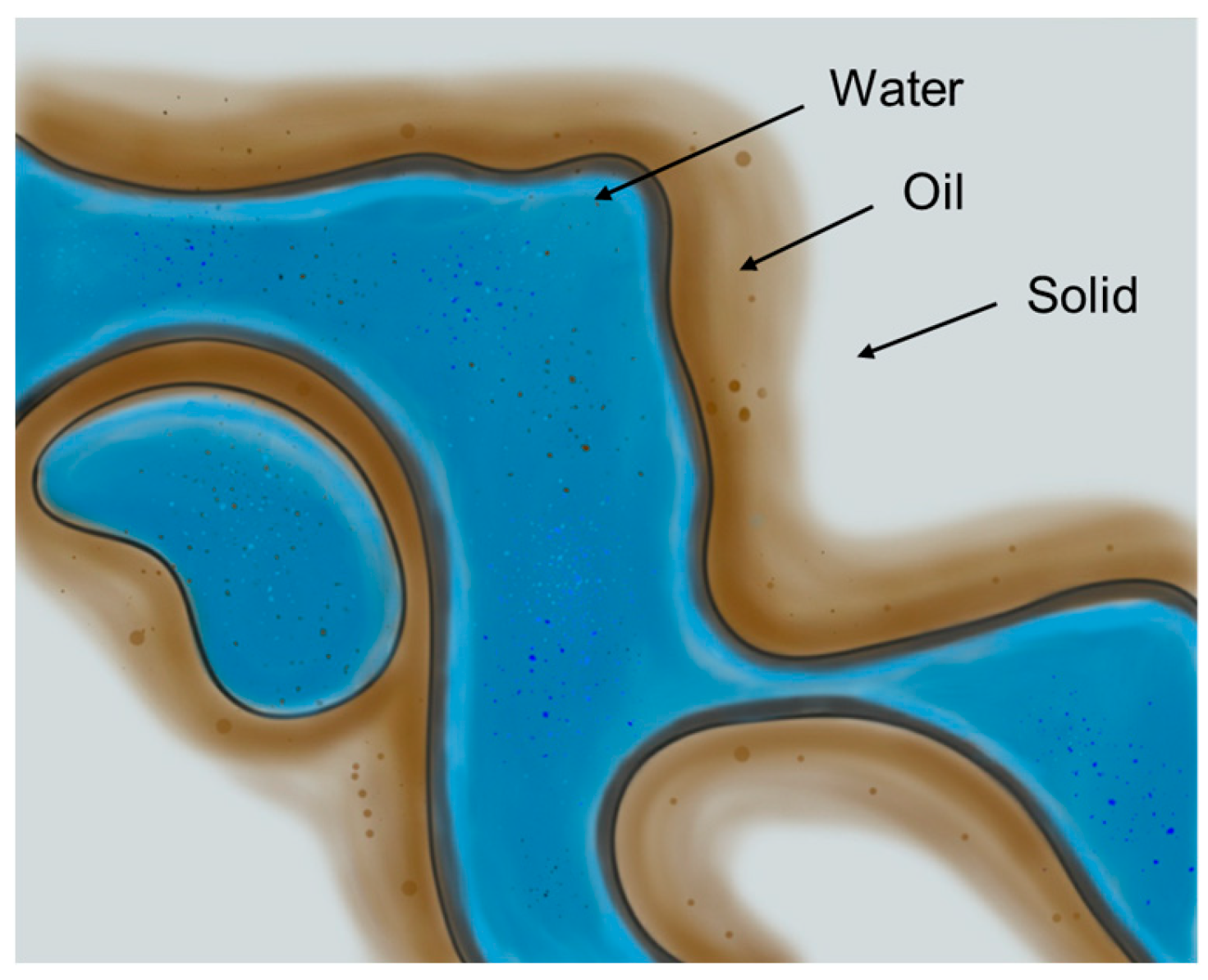
2.2.1. Mechanisms of Wettability Reversal
- Initial State: In an oil-wet system, polar organic components from crude oil firmly adhere onto the rock surface, creating a hydrophobic layer. Oil spreads across this surface, the water phase contact angle (θ) is greater than 90°.
- Biosurfactant Introduction: When a biosurfactant solution is introduced, the amphiphilic molecules migrate to the interfaces.
- Adsorption and Displacement: The biosurfactant molecules then adsorb onto the rock surface. The specific interaction depends on the nature of the biosurfactant and the rock mineralogy. The hydrophobic tail of the biosurfactant can interact with the adsorbed oil layer via hydrophobic interactions, while the hydrophilic head has an affinity for the water phase. In many cases, the biosurfactant can insert itself between the rock surface and the adsorbed oil film, effectively "lifting off" or displacing the organic material.
- Formation of a Water-wet Layer: As biosurfactants accumulate at the surface, they orient themselves with their hydrophilic head groups facing outwards into the aqueous phase. This creates a new, stable hydrophilic layer on the rock surface.
- Final State: The rock now preferentially contacts water. The oil film retracts into discrete droplets, and the contact angle drops below 90°. Consequently, water can spontaneously penetrate finer pore networks, thereby mobilizing the trapped oil and improving the overall sweep efficiency of the waterflood. A study showed that wettability alteration alone could be responsible for an oil production increase of over 50% in a mature well, boosting overall recovery from around 32% of OOIP to over 62% in MEOR cases [75].
2.2.2. Adsorption Behaviors on Different Rock Surfaces
- Carbonate Surfaces (Limestone, Dolomite): Carbonate rocks are typically composed of calcite (CaCO3) and dolomite (CaMg(CO3)2). In reservoir brines, their surfaces often carry a positive charge. Anionic biosurfactants (which are common), such as the glycolipid rhamnolipid and the lipopeptide surfactin, possess negatively charged carboxylate groups (-COO-) in their hydrophilic heads. This leads to strong electrostatic attraction between the biosurfactant and the positively charged carbonate surface, promoting robust adsorption and effective wettability alteration [43]. However, this interaction can be complex. High salinity, particularly the presence of divalent cations (Ca2+, Mg2+) in seawater or formation brine, can screen the electrostatic charges or lead to biosurfactant precipitation, potentially inhibiting its effectiveness.
- Sandstone Surfaces (Quartz, Feldspars, Clays): Sandstone reservoirs are primarily composed of quartz (SiO2), with varying amounts of feldspars and clays. Under typical reservoir pH conditions, these silicate minerals carry a negative surface charge. Consequently, anionic biosurfactants would experience electrostatic repulsion, hindering their adsorption. In such cases, adsorption is driven by other forces like hydrophobic interactions between the biosurfactant tail and any adsorbed organic matter, or van der Waals forces. A major challenge in sandstone reservoir is the significant biosurfactant loss through adsorption onto clays, which possess high surface area and complex charge distribution. Non-ionic or cationic biosurfactants may be more effective in these formations, although they are less commonly studied for MEOR. Despite this challenge, biosurfactants demonstrably alter sandstone wettability: contact angle measurements on sandstone slices treated with a microbial consortium show a decrease from >120°(oil-wet) to 60°(water-wet) within 3–4 days, corresponding to a huge capillary pressure change of ~5000 Pa sufficient to increase oil recovery via spontaneous imbibition [44]. This mechanistic gap was later addressed by Zihui Chen et al. [76], whose AFM and molecular simulation study elucidated the distinct actions of two key biosurfactants, providing a quantitative and structural explanation for their differing efficacies. They found that rhamnolipid reduces the adhesion force between oil and glass by adsorbing onto the oil film and presenting hydrophilic head groups outward, thereby lowering surface hydrophobicity to create a weakly water-wet state. In contrast, surfactin can peel the oil film away from the rock surface and reverse the interaction force between oil and a glass surface from attractive to repulsive, leaving a strongly water-wet rock surface. The more effective and efficient wettability alteration capability of surfactin stems from its much higher adsorption energy (-38.01 eV) compared to rhamnolipid (-12.23 eV) on hydrophobic surfaces. This difference of the adsorption energy is attributed to their different molecular structure: the longer alkyl chain (C12) of surfactin strengthens hydrophobic-hydrophobic interactions, whereas shorter tails (C7) and the presence of a carboxyl group of rhamnolipid diminish its hydrophobicity and adsorption stability. This fundamental insight confirms that overcoming sandstone's mineralogical constraints requires biosurfactants with molecular structures specifically tailored for strong hydrophobic adsorption—directly linking nanoscale interactions to macroscopic recovery efficacy, which is critical for designing effective MEOR strategies.
2.3. Emulsification and Mobility Control
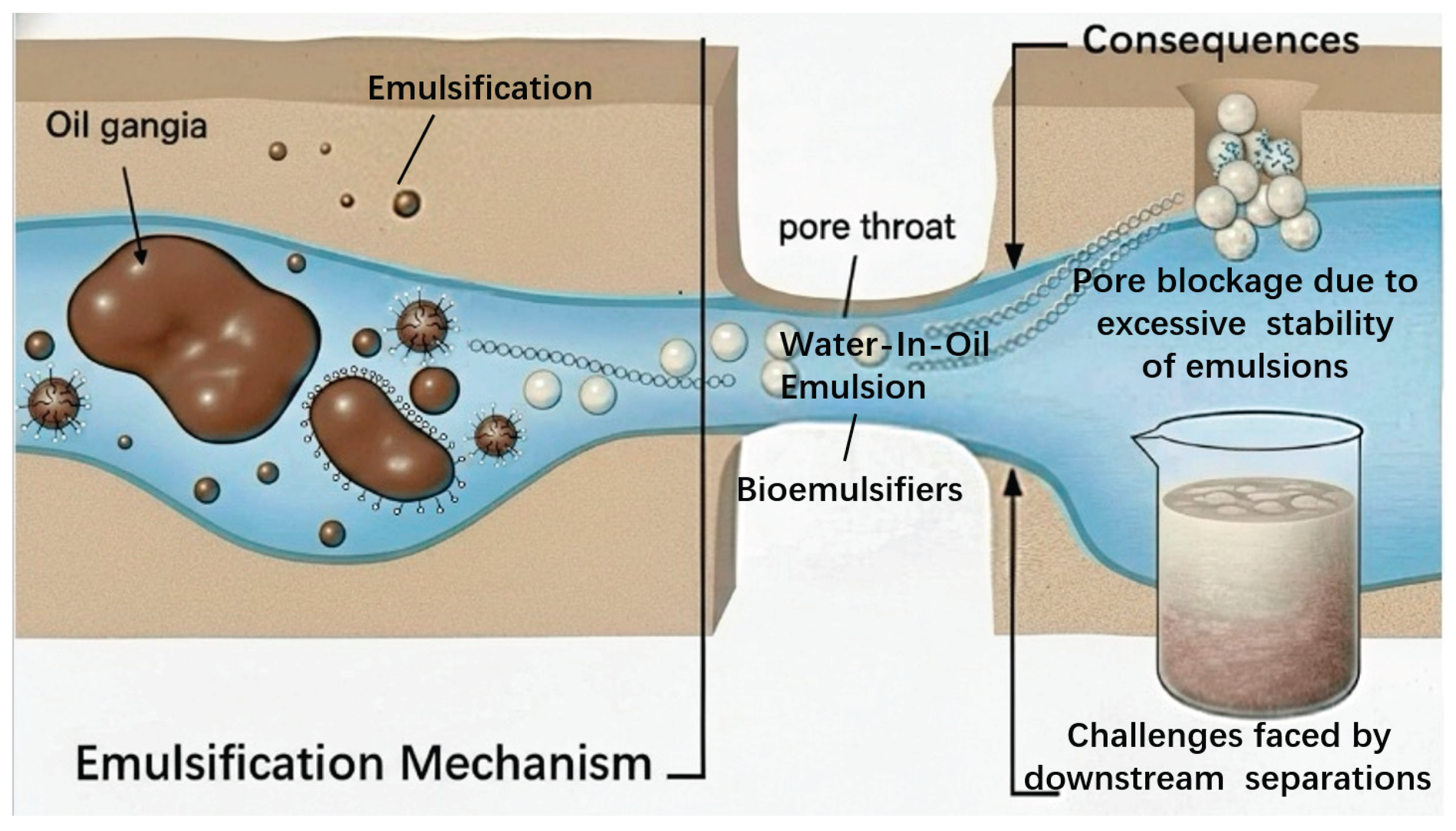
- Emulsification: By drastically lowering IFT, biosurfactants can promote the formation of emulsions, typically O/W emulsions. These emulsions break down large, continuous oil ganglia into smaller, mobile droplets that can be more easily transported through the porous medium. High-MW polymeric biosurfactants, often called bio-emulsifiers, are particularly effective at creating stable emulsions that can be flushed out of the reservoir [55]. However, the formation of overly stable emulsions can be detrimental, causing pore plugging and posing challenges for downstream oil-water separation facilities.
- Mobility Control: A successful waterflood requires favorable mobility control, meaning the displacing fluid (water) should’t advance much faster than the displaced fluid (oil). An unfavorable viscosity ratio leads to ”viscous fingering”, where water bypasses large regions of oil, severely reducing sweep efficiency [77]. During in-situ MEOR applications, biosurfactants are usually not the only product generated by microorganisms, biopolymers like xanthan gum and scleroglucan can also be produced. The co-produced biopolymers improve the overall rheological behavior of the injected fluid via enhancing the viscosity of the injected water [78,79]. This enhances the mobility ratio and leads to a more stable displacement front and a much higher volumetric sweep efficiency, ensuring that the mobilized oil is effectively pushed towards the production wells. The synergy between biosurfactants mobilizing the oil and biopolymers improving the sweep is a powerful combination for maximizing recovery [4,80].
3. Biosurfactant Categories and Formulations
| Biosurfactant Class | Sub-Type Example | Producing Microorganism (Typical) | Key Structural Features | Properties & MEOR Relevance |
| Glycolipids | Rhamnolipids | Pseudomonas aeruginosa | One or two rhamnose sugar molecules linked to one or two β-hydroxy fatty acid chains. Anionic. |
Excellent IFT reduction. Proven wettability alteration capability. High stability in a wide range of T, pH, and salinity [89]. |
| Sophorolipids | Starmerella bombicola (Yeast) |
Sophorose (a disaccharide) linked to a long-chain hydroxy fatty acid. Can be lactonic (cyclic) or acidic (linear). Non-ionic/Anionic. |
Good emulsifying properties. Production yields are often high. Potential for cost-effective EOR applications [90]. |
|
| Lipopeptides | Surfactin | Bacillus subtilis | A cyclic lipopeptide of 7 amino acids linked to a C12-C16 β-hydroxy fatty acid chain. Anionic. |
Extremely high surface activity; one of the most effective biosurfactants known. Reduces surface tension of water to ~27 mN/m [3]. Good thermal stability, but can be sensitive to divalent cations/high salinity [43]. |
| Lichenysin | Bacillus licheniformis | Similar structure to surfactin; cyclic heptapeptide linked to a β-hydroxy fatty acid. Anionic. |
High thermal and pH stability. Particularly effective and stable in high-salinity environments, making it a strong candidate for offshore EOR [91]. |
3.1. Glycolipids
3.1.1. Rhamnolipids
3.1.2. Sophorolipids
3.2. Lipopeptides
3.2.1. Surfactin
3.2.2. Lichenysin
3.3. High Molecular Weight Bioemulsifiers
3.4. Formulation of Biosurfactants
4. Synergistic Biopolymer and Bio-Complex Formulations
4.1. Synergy with Biopolymers: Integrating Pore-Scale Mobilization with Macroscopic Sweep
- Role of Biopolymers: This macroscopic challenge is addressed by co-injecting biosurfactants with viscosity-enhancing biopolymers, such as xanthan gum or scleroglucan. These are high-MW polysaccharides produced by microorganisms—xanthan gum by the bacterium Xanthomonas campestris and scleroglucan by the fungus Sclerotium rolfsii., respectively. By significantly thickening the aqueous phase at low concentrations, they function as highly effective viscosifiers, improve the oil-water mobility ratio and establish a stable displacement front [108,109]. Their excellent shear-thinning behavior ensures injectability, while their relative stability at reservoir temperatures underpins their widespread use in EOR for reliable mobility control [110,111,112].
- Synergistic Mechanism: The composite system of biosurfactants and biopolymers creates a synergistic “release-and-sweep” mechanism. In the combined flooding process, biosurfactants work at the leading edge by reducing IFT and altering wettablility to remove the aged oil from rock surfaces, then biopolymers enhance the mobility of the displaced oil toward production well by increasing the viscosity of the injected fluid [113]. This synergy enhances both pore-scale displacement and reservoir-scale sweep efficiency, yielding significantly EOR results than using either agent alone [112,113,114].
4.2. Synergy with Nanoparticles and Chemicals: Augmenting Interfacial Activity and Stability
- Biosurfactant-Nanoparticle Hybrids: Recently, the combination of biosurfactants with nanoparticles (e.g., silica, ZnO) has become a research focus, as nanoparticles can synergistically improve the interfacial performance at both fluid-rock and fluid-fluid interfaces to a higher level [116,117]. Studies have shown that nanoparticles and biosurfactants could co-adsorb, form a denser and more resilient film, thus achieving ultra-low IFT [118,119]. Azarshin et al. revealed that nanoparticles can generate structural disjoining pressure to peel oil films from rock surfaces, which accounts for their highly effective wettability alteration ability [120]. In the meantime, biosurfactants act as dispersants, preventing nanoparticles from aggregation and ensuring their propagation in deep reservoir. Further, Yulong Liu et al. synthesized a promising biological nanocomposite fluid for offshore oil field application with nano-Fe3O4, nano-Ag and biopolymer sodium alginate, and found that nanoparticles can interact with polymer chains in this tertiary formulations, enhancing the thermal and mechanical stability of the entire displacing system [121]. The outstanding synergy effect was also confirmed by a recent work by Amr Gazem et al., who integrated ZnO nanoparticles with a rhamnolipid/sophorolipid blend and xanthan gum, which achieved 95.14% oil recovery at 70 °C—significantly outperforming the the base biosurfactant–polymer formulation without nanoparticles (80.94%) and brine flooding (58.34%) [117].
- Bio-based Alkali-Surfactant-Polymer (Bio-ASP) Formulations: Incorporating biosurfactants into the established ASP flooding framework creates an environmentally optimized Bio-ASP system. Adding a low-concentration of alkali (e.g., sodium carbonate) can generate in-situ soaps with the biosurfactant to achieve ultralow IFT. An IFT of 0.02 mN/m was achieved by Taher Al-Ghailani et al., with 0.9% (w/v) alkali added into a 20-times diluted biosurfactant solution. A synergy effect was also observed in altering wettability and reducing the adsorption loss of the biosurfactant onto negatively charged rock surfaces. Consequently, Bio-ASP significantly improves chemical utilization efficiency, owing to these combined advantages, making it a highly promising EOR strategy [113,122].
4.3. The Synergistic Outcome and Potential
5. Key Challenges for Biosurfactant Application in Subsurface Reservoirs
5.1. Resilience Under Extreme Reservoir Environments
- High Temperature: In deep formations, reservoir temperatures can easily exceed 70°C and even surpass 120°C. Such heat can denature the proteinaceous components of lipopeptides or degrade glycolipid structures, leading to irreversible loss of surface activity. Therefore, it is necessary to screen and engineer thermotolerant strains. Encouragingly, many biosurfactants, especially lipopeptides from Bacillus species, display innate thermal robustness that often surpasses synthetic surfactants. Bo Wu et al. found that lipopeptide produced by Bacillus subtilis strain kept excellent surface activity at 120 °C and pH 5-12 [54]. Marcela Nunes Argentin et al. isolated bacterial strain Ar70C7-2 from rock of a deep offshore Brazilian oil reservoir and obtained the produced biosurfactant, which showed splendid emulsification and IFT reduction ability and maintained its stability at temperature ranging from -18 to 121°C [3,125]. Surfactin from Bacillus tequilensis was also reported to show negligible decay in interfacial tension reduction after 10 days of aging at 90°C [99].
- High Salinity: Formation water is often highly saline, with total dissolved solids frequently exceeding 100,000 ppm (10% salinity) and sometimes reaching saturation levels (>20%). High salt concentration can screen electrostatic repulsions, especially the presence of divalent cations (e.g., Ca2+, Mg2+) can precipitate anionic biosurfactants by forming insoluble salts and remove them from solution [37,43,115,126]. High ionic strength can also alter the HLB of surfactant, thus impairing its performance in IFT reduction and wettability alteration. Luckily, like biosurfactants produced by thermophilic microorganisms can endure high temperature, biosurfactants produced by halophilic microorganisms often exhibit exceptional salt tolerance ability. Xiaotong Wang et al. Obtained a biosurfactant from Bacillus halotolerans and demonstrated it can maintain high activity in salinities up to 21% NaCl [127]. Notably, tolerance is not universal; the stability of common biosurfactants like rhamnolipid, for example, has been observed to decrease with increasing salinity [128]. Therefore, screening or engineering microbial strains specifically adapted to target reservoir conditions is crucial.
- pH Fluctuations: Reservoir pH can vary, though it is typically near-neutral to slightly alkaline. The activity of biosurfactants, particularly those with ionizable functional groups like carboxylates or amines, is pH-dependent. At low pH, for instance, the carboxylate groups of anionic biosurfactants become protonated, reducing their solubility in water and diminishing their surface activity [129,130]. The ideal biosurfactant should have a broad pH stability range that encompasses the conditions of the target reservoir.
5.2. Adsorption and Loss
- (1)
- Rock Type and Mineralogy
- Sandstone Reservoirs: Composed primarily of quartz, sandstone surfaces are negatively charged under typical reservoir conditions. This results in weak electrostatic adsorption of anionic biosurfactants (e.g., rhamnolipids) but strong attraction to cationic surfactants [76].
- Carbonate Reservoirs: Carbonates (e.g., limestone, dolomite) present a greater challenge. Due to the adsorption of crude oil components like asphaltenes, the rock surfaces are often oil-wet [131,132,133]. Therefore, wettability alteration is a key target for biosurfactants, but their high adsorption and loss remains a major problem. More critically, the surface charge of carbonates is highly pH-dependent. At typical formation brine pH, which is usually below the zero point of charge of calcite (8-9.5), carbonate surfaces are positively charged [134,135]. The strong electrostatic attraction between positively charged rock surface and negatively charged biosurfactants lead to much higher adsorption losses.
- (2)
- Biosurfactant Concentration
- (3)
- Brine Chemistry
- The ionic composition of the formation brine directly modulates electrostatic interactions. As earlier noted, high ionic strength can screen repulsive charges, potentially increasing adsorption. However, divalent cations can bridge negatively charged surfactant head groups and mineral surfaces, exacerbating loss. The pH of brine poses significant influence on the rock surface and the biosurfactant, thus controlling the electrostatic driving force for adsorption.
- Use of Sacrificial Adsorbing Agents: Pre-injecting agents that competitively adsorb to mineral sites can reduce subsequent biosurfactant loss. Nanoparticles (e.g., SiO2, CaCO3, TiO2, ZrO2 and Al2O3) are especially attractive because of their high surface area and tunability [138,139,140,141]. For instance, the addition of colloidal SiO2 has been shown to reduce the adsorption of an anionic surfactant on sandstone by up to 61% [140].
- Formulation Engineering: Co-injecting polymers or alkali can alter system chemistry. Polymers can provide steric hindrance, while alkali increases pH and can reverse carbonate surface charge to negative, repelling anionic surfactants [115].
- Operational Optimization: Tailoring the ionic strength or pH of the injection brine to minimize electrostatic attraction between the biosurfactant and the target rock can significantly reduce adsorption.
5.3. Scalability and Economic Hurdles
- Large-Scale Production Costs: While biosurfactants can be produced from renewable resources, the current costs of fermentation, substrate, and downstream processing (extraction and purification) are often higher than for manufacturing synthetic surfactants [128]. Low yields and the expense of nutrient media are significant cost drivers. A major area of ongoing research is the use of cheap, renewable substrates, such as agro-industrial wastes (molasses, whey, plant oil residues), to drastically reduce production costs [142].
- Purification Challenges: For ex-situ applications, the biosurfactant must be separated from the fermentation broth and purified to some extent. This downstream processing cost often consumes over 60% of the total production expense [143,144]. It is therefore crucial to develop economical separation and purification techniques, such as membrane filtration or foam fractionation.
- Nutrient Supply for In-situ Applications: The in-situ approach (detailed in the following section) avoids expensive downstream processing but introduces its own challenges. Stimulating the growth of indigenous or injected microbes necessitates a carefully designed nutrient package (e.g., molasses) [56]. However, it is very challenging to transport and uniformly distribute these nutrients throughout the target reservoir zone without causing pore plugging (biomass souring) problem [145]. Furthermore, ensuring the microbes preferentially produce biosurfactants rather than just biomass or other less useful metabolites requires precise control over nutrient composition and injection strategies.
6. Field Applications for Biosurfactant-Based MEOR
6.1. Historical Overview and Implementation Strategies
- Huff-n-Puff (Cyclic Microbial Recovery): This is a single-well stimulation technique. A formulation of microbes and/or nutrients is injected into a production well. The well is then shut-in for a period (the "huff" or "soak" phase) to allow for microbial growth and biosurfactant production. Afterwards, the well is put back on production (the "puff" phase). The produced fluids contain mobilized oil. This method is relatively low-cost and is used to treat near-wellbore damage and stimulate individual wells. Some heavy oil wells that have undergone multiple rounds of steam stimulation are also attempting to apply microbial huff-and-puff technology to extend their economic life. Currently, the success rate of microbial huff-and-puff remains relatively low, at approximately 70% [147].
- Microbial Flooding: This is a reservoir-wide process analogous to a chemical flood, where a microbial/nutrient solution is injected continuously or in slugs into injector wells to displace oil towards producers. This approach is more complex but holds significant potential for enhancing ultimate recovery [148]. Field applications of microbial huff-and-puff trial in China's extra-low permeability reservoirs achieved an average incremental oil of 149 tons per well. A microbial flooding project in the Chaoyanggou Oilfield resulted in a cumulative incremental production of 60,000 tons, enhancing oil recovery by 4.95%. These successes, alongside systematic research on reservoir microbial ecology have demonstrated the viability of this method, despite its high cost of CNY 300 to 557 per ton of incremental oil [149].
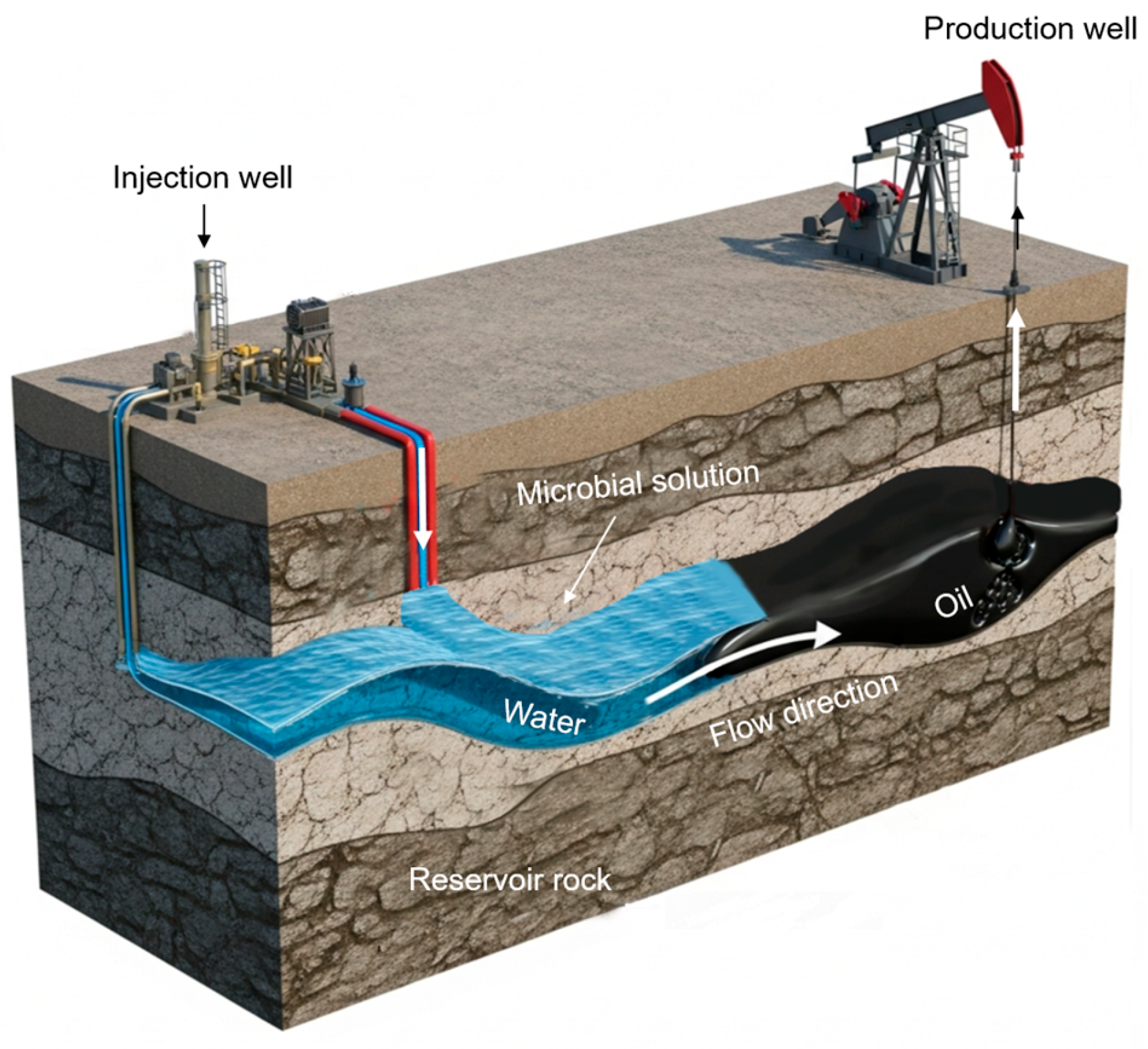
6.2. The Core Operational Decision: Ex-Situ vs. In-Situ MEOR
| Feature |
Ex-situ MEOR (Biosurfactant Injection) |
In-situ MEOR (Microbial Injection) |
| Concept | Biosurfactants are produced in industrial fermenters, separated, and injected as a chemical solution into the reservoir [150]. | A consortium of selected microorganisms and a nutrient package (e.g., molasses) are injected into the reservoir. Biosurfactants are produced downhole [151]. |
| Formulation | Purified or semi-purified biosurfactant solution, often combined with biopolymers or other agents [152]. | Microbial inoculum, carbon source (molasses, sugars), nitrogen/phosphorus sources, and minerals. |
| Advantages | High Control: Precise control over the type, concentration, and quality of the biosurfactant being injected [153,154]. Predictable: Process performance is easier to model and predict. No Bio-Plugging Risk: Avoids risks of uncontrolled biomass growth and reservoir souring [155]. |
Lower Cost: Avoid expensive downstream processing and purification. Deep Penetration: Microbes can travel deep into the reservoir and produce agents at the oil-rock interface where they are most needed [156]. Synergistic Mechanisms: Multiple EOR mechanisms (biosurfactants, biopolymers, acids, gases) function concurrently. |
| Disadvantages | High Cost: Fermentation and especially purification costs are a major economic barrier [116,128]. Adsorption Loss: Injected surfactant is subject to significant loss via adsorption onto rock surfaces [157]. Transport Issues: Ensuring the formulation reaches the target zone without degradation. |
Uncontrollable: Hard to control microbial growth and metabolic activity in subsurface reservoir. Plugging Risk: Uncontrolled biomass growth can plug pore throats and damage permeability [158,159]. Reservoir Souring: Potential H2S production by sulfate-reducing bacteria [160]. Nutrient Transport: Poor nutrient distribution uniformity. |
| Best Suited For | High-temperature reservoirs where microbial survival is difficult; reservoirs where precise chemical control is desired; testing specific biosurfactant performance. | Reservoirs with low-moderate temperature (<80°C); reservoirs with favorable geochemistry; cost-sensitive operations [161]. |
6.3. Key Insights from Field Trials
- (1)
- Comprehensive reservoir assessment. A successful MEOR project requires a deep understanding of the reservoir's geology, mineralogy, fluid properties, temperature, pressure, and indigenous microbial population, which is crucial for selecting the right MEOR strategy (in-situ vs. ex-situ) and the appropriate microbial strain or biosurfactant.
- (2)
- Tailored Strain Selection: There is no one-size-fits-all microbe. The chosen strain (for in-situ) or the production strain (for ex-situ) must be robust enough to thrive or produce metabolites that are stable under the specific reservoir's temperature, salinity, and pressure. Bioprospecting from environments analogous to oil reservoirs (e.g., hypersaline lakes, hydrothermal vents) is a promising source of suitable candidates [162].
- (3)
- Targeted Nutrient Delivery: In in-situ MEOR, the nutrient formulation directs the subsurface microbial metabolism. It must be designed to maximize the production of desired metabolites (like biosurfactants) while minimizing unwanted outcomes like excess biomass (which causes plugging) or hydrogen sulfide (H2S) from reservoir souring [163]. The inclusion of nitrate is a common strategy to outcompete sulfate-reducing bacteria [164].
- (4)
- Robust Process Monitoring: An integrated monitoring program is key to timely and effective process optimization. This includes the monitoring of injection/production rates and pressures, chemical composition analysis of produced fluids (e.g., pH, biosurfactants, polymers, microbial counts), and fluid movement.
7. Overcoming Challenges and Future Perspectives
7.1. Cost-Effective Production: The Quest for Cheaper Feedstocks
- Agro-Industrial Wastes: Substrates like molasses from sugar refining, crude glycerol from biodiesel production, whey from the dairy industry, and residues from vegetable oil processing are rich in carbohydrates and lipids, making them ideal feedstocks for many biosurfactant-producing microbes .
- Lignocellulosic Biomass: Developing microbial strains or consortia that can directly convert non-food plant biomass (e.g., corn stover, switchgrass) into biosurfactants would open up a vast and sustainable resource base.
7.2. Hybrid Technologies: The Power of Synergy
- Biosurfactants and Low Salinity Water (LSW) Flooding: LSW flooding is an emerging EOR technique where water with reduced salinity is injected into the reservoir. Low-salinity environment can induce wettability alteration of the rock surface towards a more water-wet state by modifying the ion-exchange equilibrium. This phenomenon is particularly pronounced in sandstone reservoirs containing clays. Combining LSW in MEOR can provide a favorable initial shift in wettability and a better environment for biosurfactant to function, thus lowering the consumption of biosurfactant and the overall cost [168,169].
- Biosurfactants and Smart Water Flooding: ”Smart Water” is an extension of LSW. In smart water flooding, the ionic composition (instead of only the total salinity) of the injected water is carefully customized to promote rock-fluid interactions. A smart water formulation could be designed to maximize the stability and activity of a co-injected biosurfactant, creating a highly optimized and targeted EOR fluid [170,171].
7.3. Environmental Impact and Lifecycle Assessment
7.4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ASP | Alkaline-surfactant-polymer |
| Bio-ASP | Bio-based Alkali-Surfactant-Polymer |
| cEOR | Chemical Enhanced Oil Recovery |
| CMC | Critical micelle concentration |
| EOR | Enhanced Oil Recovery |
| HLB | Hydrophilic-lipophilic balance |
| HTHS | High temperature, high salinity |
| LSW | Low Salinity Water |
| MEOR | Microbial Enhanced Oil Recovery |
| MW | Molecular weight |
| OOIP | Original oil in place |
References
- Falkowski, P.G.; Fenchel, T.; Delong, E.F. The Microbial Engines That Drive Earth's Biogeochemical Cycles. Science 2008, 320, 1034–1039. [Google Scholar] [CrossRef]
- Datta, P.; Tiwari, P.; Pandey, L.M. Isolation and characterization of biosurfactant producing and oil degrading Bacillus subtilis MG495086 from formation water of Assam oil reservoir and its suitability for enhanced oil recovery. Bioresource Technology 2018, 270, 439–448. [Google Scholar] [CrossRef]
- Argentin, M.N.; Martins, L.F.; Sousa, M.P.; Bossolan, N.R.S. Biosurfactant from a thermo-halophilic strain of Bacillus alveayuensis isolated from a Brazilian oil reservoir: Production, chemical characterization, antimicrobial activity, and efficiency in wettability reversal and oil removal from oil-soaked sand. Geoenergy Science and Engineering 2023, 231. [Google Scholar] [CrossRef]
- Niu, J.; Liu, Q.; Lv, J.; Peng, B. Review on microbial enhanced oil recovery: Mechanisms, modeling and field trials. Journal of Petroleum Science and Engineering 2020, 192. [Google Scholar] [CrossRef]
- p.l.c, B. BP Statistical Review of World Energy 2022 | 71st edition. 2022, p. 3. Available online: https://www.bp.com.cn/content/dam/bp/country-sites/zh_cn/china/home/reports/statistical-review-of-world-energy/2022/bp-stats-review-2022-full-report_zh_resized.pdf.
- IEA. Electricity Mid-Year Update 2025. Available online: https://www.iea.org/reports/electricity-mid-year-update-2025.
- Yasari, E.; Pishvaie, M.R.; Khorasheh, F.; Salahshoor, K.; Kharrat, R. Application of multi-criterion robust optimization in water-flooding of oil reservoir. Journal of Petroleum Science and Engineering 2013, 109, 1–11. [Google Scholar] [CrossRef]
- Sorbie, K.; Skauge, A. Mobilization of by-passed oil by viscous crossflow in EOR processes. In Proceedings of the IOR 2019–20th European Symposium on Improved Oil Recovery, 2019; pp. 1–16. [Google Scholar]
- de Araujo, L.L.G.C.; Sodré, L.G.P.; Brasil, L.R.; Domingos, D.F.; de Oliveira, V.M.; da Cruz, G.F. Microbial enhanced oil recovery using a biosurfactant produced by Bacillus safensis isolated from mangrove microbiota - Part I biosurfactant characterization and oil displacement test. Journal of Petroleum Science and Engineering 2019, 180, 950–957. [Google Scholar] [CrossRef]
- Sen, R. Biotechnology in petroleum recovery: the microbial EOR. Progress in Energy and Combustion Science 2008, 34, 714–724. [Google Scholar] [CrossRef]
- Belyaev, S.e.a. Use of microorganisms in the biotechnology for the enhancement of oil recovery. Microbiology 2004, 73, 590–598. [Google Scholar] [CrossRef]
- Lazar, I.P., I; Yen, T. Microbial enhanced oil recovery (MEOR). Petroleum Science and Technology 2007, 25, 1353–1366. [CrossRef]
- McInerney, M.N., DP; Knapp, RM. Microbially enhanced oil recovery: past, present, and future. In Petroleum microbiology; Magot, M.O., B, Pandey, L.M., Eds.; 2005; pp. 215–237. [Google Scholar]
- Van Hamme, J.S., A; Ward, OP. Recent advances in petroleum microbiology. Microbiology and Molecular Biology Reviews 2003, 67, 503–549. [CrossRef]
- Fletcher, A.J.P.D., J.P. How EOR Can be Transformed by Nanotechnology. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, April 24–26, 2010, 2010. [Google Scholar]
- Patel, J.; Borgohain, S.; Kumar, M.; Rangarajan, V.; Somasundaran, P.; Sen, R. Recent developments in microbial enhanced oil recovery. Renewable and Sustainable Energy Reviews 2015, 52, 1539–1558. [Google Scholar] [CrossRef]
- Asl, H.F.; Zargar, G.; Manshad, A.K.; Takassi, M.A.; Ali, J.A.; Keshavarz, A. Experimental investigation into l-Arg and l-Cys eco-friendly surfactants in enhanced oil recovery by considering IFT reduction and wettability alteration. Petroleum Science 2019, 17, 105–11. [Google Scholar] [CrossRef]
- Shirazi, M.; Mahani, H.; Tamsilian, Y.; Muggeridge, A.; Masihi, M. Full life cycle review of water-based CEOR methods from pre-injection to post-production. Fuel 2024, 356. [Google Scholar] [CrossRef]
- Bai, M.; Zhang, Z.; Cui, X.; Song, K. Studies of injection parameters for chemical flooding in carbonate reservoirs. Renewable and Sustainable Energy Reviews 2017, 75, 1464–1471. [Google Scholar] [CrossRef]
- Wang, D.S., R.; Shao, Z. Key aspects of project design for polymer flooding at the Daqing Oilfield. SPE Reservoir Evaluation & Engineering 2008, 11, 1117–1124. [Google Scholar] [CrossRef]
- Morel, D.V., M.Jouenne, S.Nahas, E. SPE 116672 Polymer Injection in Deep Offshore Field: The Dalia Angola Case. In Proceedings of the SPE Annual Technical Conference and Exhibition, 2008; pp. 4425–4436.
- Prasad, D.P., A.; Kumar, M.S.; Koduru, N. SPE-169146-MS Pilot to Full-field Polymer Application in One of the Largest Onshore Field in India. In Proceedings of the SPE/DOE SYMPOSIUM ON IMPROVED OIL RECOVERY, Richardson, Texas, 20140101, 2014; pp. 1582–1594. [Google Scholar]
- Mehta, N.K., G.; Panneer Selvam, V. SPE-179807-MS Challenges in Full Field Polymer Injection Mangala in Field. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 2016–3–21, 2016; pp. 1–11. [Google Scholar]
- Lu, Q.N., Y. G.; Wang, J. H.; Yang, X. Full Field Offshore Surfactant-Polymer Flooding in Bohai Bay China. In Proceedings of the SPE Asia Pacific Enhanced Oil Recovery Conference (SPE Enhanced Oil Recovery Conference), Kuala Lumpur, Malaysia, 2015. [Google Scholar]
- Pitts, M.J.D., E.; Wyatt, K.; Skeans, E.; Deo, D.; Galipeault, A.; Mohagen, D.; Humphry, C. Instow: A Full-Field, Multipatterned Alkaline-Surfactant-Polymer Flood—Analyses and Comparison of Phases 1 and 2. SPE Reservoir Evaluation & Engineering 2021, 24, 874–888. [CrossRef]
- Mahrouqi, D.A.S., H.; Farajzadeh, R.; Svec, Y.; Farsi, S.; Baqlani, S.; Battash, M.; Hadidi, K.; Kindi, O.; Balushi, A.; Karpan, V. Advancement Towards the Full-Field Implementation of Marmul Alkaline-Surfactant-Polymer in the Sultanate of Oman. In Proceedings of the Abu Dhabi International Petroleum Exhibition & Conference, Abu Dhabi, UAE, 2021.
- Reddick, C.E. Low Salinity Enhanced Oil Recovery - Laboratory to Day One Field Implementation - LoSal EOR into the Clair Ridge Project. In Proceedings of the Abu Dhabi International Petroleum Conference and Exhibition, Abu Dhabi, UAE, 2012. [Google Scholar]
- Baginski, V.L., D.; Waldman, T.; Manrique, E.; Ravikiran, R. Logistic Considerations for Safe Execution of Offshore Chemical EOR Projects. In Proceedings of the SPE Asia Pacific Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, August 11–13,, 2015.
- Abdullahi, M.B.R., K.; Junin, R.; Bayat, A. E. Appraising the impact of metal-oxide nanoparticles on rheological properties of HPAM in different electrolyte solutions for enhanced oil recovery. Journal of Petroleum Science and Engineering 2019, 172, 1057–1068. [CrossRef]
- Maia, A.M.S.B., R.; Balaban, R. C. Comparison between a polyacrylamide and a hydrophobically modified polyacrylamide flood in a sandstone core. Materials Science and Engineering: C 2009, 29, 505–509. [Google Scholar] [CrossRef]
- Jouenne, S. Polymer flooding in high temperature, high salinity conditions: Selection of polymer type and polymer chemistry, thermal stability. Journal of Petroleum Science and Engineering 2020, 195, 107545. [Google Scholar] [CrossRef]
- Argillier, J.-F.H., I.; Noik, C.; Viera, R.; Roca Leon, F.; Aanesen, B. Influence of Chemical EOR on Topside Produced Water Management. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, Oklahoma, USA, April 12, 2014.
- Al Shehai, A. Royal Commission at Yanbu Environmental Regulations. Procedia Engineering 2015, 125, 193–198. [Google Scholar] [CrossRef]
- Kaiser, A.W., A.; Lukman, A.; Istiyarso, I.; Gernand, M.; ShamsiJazeyi, H.; Wylde, J.; Alvarez, L. The Influence of Chemical EOR on Produced Water Separation and Quality. In Proceedings of the SPE Asia Pacific Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, August 11–13, 2015.
- Dwyer, P.D., E. Produced Water Treatment - Preparing for EOR Projects. In Proceedings of the SPE Produced Water Handling & Management Symposium, Galveston, Texas, USA, May 20–21, 2015. [Google Scholar]
- Badmus, S.O.A., H. K.; Oyehan, T. A.; Saleh, T. A. Environmental risks and toxicity of surfactants: overview of analysis, assessment, and remediation techniques. Environmental Science and Pollution Research International 2021, 28, 62085–62104. [CrossRef]
- Raouf, S.; Ismail, Y.; Gamaleldin, N.; Aboelkhair, H.; Attia, A. Screening of sustainable biosurfactant produced by indigenous bacteria isolated from Egyptian oil fields for microbial enhanced oil recovery. Geoenergy Science and Engineering 2024, 239. [Google Scholar] [CrossRef]
- Nikolova, C.G., T. Use of Microorganisms in the Recovery of Oil From Recalcitrant Oil Reservoirs: Current State of Knowledge, Technological Advances and Future Perspectives. Frontiers in Microbiology 2020, 10, 2996. [Google Scholar] [CrossRef]
- Omoniyi, O.; Abdulmalik, F. A review of microbial enhanced oil recovery: Current development and future prospects. International Journal of Scientific & Engineering Research 2015, 6, 1378–1389. [Google Scholar]
- Wu, B.X., J.; Yu, L.; Huang, L.; Yi, L.; Ma, Y. Research advances of microbial enhanced oil recovery. Heliyon 2022, 8, e11424. [CrossRef]
- Brown, L.R. Microbial enhanced oil recovery (MEOR). Current Opinion in Microbiology 2010, 13, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, T.; Samak, N.A.; Abdelhamid, M.M.; Aboulrous, A.A.; Xing, J. Modification Wettability and Interfacial Tension of Heavy Crude Oil by Green Bio-surfactant Based on Bacillus licheniformis and Rhodococcus erythropolis Strains under Reservoir Conditions: Microbial Enhanced Oil Recovery. Energy & Fuels 2021, 35, 1648–1663. [Google Scholar]
- Jahanbani Veshareh, M.; Ayatollahi, S. Microorganisms’ effect on the wettability of carbonate oil-wet surfaces: implications for MEOR, smart water injection and reservoir souring mitigation strategies. Journal of Petroleum Exploration and Production Technology 2019, 10, 1539–1550. [Google Scholar] [CrossRef]
- Alkan, H.; Szabries, M.; Dopffel, N.; Koegler, F.; Baumann, R.-P.; Borovina, A.; Amro, M. Investigation of spontaneous imbibition induced by wettability alteration as a recovery mechanism in microbial enhanced oil recovery. Journal of Petroleum Science and Engineering 2019, 182. [Google Scholar] [CrossRef]
- Fenibo, E.O.I., G. N.; Selvarajan, R.; Chikere, C. . Microbial Surfactants: The Next Generation Multifunctional Biomolecules for Applications in the Petroleum Industry and Its Associated Environmental Remediation. Microorganisms 2019, 7, 581. [CrossRef]
- Gudiña, E.J.F., E. C.; Rodrigues, A. I.; Teixeira, J. A.; Rodrigues, L. R. Biosurfactant production by Bacillus subtilis using corn steep liquor as culture medium. Frontiers in Microbiology 2015, 6, 59. [CrossRef] [PubMed]
- Purwasena, I.A.A., D. I.; Syukron, M.; Amaniyah, M.; Sugai, Y. Stability test of biosurfactant produced by Bacillus licheniformis DS1 using experimental design and its application for MEOR. Journal of Petroleum Science and Engineering 2019, 183, 106383. [CrossRef]
- Ron, E.Z.R., E. Biosurfactants and oil bioremediation. Current Opinion in Biotechnology 2002, 13, 249–252. [Google Scholar] [CrossRef]
- De Almeida, D.G.D.D.S., R. C. F. S.; Luna, J. M.; Rufino, R. D.; Santos, V. A.; Banat, I. M.; Sarubbo, L. A. Biosurfactants: Promising Molecules for Petroleum Biotechnology Advances. Frontiers in Microbiology 2016, 7, 1718. [CrossRef]
- Gudiña, E.J.P., J. F. B.; Costa, R.; Coutinho, J. A. P.; Teixeira, J. A.; Rodrigues, L. R. Biosurfactant-producing and oil-degrading Bacillus subtilis strains enhance oil recovery in laboratory sand-pack columns. Journal of Hazardous Materials 2013, 261, 106–113. [CrossRef]
- Lake, L.W. Enhanced Oil Recovery; Prentice-Hall Inc., 1989. [Google Scholar]
- Guo, H.S., K.; Hilfer, R. A Brief Review of Capillary Number and its Use in Capillary Desaturation Curves. Transport in Porous Media 2022, 144, 3–31. [Google Scholar] [CrossRef]
- Khaksar Manshad, A.R., M.; Moradi, S.; Nowrouzi, I.; Mohammadi, A. H. Wettability alteration and interfacial tension (IFT) reduction in enhanced oil recovery (EOR) process by ionic liquid flooding. Journal of Molecular Liquids 2017, 248, 153–162. [CrossRef]
- Wu, B.; Xiu, J.; Yu, L.; Huang, L.; Yi, L.; Ma, Y. Biosurfactant production by Bacillus subtilis SL and its potential for enhanced oil recovery in low permeability reservoirs. Scientific Reports 2022, 12. [Google Scholar] [CrossRef]
- Liu, Q.; Niu, J.; Liu, Y.; Li, L.; Lv, J. Optimization of lipopeptide biosurfactant production by Bacillus licheniformis L20 and performance evaluation of biosurfactant mixed system for enhanced oil recovery. Journal of Petroleum Science and Engineering 2022, 208. [Google Scholar] [CrossRef]
- Huang, L. Analysis of microbial communities in typical oil reservoirs and research on oil displacement mechanism. Doctoral Dissertation, University of Chinese Academy of Sciences, Beijing, 2014. [Google Scholar]
- Urum, K.; Pekdemir, T. Evaluation of biosurfactants for crude oil contaminated soil washing. Chemosphere 2004, 57, 1139–1150. [Google Scholar] [CrossRef] [PubMed]
- Hung, H.-c.; Shreve, G.S. Effect of the hydrocarbon phase on interfacial and thermodynamic properties of two anionic glycolipid biosurfactants in hydrocarbon/water systems. The Journal of Physical Chemistry B 2001, 105, 12596–12600. [Google Scholar] [CrossRef]
- Daoshan, L.; Shouliang, L.; Yi, L.; Demin, W. The effect of biosurfactant on the interfacial tension and adsorption loss of surfactant in ASP flooding. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2004, 244, 53–60. [Google Scholar] [CrossRef]
- Benincasa, M.; Abalos, A.; Oliveira, I.; Manresa, A. Chemical structure, surface properties and biological activities of the biosurfactant produced by Pseudomonas aeruginosa LBI from soapstock. Antonie Van Leeuwenhoek 2004, 85, 1–8. [Google Scholar] [CrossRef]
- Huszcza, E.; Burczyk, B. Biosurfactant production by Bacillus coagulans. Journal of Surfactants and Detergents 2003, 6, 61–64. [Google Scholar] [CrossRef]
- Lin, S.-C.; Minton, M.A.; Sharma, M.M.; Georgiou, G. Structural and immunological characterization of a biosurfactant produced by Bacillus licheniformis JF-2. Applied and environmental microbiology 1994, 60, 31–38. [Google Scholar] [CrossRef]
- Kim, H.-S.; Jeon, J.-W.; Kim, S.-B.; Oh, H.-M.; Kwon, T.-J.; Yoon, B.-D. Surface and physico-chemical properties of a glycolipid biosurfactant, mannosylerythritol lipid, from Candida antarctica. Biotechnology Letters 2002, 24, 1637–1641. [Google Scholar] [CrossRef]
- Choi, K.-S.; Kim, S.-H.; Lee, T.-H. Purification and characterization of biosurfactant from Tsukamurella sp. 26A. Journal of microbiology and biotechnology 1999, 9, 32–38. [Google Scholar]
- Makkar, R.; Cameotra, S.S. Biosurfactant production by a thermophilic Bacillus subtilis strain. Journal of Industrial Microbiology and Biotechnology 1997, 18, 37–42. [Google Scholar] [CrossRef]
- Deleu, M.; Razafindralambo, H.; Popineau, Y.; Jacques, P.; Thonart, P.; Paquot, M. Interfacial and emulsifying properties of lipopeptides from Bacillus subtilis. Colloids and Surfaces A: Physicochemical and Engineering Aspects 1999, 152, 3–10. [Google Scholar] [CrossRef]
- Mousavi Moghadam, A.B.S., M. Enhancing hydrocarbon productivity via wettability alteration: A review on the application of nanoparticles. De Gruyter Reviews in Chemical Engineering 2019, 35. [Google Scholar] [CrossRef]
- Anderson, W.G. Wettability Literature Survey–Part 1: Rock/Oil/Brine Interactions and the Effects of Core Handling on Wettability. Journal of Petroleum Technology (JPT) 1986, 38, 1125–1144. [Google Scholar] [CrossRef]
- Kanj, M.S., S.; Giannelis, E. Wettability Alteration in Carbonate Reservoirs by Carbon Nanofluids. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2020, 598. [Google Scholar] [CrossRef]
- Mousavi, S.-P.H.-S., A.; Norouzi-Apourvari, S.; Jalalvand, M.; Schaffie, M.; Ranjbar, M. Toward mechanistic understanding of wettability alteration in calcite and dolomite rocks: The effects of resin, asphaltene, anionic surfactant, and hydrophilic nano particles. Journal of Molecular Liquids 2021, 321. [CrossRef]
- Mwangi, P.B., P. V.; Radonjic, M.; Thyne, G. The effect of organic acids on wettability of sandstone and carbonate rocks. Journal of Petroleum Science and Engineering 2018, 165, 428–435. [Google Scholar] [CrossRef]
- Gao, Y.X., X.; Wang, S.; Tang, L.; Geng, Z.; Li, J. Pore-scale experimental investigation of the remaining oil formation in water-wet, mixed-wet and oil-wet sandstone samples. Journal of Petroleum Science and Engineering 2022, 216. [CrossRef]
- Xu, Y.Y., G.; Hou, B.; Su, K.; Zhang, F.; Liu, B.; Li, S.; Zhao, S.; Liu, Y. Different effects of resins and asphaltenes concentration of crude oil on sandstone wettability. Fuel 2024, 370. [CrossRef]
- Khajepour, H.; Mahmoodi, M.; Biria, D.; Ayatollahi, S. Investigation of wettability alteration through relative permeability measurement during MEOR process: A micromodel study. Journal of Petroleum Science and Engineering 2014, 120, 10–17. [Google Scholar] [CrossRef]
- Alkan, H.; Szabries, M.; Dopffel, N.; Koegler, F.; Baumann, R.-P.; Borovina, A.; Amro, M. Investigation of spontaneous imbibition induced by wettability alteration as a recovery mechanism in microbial enhanced oil recovery. Journal of Petroleum Science and Engineering 2019, 182, 106163. [Google Scholar] [CrossRef]
- Chen, Z.L., Junzhang; Wang, Weidong; Wang, Lushan; Chang, Jing. Insights into wettability alteration mechanisms of microbial enhanced oil recovery by different biosurfactants in sandstone oil reservoir. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2024, 691, 133889. [CrossRef]
- Kargozarfard, Z.R., Masoud; Ayatollahi, Shahab. Viscous fingering and its effect on areal sweep efficiency during waterflooding: an experimental study. Petroleum Science 2019, 16, 105–116. [CrossRef]
- Wever, D.A.Z.P., F.; Broekhuis, A.A. Polymers for enhanced oil recovery: A paradigm for structure–property relationship in aqueous solution. Progress in Polymer Science 2011, 36, 1558–1628. [Google Scholar] [CrossRef]
- Couto, M.R.G., Eduardo J.; Ferreira, Débora; Teixeira, José A.; Rodrigues, Lígia R. The biopolymer produced by Rhizobium viscosum CECT 908 is a promising agent for application in microbial enhanced oil recovery. New Biotechnology 2019, 49, 144–150. [CrossRef]
- Correia, J.A.H. Development of (bio) molecules’ cocktails (including (bio) polymers and (bio) surfactants) to promote additional oil recovery. In Universidade do Minho (Portugal); 2024. [Google Scholar]
- Rosenberg, E.; Ron, E.Z. High-and low-molecular-mass microbial surfactants. Applied microbiology and biotechnology 1999, 52, 154–162. [Google Scholar] [CrossRef]
- Dong, H.; Xia, W.; Dong, H.; She, Y.; Zhu, P.; Liang, K.; Zhang, Z.; Liang, C.; Song, Z.; Sun, S.; et al. Rhamnolipids Produced by Indigenous Acinetobacter junii from Petroleum Reservoir and its Potential in Enhanced Oil Recovery. Frontiers in Microbiology 2016, 7. [Google Scholar] [CrossRef]
- Cooper, D.G.; Goldenberg, B.G. Surface-active agents from two Bacillus species. Applied and environmental microbiology 1987, 53, 224–229. [Google Scholar] [CrossRef]
- Xia, W.; Du, Z.; Cui, Q.; Dong, H.; Wang, F.; He, P.; Tang, Y. Biosurfactant produced by novel Pseudomonas sp. WJ6 with biodegradation of n-alkanes and polycyclic aromatic hydrocarbons. Journal of hazardous materials 2014, 276, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-F.; Mbadinga, S.M.; Yang, S.-Z.; Gu, J.-D.; Mu, B.-Z. Chemical structure, property and potential applications of biosurfactants produced by Bacillus subtilis in petroleum recovery and spill mitigation. International Journal of Molecular Sciences 2015, 16, 4814–4837. [Google Scholar] [CrossRef]
- Zheng, C.; He, J.; Wang, Y.; Wang, M.; Huang, Z. Hydrocarbon degradation and bioemulsifier production by thermophilic Geobacillus pallidus strains. Bioresource technology 2011, 102, 9155–9161. [Google Scholar] [CrossRef] [PubMed]
- Yadav, K.K.; Mandal, A.K.; Sen, I.K.; Chakraborti, S.; Islam, S.S.; Chakraborty, R. Flocculating property of extracellular polymeric substances produced by a biofilm-forming bacterium Acinetobacter junii BB1A. Applied biochemistry and biotechnology 2012, 168, 1621–1634. [Google Scholar] [CrossRef]
- Dastgheib, S.; Amoozegar, M.; Elahi, E.; Asad, S.; Banat, I. Bioemulsifier production by a halothermophilic Bacillus strain with potential applications in microbially enhanced oil recovery. Biotechnology letters 2008, 30, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Khademolhosseini, R.; Jafari, A.; Mousavi, S.M.; Hajfarajollah, H.; Noghabi, K.A.; Manteghian, M. Physicochemical characterization and optimization of glycolipid biosurfactant production by a native strain of Pseudomonas aeruginosa HAK01 and its performance evaluation for the MEOR process. RSC advances 2019, 9, 7932–7947. [Google Scholar] [CrossRef]
- Soroush, A.; Azizollah, K. Petroleum Emulsion Stability and Separation Strategies: A Comprehensive Review. ChemEngineering 2025, 9, 113. [Google Scholar] [CrossRef]
- Hassan, A.M.; Al-Shalabi, E.W.; Ayoub, M.A. Updated perceptions on polymer-based enhanced oil recovery toward high-temperature high-salinity tolerance for successful field applications in carbonate reservoirs. Polymers 2022, 14. [Google Scholar] [CrossRef]
- Geetha, S.J.; Banat, I.M.; Joshi, S.J. Biosurfactants: Production and potential applications in microbial enhanced oil recovery (MEOR). Biocatalysis and Agricultural Biotechnology 2018, 14, 23–32. [Google Scholar] [CrossRef]
- Rikalović, M.G.; Vrvić, M.M.; Karadžić, I.M. Rhamnolipid biosurfactant from Pseudomonas aeruginosa: from discovery to application in contemporary technology. Journal of the Serbian Chemical Society 2015, 80, 279–304. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Fukuoka, T. Sophorolipids, commercialized glycolipid biosurfactants: Derivatives, component analysis, and applications. Journal of the American Oil Chemists' Society 2025, 102, 251–260. [Google Scholar] [CrossRef]
- Roelants, S.L.; Bovijn, S.; Bytyqi, E.; de Fooz, N.; Luyten, G.; Castelein, M.; Van de Craen, T.; Diao, Z.; Maes, K.; Delmulle, T. Bubbling insights: unveiling the true sophorolipid biosynthetic pathway by Starmerella bombicola. Biotechnology for Biofuels and Bioproducts 2024, 17, 113. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Peñalver, P.; Castillejos, M.; Koh, A.; Gross, R.; Sánchez, A.; Font, X.; Gea, T. Production and characterization of sophorolipids from stearic acid by solid-state fermentation, a cleaner alternative to chemical surfactants. Journal of cleaner production 2018, 172, 2735–2747. [Google Scholar] [CrossRef]
- Pang, Z.; Zhang, W.; Zhang, B.; Navaz, S.; Wang, F.; Liao, Y. Biosynthesis and modification strategies of novel cyclic lipopeptide secreted by Bacillus spp.: Research progress. Process Biochemistry 2025, 151, 27–42. [Google Scholar] [CrossRef]
- Gao, L.; Du, K.; Zhou, Y.; Li, C.; Su, Y.; Xia, X. Metabolic reprogramming of Bacillus subtilis for surfactin: Maximizing titer/yield/productivity and tailoring C14/C15 homologue proportions. Chemical Engineering Journal 2025, 171675. [CrossRef]
- Datta, P.; Tiwari, P.; Pandey, L.M. Experimental investigation on suitability of Surfactin for enhanced oil recovery: Stability, adsorption equilibrium and kinetics studies. Journal of Environmental Chemical Engineering 2022, 10, 107083. [Google Scholar] [CrossRef]
- Datta, P.; Tiwari, P.; Pandey, L.M. Oil washing proficiency of biosurfactant produced by isolated Bacillus tequilensis MK 729017 from Assam reservoir soil. Journal of Petroleum Science and Engineering 2020, 195, 107612. [Google Scholar] [CrossRef]
- Anuradha S, N. Structural and molecular characteristics of lichenysin and its relationship with surface activity. Biosurfactants 2010, 304–315. [Google Scholar]
- Tao, W.; Lin, J.; Wang, W.; Huang, H.; Li, S. Designer bioemulsifiers based on combinations of different polysaccharides with the novel emulsifying esterase AXE from Bacillus subtilis CICC 20034. Microbial cell factories 2019, 18, 173. [Google Scholar] [CrossRef] [PubMed]
- Zosim, Z.; Fleminger, G.; Gutnick, D.; Rosenberg, E.; Wise, G.S. Effect of protein on the emulsifying activity of emulsan. JOURNAL OF DISPERSION SCIENCE ANDTECHNOLOGY 1989, 10, 307–317. [Google Scholar] [CrossRef]
- Fenibo, E.O.; Ijoma, G.N.; Selvarajan, R.; Chikere, C.B. Microbial surfactants: the next generation multifunctional biomolecules for applications in the petroleum industry and its associated environmental remediation. Microorganisms 2019, 7, 581. [Google Scholar] [CrossRef]
- Dong, D.; Wang, X.; Cai, M.; Wang, J.; Han, Y.; Zhang, X.; Huang, Z. Partial purification and chemical characterization of a bioemulsifier and its application in MEOR. In Proceedings of the Advances in Applied Biotechnology: Proceedings of the 2nd International Conference on Applied Biotechnology (ICAB 2014)-Volume II, 2015; pp. 587–598. [Google Scholar]
- Standnes, D.C.; Skjevrak, I. Literature review of implemented polymer field projects. Journal of petroleum science and engineering 2014, 122, 761–775. [Google Scholar] [CrossRef]
- Gbadamosi, A.O.; Junin, R.; Manan, M.A.; Agi, A.; Oseh, J.O.; Usman, J. Synergistic application of aluminium oxide nanoparticles and oilfield polyacrylamide for enhanced oil recovery. Journal of Petroleum Science and Engineering 2019, 182, 106345. [Google Scholar] [CrossRef]
- Gbadamosi, A.O.; Junin, R.; Manan, M.A.; Yekeen, N.; Augustine, A. Hybrid suspension of polymer and nanoparticles for enhanced oil recovery. Polymer Bulletin 2019, 76, 6193–6230. [Google Scholar] [CrossRef]
- Kumar, S.; Saxena, N.; Mandal, A. Synthesis and evaluation of physicochemical properties of anionic polymeric surfactant derived from Jatropha oil for application in enhanced oil recovery. Journal of Industrial and Engineering Chemistry 2016, 43, 106–116. [Google Scholar] [CrossRef]
- Wever, D.-A.Z. Synthesis and evaluation of novel linear and branched polyacrylamides for enhanced oil recovery. 2013. [Google Scholar]
- Sveistrup, M.; van Mastrigt, F.; Norrman, J.; Picchioni, F.; Paso, K. Viability of biopolymers for enhanced oil recovery. Journal of Dispersion Science and Technology 2016, 37, 1160–1169. [Google Scholar] [CrossRef]
- Machale, J.; Majumder, S.K.; Ghosh, P.; Sen, T.K. Development of a novel biosurfactant for enhanced oil recovery and its influence on the rheological properties of polymer. Fuel 2019, 257, 116067. [Google Scholar] [CrossRef]
- Olajire, A.A. Review of ASP EOR (alkaline surfactant polymer enhanced oil recovery) technology in the petroleum industry: Prospects and challenges. Energy 2014, 77, 963–982. [Google Scholar] [CrossRef]
- Sheng, J.J. A comprehensive review of alkaline-surfactant-polymer (ASP) flooding. In Proceedings of the SPE Western Regional Meeting, 2013; p. pp. SPE–165358–MS. [Google Scholar]
- Deshmukh, M.; Pathan, A. Advancements and challenges in the use of surfactants and nanoparticles for enhanced oil recovery: mechanisms, synergies, and field applications. Environmental Science and Pollution Research 2025, 1–35. [Google Scholar] [CrossRef]
- Hou, J.; Sui, H.; Du, J.; Sun, L. Synergistic effect of silica nanofluid and biosurfactant on bitumen recovery from unconventional oil. Journal of Dispersion Science and Technology 2020, 43, 704–715. [Google Scholar] [CrossRef]
- Gazem, A.; Krishna, S.; Al-Yaseri, A. Polymer formulations with binary biosurfactants and zinc oxide nanoparticles for enhanced oil recovery. Fuel 2025, 400, 135733. [Google Scholar] [CrossRef]
- Almahfood, M.; Bai, B. The synergistic effects of nanoparticle-surfactant nanofluids in EOR applications. Journal of Petroleum Science and Engineering 2018, 171, 196–210. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, X.; Mohanty, K. Effect of a nanoparticle on wettability alteration and wettability retainment of carbonate reservoirs. Journal of Petroleum Science and Engineering 2022, 215, 110684. [Google Scholar] [CrossRef]
- Azarshin, S.; Moghadasi, J.; A Aboosadi, Z. Surface functionalization of silica nanoparticles to improve the performance of water flooding in oil wet reservoirs. Energy Exploration & Exploitation 2017, 35, 685–697. [Google Scholar] [CrossRef]
- Liu, Y.; Fu, J.; Zeng, H.; Qu, R.; Feng, Q.; Li, X.; Zhang, F.; She, Y. Synthesis of biological nanocomposite fluids and enhanced crude oil recovery. Fuel 2024, 363, 130954. [Google Scholar] [CrossRef]
- Al-Ghailani, T.; Al-Wahaibi, Y.M.; Joshi, S.J.; Al-Bahry, S.N.; Elshafie, A.E.; Al-Bemani, A.S. Application of a new bio-ASP for enhancement of oil recovery: Mechanism study and core displacement test. Fuel 2021, 287, 119432. [Google Scholar] [CrossRef]
- Joshi, S.J.; Al-Farsi, H.; Al-Bahry, S.; Al-Wahaibi, Y. “Glycolipid biosurfactant-silica nanoparticles” based green application for enhancement of oil recovery. Petroleum Science and Technology 2022, 40, 2064–2081. [Google Scholar] [CrossRef]
- Ayman, A.; Abdelhamid, M.; Jaeger, P.; Mostafa, E.M. Optimizing Oil Recovery with Eco-Friendly Biopolymer-Stabilized Aluminum Oxide Nanofluids: A Comparative Investigation of Egg and Soy Proteins. Egyptian Journal of Chemistry 2025, 68, 323–331. [Google Scholar] [CrossRef]
- Sałek, K.; Euston, S.R. Sustainable microbial biosurfactants and bioemulsifiers for commercial exploitation. Process Biochemistry 2019, 85, 143–155. [Google Scholar] [CrossRef]
- Alyousef, M.H.; Kamal, M.S.; Murtaza, M.; Hussain, S.M.S.; Raza, A.; Patil, S.; Mahmoud, M. Enhancing Aqueous Stability of Anionic Surfactants in High Salinity and Temperature Conditions with SiO2 Nanoparticles. ACS omega 2024, 9, 49804–49815. [Google Scholar] [CrossRef]
- Wang, X.-T.; Liu, B.; Li, X.-Z.; Lin, W.; Li, D.-A.; Dong, H.; Wang, L. Biosurfactants produced by novel facultative-halophilic Bacillus sp. XT-2 with biodegradation of long chain n-alkane and the application for enhancing waxy oil recovery. Energy 2022, 240. [Google Scholar] [CrossRef]
- Khademolhosseini, R.; Jafari, A.; Mousavi, S.M.; Hajfarajollah, H.; Noghabi, K.A.; Manteghian, M. Physicochemical characterization and optimization of glycolipid biosurfactant production by a native strain ofPseudomonas aeruginosaHAK01 and its performance evaluation for the MEOR process. RSC Advances 2019, 9, 7932–7947. [Google Scholar] [CrossRef]
- Cooper, D.; Zajic, J.; Denis, C. Surface active properties of a biosurfactant from Corynebacterium lepus. Journal of the American Oil Chemists’ Society 1981, 58, 77–80. [Google Scholar] [CrossRef]
- Zou, C.; Wang, M.; Xing, Y.; Lan, G.; Ge, T.; Yan, X.; Gu, T. Characterization and optimization of biosurfactants produced by Acinetobacter baylyi ZJ2 isolated from crude oil-contaminated soil sample toward microbial enhanced oil recovery applications. Biochemical Engineering Journal 2014, 90, 49–58. [Google Scholar] [CrossRef]
- Ivanova, A.; Mitiurev, N.; Cheremisin, A.; Orekhov, A.; Kamyshinsky, R.; Vasiliev, A. Characterization of organic layer in oil carbonate reservoir rocks and its effect on microscale wetting properties. Scientific reports 2019, 9, 10667. [Google Scholar] [CrossRef] [PubMed]
- Standnes, D.C.; Austad, T. Wettability alteration in chalk: 2. Mechanism for wettability alteration from oil-wet to water-wet using surfactants. Journal of Petroleum Science and Engineering 2000, 28, 123–143. [Google Scholar] [CrossRef]
- Shi, Y.; Miller, C.; Mohanty, K. Surfactant-aided low-salinity waterflooding for low-temperature carbonate reservoirs. SPE Journal 2021, 26, 2214–2230. [Google Scholar] [CrossRef]
- Yao, Y.; Wei, M.; Kang, W. A review of wettability alteration using surfactants in carbonate reservoirs. Advances in Colloid and Interface Science 2021, 294, 102477. [Google Scholar] [CrossRef]
- Deng, X.; Kamal, M.S.; Patil, S.; Hussain, S.M.S.; Zhou, X. A review on wettability alteration in carbonate rocks: Wettability modifiers. Energy & Fuels 2019, 34, 31–54. [Google Scholar] [CrossRef]
- Al-Sulaimani, H.; Al-Wahaibi, Y.; Al-Bahry, S.; Elshafie, A.; Al-Bemani, A.; Joshi, S.; Ayatollahi, S. Residual-oil recovery through injection of biosurfactant, chemical surfactant, and mixtures of both under reservoir temperatures: induced-wettability and interfacial-tension effects. SPE Reservoir Evaluation & Engineering 2012, 15, 210–217. [Google Scholar]
- Scerbacova, A.; Ivanova, A.; Grishin, P.; Cheremisin, A.; Tokareva, E.; Tkachev, I.; Sansiev, G.; Fedorchenko, G.; Afanasiev, I. Application of alkalis, polyelectrolytes, and nanoparticles for reducing adsorption loss of novel anionic surfactant in carbonate rocks at high salinity and temperature conditions. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2022, 653, 129996. [Google Scholar] [CrossRef]
- Nourafkan, E.; Hu, Z.; Wen, D. Nanoparticle-enabled delivery of surfactants in porous media. Journal of colloid and interface science 2018, 519, 44–57. [Google Scholar] [CrossRef]
- Yekeen, N.; Al-Yaseri, A.; Idris, A.K.; Khan, J.A. Comparative effect of zirconium oxide (ZrO2) and silicon dioxide (SiO2) nanoparticles on the adsorption properties of surfactant-rock system: Equilibrium and thermodynamic analysis. Journal of Petroleum Science and Engineering 2021, 205, 108817. [Google Scholar] [CrossRef]
- Kesarwani, H.; Sharma, S.; Mandal, A. Application of novel colloidal silica nanoparticles in the reduction of adsorption of surfactant and improvement of oil recovery using surfactant polymer flooding. ACS omega 2021, 6, 11327–11339. [Google Scholar] [CrossRef]
- Saxena, N.; Kumar, A.; Mandal, A. Adsorption analysis of natural anionic surfactant for enhanced oil recovery: The role of mineralogy, salinity, alkalinity and nanoparticles. Journal of Petroleum Science and Engineering 2019, 173, 1264–1283. [Google Scholar] [CrossRef]
- Jahanbani Veshareh, M.; Ganji Azad, E.; Deihimi, T.; Niazi, A.; Ayatollahi, S. Isolation and screening of Bacillus subtilis MJ01 for MEOR application: biosurfactant characterization, production optimization and wetting effect on carbonate surfaces. Journal of Petroleum Exploration and Production Technology 2018, 9, 233–245. [Google Scholar] [CrossRef]
- Henkel, M.; Müller, M.M.; Kügler, J.H.; Lovaglio, R.B.; Contiero, J.; Syldatk, C.; Hausmann, R. Rhamnolipids as biosurfactants from renewable resources: concepts for next-generation rhamnolipid production. Process Biochemistry 2012, 47, 1207–1219. [Google Scholar] [CrossRef]
- Sharon, A.B.; Ahuekwe, E.F.; Nzubechi, E.G.; Oziegbe, O.; Oniha, M. Statistical optimization strategies on waste substrates for solving high-cost challenges in biosurfactants production: a review. In Proceedings of the IOP Conference Series: Earth and Environmental Science, 2023; p. 012004. [Google Scholar]
- Li, W. Polyelectrolyte complex nanoparticle nutrient delivery system for microbial enhanced oil recovery. 2015. [Google Scholar]
- Safdel, M.; Anbaz, M.A.; Daryasafar, A.; Jamialahmadi, M. Microbial enhanced oil recovery, a critical review on worldwide implemented field trials in different countries. Renewable and Sustainable Energy Reviews 2017, 74, 159–172. [Google Scholar] [CrossRef]
- Weidong, W. Research Progress and Development Trend of Microbial Enhanced Oil Recovery Technology. Petroleum Geology and Recovery Efficiency 2021, 28, 1–9. [Google Scholar]
- Nikolova, C.; Gutierrez, T. Use of microorganisms in the recovery of oil from recalcitrant oil reservoirs: Current state of knowledge, technological advances and future perspectives. Frontiers in microbiology 2020, 10, 2996. [Google Scholar] [CrossRef]
- Hou, Z.L., Wei; Le, Jianjun; Jin, Rui; Dou, Xumou. Research and application of microbial enhanced oil recovery technology in Daqing Oilfield. Petroleum Geology and Recovery Efficiency 2021, 28, 10–18.
- Perfumo, A.; Rancich, I.; Banat, I.M. Possibilities and challenges for biosurfactants use in petroleum industry. Biosurfactants 2010, 135–145. [Google Scholar]
- Youssef, N.; Elshahed, M.S.; McInerney, M.J. Microbial processes in oil fields: culprits, problems, and opportunities. Advances in applied microbiology 2009, 66, 141–251. [Google Scholar]
- Luft, L.; Confortin, T.C.; Todero, I.; Zabot, G.L.; Mazutti, M.A. An overview of fungal biopolymers: bioemulsifiers and biosurfactants compounds production. Critical reviews in biotechnology 2020, 40, 1059–1080. [Google Scholar] [CrossRef]
- Reis, R.S.; Pacheco, G.; Pereira, A.; Freire, D. Biosurfactants: production and applications. In Biodegradation-life of science; IntechOpen, 2013. [Google Scholar]
- Al-Wahaibi, Y.; Al-Hadrami, H.; Al-Bahry, S.; Elshafie, A.; Al-Bemani, A.; Joshi, S. Injection of biosurfactant and chemical surfactant following hot water injection to enhance heavy oil recovery. Petroleum Science 2016, 13, 100–109. [Google Scholar] [CrossRef]
- Landa-Marbán, D. Mathematical modeling of microbial enhanced oil recovery with focus on bio-plug technology: from the pore to the core scale. 2019. [Google Scholar]
- Park, T.; Jeon, M.-K.; Yoon, S.; Lee, K.S.; Kwon, T.-H. Modification of interfacial tension and wettability in oil–brine–quartz system by in situ bacterial biosurfactant production at reservoir conditions: implications for microbial enhanced oil recovery. Energy & Fuels 2019, 33, 4909–4920. [Google Scholar]
- Ahmadi, M.A.; Shadizadeh, S.R. Experimental investigation of a natural surfactant adsorption on shale-sandstone reservoir rocks: Static and dynamic conditions. Fuel 2015, 159, 15–26. [Google Scholar] [CrossRef]
- Stewart, T.L.; Fogler, H.S. Biomass plug development and propagation in porous media. Biotechnology and Bioengineering 2001, 72, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Stewart, T.L.; Scott Fogler, H. Pore-scale investigation of biomass plug development and propagation in porous media. Biotechnology and bioengineering 2002, 77, 577–588. [Google Scholar] [CrossRef]
- Vance, I.; Thrasher, D.R. Reservoir souring: mechanisms and prevention. Petroleum microbiology 2005, 123–142. [Google Scholar]
- Peters, K.E.; Fowler, M.G. Applications of petroleum geochemistry to exploration and reservoir management. Organic Geochemistry 2002, 33, 5–36. [Google Scholar] [CrossRef]
- Leary, D.K. Bioprospecting and the genetic resources of hydrothermal vents on the high seas: what is the existing legal position, where are we heading and what are our options. Macquarie J. Int'l & Comp. Envtl. L. 2004, 1, 137. [Google Scholar]
- Gieg, L.M.; Jack, T.R.; Foght, J.M. Biological souring and mitigation in oil reservoirs. Applied microbiology and biotechnology 2011, 92, 263–282. [Google Scholar] [CrossRef]
- Fan, F.; Zhang, B.; Liu, J.; Cai, Q.; Lin, W.; Chen, B. Towards sulfide removal and sulfate reducing bacteria inhibition: Function of biosurfactants produced by indigenous isolated nitrate reducing bacteria. Chemosphere 2020, 238, 124655. [Google Scholar] [CrossRef] [PubMed]
- Hirasaki, G.J.; Miller, C.A.; Puerto, M. Recent advances in surfactant EOR. SPE journal 2011, 16, 889–907. [Google Scholar] [CrossRef]
- Hemlata, B.; Selvin, J.; Tukaram, K. Optimization of iron chelating biosurfactant production by Stenotrophomonas maltophilia NBS-11. Biocatalysis and agricultural biotechnology 2015, 4, 135–143. [Google Scholar] [CrossRef]
- Jain, R.M.; Mody, K.; Joshi, N.; Mishra, A.; Jha, B. Effect of unconventional carbon sources on biosurfactant production and its application in bioremediation. International journal of biological macromolecules 2013, 62, 52–58. [Google Scholar] [CrossRef]
- Gazem, A.; Krishna, S.; Al-Yaseri, A. Low-salinity enhanced oil recovery using biosurfactant-ZnO nanoparticle-xanthan gum formulations: A comparative study of rhamnolipid and sophorolipid systems. Journal of Molecular Liquids 2025, 432, 127894. [Google Scholar] [CrossRef]
- Bemani, A.; Ahmadi, M.; Motamedi, H.; Soulgani, B.S. The impacts of hybrid of microbial enhanced oil recovery and low salinity water flooding on oil recovery mechanisms: an experimental and theoretical Investigation. Journal of Molecular Liquids 2025, 418, 126687. [Google Scholar] [CrossRef]
- Honarvar, B.; Rahimi, A.; Safari, M.; Khajehahmadi, S.; Karimi, M. Smart water effects on a crude oil-brine-carbonate rock (CBR) system: Further suggestions on mechanisms and conditions. Journal of Molecular Liquids 2020, 299, 112173. [Google Scholar] [CrossRef]
- Norouzpour, M.; Azdarpour, A.; Nabipour, M.; Santos, R.M.; Manshad, A.K.; Iglauer, S.; Akhondzadeh, H.; Keshavarz, A. Red beet plant as a novel source of natural surfactant combined with ‘Smart Water’for EOR purposes in carbonate reservoirs. Journal of Molecular Liquids 2023, 370, 121051. [Google Scholar] [CrossRef]
- Lars, B.A.-K., Briem; Alexander, Beck; Susanne, Zibek; Stefan, Albrecht. Life cycle assessment for early-stage process optimization of microbial biosurfactant production using kinetic models—a case study on mannosylerythritol lipids (MEL). Frontiers in Bioengineering and Biotechnology 2024, 12.
- Albrecht, A.-K.B.L.B.A.O.P.N.S.Z.S. Environmental Impacts of Biosurfactants from a Life Cycle Perspective: A Systematic Literature Review. Environmental Impacts of Biosurfactants from a Life Cycle Perspective: A Systematic Literature Review 2022, 181, 235–269. [Google Scholar]
- Elias, A.M.L., A.A.; Ellamla, H.R.; Furlan, F.F.; Ribeiro, M.P.A.; Marcelino, P.R.F.; dos Santos, J.C.; da Silva, S.S.; Giordano, R.C. Techno-Economic-Environmental Analysis of Sophorolipid Biosurfactant Production from Sugarcane Bagasse Journal: Industrial & Engineering Chemistry Research. Industrial & Engineering Chemistry Research 2021, 60, 9833–9850.
- Noll, P.; Solarte-Toro, J.C.; Restrepo-Serna, D.L.; Treinen, C.; Poveda-Giraldo, J.A.; Henkel, M.; Alzate, C.A.C.; Hausmann, R. Limits for sustainable biosurfactant production: techno-economic and environmental assessment of a rhamnolipid production process. Bioresource Technology Reports 2024, 25, 101767. [Google Scholar] [CrossRef]
- Melikoglu, M. Biosurfactants for enhanced oil recovery and bioremediation in the modern petroleum industry: A global review. JCIS Open 2026, 21. [Google Scholar] [CrossRef]
| Biosurfactant |
Interfacial tension (mN/m) |
Liquid Phase | Reference |
| Rhamnolipid PG201 | 4.5 | Ekofisk crude oil | [57] |
| 0.08 at PH 5.4 | dodecane benzene |
[58] | |
| Rhamnolipid Dyna 201 | 0.06 at PH 5.4 0.94 at PH 5.7 |
Isooctane benzene |
[58] |
| Rhamnolipid | 4 | Crude oil | [59] |
| 1.32 | hexadecane hexadecane |
[60] | |
| Bacillus biosurfactant | 4.5 | hexadecane | [61] |
| Bacillus lipopeptide JF-2 | 0.006 at PH 6 0.023 at PH 7 |
Decane/5o NaC1 |
[62] |
| Mannosylerythritol lipid | 0.1 | Kerosene | [63] |
| Trehalose glycolipid | 1.5 | hexadecane | [64] |
| Lipopeptide Surfactin | 0.2 2.03 |
Diesel oil | [65] |
| 2.03 | dodecane | [66] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




