3.1. Infection of Tubers with Common Scab
This trait measures the disease incidence – the percentage of the entire collected tuber sample showing scab symptoms. Treatments with EMsignificantly reduced the proportion of infected tubers compared to the control treatment, which did not use them. Among the treatments, treatment variant E2, with a 20-minute inoculation of tubers with effective microorganisms, proved to be the most effective, reducing the average infection level to 52.0% compared to 61.5% in control treatment (
Table 5).
The value of this trait was significantly related to the genetic characteristics of the cultivars. Nora was the cultivar that responded best to effective microorganisms. A drastic reduction in the percentage of tubers with common scab symptoms was observed from 51.7% to 21.3% after the longer, 20-minute EM inoculation (E2) (
Table 5). Cultivars such as Nora and Oktan showed the strongest response to the extended treatment with EM (E2), achieving significantly lower infection rates than the other cultivars. The Oktan cultivar showed a significant improvement from 65.5% to 32.5% after the longer treatment with this preparation (E2). The Czapla cultivar showed a reduction from 60.7% to 34.8% after the longer E2 inoculation. Anomalies: For some cultivars, such as Bellarosa and Ewelina, tuber inoculation with EM resulted in a higher percentage of tubers showing common scab symptoms compared to the control. This suggests that although the actinomycete affected more tubers, the disease severity may have been controlled (
Table 5). Significant differences between highly resistant cultivars (such as Nora E2) and susceptible cultivars (Zuzanna - control) are statistically significant (
Table 5).
Statistical analysis showed that all three factors examined – cultivar, seed potato improvement method (EM biopreparation), and weather conditions during the growing years – had a significant impact on potato tuber health with respect to common scab (
S. scabies) (
Table 6).
Cultivation year variability: This factor proved to be crucial for disease severity. The average percentage of infected tubers was significantly lowest in 2020 (39.1%), suggesting conditions least favorable for infection (e.g., optimal soil moisture during tuber setting). Maximum severity: The highest infection rate was recorded in 2021 (78.5%), which statistically significantly differed from the other study years. 2019 (53.8%) represented an intermediate variant – infection was significantly lower than in 2021, but significantly higher than in 2020 (
Table 6).
3.1.1. Analysis of the Interaction of Weather Conditions and the Effectiveness of the EM Biopreparation
The interaction between the years of study and the applied pre-planting treatments (E1, E2) significantly influenced all three health parameters of potato tubers in relation to common scab (
S. scabies) (
Table 7).
Our research has shown that the effectiveness of EM technology in reducing common scab is closely linked to the severity of infection pressure during a given growing season.
Impact of Infection Pressure (2020 vs. 2021): Extremely different infection levels were recorded depending on the year of the study, allowing for the evaluation of the preparation under extreme conditions: High Pressure Year (2021): In the control treatment, as many as 85.6% of tubers showed scab symptoms. Under these critical conditions, the E2 variant (20 min) proved most effective, reducing the percentage of diseased tubers by 17.4 percentage points (to 68.2%). Low Pressure Year (2020): With the naturally lower disease severity (control: 42.5%), the use of the E2 variant achieved a record low infection level throughout the entire study cycle – just 34.0% (
Table 7).
Optimization of exposure time (Variants E1 vs. E2): Statistical analysis showed that extending the seed potato soaking time is not always necessary, as the differences between short (E1 – 10 min) and long (E2 – 20 min) exposure to EM in 2019 were statistically insignificant. This suggests that under conditions of average pathogen risk, a shorter treatment time is sufficient to obtain a satisfactory protective effect (
Table 7).
3.1.2. Infection Level of a Tuber Sample (Commercial Quality Index)
This index determines the overall health of the batch, considering both the extent (presence) and intensity (area) of common scab symptoms. Key effects of EM technology include:
- −
Improved quality: The average health score increased from 7.3° (control) to 7.8° (E2). On a 9-point scale, an increase of 0.5° represents a statistically and commercially significant improvement in skin cleanliness.
- −
Health Leaders: The Jelly cultivar demonstrated the highest reactivity to the product (reaching 8.6° in the E1 variant). Very high scores (8.5°) were also recorded for the Czapla and Roxana cultivars, which in practice means almost symptom-free tubers.
- −
Genetic stability: The Oktan and Krasa cultivars demonstrated high initial resistance (results above 8.0° in the control group), confirming their strong genetic predisposition to limiting pathogen expansion (
Table 5).
Inter-annual variability: The years 2019–2020 were characterized by low infection pressure (average degree of infection in the tuber sample > 7.9°), with symptoms being purely cosmetic. A drastic deterioration in quality occurred in 2021 (6.6°) (
Table 6).
Protective mechanism: In the year of high infection pressure (2021), the index in the control group dropped to a critical level of 6.1°. The use of the E2 variant effectively cushioned this decline, increasing the result to 7.3°, which significantly increased the market value of the lot (
Table 7).
The analysis (
Table 5,
Table 6 and
Table 7) confirms that longer exposure to the biopreparation (E2) is an effective tool for stabilizing quality. commercial potatoes, especially in seasons with a high risk of infection, regardless of varietal conditions.
3.1.3. Severity of Common Scab Infection on Infested Tubers
EM effectively limits both the infection rate and the "depth" of the disease. This metric refers specifically to tubers already exhibiting symptoms and assesses the extent of damage (e.g., surface scab vs. pitted/cankered scab). Pre-planting tuber treatments with EM improved the average score from 6.5° to 6.8° on a 9° scale. This indicates that even when tubers were infected, the lesions were shallower and less harmful, particularly in the E2 treatment (
Table 5).
Varietal Resistance and Genetic Factors: The severity of this trait was significantly influenced by genetic characteristics. The Ewelina, despite a high percentage of infected tubers (78.8%), achieved the highest severity score of 7.7° on the 9.0° scale, indicating that the scabs were very superficial and did not penetrate deep into the tuber. Red Fantasy showed a consistent improvement in lesion depth, increasing from 6.7° (Control) to 7.3° (E2).
Overall, Ewelina exhibited the highest health status (least affected tubers), reaching the highest values on the 9° scale. Conversely, Nora was characterized by the highest stability, showing nearly identical results regardless of the treatment, which suggests lower reactivity to the tested microorganisms.
Negative Interactions and Treatment Duration: A significant negative reaction was observed in Hinga and Krasa, which showed a substantial increase in infection severity under the E1 treatment compared to the control. In many cases (e.g., Oktan, Red Fantasy, Jelly), the extended 20-minute inoculation (E2) yielded the best results, suggesting it is the most universal method for improving tuber health. The poorest results were recorded for Hinga under the shorter inoculation period (E1), where resistance dropped to 5.0°—the lowest in the entire dataset. This suggests that for this specific cultivar, the 10-minute EM inoculation (E1) may have significantly exacerbated the disease symptoms. Cultivars like Ewelina and Red Fantasy demonstrated high tissue resistance, as their infection severity scores remained significantly higher (healthier) even under intense pathogen pressure (
Table 5).
Annual Variability and Environmental Impact: The severity of common scab (S. scabies) lesions varied across the study years:
- −
2019: Results were highest (approx. 7.0°), indicating very superficial lesions.
- −
2021: Infections were not only more widespread but also deeper (Control group: 5.4°) (
Table 6).
Treatment E2: In 2021, the E2 variant reached 6.2°, confirming that a 20-minute exposure to EM not only reduces the number of infected tubers but ensures shallower, less damaging lesions.
This annual variability suggests that weather conditions in 2021—specifically drought during tuber initiation—favored the development of common scab. Nora and Roxana proved resilient to environmental stress, showing high repeatability across years, which confirms their health stability.
Extreme Cases: A notable outlier occurred in 2021 with the cultivar Krasa under the E2 treatment, where the score plummeted to 2.3
° in 9⇨ scale (
Table 7). This indicates severe negative interaction between the treatment and the specific environmental conditions of that year.
3.2. Rhizoctonia Tuber Infection
Statistical analysis showed significant differences in the response of individual genotypes to the biotechnological treatments used (
Table 8).
Effectiveness of Treatments in Reducing the Proportion of Diseased Tuber Plants: This indicator exhibits the greatest variability. The following cultivars were treated to significantly reduce the number of diseased tubers compared to the control: Nicola, which saw a dramatic decrease in the proportion of diseased tubers in the E1 treatment (35.9%) compared to the control (62.8%). Ewelina & Hinga showed a significantly positive response to the E2 treatment. In the Hinga cultivar, the proportion of infected tubers in the sample dropped from 43.6% to just 19.7%. In the Oktan cultivar, both treatments (E1 and E2) significantly reduced the spread of the disease compared to the control group (
Table 8).
Three groups of cultivars with different reactivity were distinguished:
A group of cultivars with high positive reactivity. The most spectacular biological protection effects were observed in the Ewelina and Hinga cultivars.
The use of the E2 variant (20 min exposure) allowed for a drastic reduction in the proportion of infected tubers from over 42-43% (control) to only 19.3-19.7%.
In the Hinga cultivar, the E2 variant also significantly improved the overall health of the sample (increasing from 4.6 to 6.8 on a 9-point scale), indicating a strong synergy between the EM microbiome and this cultivar's natural defense mechanisms.
Specific response of the Nicola cultivar: The Nicola cultivar demonstrated an unusual pattern of response to EM inoculation, as: It demonstrated a significant improvement in health with a shorter exposure time (E1), where the proportion of infected tubers decreased to 35.9%.
Extending exposure to 20 minutes (E2), however, resulted in a deterioration of parameters (increasing infection to 74%). This suggests that for some genotypes, excessive inoculation may lead to an imbalance in the geocaulosphere.
Stabilization of skin quality (e.g., Bellarosa, Red Fantasy): The Bellarosa cultivar maintained very high sample health (7.8–8.0) regardless of the cultivar, with E1 inoculation further reducing the number of infected tubers (a decrease to 36.3%).
In the Red Fantasy cultivar, E2 reduced the number of diseased tubers (33.9%), although a simultaneous decrease in the health of already infected tubers was noted, indicating a complex mechanism of pathogen-EM interactions in this genotype (
Table 8).
It can therefore be concluded that:
Optimization of exposure time: For most cultivars (Krasa, Nora, Ewelina, Hinga), the longer exposure (E2) proved to be more beneficial or statistically safer, resulting in a higher score on a 9-point scale.
Effect of EM on the degree of infection: EM inoculation most effectively improves the health of infected tubers (alleviates symptoms) in initially more susceptible cultivars, such as Hinga.
The high LSD value for the proportion of tubers infected with
R. solani highlights the high plasticity of the microbiome within different cultivars, suggesting the need to personalize pre-planting improvement treatments for specific potato genotypes (
Table 8).
Symptom severity (Infection degree): By analyzing the degree of infection of the sample itself and of already infected tubers, conclusions can be drawn about the "aggressiveness" of the disease under given conditions. For example, despite a relatively high percentage of diseased tubers, Bellarosa scored very high on a 9-point scale (7.3–8.0), demonstrating its high tolerance. Even when its tubers were infected, the degree of surface damage remained minimal. The Red Fantasy cultivar had the lowest degree of infection in infected tubers (5.3) with the E2 treatment, suggesting that this particular treatment, combined with the cultivar's resistance, effectively inhibits mycelium growth on the skin surface (
Table 8).
Dependence on earliness group: No clear correlation was found indicating that late cultivars are more susceptible than early cultivars. However, very early cultivars (Krasa, Korona) tended to be more severely infected under the E1 treatment. This may suggest that the early development cycle of these cultivars interferes with the timing or specificity of this treatment. Late, starchy cultivars (Hinga, Czapla) responded better to the E2 treatment, which may be due to the longer growing season and different dynamics of sclerotia formation on tubers. Unusual responses (C x T interactions): Differences between some cultivars were statistically significant. The E1 treatment in the Hinga cultivar resulted in a drastic decrease in the infection rate (to 4.6), while maintaining a relatively low percentage of diseased tubers (42.9%) (
Table 8).
Analysis of the year x cultivar interaction provided key information on the genetic stability of resistance and plasticity of the studied potato cultivars in the face of
R. solani pressure. This suggests significant dynamics of infection changes induced by hydrothermal factors in individual growing seasons (
Table 9).
Analysis of data from 2019–2021 indicates a very strong influence of weather conditions, which significantly modified the genetic resistance of cultivars.
2021 as the season with the highest pathogen pressure: 2021 is statistically distinguished by the highest percentage of tubers with R. solani for almost all cultivars (e.g., Roxana increased from 12.7% to 77.0%, Bellarosa from 0% to 71.3%). Conditions in 2021 (a cool and wet spring) favored mass infections of stolons and young tubers, eliminating the natural resistance of many cultivars.
Symptom severity vs. disease paradox Spread: There is a noticeable inverse correlation between the number of diseased tubers and their degree of infection (scale 1-9):
In 2020, the proportion of diseased tubers was moderate, but their degree of infection (infected units) was the highest (often above 8.0, e.g., in the Ewelina, Red Fantasy, and Hinga cultivars). This means that infected tubers were covered with very thick, numerous sclerotia. In 2021, although there were more diseased tubers, the degree of damage was less severe. Varietal Stability (Plasticity):
The highly stable cultivars Korona and Oktan showed relatively smaller fluctuations in infection rates compared to the others, suggesting their greater resistance to changing environmental conditions.
Extreme reaction (Bellarosa): This cultivar exhibits an extreme reaction to the year factor – from a complete absence of symptoms (0% in 2019) to very high infection rates (over 70% in 2021). This indicates that its resistance is highly dependent on weather conditions during critical growth stages.
Earliness groups and infection date: Late and starchy cultivars (Hinga, Czapla) showed lower infection rates than early cultivars in the year with the lowest overall infection pressure, 2019 (the year with the lowest overall infection pressure). This suggests that the later tuber setting date in these cultivars allowed them to escape the first wave of infection, which affected the fast-growing cultivars (
Table 9).
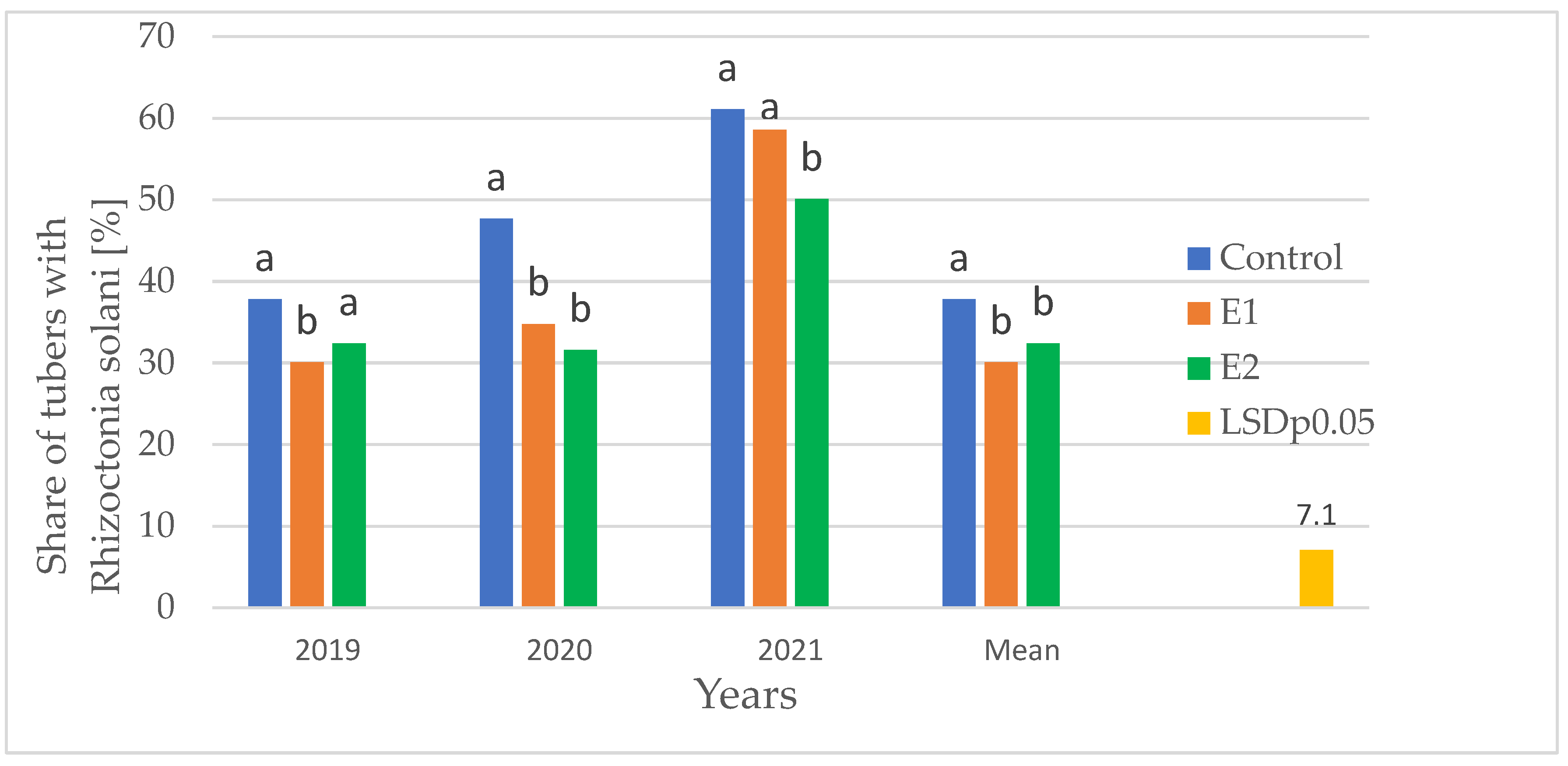
3.2.1. Analysis of the Effectiveness of Inoculation in Reducing R. solani
The analysis of the data presented in
Figure 1 indicates a significant effect of the biopreparations used (E1 and E2) on reducing the infection of potato tubers by
R. solani, and this effectiveness was strictly dependent on the conditions prevailing in the individual years of the study.
Analysis of the effectiveness of inoculation in reducing R. solani. Research results from 2019–2021 clearly indicate that engineering the geocaulosphere microbiome through seed potato inoculation (variants E1 and E2) provides an effective biological barrier against the pathogen.
Disease severity dynamics (Year effect): Pathogen pressure showed significant variability throughout the research cycle, allowing for the assessment of the stability of protective methods under various conditions:
- −
Year 2021 (Maximum pressure): The highest disease severity; infestation exceeded 60% in the control treatment.
- −
Year 2019 (Minimum pressure): The lowest infection level in the control treatment, approximately 38%.
Effectiveness of variants E1 and E2: Both forms of inoculation statistically significantly reduced the proportion of infected tubers in each year of the study.
Key observations:
- −
2020 – High stability and reduction in paralysis by over 10 percentage points in both variants (E1 and E2).
- −
2021 – Extreme infection pressure. Variant E2 demonstrated higher efficacy than E1 under severe stress conditions (
Figure 1).
3.4. Variability of Tuber Infection with Common Scab Under the Influence of Cultivar and Climatic Factors
Tuber skin quality is a key commercial parameter, determining potato suitability for washing and packaging. Common scabs, caused by S. scabies, pose a significant agrotechnical challenge due to the widespread nature of the pathogen and its strong dependence on external factors. The severity of this disease is the result of a complex genotype × environment interaction:
Varietal factors: The genetically determined skin structure and the rate of its maturation determine the plant's underlying susceptibility.
Hydrothermal conditions (HTC): Moisture deficit during the tuberization drastically increases the risk of infection, making weather patterns the primary modifier of pathogen pressure.
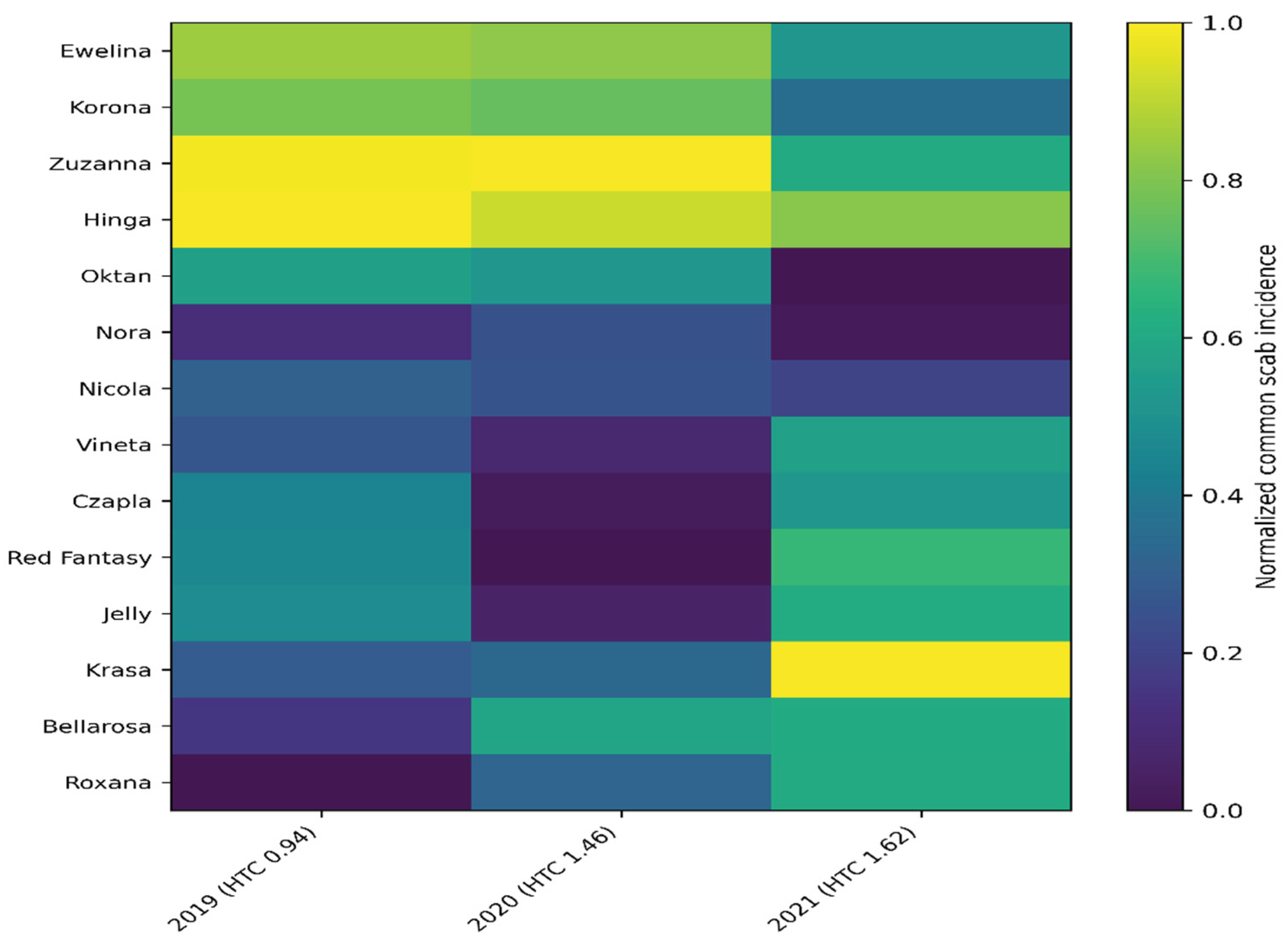
Therefore, the dynamics of tuber infection between 2019 and 2021 were analyzed. The aim of the analysis was to identify the resistance stability of the studied cultivars and determine the impact of climatic variables on yield quality, which is crucial for optimizing control strategies in precision agriculture.
Heat map analysis (
Figure 2) allows us to identify multivariate relationships between hydrothermal conditions and tuber health. Key findings:
Genotype × Environment Interaction (Cultivar and Year) Cultivar Variability: The heat map clearly differentiates cultivars into groups with varying degrees of susceptibility. Cultivars characterized by "cool" color (low %) throughout the entire study period can be considered genetically more resistant to common scab. Resistance Stability: Cultivars that maintain consistent color throughout the year demonstrate high phenotypic stability, which is crucial for precision agriculture.
Impact of Hydrothermal Coefficient (HTC) Correlation with Humidity: Color changes on the heat map correlate with HTC values for individual years (2019–2021). Typically, dry years (low HTC during tuberization) generate "hotter" areas on the map, confirming that water shortages drastically increase S. scabies pressure. Critical years: If 2021 (as suggested by previous data) was a year with a specific temperature and precipitation pattern, the map should indicate it as the year with the highest infection gradient for susceptible cultivars.
Identification of risk cultivars: The map allows for the quick identification of cultivars that reach thresholds critical infection in years with low HTC (drought), disqualifying them from the washed potato segment.
Biological barrier: The heat map provides a comparative framework – it allows for assessing whether scab pressure in given years was strong enough to verify the effectiveness of the biopreparations used.
3.7. Theoretical Health Model (Health-Shift Model)
This model assumes that EM inoculation acts as a vector force that counteracts environmental and pathogenic pressures.
Table 13 presents a theoretical model of the health-shift mechanism. It explains how EM Farming™ technology (variants E1 and E2) modifies the position of the studied objects in statistical space (PCA and cluster analysis), translating biological processes into measurable resistance indicators.
Here is a detailed interpretation of the key elements of this model:
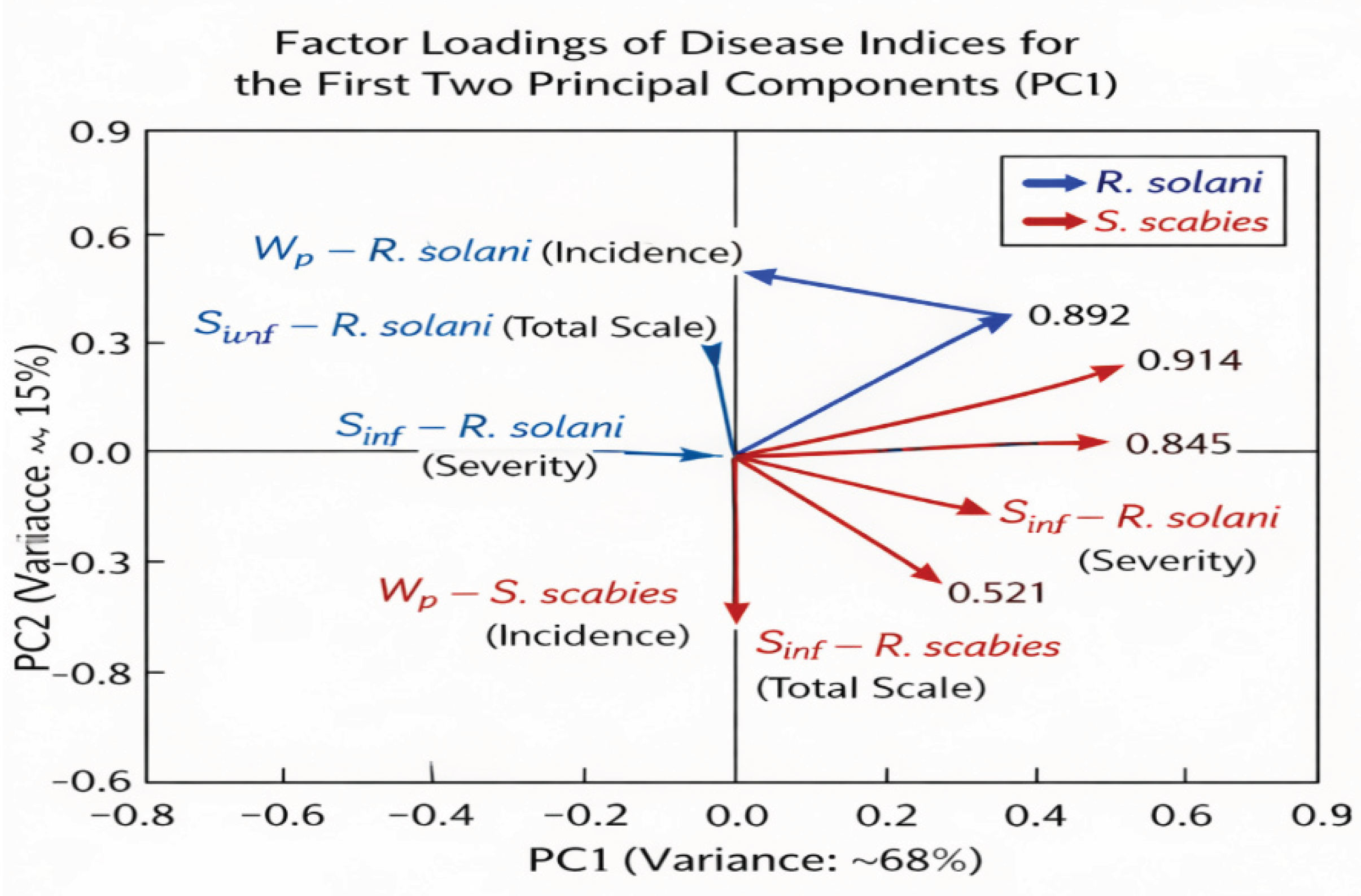
Control (C): Susceptibility and Risk:
Mechanism: The control cultivar is biologically "naked" – its health depends solely on genetic traits that fail at high HTC (high humidity favoring pathogens). Statistical interpretation: These cultivars correlate positively with the infection vector (S
inf). In the PCA plot (
Figure 4), they are in the high-risk zone, meaning their health profile is dominated by the presence of the pathogen (
Table 13).
Inoculation E1 (10 min): Initiation Phase:
Mechanism: A short exposure is sufficient to initiate biofilm formation and activate nonspecific systemic immunity (NSR). This is the "kickstart" of the biological shield. Statistical interpretation: There is a clear shift in the cultivar along the first component axis (PC1). In PCA analysis, the PC1 axis typically explains the largest proportion of variability (here: level of infection). A leftward movement indicates a real reduction in disease severity, but the object may still remain within the pathogen's sphere of influence (
Table 13).
Inoculation E2 (20 min): Full Stabilization and "Health-Shift:
Mechanism: A longer exposure time allows for full colonization of the geocaulosphere (the zone around underground stems and tubers). A stable "core" microbiome is formed, which actively displaces
R. solani. Statistical interpretation: This is the most radical change. The object is reclassified – statistically, it no longer resembles a diseased plant. An increase in the Euclidean distance from the pathogen in the cluster plot indicates that the plant's microbiological and biochemical profile has become radically different from the control group (
Table 13).
Mechanism summary: The model demonstrates that EM technology does not merely target the pathogen but changes the plant's biological status:
- −
E1 is a symptom-limiting intervention.
- −
E2 is microbiome engineering that moves the plant into a completely different health category (the so-called “Stable & Healthy” cluster) (
Table 13).
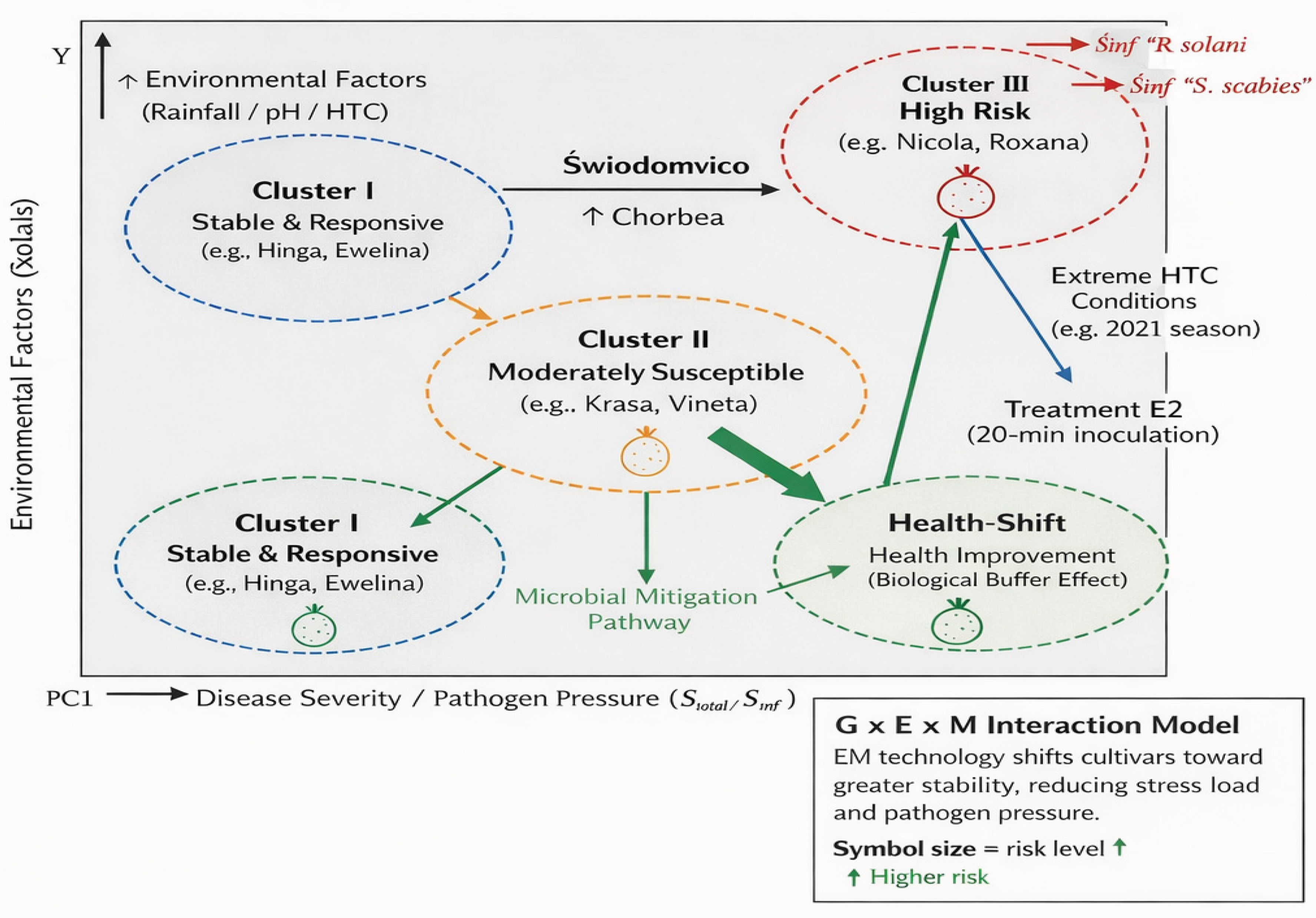
3.7.1. Model Description (for Figure Caption)
The presented G x E x M interaction model illustrates the mechanism in which EM technology acts as a biological buffer, enabling the shift of potato cultivars from high infection risk zones (Cluster III) towards groups with increased health and yield stability (
Figure 4).
Theoretical model of the G × E × M interaction (Genotype × Environment × Microbiome). The model illustrates the "Health-Shift" phenomenon where microbial engineering (EM Farming™) reclassifies susceptible and intermediate cultivars into higher health clusters. Treatment E2 (20-minute exposure) acts as a biological buffer, neutralizing the negative impact of extreme hydrothermal conditions (high HTC) and reducing the Euclidean distance between susceptible cultivars and the resistant reference group (
Figure 4).